Submitted:
12 March 2026
Posted:
13 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Trajectory Inference and Pseudotime Analysis
2.3. Differential Gene Expression Along Trajectories
2.4. Module Score Analysis
2.5. Cell-Cell Communication Analysis
3. Results
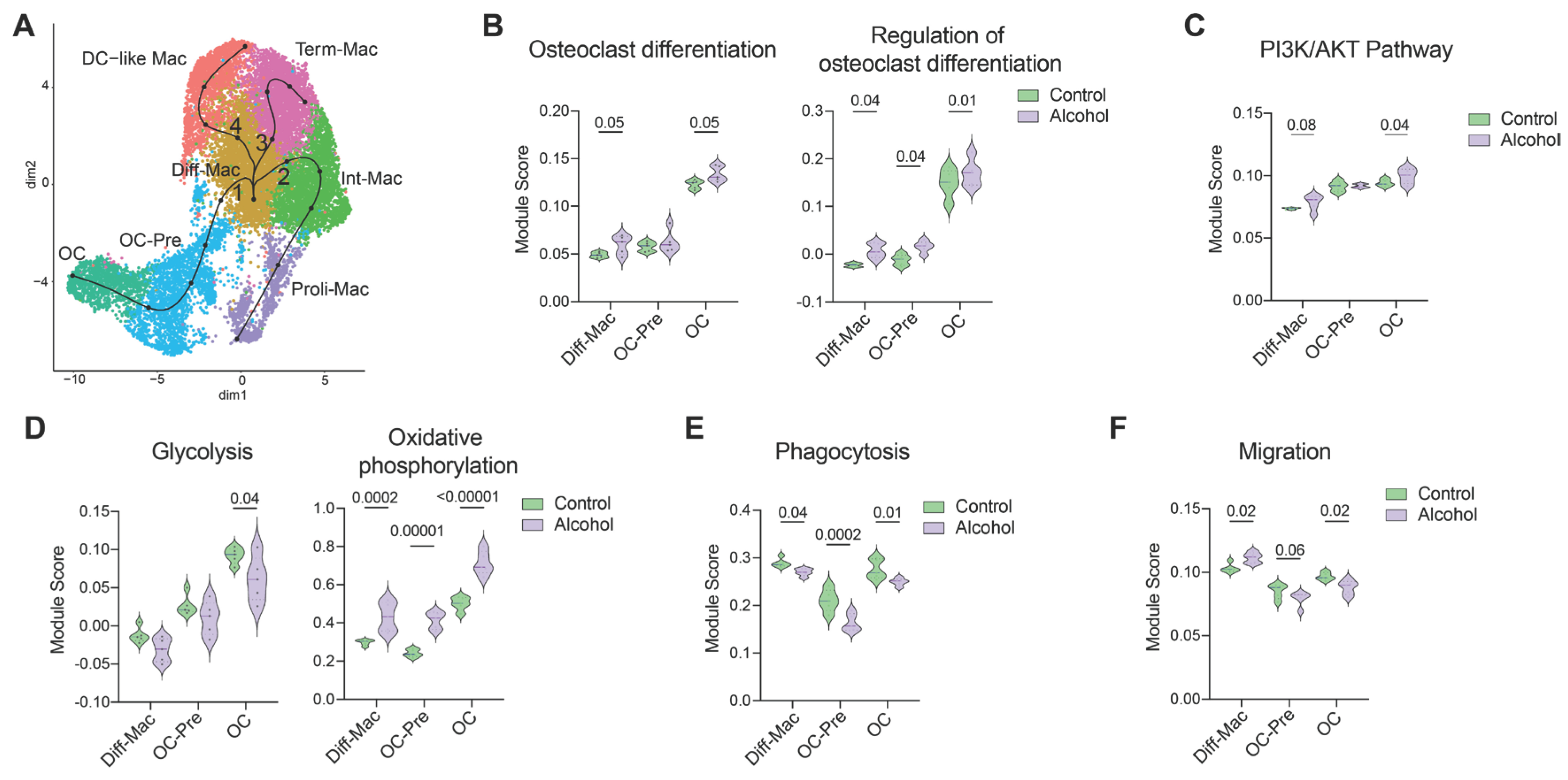
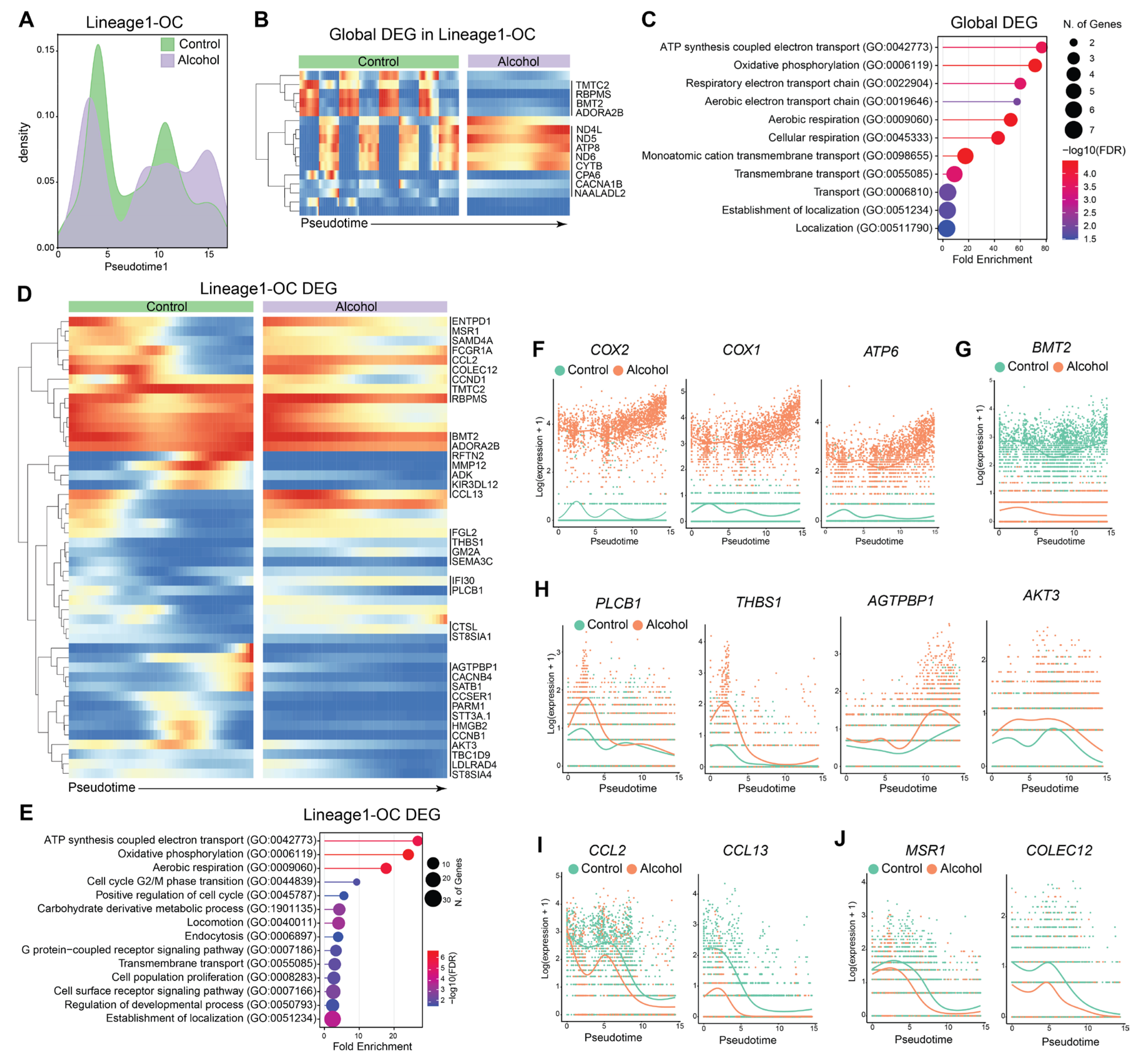
3.1. Chronic Alcohol Consumption Reprograms Cellular Metabolism During Enhanced Osteoclastogenesis.
3.2. Alcohol-Mediated Transcriptional Changes Lead to Early Fate Commitment Bias
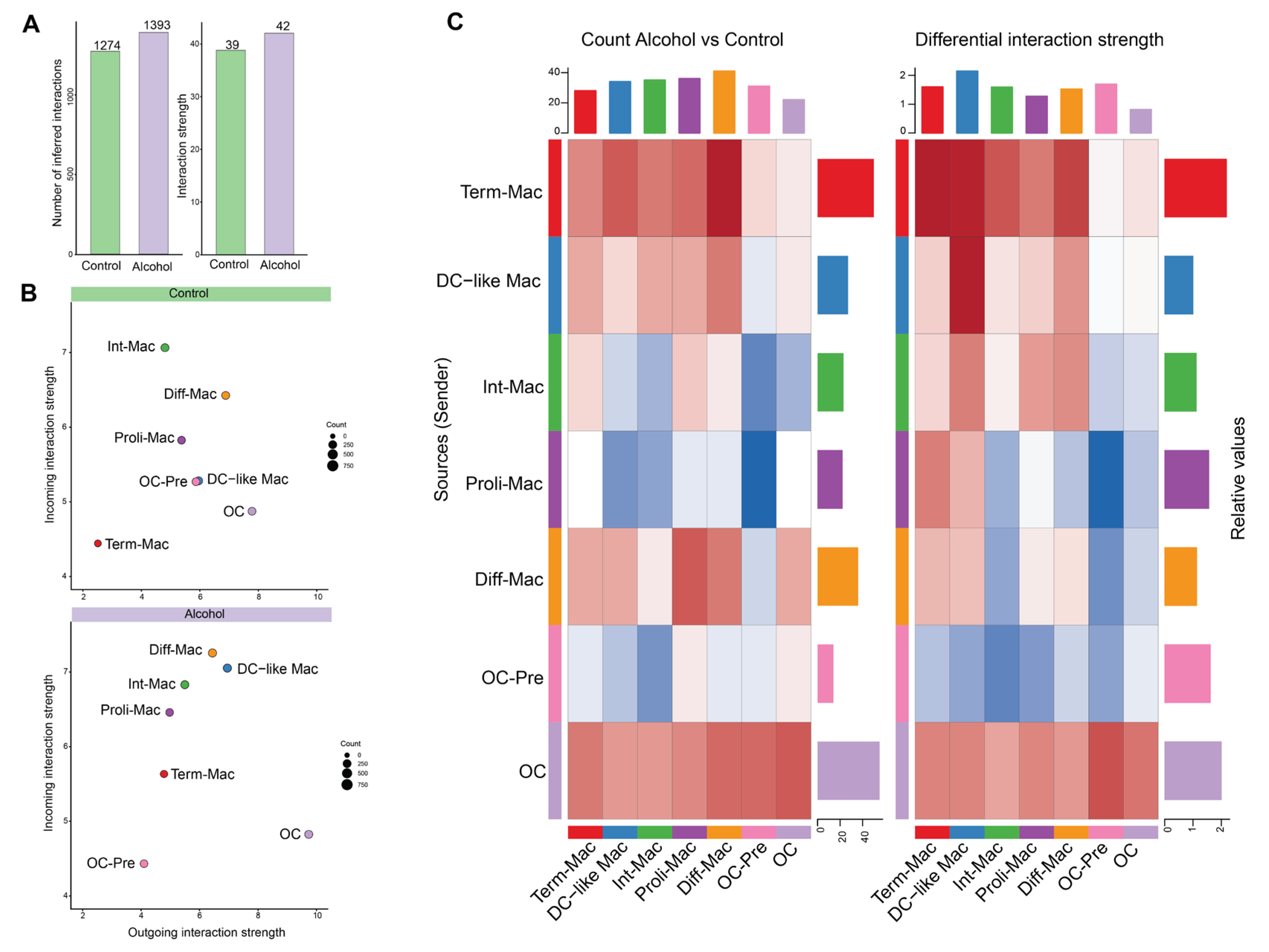
3.3. Alcohol Amplifies Intercellular Communication During Osteoclastogenesis
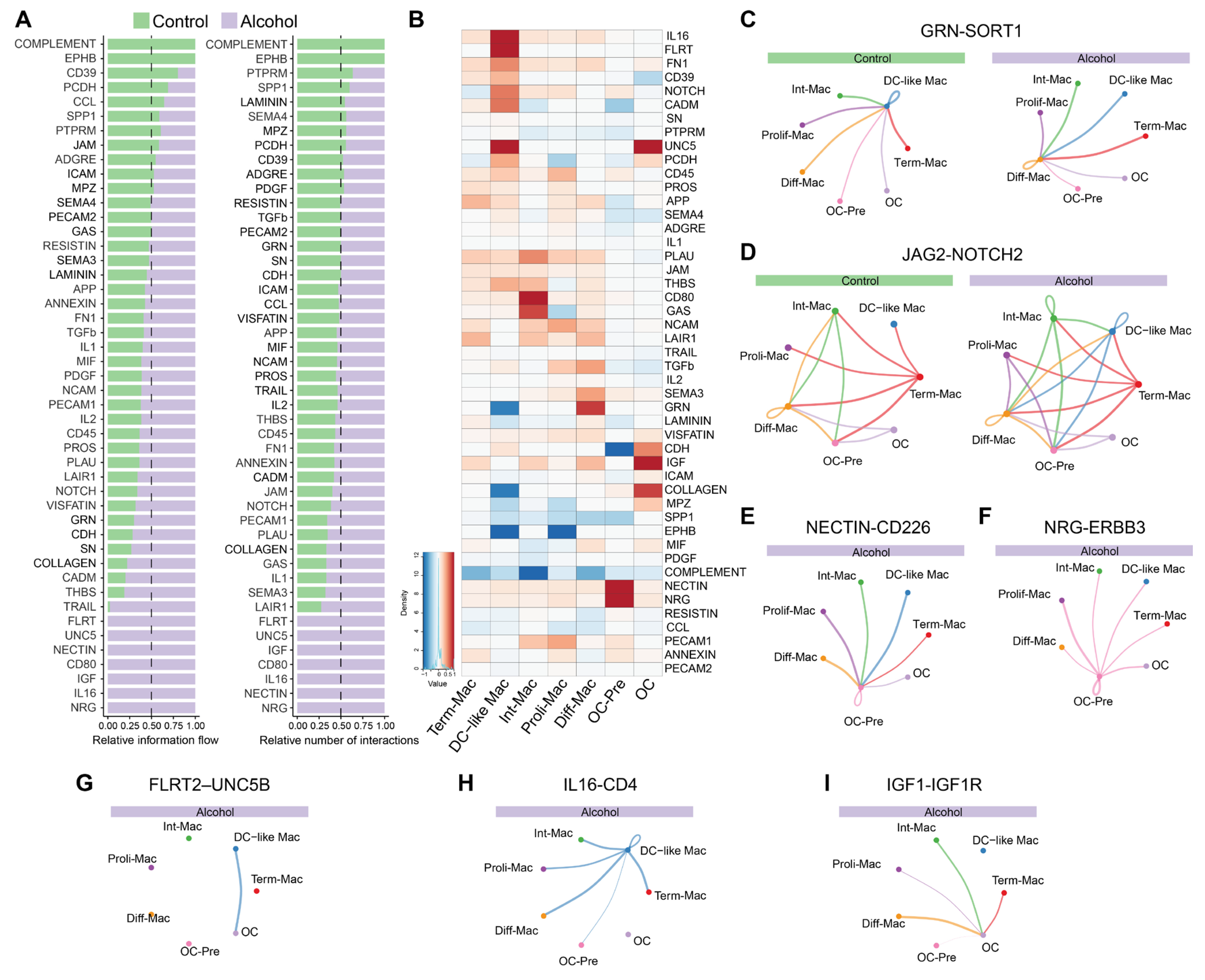
3.4. Alcohol Induces Fate-Instructive, Adhesion, and Matrix-Dependent Signaling in the Osteoclast Lineage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| scRNA-seq | Single-cell RNA sequencing |
| HSPC | Hematopoietic stem and progenitor cell |
| M-CSF | Macrophage colony-stimulating factor |
| RANKL | Receptor activator of nuclear factor κB ligand |
| GAM | Generalized additive model |
| FDR | False discovery rate |
| GO-PB | Gene Ontology Biological processes |
| DEG | Differentially expressed gene |
| Diff-Mac | Differentiating macrophage |
| OC-Pre | Osteoclast Precursor |
| OC | Mature osteoclast |
| Int-Mac | Intermediate macrophage |
| Proli-Mac | Proliferating macrophage |
| Term-Mac | Terminally differentiated macrophage |
| DC-like Mac | Dendritic-like macrophage |
| NRG | Neuregulin |
| CDH | Cadherins |
| OsteoMAC | Osteal macrophage |
References
- (NIAAA), N.I.o.A.A.a.A. Alcohol Use in the United States: Age Groups and Demographic Characteristics 2024. Available online: https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-use-united-states-age-groups-and-demographic-characteristics# (accessed on Feb 11).
- Maurel, D.B.; Boisseau, N.; Benhamou, C.L.; Jaffre, C. Alcohol and bone: review of dose effects and mechanisms. Osteoporos Int 2012, 23, 1–16. [Google Scholar] [CrossRef]
