Submitted:
16 March 2026
Posted:
18 March 2026
Read the latest preprint version here
Abstract
Keywords:
Introduction
Methods
Study Design
Inclusion Criteria
Variables Assessed
Clinical Phenotype Classification
Statistical Analysis
Ethical Considerations
Results
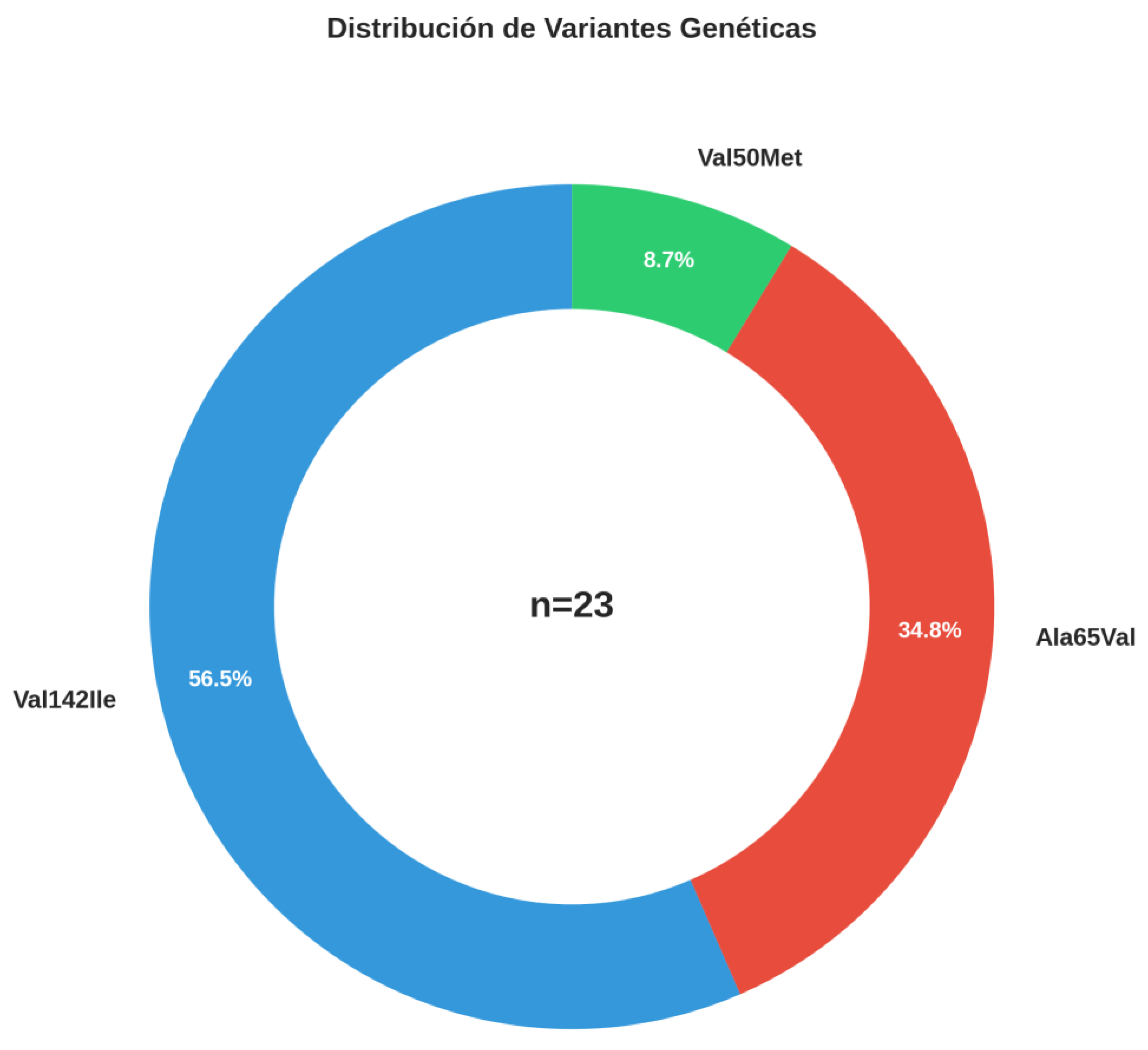
Demographic and Genetic Characteristics
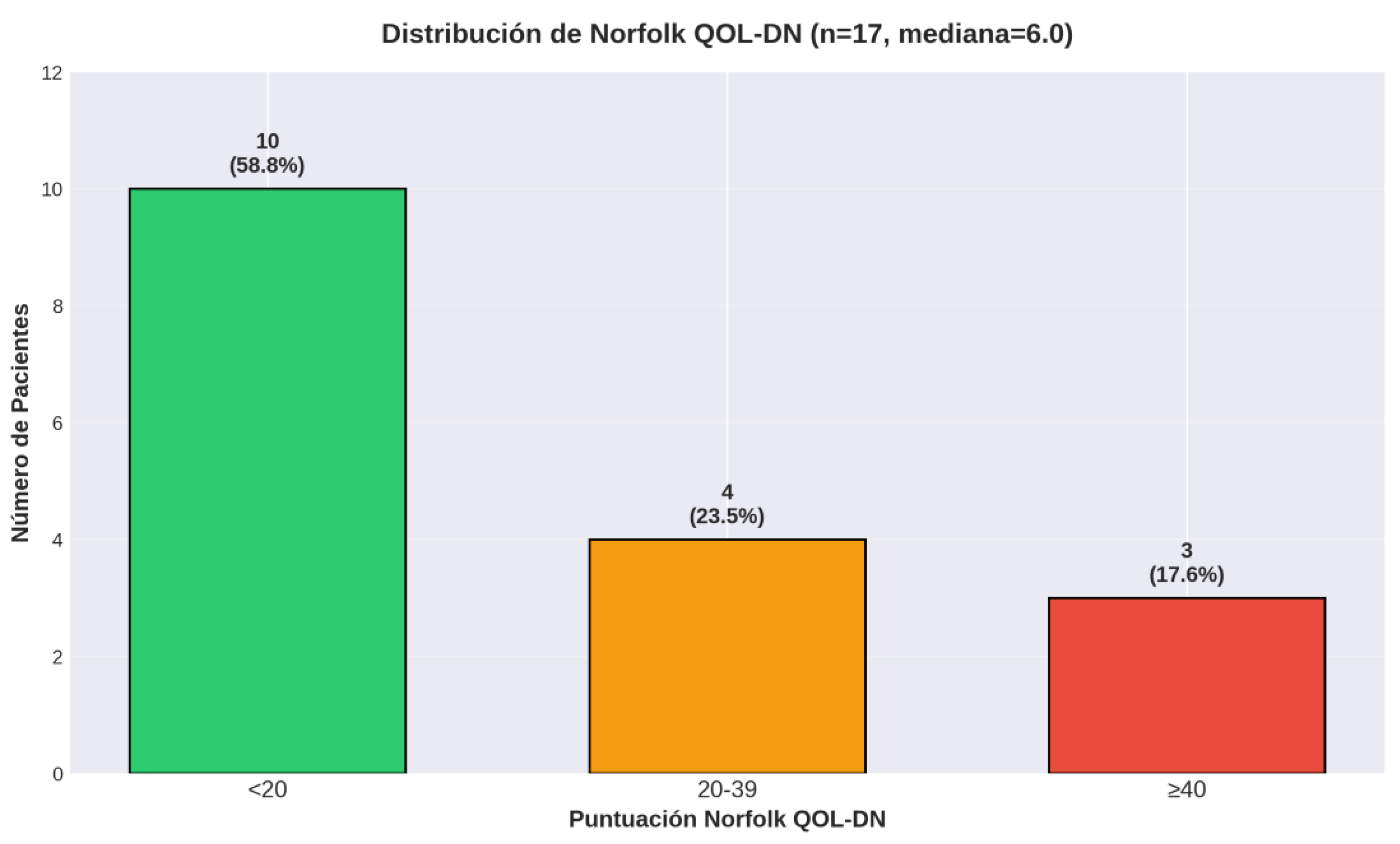
Neurological Assessment
Autonomic Assessment
Cardiovascular Assessment
Electrical Assessment and Medical History
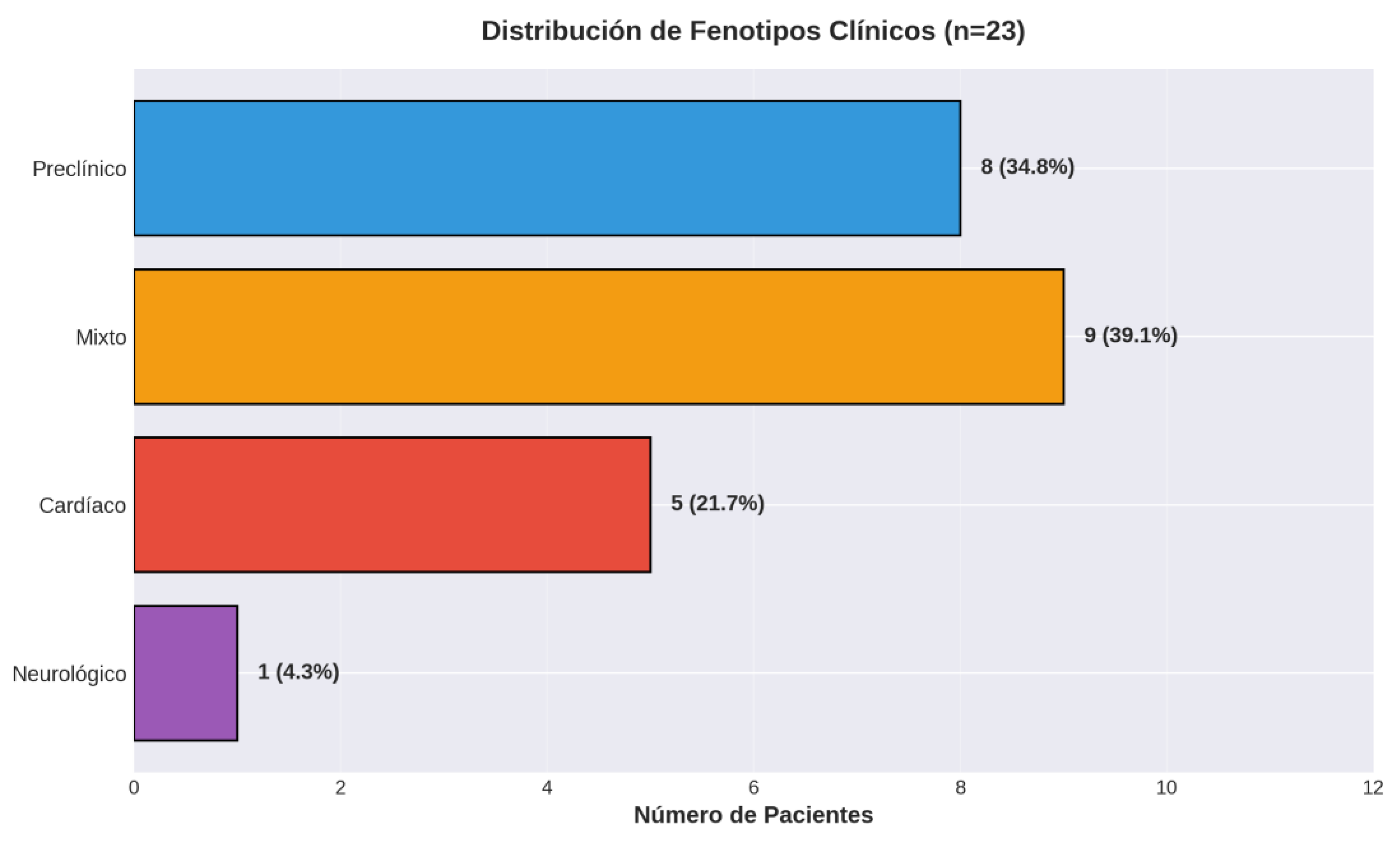
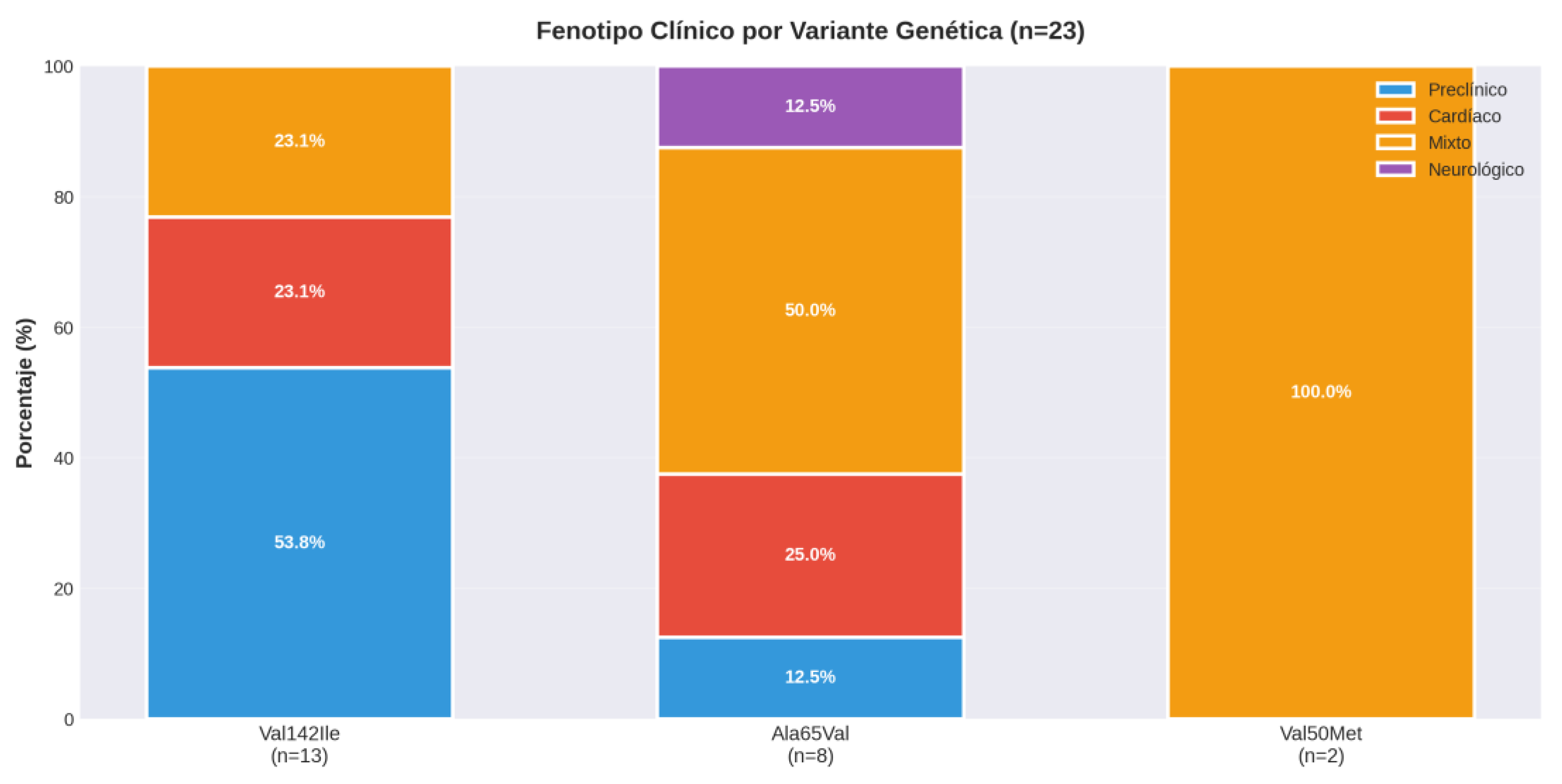
Clinical Phenotype Distribution
Discussion
Conclusions
Tables
| Variable | Total (n=23) | Val142Ile (n=13) | Ala65Val (n=8) | Val50Met (n=2) |
|---|---|---|---|---|
| Age, years - median (IQR) | 60 (45-70) | 65 (41-76) | 51 (46-66) | 64 (60-68) |
| Male sex, n (%) | 16 (69.6%) | 8 (61.5%) | 7 (87.5%) | 1 (50.0%) |
| Height, cm - mean (n=17) | 168.1 | 171.0 (n=8) | 172.8 (n=4) | 158.5 (n=2) |
| Weight, kg - mean (n=17) | 70.8 | 72.9 (n=8) | 67.5 (n=4) | 58.0 (n=2) |
| BMI - mean (n=6) | 28.3 | 24.0 (n=2) | 31.0 (n=1) | N/D |
| Zygosity - Heterozygous, n/total (%) | 16/16 (100%) | 8/8 (100%) | 5/5 (100%) | 2/2 (100%) |
| Pathogenic | 13/16 (81.3%) | 8/8 (100%) | 2/5 (40.0%) | 2/2 (100%) |
