Submitted:
12 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Biofilm Adaptation Across Various Host and Environmental Niches
2.1. Gastrointestinal Tract—Primary Infection Site
2.2. Gallbladder and Bile-Rich Environments—Chronically Persistence Niche
2.3. Food Production Environment (Poultry, Cattle, and Swine)—Transmission Niche
2.4. Clinical and Environmental Reservoirs—Secondary Reservoir Niche
3. Serovar Diversity in Biofilm Formation and AMR
3.1. Classical Phenotype Comparisons
3.2. Link to Antimicrobial Resistance
4. Genomic and Pangenomic Basis of Biofilm Traits
4.1. Core vs. Accessory Biofilm Genes
4.2. Mobile Genetic Elements (MGEs) as Genomic “Bridges” Between Biofilm Ecology and AMR
4.3. Comparative Genomics and Pangenome Analyses
5. Transcriptomic, Proteomic and Metabolomic Signatures of Biofilm in Different Serovars and Hosts
5.1. In Vitro Biofilm vs. Planktonic Omics
5.2. Serovar-Specific Omics Under Host-Mimicking Conditions
5.3. Host-Specific Omics and Salmonella Biofilm Responses
5.4. Host-Specific Omics and Salmonella Biofilm Responses
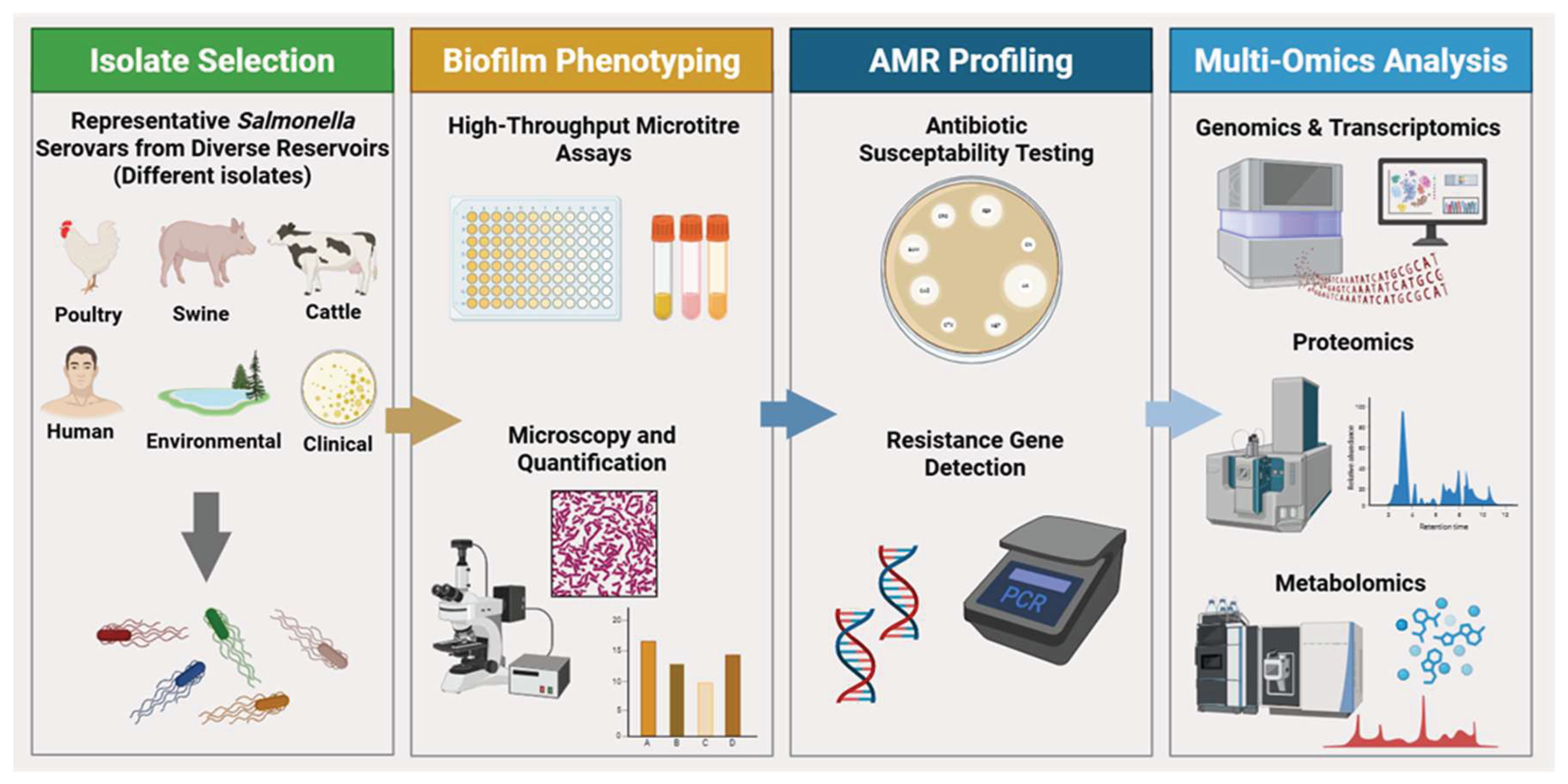
6. Methodological and Analytical Framework for Comparative Biofilmomics
6.1. Ideal Experimental Framework
6.2. Integrated Bioinformatics and Machine Learning
7. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMR | Antimicrobial resistance |
| MDR | Multidrug resistance |
| ESBL | Extended-spectrum β-lactamase |
| EPS | Extracellular polymeric substance |
| ST | Sequence type |
| RDAR | Red, dry, and rough |
| BDAR | Brown, dry, and rough |
| PDAR | Pink, dry, and rough |
| MGEs | Mobile genetic elements |
| LC-MS | Liquid chromatography and mass-spectrometry |
| ML | Machine learning |
References
- World Health Organization Salmonella (Non-Typhoidal). Available online: https://www.who.int/news-room/fact-sheets/detail/salmonella-%28non-typhoidal%29 (accessed on 28 February 2026).
