Submitted:
16 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
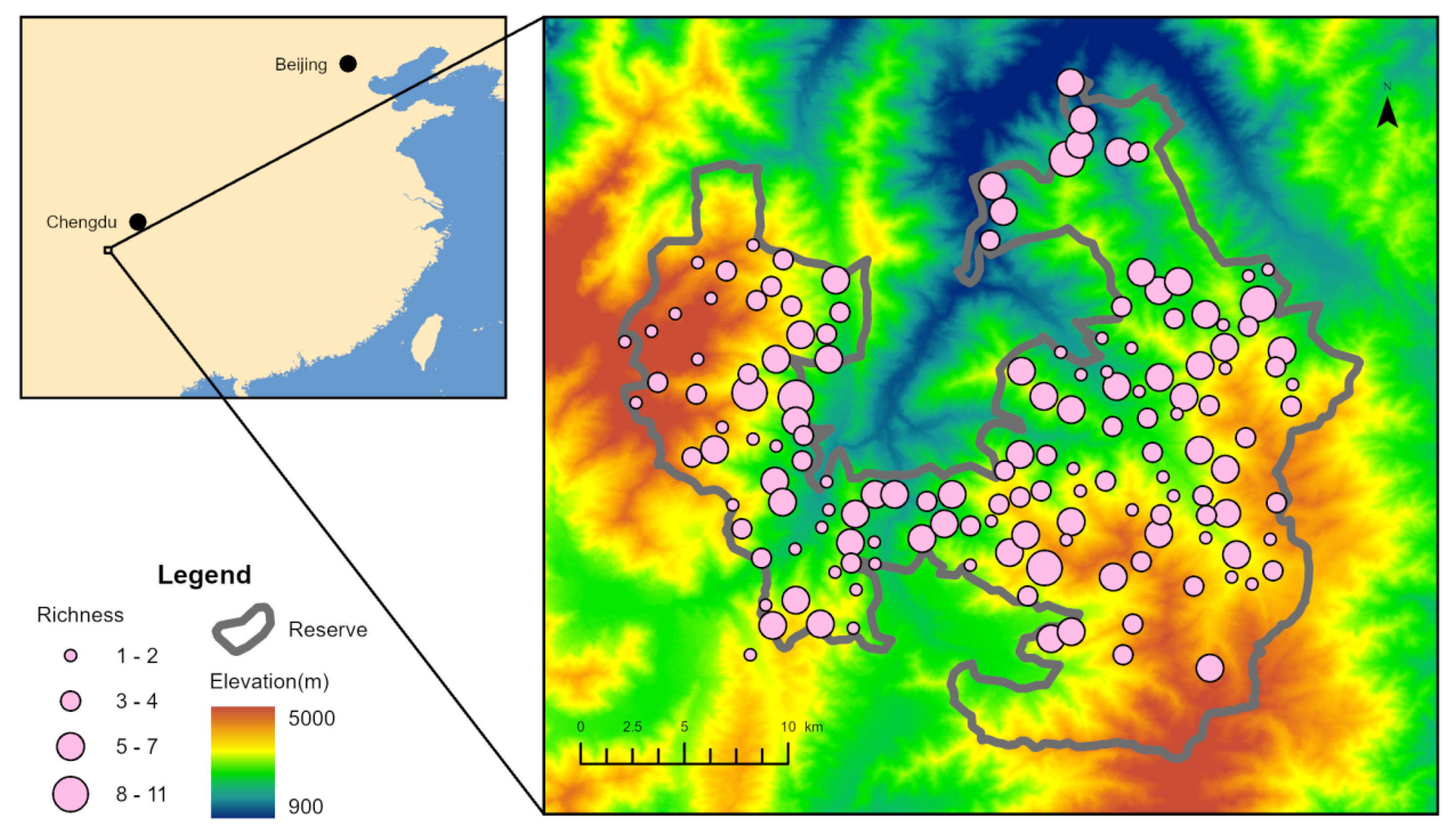
2.1. Study Area
2.2. Infrared Camera Deployment
2.3. Environmental Data
2.4. Analytical Framework and Data Structure
2.5. Beta-Diversity and Null Model Analyses
2.6. Clustering Analysis
2.7. NMDS Ordination and Environmental Fitting
2.8. Occupancy Analysis
2.9. Clustering of Species Environmental Response Coefficients
2.10. Mantel Analysis
3. Results
3.1. Community Beta Diversity
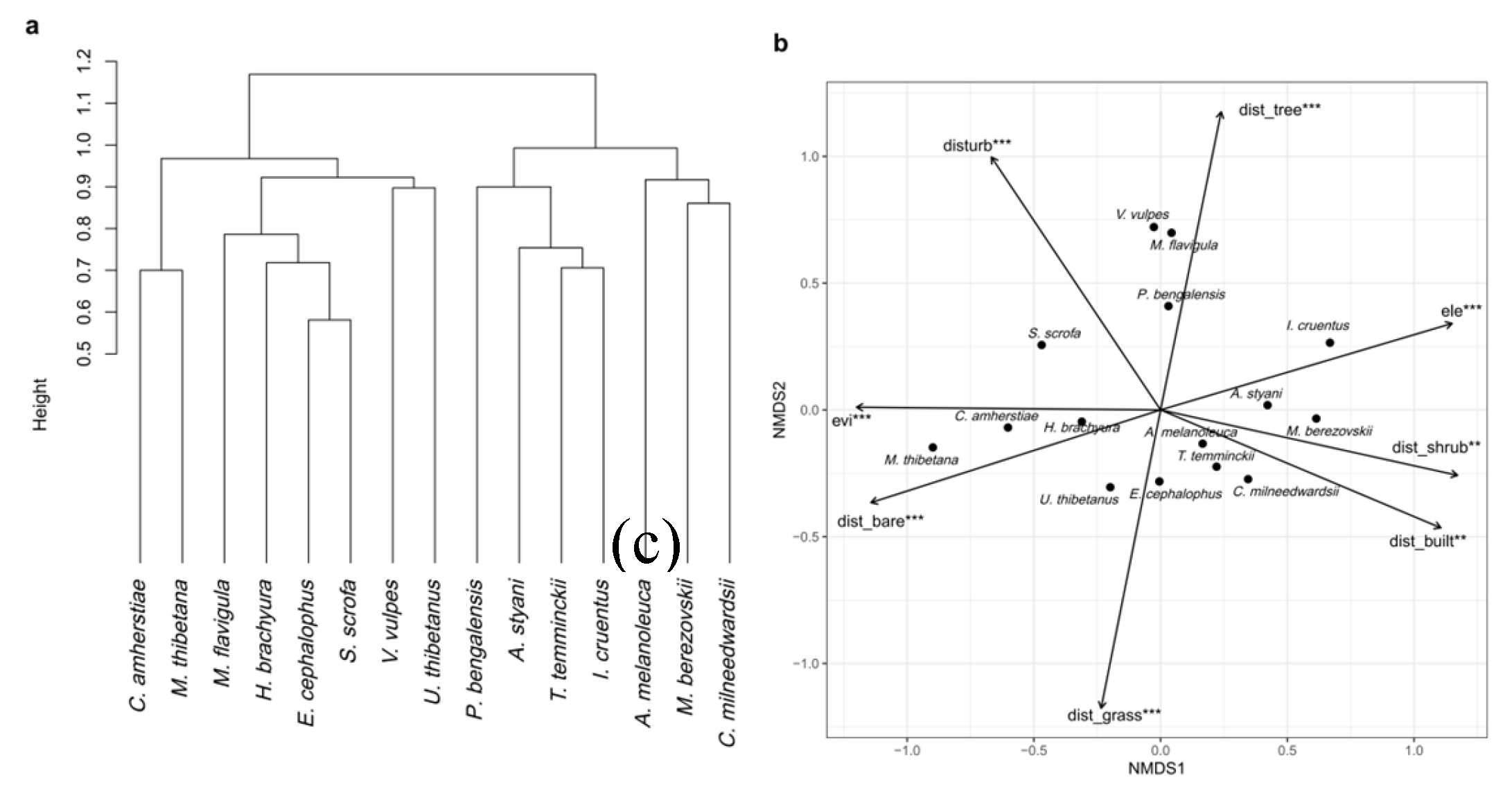
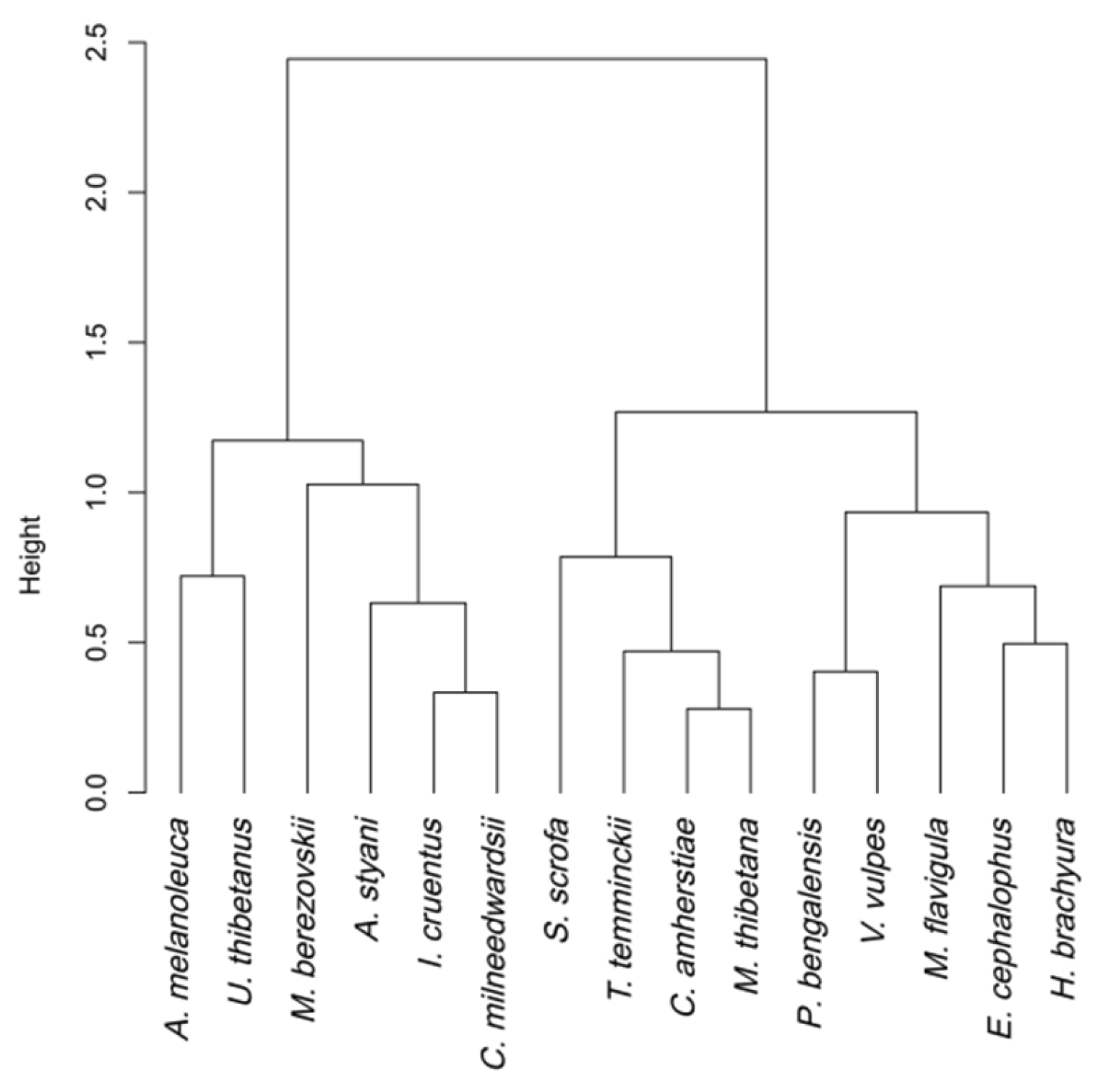
3.2. Species Similarity and Clustering
3.3. NMDS Ordination and Environmental Gradients
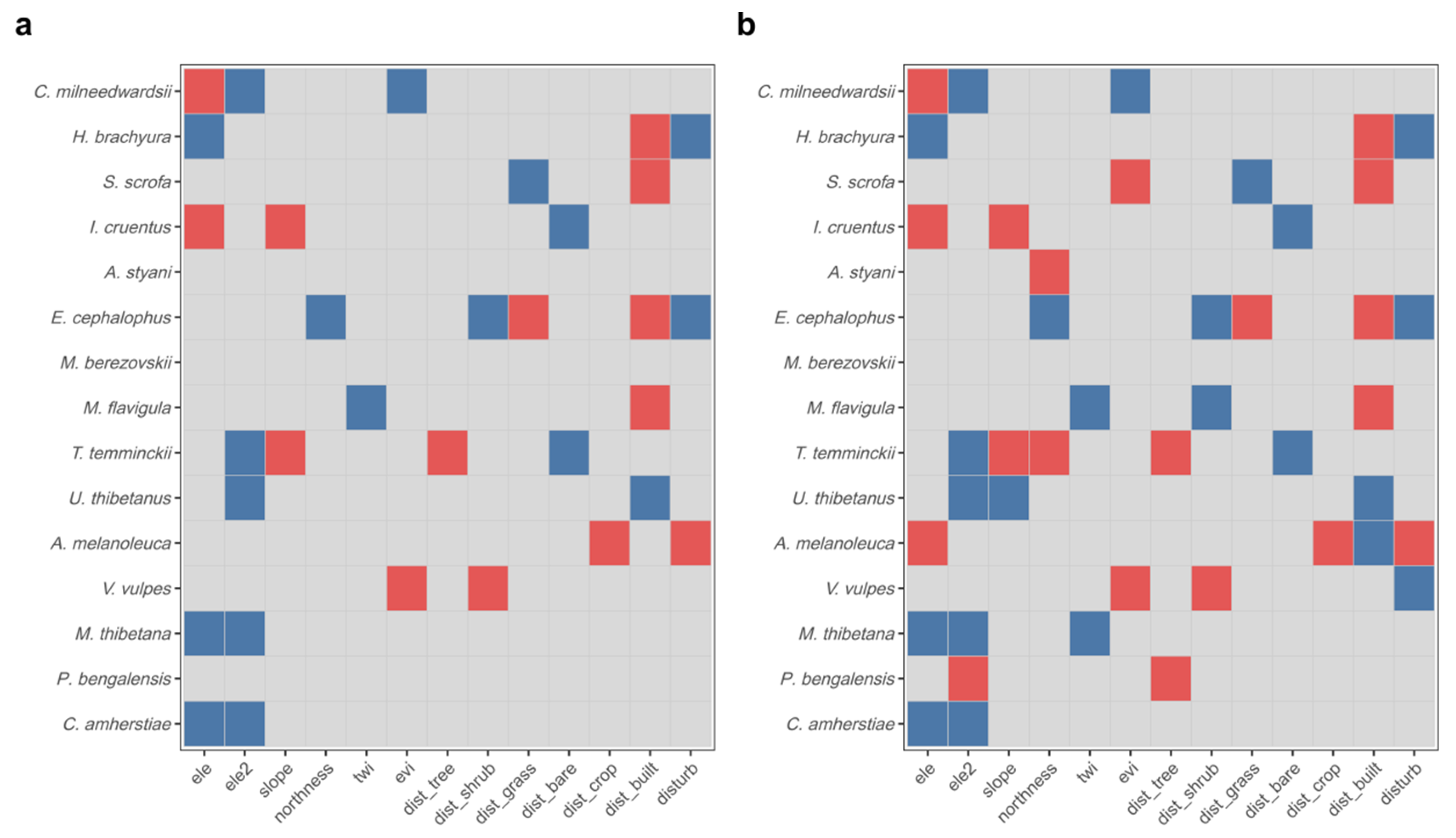
3.4. Occupancy Analysis
3.5. Mantel Test
4. Discussion
4.1. Environmental Gradients and Community Turnover
4.2. Species-Specific Environmental Responses and Niche Differentiation
4.3. Multiple Ecological Dimensions Revealed by Different Data Types
4.4. Human Disturbance
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lewis, S.L.; Maslin, M.A. Defining the Anthropocene. Nature 2015, 519, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Finn, C.; Grattarola, F.; Pincheira-Donoso, D. More losers than winners: Investigating Anthropocene defaunation through the diversity of population trends. Biological Reviews 2023, 98, 1732–1748. [Google Scholar] [CrossRef] [PubMed]
