Submitted:
17 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
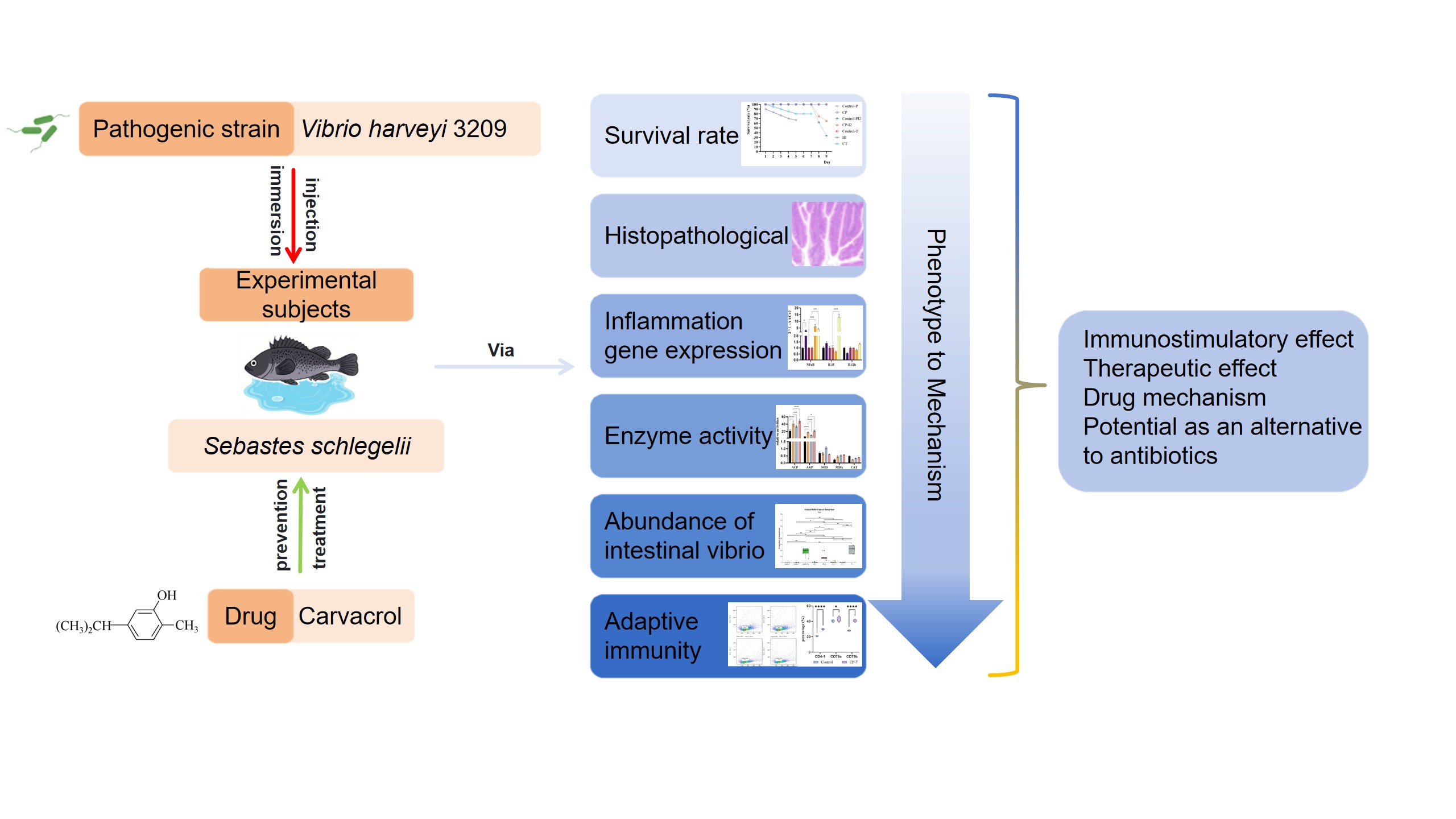
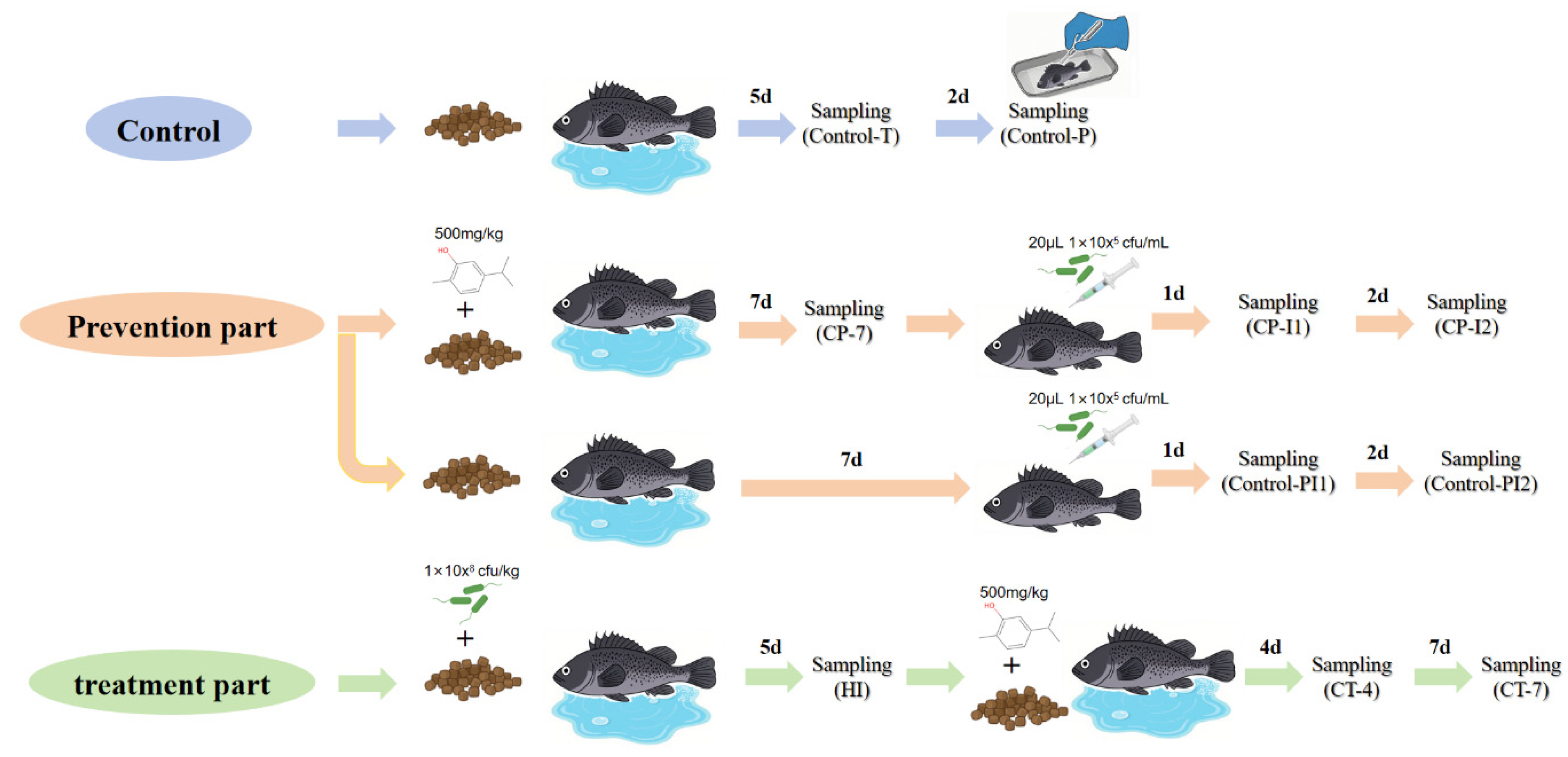
2.1. Fish Management, Sampling, Survival Rate Calculation and Pathological Observation
2.2. Carvacrol Treatment and Intestinal Pathogen Load in Sebastes schlegelii
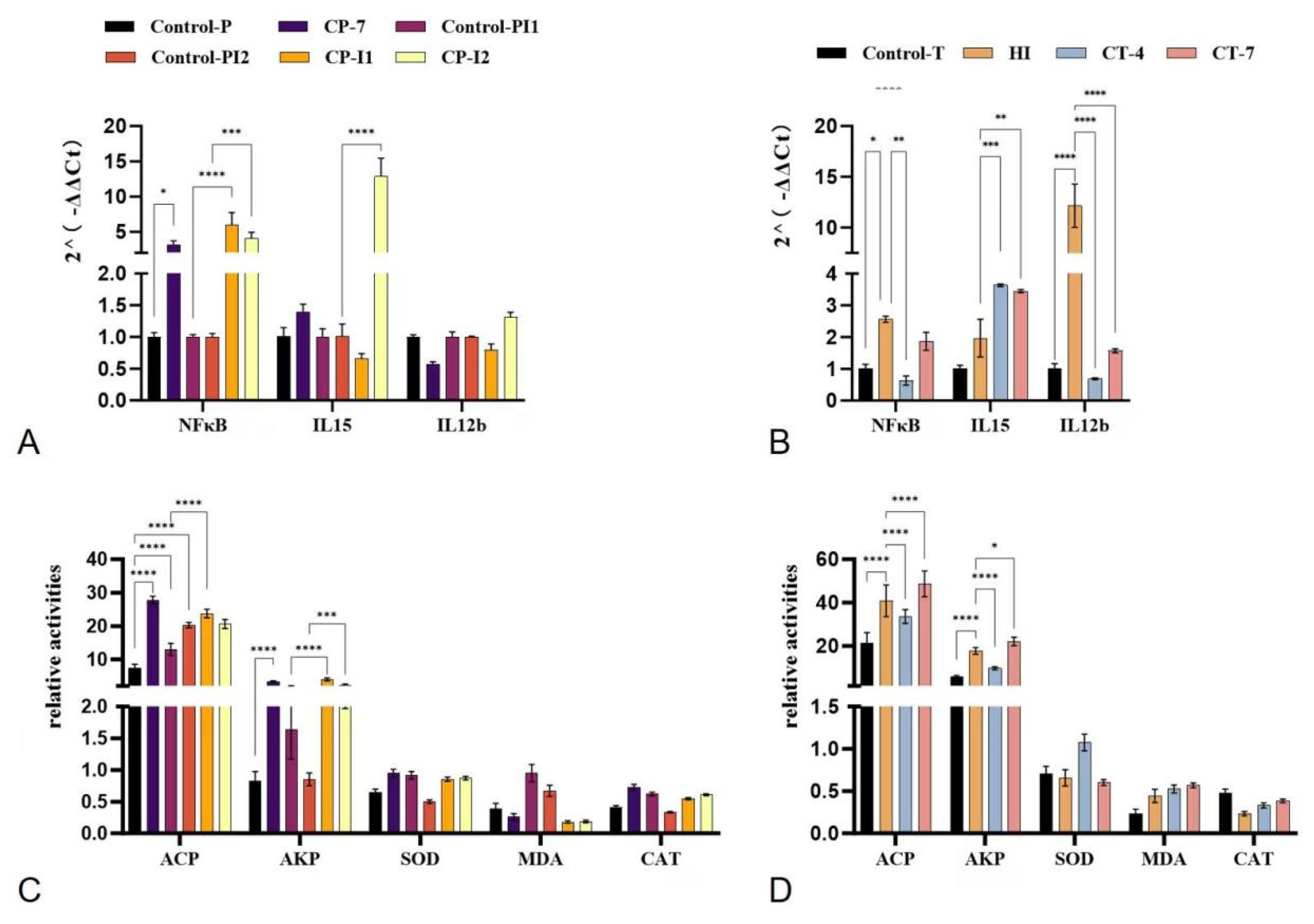
2.3. Effects of Carvacrol on Non-Specific Immunity of Sebastes schlegelii Under Vibrio harveyi Stress
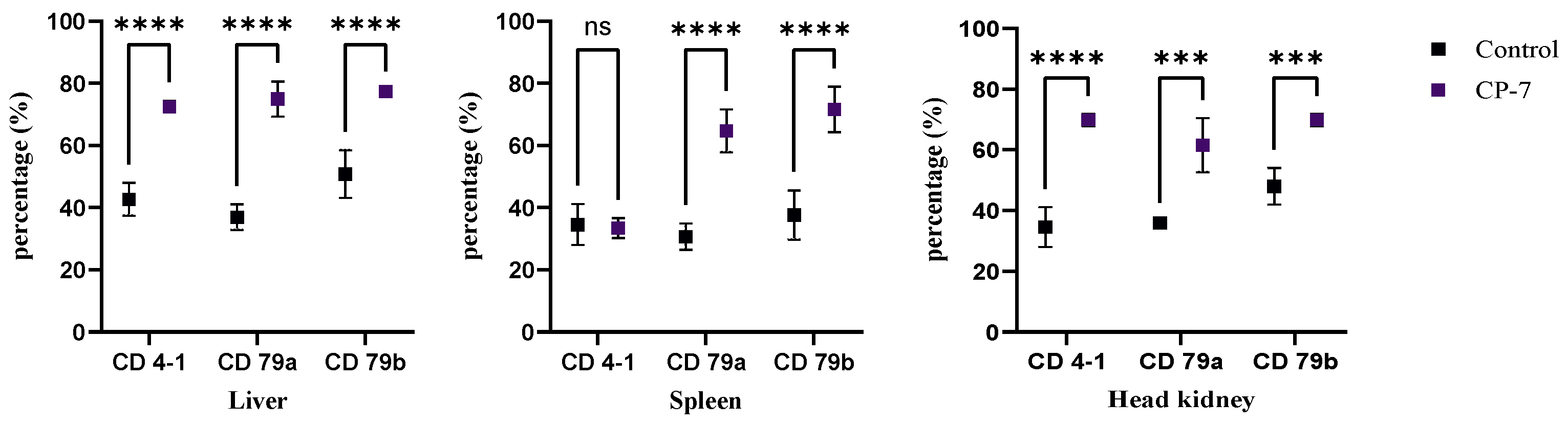
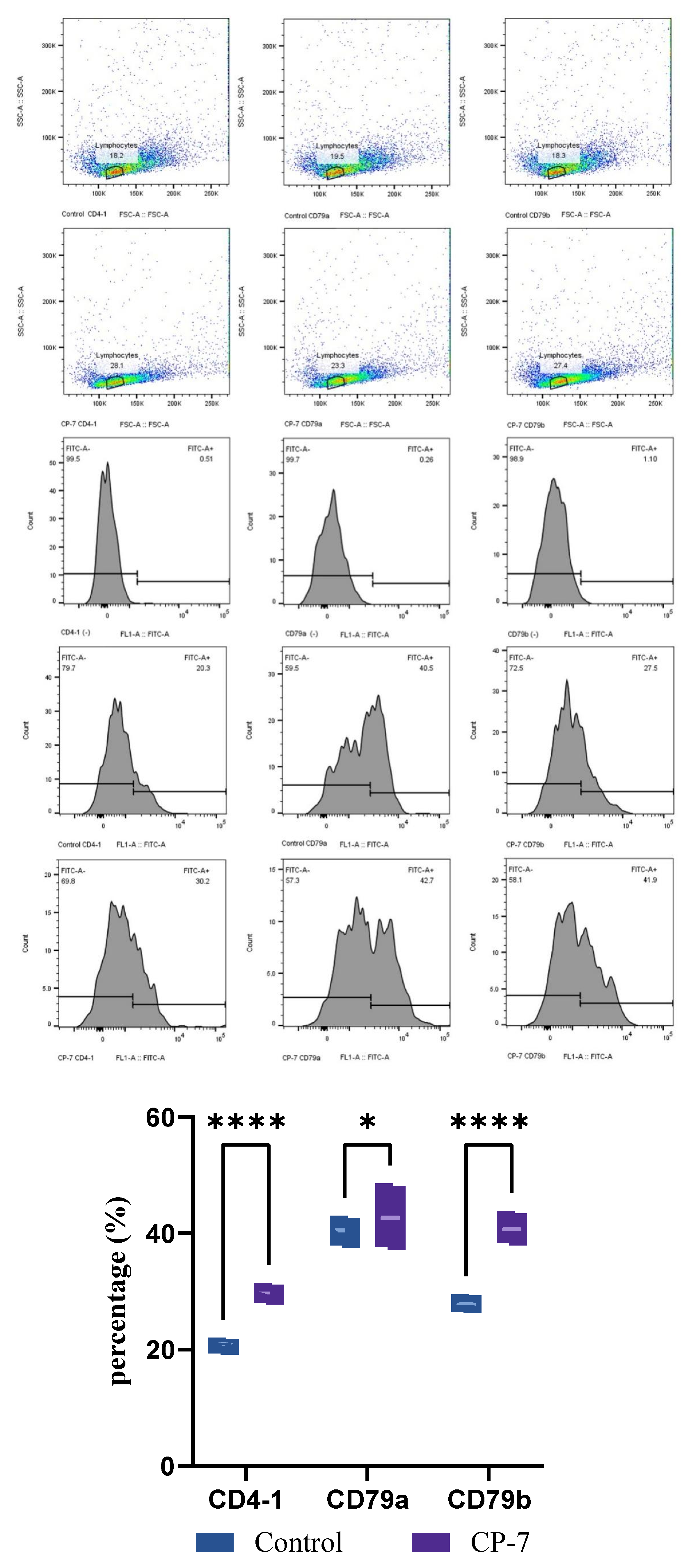
2.4. Regulatory Effect of Carvacrol Prophylactic Treatment on Specific Immunity in Sebastes schlegelii
2.5. Statistical Analysis
3. Result
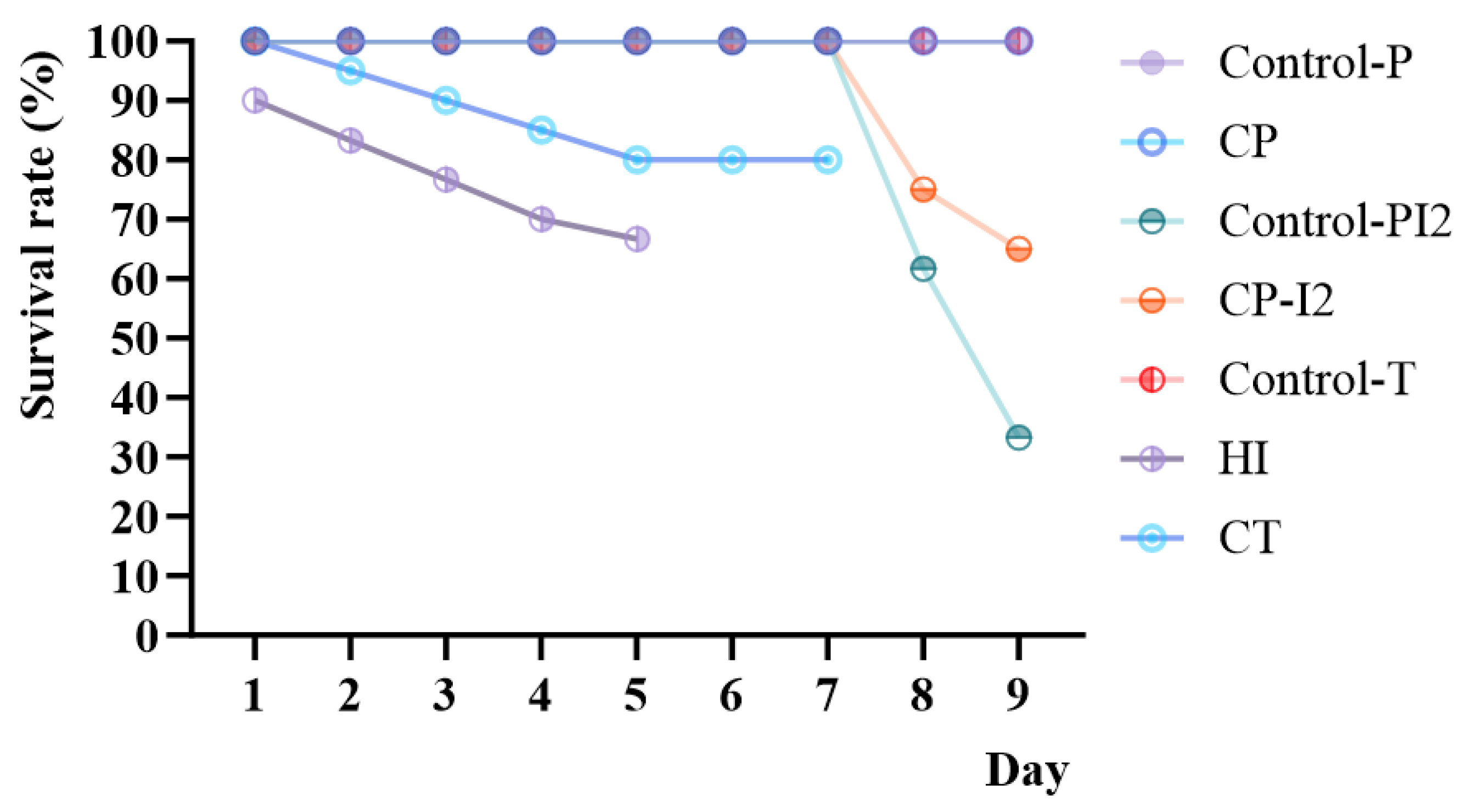
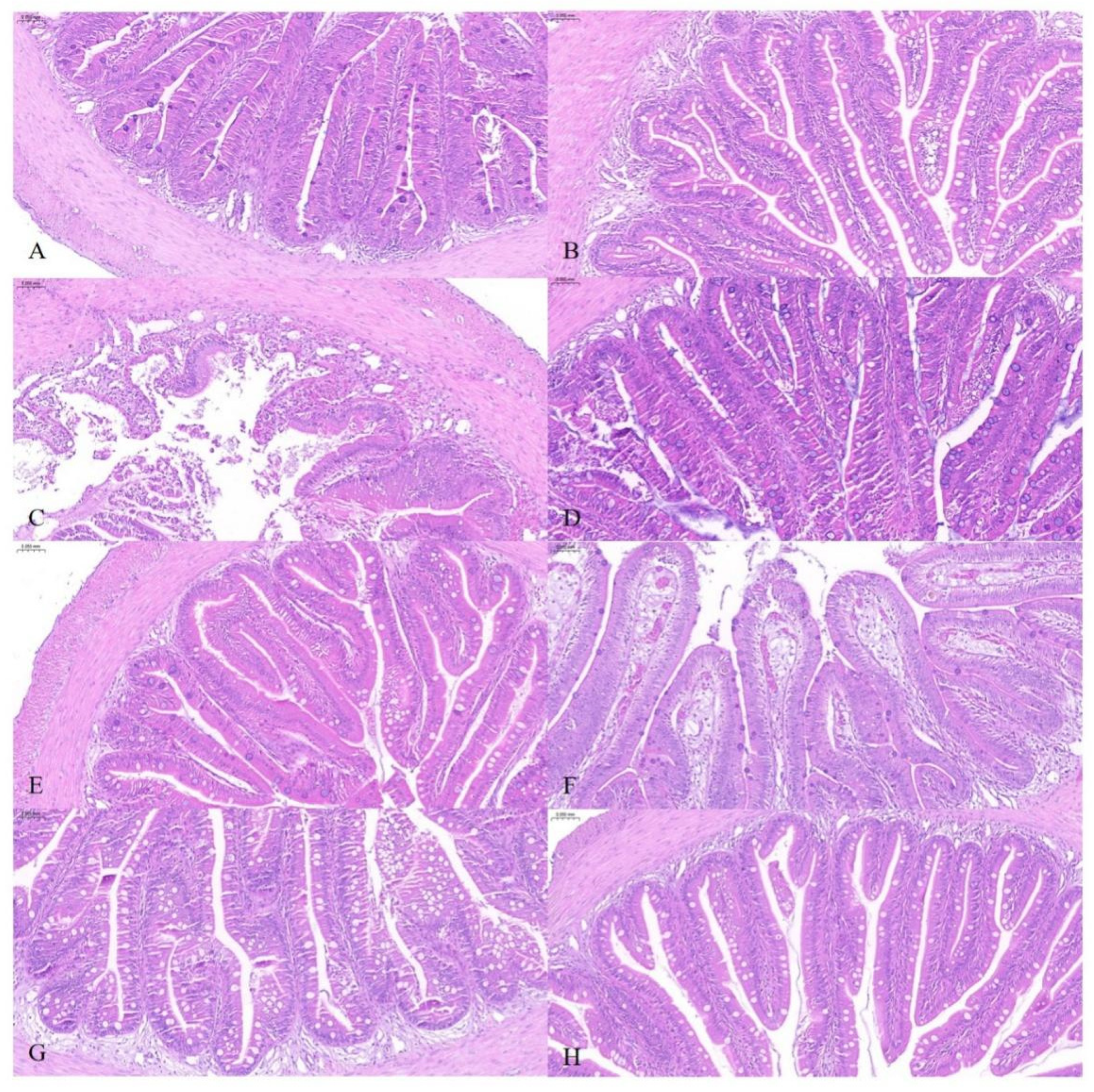
3.1. Effects of Carvacrol on Survival and Intestinal Histopathology of Fish Infected with Vibrio harveyi
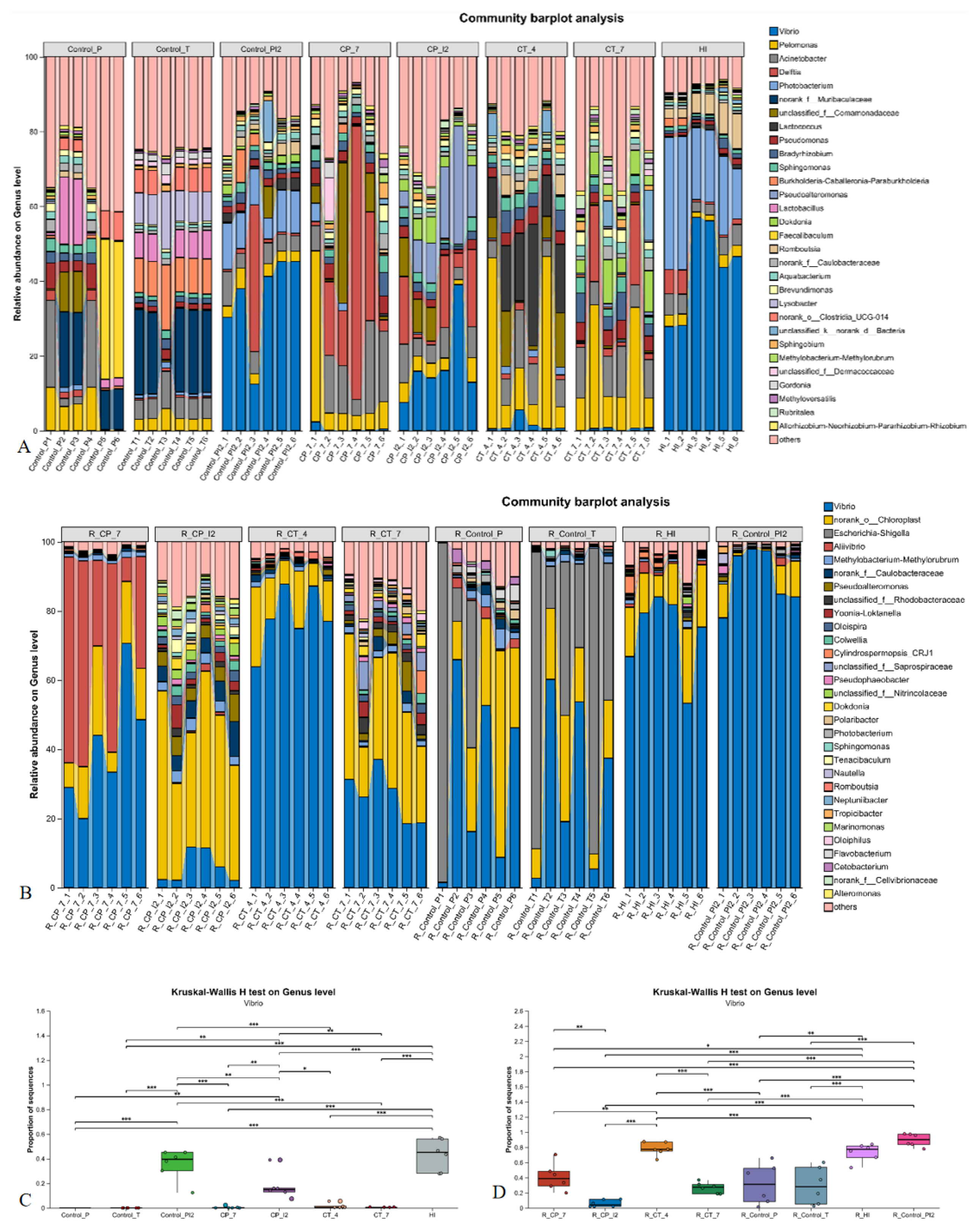
3.2. Changes in Intestinal Vibrio Load in Sebastes schlegelii Under Pathogen Infection and Carvacrol Treatment
3.3. Changes in Non-Specific Immunity of Experimental Fish in Each Treatment Group
3.4. Expression of Specific Immune Cells in Tissues and Peripheral Blood After Prophylactic Administration of Carvacrol
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
References
- Ahmadifar, E.; Mansour, M. R.; Amirkolaie, A. K.; Rayeni, M. F. Growth efficiency, survival and haematological changes in great sturgeon (Huso huso Linnaeus, 1758) juveniles fed diets supplemented with different levels of thymol-carvacrol. Animal Feed Science and Technology 2014, 198, 304–308. [Google Scholar] [CrossRef]