- World Health Organization Food Safety. Available online: https://www.who.int/news-room/fact-sheets/detail/food-safety (accessed on 28 February 2026).
- Bharat, A.; Mataseje, L.; Parmley, E.J.; Avery, B.; Cox, G.; Carson, C.; Irwin, R.; Deckert, A.; Daignault, D.; Alexander, D.; et al. One Health Genomic Analysis of Extended-Spectrum β-Lactamase‒Producing. Emerging Infectious Disease journal 2022, 28, 1410. [Google Scholar] [CrossRef]
- Harrell, J.E.; Hahn, M.M.; D’Souza, S.J.; Vasicek, E.M.; Sandala, J.L.; Gunn, J.S.; McLachlan, J.B. Salmonella Biofilm Formation, Chronic Infection, and Immunity Within the Intestine and Hepatobiliary Tract. Front. Cell. Infect. Microbiol. 2021, 10–2020. [Google Scholar] [CrossRef]
- Guo, L.; Dai, H.; Feng, S.; Zhao, Y. Contribution of GalU to Biofilm Formation, Motility, Antibiotic and Serum Resistance, and Pathogenicity of Salmonella Typhimurium. Front. Cell. Infect. Microbiol. 2023, 13–2023. [Google Scholar] [CrossRef]
- Liu, H.Y.; Prentice, E.L.; Webber, M.A. Mechanisms of Antimicrobial Resistance in Biofilms. npj Antimicrobials and Resistance 2024, 2, 27. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, M.; Morris, D.; De Lappe, N.; O’Connor, J.; Lalor, P.; Dockery, P.; Cormican, M. Commonly Used Disinfectants Fail to Eradicate Salmonella Enterica Biofilms from Food Contact Surface Materials. Appl. Environ. Microbiol. 2014, 80, 1507–1514. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, K.D.; Palmer, M.B.; Köster, W.L.; White, A.P. Examining the Link between Biofilm Formation and the Ability of Pathogenic Salmonella Strains to Colonize Multiple Host Species. Front. Vet. Sci. 2017, 4–2017. [Google Scholar] [CrossRef]
- Gal-Mor, O.; Boyle, E.C.; Grassl, G.A. Same Species, Different Diseases: How and Why Typhoidal and Non-Typhoidal Salmonella Enterica Serovars Differ. Front. Microbiol. 2014, 5. [Google Scholar] [CrossRef]
- Thomson, N.R.; Clayton, D.J.; Windhorst, D.; Vernikos, G.; Davidson, S.; Churcher, C.; Quail, M.A.; Stevens, M.; Jones, M.A.; Watson, M.; et al. Comparative Genome Analysis of Salmonella Enteritidis PT4 and Salmonella Gallinarum 287/91 Provides Insights into Evolutionary and Host Adaptation Pathways. Genome Res. 2008, 18, 1624–1637. [Google Scholar] [CrossRef] [PubMed]
- Aviv, G.; Tsyba, K.; Steck, N.; Salmon-Divon, M.; Cornelius, A.; Rahav, G.; Grassl, G.A.; Gal-Mor, O. A Unique Megaplasmid Contributes to Stress Tolerance and Pathogenicity of an Emergent Salmonella Enterica Serovar Infantis Strain. Environ. Microbiol. 2014, 16, 977–994. [Google Scholar] [CrossRef]
- Yuan, L.; Fan, L.; Dai, H.; He, G.; Zheng, X.; Rao, S.; Yang, Z.; Jiao, X. Multi-Omics Reveals the Increased Biofilm Formation of Salmonella Typhimurium M3 by the Induction of Tetracycline at Sub-Inhibitory Concentrations. Science of The Total Environment 2023, 899, 165695. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Zhu, K.; Min, K.; Zhang, Y.; Liu, J.; Liu, K.; Han, Y.; Li, X.; Du, X.; Wang, X.; et al. Characterization of a Salmonella Enterica Serovar Typhimurium Lineage with Rough Colony Morphology and Multidrug Resistance. Nat. Commun. 2024, 15. [Google Scholar] [CrossRef] [PubMed]
- Dutta, B.; Chatterjee, D.; Sarkar, N.; Lahiri, D.; Nag, M.; Ray, R.R. Multi-Omics Technology in Detection of Multispecies Biofilm. The Microbe 2024, 4, 100128. [Google Scholar] [CrossRef]
- Seneviratne, C.J.; Suriyanarayanan, T.; Widyarman, A.S.; Lee, L.S.; Lau, M.; Ching, J.; Delaney, C.; Ramage, G. Multi-Omics Tools for Studying Microbial Biofilms: Current Perspectives and Future Directions. Crit. Rev. Microbiol. 2020, 46, 759–778. [Google Scholar] [CrossRef]