| Probably pathogenic | 3/16 (18.7%) | 0/8 (0%) | 3/5 (60.0%) | 0/2 (0%) |
| Age at diagnosis, years - mean | 57.4 | 63.8 (n=9) | 42.0 (n=5) | 62.5 (n=2) |
| Age at diagnosis, years - median | 66.0 | 67.0 | 40.0 | 62.5 |
| Index case, n/total (%) | 10/21 (47.6%) | 5/12 (41.7%) | 3/7 (42.9%) | 2/2 (100%) |
| Family screening, n/total (%) | 11/21 (52.4%) | 7/12 (58.3%) | 4/7 (57.1%) | 0/2 (0%) |
| ESSALUD, n (%) | 14/21 (66.7%) | 8/12 (66.7%) | 5/7 (71.4%) | 1/2 (50.0%) |
| MINSA, n (%) | 5/21 (23.8%) | 2/12 (16.7%) | 2/7 (28.6%) | 1/2 (50.0%) |
| Private, n (%) | 2/21 (9.5%) | 2/12 (16.7%) | 0/7 (0%) | 0/2 (0%) |
| Number of hospitals | 8 | 4 | 4 | 2 |
| Mortality, n (%) | 1/23 (4.3%) | 1 (7.7%) | 0 (0%) | 0 (0%) |
| Variable | n | Value |
|---|---|---|
| NIS Total, median (range) | 15 | 0.0 (0-137) |
| NIS = 0, n (%) | 15 | 11 (73.3%) |
| NIS > 0, n (%) | 15 | 4 (26.7%) |
| Norfolk QOL-DN, median | 18 | 6.0 |
| Norfolk <20 (minimal), n (%) | 18 | 10 (55.6%) |
| Norfolk 20-39 (mild), n (%) | 18 | 4 (22.2%) |
| Norfolk ≥40 (mod-severe), n (%) | 18 | 4 (22.2%) |
| Variable | n | Value |
|---|---|---|
| COMPASS-31, median | 18 | 6.0 |
| SUDOSCAN Feet, µS - median (IQR) | 6 | 66.0 (54.0-71.0) |
| SUDOSCAN Hands, µS - median (IQR) | 6 | 62.0 (59.0-73.0) |
