Submitted:
17 March 2026
Posted:
18 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Participants
2.3. Inclusion and Exclusion Criteria
2.4. Associated Procedures
2.5. Surgical Technique
2.6. MRI Protocol and Imaging Measurements
2.7. Outcomes
2.8. Follow-Up
2.9. Statistical Analysis
2.10. Ethical Considerations
3. Results
3.1. Study Population and Baseline Characteristics
3.2. MRI-Based Pelvic Floor Parameters
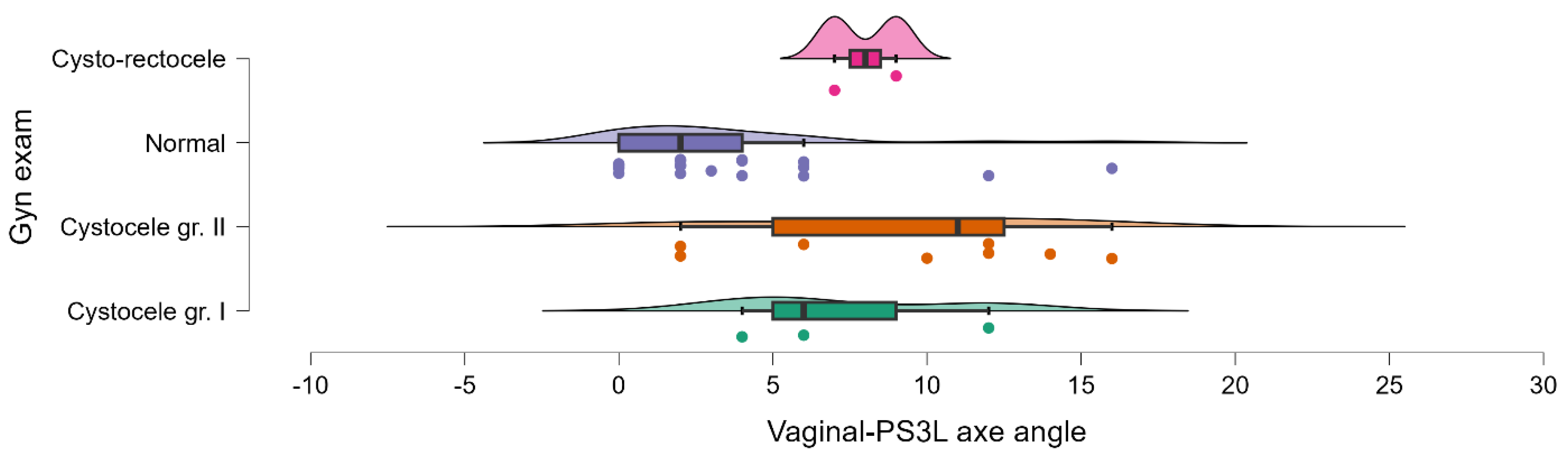
3.3. MRI-Based Vaginal Axis Evaluation
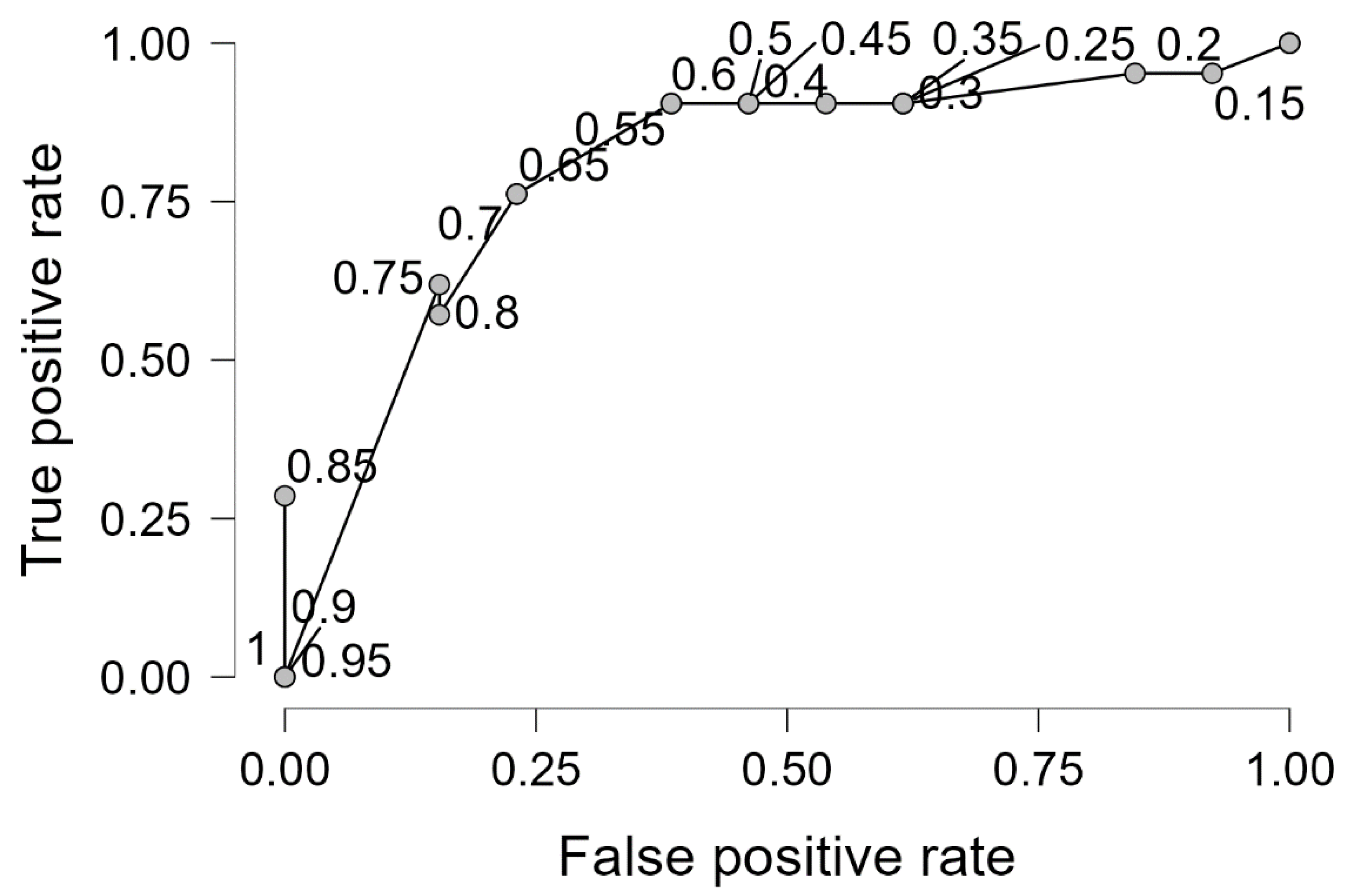
3.4. Association Between Vaginal Axis Orientation and Clinical Outcome
3.5. Postoperative Clinical Findings
4. Discussion
4.1. Principal Findings
4.2. Biomechanical Interpretation
4.3. Implications for Pectopexy Technique
4.4. Role of MRI in Outcome Assessment
4.5. Clinical Implications
4.6. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MRI | Magnetic Resonance Imaging |
| ROC | Receiver Operating Characteristic |
| AUC | Area Under The Curve |
| POP-Q | Pelvic Organ Prolapse Quantification |
References
- Maher, C.; Feiner, B.; Baessler, K.; Schmid, C. Surgical management of pelvic organ prolapse in women. Cochrane Database Syst Rev 2013. [Google Scholar] [CrossRef]
- Barber, M.D. Pelvic organ prolapse. BMJ 2016, 354, i3853. [Google Scholar] [CrossRef] [PubMed]
- Dietz, H.P. Ultrasound in the assessment of pelvic organ prolapse. Best Pr. Res. Clin. Obstet. Gynaecol. 2019, 54, 12–30. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.W.; Fernandes, R.A.; Yao, H.H.-I.; O’cOnnell, H.E.; Tse, V.; Gani, J. A systematic review of the surgical management of apical pelvic organ prolapse. Int. Urogynecology J. 2022, 34, 825–841. [Google Scholar] [CrossRef]
- DeLancey, J.O.; Mastrovito, S.; Masteling, M.; Horner, W.; Ashton-Miller, J.A.; Chen, L. A unified pelvic floor conceptual model for studying morphological changes with prolapse, age, and parity. Am. J. Obstet. Gynecol. 2023, 230, 476–484.e2. [Google Scholar] [CrossRef]
- Braga, A.; Serati, M. New Advances in Female Pelvic Floor Dysfunction Management. Medicina 2023, 59, 1010. [Google Scholar] [CrossRef]
- Hai, P.; Tian, Y.; Wang, L.; Yan, S.; Zhang, Y.; Wang, Q.; Zhao, Q.; Guo, R. Clinical application of laparoscopic pectopexy in the treatment of pelvic organ prolapse: Efficacy and safety. Eur. J. Med Res. 2025, 31. [Google Scholar] [CrossRef]
