Submitted:
26 February 2026
Posted:
16 March 2026
You are already at the latest version
Abstract
Introduction Overactive bladder (OAB) frequently co-occurs with cardiovascular-kidney-metabolic (CKM) syndrome; however, the complex interplay of systemic inflammation, psychological distress, and metabolic dysregulation driving this connection remains poorly defined. This study aimed to elucidate these multidimensional associations and identify shared metabolic patterns between OAB and CKM-related conditions. Methods We analyzed data from 11,836 participants in the National Health and Nutrition Examination Survey (2005–2018). CKM stages were classified using American Heart Association criteria, while OAB severity, systemic inflammation, and depression were assessed via the Overactive Bladder Symptom Score, neutrophil-to-high-density lipoprotein cholesterol ratio (NHR), and Patient Health Questionnaire-9, respectively. We utilized survey-weighted multivariable regression and mediation analysis. Furthermore, two-sample Mendelian randomization (MR) analyses using genome-wide association study datasets were conducted to identify causal metabolites. Results Higher CKM stages were significantly associated with increased OAB severity. Elevated NHR and depression scores were independently linked to OAB. Notably, a significant synergistic interaction was observed: moderate inflammation amplified the impact of depressive symptoms on OAB. Mediation analyses demonstrated that NHR, depression, and their interaction significantly mediated the relationship between CKM stage and OAB. MR analysis identified specific causal lipid, amino acid, and energy-related metabolites for OAB, exhibiting substantial overlap with CKM metabolic signatures. Discussion & Conclusion CKM progression, systemic inflammation, and depression are robustly associated with OAB, linked through neuro-inflammatory and psychological pathways. OAB appears to be a manifestation of systemic dysregulation shared with CKM syndrome, necessitating integrated management strategies addressing cardiometabolic health and psychological well-being.
Keywords:
1. Introduction
2. Materials and Methods
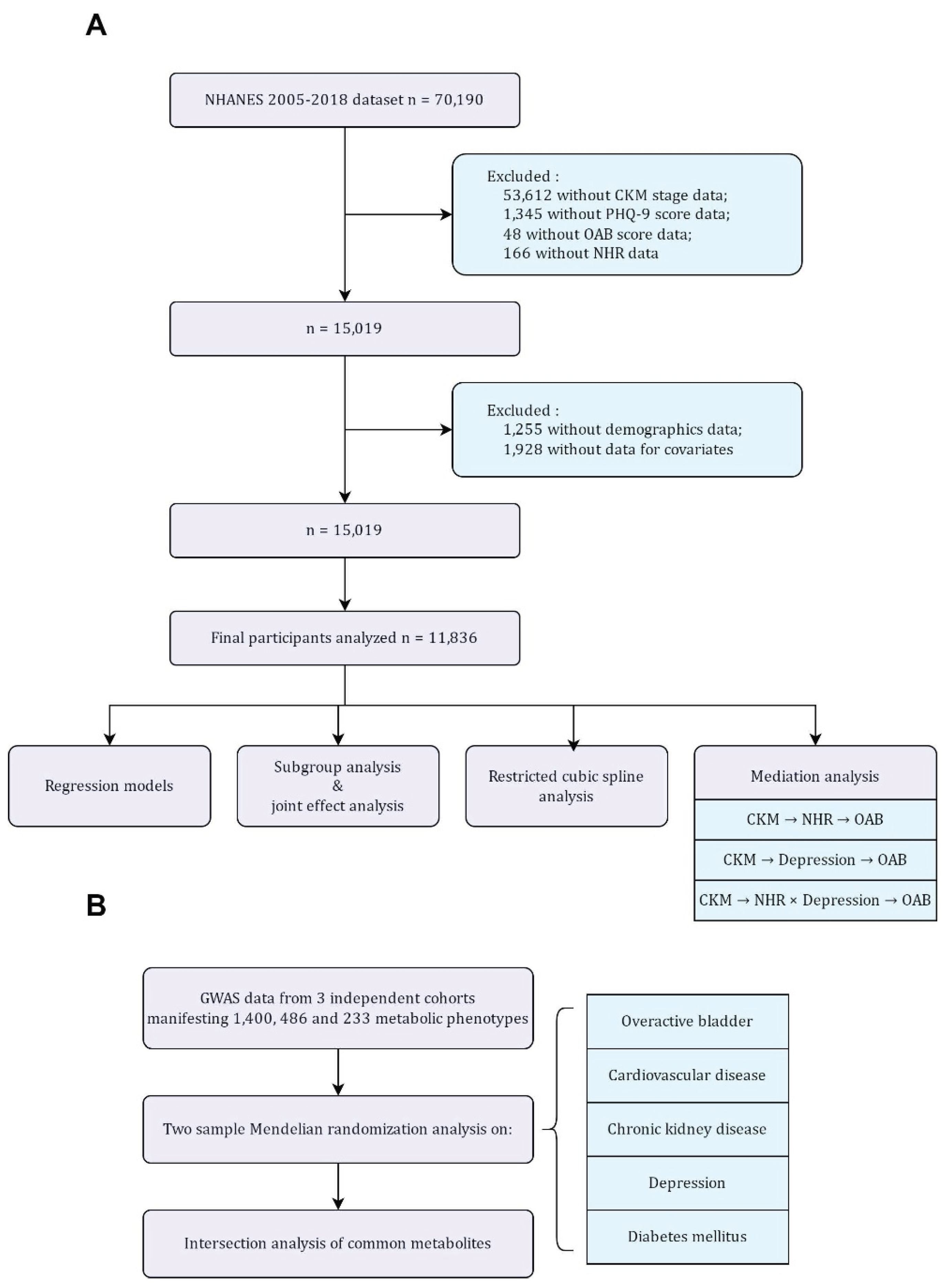
2.1. Study Design and Population
2.2. Definition and Measurement
2.2.1. CKM Stage
2.2.2. NHR, Inflammation Indicator
2.2.3. PHQ-9 Score and Depression Grade
2.2.4. OAB Score
2.2.5. Covariates
2.2.6. GWAS Data for Metabolomic Features
2.2.7. GWAS Data for Disease Outcomes
2.3. Data Analysis