- Alagawany, M.; Farag, M. R.; Salah, A. S.; Mahmoud, M. A. The role of oregano herb and its derivatives as immunomodulators in fish. Reviews in Aquaculture 2020, 12(4), 2481–2492. [Google Scholar] [CrossRef]
- Bhat, R. A. H.; Khangembam, V. C. C.; Pant, V.; Tandel, R. S.; Pandey, P. K.; Thakuria, D. Antibacterial Potential of a De-Novo Designed Peptide Against Bacterial Fish Pathogens. International Journal of Peptide Research and Therapeutics 2023, 29(5). [Google Scholar] [CrossRef]
- Bloomfield, S. J.; Palau, R.; Janecko, N.; Baker-Austin, C.; Mather, A. E. Association of antimicrobial resistant Vibrio and species pathogenic to humans with aquacultured seafood. Food Microbiology 2025, 132. [Google Scholar] [CrossRef] [PubMed]
- Clark, S. E.; Burrack, K. S.; Jameson, S. C.; Hamilton, S. E.; Lenz, L. L. NK Cell IL-10 Production Requires IL-15 and IL-10 Driven STAT3 Activation. Frontiers in Immunology 2019, 10. [Google Scholar] [CrossRef]
- Coelho, J. D.; Rosa, K. V.; Rocha, J. S.; Ramirez, N. C. B.; Maraschin, M.; Vieira, F. D. IN VITRO ANTIMICROBIAL ACTIVITY OF CARVACROL AGAINST SHRIMP PATHOGENS AND ITS USE AS FEED ADDITIVE FOR THE PACIFIC WHITE SHRIMP; <u>Boletim Do Instituto De Pesca</u> <b>47</b>, 2021. [Google Scholar] [CrossRef]
- Das, S.; Chourashi, R.; Mukherjee, P.; Kundu, S.; Koley, H.; Dutta, M.; Mukhopadhyay, A. K.; Okamoto, K.; Chatterjee, N. S. Inhibition of growth and virulence of Vibrio cholerae by carvacrol, an essential oil component of Origanum spp. Journal of Applied Microbiology 2021, 131(3), 1147–1161. [Google Scholar] [CrossRef]