- Price, N.; Slack, A.; Jackson, S.R. Laparoscopic sacrocolpopexy: An observational study of functional and anatomical outcomes. Int. Urogynecology J. 2010, 22, 77–82. [Google Scholar] [CrossRef]
- Noé, G.K.; Alkatout, I.; Schiermeier, S.; Soltécz, S.; Anapolski, M. Laparoscopic anterior and posterior native tissue repair: A new pelvic floor approach. Minim. Invasive Ther. Allied Technol. 2018, 28, 241–246. [Google Scholar] [CrossRef]
- Noé, G.K.; Schiermeier, S.; Papathemelis, T.; Fuellers, U.; Khudyakov, A.; Altmann, H.-H.; Borowski, S.; Morawski, P.P.; Gantert, M.; De Vree, B.; et al. Prospective international multicenter pectopexy trial: Interim results and findings post surgery. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 244, 81–86. [Google Scholar] [CrossRef]
- Lei, Y.; Sun, Y.; Sun, T.; Han, X.; Zhao, Z.; Miao, Y. Mid-Term Efficacy Evaluation of Laparoscopic Sacrocolpopexy vs Laparoscopic Pectopexy for Pelvic Organ Prolapse. J. Sichuan Univ. (Med. Sci. Ed.) 2025, 56, 1104–1111. [Google Scholar]
- Kämpfer, C.; Pieper, C.C. Dynamische Magnetresonanztomographie des Beckenbodens: Klinische Anwendung. Die Radiol. 2023, 63, 799–807. [Google Scholar] [CrossRef] [PubMed]
- DeLancey, J.O.L. What’s new in the functional anatomy of pelvic organ prolapse? Curr. Opin. Obstet. Gynecol. 2016, 28, 420–429. [Google Scholar] [CrossRef]
- Shek, K.L.; Dietz, H.P. Coronal Plane Assessment for Levator Trauma. J. Ultrasound Med. 2024, 43, 1627–1633. [Google Scholar] [CrossRef] [PubMed]
- Sarpietro, G.; Foti, P.V.; Conte, C.; Matarazzo, M.G. Role of Magnetic Resonance Imaging in Pelvic Organ Prolapse Evaluation. Medicina 2023, 59, 2074. [Google Scholar] [CrossRef] [PubMed]
- Kieserman-Shmokler, C.; Swenson, C.W.; Chen, L.; Desmond, L.M.; Ashton-Miller, J.A.; DeLancey, J.O. From molecular to macro: The key role of the apical ligaments in uterovaginal support. Am. J. Obstet. Gynecol. 2020, 222, 427–436. [Google Scholar] [CrossRef]
- Ram, R.; Jambhekar, K.; Glanc, P.; Steiner, A.; Sheridan, A.D.; Arif-Tiwari, H.; Palmer, S.L.; Khatri, G. Meshy business: MRI and ultrasound evaluation of pelvic floor mesh and slings. Abdom. Imaging 2020, 46, 1414–1442. [Google Scholar] [CrossRef]