- Cheraghi, Z.; Doosti-Irani, A.; Almasi-Hashiani, A.; Baigi, V.; Mansournia, N.; Etminan, M.; Mansournia, M.A. The effect of alcohol on osteoporosis: A systematic review and meta-analysis. Drug Alcohol Depend 2019, 197, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Giampieri, F.; Chisari, E.; Micek, A.; Paladino, N.; Forbes-Hernandez, T.Y.; Quiles, J.L.; Battino, M.; La Vignera, S.; Musumeci, G.; et al. Alcohol Consumption, Bone Mineral Density, and Risk of Osteoporotic Fractures: A Dose-Response Meta-Analysis. Int J Environ Res Public Health 2022, 19. [Google Scholar] [CrossRef] [PubMed]
- Hemati, H.; Blanton, M.B.; Koura, J.; Khadka, R.; Grant, K.A.; Messaoudi, I. Chronic Alcohol Consumption Enhances the Differentiation Capacity of Hematopoietic Stem and Progenitor Cells into Osteoclast Precursors. Am J Pathol 2025. [Google Scholar] [CrossRef] [PubMed]
- Eby, J.M.; Sharieh, F.; Azevedo, J.; Callaci, J.J. Episodic alcohol exposure attenuates mesenchymal stem cell chondrogenic differentiation during bone fracture callus formation. Alcohol Clin Exp Res 2022, 46, 915–927. [Google Scholar] [CrossRef]
- Eby, J.M.; Sharieh, F.; Callaci, J.J. Impact of Alcohol on Bone Health, Homeostasis and Fracture repair. Curr Pathobiol Rep 2020, 8, 75–86. [Google Scholar] [CrossRef]
- Jacome-Galarza, C.E.; Percin, G.I.; Muller, J.T.; Mass, E.; Lazarov, T.; Eitler, J.; Rauner, M.; Yadav, V.K.; Crozet, L.; Bohm, M.; et al. Developmental origin, functional maintenance and genetic rescue of osteoclasts. Nature 2019, 568, 541–545. [Google Scholar] [CrossRef]
- Tsai, J.; Kaneko, K.; Suh, A.J.; Bockman, R.; Park-Min, K.H. Origin of Osteoclasts: Osteoclast Precursor Cells. J Bone Metab 2023, 30, 127–140. [Google Scholar] [CrossRef]
- Tsukasaki, M.; Takayanagi, H. Osteoclast biology in the single-cell era. Inflamm Regen 2022, 42, 27. [Google Scholar] [CrossRef]
- Tsukasaki, M.; Huynh, N.C.; Okamoto, K.; Muro, R.; Terashima, A.; Kurikawa, Y.; Komatsu, N.; Pluemsakunthai, W.; Nitta, T.; Abe, T.; et al. Stepwise cell fate decision pathways during osteoclastogenesis at single-cell resolution. Nat Metab 2020, 2, 1382–1390. [Google Scholar] [CrossRef]
- Ponzetti, M.; Rucci, N. Updates on Osteoimmunology: What’s New on the Cross-Talk Between Bone and Immune System. Front Endocrinol (Lausanne) 2019, 10, 236. [Google Scholar] [CrossRef]