- Barnosky, A.D.; Matzke, N.; Tomiya, S.; Wogan, G.O.U.; Swartz, B.; Quental, T.B.; Marshall, C.; McGuire, J.L.; Lindsey, E.L.; Maguire, K.C.; et al. Has the Earth’s sixth mass extinction already arrived? Nature 2011, 471, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Cowie, R.H.; Bouchet, P.; Fontaine, B. The Sixth Mass Extinction: Fact, fiction or speculation? Biological Reviews 2022, 97, 640–663. [Google Scholar] [CrossRef]
- Wang, S.; Yoder, A.D. Synergies between speciation and conservation science yield novel insights for mitigating the biodiversity crisis of the Anthropocene. Proc. Natl. Acad. Sci. U.S.A. 2025, 122, e2500713122. [Google Scholar] [CrossRef]
- Maxwell, S.L.; Cazalis, V.; Dudley, N.; Hoffmann, M.; Rodrigues, A.S.L.; Stolton, S.; Visconti, P.; Woodley, S.; Kingston, N.; Lewis, E.; et al. Area-based conservation in the twenty-first century. Nature 2020, 586, 217–227. [Google Scholar] [CrossRef]
- Gaston, K.J.; Jackson, S.F.; Nagy, A.; Cantú-Salazar, L.; Johnson, M. Protected Areas in Europe: Principle and Practice. Annals of the New York Academy of Sciences 2008, 1134, 97–119. [Google Scholar] [CrossRef]
- Xu, W.; Viña, A.; Kong, L.; Pimm, S.L.; Zhang, J.; Yang, W.; Xiao, Y.; Zhang, L.; Chen, X.; Liu, J.; et al. Reassessing the conservation status of the giant panda using remote sensing. Nature Ecology & Evolution 2017, 1, 1635–1638. [Google Scholar] [CrossRef]