- Kale, A.; Biler, A.; Terzi, H.; Usta, T.; Kale, E. Laparoscopic pectopexy: Initial experience of single center with a new technique for apical prolapse surgery. Int. braz j urol 2017, 43, 903–909. [Google Scholar] [CrossRef]
- Pirtea, M.; Bălulescu, L.; Pirtea, L.; Brasoveanu, S.; Secosan, C.; Balan, L.; Olaru, F.; Dabica, A.; Margan, M.-M.; Navolan, D. The Effectiveness of Mesh-Less Pectopexy in the Treatment of Vaginal Apical Prolapse—A Prospective Study. Diagnostics 2025, 15, 526. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, L.; He, L.; Lin, W.; Yu, B.; Yu, X.; Lin, Y. Roles and mechanisms of biomechanical-biochemical coupling in pelvic organ prolapse. Front. Med. 2024, 11, 1303044. [Google Scholar] [CrossRef]
- Wang, H.; Shen, J.; Li, S.; Gao, Z.; Ke, K.; Gu, P. The feasibility of uterine-vaginal axis MRI-based as evaluation of surgical efficacy in women with pelvic organ prolapse. Ann. Transl. Med. 2022, 10, 447. [Google Scholar] [CrossRef] [PubMed]
- Egorov, V.; Takacs, P.; Shobeiri, S.A.; Hoyte, L.; Lucente, V.; van Raalte, H.; Sarvazyan, N. Predictive Value of Biomechanical Mapping for Pelvic Organ Prolapse Surgery. Urogynecology 2021, 27, e28–e38. [Google Scholar] [CrossRef] [PubMed]
- Babayi, M.; Azghani, M.; Hajebrahimi, S.; Berghmans, B. Three-dimensional finite element analysis of the pelvic organ prolapse: A parametric biomechanical modeling. Neurourol. Urodynamics 2018, 38, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.; Wang, G.; Zhao, Z. Advancements in Magnetic Resonance Imaging for the Evaluation of Pelvic Organ Prolapse: A Comprehensive Review. Acad. Radiol. 2025, 32, 4689–4704. [Google Scholar] [CrossRef]
- Maher, C.; Feiner, B.; Baessler, K.; Christmann-Schmid, C.; Haya, N.; Marjoribanks, J. Transvaginal mesh or grafts compared with native tissue repair for vaginal prolapse. Cochrane Database Syst. Rev. 2016, 2016, CD012079. [Google Scholar] [CrossRef]
- Dabica, A.; Balint, O.; Olaru, F.; Secosan, C.; Balulescu, L.; Brasoveanu, S.; Pirtea, M.; Popin, D.; Bacila, I.F.; Pirtea, L. Complications of Pelvic Prolapse Surgery Using Mesh: A Systematic Review. J. Pers. Med. 2024, 14, 622. [Google Scholar] [CrossRef]
