Submitted:
13 March 2026
Posted:
16 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Chronic Wounds: Definitions and Challenges
1.2. Infection as a Barrier to Wound Healing
1.3. Role of Biomaterials in Wound Management
1.3.1. Structural and Protective Functions
1.3.2. Antimicrobial and Anti-Biofilm Capabilities
1.3.3. Drug and Growth Factor Delivery
1.3.4. Immunomodulation and Microenvironment Regulation
1.3.5. Advanced and Smart Biomaterials
1.3.6. Clinical Potential and Limitations
2. Pathophysiology of Chronic Wound Infections
3. Biomaterial Platforms for Wound Healing Applications
3.1. Natural Polymers
3.2. Synthetic Polymers
3.3. Hybrid and Composite Biomaterials
4. Antimicrobial Strategies Using Biomaterials
4.1. Drug- and Antibiotic-Loaded Biomaterials
4.2. Antimicrobial Peptide-Loaded Systems
4.3. Metal-Based Nanomaterials
4.4. Bacteriophages-Loaded Biomaterials
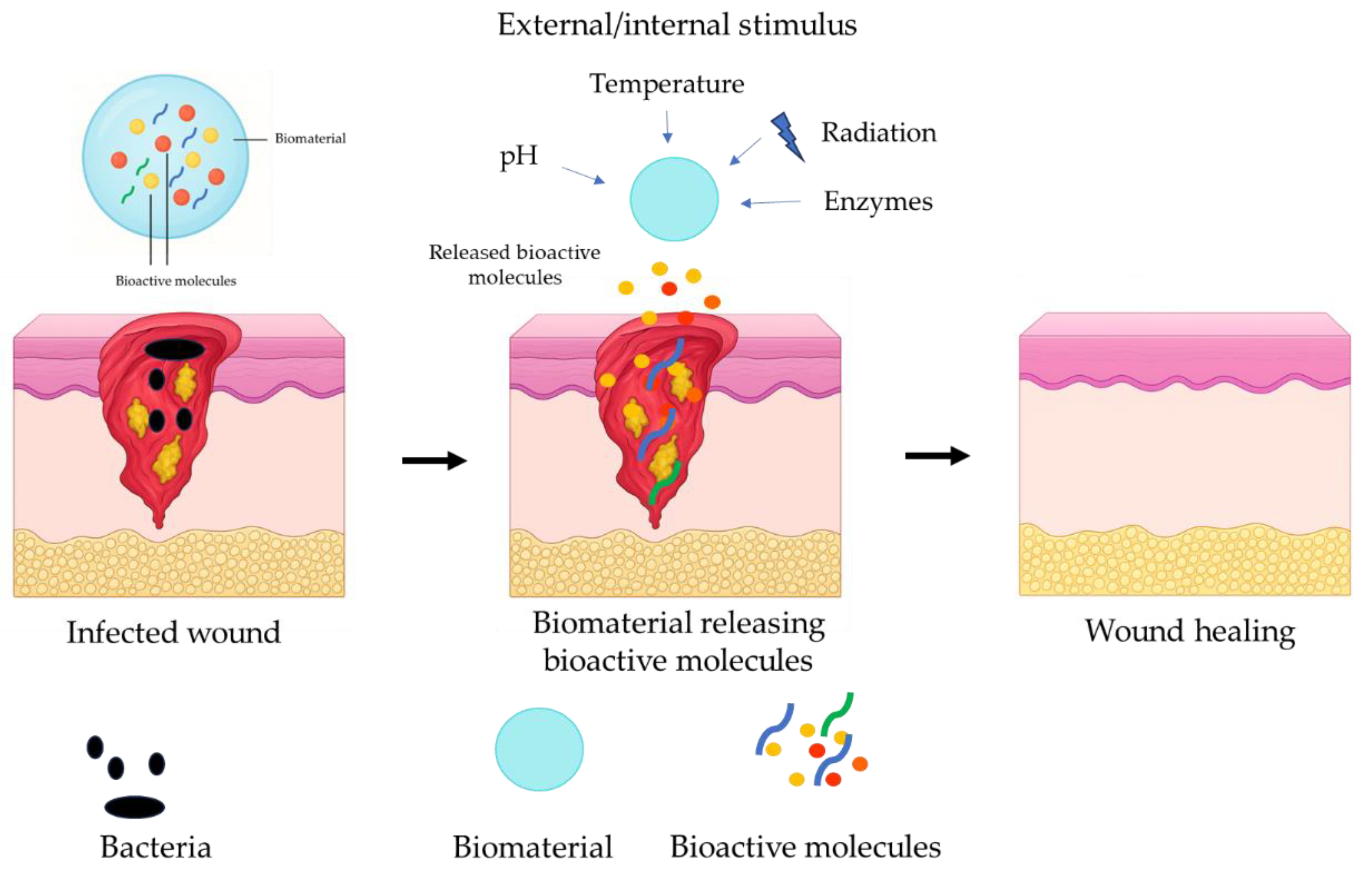
5. Smart and Responsive Biomaterials
6. Role of Biomaterials in Modulating the Wound Microenvironment
7. Surfactants and Antioxidant-Based Biomaterials for Infection Control in Chronic Wound Healing
7.1. Poloxamers as Functional Surfactants in Wound Biomaterials
7.2. Melatonin as an Antioxidant and Immunomodulatory Agent
7.3. Curcumin and Other Antioxidants
8. Translational Challenges and Clinical Applications
9. Future Perspectives
10. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Falanga, V.; Isseroff, R.R.; Soulika, A.M.; Romanelli, M.; Margolis, D.; Kapp, S.; Granick, M.; Harding, K. Chronic wounds. Nat Rev Dis Primers 2022, 8, 50. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Banks, J. Challenges in the Treatment of Chronic Wounds. Adv Wound Care (New Rochelle) 2015, 4, 560–582. [Google Scholar] [CrossRef]
- Hunt, M.; Torres, M.; Bachar-Wikstrom, E.; Wikstrom, J.D. Multifaceted roles of mitochondria in wound healing and chronic wound pathogenesis. Front Cell Dev Biol 2023, 11, 1252318. [Google Scholar] [CrossRef]
- Monika, P.; Chandraprabha, M.N.; Rangarajan, A.; Waiker, P.V.; Chidambara Murthy, K.N. Challenges in Healing Wound: Role of Complementary and Alternative Medicine. Front Nutr 2021, 8, 791899. [Google Scholar] [CrossRef]
- Papadopoulou, V.; Sidders, A.E.; Lu, K.Y.; Velez, A.Z.; Durham, P.G.; Bui, D.T.; Angeles-Solano, M.; Dayton, P.A.; Rowe, S.E. Overcoming biological barriers to improve treatment of a Staphylococcus aureus wound infection. Cell Chem Biol 2023, 30, 513–526 e515. [Google Scholar] [CrossRef]
- Zhang, S.J.; Xu, R.; He, S.B.; Sun, R.; Wang, G.N.; Wei, S.Y.; Yan, X.Y.; Fan, K.L. Nanozyme-driven multifunctional dressings: moving beyond enzyme-like catalysis in chronic wound treatment. Mil Med Res 2025, 12, 27. [Google Scholar] [CrossRef]
- Nguyen, H.M.; Ngoc Le, T.T.; Nguyen, A.T.; Thien Le, H.N.; Pham, T.T. Biomedical materials for wound dressing: recent advances and applications. RSC Adv 2023, 13, 5509–5528. [Google Scholar] [CrossRef]
- Pereira, A.B.V.; Terroso, M.S.; Gonçalves, H.; Catita, J.; Santos, D.; Baptista, R.M.F.; Monteiro, F.J.; Lopes, C.M.; Lúcio, M.; Ferraz, M.P. Hybrid nano-scaffolds loaded with resveratrol and Omega-3 fatty Acids: An innovative antimicrobial strategy against biofilm. Int J Pharmaceut 2025, 680. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.B.V.; Terroso, M.S.; Gonçalves, H.; Catita, J.; Santos, D.; Baptista, R.M.F.; Monteiro, F.J.; Lúcio, M.; Lopes, C.M.; Ferraz, M.P. Quercetin and omega-3 fatty acids-loaded lipid hybrid nanoassemblies as promising antimicrobial biofilm strategies. Materials Today Nano 2025, 32. [Google Scholar] [CrossRef]
