5. Discussion
Accessory pathway ablation is one of the most frequent procedures in pediatric electrophysiology, especially in patients with Wolff–Parkinson–White syndrome. Most of these procedures are performed with sedation or general anesthesia in children, due to limited patient cooperation and the need for adequate immobility during the electrophysiological study (9). However, the possibility of drug-induced arrhythmia suppression remains a relevant concern, as it may compromise both the performance of the EPS and the success of the ablation.
5.1. Primary Findings: Arrhythmia Inducibility
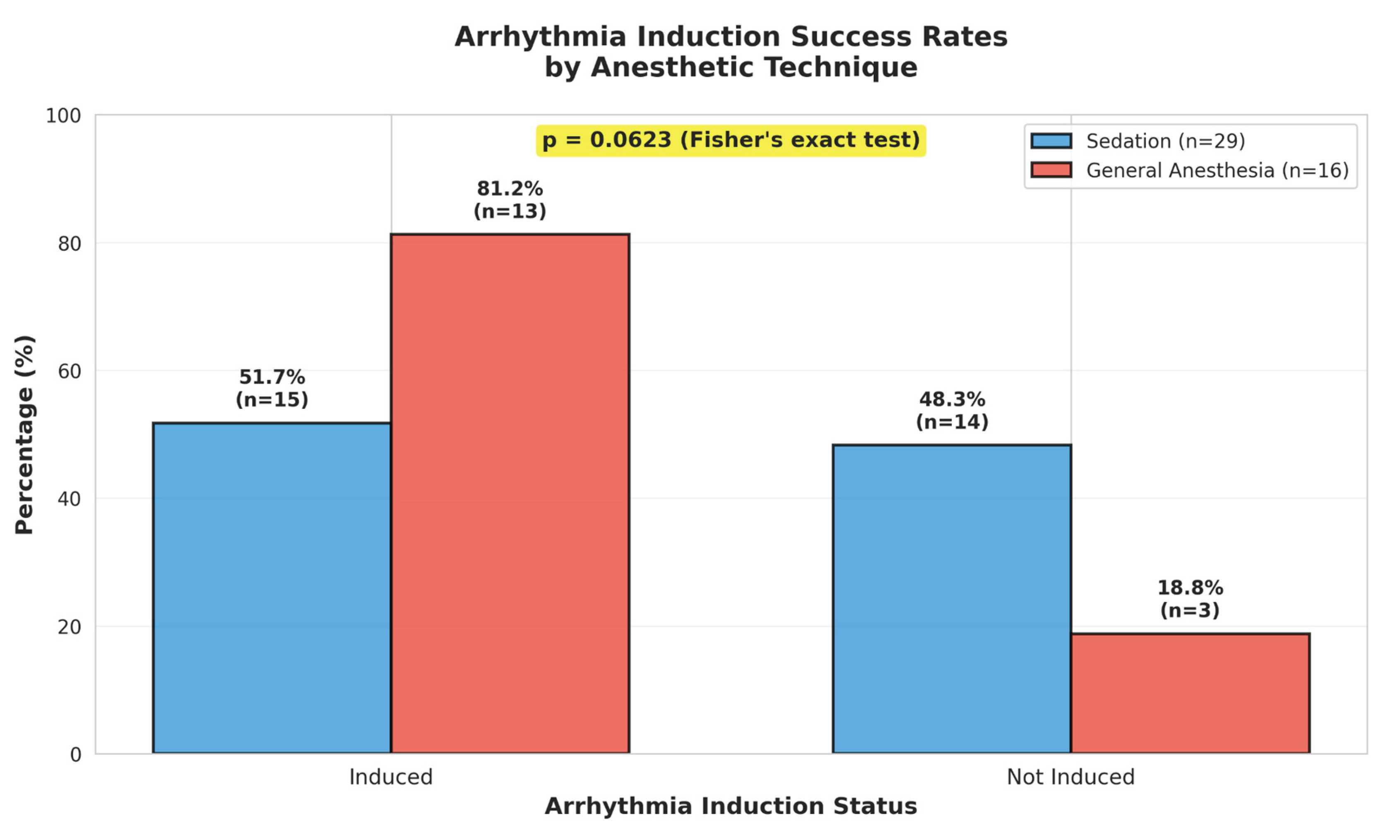
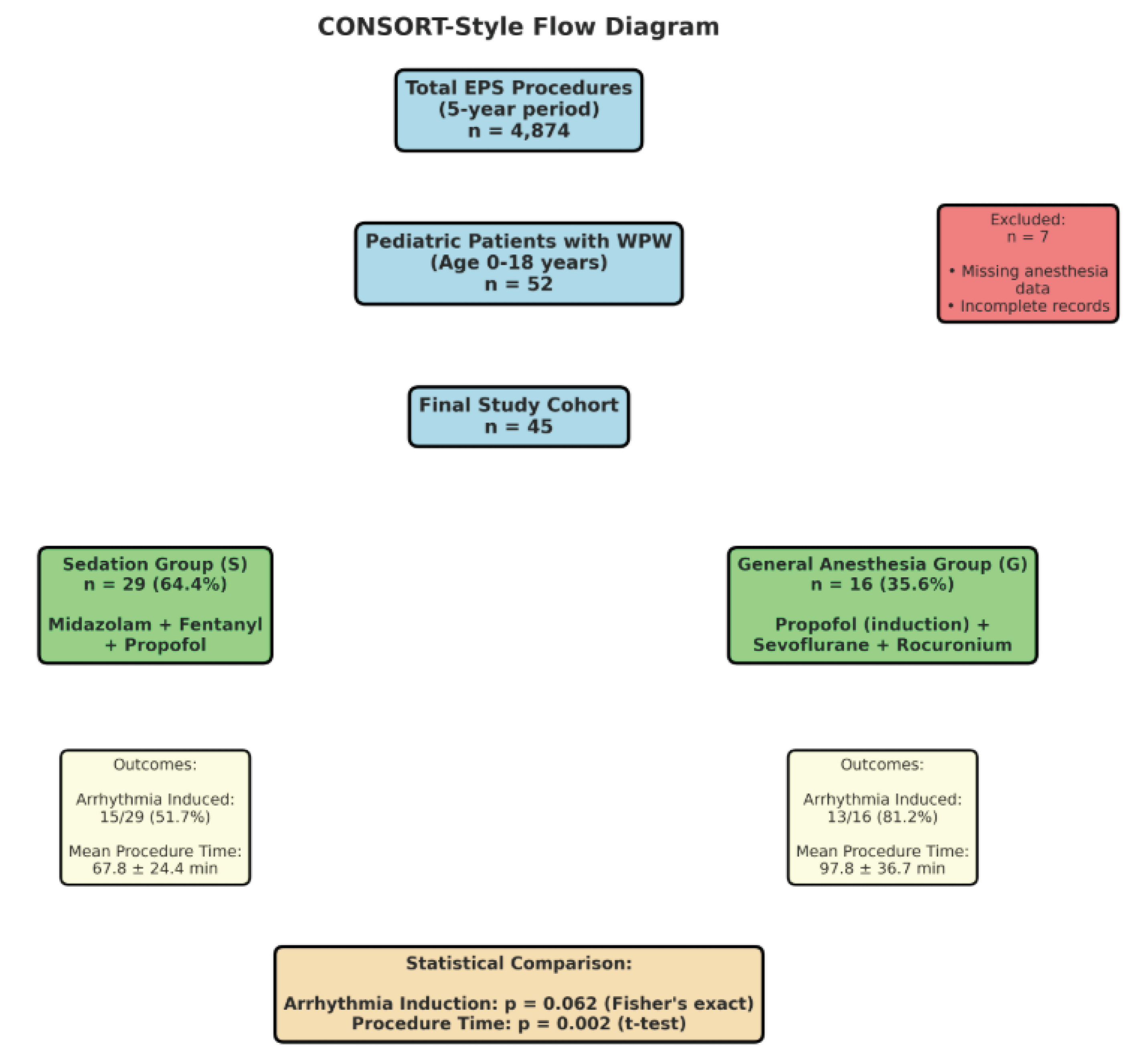
The central finding of our study is the observed trend toward lower arrhythmia inducibility in the sedation group (51.7%) compared to the general anesthesia group (81.2%), representing a 29.5% difference. Although this difference did not achieve statistical significance (p = 0.062), it approaches the conventional threshold and warrants careful consideration.
Critically, our post-hoc power analysis revealed that the study had only 49.8% statistical power to detect this observed difference. This low power substantially increases the risk of a Type II error—that is, failing to detect a true difference when one exists. Therefore, the lack of statistical significance should not be interpreted as definitive evidence of no effect. Rather, it suggests that a larger sample size would be necessary to conclusively determine whether propofol-based sedation genuinely reduces arrhythmia inducibility in this population.