- Peters, A.; Smith, A.F.; Henrich, M.; Dormann, C.F.; Heurich, M. Temporal displacement of the mammal community in a protected area due to hunting and recreational activities. Ecological Applications 2025, 35, e70118. [Google Scholar] [CrossRef]
- Pang, D.; Wan, Y.; Chen, Y.; Li, Y.; Wang, L.; Chen, G.; Pu, F.; Ding, J.; Li, J.; Dai, Q.; et al. Climate change and conservation strategies for the Anhui musk deer: Habitat suitability and climate refuges in the Dabie Mountains. Landscape Ecology 2025, 40, 94. [Google Scholar] [CrossRef]
- Zanón Martínez, J.I.; Iranzo, E.C.; Travaini, A.; McNitt, D.C.; Mansilla, A.P.; Llanos, R.; Kelly, M.J. Puma density, habitat use, and activity patterns across a mosaic landscape of ranches, game reserves, and a protected area in central Argentina. European Journal of Wildlife Research 2023, 69, 89. [Google Scholar] [CrossRef]
- Yoon, H.S.; Vijay, V.; Armsworth, P.R. Accounting for spatial heterogeneity in the added conservation value of land protection when prioritizing protected areas. Conservation Biology 2022, 36, e13960. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Liang, Z.; Ye, J.; Li, J.; Yang, F.; Li, Z.; Cui, D.; Yan, L.; Li, B.; Hu, J. Conservation implications of climatically heterogeneous areas for species diversity in a biodiversity hotspot. Journal of Environmental Management 2024, 371, 123275. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, R.J.; Fortin, M.