| Dysautonomia (COMPASS-31 >31), n (%) | 13 | 3 (23.1%) |
| Variable | n | Value | Abnormal % |
|---|---|---|---|
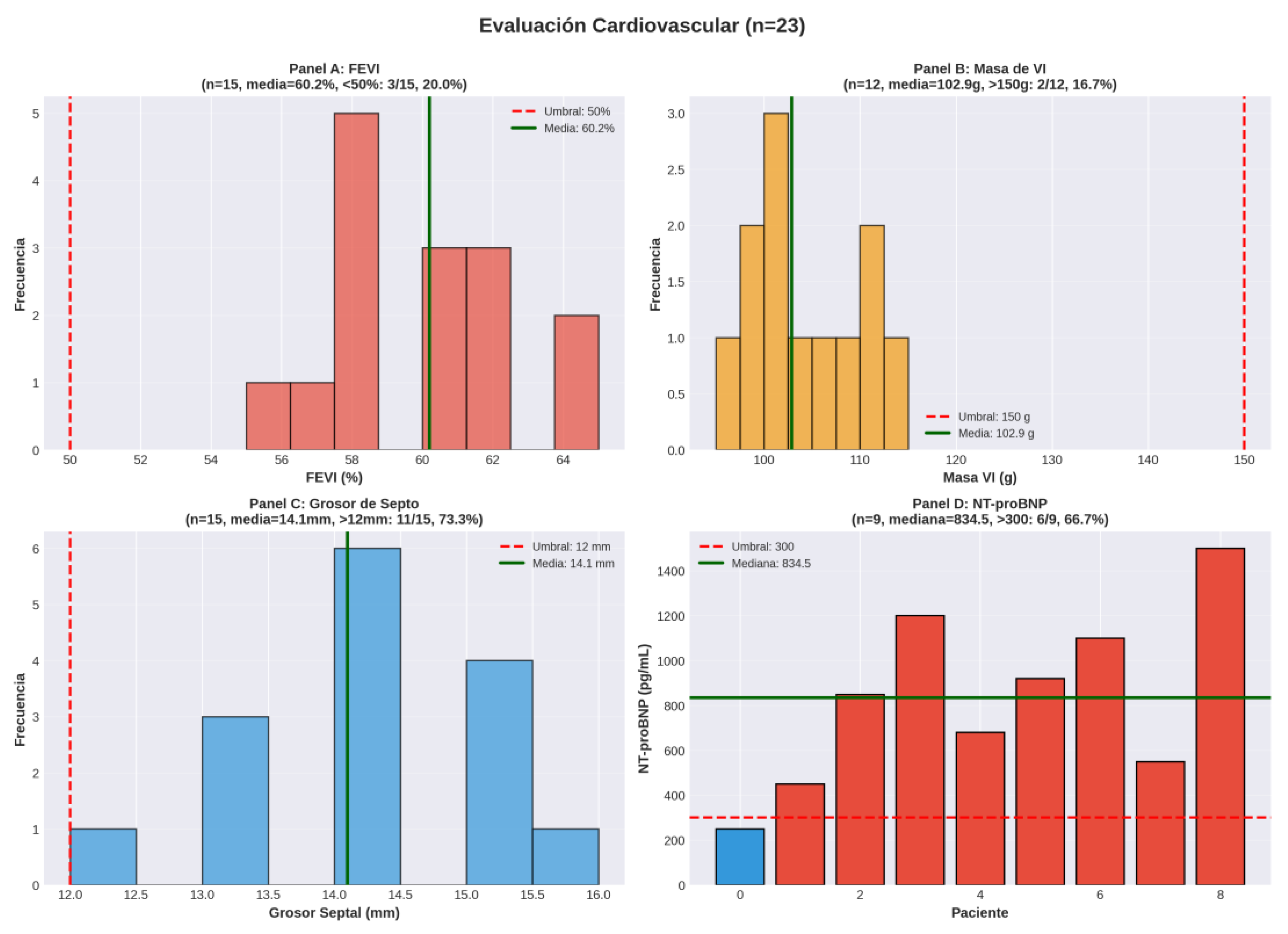
| LVEF %, mean (median) | 15 | 60.2% (61.0%) | |
| LVEF <50% | 16 | 3/15 (20.0%) | 20.0% |
| GLS %, mean | 15 | -15.5% | |
| LV Mass, g - mean (median) | 12 | 102.9 (84.5) | |
| LV Mass >150g | 13 | 2/12 (16.7%) | 16.7% |
| TAPSE, mm - mean | 15 | 18.8 | |
| Septal thickness, mm - mean (median) | 15 | 14.1 (15.0) | |
| Posterior wall, mm - mean | 15 | 12.9 | |
| E/e’ ratio - mean (median) | 15 | 15.0 (13.8) | |
| Apical sparing (cherry on top) | 16 | 9/15 (60.0%) | 60.0% |
| Troponin T, ng/L - median | 9 | 47.0 | |
| Troponin T >14 ng/L | 9 | 8/9 (88.9%) | 88.9% |
| NT-proBNP, pg/mL - median (range) | 9 | 834.5 (85.9-5096.0) | |
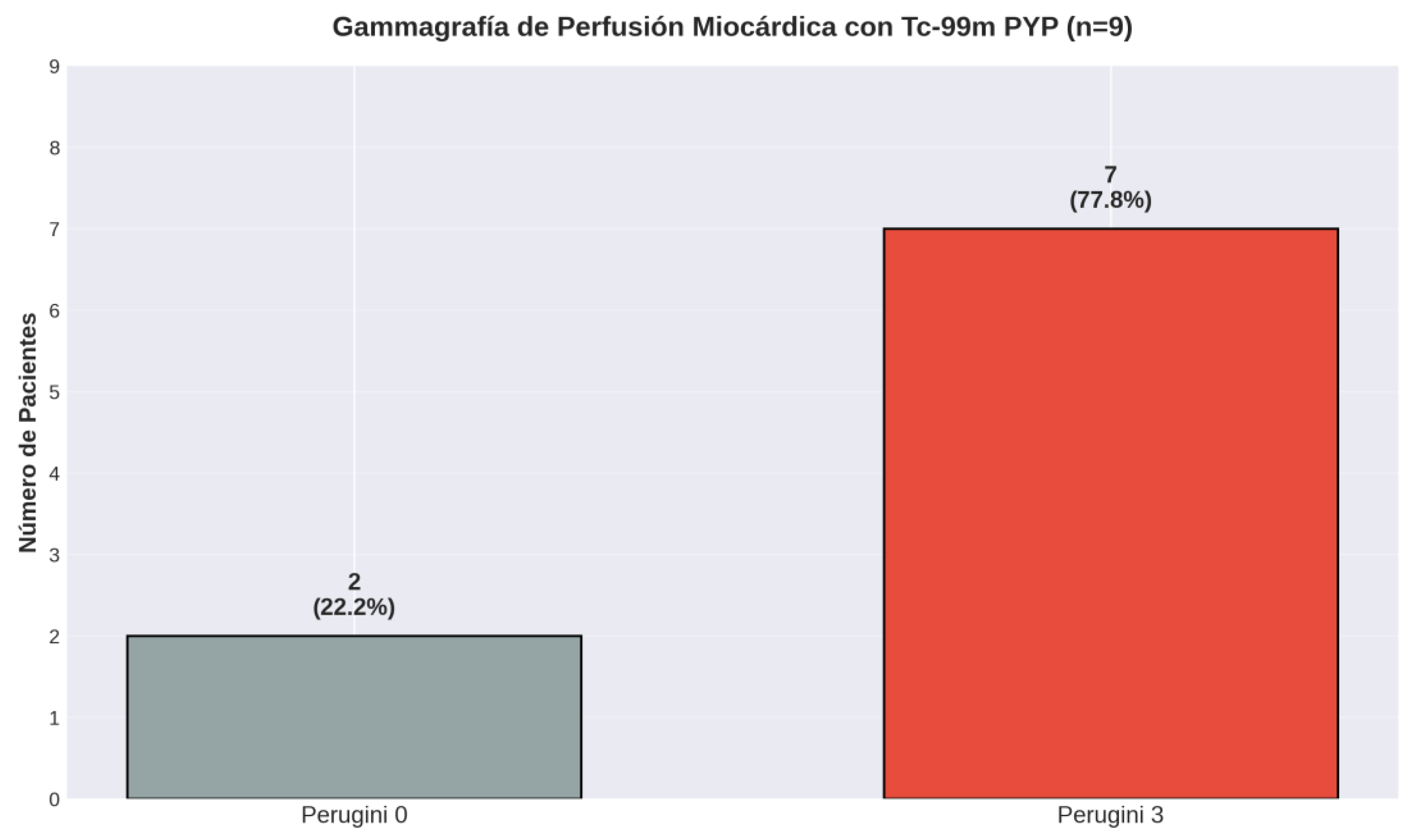
| Tc99m-PYP Perugini grade 3 | 9 | 7/9 (77.8%) | 77.8% |
| Tc99m-PYP Perugini grade 0 | 9 | 2/9 (22.2%) | 22.2% |
| ECG Sinus rhythm | 12 | 10/12 (83.3%) | 83.3% |
| ECG Atrial fibrillation | 12 | 1/12 (8.3%) | 8.3% |
| ECG Atrial flutter | 12 | 1/12 (8.3%) | 8.3% |
| Low voltage ECG | 12 | 7/12 (58.3%) | 58.3% |
| Pacemaker implantation | 11 | 1/11 (9.1%) | 9.1% |
| Phenotype | Total n=23 (%) | Val142Ile (n=13) | Ala65Val (n=8) | Val50Met (n=2) |
|---|---|---|---|---|
