Submitted:
13 March 2026
Posted:
16 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Asymmetric Organocatalysis in Deep-Eutectic-Solvents
2.1. In DESs and NADES
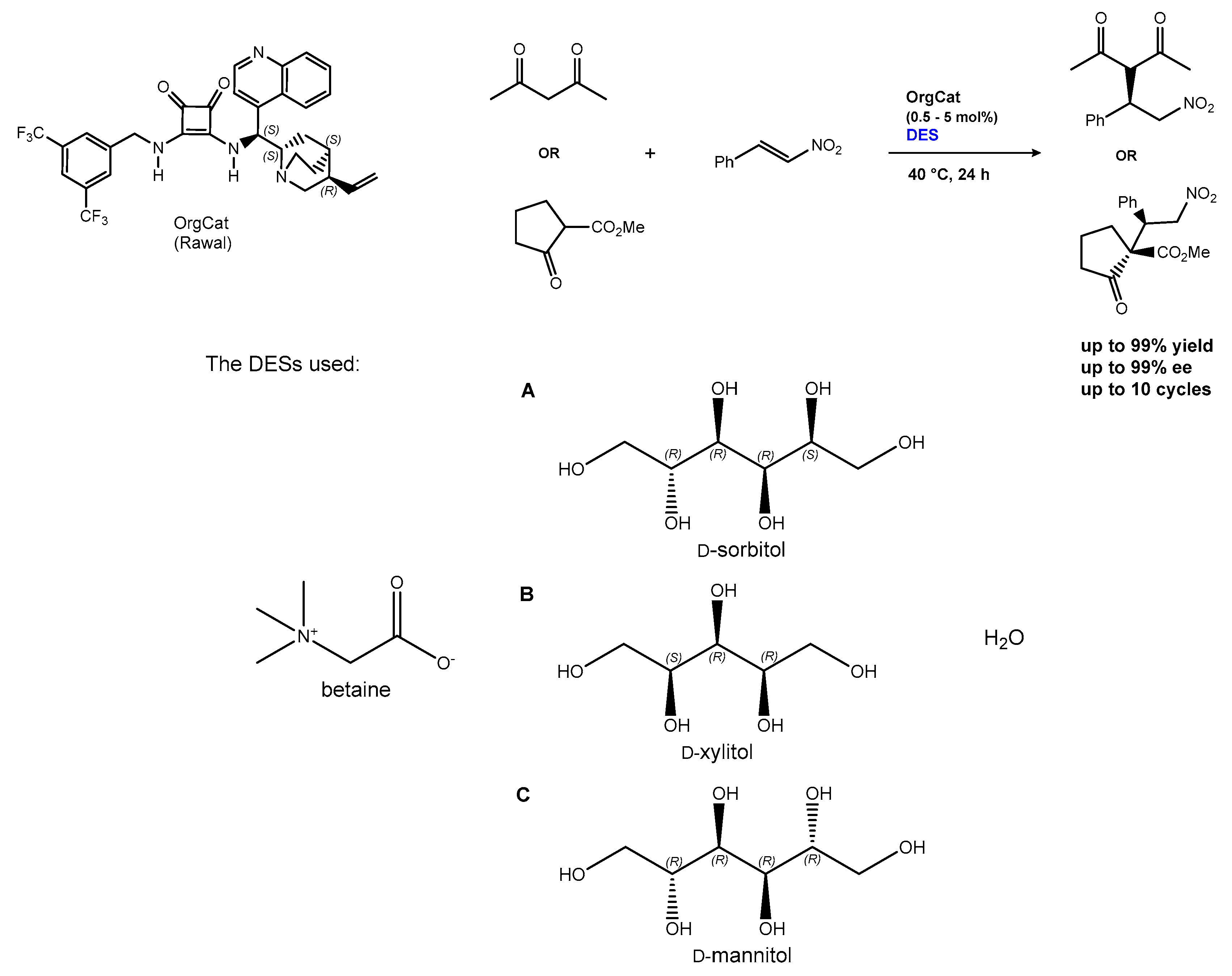
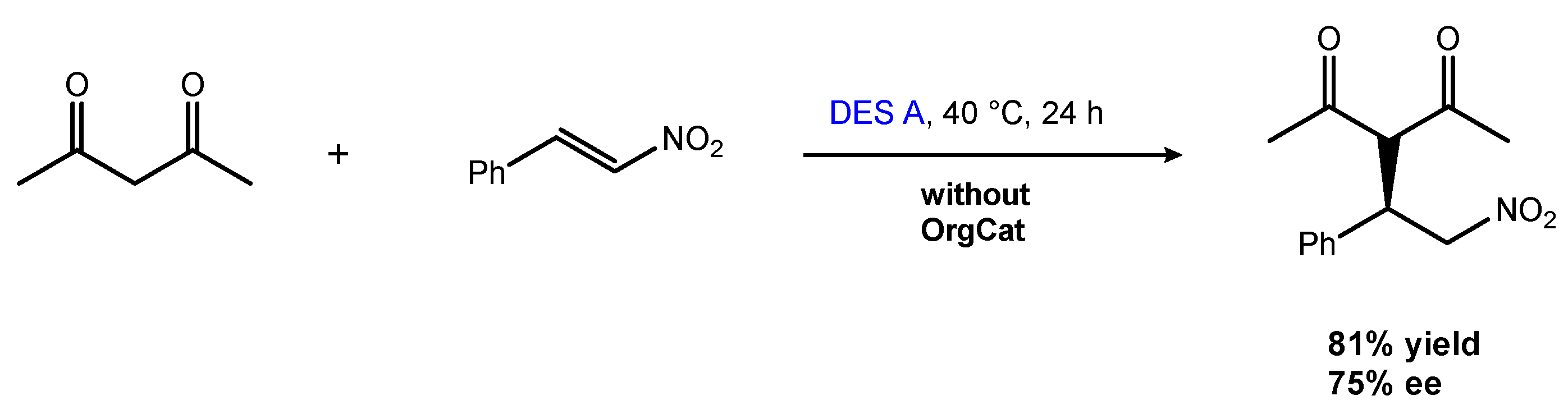
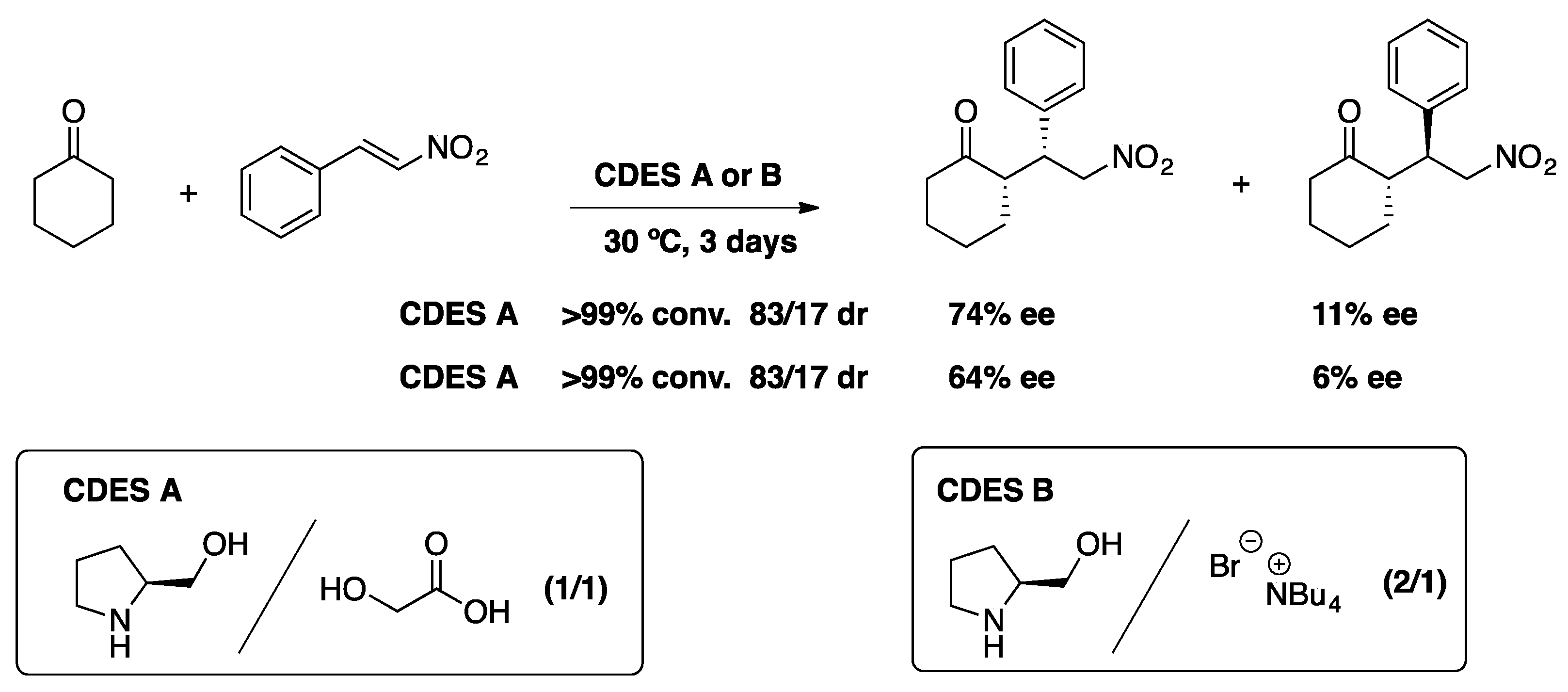
2.1.1. The Michael and Other Conjugate Additions
2.1.2. The Aldol Condensation
2.1.3. Carbonyl α-Aminations
2.1.5. Multi-Component Reactions
2.2. In Eutectogels
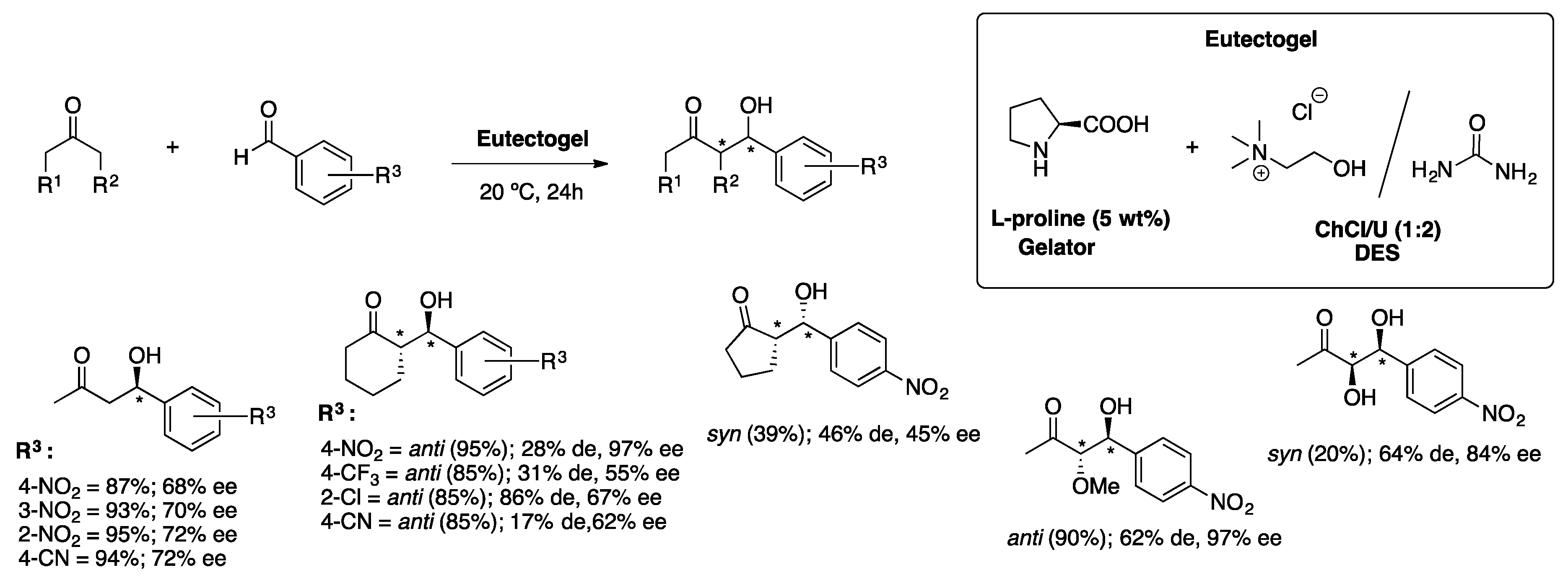
2.2.1. The Aldol Reaction
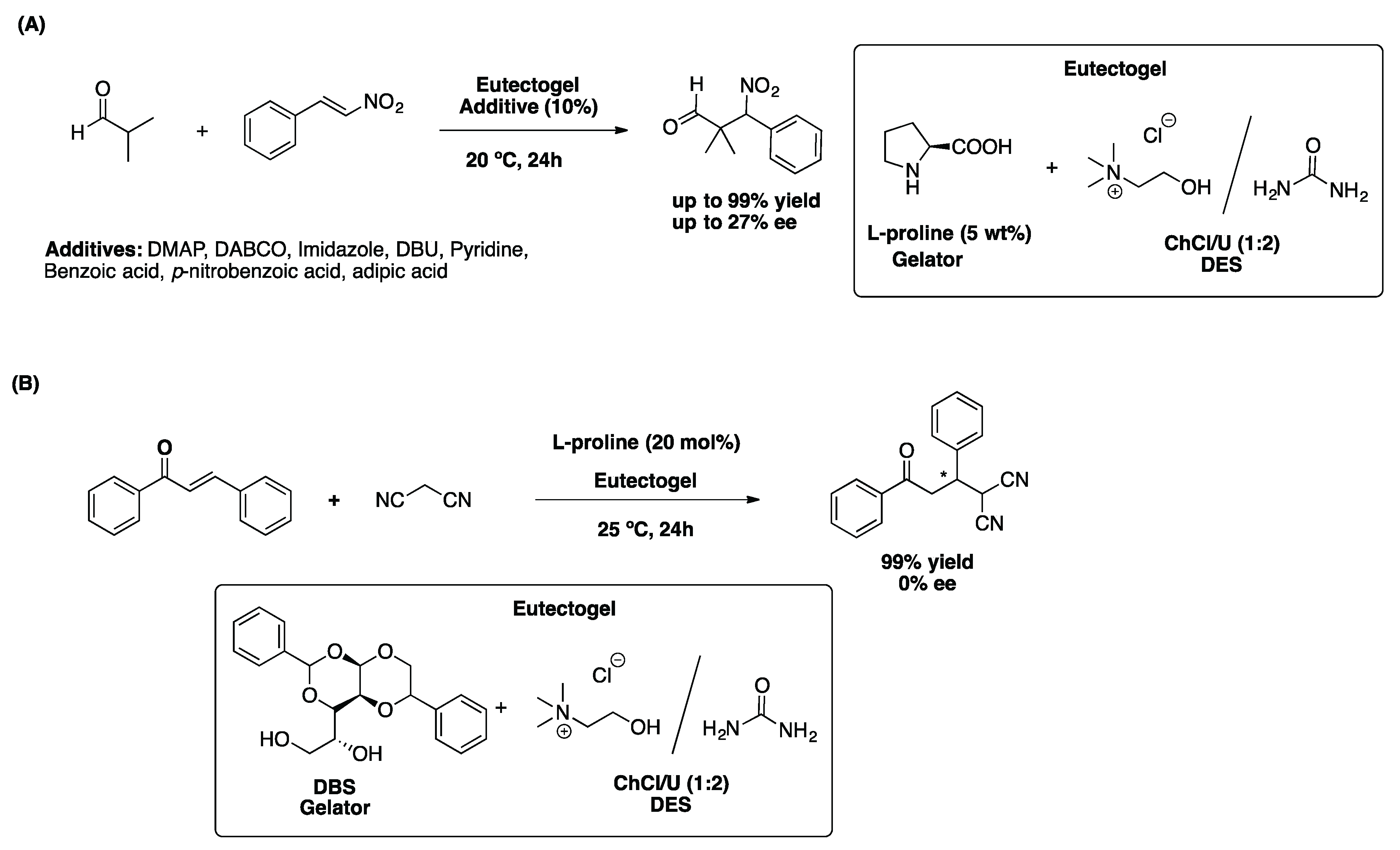
2.2.2. The Michael Reaction
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Hansen, B.B.; Spittle, S.; Chen, B.; Poe, D.; Zhang, Y.; Klein, J.M.; Horton, A.; Adhikari, L.; Zelovich, T.; Doherty, B.W.; et al. Deep Eutectic Solvents: A Review of Fundamentals and Applications. Chem. Rev. 2021, 121, 1232–1285. [Google Scholar] [CrossRef]
- Carreiro, E.P.; Federsel, H.-J.; Hermann, G.J.; Burke, A.J. Stereoselective Catalytic Synthesis of Bioactive Compounds in Natural Deep Eutectic Solvents (NADESs): A Survey across the Catalytic Spectrum. Catalysts 2024, 14, 160. [Google Scholar] [CrossRef]
- Burke, A.J.; Carreiro, E.P.; Federsel, H.-J. How Deep Eutectic Solvents Are Currently Shaping Organocatalytic and Enzymatic Asymmetric Catalysis. RSC Sustainability 2025, 3, 3883–3890. [Google Scholar] [CrossRef]