- Lewis, S.A.; Doratt, B.M.; Qiao, Q.; Blanton, M.; Grant, K.A.; Messaoudi, I. Integrated single cell analysis shows chronic alcohol drinking disrupts monocyte differentiation in the bone marrow. Stem Cell Reports 2023. [Google Scholar] [CrossRef]
- Sattgast, L.H.; Branscum, A.J.; Walter, N.A.R.; Newman, N.; Gonzales, S.W.; Grant, K.A.; Turner, R.T.; Iwaniec, U.T. Effects of graded increases in ethanol consumption on biochemical markers of bone turnover in young adult male cynomolgus macaques. Alcohol 2021, 91, 53–59. [Google Scholar] [CrossRef]
- Zhakubayev, A.; Sattgast, L.H.; Lewis, A.D.; Grant, K.A.; Turner, R.T.; Iwaniec, U.T.; Benton, M.L. Ethanol consumption in non-human primates alters plasma markers of bone turnover but not tibia architecture. Sci Rep 2024, 14, 14137. [Google Scholar] [CrossRef]
- Gaddini, G.W.; Grant, K.A.; Woodall, A.; Stull, C.; Maddalozzo, G.F.; Zhang, B.; Turner, R.T.; Iwaniec, U.T. Twelve months of voluntary heavy alcohol consumption in male rhesus macaques suppresses intracortical bone remodeling. Bone 2015, 71, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Kahler-Quesada, A.M.; Grant, K.A.; Walter, N.A.R.; Newman, N.; Allen, M.R.; Burr, D.B.; Branscum, A.J.; Maddalozzo, G.F.; Turner, R.T.; Iwaniec, U.T. Voluntary Chronic Heavy Alcohol Consumption in Male Rhesus Macaques Suppresses Cancellous Bone Formation and Increases Bone Marrow Adiposity. Alcohol Clin Exp Res 2019, 43, 2494–2503. [Google Scholar] [CrossRef]
- Kuah, A.H.; Sattgast, L.H.; Grant, K.A.; Gonzales, S.W.; Khadka, R.; Damrath, J.G.; Allen, M.R.; Burr, D.B.; Wallace, J.M.; Maddalozzo, G.F.; et al. Six months of voluntary alcohol consumption in male cynomolgus macaques reduces intracortical bone porosity without altering mineralization or mechanical properties. Bone 2024, 185. [Google Scholar] [CrossRef]
- Baker, E.J.; Farro, J.; Gonzales, S.; Helms, C.; Grant, K.A. Chronic alcohol self-administration in monkeys shows long-term quantity/frequency categorical stability. Alcohol Clin Exp Res 2014, 38, 2835–2843. [Google Scholar] [CrossRef] [PubMed]
- Grant, K.A.; Leng, X.; Green, H.L.; Szeliga, K.T.; Rogers, L.S.; Gonzales, S.W. Drinking typography established by scheduled induction predicts chronic heavy drinking in a monkey model of ethanol self-administration. Alcohol Clin Exp Res 2008, 32, 1824–1838. [Google Scholar] [CrossRef]
