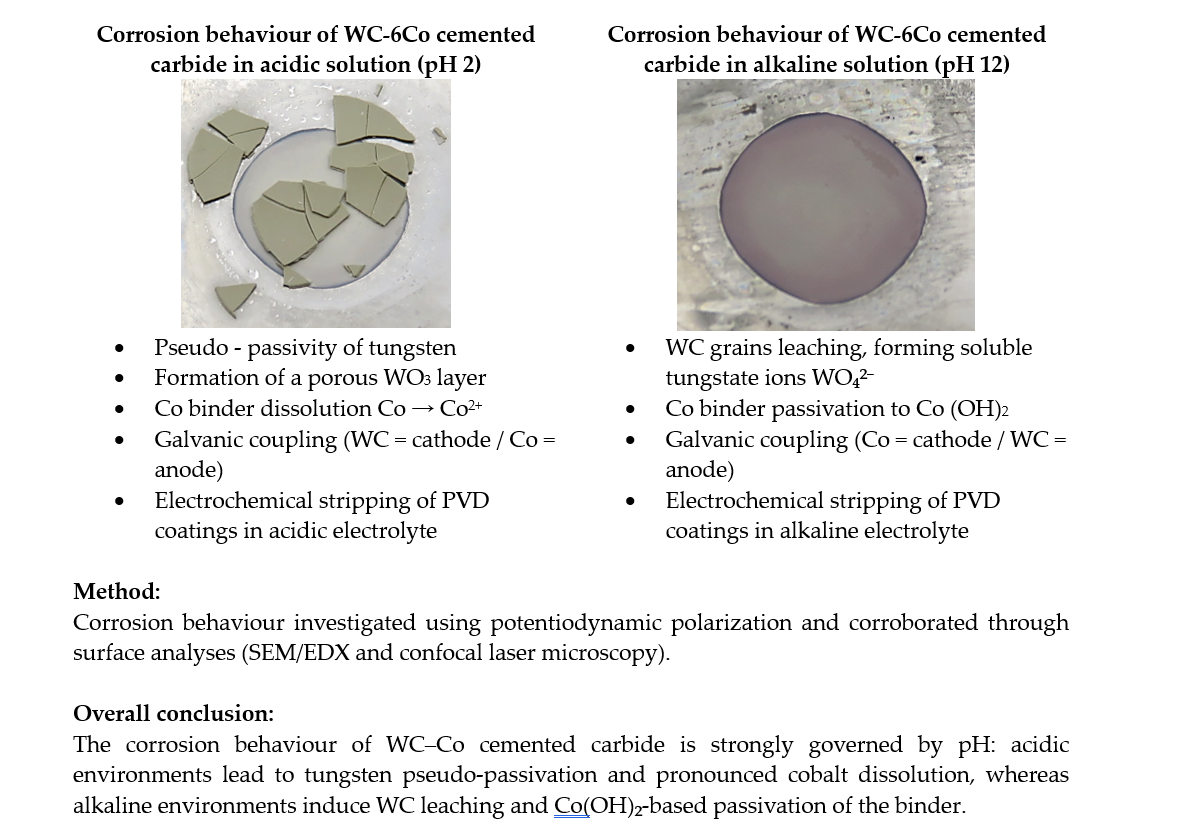
4.1. Electrochemical Investigation of Alkaline and Acidic Electrolytes Using a Pt Working Electrode
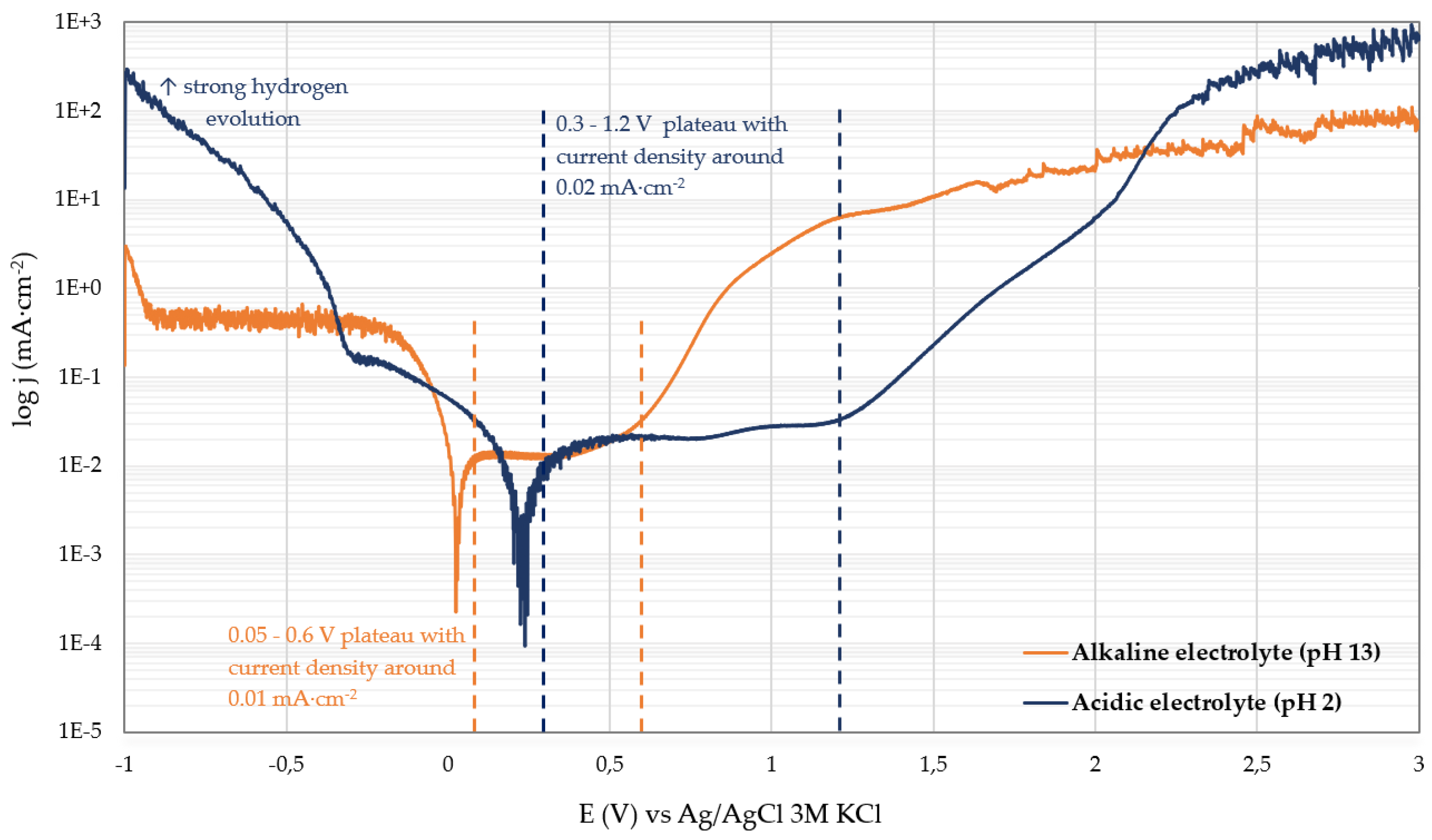
To determine the electrochemical stability windows of both electrolytes and to identify potential ranges that should be avoided during polarization measurements on WC–6Co, potentiodynamic polarization was first performed on an inert platinum working electrode (Pt). Potentiodynamic polarization measurements were conducted from −1 V to +3 V at a scan rate of 1 mV·s⁻¹ under stirred conditions in a 1 L corrosion cell.
Figure 2 shows the resulting polarization curves recorded in the alkaline (pH 13) and acidic (pH 2) electrolytes.