-J. Spatial Prioritization and Conservation Planning. In Spatial Ecology and Conservation Modeling: Applications with R; Fletcher, J.R.J., Fortin, M.-J., Eds.; Springer Nature Switzerland: Cham, 2026; pp. 609–654. [Google Scholar]
- Cumming, G.S.; Allen, C.R.; Ban, N.C.; Biggs, D.; Biggs, H.C.; Cumming, D.H.M.; De Vos, A.; Epstein, G.; Etienne, M.; Maciejewski, K.; et al. Understanding protected area resilience: A multi-scale, social-ecological approach. Ecological Applications 2015, 25, 299–319. [Google Scholar] [CrossRef]
- Hartig, F.; Abrego, N.; Bush, A.; Chase, J.M.; Guillera-Arroita, G.; Leibold, M.A.; Ovaskainen, O.; Pellissier, L.; Pichler, M.; Poggiato, G.; et al. Novel community data in ecology-properties and prospects. Trends in Ecology & Evolution 2024, 39, 280–293. [Google Scholar] [CrossRef]
- Patterson, B.D. The Principle of Nested Subsets and Its Implications for Biological Conservation. Conservation Biology 1987, 1, 323–334. [Google Scholar] [CrossRef]
- Nicholson, E.; Possingham, H.P. Objectives for Multiple-Species Conservation Planning. Conservation Biology 2006, 20, 871–881. [Google Scholar] [CrossRef]
- Jones, H.H.; Barreto, E.; Murillo, O.; Robinson, S.K. Turnover-driven loss of forest-dependent species changes avian species richness, functional diversity, and community composition in Andean forest fragments. Global Ecology and Conservation 2021, 32, e01922. [Google Scholar] [CrossRef]