- Jimenez, V.A.; Helms, C.M.; Cornea, A.; Meshul, C.K.; Grant, K.A. An ultrastructural analysis of the effects of ethanol self-administration on the hypothalamic paraventricular nucleus in rhesus macaques. Front Cell Neurosci 2015, 9, 260. [Google Scholar] [CrossRef] [PubMed]
- Street, K.; Risso, D.; Fletcher, R.B.; Das, D.; Ngai, J.; Yosef, N.; Purdom, E.; Dudoit, S. Slingshot: cell lineage and pseudotime inference for single-cell transcriptomics. BMC Genomics 2018, 19, 477. [Google Scholar] [CrossRef]
- Roux de Bezieux, H.; Van den Berge, K.; Street, K.; Dudoit, S. Trajectory inference across multiple conditions with condiments. Nat Commun 2024, 15, 833. [Google Scholar] [CrossRef]
- Van den Berge, K.; Roux de Bézieux, H.; Street, K.; Saelens, W.; Cannoodt, R.; Saeys, Y.; Dudoit, S.; Clement, L. Trajectory-based differential expression analysis for single-cell sequencing data. Nature Communications 2020, 11. [Google Scholar] [CrossRef]
- Ge, S.X.; Jung, D.; Yao, R. ShinyGO: a graphical gene-set enrichment tool for animals and plants. Bioinformatics 2020, 36, 2628–2629. [Google Scholar] [CrossRef]
- Jin, S.; Guerrero-Juarez, C.F.; Zhang, L.; Chang, I.; Ramos, R.; Kuan, C.H.; Myung, P.; Plikus, M.V.; Nie, Q. Inference and analysis of cell-cell communication using CellChat. Nat Commun 2021, 12, 1088. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Huang, M.; Tan, J.Y.; Chen, X.T.; Lei, L.H.; Wu, Y.M.; Zhang, D.Y. PI3K/AKT pathway involvement in the osteogenic effects of osteoclast culture supernatants on preosteoblast cells. Tissue Eng Part A 2013, 19, 2226–2232. [Google Scholar] [CrossRef]
- Park-Min, K.H. Metabolic reprogramming in osteoclasts. Semin Immunopathol 2019, 41, 565–572. [Google Scholar] [CrossRef]
- Srivastava, R.K.; Sapra, L.; Mishra, P.K. Osteometabolism: Metabolic Alterations in Bone Pathologies. Cells 2022, 11. [Google Scholar] [CrossRef]
