Submitted:
16 March 2026
Posted:
17 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Patients and Methods
2.1. Patients
2.2. Clinical and Biological Data
2.3. Nivolumab Administration
2.4. Ethical Approval and Health Authorities Approval
2.5. Statistical Analysis
3. Results
3.1. Description of Patients
3.1.1. Response Rates, Survival and Toxicity
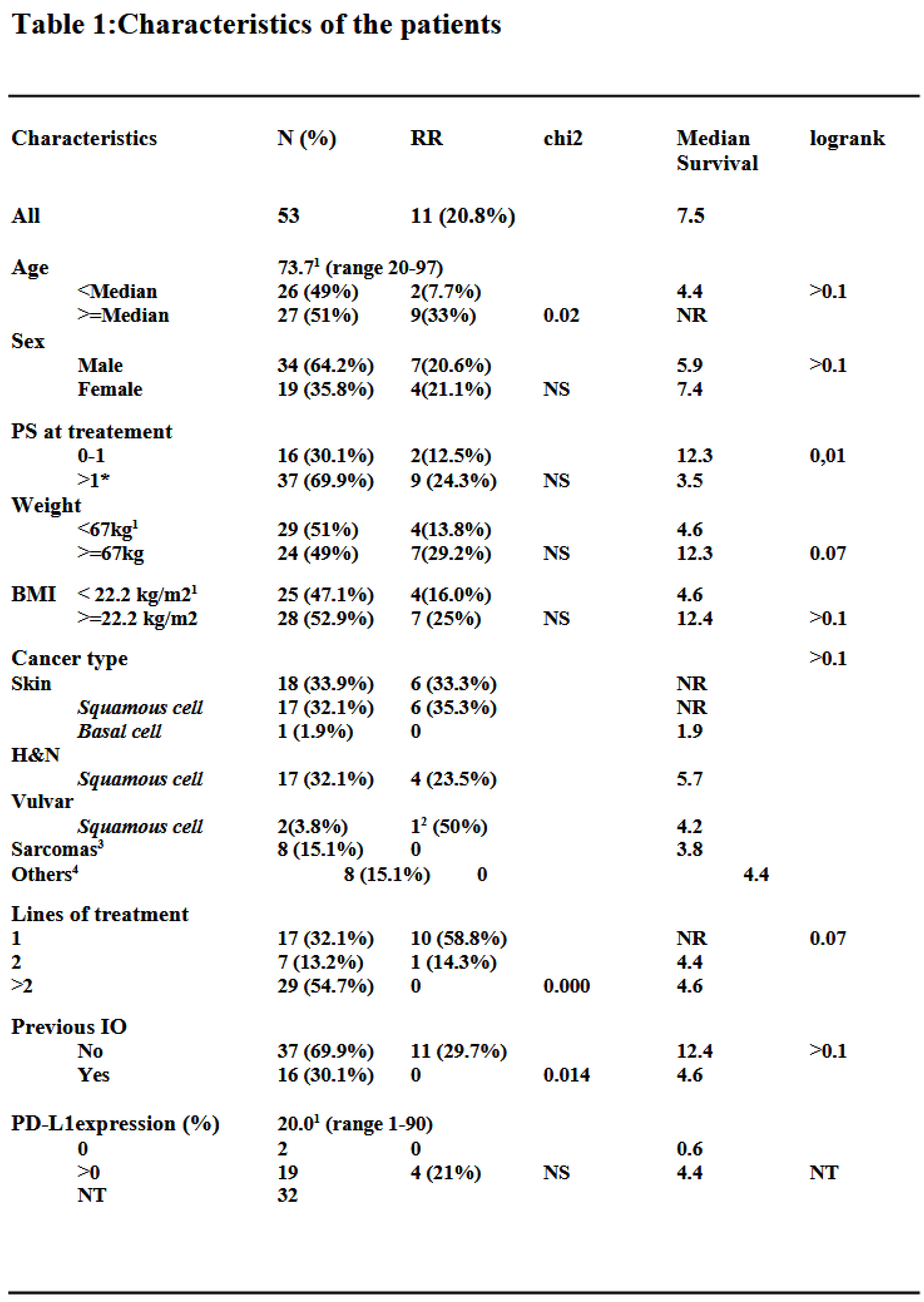
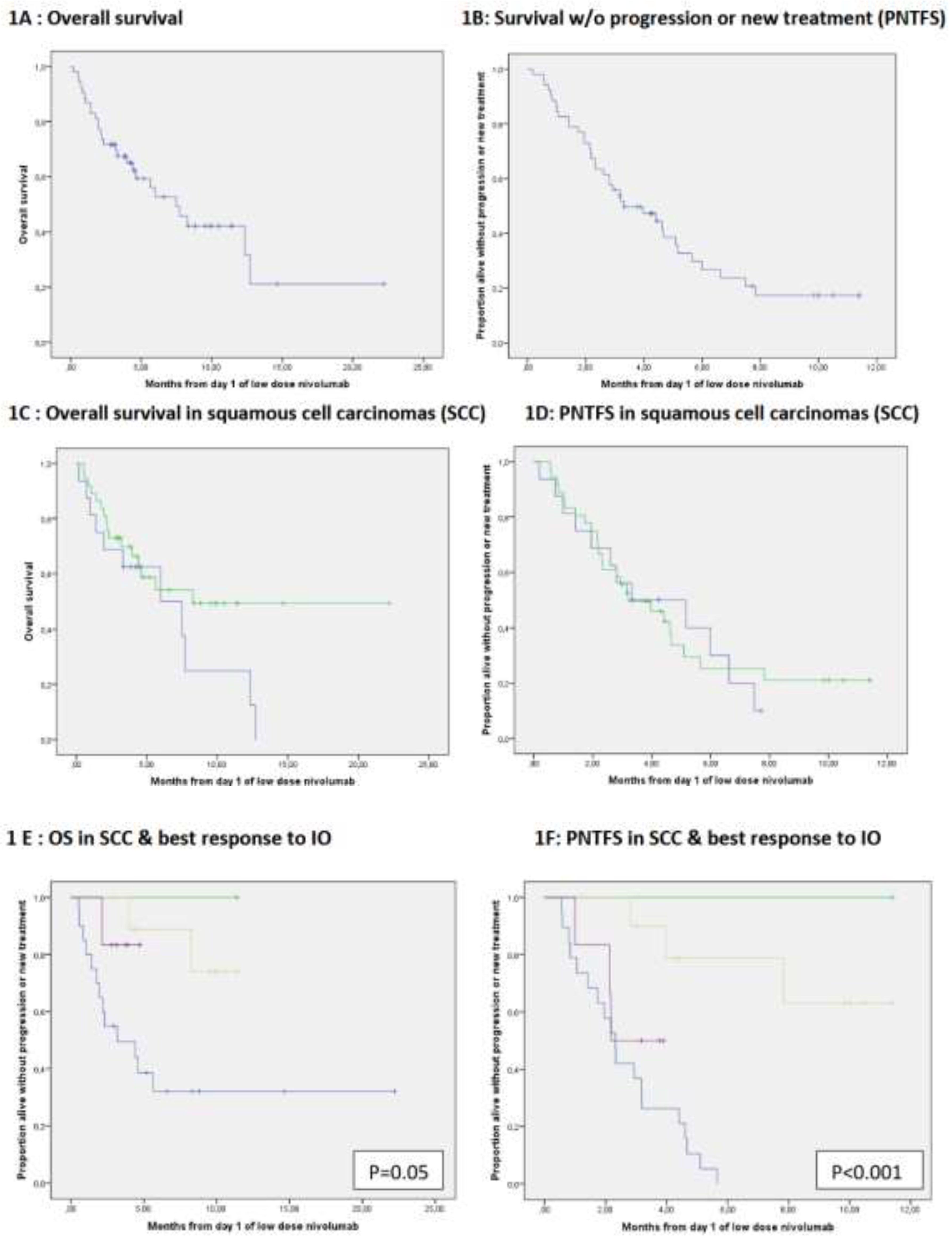
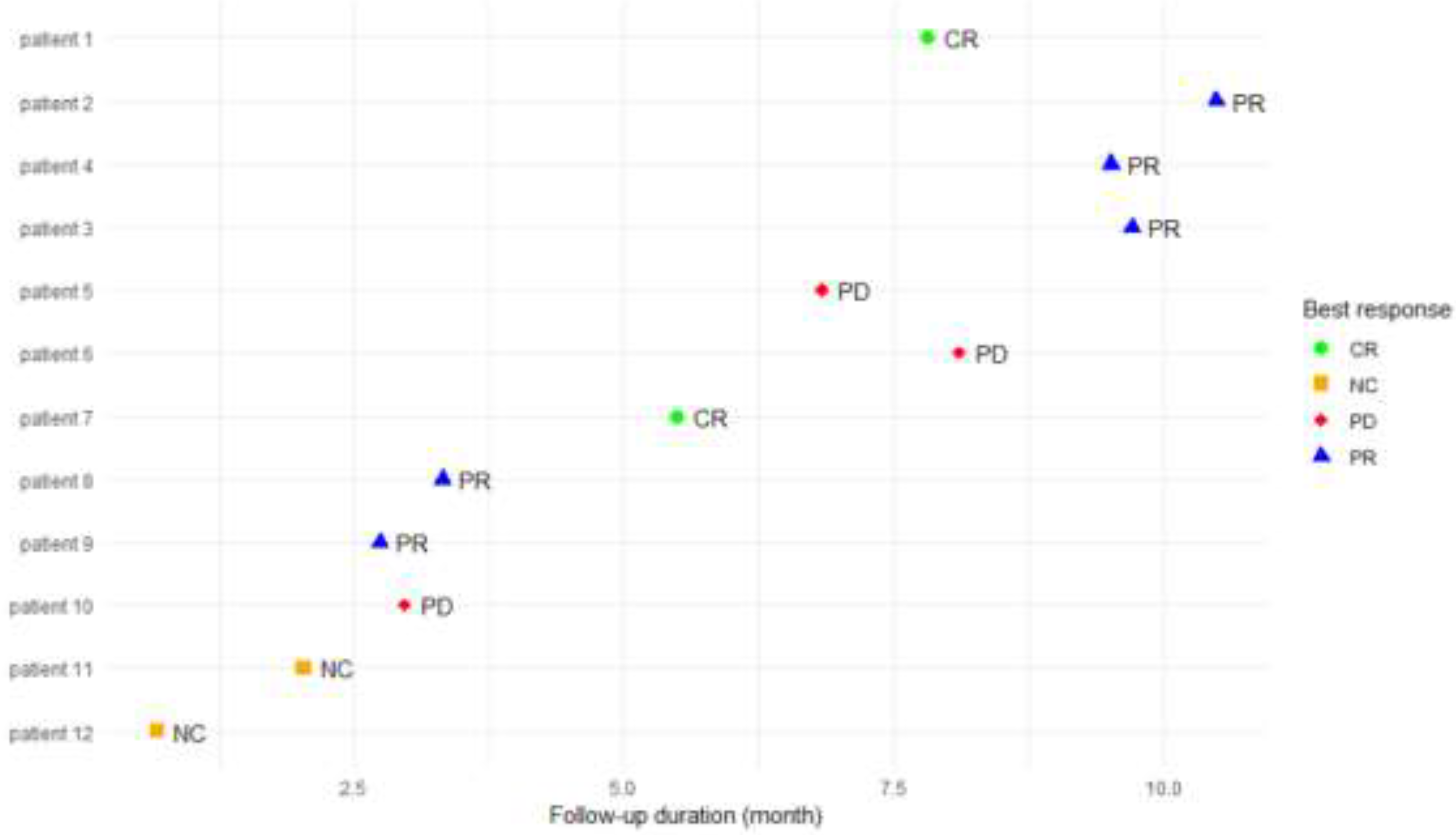
3.1.2. Subgroup Analysis of Cutaneous Squamous Cell Carcinoma
4. Discussion
4.1. Efficacy in a Frail Population
4.2. Safety and Tolerability
4.3. Clinical and Economic Implications
4.4. Limitations and Future Directions
5. Conclusion
Funding
References
- Pardoll, D.M. The blockade of immune checkpoints in cancerimmunotherapy. Nat. Rev. Cancer 12, 252–264 (2012).