- Gossner, M.M.; Getzin, S.; Lange, M.; Pašalić, E.; Türke, M.; Wiegand, K.; Weisser, W.W. The importance of heterogeneity revisited from a multiscale and multitaxa approach. Biological Conservation 2013, 166, 212–220. [Google Scholar] [CrossRef]
- Ramírez-Sánchez, O.; Rodríguez-Ávila, D.; Santana-Pérez, P.; Santana-Pérez, V.; Benítez-Malvido, J.; Arena-Ortíz, M.L.; Alvarez-Añorve, M.Y.; Avila-Cabadilla, L.D. Unstructured spatial gradients in anthropogenic landscapes drive contrasting rodent and bat metacommunities, underscoring the need for multiscale conservation planning. Biological Conservation 2025, 312, 111484. [Google Scholar] [CrossRef]
- Bisht, S.; Rawat, G.S. Avian diversity and responses to altitude and human disturbance in Askot Wildlife Sanctuary, Western Himalaya. Journal for Nature Conservation 2026, 91, 127234. [Google Scholar] [CrossRef]
- Barceló, G.; Donadio, E.; Alldredge, M.W.; Pauli, J.N. Human disturbance alters the foraging and spatiotemporal activity of a large carnivore. Oecologia 2025, 207, 112. [Google Scholar] [CrossRef] [PubMed]
- Zaccarelli, N.; Riitters, K.H.; Petrosillo, I.; Zurlini, G. Indicating disturbance content and context for preserved areas. Ecological Indicators 2008, 8, 841–853. [Google Scholar] [CrossRef]