- Ledesma-Colunga, M.G.; Passin, V.; Lademann, F.; Hofbauer, L.C.; Rauner, M. Novel Insights into Osteoclast Energy Metabolism. Curr Osteoporos Rep 2023, 21, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Zhang, Y.; Wang, G.; Zhang, C.; Wang, F.; Shi, J.; Zhang, T.; Ding, J. Molecular mechanism of S-adenosylmethionine sensing by SAMTOR in mTORC1 signaling. Sci Adv 2022, 8, eabn3868. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Lin, D.; Zhang, J.; Habib, P.; Smith, P.; Murtha, J.; Fu, Z.; Yao, Z.; Qi, Y.; Keller, E.T. Chronic alcohol ingestion induces osteoclastogenesis and bone loss through IL-6 in mice. J Clin Invest 2000, 106, 887–895. [Google Scholar] [CrossRef]
- Iitsuka, N.; Hie, M.; Nakanishi, A.; Tsukamoto, I. Ethanol increases osteoclastogenesis associated with the increased expression of RANK, PU.1 and MITF in vitro and in vivo. Int J Mol Med 2012, 30, 165–172. [Google Scholar] [CrossRef]
- Zhu, S.; Yan, M.Q.; Masson, A.; Chen, W.; Li, Y.P. Cell signaling and transcriptional regulation of osteoclast lineage commitment, differentiation, bone resorption and diseases. Cell Discov 2026, 12, 6. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, N. Signaling Pathways in Osteoclast Differentiation. Chonnam Med J 2016, 52, 12–17. [Google Scholar] [CrossRef]
- Fang, C.; Qiao, Y.; Mun, S.H.; Lee, M.J.; Murata, K.; Bae, S.; Zhao, B.; Park-Min, K.H.; Ivashkiv, L.B. Cutting Edge: EZH2 Promotes Osteoclastogenesis by Epigenetic Silencing of the Negative Regulator IRF8. J Immunol 2016, 196, 4452–4456. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.; Takeshita, S.; Barker, J.E.; Kanagawa, O.; Ross, F.P.; Teitelbaum, S.L. TNF-alpha induces osteoclastogenesis by direct stimulation of macrophages exposed to permissive levels of RANK ligand. J Clin Invest 2000, 106, 1481–1488. [Google Scholar] [CrossRef]
- Liao, T.; Chen, W.; Sun, J.; Zhang, Y.; Hu, X.; Yang, S.; Qiu, H.; Li, S.; Chu, T. CXCR4 Accelerates Osteoclastogenesis Induced by Non-Small Cell Lung Carcinoma Cells Through Self-Potentiation and VCAM1 Secretion. Cell Physiol Biochem 2018, 50, 1084–1099. [Google Scholar] [CrossRef]
- Faccio, R.; Choi, Y.; Teitelbaum, S.L.; Takayanagi, H. The Osteoclast: The Pioneer of Osteoimmunology. In Osteoimmunology; Lorenzo, J., Choi, Y., Horowitz, M., Takayanagi, H., Eds.; Academic Press: Cambridge, MA, USA, 2011; pp. 141–185. [Google Scholar]