- Deng, H.; Deng, Y.; Song, T.; Pang, L.; Zhu, S.; Ren, Z.; Guo, H.; Xu, Z.; Zhu, L.; Geng, Y.; Ouyang, P.; He, R.; Deng, J. Evaluation of the activity and mechanisms of oregano essential oil against PRV in vivo and in vitro. Microbial Pathogenesis 2024, 194. [Google Scholar] [CrossRef]
- Drake, A.; Kaur, M.; Iliopoulou, B. P.; Phennicie, R.; Hanson, A.; Chen, J. Z. Interleukins 7 and 15 Maintain Human T Cell Proliferative Capacity through STAT5 Signaling. Plos One 2016, 11(11). [Google Scholar] [CrossRef]
- Hirose, S.; Horiyama, S.; Morikami, A.; Fujiwara, K.; Tsukagoshi, H. Eugenol and basil essential oil as priming agents for enhancing Arabidopsis immune response. Bioscience Biotechnology and Biochemistry 2024, 89(1), 41–50. [Google Scholar] [CrossRef]
- Huang, X. G.; Takata, K.; Sato, Y.; Tanaka, T.; Ichimura, K.; Tamura, M.; Oka, T.; Yoshino, T. Downregulation of the B-cell receptor signaling component CD79b in plasma cell myeloma: A possible post transcriptional regulation. Pathology International 2011, 61(3), 122–129. [Google Scholar] [CrossRef]
- Huse, K.; Bai, B. Y.; Hilden, V. I.; Bollum, L. K.; Våtsveen, T. K.; Munthe, L. A.; Smeland, E. B.; Irish, J. M.; Wälchli, S.; Myklebust, J. H. Mechanism of CD79A and CD79B Support for IgM plus B Cell Fitness through B Cell Receptor Surface Expression. Journal of Immunology 2022, 209(10), 2042–2053. [Google Scholar] [CrossRef]
- Imtiaz, N.; Anwar, Z.; Waiho, K.; Shi, C.; Mu, C.; Wang, C.; Wu, Q. A review on aquaculture adaptation for fish treatment from antibiotic to vaccine prophylaxis. Aquaculture International 2024, 32(3), 2643–2668. [Google Scholar] [CrossRef]