3. Results
3.1. Baseline Characteristics
3.2. Associations of CKM Stage, Inflammation, and Depression with OAB Score
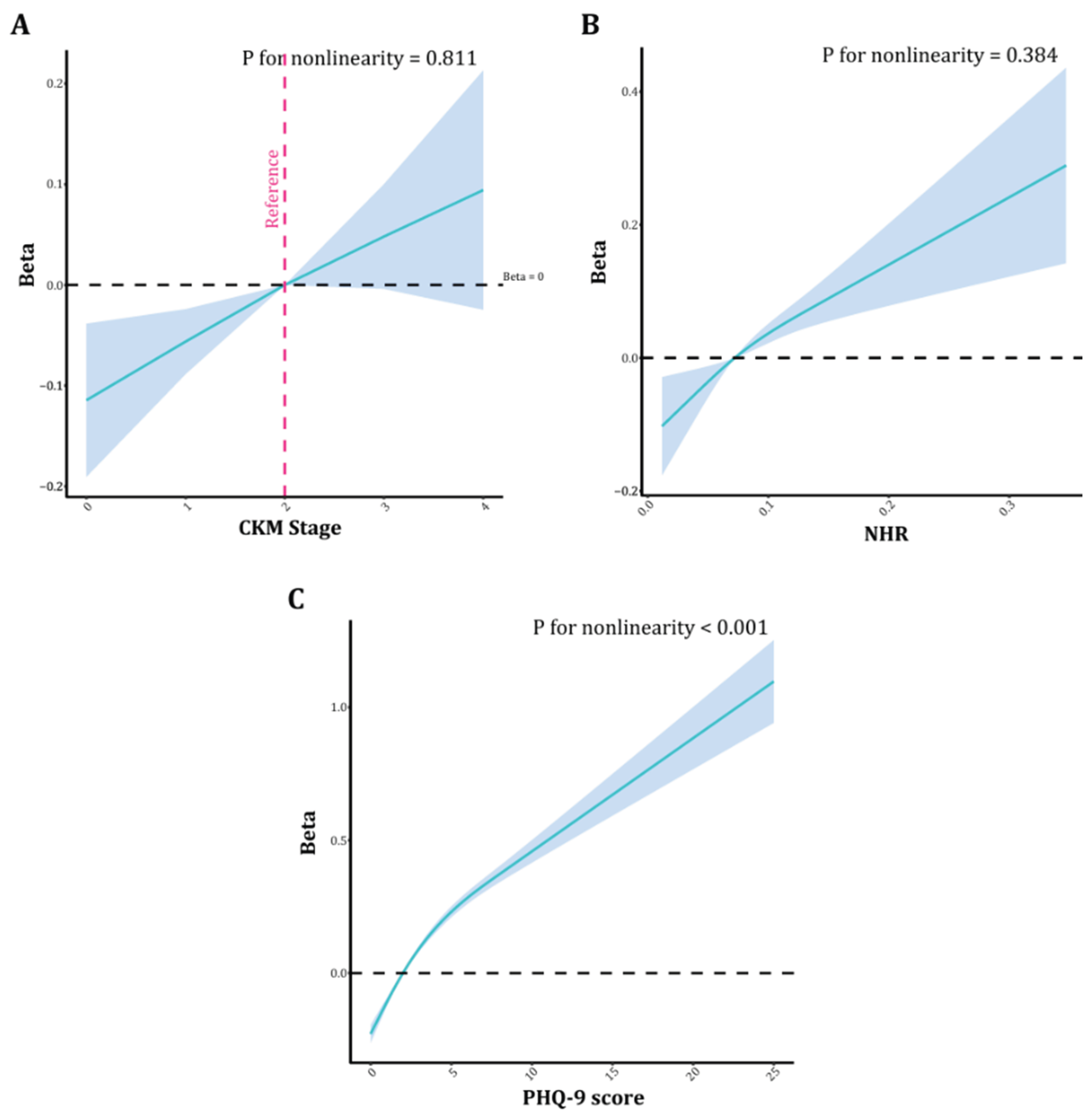
3.3. RCS Analysis
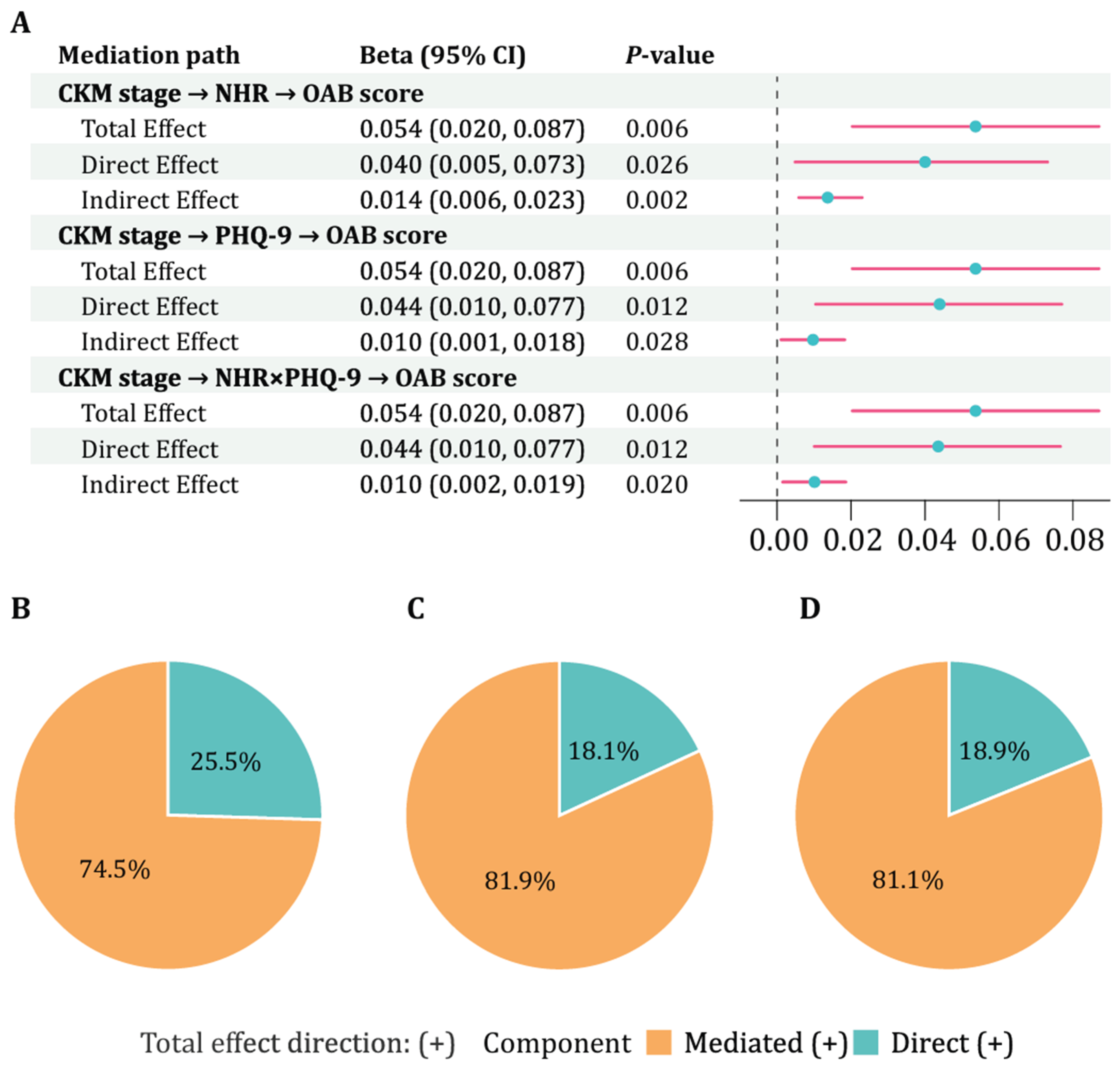
3.4. Mediation Analysis
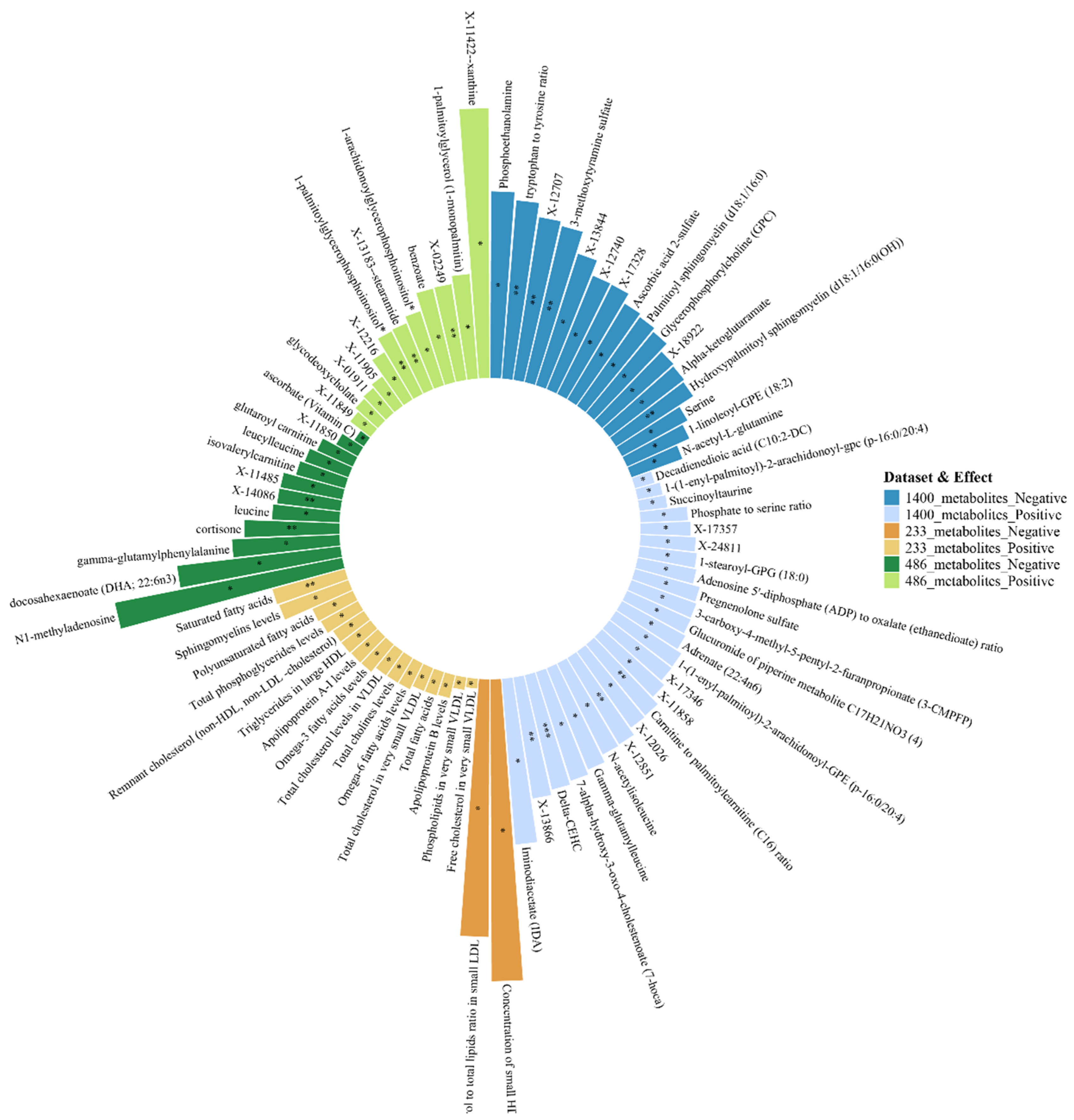
3.5. Metabolomic Signatures Associated with OAB
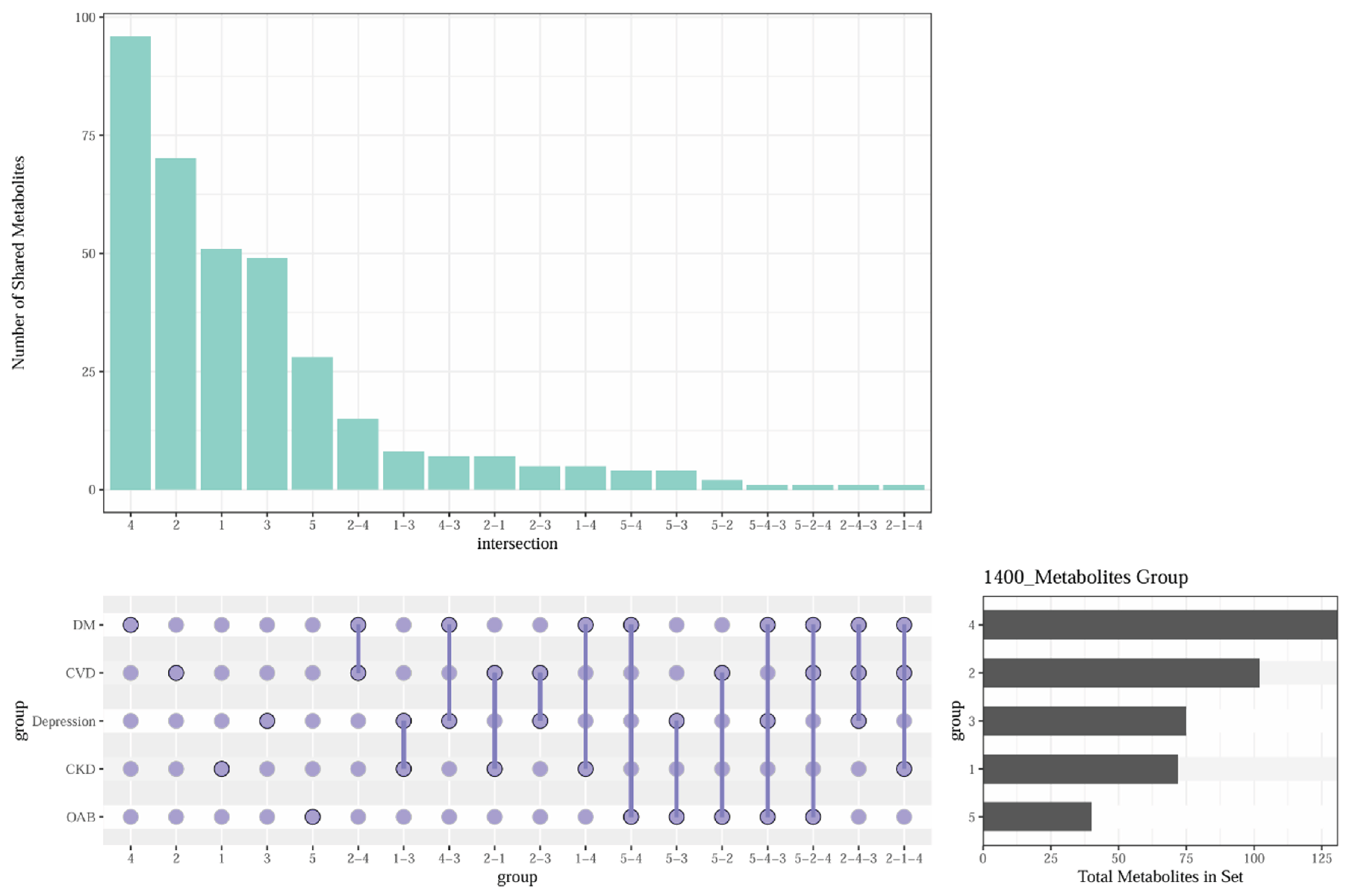
3.6. Shared Metabolic Patterns Across Multiple Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Ethical approval
Informed consent
Data Availability Statement
Acknowledgments
Declaration of generative AI and AI-assisted technologies in the writing process
Competing Interests
Conflicts of Interest
Disclosure Statement
Abbreviations
| CKM | Cardiovascular-kidney-metabolic syndrome |
| OAB | Overactive bladder |
| CKD | Chronic kidney disease |
| PHQ-9 | Patient health questionnaire-9 |
| NHR | Neutrophil to high-density lipoprotein cholesterol ratio |
| NHANES | National Health and Nutrition Examination Survey |
| NCHS | National Center for Health Statistics |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology |
| AHA | American Heart Association |
| CVD | Clinical/subclinical cardiovascular disease |
| KIDGO | Kidney Disease Improving Global Outcomes |
| eGFR | Estimated glomerular filtration rate |
| PREVENT | Predicting Risk of CVD EVENTs |
| UUI | Urge urinary incontinence |
| OABSS | Overactive bladder symptom score |
| PIR | Poverty-to-income ratio |
| CDC | Centers for Disease Control and Prevention |
| SD | Standard deviation |
| RCS | Restricted cubic spline |
| CI | Confidence interval |
| BDNF | Brain-derived neurotrophic factor |
References
- Nambiar, AK; Arlandis, S; Bø, K; et al. European Association of Urology Guidelines on the Diagnosis and Management of Female Non-neurogenic Lower Urinary Tract Symptoms. Part 1: Diagnostics, Overactive Bladder, Stress Urinary Incontinence, and Mixed Urinary Incontinence. Eur Urol. 2022, 82, 49–59. [Google Scholar] [PubMed]
- Tang, F; Zhang, J; Huang, R; et al. The association between wet overactive bladder and consumption of tea, coffee, and caffeine: Results from 2005–2018 National Health and Nutrition Examination Survey. Clin Nutr. 2024, 43, 1261–1269. [Google Scholar] [CrossRef]
- Huang, J; Fan, Y; Zhao, K; et al. Comparative Efficacy of Neuromodulation Technologies for Overactive Bladder in Adults: A Network Meta-Analysis of Randomized Controlled Trials. Neuromodulation Technol Neural Interface 2023, 26, 1535–1548. [Google Scholar] [CrossRef] [PubMed]