- Perna, F.M.; Vitale, P.; Capriati, V. Deep Eutectic Solvents and Their Applications as Green Solvents. Current Opinion in Green and Sustainable Chemistry 2020, 21, 27–33. [Google Scholar] [CrossRef]
- Yu, D.; Xue, Z.; Mu, T. Deep Eutectic Solvents as a Green Toolbox for Synthesis. Cell Reports Physical Science 2022, 3, 100809. [Google Scholar] [CrossRef]
- Khan, L.; Hasan, A.; Alvi, S.; Abbasi, M.; Ali, R. Organic Named Reactions in Green Deep Eutectic Solvents (DESs). Top Curr Chem (Z) 2025, 383, 36. [Google Scholar] [CrossRef]
- Zlotin, S.G.; Egorova, K.S.; Ananikov, V.P.; Akulov, A.A.; Varaksin, M.V.; Chupakhin, O.N.; Charushin, V.N.; Bryliakov, K.P.; Averin, A.D.; Beletskaya, I.P.; et al. The Green Chemistry Paradigm in Modern Organic Synthesis. RUSS CHEM REV 2023, 92, RCR5104. [Google Scholar] [CrossRef]
- Alonso, D.A.; Burlingham, S.; Chinchilla, R.; Guillena, G.; Ramón, D.J.; Tiecco, M. Asymmetric Organocatalysis in Deep Eutectic Solvents. Eur J Org Chem 2021, 2021, 4065–4071. [Google Scholar] [CrossRef]
- Fanjul-Mosteirín, N.; Del Amo, V. Organocatalytic Transformations in Deep Eutectic Solvents: Green Methodologies Made Greener. Tetrahedron 2021, 84. [Google Scholar] [CrossRef]
- Tolmachev, D.; Lukasheva, N.; Ramazanov, R.; Nazarychev, V.; Borzdun, N.; Volgin, I.; Andreeva, M.; Glova, A.; Melnikova, S.; Dobrovskiy, A.; et al. Computer Simulations of Deep Eutectic Solvents: Challenges, Solutions, and Perspectives. IJMS 2022, 23, 645. [Google Scholar] [CrossRef]