- Jeong-hyeon, C.; Gyeong-mi, W.; Sae-bom, S.; Hyo-jin, P.; Soon-kyu, B.; Bae-ik, L.; Jong-ha, L.; I-cheong, K.; Su-il, P. Pathogenicity of Vibrio harveyi to black rockfish, Sebastes schlegeli. Journal of Fish Pathology 2007, 20(2), 99–108. [Google Scholar]
- Ma, P.; Chen, Y.; Shao, N.; Zhang, J.; Wu, Y.; Liu, R. Synergistic Combination of Biodegradable Peptide Polymer and Curcumin as Promising Antibiotic Substitution in Aquaculture to Alleviate the Global Challenge of Antimicrobial Resistance. Chinese Journal of Chemistry 2022, 40(24), 2947–2955. [Google Scholar] [CrossRef]
- Maisey, K.; Montero, R.; Corripio-Miyar, Y.; Toro-Ascuy, D.; Valenzuela, B.; Reyes-Cerpa, S.; Sandino, A. M.; Zou, J.; Wang, T. H.; Secombes, C. J.; Imarai, M. Isolation and Characterization of Salmonid CD4+ T Cells. Journal of Immunology 2016, 196(10), 4150–4163. [Google Scholar] [CrossRef]
- Manchanayake, T.; Mohamad, A.; Amir-Danial, Z.; Abu Nor, N.; Yong-Kit, C.; Nazarudin, M. F.; Nor, R. M.; Hasnan, Q.; Zamri-Saad, M.; Amal, M. N. A.; Ina-Salwany, M. Y.; Salleh, A. Oral adjuvanted Vibrio vaccine enhances antibody production and lysozyme activity in the serum and mucus of marine-cultured red hybrid tilapia (Oreochromis sp.) against V. harveyi and V. alginolyticus. Fish & Shellfish Immunology 2025, 165. [Google Scholar] [CrossRef] [PubMed]
- Márquez, A.; Mendoza, J. L.; Toxonera, C.; Díaz-Rubio, M.; de la Concha, E. G.; Urcelay, E.; Martínez, A. IL23R and IL12B polymorphisms in Spanish IBD patients:: No evidence of interaction. Inflammatory Bowel Diseases 2008, 14(9), 1192–1196. [Google Scholar] [CrossRef]