In the cathodic region (E <
Ecorr), the acidic electrolyte exhibits significantly higher current densities than the alkaline electrolyte. This behaviour is characteristic of intensified hydrogen evolution, which is strongly favoured at low pH. The high cathodic currents therefore indicate accelerated proton reduction (2H⁺ + 2e⁻ → H₂). Platinum is an excellent hydrogen evolution catalyst, which further amplifies cathodic activity in acidic media. In contrast, the alkaline electrolyte shows much lower cathodic currents, consistent with the slower hydrogen evolution kinetics associated with water reduction (2H₂O + 2e⁻ → H₂ + 2OH⁻), a well-established effect reflecting the two-orders-of-magnitude decrease in HER rates on Pt in alkaline compared to acidic conditions [
18].
In the anodic region, the acidic electrolyte displays a relatively broad passive-like plateau between approximately 0.3 and 1.2 V with current densities around 0.02 mA/cm². Beyond ~2.3 V, a steep rise in current density occurs, corresponding to oxygen evolution and the onset of electrolyte decomposition. The alkaline electrolyte exhibits a narrower passive region between ~0.05 and 0.6 V at approximately 0.01 mA/cm². At higher potentials, a gradual increase in anodic current is observed, but the overall current remains lower than in the acidic medium. This enhanced stability may be attributed to the presence of surfactants and phosphate-based inhibitors, which adsorb onto the platinum surface, suppressing charge transfer and mitigating oxygen evolution.
Based on the stability-window analysis obtained from the Pt electrode, the potential range for the WC–6Co measurements was adjusted to start at OCP − 0.2 V to avoid excessive hydrogen evolution in the acidic electrolyte and prevent unintended surface alterations.
The upper limit was set to +3 V, as the focus of these measurements was to examine anodic processes relevant to electrochemical stripping such as passivation and transpassive dissolution. In this context, precise corrosion-rate determination was of secondary importance, since electrochemical stripping involves the application of an external potential rather than free-corrosion conditions.
4.2. Corrosion Behaviour of WC-6Co in Alkaline and Acidic Electrolytes
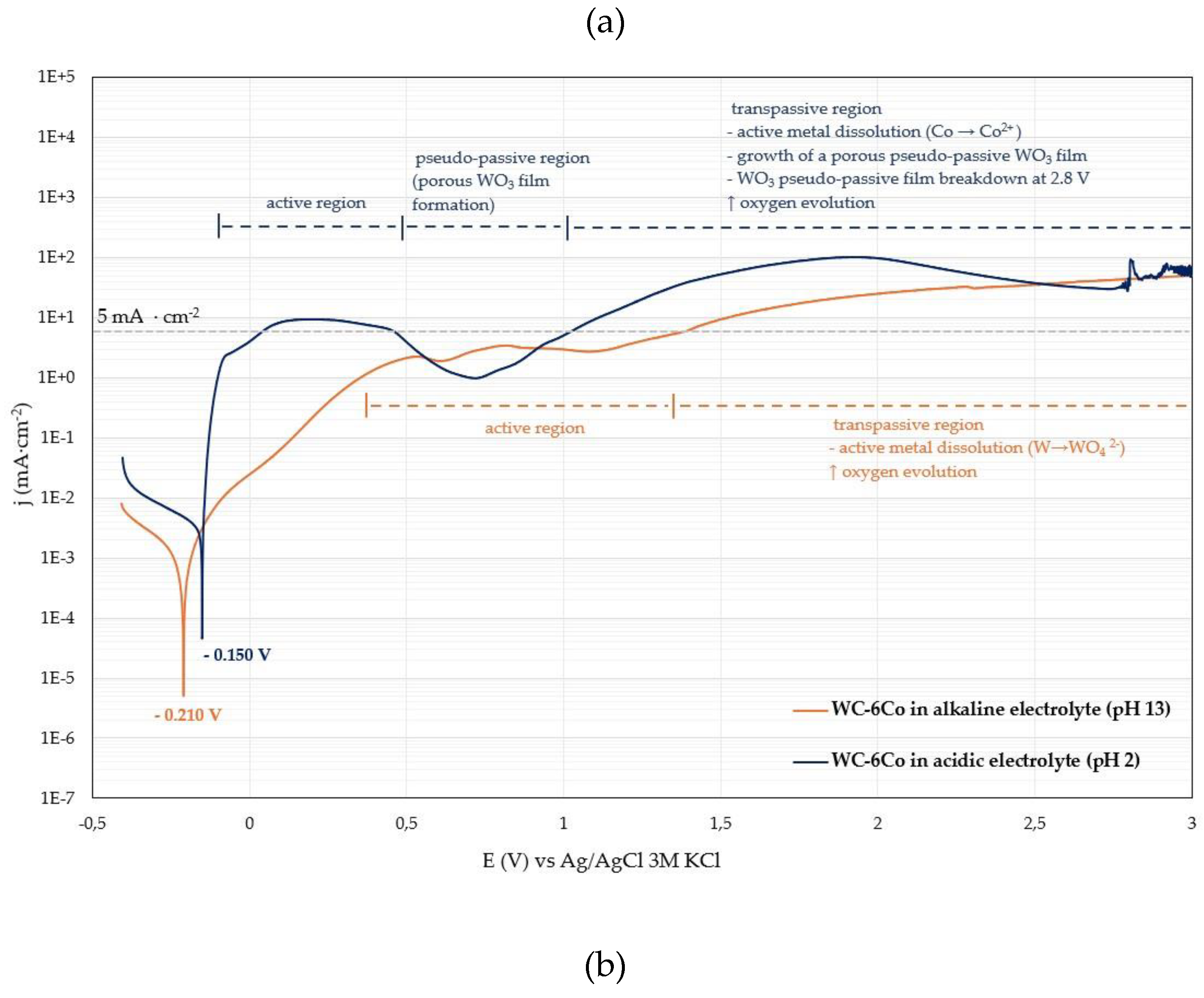