- Ndumele, CE; Rangaswami, J; Chow, SL; et al. Cardiovascular-kidney-metabolic health: A presidential advisory from the American heart association. Circulation 2023, 148, 1606–1635. [Google Scholar] [CrossRef]
- Malhi, GS; Mann, JJ. Depression. The Lancet 2018, 392, 2299–2312. [Google Scholar] [CrossRef]
- Huang, AA; Huang, SY. Increased vigorous exercise and decreased sedentary activities are associated with decreased depressive symptoms in United States adults: Analysis of The National Health and Nutrition Examination Survey (NHANES) 2017–2020. Health Sci Rep. 2023, 6, e1473. [Google Scholar]
- Boothroyd, L; Dagnan, D; Muncer, S. PHQ-9: One factor or two? Psychiatry Res. 2019, 271, 532–534. [Google Scholar] [CrossRef]
- Wittkampf, KA; Naeije, L; Schene, AH; Huyser, J; Van Weert, HC. Diagnostic accuracy of the mood module of the Patient Health Questionnaire: a systematic review. Gen Hosp Psychiatry 2007, 29, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Beard, C; Hsu, KJ; Rifkin, LS; Busch, AB; Björgvinsson, T. Validation of the PHQ-9 in a psychiatric sample. J Affect Disord. 2016, 193, 267–273. [Google Scholar] [CrossRef]
- Lee, KS; Yoo, TK; Liao, L; et al. Association of lower urinary tract symptoms and OAB severity with quality of life and mental health in China, Taiwan and South Korea: results from a cross-sectional, population-based study. BMC Urol. 2017, 17, 108. [Google Scholar] [CrossRef]
- Zhang, Y; Wu, X; Liu, G; Feng, X; Jiang, H; Zhang, X. Association between overactive bladder and depression in American adults: A cross-sectional study from NHANES 2005–2018. J Affect Disord. 2024, 356, 545–553. [Google Scholar]
- Li, M; Xu, M; Zhou, H; Pan, Y; Liu, X. The association between different levels of depression and overactive bladder: A cross-sectional study of the 2005–2018 National Health and Nutrition Examination Survey. Neurourol Urodyn. 2024, 43, 1826–1834. [Google Scholar]
- Qing, G; Bao, C; Yang, Y; Wei, B. Association between neutrophil to high-density lipoprotein cholesterol ratio (NHR) and depression symptoms among the United States adults: a cross-sectional study. Lipids Health Dis. 2024, 23, 215. [Google Scholar]
- Qiu, M; Zhang, C; Zhang, H; et al. Retrospective evaluation of novel serum inflammatory biomarkers in first-episode psychiatric disorders: diagnostic potential and immune dysregulation. Front Psychiatry 2024, 15, 1442954. [Google Scholar] [PubMed]