From a clinical perspective, a 30% reduction in inducibility could have meaningful implications for procedural success. Inability to induce arrhythmia during EPS significantly complicates mapping and ablation, potentially leading to incomplete procedures, need for repeat interventions, or alternative management strategies. While our data cannot definitively establish causation, they raise important considerations for anesthesia selection in pediatric WPW ablation procedures.
5.2. Comparison with Existing Literature
Our findings align with several previous reports suggesting potential interference of propofol with arrhythmia inducibility, while also acknowledging contradictory evidence in the literature. Some authors suggest that propofol may influence parameters of the cardiac conduction system. Matsushima et al. studied the effect of propofol in pediatric patients during RF ablation and found that this drug significantly prolonged the HV interval but did not interfere with the Atrium–His interval or other intervals measured in the study; however, they observed that propofol directly suppresses parasympathetic activity as well as sympathetic activity, concluding that this result may help elucidate the mechanism by which lethal AV blocks are induced by propofol (10).
Similarly, Paech C et al., in their retrospective cohort study of patients with WPW, observed the occurrence of loss of pre-excitation in 4 patients after initiating sedation with propofol, demonstrating a possible suppression of the accessory pathway. These authors reported that they were not able to induce arrhythmia in all patients when propofol was used in the anesthetic regimen, concluding that propofol may have interfered negatively with the inducibility of the arrhythmias (1).
On the other hand, other studies attempted to demonstrate some effect of propofol on the CCS without success, such as that of Warpechowski et al., who evaluated the effects of propofol on the AV conduction system of patients with nodal reentrant tachycardia by analyzing the effective refractory periods of the slow pathway and the fast pathway of the AVN during EPS.The authors concluded that propofol did not cause significant alterations in the electrophysiological variables of the atrioventricular node (11). These findings were similar to those reported by Sharpe et al., who studied patients with Wolff–Parkinson–White syndrome; in both studies, the arrhythmia mechanism was reentry (12).Based on these studies and our findings, it appears that propofol’s effects may be substrate-dependent or dose-dependent, potentially explaining the conflicting results in the literature.
Perez et al. evaluated the electrophysiological effects in children with WPW undergoing RF ablation and found that the administration of sevoflurane produced a widening of the effective refractory period of the accessory pathway (APERP) (propofol-ERP 283 ± 22 ms; sevoflurane-ERP 298 ± 25 ms; p = 0.004), but it did not prevent its ablation, and no other significant change was found during the procedure (4).
Furthermore, the results we found are also aligned with the study by Hino et al. (2018), which demonstrated that propofol preserves the inducibility of supraventricular tachycardia in children with WPW, whereas desflurane, at concentrations of 0.5 MAC and 1.0 MAC, reduced this capability. In the study, the arrhythmia was induced in all children under propofol, but could not be induced in 1 child with 0.5 MAC and in 4 children with 1.0 MAC of desflurane. However, it is important to note that in the Hino study, propofol was used in isolation for sedation, whereas in our general anesthesia group, propofol was only used for induction followed by sevoflurane maintenance. This methodological difference may partly explain the divergent findings and highlights the challenge of attributing effects to specific agents in multi-drug anesthesia protocols.(13).
More recent data further support the potential neutrality of propofol with respect to arrhythmia inducibility. In a study by Zhang et al. (2025), which compared propofol with next-generation sedatives such as ciprofol and remimazolam in children undergoing electrophysiological study for supraventricular tachyarrhythmias, no significant differences were observed in arrhythmia induction rates among the three agents. These findings reinforce that, when compared with modern sedative agents, propofol maintains similar performance in preserving inducibility, though our study suggests this may not be the case when compared to volatile anesthetic-based general anesthesia.(14).