- Greco, I.; Beaudrot, L.; Sutherland, C.; Tenan, S.; Hsieh, C.; Gorczynski, D.; Sheil, D.; Brodie, J.; Firoz Ahmed, M.; Ahumada, J.; et al. Landscape-level human disturbance results in loss and contraction of mammalian populations in tropical forests. PLOS Biology 2025, 23, e3002976. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, B.R.; Keller, A.G.; Calhoun, K.L.; Barker, K.J.; Montealegre-Mora, F.; Serota, M.W.; Van Scoyoc, A.; Parker-Shames, P.; Andreozzi, C.L.; de Valpine, P. How do ecologists estimate occupancy in practice? Ecography 2024, e07402. [Google Scholar] [CrossRef]
- Traba, J.; Morales, M.B.; Silva, J.P.; Bretagnolle, V.; Devoucoux, P. Habitat Selection and Space Use. In Little Bustard: Ecology and Conservation; Bretagnolle, V., Traba, J., Morales, M.B., Eds.; Springer International Publishing: Cham, 2022; pp. 101–121. [Google Scholar]
- Perea, S.; Fandos, G.; Larsen-Gray, A.; Greene, D.U.; Chandler, R.; Castleberry, S.B. Bat winter foraging habitat use in working forests: A multispecies spatial occupancy approach. Animal Conservation 2024, 27, 478–491. [Google Scholar] [CrossRef]
- Van der Weyde, L.K.; Mbisana, C.; Klein, R. Multi-species occupancy modelling of a carnivore guild in wildlife management areas in the Kalahari. Biological Conservation 2018, 220, 21–28. [Google Scholar] [CrossRef]
- Institute of Rare Animals & Plants; China West Normal University. The Scientific Survey Report on Liziping Nature Reserve in Sichuan Province, China; China West Normal University: Nanchong, China, 2011.