- Khan, C.M.A. The Dynamic Interactions between Salmonella and the Microbiota, within the Challenging Niche of the Gastrointestinal Tract. Int. Sch. Res. Notices 2014, 2014, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Nesse, L.L.; Osland, A.M.; Vestby, L.K. The Role of Biofilms in the Pathogenesis of Animal Bacterial Infections. Microorganisms 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Sivula, C.P.; Bogomolnaya, L.M.; Andrews-Polymenis, H.L. A Comparison of Cecal Colonization of Salmonella Enterica Serotype Typhimurium in White Leghorn Chicks and Salmonella-Resistant Mice. BMC Microbiol. 2008, 8. [Google Scholar] [CrossRef] [PubMed]
- Jonas, K.; Tomenius, H.; Kader, A.; Normark, S.; Römling, U.; Belova, L.M.; Melefors, Ö. Roles of Curli, Cellulose and BapA in Salmonella Biofilm Morphology Studied by Atomic Force Microscopy. BMC Microbiol. 2007, 7. [Google Scholar] [CrossRef] [PubMed]
- Tursi, S.A.; Puligedda, R.D.; Szabo, P.; Nicastro, L.K.; Miller, A.L.; Qiu, C.; Gallucci, S.; Relkin, N.R.; Buttaro, B.A.; Dessain, S.K.; et al. Salmonella Typhimurium Biofilm Disruption by a Human Antibody That Binds a Pan-Amyloid Epitope on Curli. Nat. Commun. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Punchihewage-Don, A.J.; Ranaweera, P.N.; Parveen, S. Defense Mechanisms of Salmonella against Antibiotics: A Review. Frontiers in Antibiotics 2024, 3. [Google Scholar] [CrossRef]
- Gonzalez-Escobedo, G.; Gunn, J.S. Gallbladder Epithelium as a Niche for Chronic Salmonella Carriage. Infect. Immun. 2013, 81, 2920–2930. [Google Scholar] [CrossRef]
- González, J.F.; Tucker, L.; Fitch, J.; Wetzel, A.; White, P.; Gunn, J.S. Human Bile-Mediated Regulation of Salmonella Curli Fimbriae. In Proceedings of the Journal of Bacteriology; American Society for Microbiology, 1 September 2019; Vol. 201. [Google Scholar]
- González, J.F.; Alberts, H.; Lee, J.; Doolittle, L.; Gunn, J.S. Biofilm Formation Protects Salmonella from the Antibiotic Ciprofloxacin in Vitro and in Vivo in the Mouse Model of Chronic Carriage. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef]
- Fritz, H.M.; Pereira, R. V.; Toohey-Kurth, K.; Marshall, E.; Tucker, J.; Clothier, K.A. Salmonella Enterica Serovar Dublin from Cattle in California from 1993–2019: Antimicrobial Resistance Trends of Clinical Relevance. Antibiotics 2022, 11. [Google Scholar] [CrossRef]
- Ivers, C.; Kaya, E.C.; Yucel, U.; Boyle, D.; Trinetta, V. Evaluation of Salmonella Biofilm Attachment and Hydrophobicity Characteristics on Food Contact Surfaces. BMC Microbiol. 2024, 24. [Google Scholar] [CrossRef] [PubMed]
- Counihan, K.L.; Tilman, S.; Uknalis, J.; Mukhopadhyay, S.; Niemira, B.A.; Bermudez-Aguirre, D. Attachment and Biofilm Formation of Eight Different Salmonella Serotypes on Three Food-Contact Surfaces at Different Temperatures. Microorganisms 2025, 13. [Google Scholar] [CrossRef]
- Kotay, S.M.; Parikh, H.I.; Gweon, H.S.; Barry, K.; Stoesser, N.; Sarah Walker, A.; Crook, D.W.; Vegesana, K.; Mathers, A.J. Biofilm Removal in Hospital Sink Drains Drives Unintended Surges in Antibiotic Resistance. npj Antimicrobials and Resistance 2026, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Metzger, G.A.; Ridenhour, B.J.; France, M.; Gliniewicz, K.; Millstein, J.; Settles, M.L.; Forney, L.J.; Stalder, T.; Top, E.M. Biofilms Preserve the Transmissibility of a Multi-Drug Resistance Plasmid. NPJ Biofilms Microbiomes 2022, 8. [Google Scholar] [CrossRef]
- Xu, J.; Wang, J.; Kang, Z.; Xu, H.; Tang, C.; Chen, J.; Hu, H. Mixed-Species Biofilm with Salmonella in Food Industry: Persistence, Interspecies Interaction, and Control. Food Research International 2025, 221, 117348. [Google Scholar] [CrossRef]
- Álvarez-Espejo, D.M.; Fredes-García, D.; Díaz-Gavidia, C.; Gutiérrez, S.; Barron-Montenegro, R.; Álvarez, F.P.; Constenla-Albornoz, R.; Cadet-Arenas, V.; Reyes-Jara, A.; Olivares-Pacheco, J.; et al. Tracking Antimicrobial Resistance in Salmonella via Poultry Supply Chains, Human Clinical Samples, and Environmental Reservoirs. Foods 2026, 15. [Google Scholar] [CrossRef]