- Huang, X; Liang, J; Zhang, J; Fu, J; Xie, W; Zheng, F. Association of cardiovascular-kidney-metabolic health and social connection with the risk of depression and anxiety. Psychol Med. 2024, 54, 4203–4211. [Google Scholar]
- Zipf G, Chiappa M, Porter KS, Ostchega Y, Lewis BG, Dostal J. National health and nutrition examination survey: Plan and operations, 1999-2010 (vital and health statistics series 1, no. 56). Natl Cent Health Stat Httpswww Cdc Govnchsdataseriessr01sr01056 Pdf. Published online 2013.
- Von Elm, E; Altman, DG; Egger, M; Pocock, SJ; Gøtzsche, PC; Vandenbroucke, JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. The Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- Ndumele, CE; Rangaswami, J; Chow, SL; et al. Cardiovascular-Kidney-Metabolic Health: A Presidential Advisory From the American Heart Association. Circulation 2023, 148, 1606–1635. [Google Scholar]
- Khan, SS; Matsushita, K; Sang, Y; et al. Development and Validation of the American Heart Association’s PREVENT Equations. Circulation 2024, 149, 430–449. [Google Scholar]
- Kroenke, K; Spitzer, RL; Williams, JBW. The PHQ-9: Validity of a brief depression severity measure. J Gen Intern Med. 2001, 16, 606–613. [Google Scholar] [PubMed]
- Homma, Y; Yoshida, M; Seki, N; et al. Symptom assessment tool for overactive bladder syndrome—overactive bladder symptom score. Urology 2006, 68, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Blaivas, JG; Panagopoulos, G; Weiss, JP; Somaroo, C. Validation of the Overactive Bladder Symptom Score. J Urol. 2007, 178, 543–547. [Google Scholar] [PubMed]
- Li, J; Wei, X. Association of cardiovascular-kidney-metabolic syndrome with all-cause and cardiovascular mortality: A prospective cohort study. Am J Prev Cardiol. 2025, 22, 100985. [Google Scholar]
- Mahemuti, N; Zou, J; Liu, C; Xiao, Z; Liang, F; Yang, X. Urinary Albumin-to-Creatinine Ratio in Normal Range, Cardiovascular Health, and All-Cause Mortality. JAMA Netw Open. 2023, 6, e2348333. [Google Scholar]
- Chen, Y; Lu, T; Pettersson-Kymmer, U; et al. Genomic atlas of the plasma metabolome prioritizes metabolites implicated in human diseases. Nat Genet. 2023, 55, 44–53. [Google Scholar]
- Shin, SY; Fauman, EB; Petersen, AK; et al. An atlas of genetic influences on human blood metabolites. Nat Genet. 2014, 46, 543–550. [Google Scholar] [CrossRef]
- Karjalainen, MK; Karthikeyan, S; Oliver-Williams, C; et al. Genome-wide characterization of circulating metabolic biomarkers. Nature 2024, 628, 130–138. [Google Scholar] [CrossRef]