- Milijasevic, M.; Veskovic-Moracanin, S.; Milijasevic, J. B.; Petrovic, J.; Nastasijevic, I. Antimicrobial Resistance in Aquaculture: Risk Mitigation within One Health Context. Preprints 2024. [Google Scholar] [CrossRef]
- Pan, T.; Yan, M. The screening of traditional Chinese herbs on nonspecific immune response and protection of Pacific white shrimp (Litopenaeus vannamei) from Vibrio harveyi infection. Aquaculture International 2020, 28(2), 767–776. [Google Scholar] [CrossRef]
- Pittet, L. A.; Quinton, L. J.; Yamamoto, K.; Robson, B. E.; Ferrari, J. D.; Algül, H.; Schmid, R. M.; Mizgerd, J. P. Earliest Innate Immune Responses Require Macrophage RelA during Pneumococcal Pneumonia. American Journal of Respiratory Cell and Molecular Biology 2011, 45(3), 573–581. [Google Scholar] [CrossRef] [PubMed]
- Prazdnova, E.; Zaikina, A.; Neurov, A.; Mazanko, M.; Ranjan, A.; Rudoy, D. Bacillibactin, a Potential Bacillus-Based Antibacterial Non-Ribosomal Peptide: In Silico Studies for Targeting Common Fish Pathogens. International Journal of Molecular Sciences 2025, 26(12). [Google Scholar] [CrossRef] [PubMed]
- Rossi, B.; Esteban, M. A.; Garcia-Beltran, J. M.; Giovagnoni, G.; Cuesta, A.; Piva, A.; Grilli, E. Antimicrobial Power of Organic Acids and Nature-Identical Compounds against Two Vibrio spp.: An In Vitro Study. Microorganisms 2021, 9(5). [Google Scholar] [CrossRef]
- Strem, R.; Meiri-Ashkenazi, I.; Segal, N. a.; Ehrlich, R.; Shashar, N.; Sharon, G. Evaluation of Flathead Grey Mullets (Mugil cephalus) Immunization and Long-Term Protection against Vibrio harveyi Infection Using Three Different Vaccine Preparations. International Journal of Molecular Sciences 2023, 24(9). [Google Scholar] [CrossRef]