- Ribeiro, M.; Ferraz, M.P.; Monteiro, F.J.; Fernandes, M.H.; Beppu, M.M.; Mantione, D.; Sardon, H. Antibacterial silk fibroin/nanohydroxyapatite hydrogels with silver and gold nanoparticles for bone regeneration. Nanomed-Nanotechnol 2017, 13, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Grenho, L.; Monteiro, F.J.; Ferraz, M.P. In vitro analysis of the antibacterial effect of nanohydroxyapatite-ZnO composites. J Biomed Mater Res A 2014, 102, 3726–3733. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, S.; Martin, V.; Pereira, J.C.; David, A.B.; Araújo, R.; Gonçalves, L.; Landin, M.; Gomes, P.; Ferraz, M.P.; Ribeiro, I.A.C.; et al. 3D-printed chitosan-starch mesh filled with minocycline-alginate hydrogel for dual anti-Staphylococcus aureus and osteogenic effects. Carbohydrate Polymers 2025, 370, 124311. [Google Scholar] [CrossRef] [PubMed]
- Barros, J.; Monteiro, F.J.; Ferraz, M.P. Bioengineering Approaches to Fight against Orthopedic Biomaterials Related-Infections. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Kasapgil, E.; Garay-Sarmiento, M.; Rodriguez-Emmenegger, C. Advanced Antibacterial Strategies for Combatting Biomaterial-Associated Infections: A Comprehensive Review. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2024, 16, e2018. [Google Scholar] [CrossRef] [PubMed]
- Landa, G.; Mendoza, G.; Irusta, S.; Arruebo, M. Bacteria-Responsive Nanostructured Drug Delivery Systems for Targeted Antimicrobial Therapy. Adv Mater 2025, e10355. [CrossRef]
- Chabria, Y.; Duffy, G.P.; Lowery, A.J.; Dwyer, R.M. Hydrogels: 3D Drug Delivery Systems for Nanoparticles and Extracellular Vesicles. Biomedicines 2021, 9. [Google Scholar] [CrossRef]
- Zegre, M.; Barros, J.; Ribeiro, I.A.C.; Santos, C.; Caetano, L.A.; Goncalves, L.; Monteiro, F.J.; Ferraz, M.P.; Bettencourt, A. Poly(DL-lactic acid) scaffolds as a bone targeting platform for the co-delivery of antimicrobial agents against mixed biofilms. Int J Pharmaceut 2022, 622. [Google Scholar] [CrossRef]
- Beheshtizadeh, N.; Gharibshahian, M.; Bayati, M.; Maleki, R.; Strachan, H.; Doughty, S.; Tayebi, L. Vascular endothelial growth factor (VEGF) delivery approaches in regenerative medicine. Biomed Pharmacother 2023, 166. [Google Scholar] [CrossRef]
- Ferraz, M.P. Advanced Nanotechnological Approaches for Biofilm Prevention and Control. Appl Sci-Basel 2024, 14. [Google Scholar] [CrossRef]
- Saberian, M.; Abak, N. Hydrogel-mediated delivery of platelet-derived exosomes: Innovations in tissue engineering. Heliyon 2024, 10, e24584. [Google Scholar] [CrossRef]
- Ferraz, M.P. Wound Dressing Materials: Bridging Material Science and Clinical Practice. Appl Sci-Basel 2025, 15. [Google Scholar] [CrossRef]
- Sousa, A.B.; Aguas, A.P.; Barbosa, M.A.; Barbosa, J.N. Immunomodulatory biomaterial-based wound dressings advance the healing of chronic wounds via regulating macrophage behavior. Regen Biomater 2022, 9, rbac065. [Google Scholar] [CrossRef]
- Zhang, S.; Ge, G.; Qin, Y.; Li, W.; Dong, J.; Mei, J.; Ma, R.; Zhang, X.; Bai, J.; Zhu, C.; et al. Recent advances in responsive hydrogels for diabetic wound healing. Mater Today Bio 2023, 18, 100508. [Google Scholar] [CrossRef]