- Zhang, X; Ma, L; Li, J; Zhang, W; Xie, Y; Wang, Y. Mental health and lower urinary tract symptoms: Results from the NHANES and mendelian randomization study. J Psychosom Res. 2024, 178, 111599. [Google Scholar]
- Kurki, MI; Karjalainen, J; Palta, P; et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature 2023, 613, 508–518. [Google Scholar]
- Johnson, CL; Paulose-Ram, R; Ogden, CL; et al. National Health and Nutrition Examination Survey: Analytic guidelines, 1999–2010. National Center for Health Statistics. Vital Health Stat 2013, 2. [Google Scholar]
- Coelho, A; Wolf-Johnston, AS; Shinde, S; et al. Urinary bladder inflammation induces changes in urothelial nerve growth factor and TRPV 1 channels. Br J Pharmacol. 2015, 172, 1691–1699. [Google Scholar] [PubMed]
- Verma, V; Gupta, S; Kumar, P; et al. Involvement of NLRP3 and NLRC4 Inflammasome in Uropathogenic E. coli Mediated Urinary Tract Infections. Front Microbiol. 2019, 10, 2020. [Google Scholar]
- Song, QX; Chermansky, CJ; Birder, LA; Li, L; Damaser, MS. Brain-derived neurotrophic factor in urinary continence and incontinence. Nat Rev Urol. 2014, 11, 579–588. [Google Scholar]
- Kashyap, MP; Pore, SK; De Groat, WC; Chermansky, CJ; Yoshimura, N; Tyagi, P. BDNF overexpression in the bladder induces neuronal changes to mediate bladder overactivity. Am J Physiol-Ren Physiol. 2018, 315, F45–F56. [Google Scholar]
- Antunes-Lopes, T; Cruz, F. Urinary Biomarkers in Overactive Bladder: Revisiting the Evidence in 2019. Eur Urol Focus. 2019, 5, 329–336. [Google Scholar] [CrossRef]
- Stemberger Maric, L; Kozmar, A; Lenicek Krleza, J; Rogic, D; Colic, M; Abdovic, S. Urinary brain-derived neurotrophic factor and nerve growth factor as noninvasive biomarkers of overactive bladder in children. Biochem Medica 2022, 32, 405–413. [Google Scholar]
- Zarza-Rebollo, JA; López-Isac, E; Rivera, M; Gómez-Hernández, L; Pérez-Gutiérrez, AM; Molina, E. The relationship between BDNF and physical activity on depression. Prog Neuropsychopharmacol Biol Psychiatry 2024, 134, 111033. [Google Scholar]
- Phillips, C. Brain-Derived Neurotrophic Factor, Depression, and Physical Activity: Making the Neuroplastic Connection. Neural Plast. 2017, 2017, 1–17. [Google Scholar] [CrossRef]
- Carniel, BP; Da Rocha, NS. Brain-derived neurotrophic factor (BDNF) and inflammatory markers: Perspectives for the management of depression. Prog Neuropsychopharmacol Biol Psychiatry 2021, 108, 110151. [Google Scholar]