- Balcázar, J.L.; Subirats, J.; Borrego, C.M. The Role of Biofilms as Environmental Reservoirs of Antibiotic Resistance. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Flores-Vargas, G.; Bergsveinson, J.; Lawrence, J.R.; Korber, D.R. Environmental Biofilms as Reservoirs for Antimicrobial Resistance. Front. Microbiol. 2021, 12. [Google Scholar] [CrossRef]
- Nahum, Y.; Muhvich, J.; Morones-Ramirez, J.R.; Casillas-Vega, N.G.; Zaman, M.H. Biofilms as Potential Reservoirs of Antimicrobial Resistance in Vulnerable Settings. Front. Public Health 2025, 13. [Google Scholar] [CrossRef] [PubMed]
- White, A.P.; Surette, M.G. Comparative Genetics of the Rdar Morphotype in Salmonella. J. Bacteriol. 2006, 188, 8395–8406. [Google Scholar] [CrossRef] [PubMed]
- Obe, T.; Richards, A.K.; Shariat, N.W. Differences in Biofilm Formation of Salmonella Serovars on Two Surfaces under Two Temperature Conditions. J. Appl. Microbiol. 2022, 132, 2410–2420. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, G.; Aheto, K.; Shirtliff, M.E.; Tennant, S.M. Poor Biofilm-Forming Ability and Long-Term Survival of Invasive Salmonella Typhimurium ST313. Pathog. Dis. 2016, 74, ftw049. [Google Scholar] [CrossRef] [PubMed]
- Vestby, L.K.; Møretrø, T.; Langsrud, S.; Heir, E.; Nesse, L.L. Biofilm Forming Abilities of Salmonella Are Correlated with Persistence in Fish Meal- and Feed Factories. BMC Vet. Res. 2009, 5. [Google Scholar] [CrossRef] [PubMed]
- Lianou, A.; Koutsoumanis, K.P. Strain Variability of the Biofilm-Forming Ability of Salmonella Enterica under Various Environmental Conditions. Int. J. Food Microbiol. 2012, 160, 171–178. [Google Scholar] [CrossRef]
- Siddique, A.; Azim, S.; Ali, A.; Andleeb, S.; Ahsan, A.; Imran, M.; Rahman, A. Antimicrobial Resistance Profiling of Biofilm Forming Non Typhoidal Salmonella Enterica Isolates from Poultry and Its Associated Food Products from Pakistan. Antibiotics 2021, 10. [Google Scholar] [CrossRef]
- Voss-Rech, D.; Ziech, R.E.; Vaz, C.S.L.; Coldebella, A.; Kuchiishi, S.S.; Balzan, C.; Matter, L.; Vargas, Á.C.; Botton, S.A. Association between Antimicrobial Resistance and Biofilm Forming Ability of Salmonella Enterica Serotypes from Commercial Broiler Farms in Brazil. Br. Poult. Sci. 2023, 64, 224–230. [Google Scholar] [CrossRef]
- Ćwiek, K.; Korzekwa, K.; Tabiś, A.; Bania, J.; Bugla-Płoskońska, G.; Wieliczko, A. Antimicrobial Resistance and Biofilm Formation Capacity of Salmonella Enterica Serovar Enteritidis Strains Isolated from Poultry and Humans in Poland. Pathogens 2020, 9, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Le Hello, S.; Bekhit, A.; Granier, S.A.; Barua, H.; Beutlich, J.; Zaja̧c, M.; Münch, S.; Sintchenko, V.; Bouchrif, B.; Fashae, K.; et al. The Global Establishment of a Highly-Fluoroquinolone Resistant Salmonella Enterica Serotype Kentucky ST198 Strain. Front. Microbiol. 2013, 4. [Google Scholar] [CrossRef]
- Denagamage, T.N.; Wallner-Pendleton, E.; Jayarao, B.M.; Xiaoli, L.; Dudley, E.G.; Wolfgang, D.; Kariyawasam, S. Detection of CTX-M-1 Extended-Spectrum Beta-Lactamase among Ceftiofur-Resistant Salmonella Enterica Clinical Isolates of Poultry. Journal of Veterinary Diagnostic Investigation 2019, 31, 681–687. [Google Scholar] [CrossRef]
- Cejas, D.; Vignoli, R.; Quinteros, M.; Marino, R.; Callejo, R.; Betancor, L.; Gutkind, G.O.; Radice, M.A. First Detection of CMY-2 Plasmid Mediated β-Lactamase in Salmonella Heidelberg in South America. Rev. Argent. Microbiol. 2014, 46, 30–33. [Google Scholar] [CrossRef]
- Hall, C.W.; Mah, T.-F. Molecular Mechanisms of Biofilm-Based Antibiotic Resistance and Tolerance in Pathogenic Bacteria. FEMS Microbiol. Rev. 2017, 41, 276–301. [Google Scholar] [CrossRef] [PubMed]
- Dutt, Y.; Dhiman, R.; Singh, T.; Vibhuti, A.; Gupta, A.; Pandey, R.P.; Raj, V.S.; Chang, C.M.; Priyadarshini, A. The Association between Biofilm Formation and Antimicrobial Resistance with Possible Ingenious Bio-Remedial Approaches. Antibiotics 2022, 11. [Google Scholar] [CrossRef]