- Yue, Y.; Wang, Y.; Ye, Z.; Zhang, C.; Qiu, L.; Xu, Q.; He, X.; Ma, C.; Yang, B.; Yang, Z.; et al. Ecological and Public Advantages of a Dual Flagship Strategy: Giant Panda and Snow Leopard. Diversity 2024, 16, 76. [Google Scholar] [CrossRef]
- Yue, Y.; Yang, Z.; Wei, W.; Yang, B.; Qi, D.; Gu, X.; Yang, X.; Lu, S.; Zhang, W.; Dai, Q.; et al. The effectiveness of using giant panda as a surrogate for protecting sympatric species. Journal of Environmental Management 2024, 351, 119803. [Google Scholar] [CrossRef]
- Annals of Sichuan Province Compilation Committee. Annals of Sichuan province: Geography; Chengdu Map Publishing House: Chengdu, China, 1996. [Google Scholar]
- Zanaga, D.; Van De Kerchove, R.; Daems, D.; De Keersmaecker, W.; Brockmann, C.; Kirches, G.; Wevers, J.; Cartus, O.; Santoro, M.; Fritz, S.; et al. ESA WorldCover 10 m 2021 v200.; The European Space Agency: Paris, France, 2022. [Google Scholar] [CrossRef]
- Forestry Department of Sichuan Province. The Pandas of Sichuan: The 4th survey report on giant panda in Sichuan Province; Sichuan Science and Technology Press: Chengdu, China, 2015. [Google Scholar]
- Baselga, A. Partitioning the turnover and nestedness components of beta diversity. Global Ecology and Biogeography 2010, 19, 134–143. [Google Scholar] [CrossRef]
- Baselga, A.; Orme, C.D.L. betapart: An R package for the study of beta diversity. Methods in Ecology and Evolution 2012, 3, 808–812. [Google Scholar] [CrossRef]
- Oksanen, J.; Simpson, G.L.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O'Hara, R.B.; Solymos, P.; Stevens, M.H.H.; Szoecs, E.; et al. vegan: Community Ecology Package.; 2001. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2025. [Google Scholar]
- Maechler, M.; Rousseeuw, P.; Struyf, A.; Hubert, M.; Hornik, K. cluster: Cluster Analysis Basics and Extensions. R package version 2.1.8.1 2025. [Google Scholar]
- Stan Development Team. RStan: The R interface to Stan. R package version 2.32.7. 2025. Available online: https://mc-stan.org/.
