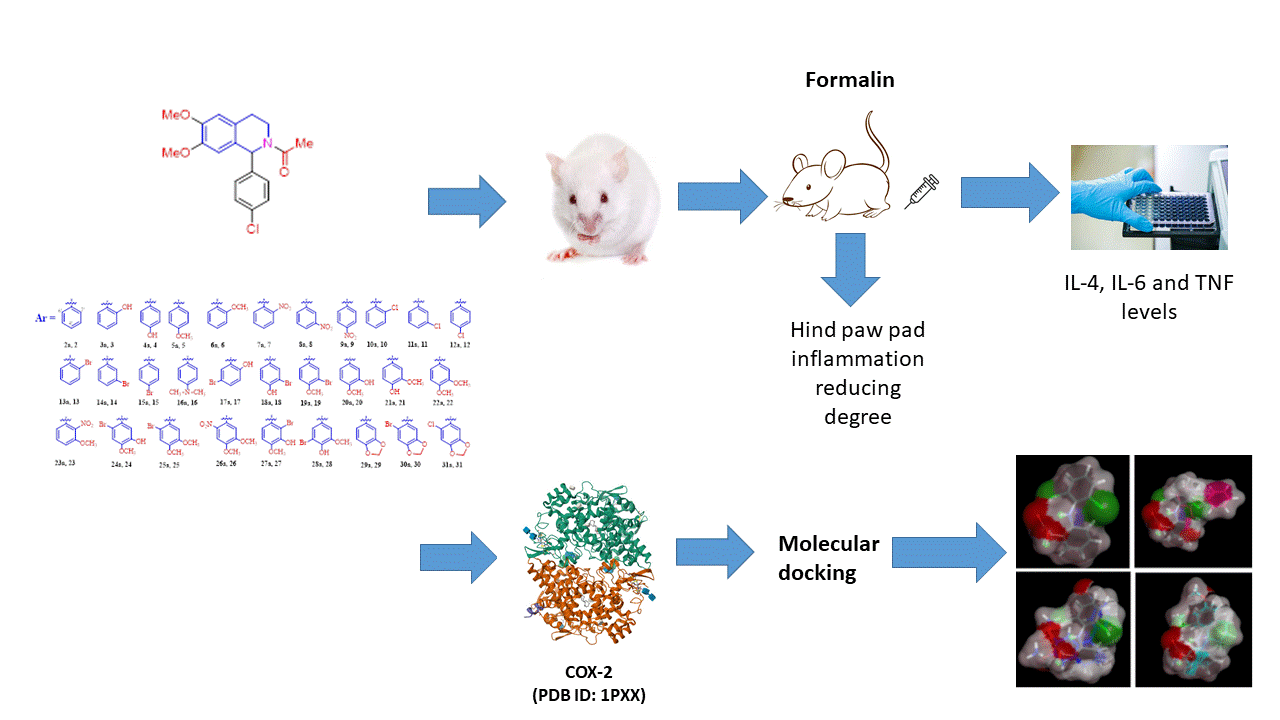
Non-steroidal anti-inflammatory drugs (NSAIDs) are widely used around the world for their pain-relieving and fever-reducing properties. However, excessive intake of NSAIDs can lead to harmful effects on multiple body systems, including the cardiovascular, gastrointestinal, hepatic, renal, and nervous systems. The anti-inflammatory activity of 34 derivatives of 1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline was investigated in vivo. A relationship between the activity of the compounds and the nature of the substituents, as well as their positional and mutual arrangement in the C ring (1-Ar-), was established. In silico modelling of 1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline derivatives interaction with COX-2 (PDB ID: 1PXX) active site indicated that the nitro-derivatives exhibited the highest stability owing to their superior capacity for electrostatic and hydrogen bond formation compare to brominated compounds. The presented data on the effect of substituents –NH₂, –OH, and –OCH₃ in ring C (1-Ar-) of 1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinolines on anti-inflammatory activity further stimulate the search for new highly effective derivatives within this series.