Submitted:
17 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
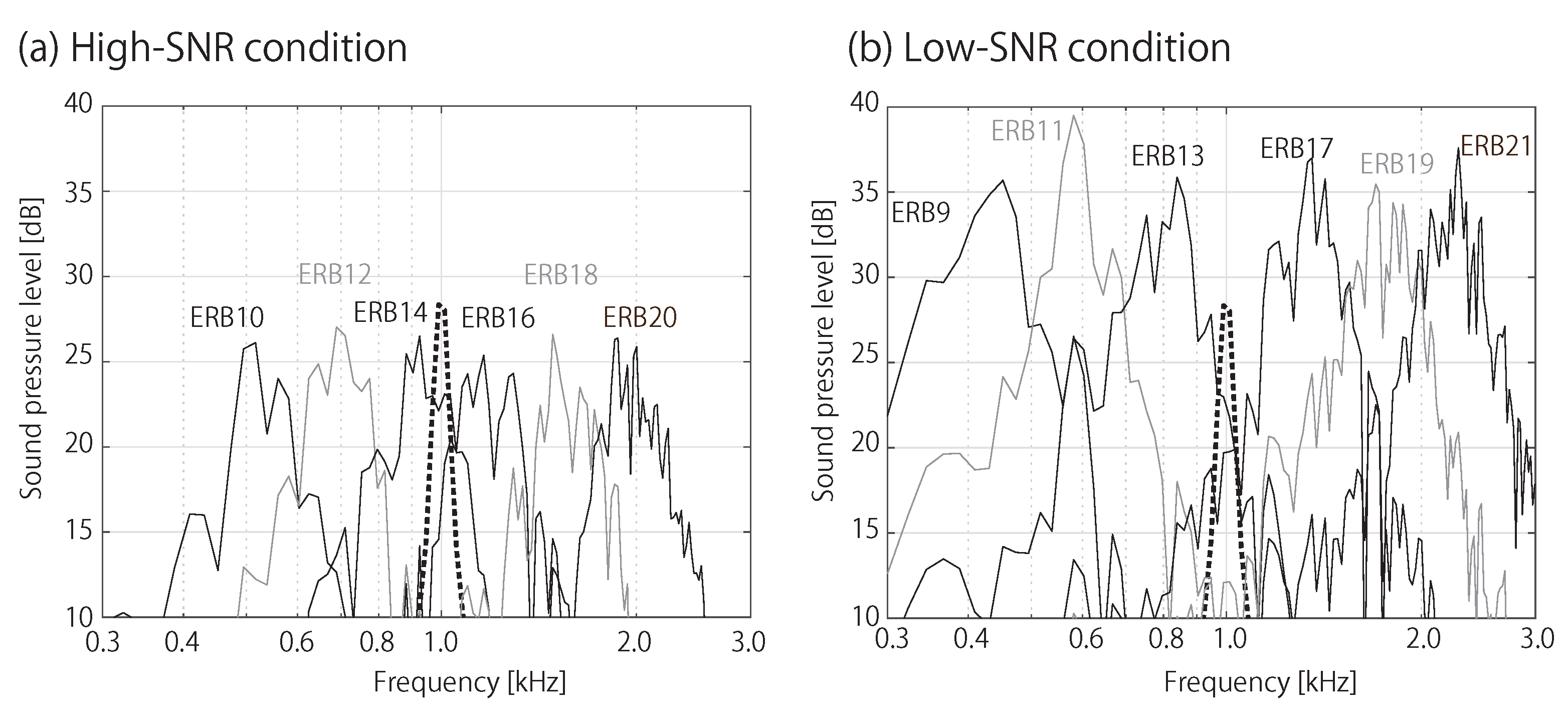
2.2. Acoustic Stimuli and Spectral Masking Conditions
2.3. Apparatus
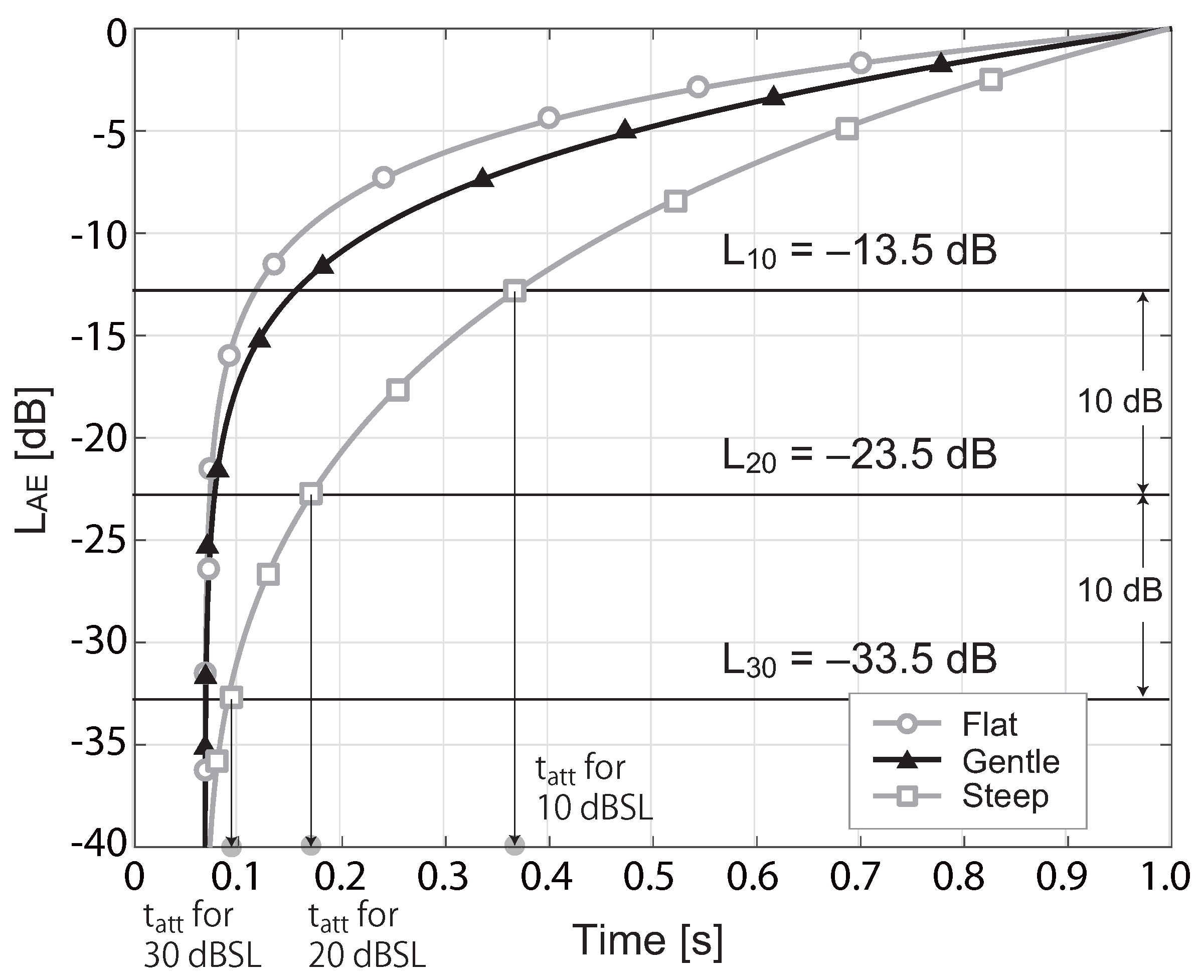
2.4. Estimation of RT
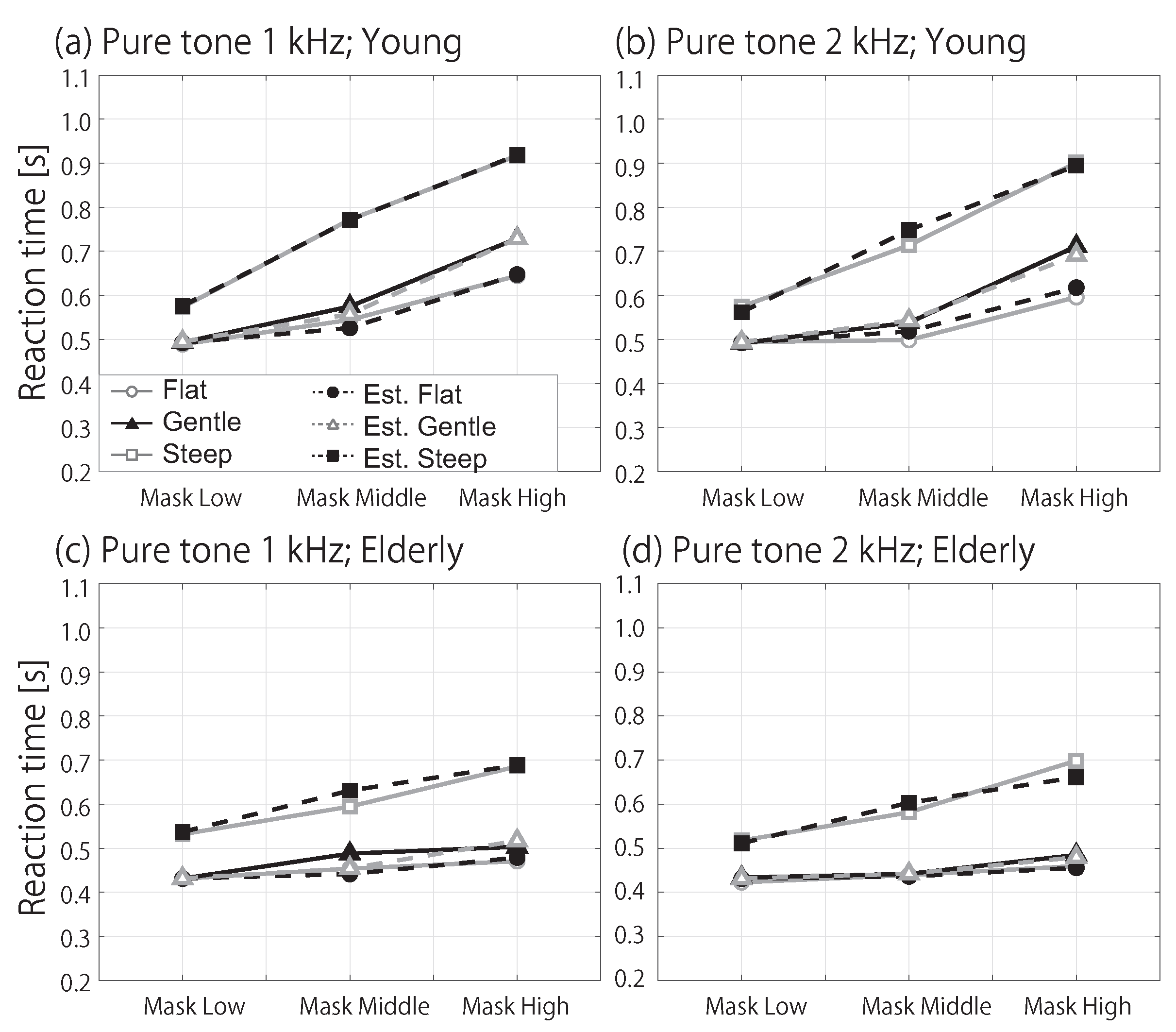
3. Results
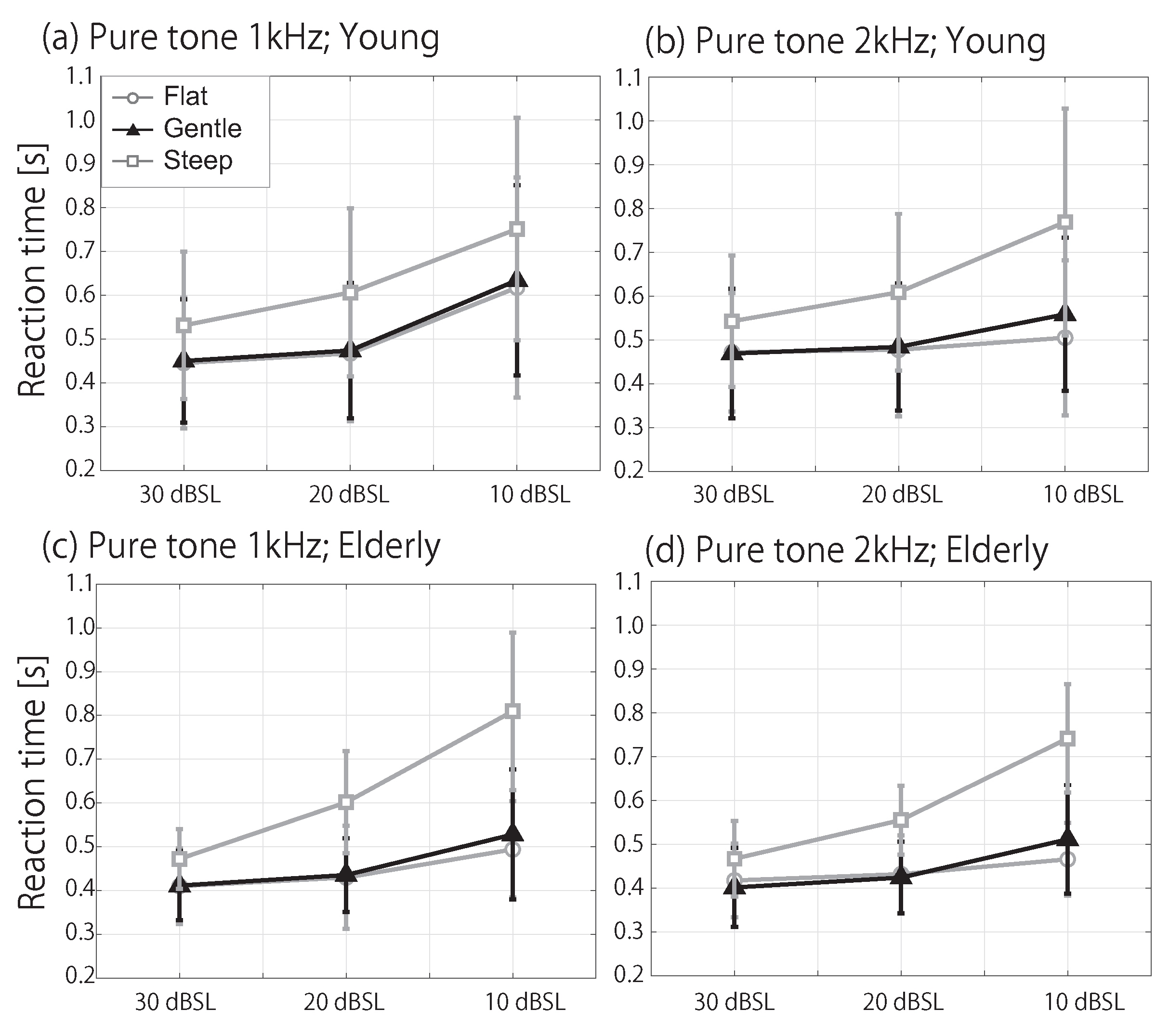
3.1. RT in the Silent Condition
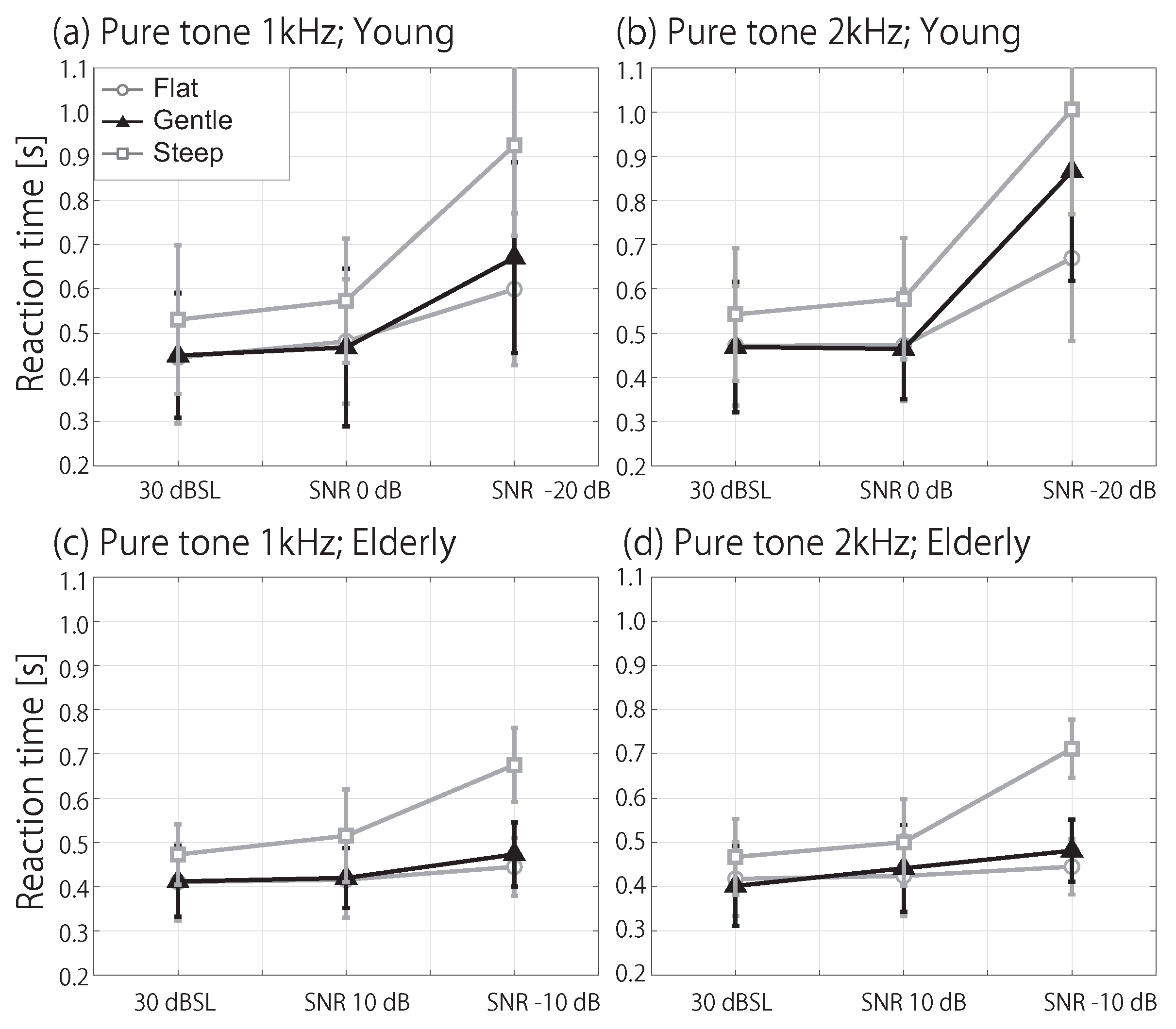
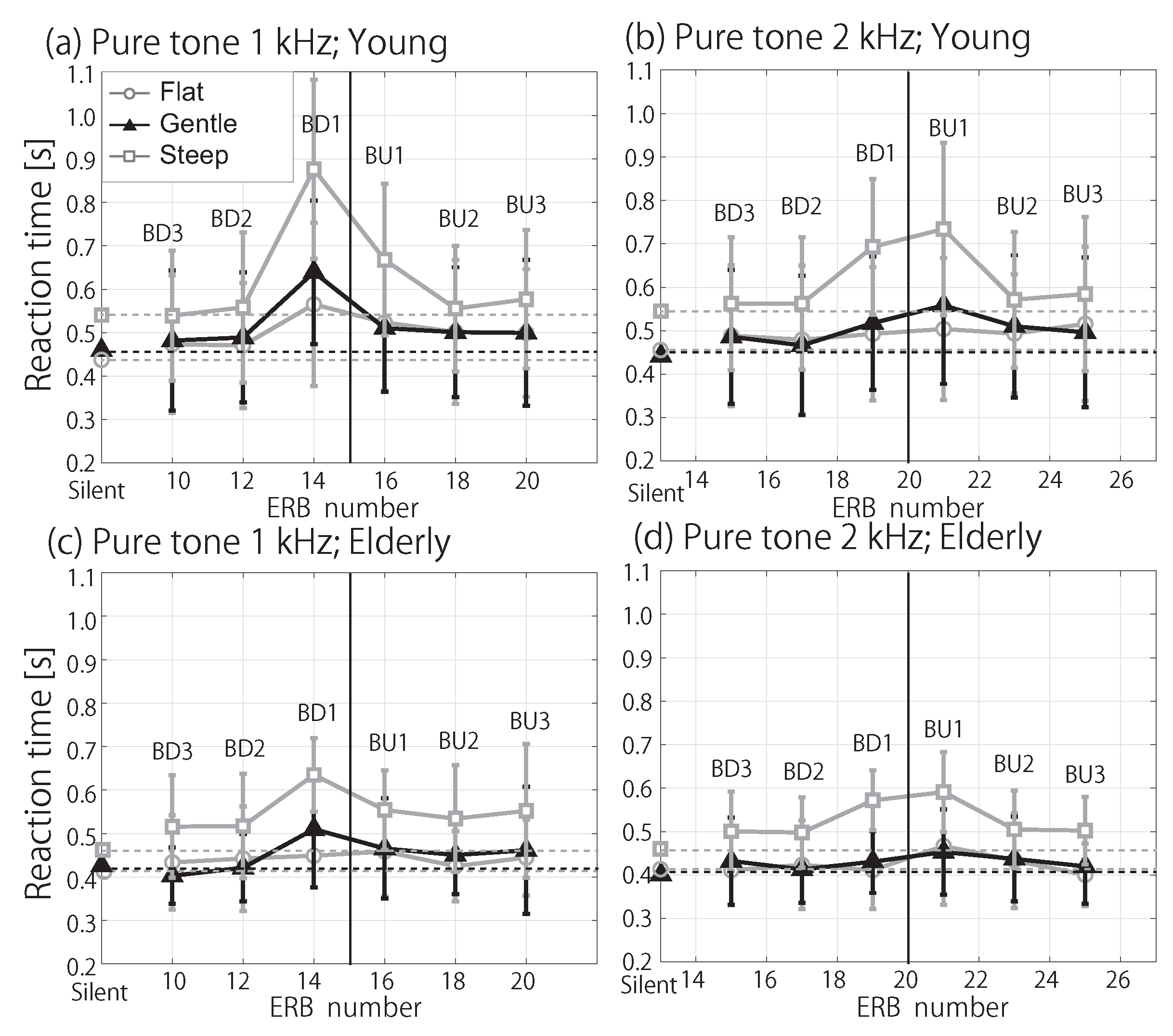
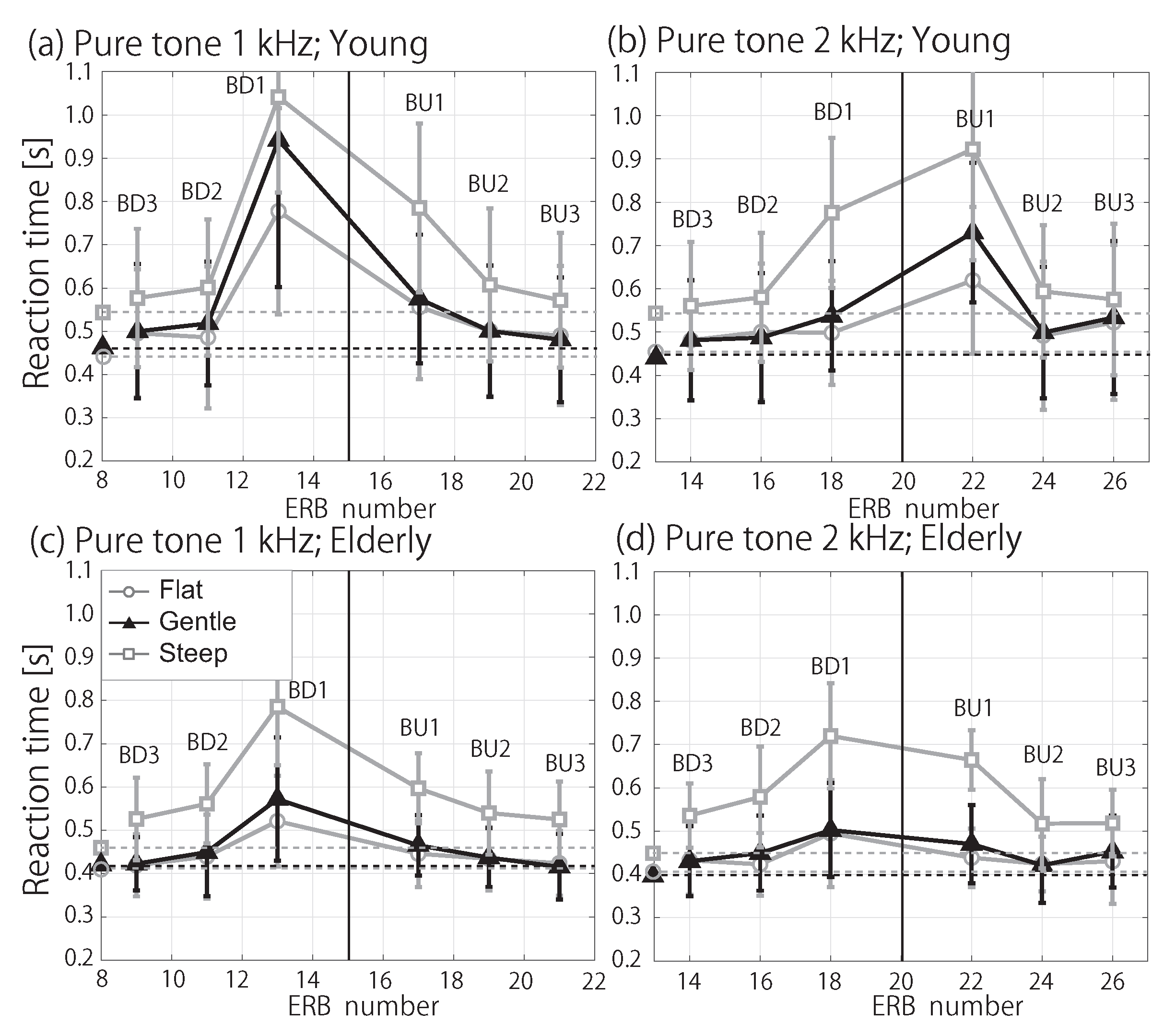
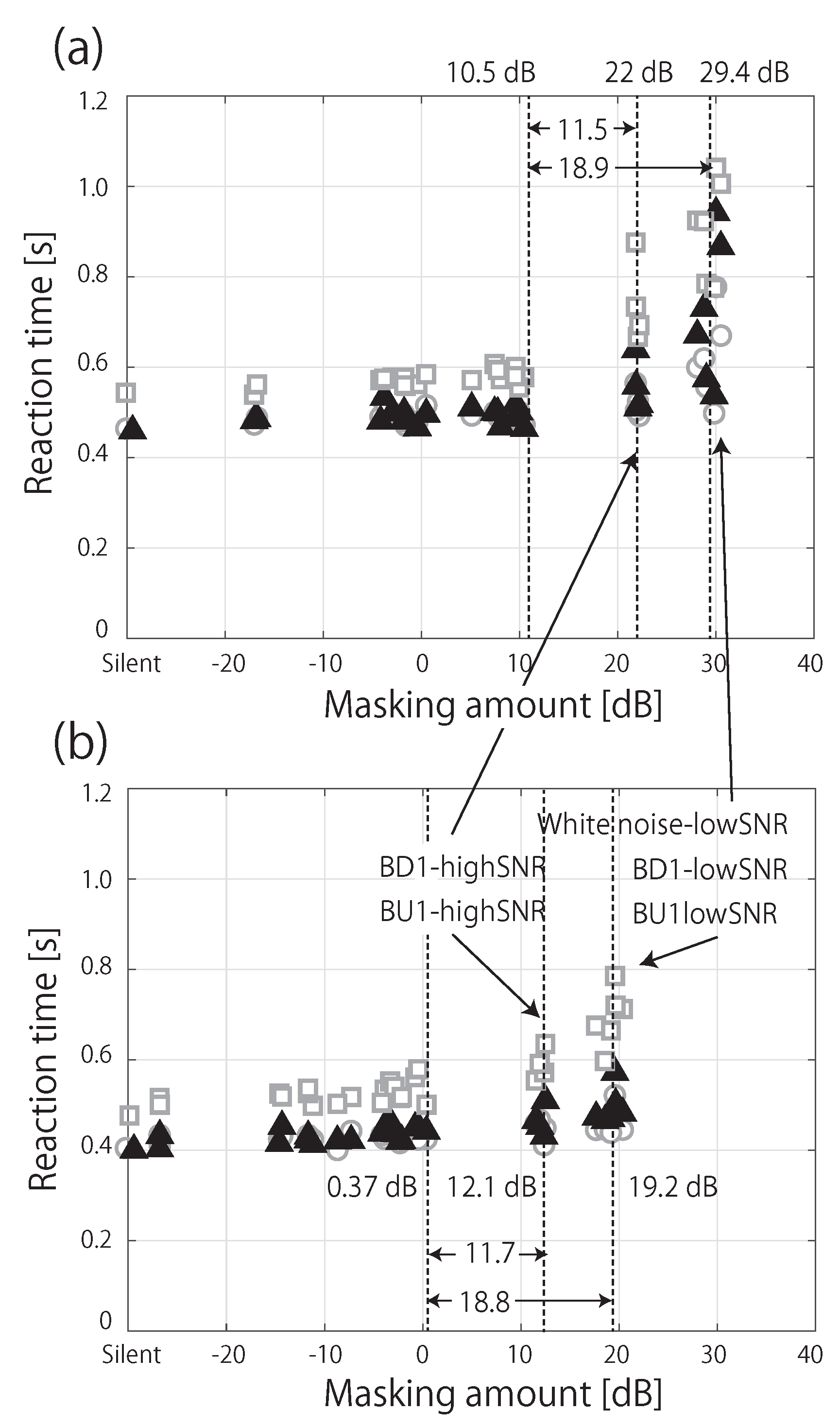
3.2. RT in Noise Conditions
4. Discussion
4.1. Temporal Integration Mechanism and the Model