- Patkowski, J.B.; Dahlberg, T.; Amin, H.; Gahlot, D.K.; Vijayrajratnam, S.; Vogel, J.P.; Francis, M.S.; Baker, J.L.; Andersson, M.; Costa, T.R.D. The F-Pilus Biomechanical Adaptability Accelerates Conjugative Dissemination of Antimicrobial Resistance and Biofilm Formation. Nat. Commun. 2023, 14. [Google Scholar] [CrossRef]
- González, J.F.; Laipply, B.; Sadowski, V.A.; Price, M.; Gunn, J.S. Functional Role of the Biofilm Regulator CsgD in Salmonella Enterica Sv. Typhi. Front. Cell. Infect. Microbiol. 2024, 14. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Johnston, R.N.; Liu, G.R.; Liu, S.L. Genomic Comparison between Salmonella Gallinarum and Pullorum: Differential Pseudogene Formation under Common Host Restriction. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Townsend, S.M.; Kramer, N.E.; Edwards, R.; Baker, S.; Hamlin, N.; Simmonds, M.; Stevens, K.; Maloy, S.; Parkhill, J.; Dougan, G.; et al. Salmonella Enterica Serovar Typhi Possesses a Unique Repertoire of Fimbrial Gene Sequences. Infect. Immun. 2001, 69, 2894–2901. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Aljahdali, N.; Zhao, S.; Tang, H.; Harbottle, H.; Hoffmann, M.; Frye, J.G.; Foley, S.L. Infection Biology of Salmonella Enterica. EcoSal Plus 2024, 12. [Google Scholar] [CrossRef]
- Aviv, G.; Elpers, L.; Mikhlin, S.; Cohen, H.; Vitman Zilber, S.; Grassl, G.A.; Rahav, G.; Hensel, M.; Gal-Mor, O. The Plasmid-Encoded Ipf and Klf Fimbriae Display Different Expression and Varying Roles in the Virulence of Salmonella Enterica Serovar Infantis in Mouse vs. Avian Hosts. PLoS Pathog. 2017, 13. [Google Scholar] [CrossRef]
- Ba, A.J.; M Tsolis, R.E.; Bowe, F.A.; Kusters, J.G.; Hoffmann, S.; Heffron, F. The Pef Fimbrial Operon of Salmonella Typhimurium Mediates Adhesion to Murine Small Intestine and Is Necessary for Fluid Accumulation in the Infant Mouse; 1996; Vol. 64. [Google Scholar]
- Khajanchi, B.K.; Foley, S.L. Antimicrobial Resistance and Increased Virulence of Salmonella. Microorganisms 2022, 10. [Google Scholar] [CrossRef]
- Wiesner, M.; Zaidi, M.B.; Calva, E.; Fernández-Mora, M.; Calva, J.J.; Silva, C. Association of Virulence Plasmid and Antibiotic Resistance Determinants with Chromosomal Multilocus Genotypes in Mexican Salmonella Enterica Serovar Typhimurium Strains. BMC Microbiol. 2009, 9. [Google Scholar] [CrossRef]
- Yates, C.R.; Nguyen, A.; Liao, J.; Cheng, R.A. What’s on a Prophage: Analysis of Salmonella Spp. Prophages Identifies a Diverse Range of Cargo with Multiple Virulence- and Metabolism-Associated Functions. mSphere 2024, 9. [Google Scholar] [CrossRef]
- Figueroa-Bossi, N.; Bossi, L. Inducible Prophages Contribute to Salmonella Virulence in Mice. Mol. Microbiol. 1999, 33, 167–176. [Google Scholar] [CrossRef]
- Betancor, L.; Yim, L.; Martínez, A.; Fookes, M.; Sasias, S.; Schelotto, F.; Thomson, N.; Maskell, D.; Chabalgoity, J.A. Genomic Comparison of the Closely Related Salmonella Enterica Serovars Enteritidis and Dublin; 2012; Vol. 6. [Google Scholar]
- Martinez-Sanguiné, A.Y.; D’Alessandro, B.; Langleib, M.; Traglia, G.M.; Mónaco, A.; Durán, R.; Chabalgoity, J.A.; Betancor, L.; Yim, L. Salmonella Enterica Serovars Dublin and Enteritidis Comparative Proteomics Reveals Differential Expression of Proteins Involved in Stress Resistance, Virulence, and Anaerobic Metabolism. Infect. Immun. 2021, 89. [Google Scholar] [CrossRef]
- Arrieta-Gisasola, A.; Martínez-Ballesteros, I.; Martinez-Malaxetxebarria, I.; Garrido, V.; Grilló, M.J.; Bikandi, J.; Laorden, L. Pan-Genome-Wide Association Study Reveals a Key Role of the Salmochelin Receptor IroN in the Biofilm Formation of Salmonella Typhimurium and Its Monophasic Variant 4,[5],12:I:-. Int. J. Food Microbiol. 2024, 419, 110753. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.S.; Hahn, T.W. Comparative Proteomic Analysis of Salmonella Enterica Serovars Enteritidis, Typhimurium and Gallinarum. Journal of Veterinary Medical Science 2012, 74, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Jia, K.; Wang, G.; Liang, L.; Wang, M.; Wang, H.; Xu, X. Preliminary Transcriptome Analysis of Mature Biofilm and Planktonic Cells of Salmonella Enteritidis Exposure to Acid Stress. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Giaouris, E.; Samoilis, G.; Chorianopoulos, N.; Ercolini, D.; Nychas, G.J. Differential Protein Expression Patterns between Planktonic and Biofilm Cells of Salmonella Enterica Serovar Enteritidis PT4 on Stainless Steel Surface. Int. J. Food Microbiol. 2013, 162, 105–113. [Google Scholar] [CrossRef]
