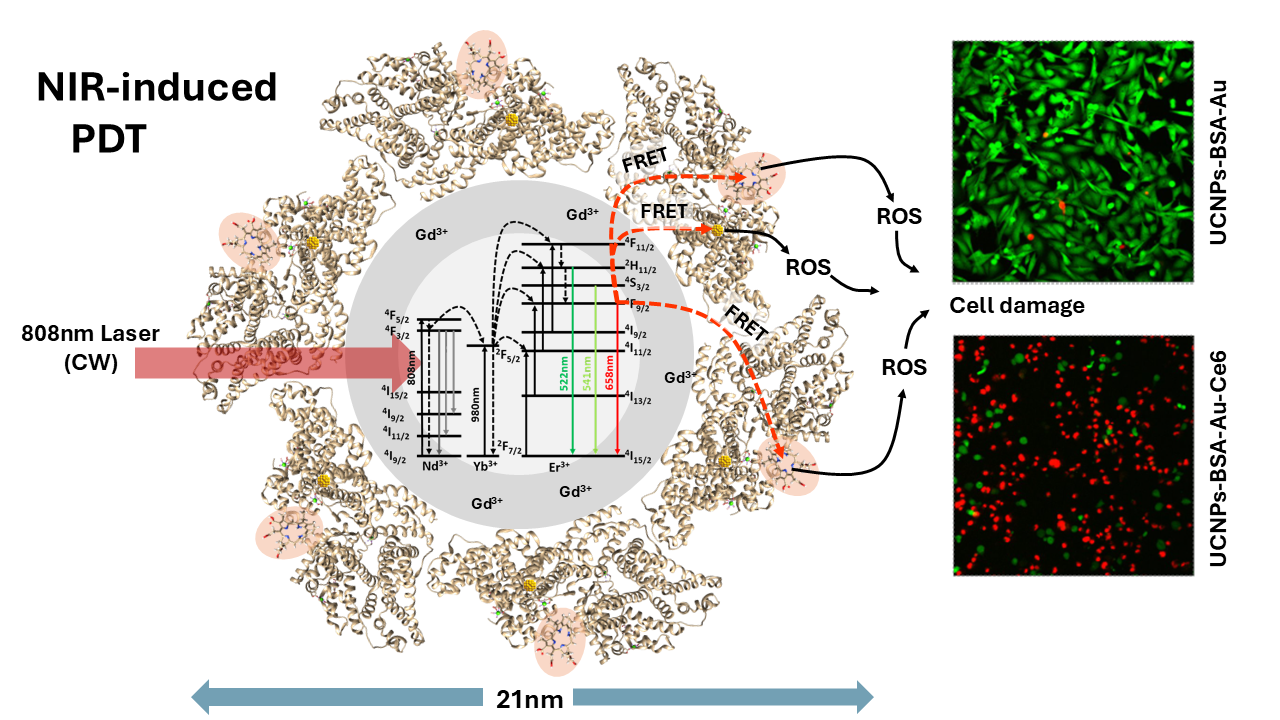
Current efforts in improving photodynamic therapy focus on nanomaterials that integrate deep-tissue imaging with efficient reactive oxygen species generation. Gold nanoclusters (Au NCs) are promising alternatives to conventional photosensitizers due to their effective ROS production and enhanced biocompatibility when stabilized by protein corona. However, both photosensitizers and Au NCs are typically activated by ultraviolet or visible light, which cannot penetrate deeper into tissues and is limited to superficial applications. Here, we report a near-infrared (NIR)-activated photodynamic nanoplatform based on core-shell upconverting nanoparticles (UCNPs; NaGdF₄:Yb³⁺,Er³⁺@NaGdF₄:Yb³⁺,Nd³⁺), functionalized with a protein corona containing bovine serum albumin-stabilized Au NCs (BSA-Au NCs) and photosensitizer chlorin e6 (Ce6). Spectroscopic data confirmed the formation of the UCNP-BSA-Au-Ce6 nanoplatform and demonstrated 32% energy transfer efficiency from UCNPs to Ce6, resulting in efficient reactive oxygen species generation under 808 nm irradiation. Cellular experiments confirmed effective internalization and optimal biocompatibility of the nanoplatform in human breast cancer and healthy cells. Upon 808 nm irradiation, the nanoplatform significantly reduced viability of MDA-MB-231 cancer cells. These findings indicate that the UCNP-BSA-Au-Ce6 nanoplatform couples NIR activation with enhanced singlet oxygen production, providing a multifunctional platform for deep-tissue imaging and NIR-activated photodynamic therapy.