- Delgado-Pujol, E.J.; Martínez, G.; Casado-Jurado, D.; Vázquez, J.; León-Barberena, J.; Rodríguez-Lucena, D.; Torres, Y.; Alcudia, A.; Begines, B. Hydrogels and Nanogels: Pioneering the Future of Advanced Drug Delivery Systems. Pharmaceutics 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Fadilah, N.I.M.; Maarof, M.; Motta, A.; Tabata, Y.; Fauzi, M.B. The Discovery and Development of Natural-Based Biomaterials with Demonstrated Wound Healing Properties: A Reliable Approach in Clinical Trials. Biomedicines 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Alberts, A.; Tudorache, D.I.; Niculescu, A.G.; Grumezescu, A.M. Advancements in Wound Dressing Materials: Highlighting Recent Progress in Hydrogels, Foams, and Antimicrobial Dressings. Gels 2025, 11. [Google Scholar] [CrossRef]
- Tian, S.; Bian, W. Advanced biomaterials in pressure ulcer prevention and care: from basic research to clinical practice. Front Bioeng Biotechnol 2025, 13, 1535588. [Google Scholar] [CrossRef]
- Diban, F.; Di Lodovico, S.; Di Fermo, P.; D'Ercole, S.; D'Arcangelo, S.; Di Giulio, M.; Cellini, L. Biofilms in Chronic Wound Infections: Innovative Antimicrobial Approaches Using the In Vitro Lubbock Chronic Wound Biofilm Model. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Wilkinson, H.N.; Hardman, M.J. Wound healing: cellular mechanisms and pathological outcomes. Open Biol 2020, 10, 200223. [Google Scholar] [CrossRef]
- Pastar, I.; Balukoff, N.C.; Marjanovic, J.; Chen, V.Y.; Stone, R.C.; Tomic-Canic, M. Molecular Pathophysiology of Chronic Wounds: Current State and Future Directions. Cold Spring Harb Perspect Biol 2023, 15. [Google Scholar] [CrossRef]
- Oliveira, C.; Sousa, D.; Teixeira, J.A.; Ferreira-Santos, P.; Botelho, C.M. Polymeric biomaterials for wound healing. Front Bioeng Biotechnol 2023, 11, 1136077. [Google Scholar] [CrossRef]
- Ansari, M.; Darvishi, A. A review of the current state of natural biomaterials in wound healing applications. Front Bioeng Biotechnol 2024, 12, 1309541. [Google Scholar] [CrossRef]
- Zhang, H.; Lin, X.; Cao, X.; Wang, Y.; Wang, J.; Zhao, Y. Developing natural polymers for skin wound healing. Bioact Mater 2024, 33, 355–376. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Raines, R.T. Review collagen-based biomaterials for wound healing. Biopolymers 2014, 101, 821–833. [Google Scholar] [CrossRef] [PubMed]
- Rajinikanth, B.S.; Rajkumar, D.S.R.; K, K.; Vijayaragavan, V. Chitosan-Based Biomaterial in Wound Healing: A Review. Cureus 2024, 16, e55193. [Google Scholar] [CrossRef] [PubMed]
- Aderibigbe, B.A.; Buyana, B. Alginate in Wound Dressings. Pharmaceutics 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Zhong, C.; Tian, X.; Zha, L.; Hou, L.; Feng, X. Emerging roles of hyaluronic acid hydrogels in cancer treatment and wound healing: A review. Int J Biol Macromol 2025, 303, 140442. [Google Scholar] [CrossRef]
- Gill, A.S.; Sood, M.; Deol, P.K.; Kaur, I.P. Synthetic polymer based electrospun scaffolds for wound healing applications. J Drug Deliv Sci Tec 2023, 89. [Google Scholar] [CrossRef]
- Liang, W.; Ni, N.; Huang, Y.; Lin, C. An Advanced Review: Polyurethane-Related Dressings for Skin Wound Repair. Polymers (Basel) 2023, 15. [Google Scholar] [CrossRef]
- Robles, K.N.; Zahra, F.T.; Mu, R.; Giorgio, T. Advances in Electrospun Poly(ε-caprolactone)-Based Nanofibrous Scaffolds for Tissue Engineering. Polymers (Basel) 2024, 16. [Google Scholar] [CrossRef]