- Galson, D.L.; Roodman, G.D. Origins of Osteoclasts. In Osteoimmunology; Lorenzo, J., Choi, Y., Horowitz, M., Takayanagi, H., Eds.; Academic Press: Cambridge, MA, USA, 2011; pp. 7–41. [Google Scholar]
- Boyce, B.F.; Xing, L.; Schwarz, E.M. The Role of the Immune System and Bone Cells in Acute and Chronic Osteomyelitis. In Osteoimmunology; Lorenzo, J., Choi, Y., Horowitz, M., Takayanagi, H., Eds.; Academic Press: Cambridge, MA, USA, 2011; pp. 369–389. [Google Scholar]
- Lemma, S.; Sboarina, M.; Porporato, P.E.; Zini, N.; Sonveaux, P.; Di Pompo, G.; Baldini, N.; Avnet, S. Energy metabolism in osteoclast formation and activity. Int J Biochem Cell Biol 2016, 79, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Bertels, J.C.; He, G.; Long, F. Metabolic reprogramming in skeletal cell differentiation. Bone Res 2024, 12, 57. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Li, J.; Xie, X.; Gu, F.; Sui, Z.; Zhang, K.; Yu, T. Macrophage-Osteoclast Associations: Origin, Polarization, and Subgroups. Front Immunol 2021, 12, 778078. [Google Scholar] [CrossRef]
- Xiao, Y.; Palomero, J.; Grabowska, J.; Wang, L.; de Rink, I.; van Helvert, L.; Borst, J. Macrophages and osteoclasts stem from a bipotent progenitor downstream of a macrophage/osteoclast/dendritic cell progenitor. Blood Adv 2017, 1, 1993–2006. [Google Scholar] [CrossRef]
- Halper, J.; Dolfi, B.; Ivanov, S.; Madel, M.B.; Blin-Wakkach, C. Macrophages and osteoclasts: similarity and divergence between bone phagocytes. Front Immunol 2025, 16, 1683872. [Google Scholar] [CrossRef]
- Gudgeon, J.; Marin-Rubio, J.L.; Trost, M. The role of macrophage scavenger receptor 1 (MSR1) in inflammatory disorders and cancer. Front Immunol 2022, 13, 1012002. [Google Scholar] [CrossRef] [PubMed]
- Georgess, D.; Machuca-Gayet, I.; Blangy, A.; Jurdic, P. Podosome organization drives osteoclast-mediated bone resorption. Cell Adh Migr 2014, 8, 191–204. [Google Scholar] [CrossRef]
- Jurdic, P.; Saltel, F.; Chabadel, A.; Destaing, O. Podosome and sealing zone: specificity of the osteoclast model. Eur J Cell Biol 2006, 85, 195–202. [Google Scholar] [CrossRef]
- Novack, D.V.; Faccio, R. Osteoclast motility: putting the brakes on bone resorption. Ageing Res Rev 2011, 10, 54–61. [Google Scholar] [CrossRef]