4.2. Influence of Spectral Masking
4.3. Start Time for Calculating
4.4. Age-Related Effects
4.5. Practical Implications for Architectural Sound Design
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Luce, R.D. Response times: Their role in inferring elementary mental organization.; Oxford University Press, 1986. [Google Scholar]
- Chocholle, R. Variation des temps de réaction auditif en function de l’intensité à diverses frequencies. Annee. Psychol. 1940, 41, 65–124. [Google Scholar] [CrossRef]
- Burke, K.S.; Crestone, M.E. Shutts, R.E. Hearing loss and reaction time. Arch. Otolaryngol. 1965, 81, 49–56. [CrossRef] [PubMed]
- Chocholle, R.; Greenbaum, H. La sonie de sons purs partiellement masqués. Étude comparatif par une méthode d’ égalisation et par la méthode des temps de reaction. Journal de Psychologie Normal et Pathologique 1966, 63, 385–414. [Google Scholar]
- Warm, J.S.; Foulke, E. Effects of rate of signal rise and decay on reaction time to the onset and offset of acoustic stimuli. Percept. Psychophys. 1970, 7, 159–160. [Google Scholar] [CrossRef]
- Emmerich, D.S.; Pitchford, L.J.; Becker, C.A. Reaction time to tones in tonal backgrounds and a comparison of reaction time to signal onset and offset. Percept. Psychophys. 1976, 20, 210–214. [Google Scholar] [CrossRef]