- Tayyab, M.; Islam, W.; Waqas, W.; Zhang, Y. Probiotic-Vaccine Synergy in Fish Aquaculture: Exploring Microbiome-Immune Interactions for Enhanced Vaccine Efficacy. Biology-Basel 2025, 14(6). [Google Scholar] [CrossRef]
- van Gelderen, T. A.; Debnath, P.; Joly, S.; Bertomeu, E.; Duncan, N.; Furones, D.; Ribas, L. Gonadal miRNomes and transcriptomes in infected fish reveal sexually dimorphic patterns of the immune response. Functional & Integrative Genomics 2025, 25(1). [Google Scholar] [CrossRef]
- Wan, X.; Da, F.; Lin, G.; Huang, Y.; Cai, S.; Jian, J. Transcriptome Analysis of the Cultured Hybrid Grouper (♀Epinephelus fuscoguttatus x♂E. lanceolatus) Immunized with Vibrio harveyi formalin-killed cells vaccine (FKC) combined with chitosan oligosaccharide. Israeli Journal of Aquaculture-Bamidgeh 2023, 75(2). [Google Scholar] [CrossRef]
- Wang, J.; Mai, K.; Ai, Q. Conventional Soybean Meal as Fishmeal Alternative in Diets of Japanese Seabass (Lateolabrax japonicus): Effects of Functional Additives on Growth, Immunity, Antioxidant Capacity and Disease Resistance. Antioxidants 2022, 11(5). [Google Scholar] [CrossRef]
- Wang, Q.; Huang, J.; Li, Y.; Wu, S.; Zhao, L.; Pan, Y.; Kang, Y.; Liu, Z. Chinese herbal medicines mixture improved antioxidant enzymes, immunity and disease resistance to infectious hematopoietic necrosis virus infection in rainbow trout (Oncorhynchus mykiss). Aquaculture International 2024, 32(3), 3217–3232. [Google Scholar] [CrossRef]