- Hassamal, S. Chronic stress, neuroinflammation, and depression: an overview of pathophysiological mechanisms and emerging anti-inflammatories. Front Psychiatry 2023, 14. [Google Scholar]
- Kittelson, KS; Junior, AG; Fillmore, N; Da Silva Gomes, R. Cardiovascular-kidney-metabolic syndrome – An integrative review. Prog Cardiovasc Dis. 2024, 87, 26–36. [Google Scholar] [CrossRef] [PubMed]
- He, H; Luo, H; Qian, B; et al. Autonomic Nervous System Dysfunction Is Related to Chronic Prostatitis/Chronic Pelvic Pain Syndrome. World J Mens Health 2024, 42, 1. [Google Scholar] [CrossRef]
- Preacher, KJ; Rucker, DD; Hayes, AF. Addressing Moderated Mediation Hypotheses: Theory, Methods, and Prescriptions. Multivar Behav Res. 2007, 42, 185–227. [Google Scholar] [CrossRef]
- Jiao, W; Lin, J; Deng, Y; et al. The immunological perspective of major depressive disorder: unveiling the interactions between central and peripheral immune mechanisms. J Neuroinflammation 2025, 22. [Google Scholar] [CrossRef] [PubMed]
- Li, J; He, M; Zhou, Y. Association between cardiometabolic index and overactive bladder in adult American women: A cross-sectional study. PLOS ONE 2025, 20, e0314594. [Google Scholar] [CrossRef]
- Alemi, F; Kwon, E; Poole, DP; et al. The TGR5 receptor mediates bile acid–induced itch and analgesia. J Clin Invest. 2013, 123, 1513–1530. [Google Scholar]
- Shimura, H; Mitsui, T; Kira, S; et al. Metabolomic analysis of overactive bladder in male patients: Identification of potential metabolite biomarkers. Urology 2018, 118, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Hsu, LN; Hu, JC; Chen, PY; Lee, WC; Chuang, YC. Metabolic syndrome and overactive bladder syndrome may share common pathophysiologies. Biomedicines 2022, 10, 1957. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | CKM Stage | P-valueb | |||||
|
Overall N = 11,836a |
0 N = 983a |
1 N = 2,209a |
2 N = 4,885a |
3 N = 2,526a |
4 N = 1,233a |
||
| Age | 47.98±(16.84) | 35.98±(13.38) | 41.46±(14.40) | 54.15±(15.28) | 42.26±(15.00) | 65.14±(12.37) | <0.001 |
| Gender | <0.001 | ||||||
| Male | 5,686(48.22%) | 298(31.02%) | 763(36.81%) | 1,732(34.30%) | 2,189(88.16%) | 704(52.48%) | |
| Female | 6,150(51.78%) | 685(68.98%) | 1,446(63.19%) | 3,153(65.70%) | 337(11.84%) | 529(47.52%) | |
| Race | <0.001 | ||||||
| Mexican American | 1,766(7.994%) | 110(5.296%) | 391(9.530%) | 710(6.885%) | 464(10.95%) | 91(4.430%) | |
| Non-Hispanic White | 5,542(69.68%) | 490(72.34%) | 943(67.03%) | 2,230(70.77%) | 1,165(66.92%) | 714(75.55%) | |