- Chen, D.S.; Mellman, I. Oncology Meets Immunology: The Cancer-Immunity Cycle. Immunity 2013, 39, 1–10. [CrossRef]
- Weber, J.; Mandalà, M.; Del Vecchio, M.; Gogas, H.J.; Arance, A.M.; Cowey, C.L.; Dalle, S.; Schenker, M.; Chiarion-Sileni, V.; Marquez-Rodas, I.; et al. Adjuvant Nivolumab versus Ipilimumab in Resected Stage III or IV Melanoma. N. Engl. J. Med. 2017, 377, 1824–1835. [CrossRef]
- Brahmer J, eckamp KL, Baas P, Crinò L, Eberhardt WE, Poddubskaya E,et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N. Engl. J. Med. 2015; 373, 123–135.
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.R.; Steins, M.; Ready, N.E.; Chow, L.Q.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Nonsquamous Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [CrossRef]
- Motzer, R.J. Escudier B, McDermott DF, George S, Hammers HJ, Srinivas S,et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N. Engl. J. Med. 373, 1803–1813.
- Ansell SM. Nivolumab in the Treatment of Hodgkin Lymphoma. Clin Cancer Res. 2017; 23:1623-1626.).
- Patel, S.P.; Othus, M.; Chen, Y.; Wright, G.P.; Yost, K.J.; Hyngstrom, J.R.; Hu-Lieskovan, S.; Lao, C.D.; Fecher, L.A.; Truong, T.-G.; et al. Neoadjuvant–Adjuvant or Adjuvant-Only Pembrolizumab in Advanced Melanoma. New Engl. J. Med. 2023, 388, 813–823. [CrossRef]
- Blank CU, Lucas MW, Scolyer RA, van de Wiel BA, Menzies AM, Lopez-Yurda M,et al. Neoadjuvant Nivolumab and Ipilimumab in Resectable Stage III Melanoma. N Engl J Med. 2024; 391:1696-1708.
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N. Engl. J. Med. 2015, 372, 2509–2520. [CrossRef]
- Brahmer JR, Drake CG, Wollner I, et al.: Phase I study of single-agent anti–programmed death-1 (MDX-1106) in refractory solid tumors: Safety, clinical activity, pharmacodynamics, and immunologic correlates. J Clin Oncol 28:3167-3175, 2010.
- Agrawal, S.; Feng, Y.; Roy, A.; Kollia, G.; Lestini, B. Nivolumab dose selection: challenges, opportunities, and lessons learned for cancer immunotherapy. J. Immunother. Cancer 2016, 4, 72. [CrossRef]
- Sheng J Srivastava S, Sanghavi K, et al.: Clinical pharmacology considerations for the development of immune checkpoint inhibitors. J Clin Pharmacol 57:S26-S42, 2017 (suppl 10).
- Feng Y, Wang X, Bajaj G, et al.: Nivolumab exposure–response analyses of efficacy and safety in previously treated squamous or nonsquamous non–small cell lung cancer. Clin Cancer Res 23: 5394-5405, 2017.
- Prabhash, K.; Patil, V.M.; Noronha, V.; Joshi, A.; Abhyankar, A.; Menon, N.; Banavali, S.; Gupta, S. Low doses in immunotherapy: Are they effective? Cancer Res. Stat. Treat. 2019, 2 : 54.
- Yoo, S.H.; Keam, B.; Kim, M.; Kim, S.H.; Kim, Y.J.; Kim, T.M.; Kim, D.-W.; Lee, J.S.; Heo, D.S. Low-dose nivolumab can be effective in non-small cell lung cancer: alternative option for financial toxicity. ESMO Open 2018, 3, e000332. [CrossRef]
- Patil, V.M.; Noronha, V.; Menon, N.; Rai, R.; Bhattacharjee, A.; Singh, A.; Nawale, K.; Jogdhankar, S.; Tambe, R.; Dhumal, S.; et al. Low-Dose Immunotherapy in Head and Neck Cancer: A Randomized Study. J. Clin. Oncol. 2023, 41, 222–232. [CrossRef]
- Vega Mateos A, Vaquera Alfaro HA, Tejada Vasquez AC, Lopez-Garcia YK, Garcia-Salas G, Cantu O, et al. Low-Dose Nivolumab Plus AVD As Front-Line Therapy for Classical Hodgkin’s Lymphoma: Preliminary Results of a Phase 2 Trial. Blood. 5 nov 2024;144(Supplement 1):1665.