- Alt, C.D.; Benner, L.; Mokry, T.; Lenz, F.; Hallscheidt, P.; Sohn, C.; Kauczor, H.-U.; A Brocker, K. Five-year outcome after pelvic floor reconstructive surgery: Evaluation using dynamic magnetic resonance imaging compared to clinical examination and quality-of-life questionnaire. Acta Radiol. 2018, 59, 1264–1273. [Google Scholar] [CrossRef]
- Luo, J.; Betschart, C.; Ashton-Miller, J.A.; DeLancey, J.O.L. Quantitative analyses of variability in normal vaginal shape and dimension on MR images. Int. Urogynecology J. 2016, 27, 1087–1095. [Google Scholar] [CrossRef]
- van Oudheusden, A.M.J.; Eissing, J.; Terink, I.M.; Vink, M.D.H.; van Kuijk, S.M.J.; Bongers, M.Y.; Coolen, A.-L.W.M. Laparoscopic sacrocolpopexy versus abdominal sacrocolpopexy for vaginal vault prolapse: Long-term follow-up of a randomized controlled trial. Int. Urogynecology J. 2022, 34, 93–104. [Google Scholar] [CrossRef]
- Padoa, A.; Braga, A.; Brecher, S.; Fligelman, T.; Mesiano, G.; Serati, M. Pelvic Organ Prolapse: Current Challenges and Future Perspectives. J. Clin. Med. 2025, 14, 7313. [Google Scholar] [CrossRef]
| Characteristic | Mesh Pectopexy (n = 50) | Wire Pectopexy (n = 50) |
|---|---|---|
| Age, years | 55.0 (52.0–59.0) | 55.0 (52.0–59.0) |
| BMI, kg/m2 | 28.0 (26.6–29.8) | 28.0 (25.9–29.4) |
| Urban residence, n (%) | 28 (56%) | 24 (48%) |
| Rural residence, n (%) | 22 (44%) | 26 (52%) |
| Apical POP-Q stage > II, n (%) | 50 (100%) | 50 (100%) |
| Total hysterectomy ± adnexectomy, n (%) | 9 (18%) | 47 (94%) |
| Rectocele repair, n (%) | 9 (18%) | 6 (12%) |
| McCall culdoplasty, n (%) | 1 (2%) | 2 (4%) |
| MRI Parameter | Mesh Pectopexy (n = 50) | Wire Pectopexy (n = 50) |
|---|---|---|
| H-line at rest (cm) | 5.9 (5.1–6.5) | 6.3 (5.0–7.2) |
| M-line at rest (cm) | 3.9 (3.5–5.6) | 3.9 (3.2–5.3) |
| H-line at Valsalva (cm) | 6.5 (5.6–7.9) | 6.7 (5.8–8.9) |
| M-line at Valsalva (cm) | 4.0 (3.9–6.8) | 5.6 (4.0–7.1) |
| Source | Sum of Squares | df | Mean Square | F | p Value |
|---|---|---|---|---|---|
| Gynecological examination | 220.978 | 3 | 73.659 | 3.867 | 0.019 |
| Residuals | 571.405 | 30 | 19.047 | — | — |
| Predictor | Estimate (β) | Standard Error | Wald z | p Value |
|---|---|---|---|---|
| Intercept | 1.951 | 0.687 | 2.841 | 0.004 |
| Vaginal PS3L axis angle | −0.257 | 0.096 | −2.664 | 0.008 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