- Marshall, L.; Brandt, J. The relationship between loudness and reaction time in normal hearing listeners. Acta Otoloryngol. 1980, 90, 244–249. [Google Scholar] [CrossRef]
- Kohfeld, D.L.; Santee, J.L.; Wallace, N.D. Loudness and reaction time: I. Percept. Psychophys. 1981, 29, 535–549. [Google Scholar] [CrossRef]
- Kemp, S. Reaction time to a tone in noise as a function of the signal-to-noise ratio and tone level. Percept. Psychophys. 1984, 36, 473–476. [Google Scholar] [CrossRef]
- Epstein, M.; Florentine, M. Reaction time to 1- and 4-kHz tones as a function of level. Ear and Hearing 2006, 27, 424–429. [Google Scholar] [CrossRef]
- Schlittenlacher, J.; Ellermeier, W. Simple reaction time to the onset of time-varying sounds. Atten. Percept. Psychophys. 2015, 77, 2424–2437. [Google Scholar] [CrossRef]
- Schlittenlacher, J.; Ellermeier, W.; Avci, G. Simple reaction time to the onset of time-varying sounds. Atten. Percept. Psychophys. 2017, 79, 628–636. [Google Scholar] [CrossRef] [PubMed]
- ISO 532-1; Acoustics-Methods for calculating loudness-Part 1: Zwicker method. International Organization for Standardization: Geneva, 2017.
- Florentine, M.; Buus, S.; Poulsen, T. Temporal integration of loudness as a function of level. J. Acoust. Soc. Am. 1996, 99, 1633–1644. [Google Scholar] [CrossRef]
- Buus, S.; Florentine, M.; Paulsen, T. Temporal integration of loudness, loudness discrimination and the form of the loudness function. J. Acoust. Soc. Am. 1997, 101, 669–680. [Google Scholar] [CrossRef]
- Shimokura, R.; Soeta, Y. Estimation of reaction time for birdsongs and effects of background noise and listener’s age. Appl. Acoust. 2022, 194, 1–11 (108785. [Google Scholar] [CrossRef]
- ISO 1996-1:2016; Acoustics-Description, measurement and assessment of environmental noise- Part 1: Basic quantities and assessment procedures. International Organization for Standardization: Geneva, 2016.