- Hamilton, S.; Bongaerts, R.J.M.; Mulholland, F.; Cochrane, B.; Porter, J.; Lucchini, S.; Lappin-Scott, H.M.; Hinton, J.C.D. The Transcriptional Programme of Salmonella Enterica Serovar Typhimurium Reveals a Key Role for Tryptophan Metabolism in Biofilms. BMC Genomics 2009, 10. [Google Scholar] [CrossRef]
- Zheng, L.; Zhang, X.; Lu, Z.; Ma, W.; Hu, A.; Zhou, H.; Bie, X. Transcriptome Sequencing Reveals the Difference in the Expression of Biofilm and Planktonic Cells between Two Strains of Salmonella Typhimurium. Biofilm 2022, 4. [Google Scholar] [CrossRef] [PubMed]
- Osman, K.M.; Ali, M.M.; Radwan, M.I.; Kim, H.K.; Han, J. Comparative Proteomic Analysis on Salmonella Gallinarum and Salmonella Enteritidis Exploring Proteins That May Incorporate Host Adaptation in Poultry. J. Proteomics 2009, 72, 815–821. [Google Scholar] [CrossRef]
- Encheva, V.; Wait, R.; Gharbia, S.E.; Begum, S.; Shah, H.N. Proteome Analysis of Serovars Typhimurium and Pullorum of Salmonella Enterica Subspecies I. BMC Microbiol. 2005, 5. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Li, H.; Yang, J.; Yang, X.; Miao, X.; Fan, X.; Liu, L.; Li, X. Temporal Transcriptome Profiling in the Response to Salmonella Enterica Serovar Enteritidis Infection in Chicken Cecum. Poult. Sci. 2025, 104, 104773. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, L.; Zhao, Y.; Ren, Y.; Miao, X.; Dong, Y.; Liu, L.; Li, X. Transcriptomic and Proteomic Analysis Reveals the Mechanism of Chicken Cecum Response to Salmonella Enterica Serovar Enteritidis Inoculation. iScience 2025, 28. [Google Scholar] [CrossRef] [PubMed]
- Harrell, J.E.; Hahn, M.M.; D’Souza, S.J.; Vasicek, E.M.; Sandala, J.L.; Gunn, J.S.; McLachlan, J.B. Salmonella Biofilm Formation, Chronic Infection, and Immunity Within the Intestine and Hepatobiliary Tract. Front. Cell. Infect. Microbiol. 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Biotechnology, S.N. A Comprehensive Review on the Development of Salmonella Biofilm on Gallbladder Surface. 2022, Vol. 7. [Google Scholar]
- Wheeler, K.M.; Gold, M.A.; Stevens, C.A.; Tedin, K.; Wood, A.M.; Uzun, D.; Cárcamo-Oyarce, G.; Turner, B.S.; Fulde, M.; Song, J.; et al. Mucus-Derived Glycans Are Inhibitory Signals for Salmonella Typhimurium SPI-1-Mediated Invasion. Cell Rep. 2025, 44, 116304. [Google Scholar] [CrossRef] [PubMed]
- Guillín, Y.; Ortiz, C.; Hidalgo, W. Comparative Metabolic Study of Planktonic and Sessile Cells in Salmonella Enteritidis ATCC 13076: Elucidating Metabolic Pathways Driving Biofilm Formation. PLoS One 2025, 20. [Google Scholar] [CrossRef] [PubMed]
- Sarjit, A.; Cheah, Y.; Dykes, G.A. The Basis for Variations in the Biofilm Formation by Different Salmonella Species and Subspecies: An In Vitro and In Silico Scoping Study. Appl. Microbiol. 2023, 3, 841–855. [Google Scholar] [CrossRef]
- Meng, F.; Lyu, F.; Bie, X.; Lu, Y.; Lu, Z. Advances in Transcriptomic Analysis of Salmonella Biofilms and Their Correlation with Food Safety. Curr. Opin. Food Sci. 2024, 55, 101110. [Google Scholar] [CrossRef]
- Desai, P.T.; Porwollik, S.; Long, F.; Cheng, P.; Wollam, A.; Clifton, S.W.; Weinstock, G.M.; McClelland, M. Evolutionary Genomics of Salmonella Enterica Subspecies. mBio 2013, 4. [Google Scholar] [CrossRef]