| Non-Hispanic Black | 2,352(9.999%) | 131(7.753%) | 470(11.84%) | 1,090(10.71%) | 378(7.302%) | 283(12.07%) | |
| Other | 2,176(12.33%) | 252(14.61%) | 405(11.60%) | 855(11.64%) | 519(14.83%) | 145(7.949%) | |
| Education | <0.001 | ||||||
| Below high school | 2,608(14.61%) | 107(8.222%) | 415(12.02%) | 1,134(14.95%) | 592(15.85%) | 360(23.59%) | |
| High School graduate |
2,708(23.35%) | 180(19.25%) | 443(21.00%) | 1,146(24.80%) | 612(23.58%) | 327(26.66%) | |
| Some college or AA degree |
3,607(31.53%) | 300(27.83%) | 702(31.33%) | 1,508(32.76%) | 747(31.90%) | 350(29.62%) | |
| College graduate or above |
2,913(30.51%) | 396(44.69%) | 649(35.64%) | 1,097(27.49%) | 575(28.67%) | 196(20.14%) | |
| Marital status | <0.001 | ||||||
| Married | 6,328(55.61%) | 461(46.55%) | 1,128(53.97%) | 2,633(58.34%) | 1,440(55.45%) | 666(58.06%) | |
| Single | 4,545(36.03%) | 427(41.32%) | 852(36.56%) | 1,962(36.07%) | 790(33.07%) | 514(36.43%) | |
| Living with a partner |
963(8.361%) | 95(12.13%) | 229(9.468%) | 290(5.594%) | 296(11.49%) | 53(5.505%) | |
| Poverty status | 0.008 | ||||||
| Poor | 2,284(13.65%) | 164(11.80%) | 447(13.47%) | 911(12.34%) | 502(15.58%) | 260(17.03%) | |
| Not poor | 9,552(86.35%) | 819(88.20%) | 1,762(86.53%) | 3,974(87.66%) | 2,024(84.42%) | 973(82.97%) | |
| eGFR | 94.00±(21.74) | 104.46±(17.81) | 101.73±(18.08) | 89.87±(20.03) | 97.30±(21.41) | 72.82±(23.47) | <0.001 |
| Smoking | 2,282(19.78%) | 176(20.67%) | 372(16.25%) | 876(17.90%) | 597(24.60%) | 261(23.14%) | <0.001 |
| Alcohol | 9.03±(20.94) | 11.33±(22.19) | 8.21±(19.54) | 8.52±(20.57) | 10.67±(23.02) | 6.27±(17.79) | <0.001 |
| Hypertension | 4,451(33.98%) | 0(0%) | 0(0%) | 2,586(51.62%) | 927(34.36%) | 938(75.57%) | <0.001 |
| CVD | 927(6.364%) | 0(0%) | 0(0%) | 0(0%) | 0(0%) | 927(75.78%) | <0.001 |
| Stroke | 457(2.986%) | 0(0%) | 0(0%) | 0(0%) | 0(0%) | 457(35.56%) | <0.001 |
| Diabetes | 1,855(11.63%) | 25(2.071%) | 100(4.196%) | 803(12.58%) | 462(13.53%) | 465(31.60%) | <0.001 |
| NHR | 0.08±(0.05) | 0.06±(0.03) | 0.07±(0.03) | 0.08±(0.05) | 0.11±(0.05) | 0.10±(0.05) | <0.001 |
| PHQ-9 score | 2.99±(4.02) | 2.52±(3.45) | 2.80±(3.80) | 3.04±(4.06) | 2.87±(3.91) | 4.15±(4.98) | <0.001 |
| Depression grade | <0.001 | ||||||
| None | 9,029(77.22%) | 800(81.41%) | 1,746(79.38%) | 3,687(76.59%) | 1,987(78.09%) | 809(67.62%) | |
| Mild | 1,845(15.25%) | 128(14.08%) | 329(14.10%) | 791(15.49%) | 360(15.17%) | 237(18.56%) | |
| Moderate | 610(4.897%) | 42(3.159%) | 83(3.994%) | 261(5.270%) | 114(4.395%) | 110(8.789%) | |