- Numba, S.; Kuwano, S.; Fastl, H. Loudness of non-steady-state sound. Jpn. Psychol. Res. 2008, 50, 154–166. [Google Scholar] [CrossRef]
- Munson, W.A. The growth of auditory sensation. J. Acoust. Soc. Am. 1947, 19, 584–591. [Google Scholar] [CrossRef]
- Florentine, M.; Buus, S.; Poulsen, T. Temporal integration of loudness as a function of level. J. Acoust. Soc. Am. 1996, 99, 1633–1644. [Google Scholar] [CrossRef]
- Buus, S.; Florentine, M.; Poulsen, T. Temporal integration of loudness, loudness discrimination, and the form of the loudness function. J. Acoust. Soc. Am. 1997, 101, 669–680. [Google Scholar] [CrossRef]
- Glasberg, B.R.; Moore, B.C. A model of loudness applicable to time-varying sounds. J. Acoust. Soc. Am. 2002, 50, 331–342. [Google Scholar]
- Miller, J.; Ulrich, R. Simple reaction time and statistical facilitation: A parallel grains model. Cogn. Psychol. 2003, 46, 101–151. [Google Scholar] [CrossRef] [PubMed]
- Plomp, R.; Bouman, M.A. Relationship between hearing threshold and duration for tone pulse. J. Acoust. Soc. Am. 1959, 31, 749–758. [Google Scholar] [CrossRef]
- Zwislocki, J.J. Theory of temporal auditory summation. J. Acoust. Soc. Am. 1960, 32, 1046–1060. [Google Scholar] [CrossRef]
- Zwislocki, J.J. Temporal summation of loudness: An analysis. J. Acoust. Soc. Am. 1969, 46, 431–441. [Google Scholar] [CrossRef]
- Poulsen, T. Loudness of tone pulses in a free field. J. Acoust. Soc. Am. 1981, 69, 1786–1790. [Google Scholar] [CrossRef]
- Hots, J.; Rennies, J.; Verhey, J.L. Influence of time constants and comparison on the prediction of temporal integration of loudness. Proc. Conference on Acoustics AIA-DAGA., 2013; pp. 1266–1268. [Google Scholar]
- Heil, P.; Neubauer, H. A unifying basis of auditory thresholds based on temporal summation. Proc. Natl. Acad. Sci. U.S.A. 2003, 100, 6151–6156. [Google Scholar] [CrossRef]
- Heil, P.; Matysiak, A.; Neubauer, H. A probabilistic Poisson-based model accounts for an extensive set of absolute auditory threshold measurement. Hear. Res. 2017, 353, 135–161. [Google Scholar] [CrossRef]
- Jaramillo, F.; Wiesenfeld, K. Mechanoelectrical transduction assisted by Brownian motion: a role for noise in the auditory system. Nat. Neurosci. 1998, 1, 384–388. [Google Scholar] [CrossRef]
- Henry, K.R. Noise improves transfer of near-threshold, phase-locked activity of the cochlear nerve: evidence for stochastic resonance? J. Comp. Physiol. A. 1999, 184, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.G.; Fu, Q.J.; Morse, R. Human hearing enhanced by noise. Brain. Res. 2000, 869, 251–255. [Google Scholar] [CrossRef]
- Moss, F.; Ward, L.M.; Sannita, W.G. Stochastic resonance and sensory information processing: a tutorial and review of application. Clin. Neurophysiol. 2004, 115, 267–281. [Google Scholar] [CrossRef] [PubMed]
- Ries, DT. The influence of noise type and level upon stochastic resonance in human audition. Hear. Res. 2007, 228, 136–143. [Google Scholar] [CrossRef]
- Ward, L.M.; MacLean, S.E.; Kirschner, A. Stochastic resonance modulates neural synchronization within and between cortical sources. PLoS ONE 2010, 5, e14371. [Google Scholar] [CrossRef]
- Yerkers, R.M.; Dodson, J.D. The relation of strength of stimulus to rapidity of habit-formation. J. Comp. Neurol. Psychol. 1908, 18, 459–482. [Google Scholar] [CrossRef]
- Broadbent, D.E. A reformulation of the Yerkes-Dodson low. Br. J. Moth. Stat. Psychol. 1965, 18, 145–157. [Google Scholar] [CrossRef]
- Mendl, M. Performing under pressure: Stress and cognitive function. Appl. Anim. Behav. Sci. 1999, 65, 221–244. [Google Scholar] [CrossRef]
- Heeren, W.; Hohmann, V.; Appell, J.E.; Verhey, J.L. Relation between loudness in categorical units and loudness in phons and sones. J. Acoust. Soc. Am. 2013, 133, EL314–319. [Google Scholar] [CrossRef]