- Azevedo, N.F.; Lopes, S.P.; Keevil, C.W.; Pereira, M.O.; Vieira, M.J. Time to “Go Large” on Biofilm Research: Advantages of an Omics Approach. Biotechnol. Lett. 2009, 31, 477–485. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhang, H.; Gao, Y.; Sun, Y. Multi-Omics Strategies for Biomarker Discovery and Application in Personalized Oncology. Molecular Biomedicine 2025, 6. [Google Scholar] [CrossRef]
- Agamah, F.E.; Bayjanov, J.R.; Niehues, A.; Njoku, K.F.; Skelton, M.; Mazandu, G.K.; Ederveen, T.H.A.; Mulder, N.; Chimusa, E.R.; t Hoen, P.A.C. Computational Approaches for Network-Based Integrative Multi-Omics Analysis. Front. Mol. Biosci. 2022, 9. [Google Scholar] [CrossRef]
- Baião, A.R.; Cai, Z.; Poulos, R.C.; Robinson, P.J.; Reddel, R.R.; Zhong, Q.; Vinga, S.; Gonçalves, E. A Technical Review of Multi-Omics Data Integration Methods: From Classical Statistical to Deep Generative Approaches. Brief. Bioinform. 2025, 26. [Google Scholar] [CrossRef]
- Kwoji, I.D.; Aiyegoro, O.A.; Okpeku, M.; Adeleke, M.A. ‘Multi-Omics’ Data Integration: Applications in Probiotics Studies. NPJ Sci. Food 2023, 7. [Google Scholar] [CrossRef]
- Wang, C.-C.; Hung, Y.-T.; Chou, C.-Y.; Hsuan, S.-L.; Chen, Z.-W.; Chang, P.-Y.; Jan, T.-R.; Tung, C.-W. Using Random Forest to Predict Antimicrobial Minimum Inhibitory Concentrations of Nontyphoidal Salmonella in Taiwan. Vet. Res. 2023, 54. [Google Scholar] [CrossRef]
- Dickinson, Q.; Aufschnaiter, A.; Ott, M.; Meyer, J.G. Multi-Omic Integration by Machine Learning (MIMaL). Bioinformatics 2022, 38, 4908–4918. [Google Scholar] [CrossRef]
- Ghosh, A.; Vang, C.K.; Brenner, E.P.; Ravi, J. Unlocking Antimicrobial Resistance with Multiomics and Machine Learning. Trends Microbiol. 2025, 33, 1048–1051. [Google Scholar] [CrossRef]
- Liu, C.; Huang, H.; Yang, P. Multi-Task Learning from Multimodal Single-Cell Omics with Matilda. Nucleic Acids Res. 2023, 51, e45. [Google Scholar] [CrossRef] [PubMed]
- Ranganathan, S.; Nagarajan, H.; Busi, S.; Esakkiraj, P.; Parasuraman, P.; Mariasoosai, R.C.C.; Vetrivel, U. Editorial: Decoding Biofilm Resilience: Integrative Multi-Omics Approaches and Novel Disruption Strategies. Front. Cell. Infect. Microbiol. 2026, 15. [Google Scholar] [CrossRef]
- Li, L.; Gao, X.; Li, M.; Liu, Y.; Ma, J.; Wang, X.; Yu, Z.; Cheng, W.; Zhang, W.; Sun, H.; et al. Relationship between Biofilm Formation and Antibiotic Resistance of Klebsiella Pneumoniae and Updates on Antibiofilm Therapeutic Strategies. Front. Cell. Infect. Microbiol. 2024, 14. [Google Scholar] [CrossRef] [PubMed]
| Salmonella Serovar | Host Association |
|---|---|
| Typhimurium | Broad host range |
| Enteritidis | Broad host range |
| Heidelberg | Broad host range |
| Kentucky | Broad host range |
| Infantis | Broad host range |
| Newport | Broad host range |
| Dublin | Cattle-adapted, occasionally humans |
| Gallinarum | Poultry-restricted |
| Pullorum | Poultry-restricted |
| Typhi | Human-restricted |
| Paratyphi A | Human restricted |
| Monophasic 4,[5],12:i:- | Broad host range |
| Hadar | Broad host range |
| Schwarzengrund | Broad host range |
| Thompson | Broad host range |
| Agona | Broad host range |
| Braenderup | Broad host range |
| Montevideo | Broad host range |
| Senftenberg | Broad host range |
| Derby | Broad host range |
| Braenderup | Broad host range |
| Muenchen | Broad host range |
| Lagos | Broad host range |
| Labadi | Broad host range |
| Anatum | Broad host range |
| Give | Broad host range |
| Mbandaka | Broad host range |
| Salmonella Serovar | Surface Type/temperature | Biofilm assay | Findings | References |
|---|---|---|---|---|
| Schwarzengrund, Heidelberg, Newport, Braenderup, Hadar, Infantis, Kentucky, Thompson, Typhimurium, Enteritidis (Total 20 isolates) | Plastic vs stainless steel; 25 °C and 15 °C | Crystal violet microtiter biofilm assay | Schwarzengrund – strong biofilms; Braenderup – strong/moderate biofilms; Enteritidis – moderate biofilms; Infantis – moderate biofilms; Typhimurium – moderate biofilms; Thompson – moderate biofilms; Hadar – moderate/weak biofilms; Kentucky – weak biofilms; Heidelberg – weak biofilms; Newport – weak biofilms; plastic surfaces generally supported stronger biofilm than stainless steel, especially at 15 °C. | [36] |