| Preclinical | 8 (34.8%) | 7 (53.8%) | 1 (12.5%) | 0 |
| Mixed | 9 (39.1%) | 3 (23.1%) | 4 (50.0%) | 2 (100%) |
| Cardiac | 5 (21.7%) | 3 (23.1%) | 2 (25.0%) | 0 |
| Neurological | 1 (4.3%) | 0 | 1 (12.5%) | 0 |
| Variable | n | Value |
|---|---|---|
| Norfolk QOL-DN - median (IQR) | 18 | 6.0 (0.0-32.0) |
| <20 (minimal), n (%) | 18 | 10 (55.6%) |
| 20-39 (mild), n (%) | 18 | 4 (22.2%) |
| ≥40 (moderate-severe), n (%) | 18 | 4 (22.2%) |
| KCCQ - Positive (normal), n (%) | 6 | 5 (83.3%) |
| KCCQ - Negative (abnormal), n (%) | 6 | 1 (16.7%) |
| Variable | n evaluated | Positive n (%) |
|---|---|---|
| Hypertension | 14 | 7 (50.0%) |
| Diabetes mellitus | 13 | 1 (7.7%) |
| Arrhythmia | 13 | 3 (23.1%) |
| Renal disease | 13 | 2 (15.4%) |
| Dyslipidemia | 13 | 0 (0%) |
| Hypothyroidism | 13 | 0 (0%) |
| Stroke | 13 | 1 (7.7%) |
| Smoking history | 13 | 0 (0%) |
| Carpal tunnel syndrome | 13 | 3 (23.1%) |
| Macroglossia | 13 | 1 (7.7%) |
| Purpura | 13 | 0 (0%) |
Figures
References
- Adams, D; Koike, H; Slama, M; Coelho, T. Hereditary transthyretin amyloidosis: a model of medical progress for a fatal disease. Nat Rev Neurol. 2019, 15, 387–404. [Google Scholar] [CrossRef]