The corrosion behavior of WC-6Co cemented carbide in the alkaline (pH 13) and acidic (pH 2) electrolytes used for electrochemical stripping was investigated using potentiodynamic polarization. The potential was scanned from OCP – 0.2 V to +3 V at 1 mV·s⁻¹, a range selected based on the stability-window analysis obtained from the Pt electrode. All measurements were performed in a flat corrosion cell containing 100 mL of electrolyte, under unstirred conditions at 20 °C. Following the electrochemical tests, surface analyses were carried out to compare corrosion mechanisms in two electrolytes with different pH values. The resulting polarisation curves are shown in
Figure 3 in (a) logarithmic and (b) linear form to facilitate comparison of cathodic and anodic reaction regions.
To complement the qualitative evaluation of the polarization curves, Tafel parameters derived from the near-OCP region are summarized in
Table 1. Although these values provide a useful indication of the relative corrosion tendency close to corrosion potential, they are less relevant for interpreting the corrosion mechanisms that occur at the higher anodic potentials typical of electrochemical stripping, where metal dissolution, pseudo-passivation and oxide-film breakdown govern material removal.
The corrosion current density (jcorr) is higher in the acidic electrolyte than in the alkaline solution, which indicates a greater tendency for dissolution near the corrosion potential. This trend is consistent with the significantly higher anodic current densities observed in the active dissolution region of the acidic electrolyte. Although the corrosion rate is temporarily attenuated at intermediate potentials (at ~0.65 to 0.85 V) due to the formation of a WO₃-based pseudo-passive layer, the porous and mechanically unstable nature of this oxide limits its protective effect. It should also be noted that the electrolytes used for electrochemical stripping do not contain oxidizing agents and primarily serve as highly conductive transport media at the respective pH values. Consequently, the corrosion behaviour relevant to electrochemical stripping is dominated by the mechanisms occurring at higher anodic potentials - such as leaching, passivation, oxide growth and subsequent oxide – film breakdown or partial film flaking - so the Tafel parameters serve only as supportive indicators rather than primary descriptors.