- Müller, C.R.; Meiners, I.; Domínguez de María, P. Highly enantioselective tandem enzyme–organocatalyst crossed aldol reactions with acetaldehyde in deep-eutectic solvents. RSC Adv. 2014, 4, 46097–46101. [Google Scholar] [CrossRef]
- Massolo, E.; Palmieri, S.; Benaglia, M.; Capriati, V.; Perna, F.M. Stereoselective Organocatalysed Reactions in Deep Eutectic Solvents: Highly Tunable and Biorenewable Reaction Media for Sustainable Organic Synthesis. Green Chem. 2016, 18, 792–797. [Google Scholar] [CrossRef]
- Brenna, D.; Massolo, E.; Puglisi, A.; Rossi, S.; Celentano, G.; Benaglia, M.; Capriati, V. Towards the Development of Continuous, Organocatalytic, and Stereoselective Reactions in Deep Eutectic Solvents. Beilstein J. Org. Chem. 2016, 12, 2620–2626. [Google Scholar] [CrossRef]
- Martínez, R.; Berbegal, L.; Guillena, G.; Ramón, D.J. Bio-Renewable Enantioselective Aldol Reaction in Natural Deep Eutectic Solvents. Green Chem. 2016, 18, 1724–1730. [Google Scholar] [CrossRef]
- Fanjul-Mosteirín, N.; Concellón, C.; Del Amo, V. L -Isoleucine in a Choline Chloride/Ethylene Glycol Deep Eutectic Solvent: A Reusable Reaction Kit for the Asymmetric Cross-Aldol Carboligation. Org. Lett. 2016, 18, 4266–4269. [Google Scholar] [CrossRef]
- Flores-Ferrándiz, J.; Chinchilla, R. Organocatalytic Enantioselective Conjugate Addition of Aldehydes to Maleimides in Deep Eutectic Solvents. Tetrahedron: Asymmetry 2017, 28, 302–306. [Google Scholar] [CrossRef]