- Mo, Y.; Zhou, T.; Li, W.; Niu, Y.; Sheu, C. Advances in Nanohybrid Hydrogels for Wound Healing: From Functional Mechanisms to Translational Prospects. Gels 2025, 11, 483. [Google Scholar] [CrossRef]
- Prete, S.; Dattilo, M.; Patitucci, F.; Pezzi, G.; Parisi, O.I.; Puoci, F. Natural and Synthetic Polymeric Biomaterials for Application in Wound Management. Journal of Functional Biomaterials 2023, 14, 455. [Google Scholar] [CrossRef] [PubMed]
- Phadtare, P.; Viswapriya, V.; Shinde, V.; Mathure, D.; Salunke, M.R. Hybrid bionanocomposites as the advancements in biomedical utility. Hybrid Advances 2025, 8, 100365. [Google Scholar] [CrossRef]
- Attasgah, R.B.; Velasco-Rodríguez, B.; Pardo, A.; Fernández-Vega, J.; Arellano-Galindo, L.; Rosales-Rivera, L.C.; Prieto, G.; Barbosa, S.; Soltero, J.F.A.; Mahmoudi, M.; et al. Development of functional hybrid scaffolds for wound healing applications. iScience 2022, 25, 104019. [Google Scholar] [CrossRef]
- Miron, A.; Giurcaneanu, C.; Mihai, M.M.; Beiu, C.; Voiculescu, V.M.; Popescu, M.N.; Soare, E.; Popa, L.G. Antimicrobial Biomaterials for Chronic Wound Care. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Liang, Y.; Zhang, H.; Guo, B. Antibacterial biomaterials for skin wound dressing. Asian Journal of Pharmaceutical Sciences 2022, 17, 353–384. [Google Scholar] [CrossRef]
- Fadilah, N.I.M.; Nizam, N.A.A.K.; Fauzi, M.B. Antibacterial compounds-incorporated functional biomaterials for chronic wound healing application via 3D bioprinting: The mechanism of action. IJB 2024, 10. [Google Scholar] [CrossRef]
- Kalelkar, P.P.; Riddick, M.; García, A.J. Biomaterial-based delivery of antimicrobial therapies for the treatment of bacterial infections. Nat Rev Mater 2022, 7, 39–54. [Google Scholar] [CrossRef]
- Fenton, O.S.; Olafson, K.N.; Pillai, P.S.; Mitchell, M.J.; Langer, R. Advances in Biomaterials for Drug Delivery. Adv Mater 2018, e1705328. [Google Scholar] [CrossRef]
- Trucillo, P. Biomaterials for Drug Delivery and Human Applications. Materials (Basel) 2024, 17. [Google Scholar] [CrossRef]
- Kanaujia, K.A.; Mishra, N.; Rajinikanth, P.S.; Saraf, S.A. Antimicrobial peptides as antimicrobials for wound care management: A comprehensive review. J Drug Deliv Sci Tec 2024, 95, 105570. [Google Scholar] [CrossRef]
- Adnan, S.B.; Maarof, M.; Fauzi, M.B.; Md Fadilah, N.I. Antimicrobial Peptides in Wound Healing and Skin Regeneration: Dual Roles in Immunity and Microbial Defense. Int J Mol Sci 2025, 26. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.; Ferrao, R.; Marta, D.; Vilaca, A.; Lino, M.; Rondao, T.; Ji, J.; Paiva, A.; Ferreira, L. Antimicrobial Peptide-Tether Dressing Able to Enhance Wound Healing by Tissue Contact. ACS Appl Mater Interfaces 2022, 14, 24213–24228. [Google Scholar] [CrossRef]
- Liang, C.; Wang, H.; Lin, Z.; Zhang, C.; Liu, G.; Hu, Y. Augmented wound healing potential of photosensitive GelMA hydrogel incorporating antimicrobial peptides and MXene nanoparticles. Front Bioeng Biotechnol 2023, 11, 1310349. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Meng, L.; Sethi, G.; Wang, J.; Li, B. Peptide-driven approaches in advanced wound healing materials. Drug Discov Today 2025, 30, 104440. [Google Scholar] [CrossRef]