5.3. Procedural Time Differences
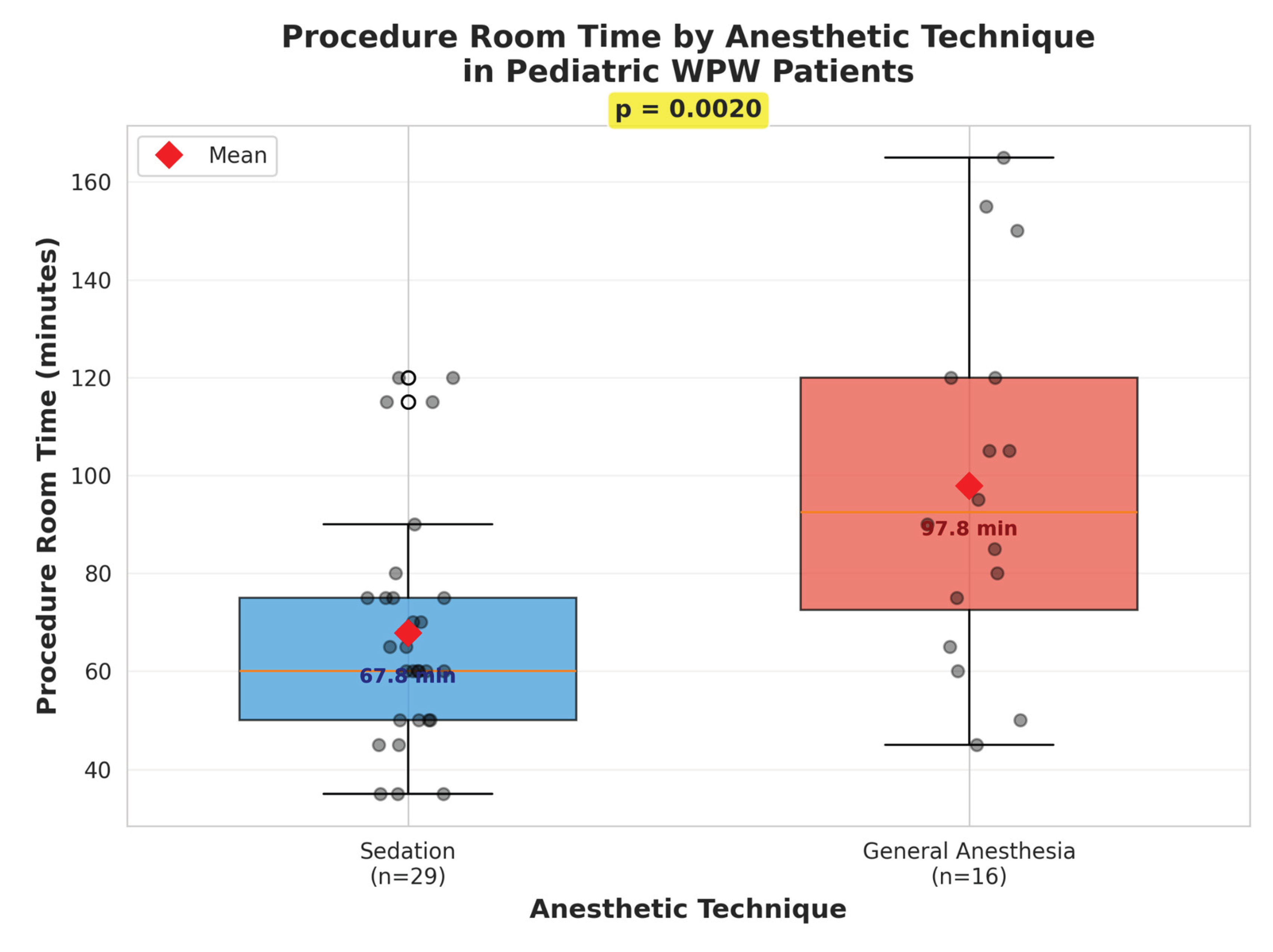
Regarding procedure duration, we observed significantly longer procedure times in the general anesthesia group (97.8 ± 36.7 minutes) compared to the sedation group (67.8 ± 24.4 minutes, p = 0.002). This 30-minute difference likely reflects multiple factors: (1) the time required for endotracheal intubation and airway management in the general anesthesia group, (2) the higher arrhythmia induction rate in the general anesthesia group potentially allowing more complete mapping and ablation procedures, and (3) possible differences in procedural complexity between the younger patients requiring general anesthesia and older patients managed with sedation.
Interestingly, the longer procedure time in the general anesthesia group occurred despite—or perhaps because of—the higher induction success rate. This paradoxical finding suggests that successful arrhythmia induction may lead to more comprehensive electrophysiological assessment and ablation, ultimately requiring more time but potentially resulting in more complete treatment.