| Agona, Montevideo, Senftenberg, Typhimurium (Total 111 isolates) | Polystyrene microtiter plates and liquid-air interface; ~20 °C | Crystal violet microtiter biofilm assay; pellicle formation assay | Agona – strong biofilms; Montevideo – strong biofilms; Senftenberg – moderate biofilms; Typhimurium – weak biofilms; persistent factory serovars (Agona and Montevideo) showed greater biofilm formation than non-persistent serovars, suggesting biofilm ability contributes to environmental persistence. | [38] |
| Typhimurium, Enteritidis, Newport, Heidelberg, Agona, Montevideo, Infantis, Senftenberg, Derby (Total 60 isolates) | Different environmental conditions: pH 3.8-7.0; NaCl 0.5-8%; temperature 4-37 °C | Crystal violet biofilm assays under factorial environmental conditions | Typhimurium – variable biofilms; Enteritidis – variable biofilms; Newport – variable biofilms; Heidelberg – variable biofilms; Agona – strong biofilms in some strains; Montevideo – moderate/variable biofilms; Infantis – variable biofilms; Senftenberg – moderate biofilms; Derby – limited data; biofilms formation strongly influenced by environmental factors (pH, NaCl, temperature) rather than serovar identity. | [39] |
| Typhimurium (sequence types ST19 and ST313 clinical isolates) (Total 16 isolates) | Polystyrene microtiter plates; glass flow cells; polycarbonate membranes; 25 °C, 28 °C, and 37 °C | Crystal violet microtiter assay; Congo-red RDAR* morphotype assay; pellicle formation assay; continuous-flow biofilm system | Typhimurium ST19 – strong biofilms; Typhimurium ST313 – weak/poor biofilms; ST19 isolates produced RDAR morphotype and thick biofilms in flow-cell systems, whereas ST313 isolates showed weak attachment and minimal biofilm formation; ST19 strains also survived desiccation and disinfectant treatment better than ST313 strains. | [37] |
| Typhimurium, Enteritidis, and other Salmonella spp. (Total 95 isolates) | Polystyrene 96-well microtiter plates; 30 °C and 37 °C | Crystal violet microtiter plate biofilm assay | Typhimurium – strong biofilms; Enteritidis – moderate/strong biofilms; other Salmonella spp. – moderate/strong biofilms; most isolates showed stronger biofilm formation at 37 °C, with 78.5% of Typhimurium isolates producing strong biofilms, while Enteritidis showed ~41.7% strong biofilm at 37 °C; strong biofilm formation was correlated with multidrug resistance (MDR). | [40] |
| Heidelberg, Muenchen, Schwarzengrund, Lagos, Labadi, Anatum, Give, Derby, Mbandaka, 0:a,5:i:-, 0:-:r:2, 0:4,5:b:- (Total 37 isolates) | Polystyrene microtiter plates; 35 °C incubation for 24 h | Crystal violet microtiter plate biofilm assay | Schwarzengrund – moderate biofilms; Muenchen – moderate/weak biofilms; Derby – moderate biofilms; Mbandaka – moderate biofilms; Heidelberg – weak or non-biofilms; Lagos – weak biofilms; Labadi – weak biofilms; Anatum – weak biofilms; Give – weak biofilms; 0:a,5:i:- – weak biofilms; 0:-:r:2 – non-biofilms; 0:4,5:b:- – weak biofilms. Overall, 65% of isolates produced biofilm, mostly weak to moderate producers, and multidrug-resistant isolates tended to form weaker biofilms. |
[41] |
| Enteritidis (Total 95 isolates; 51 from poultry and 44 from human feces) | Congo red agar medium; 28 °C | RDAR*/BDAR#/PDAR¥ colony morphotype analysis; gene expression analysis of biofilm-related genes (adrA, csgD, luxS, sdiA) | Enteritidis – strong/weak biofilms; RDAR morphotype (curli + cellulose) observed in ~39% of poultry isolates and ~34% of human isolates; BDAR and PDAR morphotypes also detected; strong biofilm formation associated with increased expression of adrA and csgD genes, whereas some strains showed weak biofilm formation despite the expression of quorum sensing-associated genes. |
[42] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