- Ros Ñíguez, D.; Guillena, G.; Alonso, D.A. Chiral 2-Aminobenzimidazoles in Deep Eutectic Mixtures: Recyclable Organocatalysts for the Enantioselective Michael Addition of 1,3-Dicarbonyl Compounds to β-Nitroalkenes. ACS Sustain. Chem. Eng. 2017, 5, 10649–10656. [Google Scholar] [CrossRef]
- Ros Ñíguez, D.; Khazaeli, P.; Alonso, D.A.; Guillena, G. Deep Eutectic Mixtures as Reaction Media for the Enantioselective Organocatalyzed α-Amination of 1,3-Dicarbonyl Compounds. Catalysts 2018, 8, 217. [Google Scholar] [CrossRef]
- Palomba, T.; Ciancaleoni, G.; Del Giacco, T.; Germani, R.; Ianni, F.; Tiecco, M. Deep Eutectic Solvents Formed by Chiral Components as Chiral Reaction Media and Studies of Their Structural Properties. Journal of Molecular Liquids 2018, 262, 285–294. [Google Scholar] [CrossRef]
- Tiecco, M.; Alonso, D.A.; Ñíguez, D.R.; Ciancaleoni, G.; Guillena, G.; Ramón, D.J.; Bonillo, A.A.; Germani, R. Assessment of the Organocatalytic Activity of Chiral L-Proline-Based Deep Eutectic Solvents Based on Their Structural Features. Journal of Molecular Liquids 2020, 313, 113573. [Google Scholar] [CrossRef]
- Torregrosa-Chinillach, A.; Sánchez-Laó, A.; Santagostino, E.; Chinchilla, R. Organocatalytic Asymmetric Conjugate Addition of Aldehydes to Maleimides and Nitroalkenes in Deep Eutectic Solvents. Molecules 2019, 24, 4058. [Google Scholar] [CrossRef]
- Fonseca, D.P.; Amorim, A.C.; Carreiro, E.P.; Ramalho, J.P.P.; Hermann, G.J.; Federsel, H.-J.; Duarte, A.R.C.; Burke, A.J. Sustainable Organocatalyzed Enantioselective Catalytic Michael Additions in Betaine-Derived Deep Eutectic Solvents. SynOpen 2023, 07, 374–380. [Google Scholar] [CrossRef]