- Alzain, M.; Daghistani, H.; Shamrani, T.; Almoghrabi, Y.; Daghistani, Y.; Alharbi, O.S.; Sait, A.M.; Mufrrih, M.; Alhazmi, W.; Alqarni, M.A.; et al. Antimicrobial Peptides: Mechanisms, Applications, and Therapeutic Potential. Infect Drug Resist 2025, 18, 4385–4426. [Google Scholar] [CrossRef]
- Zheng, Q.; Chen, C.; Liu, Y.; Gao, J.; Li, L.; Yin, C.; Yuan, X. Metal Nanoparticles: Advanced and Promising Technology in Diabetic Wound Therapy. Int J Nanomedicine 2024, 19, 965–992. [Google Scholar] [CrossRef] [PubMed]
- Mendes, C.; Thirupathi, A.; Correa, M.; Gu, Y.; Silveira, P.C.L. The Use of Metallic Nanoparticles in Wound Healing: New Perspectives. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Faghani, G.; Azarniya, A. Emerging nanomaterials for novel wound dressings: From metallic nanoparticles and MXene nanosheets to metal-organic frameworks. Heliyon 2024, 10, e39611. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, Y. Phage therapy as a revitalized weapon for treating clinical diseases. Microbiome Res Rep 2025, 4, 35. [Google Scholar] [CrossRef]
- Pinto, A.M.; Cerqueira, M.A.; Banobre-Lopes, M.; Pastrana, L.M.; Sillankorva, S. Bacteriophages for Chronic Wound Treatment: from Traditional to Novel Delivery Systems. Viruses 2020, 12. [Google Scholar] [CrossRef]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.P.; Que, Y.A.; Resch, G.; Rousseau, A.F.; Ravat, F.; Carsin, H.; Le Floch, R.; et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): a randomised, controlled, double-blind phase 1/2 trial. Lancet Infect Dis 2019, 19, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Fukaya-Shiba, A.; Ogata, A.; Kuribayashi, R.; Sakurai, A.; Suzuki, K.; Takadama, S.; Nishimura, J.; Uchiyama, J.; Ohge, H.; Takeuchi, T.; et al. Regulatory considerations for developing phage therapy medicinal products for the treatment of antimicrobial resistant bacterial infections. Front Pharmacol 2025, 16. [Google Scholar] [CrossRef]
- Ferraz, M.P. Phage Therapy for Bone and Joint Infections: Challenges, Biological Dynamics, and Therapeutic Prospects in the Era of Antimicrobial Resistance. Applied Sciences 2026, 16, 2002. [Google Scholar] [CrossRef]
- Saghazadeh, S.; Rinoldi, C.; Schot, M.; Kashaf, S.S.; Sharifi, F.; Jalilian, E.; Nuutila, K.; Giatsidis, G.; Mostafalu, P.; Derakhshandeh, H.; et al. Drug delivery systems and materials for wound healing applications. Adv Drug Deliv Rev 2018, 127, 138–166. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, B.; Lv, D.; Li, W.; Lu, Y.; Luo, G. Biomaterials releasing drug responsively to promote wound healing via regulation of pathological microenvironment. Advanced Drug Delivery Reviews 2023, 196, 114778. [Google Scholar] [CrossRef]
- Shi, Y.; Guo, S.; Tian, J.; Xie, X.; Shi, J.; Zhang, X.; Wang, C. Biomaterials-mediated sequential drug delivery: emerging trends for wound healing. Asian Journal of Pharmaceutical Sciences 2025, 101088. [CrossRef]
- Kusnadi, K.; Herdiana, Y.; Rochima, E.; Putra, O.N.; Mohd Gazzali, A.; Muchtaridi, M. Collagen-Based Nanoparticles as Drug Delivery System in Wound Healing Applications. Int J Nanomedicine 2024, 19, 11321–11341. [Google Scholar] [CrossRef]
- He, X.; Han, Z.; Ruan, Y.; Wang, Z.; Liao, B.; Li, X.; Tan, J.; Han, X.; Shen, J.; Bai, D. Smart responsive biomaterials for spatiotemporal modulation of functional tissue repair. Mater Today Bio 2025, 33, 102063. [Google Scholar] [CrossRef]