- Wu, Q.; Jiang, Y.; Chen, E.; Mu, C.; Waiho, K. Chinese gallnut (Gall chinensis) against Vibrio parahaemolyticus: In vitro activity and the use of medicated bath method to treat infected mud crab Scylla paramamosain. Aquaculture 2021, 539. [Google Scholar] [CrossRef]
- Xu, M.; Li, H.; Wu, X.; Dai, L.; Yang, R.; Zhang, Q.; Zhang, Z.; Lei, L.; Zhang, F. Inhibitory effect of arctigenin on reovirus infection in grass carp. Aquaculture 2024, 593. [Google Scholar] [CrossRef]
- Xu, Z.; Pei, L.; Zhang, F.; Hu, X.; Gui, Y.; Wang, L.; Wu, B. A functional variant in IL12B promoter modulates its expression and associates with increased risk of allergic asthma. Genes and Immunity 2013, 14(4), 238–243. [Google Scholar] [CrossRef]
- Yang, X.; Cheng, Y.; Zhao, Y.; Zhu, H.; Zhang, Y.; Zhao, Y.; Liu, X. Comparative transcriptome analysis reveals immunoregulation mechanism in the spleen of large yellow croaker (Larimichthys crocea) in response to Vibrio harveyi infection. Aquaculture Reports 2023, 31. [Google Scholar] [CrossRef]
- Yu, B.; Sun, Z.; Li, X. C.; Qv, A.; Sohail, M.; Li, Y. L.; Xu, H.; Xiang, P. Research Progress of Novel Drug Delivery Systems of Chinese Medicine Monomers based on Natural Silk Fibroin: A Mini-Review. Current Drug Delivery 2023, 20(3), 211–222. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B. Z.; Qiu, Y. L.; Shi, C. X.; Zhang, J. Development of Multiple Real-Time Fluorescent Quantitative PCR for Vibrio Pathogen Detection in Aquaculture. Veterinary Sciences 2025, 12(4). [Google Scholar] [CrossRef]