- Burlingham, S.J.; Ñíguez, J.A.; Torregrosa-Chinillach, A.; Ros Ñíguez, D.; Chinchilla, R.; Such-Basáñez, I.; Tiecco, M.; Alonso, D.A. Synthesis, Characterization and Organocatalytic Activity of Novel Chiral (Ammoniummethyl)Pyrrolidine-Derived Deep Eutectic Solvents. Journal of Molecular Liquids 2024, 411, 125724. [Google Scholar] [CrossRef]
- Ñíguez, J.A.; Burlingham, S.J.; Chinchilla, R.; Pérez, J.M.; Fernández, I.; Alonso, D.A. Synthesis and Structural Characterization of L -Prolinol Derived Chiral Eutectic Mixtures as Sustainable Solvents in Asymmetric Organocatalysis. RSC Sustainability 2024, 2, 499–509. [Google Scholar] [CrossRef]
- Al Beiruty, H.; Zhylinska, S.-S.; Kutateladze, N.; Cheong, H.K.T.; Ñíguez, J.A.; Burlingham, S.J.; Marset, X.; Guillena, G.; Chinchilla, R.; Alonso, D.A.; et al. Enantioselective catalytic aldol reactions in the presence of Knoevenagel nucleophiles: A chemoselective switch optimized in deep eutectic solvents using mechanochemistry. Molecules 2024, 29, 4. [Google Scholar] [CrossRef]
- Zárate-Roldán, S.; Trujillo-Rodríguez, M.J.; Gimeno, M.C.; Herrera, R.P. L-Proline-Based Deep Eutectic Solvents as Green and Enantioselective Organocatalyst/Media for Aldol Reaction. Journal of Molecular Liquids 2024, 396, 123971. [Google Scholar] [CrossRef]
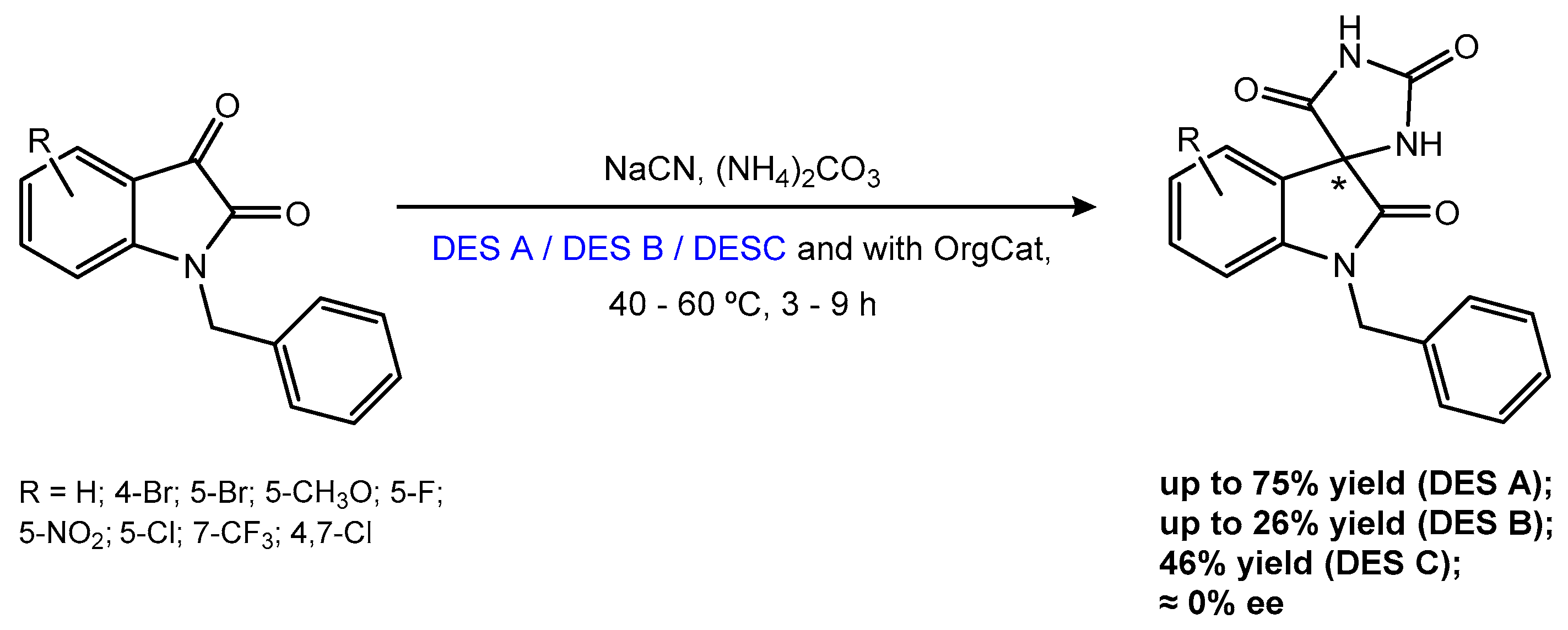