- Takegahara, N.; Kim, H.; Choi, Y. Unraveling the intricacies of osteoclast differentiation and maturation: insight into novel therapeutic strategies for bone-destructive diseases. Exp Mol Med 2024, 56, 264–272. [Google Scholar] [CrossRef]
- Han, S.Y.; Lee, N.K.; Kim, K.H.; Jang, I.W.; Yim, M.; Kim, J.H.; Lee, W.J.; Lee, S.Y. Transcriptional induction of cyclooxygenase-2 in osteoclast precursors is involved in RANKL-induced osteoclastogenesis. Blood 2005, 106, 1240–1245. [Google Scholar] [CrossRef]
- Boyce, B.F. Advances in the regulation of osteoclasts and osteoclast functions. J Dent Res 2013, 92, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Rolph, D.; Das, H. Transcriptional Regulation of Osteoclastogenesis: The Emerging Role of KLF2. Front Immunol 2020, 11, 937. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Luo, F.; Du, X.; Xian, F.; Li, X. The immune cells in modulating osteoclast formation and bone metabolism. Int Immunopharmacol 2024, 133, 112151. [Google Scholar] [CrossRef]
- Ron, D.; Messing, R.O. Signaling pathways mediating alcohol effects. Curr Top Behav Neurosci 2013, 13, 87–126. [Google Scholar] [CrossRef]
- Navarro, S.; Louache, F.; Debili, N.; Vainchenker, W.; Doly, J. Autocrine regulation of terminal differentiation by interleukin-6 in the pluripotent KU812 cell line. Biochem Biophys Res Commun 1990, 169, 184–191. [Google Scholar] [CrossRef]
- Kopesky, P.; Tiedemann, K.; Alkekhia, D.; Zechner, C.; Millard, B.; Schoeberl, B.; Komarova, S.V. Autocrine signaling is a key regulatory element during osteoclastogenesis. Biol Open 2014, 3, 767–776. [Google Scholar] [CrossRef]
- Weivoda, M.M.; Bradley, E.W. Macrophages and Bone Remodeling. J Bone Miner Res 2023, 38, 359–369. [Google Scholar] [CrossRef]
- Batoon, L.; Millard, S.M.; Raggatt, L.J.; Pettit, A.R. Osteomacs and Bone Regeneration. Curr Osteoporos Rep 2017, 15, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Wakkach, A.; Rouleau, M.; Blin-Wakkach, C. Osteoimmune Interactions in Inflammatory Bowel Disease: Central Role of Bone Marrow Th17 TNFalpha Cells in Osteoclastogenesis. Front Immunol 2015, 6, 640. [Google Scholar] [CrossRef]