- Xu, Z.; Fan, J.L.; Tian, W.G.; Ji, X.; Cui, Y.Q.; Nan, Q.Y.; Sun, F.F.; Zhang, J. Cellulose-Based pH-Responsive Janus Dressing with Unidirectional Moisture Drainage for Exudate Management and Diabetic Wounds Healing. Adv Funct Mater 2024, 34. [Google Scholar] [CrossRef]
- Blackburn, J.; Ousey, K.; Rippon, M.; Rogers, A.; Pastar, I.; Lev-Tov, H. Applying Antimicrobial Strategies in Wound Care Practice: A Review of the Evidence. Int Wound J 2025, 22, e70684. [Google Scholar] [CrossRef]
- Franco, D.; Calabrese, G.; Guglielmino, S.P.P.; Conoci, S. Metal-Based Nanoparticles: Antibacterial Mechanisms and Biomedical Application. Microorganisms 2022, 10. [Google Scholar] [CrossRef]
- Barros, J.; Melo, L.D.R.; Poeta, P.; Igrejas, G.; Ferraz, M.P.; Azeredo, J.; Monteiro, F.J. Lytic bacteriophages against multidrug-resistant, and isolates from orthopaedic implant-associated infections. Int J Antimicrob Ag 2019, 54, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Barros, J.A.R.; de Melo, L.D.R.; da Silva, R.A.R.; Ferraz, M.P.; Azeredo, J.C.V.D.; Pinheiro, V.M.D.; Colaço, B.J.A.; Fernandes, M.H.R.; Gomes, P.D.; Monteiro, F.J. Encapsulated bacteriophages in alginate-nanohydroxyapatite hydrogel as a novel delivery system to prevent orthopedic implant-associated infections. Nanomed-Nanotechnol 2020, 24. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Chang, Q.; Lu, F. Oxygen-releasing biomaterials for chronic wounds breathing: From theoretical mechanism to application prospect. Mater Today Bio 2023, 20, 100687. [Google Scholar] [CrossRef]
- Bayraktar, S.; Ustun, C.; Kehr, N.S. Oxygen Delivery Biomaterials in Wound Healing Applications. Macromol Biosci 2024, 24, e2300363. [Google Scholar] [CrossRef]
- Wang, Y.; Vizely, K.; Li, C.Y.; Shen, K.; Shakeri, A.; Khosravi, R.; Smith, J.R.; Alteza, E.; Zhao, Y.; Radisic, M. Biomaterials for immunomodulation in wound healing. Regen Biomater 2024, 11, rbae032. [Google Scholar] [CrossRef]
- Percival, S.L.; Mayer, D.; Kirsner, R.S.; Schultz, G.; Weir, D.; Roy, S.; Alavi, A.; Romanelli, M. Surfactants: Role in biofilm management and cellular behaviour. Int Wound J 2019, 16, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Di Spirito, N.A.; Grizzuti, N.; Lutz-Bueno, V.; Urciuoli, G.; Auriemma, F.; Pasquino, R. Pluronic F68 Micelles as Carriers for an Anti-Inflammatory Drug: A Rheological and Scattering Investigation. Langmuir 2024, 40, 1544–1554. [Google Scholar] [CrossRef]
- Makabenta, J.M.V.; Nabawy, A.; Chattopadhyay, A.N.; Jeon, T.; Park, J.; Lo, P.C.; Nosovitski, S.; Huang, R.; Li, C.H.; Jiang, M.; et al. Antimicrobial polymer-loaded hydrogels for the topical treatment of multidrug-resistant wound biofilm infections. J Control Release 2023, 362, 513–523. [Google Scholar] [CrossRef]
- Visvalingam, J.; Yakandawala, N.; Regmi, S.; Adeniji, A.; Sharma, P.; Sailer, M. Wound Gel Formulations Containing Poloxamer 407 and Polyhexanide Have In Vitro Antimicrobial and Antibiofilm Activity Against Wound-Associated Microbial Pathogens. Microorganisms 2024, 12. [Google Scholar] [CrossRef]
- Sohn, E.H.; Kim, S.N.; Lee, S.R. Melatonin's Impact on Wound Healing. Antioxidants (Basel) 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek-Szczepańska, B.; Grabska-Zielińska, S. Biopolymeric Scaffolds with Melatonin for Tissue Engineering-A Review. Int J Mol Sci 2025, 26. [Google Scholar] [CrossRef]