- Wan, J.-Z.; Wang, C.-J.; Wang, X. Assessing the effectiveness of global protected areas in conserving plant community completeness. Journal of Applied Ecology 2026, 63, e70223. [Google Scholar] [CrossRef]
- García-Navas, V.; Martínez-Núñez, C.; Christidis, L.; Ozgul, A. Turnover of bird species along the Nullarbor Plain: Insights from taxonomic, phylogenetic, and functional beta diversity. Ecosphere 2024, 15, e4910. [Google Scholar] [CrossRef]
- Lin, H.; Wang, X.; Zhong, M.; Tang, K.; Du, Y.; Xu, H.; Yi, J.; Liu, W.; Hu, J. Elevational patterns and assembly processes of multifaceted bird diversity in a subtropical mountain system. Journal of Biogeography 2024, 51, 1276–1289. [Google Scholar] [CrossRef]
- Körner, C. The use of ‘altitude’ in ecological research. Trends in Ecology & Evolution 2007, 22, 569–574. [Google Scholar] [CrossRef]
- Rahbek, C.; Borregaard, M.K.; Colwell, R.K.; Dalsgaard, B.; Holt, B.G.; Morueta-Holme, N.; Nogues-Bravo, D.; Whittaker, R.J.; Fjeldså, J. Humboldt’s enigma: What causes global patterns of mountain biodiversity? Science 2019, 365, 1108–1113. [Google Scholar] [CrossRef]
- Questad, E.J.; Foster, B.L. Coexistence through spatio-temporal heterogeneity and species sorting in grassland plant communities. Ecology Letters 2008, 11, 717–726. [Google Scholar] [CrossRef]
- Mausberg, N.; Dausmann, K.H.; Glos, J. In Search of Suitable Breeding Sites: Habitat Heterogeneity and Environmental Filters Determine Anuran Diversity of Western Madagascar. Animals 2023, 13, 3744. [Google Scholar] [CrossRef]
- Báldi, A. Habitat heterogeneity overrides the species–area relationship. Journal of Biogeography 2008, 35, 675–681. [Google Scholar] [CrossRef]
- Cerini, F.; Chiatante, G.; Chiocchio, A.; Mazza, V.; Piovesan, G.; Chiarucci, A.; Musiani, M.; Milazzo, M.; Maiorano, L.; Rondinini, C.; et al. Are we zoning out? Biases in the assessment of protected area zoning and a blueprint for a way forward. One Earth 2025, 8. [Google Scholar] [CrossRef]
- Linck, P.; Palomares, F.; Negrões, N.; Rossa, M.; Fonseca, C.; Couto, A.; Carvalho, J. Increasing homogeneity of Mediterranean landscapes limits the co-occurrence of mesocarnivores in space and time. Landscape Ecology 2023, 38, 3657–3673. [Google Scholar] [CrossRef]
- Zhong, H.; Liu, Y.; Sartor, C.C.; Sillero-Zubiri, C.; Cering, L.; Shi, K.; Atzeni, L. Co-existence of predators, prey, and livestock in the Central Himalaya: Insights from multi-species occupancy modeling. The Journal of Wildlife Management 2026, 90, e70124. [Google Scholar] [CrossRef]
- Willey, L.L.; Jones, M.T.; Sievert, P.R.; Akre, T.S.B.; Marchand, M.; deMaynadier, P.; Yorks, D.; Mays, J.; Dragon, J.; Erb, L.; et al. Distribution models combined with standardized surveys reveal widespread habitat loss in a threatened turtle species. Biological Conservation 2022, 266, 109437. [Google Scholar] [CrossRef]
- Farr, M.T.; O'Brien, T.; Yackulic, C.B.; Zipkin, E.F. Quantifying the conservation status and abundance trends of wildlife communities with detection–nondetection data. Conservation Biology 2022, 36, e13934. [Google Scholar] [CrossRef]
- Li, B.V.; Pimm, S.L.; Li, S.; Zhao, L.; Luo, C. Free-ranging livestock threaten the long-term survival of giant pandas. Biological Conservation 2017, 216, 18–25. [Google Scholar] [CrossRef]
- Chen, Z.; Peng, K.; Lv, X.; Luo, G.; Hu, Y.; Li, D.; Peng, B.; Ran, J. Effects of free-ranging livestock on occurrences and interspecific interactions of a wildlife community in a temperate forest. Global Ecology and Conservation 2024, 50, e02826. [Google Scholar] [CrossRef]
- Qing, J.; Xu, C.; Yang, B.; Yang, Z.; Qi, D.; Yang, X.; Gu, X.; Dai, D. Corridor design for the giant panda in the Xiaoxiangling Mountains. Acta Ecologica Sinica 2016, 36, 1125–1133. [Google Scholar] [CrossRef]
- Huang, F.; He, L.; He, K.; Dai, Q.; Zhang, K.; Tang, B.; Gu, X.; Yang, Z. Spatial and Temporal Distribution of Human Disturbance in Tuowushan Giant Panda Corridor: Survey by Camera Trap Array. Chinese Journal of Zoology 2017, 52, 403–410. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