- Sekijima, Y. Transthyretin (ATTR) amyloidosis: clinical spectrum, molecular pathogenesis and disease-modifying treatments. J Neurol Neurosurg Psychiatry 2015, 86, 1036–1043. [Google Scholar] [CrossRef]
- Andrade, C. A peculiar form of peripheral neuropathy: familiar atypical generalized amyloidosis with special involvement of the peripheral nerves. Brain 1952, 75, 408–427. [Google Scholar] [CrossRef]
- Maurer, MS; Hanna, M; Grogan, M; et al. Genotype and phenotype of transthyretin cardiac amyloidosis: THAOS (Transthyretin Amyloid Outcome Survey). J Am Coll Cardiol. 2016, 68, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Araujo, BN; et al. Clinical and genetic profiles of patients with hereditary and wild-type transthyretin amyloidosis: the REACT-SP registry, São Paulo, Brazil. Orphanet J Rare Dis. 2024, 19, 281. [Google Scholar]
- Saez, MS; Aguirre, MA; Pérez de Arenaza, D; Sorroche, P; Nucifora, E; Posadas-Martinez, ML. Epidemiology of variant transthyretin amyloidosis at a reference center in Argentina. Mol Genet Genomic Med. 2023, 11, e2196. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Duarte, A; Soto, KC; Martinez-Banos, D; et al. Amyloidosis due to TTR mutations in Mexico with 4 distinct genotypes in the index cases. Orphanet J Rare Dis. 2018, 13, 107. [Google Scholar] [CrossRef]