- Bulut, O.; Sorucu, A.; Akkoç, A.N.; Tinas, I.S. Comparison of the efficacy of royal jelly and melatonin combinations in experimentally induced wounds in geriatric and young mice. Ulus Travma Acil Cer 2024, 30, 397–405. [Google Scholar] [CrossRef]
- Chen, K.; Tong, C.C.; Yang, J.G.; Cong, P.F.; Liu, Y.; Shi, X.Y.; Liu, X.; Zhang, J.; Zou, R.F.; Xiao, K.S.; et al. Injectable melatonin-loaded carboxymethyl chitosan (CMCS)-based hydrogel accelerates wound healing by reducing inflammation and promoting angiogenesis and collagen deposition. J Mater Sci Technol 2021, 63, 236–245. [Google Scholar] [CrossRef]
- Romic, M.D.; Klaric, M.S.; Lovric, J.; Pepic, I.; Cetina-Cizmek, B.; Filipovic-Grcic, J.; Hafner, A. Melatonin-loaded chitosan/Pluronic® F127 microspheres as in situ forming hydrogel: An innovative antimicrobial wound dressing. Eur J Pharm Biopharm 2016, 107, 67–79. [Google Scholar] [CrossRef]
- Mohanty, C.; Sahoo, S.K. Curcumin and its topical formulations for wound healing applications. Drug Discovery Today 2017, 22, 1582–1592. [Google Scholar] [CrossRef] [PubMed]
- Sideek, S.A.; El-Nassan, H.B.; Fares, A.R.; ElMeshad, A.N.; Elkasabgy, N.A. Different Curcumin-Loaded Delivery Systems for Wound Healing Applications: A Comprehensive Review. Pharmaceutics 2023, 15, 38. [Google Scholar] [CrossRef] [PubMed]
- Hecker, A.; Schellnegger, M.; Hofmann, E.; Luze, H.; Nischwitz, S.P.; Kamolz, L.P.; Kotzbeck, P. The impact of resveratrol on skin wound healing, scarring, and aging. Int Wound J 2022, 19, 9–28. [Google Scholar] [CrossRef]
- Pereira, A.B.V.; Terroso, M.S.; Goncalves, H.; Catita, J.; Santos, D.; Baptista, R.M.F.; Monteiro, F.J.; Lopes, C.M.; Lucio, M.; Ferraz, M.P. Hybrid nano-scaffolds loaded with resveratrol and Omega-3 fatty Acids: An innovative antimicrobial strategy against biofilm. Int J Pharm 2025, 680, 125784. [Google Scholar] [CrossRef] [PubMed]
- Hiruthyaswamy, S.P.; Deepankumar, K. Suckerin based biomaterials for wound healing: a comparative review with natural protein-based biomaterials. Mater Adv 2025, 6, 1262–1277. [Google Scholar] [CrossRef]
- Sawaragi, E.; Sakamoto, M.; Katayama, Y.; Kawabata, S.; Somamoto, S.; Noda, K.; Morimoto, N. A prospective multicenter phase III clinical trial evaluating the efficacy and safety of silk elastin sponge in patients with skin defects. Sci Rep 2025, 15, 11279. [Google Scholar] [CrossRef] [PubMed]
- Kolimi, P.; Narala, S.; Nyavanandi, D.; Youssef, A.A.A.; Dudhipala, N. Innovative Treatment Strategies to Accelerate Wound Healing: Trajectory and Recent Advancements. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, D.; Kumar, S.; Agrawal, G. Multifunctional bilayer dressings: a next generation biomaterial for enhanced wound healing. Biomater Sci 2025, 13, 4639–4662. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zu, L.; Wang, S.; Li, J.; Fei, X.; Geng, M.; Zhu, C.; Shi, H. Tailored biomedical materials for wound healing. Burns Trauma 2023, 11, tkad040. [Google Scholar] [CrossRef]
| Advantages | Disadvantages | References | |
| Drug- and antibiotic-loaded biomaterials | Enable localized, sustained, and controlled delivery of antibiotics or drugs directly to the wound site, reducing the risk of systemic side effects and improving local concentrations. Protect fragile therapeutic agents from degradation and extend their activity within the wound environment. Enhance patient compliance by reducing the frequency of dressing changes and systemic medication. |