- 19th February 2026. Available online: https://jp.mathworks.com/help/audio/ref/gammatonefilterbank-system-object.html.
- Moore, B.C.J.; Glasberg, B.R. Derivation of auditory filter shapes from notched-noise data. Hear. Res. 1990, 47, 103–138. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, W.M. Signals, Sound, and Sensation.; Springer Science & Business Media, 2004; p. 251. [Google Scholar]
- Levitt, H. Transformed up-down methods in psychoacoustics. J. Acoust. Soc. Am. 1971, 49, 467–477. [Google Scholar] [CrossRef]
- Garner, W.R.; Miller, G.A. The masked threshold of pure tones as a function of duration. J. Exp. Psychol. 1947, 37, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, J.E.; Stevens, S.S. The masking of pure tones and of speech by white noise. J. Acoust. Soc. Am. 1950, 22, 6–13. [Google Scholar] [CrossRef]
- Wagner, E.; Florentine, M.; Buus, S.; McCormack, J. Spectral loudness summation and simple reaction time. J. Acoust. Soc. Am. 2004, 116, 1681–1686. [Google Scholar] [CrossRef]
- Raab, D.H.; Grossberg, M. Reaction time to changes in the intensity of white noise. J. Exp. Psychol. 1965, 69, 609–612. [Google Scholar] [CrossRef] [PubMed]
- Kohfeld, D.L.; Goedecke, D.W. Intensity and predictability of background noise as determinants of simple reaction time. Bulletin of the Psychonomic Society 1978, 12, 129–132. [Google Scholar] [CrossRef]
- Glasberg, B.R.; Moore, B.C.J. Auditory filter shapes in subjects with unilateral and bilateral cochlear impairments. J. Acoust. Soc. Am. 1986, 79, 1020–1033. [Google Scholar] [CrossRef] [PubMed]
- Soeta, Y.; Ariki, A. Subjective salience of birdsong and insect song with equal sound pressure level and loudness. Int. J. Environ. Res. Public Health 2020, 17, 8858, 12 pages. [Google Scholar] [CrossRef]
- Shimokura, R.; Soeta, Y. Characteristics of train noise in above-ground and underground stations with side and island platforms. J. Sound Vib. 2011, 330, 1621–1633. [Google Scholar] [CrossRef]
| 1 | The curves shown in Figure 5 and Figure 6 of the RT study with birdsongs [16] could not be divided by the sampling rate after the discrete integrated energy was calculated, and the was normalized by the minimum audible pressure ( Pa). Consequently, the values on the vertical axis differ from those in Figure 3 of this paper. |
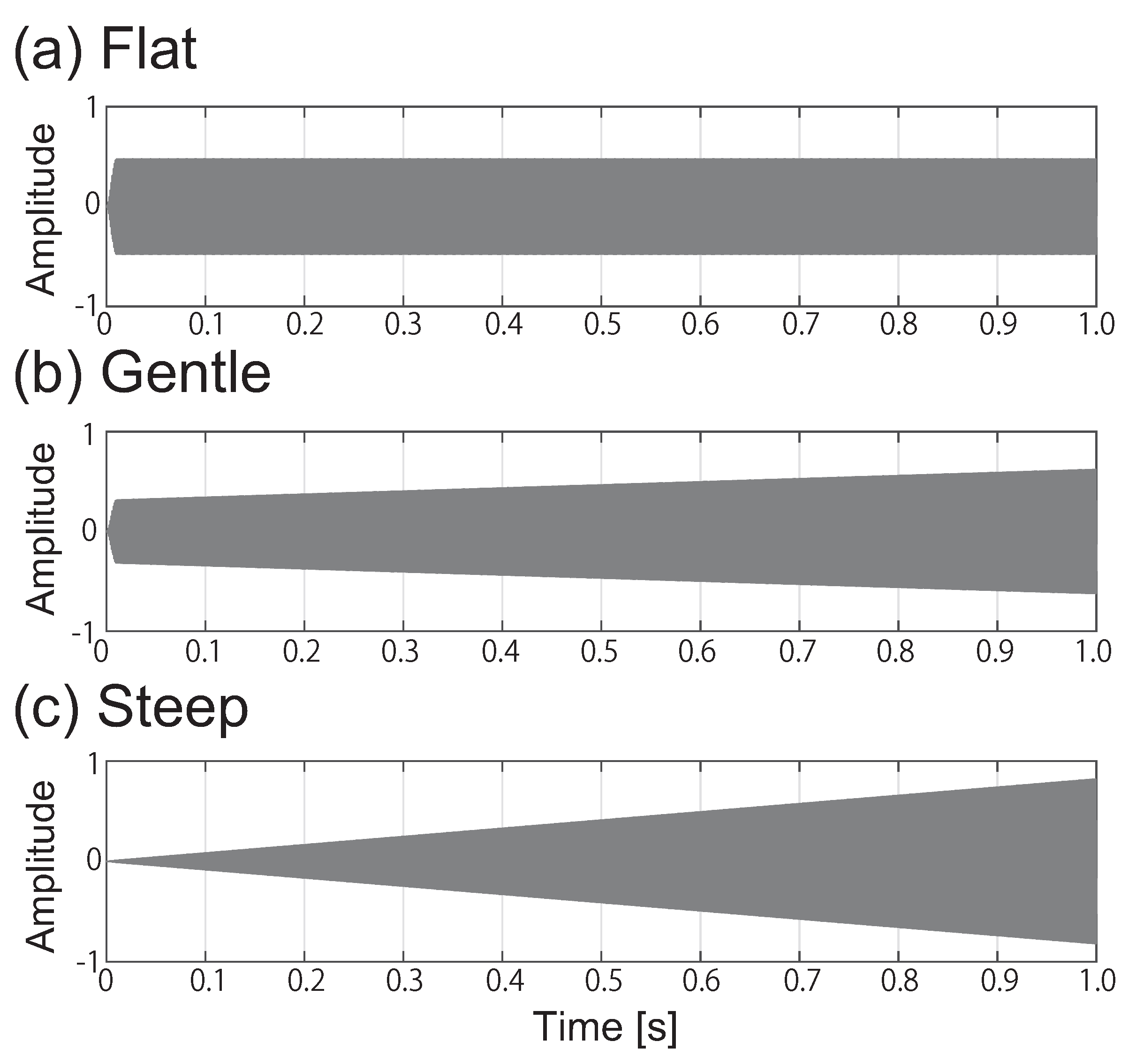
| Signal | Frequency: | 1 and 2 kHz |
|---|---|---|
| Envelope: | Flat, Gentle and Steep | |
| Silent condition | Signal level: | 10, 20 and 30 dBSL |
| Noise condition | Signal level: | 30 dBSL |
| Noise type: | White noise and Six bandpass noises | |
| SNR: | High and Low |
| 1 kHz Pure tone | |||||||
|---|---|---|---|---|---|---|---|
| Name | BD3 | BD2 | BD1 | BU1 | BU2 | BU3 | |
| High-SNR | ERB number | 9 | 11 | 13 | 17 | 19 | 21 |
| Frequency [Hz] | 439 | 602 | 805 | 1371 | 1762 | 2247 | |
| Low-SNR | ERB number | 10 | 12 | 14 | 16 | 18 | 20 |
| Frequency [Hz] | 516 | 698 | 924 | 1206 | 1556 | 1991 | |
| 2 kHz Pure tone | |||||||
| Name | BD3 | BD2 | BD1 | BU1 | BU2 | BU3 | |
| High-SNR | ERB number | 15 | 17 | 19 | 21 | 23 | 25 |
| Frequency [Hz] | 1057 | 1371 | 1762 | 2247 | 2852 | 3603 | |
| Low-SNR | ERB number | 14 | 16 | 18 | 22 | 24 | 26 |
| Frequency [Hz] | 924 | 1206 | 1556 | 2533 | 3207 | 4045 | |
| Start time [sec] | [dB] | a | b [sec] | Correlation coefficient | Averaged error [sec] | ||
|---|---|---|---|---|---|---|---|
| Silent condition | Young | 0.06 | 0.38 | 0.92 | 0.96 | 0.02 | |
| Elder | 0.06 | 0.57 | 1.11 | 0.99 | 0.01 | ||
| Noise condition | Young | 0.08 | 0.60 | 1.14 | 0.90 | 0.04 | |
| Elder | 0.02 | 0.25 | 0.85 | 0.93 | 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