- Maurer, MS; Schwartz, JH; Gundapaneni, B; et al. Tafamidis treatment for patients with transthyretin amyloid cardiomyopathy. N Engl J Med. 2018, 379, 1007–1016. [Google Scholar] [CrossRef]
- Adams, D; Gonzalez-Duarte, A; O’Riordan, WD; et al. Patisiran, an RNAi therapeutic, for hereditary transthyretin amyloidosis. N Engl J Med. 2018, 379, 11–21. [Google Scholar] [CrossRef]
- Benson, MD; Waddington-Cruz, M; Berk, JL; et al. Inotersen treatment for patients with hereditary transthyretin amyloidosis. N Engl J Med. 2018, 379, 22–31. [Google Scholar] [CrossRef]
- Gillmore, JD; Gane, E; Taubel, J; et al. CRISPR-Cas9 in vivo gene editing for transthyretin amyloidosis. N Engl J Med. 2021, 385, 493–502. [Google Scholar]
- Sletten, DM; Suarez, GA; Low, PA; et al. COMPASS 31: a refined and abbreviated Composite Autonomic Symptom Score. Mayo Clin Proc. 2012, 87, 1196–1201. [Google Scholar] [CrossRef]
- Vinik, AI; Nevoret, ML; Casellini, C. The new age of sudomotor function testing: a sensitive and specific biomarker for diagnosis, estimation of severity, monitoring progression, and regression in response to intervention. Rev Endocr Metab Disord. 2008, 9, 149–159. [Google Scholar] [CrossRef]
- Phelan, D; Collier, P; Thavendiranathan, P; et al. Relative apical sparing of longitudinal strain using two-dimensional speckle-tracking echocardiography is both sensitive and specific for the diagnosis of cardiac amyloidosis. Heart 2012, 98, 1442–1448. [Google Scholar] [CrossRef]