5.4. Role of Isoproterenol
In this context, it is also important to note that, in situations of difficulty in inducing the arrhythmia during the electrophysiological study, especially in pediatric patients with WPW under general anesthesia, pharmacological strategies may assist in restoring conduction through the accessory pathway. In this context, the findings of Moore et al. (2011) are notably relevant: in a pediatric cohort, the authors demonstrated that the administration of isoproterenol significantly increased anterograde and retrograde conduction through the accessory pathways, facilitating the induction of reentrant tachyarrhythmias under general anesthesia. Thus, the use of isoproterenol emerges as a possible alternative to overcome induction difficulties potentially related to propofol sedation, allowing mapping and ablation to be completed safely and accurately. Future studies should systematically document the use of isoproterenol and other provocative agents to better understand their role in overcoming anesthesia-related suppression of arrhythmia inducibility.(15).
5.5. Methodological Considerations and Confounding Factors
A critical limitation of our study design is that propofol was not used in isolation in either group. In the sedation group, propofol was administered together with midazolam and fentanyl, while in the general anesthesia group it was combined with fentanyl, neuromuscular blockade, and sevoflurane. Consequently, any observed effects cannot be attributed solely to propofol. This polypharmacy approach, while reflecting real-world clinical practice, limits our ability to definitively isolate propofol’s specific contribution to the observed differences in arrhythmia inducibility.
Furthermore, the depth of sedation or anesthesia was not quantified using validated scales or monitoring tools such as Bispectral Index (BIS), Ramsay Sedation Scale, or Richmond Agitation-Sedation Scale (RASS). Given that autonomic tone and depth of anesthesia may influence accessory pathway conduction and arrhythmia inducibility, this represents an important methodological limitation that should be addressed in future prospective studies.
Additionally, we did not systematically document whether patients were receiving antiarrhythmic medications prior to the procedure, nor whether such medications were discontinued and for how long before the EPS. Antiarrhythmic drugs can significantly affect arrhythmia inducibility and should be carefully controlled in future studies.
An important mechanistic consideration is the role of autonomic tone in WPW arrhythmia inducibility. Young patients with WPW often exhibit heightened sympathetic activity and catecholamine prevalence, which can facilitate accessory pathway conduction and arrhythmia induction. Both sedation and general anesthesia suppress autonomic nervous system activity, though potentially through different mechanisms and to different degrees. Propofol has been shown to suppress both sympathetic and parasympathetic activity (10), while volatile anesthetics like sevoflurane have distinct autonomic effects. Therefore, any observed differences in arrhythmia inducibility between our groups may reflect differential autonomic suppression rather than direct electrophysiological effects on the cardiac conduction system. This autonomic confounding makes it impossible to isolate propofol’s direct cardiac effects in our study design.
5.6. Study Limitations
Several important limitations of our study must be acknowledged:
1. Small Sample Size and Statistical Power: The most significant limitation is the small number of patients (n=45), resulting in inadequate statistical power (49.8%) to detect the observed 29.5% difference in induction rates. This substantially increases the risk of Type II error and limits the generalizability of our findings.
2. Retrospective Design: As a retrospective observational study, our analysis is subject to selection bias and unmeasured confounding. The choice of anesthesia technique was not randomized but rather determined by clinical factors, particularly patient age and cooperation ability.
3. Lack of Detailed Electrophysiological Parameters: We did not evaluate specific intervals of the cardiac conduction system, such as the AH interval, HV interval, accessory pathway effective refractory periods, or other electrophysiological parameters that might provide mechanistic insights into propofol’s effects.
4. Absence of Drug Level Monitoring: Serum concentrations of propofol were not measured in the patients, limiting our ability to establish dose-response relationships.