- Okamoto, K. Crosstalk between bone and the immune system. J Bone Miner Metab 2024, 42, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Odhiambo, W.O.; Qin, M.; To, T.T.; Brewer, G.M.; Kheshvadjian, A.R.; Cheng, C.; Agak, G.W. Analysis of intracellular communication reveals consistent gene changes associated with early-stage acne skin. Cell Commun Signal 2024, 22, 400. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, M.B.; Kjolby, M.; Gunnersen, S.; Larsen, J.V.; Palmfeldt, J.; Falk, E.; Nykjaer, A.; Bentzon, J.F. Targeting sortilin in immune cells reduces proinflammatory cytokines and atherosclerosis. J Clin Invest 2014, 124, 5317–5322. [Google Scholar] [CrossRef]
- Xia, Y.; Inoue, K.; Du, Y.; Baker, S.J.; Reddy, E.P.; Greenblatt, M.B.; Zhao, B. TGFbeta reprograms TNF stimulation of macrophages towards a non-canonical pathway driving inflammatory osteoclastogenesis. Nat Commun 2022, 13, 3920. [Google Scholar] [CrossRef] [PubMed]
- Jo, Y.; Sim, H.I.; Yun, B.; Park, Y.; Jin, H.S. Revisiting T-cell adhesion molecules as potential targets for cancer immunotherapy: CD226 and CD2. Exp Mol Med 2024, 56, 2113–2126. [Google Scholar] [CrossRef]
- Berrocal-Rubio, M.A.; Pawer, Y.D.J.; Dinevska, M.; De Paoli-Iseppi, R.; Widodo, S.S.; Gleeson, J.; Rajab, N.; De Nardo, W.; Hallab, J.; Li, A.; et al. Discovery of NRG1-VII: the myeloid-derived class of NRG1. BMC Genomics 2024, 25, 814. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Nishida, S.; Elalieh, H.Z.; Long, R.K.; Halloran, B.P.; Bikle, D.D. Role of IGF-I signaling in regulating osteoclastogenesis. J Bone Miner Res 2006, 21, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, J.; Takegahara, N.; Kim, H.; Lee, S.H.; Sato, K.; Yamagishi, S.; Choi, Y. Flrt2 is involved in fine-tuning of osteoclast multinucleation. BMB Rep 2019, 52, 514–519. [Google Scholar] [CrossRef]
- Chang, Y.; Hsiao, Y.M.; Hu, C.C.; Chang, C.H.; Li, C.Y.; Ueng, S.W.N.; Chen, M.F. Synovial Fluid Interleukin-16 Contributes to Osteoclast Activation and Bone Loss through the JNK/NFATc1 Signaling Cascade in Patients with Periprosthetic Joint Infection. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Yu, J.; Canalis, E. Notch and the regulation of osteoclast differentiation and function. Bone 2020, 138, 115474. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