In the acidic electrolyte (pH 2), WC-6Co exhibits a pseudo-passive plateau between approximately 0.65 to 0.85 V vs Ag/AgCl, within gas evolution ceases. Although the current density appears to stabilize within this potential range, the measured values (~0.6–0.7 mA cm⁻²) remain far higher than typical passive current densities (~10 µA cm⁻² [
15]). indicating that the material does not achieve true passivation. This behaviour is therefore classified as pseudo-passivity and is associated with the onset of a porous WO₃ layer formation, on the tungsten rich surface. The growth of the passive film follows the mechanism described by Sato [
11], involving place-exchange and/or field-assisted ionic migration. Oxygen ions migrate toward the metal/oxide interface to form the inner oxide layer, while metal ions move toward the oxide/electrolyte interface resulting in the formation of an outer oxide layer that may incorporate electrolyte species.
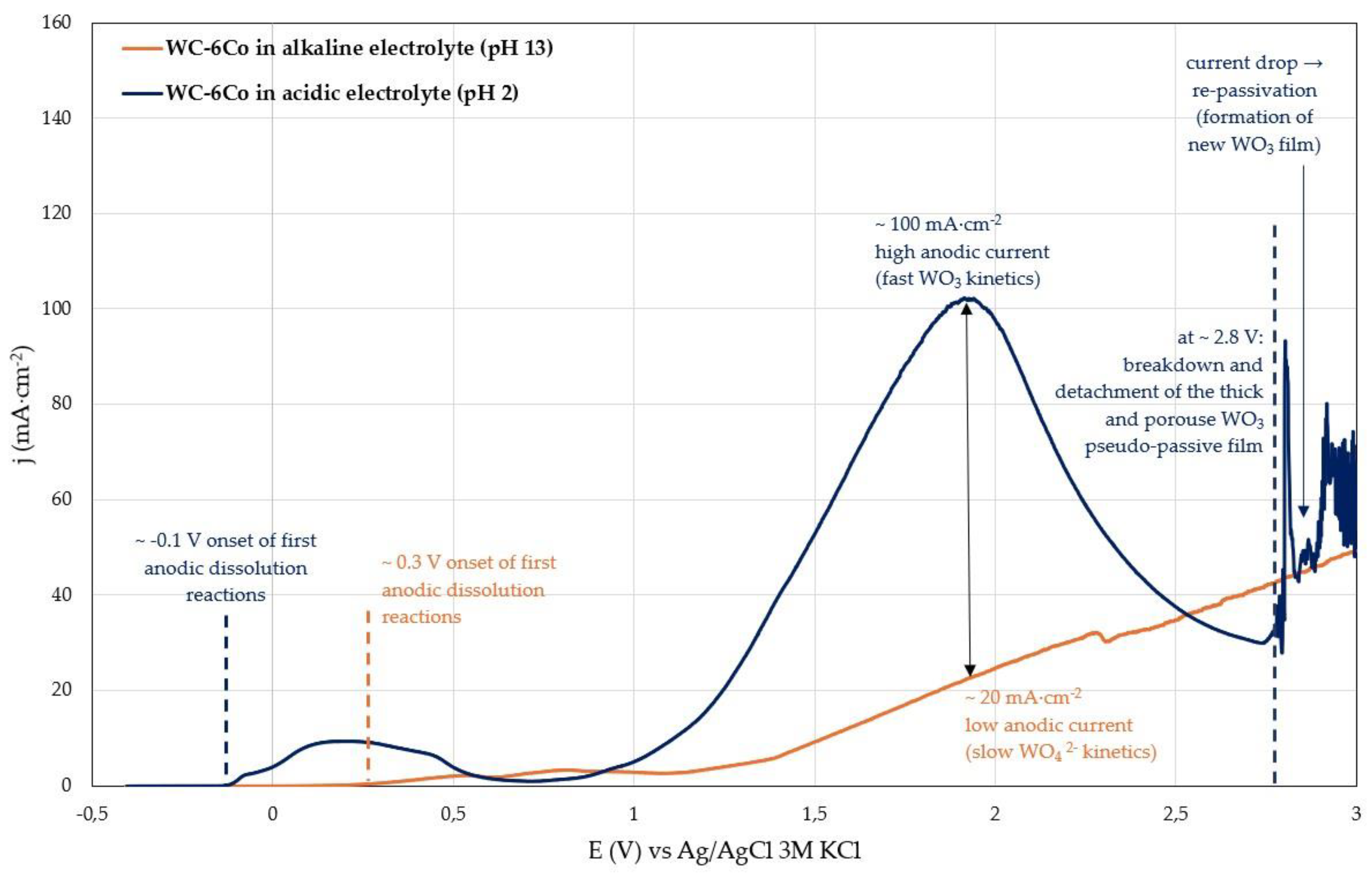
As the anodic potential increases beyond approx. 0.85 V, the current density rises again, marking the onset of transpassive behaviour. In this regime, continued tungsten oxidation thickens the WO₃ layer, which becomes increasingly porous and permeable to ionic transport. Consequently, the anodic current density increases further as shown in
Figure 3b. At potentials around 1.9 V, a temporary decrease in the current density is observed, which can be attributed to short-term densification or local stabilization of thick WO
3 layer. At approx. 2.8 V, the oxide undergoes breakdown, accompanied by fracturing and detachment of the thick and porous pseudo-passive layer. This is reflected in a sharp current spike from ~25 mA·cm⁻² to ~95 mA·cm⁻², indicating the exposure of fresh substrate material. The subsequent decrease in current density corresponds to the formation of a new oxide layer, after which the cycle of oxide growth and breakdown may repeat. This sequence of porous oxide formation, transient stabilisation, and mechanical failure is characteristic of transpassive behaviour and highlights the limited protective capability of the WO₃ layer under acidic conditions.