- Melemadathil, K.; Pai, A.; Udupa, K.; Mailankody, S. 472P Exploring the efficacy of low-dose nivolumab in metastatic settings. Ann. Oncol. 2024, 35, S1577. [CrossRef]
- Gandhi, K.A.; Shirsat, A.; Hj, S.K.; Chavan, A.; Dicholkar, P.; Shah, S.; Menon, N.; Noronha, V.; Joshi, A.; Prabhash, K.; et al. Pharmacokinetics and clinical outcomes of low-dose nivolumab relative to conventional dose in patients with advanced cancer. Cancer Chemother. Pharmacol. 2024, 94, 659–668. [CrossRef]
- Biswal, N. 613TiP Evaluation of low dose Nivolumab with neoadjuvant chemotherapy in non-small cell lung cancer: A phase II open label randomised clinical trial (ELON). Ann. Oncol. 2024, 35, S1622. [CrossRef]
- Prabhash, K.; Abraham, G.; Menon, N.; Patil, V.M.; Joshi, A.P. The efficacy of low-dose immunotherapy in head-and-neck cancer. Cancer Res. Stat. Treat. 2019, 2, 268. [CrossRef]
- Zhao, J.J.; Kumarakulasinghe, N.B.; Muthu, V.; Lee, M.; Walsh, R.; Low, J.L.; Choo, J.; Tan, H.L.; Chong, W.Q.; Ang, Y.; et al. Low-Dose Nivolumab in Renal Cell Carcinoma: A Real-World Experience. Oncology 2021, 99, 192–202. [CrossRef]
- Kumar, A.; Noronha, V.; Patil, V.; Joshi, A.; Menon, N.; Kapoor, A.; Janu, A.; Mahajan, A.; Rajendra, A.; Prabhash, K. 1049P Efficacy and safety of low dose immunotherapy in palliative setting of advanced solid tumours. Ann. Oncol. 2020, 31, S718–S718. [CrossRef]
- Zhao, J.J.; Kumarakulasinghe, N.B.; Muthu, V.; Lee, M.; Walsh, R.; Low, J.L.; Choo, J.; Tan, H.L.; Chong, W.Q.; Ang, Y.; et al. Low-Dose Nivolumab in Renal Cell Carcinoma: A Real-World Experience. Oncology 2021, 99, 192–202. [CrossRef]
- Chen, Y.-H.; Wang, C.-C.; Chen, Y.-Y.; Wang, J.-H.; Hung, C.-H.; Kuo, Y.-H. Low-dose nivolumab in advanced hepatocellular carcinoma. BMC Cancer 2022, 22, 1–12. [CrossRef]
- Meriggi, F.; Zaniboni, A.; Zaltieri, A. Low-Dose Immunotherapy: Is It Just an Illusion?. Biomedicines 2023, 11, 1032. [CrossRef]
- Trikha, M.; Sarkar, L.; Dhanawat, A.; Syed, N.; Gujarathi, H.; Vora, M.; Raja, A.S.S.; Bhargava, P.; Ramaswamy, A.; Mandavkar, S.; et al. Performance of Low-Dose Immunotherapy and Standard-Dose Immunotherapy in Microsatellite Instability-High Metastatic Colorectal Cancer: Real-World Data (CLouD—High Study). JCO Glob. Oncol. 2024, 10, e2400141. [CrossRef]
- Guérin, J.; Nahid, A.; Tassy, L.; Deloger, M.; Bocquet, F.; Thézenas, S.; Desandes, E.; Le Deley, M.-C.; Durando, X.; Jaffré, A.; et al. Consore: A Powerful Federated Data Mining Tool Driving a French Research Network to Accelerate Cancer Research. Int. J. Environ. Res. Public Heal. 2024, 21, 189. [CrossRef]
- Heudel, P.; Crochet, H.; Durand, T.; Zrounba, P.; Blay, J.-Y. From data strategy to implementation to advance cancer research and cancer care: A French comprehensive cancer center experience. PLOS Digit. Heal. 2023, 2, e0000415. [CrossRef]
- Heudel, P.-E.; Fervers, B.; Durand, T.; Chabaud, S.; Michallet, A.-S.; Gomez, F.; Rivoire, M.; Bachelot, T.; Claude, L.; Chassagne-Clement, C.; et al. Second primary cancers: a retrospective analysis of real world data using the enhanced medical research engine ConSoRe in a French comprehensive cancer center. Int. J. Clin. Oncol. 2021, 26, 1793–1804. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