- Zhou, F.; Ji, B. P.; Zhang, H.; Jiang, H.; Yang, Z. W.; Li, J. J.; Li, J. H.; Yan, W. J. The antibacterial effect of cinnamaldehyde, thymol, carvacrol and their combinations against the foodborne pathogen Salmonella typhimurium. Journal of Food Safety 2007, 27(2), 124–133. [Google Scholar] [CrossRef]
- Zhu, X. L.; Zhu, J. F. CD4 T Helper Cell Subsets and Related Human Immunological Disorders. International Journal of Molecular Sciences 2020, 21(21). [Google Scholar] [CrossRef]
- Zhu, Z.-X.; Yao, Y.-Y.; Ai, C.-H.; Yang, G.; Liang, X.-Y.; Liu, T.-D.; He, M.-L.; Xia, J.-H. Dietary supplementation of Patchouli oil increases the resistance against Streptococcus agalactiae infection in GIFT tilapia as revealed by transcriptome and 16s amplicon profiling. Aquaculture Reports 2023, 33. [Google Scholar] [CrossRef]

| Target Gene | Primer | Nucleotide Sequence 5′-3′ |
|---|---|---|
| RPL-17 | RPL17-F | AGGCGACGCACCTACCG |
| RPL17-R | CCTCTGGTTTGGGGACGA | |
| NFκB/RelA | NFκB/RelA-F | TGTCGTAGATGGGGTTGGA |
| NFκB/RelA-R | AGGAGCTGGGGAAGGTGAT | |
| IL-12b | IL-12b-F | CTCTGGCATCCTTATCAGTTCA |
| IL-12b-R | GTCTTGGTTGCTGGCGTAG | |
| IL-15 | IL-15-F | CGCCTACAATACAACTAAAGAGC |
| IL-15-R | AGATGACGGAGCATACAGCA |
| Group | Vibrio Abundance Determined by DNA Extraction Method (%) | Vibrio Abundance Determined by RNA Extraction and Reverse Transcription (%) |
|---|---|---|
| Control-P | <0.06 | 42.08 |
| CP-7 | 0.32 | 38.25 |
| Control-PI2 | 46.59 | 92.54 |
| CP-I2 | 16.88 | 4.59 |
| Control-T | 0.06 | 31.12 |
| HI | 48.55 | 78.27 |
| CT-4 | 0.84 | 80.14 |
| CT-7 | 0.95 | 29.71 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