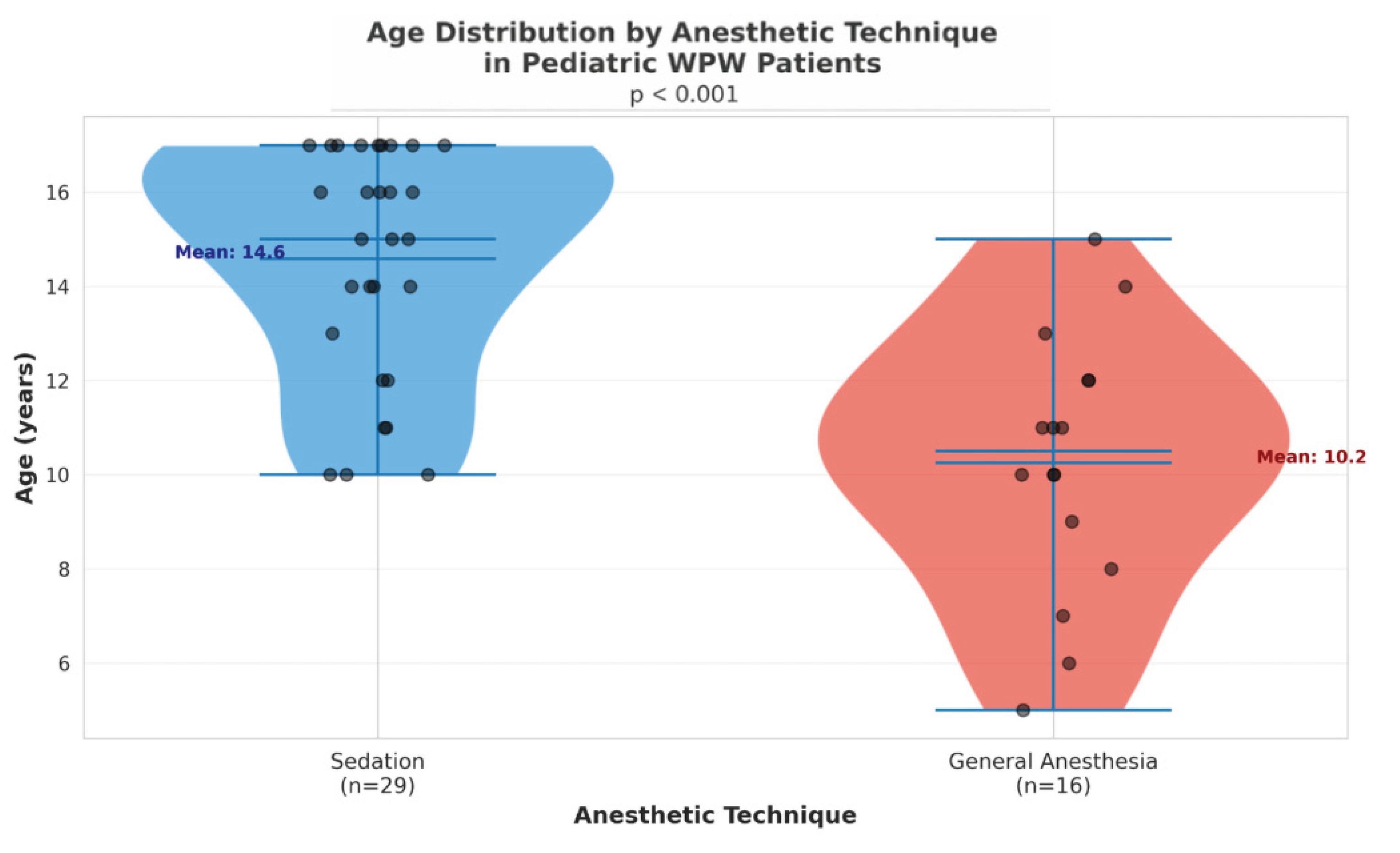
5. Age Disparity Between Groups: The significant age difference between groups (14.6 vs 10.3 years) introduces potential confounding. Younger patients may have different electrophysiological properties, accessory pathway characteristics, or arrhythmia substrates compared to older children, independent of anesthesia type.
6. Wide Age Range: Our study included patients from 0 to 18 years, encompassing infants, children, and adolescents. The electrophysiological characteristics and responses to anesthetic agents may differ substantially across this developmental spectrum, and pooling such a heterogeneous age range may obscure age-specific effects.
7. Multi-Drug Protocols: Both anesthetic protocols involved multiple agents, making it impossible to attribute observed effects specifically to propofol versus other medications in the regimen.
8. Lack of Standardized Induction Protocols: Electrophysiological stimulation protocols, use of isoproterenol, and other procedural details were not standardized or systematically documented, introducing potential variability in arrhythmia induction attempts.
9. Autonomic Tone Confounding: We did not measure autonomic nervous system activity (e.g., heart rate variability, catecholamine levels) or control for baseline sympathetic tone. Given that both anesthetic techniques suppress autonomic activity—potentially the primary driver of WPW inducibility in young patients—we cannot distinguish between direct cardiac effects and indirect autonomic effects.