- Kittleson, MM; Maurer, MS; Ambardekar, AV; et al. Cardiac amyloidosis: evolving diagnosis and management: a scientific statement from the American Heart Association. Circulation 2020, 142, e7–e22. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, DR; Alexander, AA; Tagoe, C; et al. Prevalence of the amyloidogenic transthyretin (TTR) V122I allele in 14,333 African-Americans. Amyloid. 2015, 22, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Castellar-Leones, SM; et al. Clinical differential factors in patients with hATTR carrying Val142Ile and Ser43Asn variants. Orphanet J Rare Dis. 2024, 19, 156. [Google Scholar]
- Ruberg, FL; Grogan, M; Hanna, M; Kelly, JW; Maurer, MS. Transthyretin amyloid cardiomyopathy: JACC state-of-the-art review. J Am Coll Cardiol. 2019, 73, 2872–2891. [Google Scholar] [CrossRef]
- Thimm, A; Oubari, S; Hoffmann, J; et al. A novel TTR mutation (p.Ala65Val) underlying late-onset hereditary transthyretin (ATTRv) amyloidosis with mixed cardiac and neuropathic phenotype: a case report and review. Amyloid 2022, 29, 67–71. [Google Scholar] [CrossRef]
- Rapezzi, C; Quarta, CC; Obici, L; et al. Disease profile and differential diagnosis of hereditary transthyretin-related amyloidosis with exclusively cardiac phenotype: an Italian perspective. Eur Heart J. 2013, 34, 520–528. [Google Scholar] [CrossRef]
- Vinik, AI; Casellini, CM; Parson, HK; et al. Norfolk QOL-DN: validation of a patient reported outcome measure in transthyretin familial amyloid polyneuropathy. J Peripher Nerv Syst. 2014, 19, 104–114. [Google Scholar] [CrossRef]
- Conceição, I; González-Duarte, A; Obici, L; et al. “Red-flag” symptom clusters in transthyretin familial amyloid polyneuropathy. J Peripher Nerv Syst. 2016, 21, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Perugini, E; Guidalotti, PL; Salvi, F; et al. Noninvasive etiologic diagnosis of cardiac amyloidosis using 99mTc-3,3-diphosphono-1,2-propanodicarboxylic acid scintigraphy. J Am Coll Cardiol. 2005, 46, 1076–1084. [Google Scholar] [CrossRef] [PubMed]
- Gillmore, JD; Maurer, MS; Falk, RH; et al. Nonbiopsy diagnosis of cardiac transthyretin amyloidosis. Circulation 2016, 133, 2404–2412. [Google Scholar] [CrossRef] [PubMed]
- Conceição, I; Damy, T; Obici, L; et al. Genetic counselling and testing for familial transthyretin (TTR) amyloidosis. J Neurol Sci. 2019, 404, 63–69. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