| Moderate to severe | 248(1.864%) | 10(0.861%) | 40(2.083%) | 108(1.946%) | 45(1.707%) | 45(2.536%) | |
| Severe | 104(0.767%) | 3(0.494%) | 11(0.440%) | 38(0.707%) | 20(0.643%) | 32(2.497%) | |
| OAB score | 1.30 (1.23) | 0.85 (0.95) | 1.08 (1.08) | 1.49 (1.24) | 1.05 (1.11) | 2.14 (1.48) | <0.001 |
| OAB group | <0.001 | ||||||
| Non-overactive bladder | 9,383(83.57%) | 919(94.02%) | 1,927(88.96%) | 3,657(79.99%) | 2,161(88.40%) | 719(61.46%) | |
| Overactive bladder | 2,453(16.43%) | 64(5.976%) | 282(11.04%) | 1,228(20.01%) | 365(11.60%) | 514(38.54%) | |
| Characteristic | Model 1 | Model 2 | Model 3 | |||
| Beta(95% CI) | P-value | Beta(95% CI) | P-value | Beta(95% CI) | P-value | |
| CKM stage | ||||||
| 0 | Reference | Reference | Reference | |||
| 1 | 0.230 (0.134, 0.326) | <0.001 | 0.095 (0.002, 0.189) | 0.048 | 0.105 (0.012, 0.198) | 0.029 |
| 2 | 0.639 (0.551, 0.728) | <0.001 | 0.201 (0.111, 0.292) | <0.001 | 0.131 (0.034, 0.228) | 0.010 |
| 3 | 0.200 (0.100, 0.299) | <0.001 | 0.264 (0.154, 0.374) | <0.001 | 0.175 (0.064, 0.286) | 0.003 |
| 4 | 1.281 (1.150, 1.411) | <0.001 | 0.635 (0.492, 0.777) | <0.001 | 0.401 (-0.057, 0.858) | 0.089 |
| P for trend | 0.186 (0.157, 0.216) | <0.001 | 0.127 (0.095, 0.159) | <0.001 | 0.054 (0.019, 0.088) | 0.003 |
| NHR | 1.317 (0.655, 1.979) | <0.001 | 1.992 (1.195, 2.789) | <0.001 | 1.192 (0.577, 1.806) | <0.001 |
| PHQ-9 score | 0.077 (0.068, 0.085) | <0.001 | 0.067 (0.059, 0.076) | <0.001 | 0.062 (0.054, 0.071) | <0.001 |
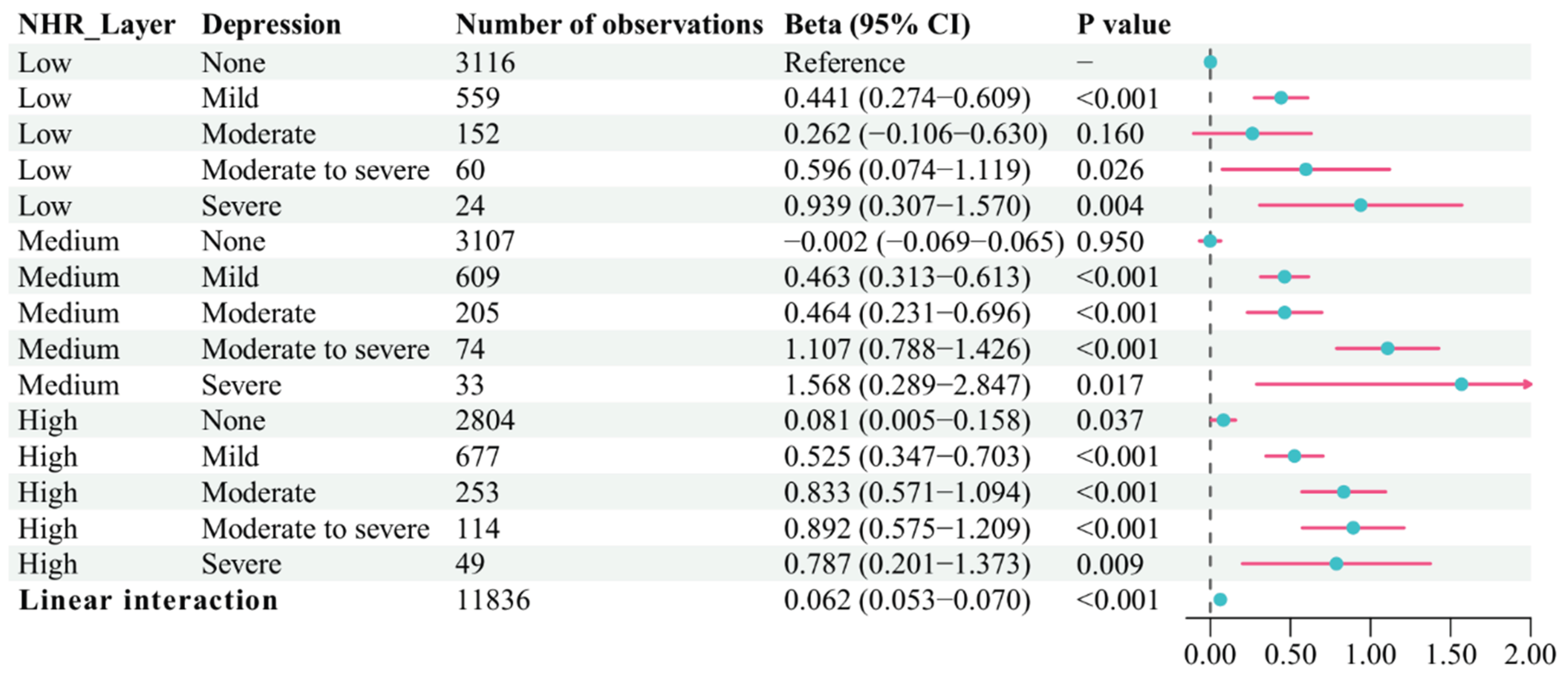
| NHR layer | Depression grades |
Number of observations |
Beta (95% CI) | P-valuea |
| Low | None | 3116 | Reference | — |
| Low | Mild | 559 | 0.458 (0.297, 0.619) | <0.001 |
| Low | Moderate | 152 | 0.280 (-0.073, 0.633) | 0.118 |
| Low | Moderate to severe | 60 | 0.622 (0.097, 1.147) | 0.021 |
| Low | Severe | 24 | 0.966 (0.328, 1.604) | 0.003 |
| Medium | None | 3107 | Reference | — |
| Medium | Mild | 609 | 0.470 (0.315, 0.624) | <0.001 |
| Medium | Moderate | 205 | 0.473 (0.252, 0.693) | <0.001 |
| Medium | Moderate to severe | 74 | 1.101 (0.794, 1.407) | <0.001 |
| Medium | Severe | 33 | 1.616 (0.377, 2.856) | 0.011 |
| High | None | 2804 | Reference | — |
| High | Mild | 677 | 0.428 (0.269, 0.587) | <0.001 |
| High | Moderate | 253 | 0.722 (0.469, 0.975) | <0.001 |
| High | Moderate to severe | 114 | 0.814 (0.507, 1.122) | <0.001 |
| High | Severe | 49 | 0.699 (0.103, 1.294) | 0.022 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