- Moura, M.B.V.; Monteiro, H.; Alves, C.; Fernandes, C.; Amorim, A.C.; Duarte, A.R.C.; Burke, A.J. The Bucherer–Bergs reaction in deep eutectic solvents: Accessing spiro-oxindole-hydantoins. J. Mol. Liq. 2026, 129390. in press. [Google Scholar] [CrossRef]
- Mercadal, P.A.; González, A.; Beloqui, A.; Tomé, L.C.; Mecerreyes, D.; Calderón, M.; Picchio, M.L. Eutectogels: The Multifaceted Soft Ionic Materials of Tomorrow. JACS Au 2024, 4, 3744–3758. [Google Scholar] [CrossRef]
- Marullo, S.; Meli, A.; Dintcheva, N.T.; Infurna, G.; Rizzo, C.; D’Anna, F. Environmentally friendly eutectogels comprising L-amino acids and deep eutectic solvents: Efficient materials for wastewater treatment. ChemPlusChem 2020, 85, 301–308. [Google Scholar] [CrossRef]
- Zhang, Z.; Yao, A.; Raffa, P. Physically Crosslinked Amphiphilic Eutectogels with Underwater Self-Healing, Strong Adhesion, Environmental Stability and Closed-Loop Recyclability for Underwater Sensing and Information Transmission. Chemical Engineering Journal 2025, 522, 167958. [Google Scholar] [CrossRef]
- Zhang, P.; Xiong, W.; Shi, M.; Tu, Z.; Hu, X.; Zhang, X.; Wu, Y. Natural Deep Eutectic Solvent-Based Gels with Multi-Site Interaction Mechanism for Selective Membrane Separation of SO2 from N2 and CO2. Chemical Engineering Journal 2022, 438, 135626. [Google Scholar]
- Li, M.; Guan, C.; Zhi, Y.; Li, J.; Sun, S.; Zhuang, X.; Fang, Y.; Peng, J. Water-Resistant Supramolecular Eutectogel Lubricant with MoS2 : Thixotropic Behavior and High-Efficiency Lubrication. ACS Appl. Mater. Interfaces 2025, 17, 46197–46207. [Google Scholar] [CrossRef]