Risk of developing local antibiotic resistance, especially with long-term or repeated use. Potential cytotoxicity or irritation depending on drug type, release rate, or carrier composition. Challenges with production scalability, maintaining reproducibility, and ensuring consistent drug loading and release profiles. Regulatory hurdles due to the combination nature of the device and drug. |
[32,65,66,67] |
| Antimicrobial peptide-loaded systems | Broad-spectrum antimicrobial activity, often effective against drug-resistant bacteria and biofilms at the wound site. Peptides often possess immunomodulatory functions, promoting wound healing by stimulating cell migration and reducing inflammatory responses. Usually demonstrate low propensity to induce resistance compared to traditional antibiotics. |
Peptides may be susceptible to enzymatic degradation within the wound, limiting their practical effectiveness unless protected by the delivery vehicle. Potential for cytotoxic effects at higher concentrations or with prolonged exposure. Higher manufacturing costs and stability issues compared to small molecule drugs. Limited large-scale clinical validation to date. |
[32,68] |
| Metal-based nanomaterials | Exhibit potent and broad-spectrum antimicrobial activity, including efficacy against multidrug-resistant organisms and biofilm-associated bacteria. Can synergize with other wound healing mechanisms, such as anti-inflammatory effects and promotion of tissue regeneration. Typically stable and can be incorporated into a variety of biomaterial matrices (hydrogels, nanofibers, sponges). |
Cytotoxicity and potential local or systemic toxicity, especially with metals like silver, copper, or high concentrations of nanoparticles. Long-term safety concerns regarding accumulation or leaching of metal ions. Manufacturing complexity and cost considerations for consistent size, dispersion, and controlled release. Risk of impaired healing or foreign body reactions in case of improper dosing or formulation. |
[10,11,19,21] |
| Bacteriophages-loaded biomaterials | Provide targeted antibacterial activity against specific pathogens, reducing damage to commensal microbiota and helping maintain a balanced wound microenvironment. Enable localized, sustained release of active phages from dressings, hydrogels, or scaffolds, improving biofilm penetration and bacterial clearance while limiting systemic exposure and side effects. Can be engineered as multifunctional systems combining phages with other antimicrobial or pro-regenerative agents (e.g., antiseptics, growth factors), thereby simultaneously controlling infection and supporting tissue repair. |
Phages may be unstable within some biomaterial matrices and are sensitive to temperature, pH, and storage conditions, which can lead to titer loss and reduced therapeutic efficacy over time. Bacterial populations can evolve phage resistance, especially if formulations use narrow phage spectra or are not periodically updated to match circulating clinical isolates. Clinical translation is hindered by limited standardization in manufacturing, challenges in quality control, and unclear or complex regulatory pathways for products that combine biologics with medical devices |
[60,61,62] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