- Mercken, J.; De Sloovere, D.; Joos, B.; Ghogare, D.; Verhille, Y.; Smeets, S.; Derveaux, E.; Adriaensens, P.; Van Bael, M.; Hardy, A. Organic modification of eutectogels enhances electrolyte/electrode contact in sodium-ion batteries. ChemSusChem 2025, 18, e202500427. [Google Scholar] [CrossRef]
- Tian, Y.; Sun, D.-W.; Xu, L.; Fan, T.-H.; Zhu, Z. Bio-Inspired Eutectogels Enabled by Binary Natural Deep Eutectic Solvents (NADESs): Interfacial Anti-Frosting, Freezing-Tolerance, and Mechanisms. Food Hydrocolloids 2022, 128, 107568. [Google Scholar] [CrossRef]
- Cassano, R.; Sole, R.; Siciliano, C.; Baldino, N.; Mileti, O.; Procopio, D.; Curcio, F.; Calviello, G.; Serini, S.; Trombino, S.; et al. Eutectogel-Based Drug Delivery: An Innovative Approach for Atenolol Administration. Pharmaceutics 2024, 16, 1552. [Google Scholar] [CrossRef]
- Meneses, L.; Jesus, A.R. Eutectogels: Recent Advances, Design Strategies, and Emerging Applications in Biotechnology. Gels 2025, 11, 1013. [Google Scholar] [CrossRef]
- Kumar, K.; Calderón, M.; Beloqui, A.; Picchio, M.L. Eutectogels as Promising Materials in Biocatalysis. ChemCatChem 2024, 16, e202400204. [Google Scholar]
- Cheng, Q.; Hao, A.; Xing, P. Eutectogels as Matrices to Manipulate Supramolecular Chirality and Circularly Polarized Luminescence. ACS Nano 2022, 16, 6825–6834. [Google Scholar] [CrossRef]
- Saavedra, B.; Meli, A.; Rizzo, C.; Ramón, D.J.; D’Anna, F. Natural Eutectogels: Sustainable Catalytic Systems for C–C Bond Formation Reactions. Green Chem. 2021, 23, 6555–6565. [Google Scholar] [CrossRef]
- Rizzo, C.; Marullo, S.; Benaglia, M.; D’Anna, F. DBS-based eutectogels: Organized vessels to perform the Michael addition reaction. Eur. J. Org. Chem. 2023, 26, e202300263. [Google Scholar] [CrossRef]
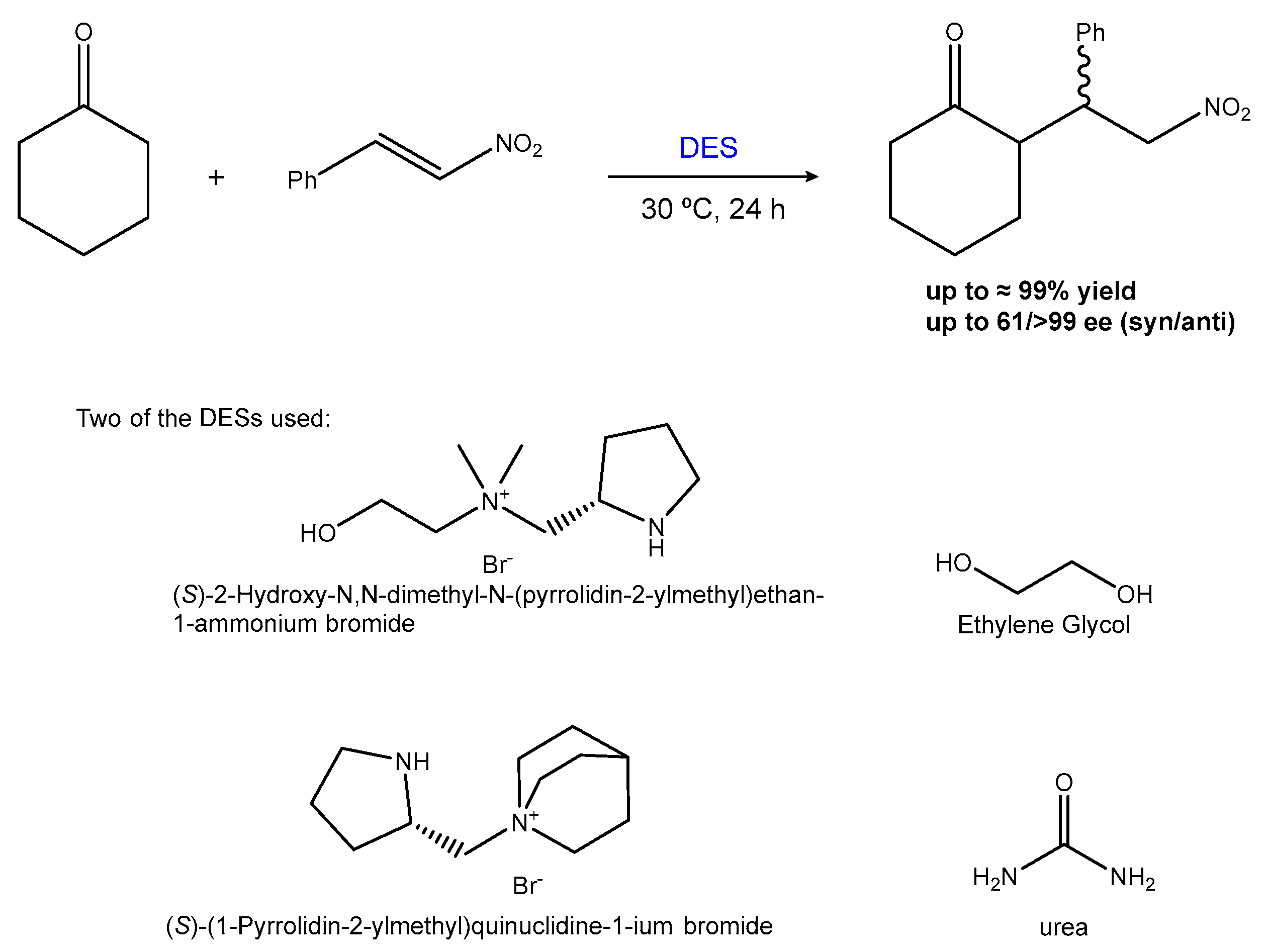
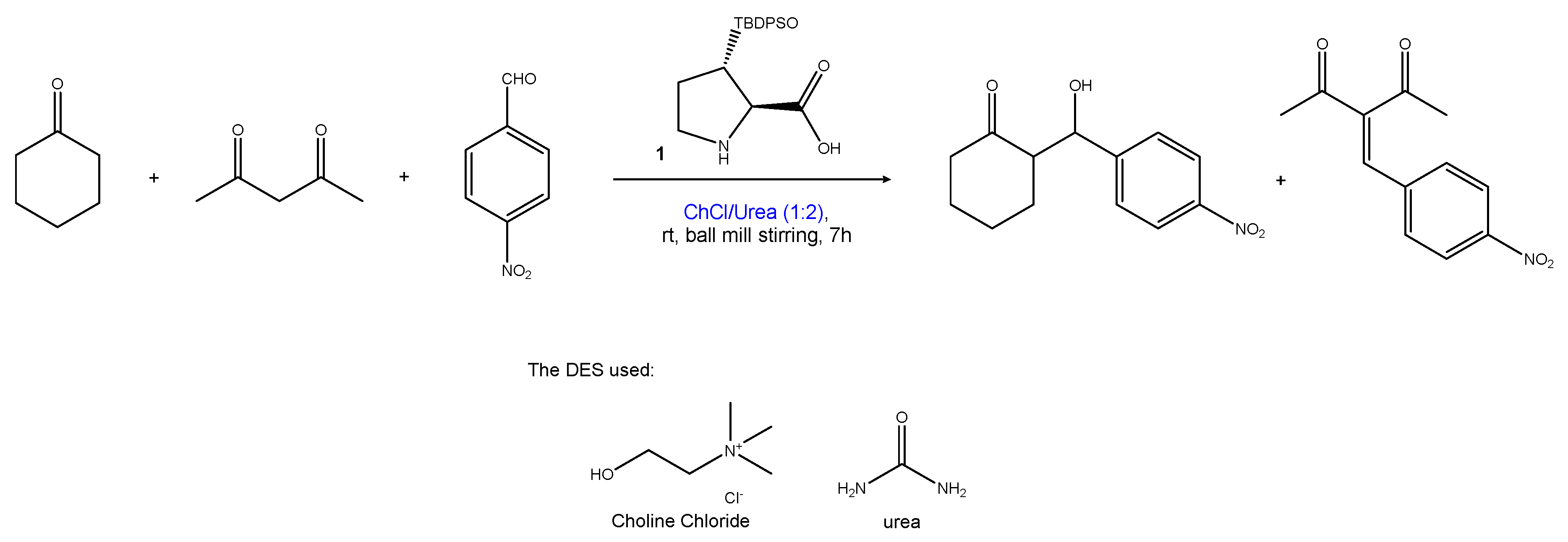
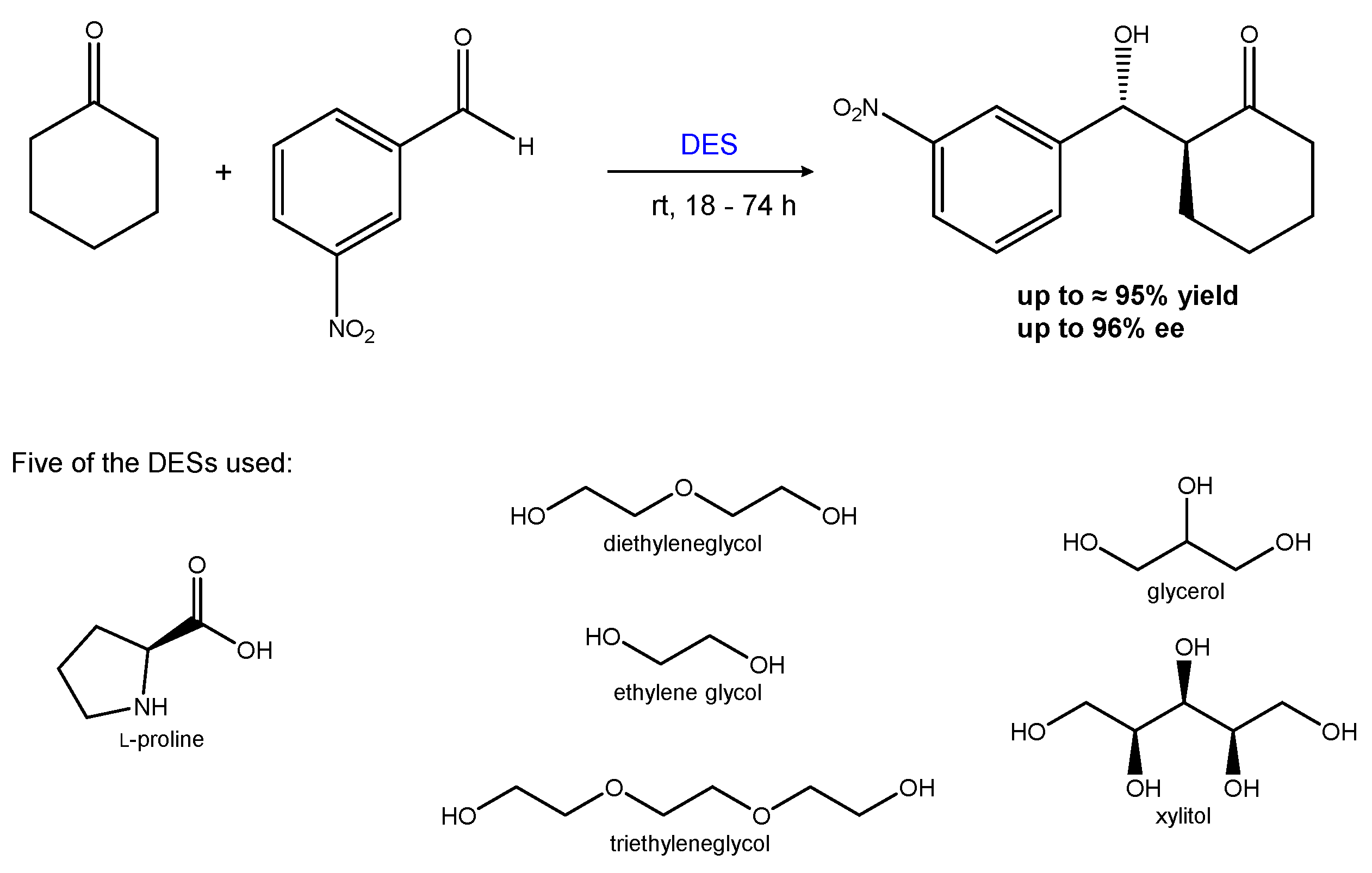
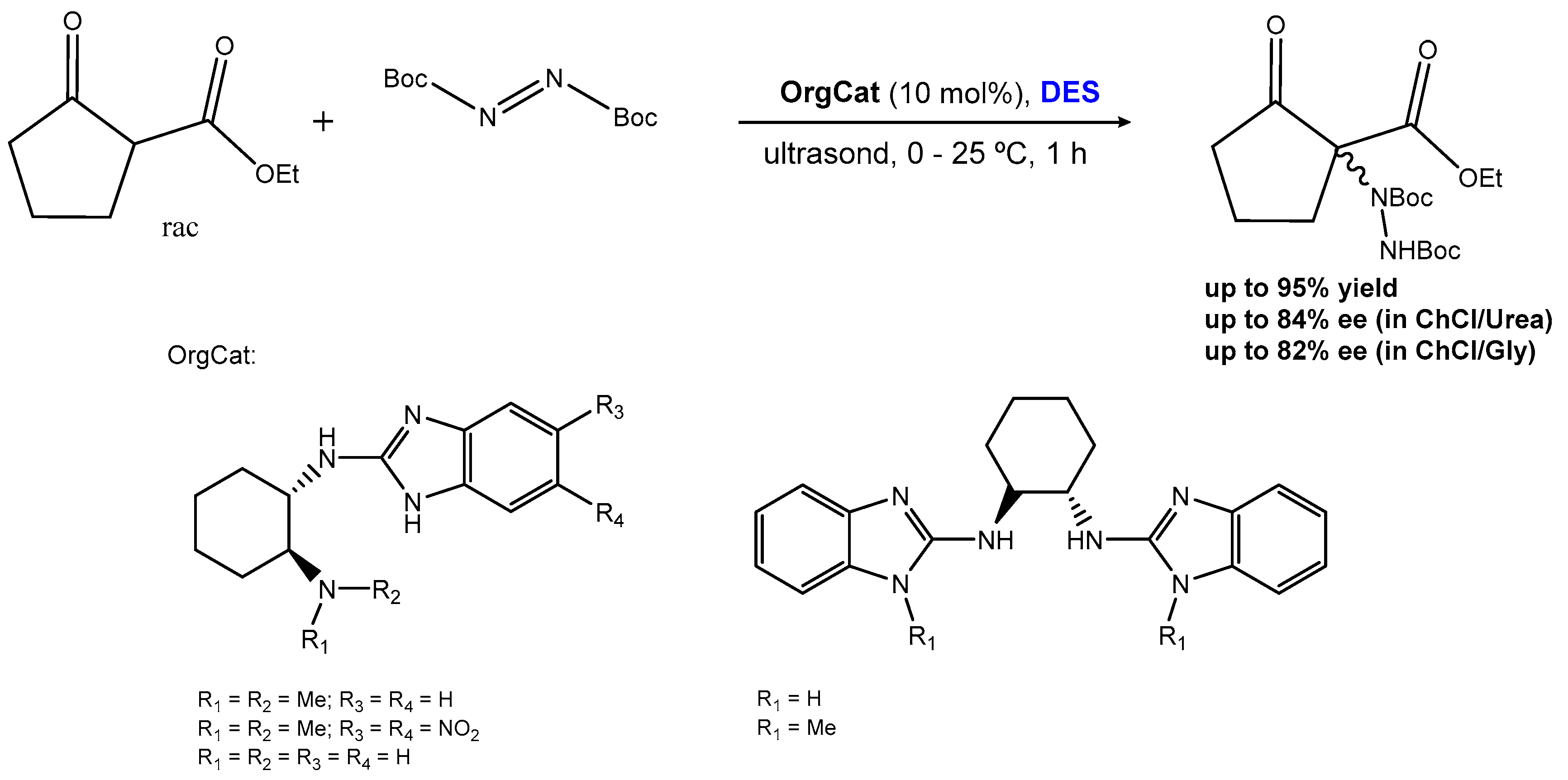
| Entry | Hydrogen-Bond Donor | Hydrogen-Bond Acceptor | Reaction Type | Reference |
|---|---|---|---|---|
| 1 | Glycerol | Choline Chloride | Cross-Aldol Reaction |
[11] |
| 2 | Fructose | Choline Chloride | Michael Reaction |
[12] |
| 3 | Urea | Choline Chloride | Aldol Reaction | [13] |
| 4 | D-Glucose | (D/L)-malic acid | Aldol Reaction | [14] |
| 5 | Ethylene glycol | ChCl | Aldol Reaction | [15] |
| 6 | Glycine | Ph3MePBr | Michael Reaction |
[16] |
| 7 | Glycine | ChCl |
Michael Reaction |
[17] |
| 8 | Glycerol | ChCl | α-amination | [18] |
| 9 | (+)-camphor-sulfonic acid | (S) or (R)-N,N,N-trimethyl-(1-phenylethyl)ammonium methanesulfonate | Michael-type Friedel-Crafts reaction | [19] |
| 10 | L-proline | Glycolic acid | Michael Reaction |
[20] |
| 11 | Ethylene Glycol | ChCl | Conjugate additions |
[21] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