In contrast, the alkaline electrolyte does not exhibit a pronounced pseudo-passive region. Tungsten is thermodynamically unstable under alkaline conditions and dissolves as soluble tungstate ions (WO₄²⁻), while cobalt forms a thin Co(OH)₂ surface layer that provides partial passivation [
6]. This behaviour is reflected in the anodic branch, where the current density increases steadily from the onset of anodic dissolution (~0.3 V) into the transpassive region, indicating predominantly active metal dissolution without the establishment of a stable protective surface film. Transpassivation, as described by Song [
13], is characterised by a sustained increase in anodic current density and is generally considered a form of corrosion damage. Compared to the acidic system, in the alkaline electrolyte WC-6Co material exhibits transpassive behaviour at comparatively lower anodic potentials, but with much smaller current densities due to slower WO
42- formation kinetics.
A clear distinction between both electrolytes is reflected in the anodic current densities relative to the commonly referenced 5 mA·cm⁻² threshold defined in ASTM G61 [
19] as an indicator of significant anodic activity: the acidic electrolyte exceeds this value already within the active dissolution region, whereas the alkaline electrolyte remains far below it, highlighting the fundamentally faster WO₃-film growth kinetics in acidic media compared to the slower WO₄²⁻-formation kinetics in alkaline solution.
These differences arise from the microstructural composition of the WC–6Co substrate. Due to the high WC content (94 wt%) and the relatively low cobalt fraction (6 wt%), the electrochemical response is governed predominantly by the WC phase. In the acidic electrolyte, the oxidation of WC produces a WO₃-based pseudo-passive layer, whereas in alkaline media tungsten dissolves directly as WO₄²⁻. Although cobalt can form a passive hydroxide film in alkaline conditions, its low surface fraction is insufficient to stabilize the overall surface. As a result, the acidic electrolyte exhibits pseudo-passivity followed by transpassive breakdown, while the alkaline electrolyte shows a direct transition from active dissolution to transpassive behaviour without the formation of a protective film.
4.3. Surface Characterization of WC-6Co After Potentiodynamic Polarisation in Acidic Electrolyte
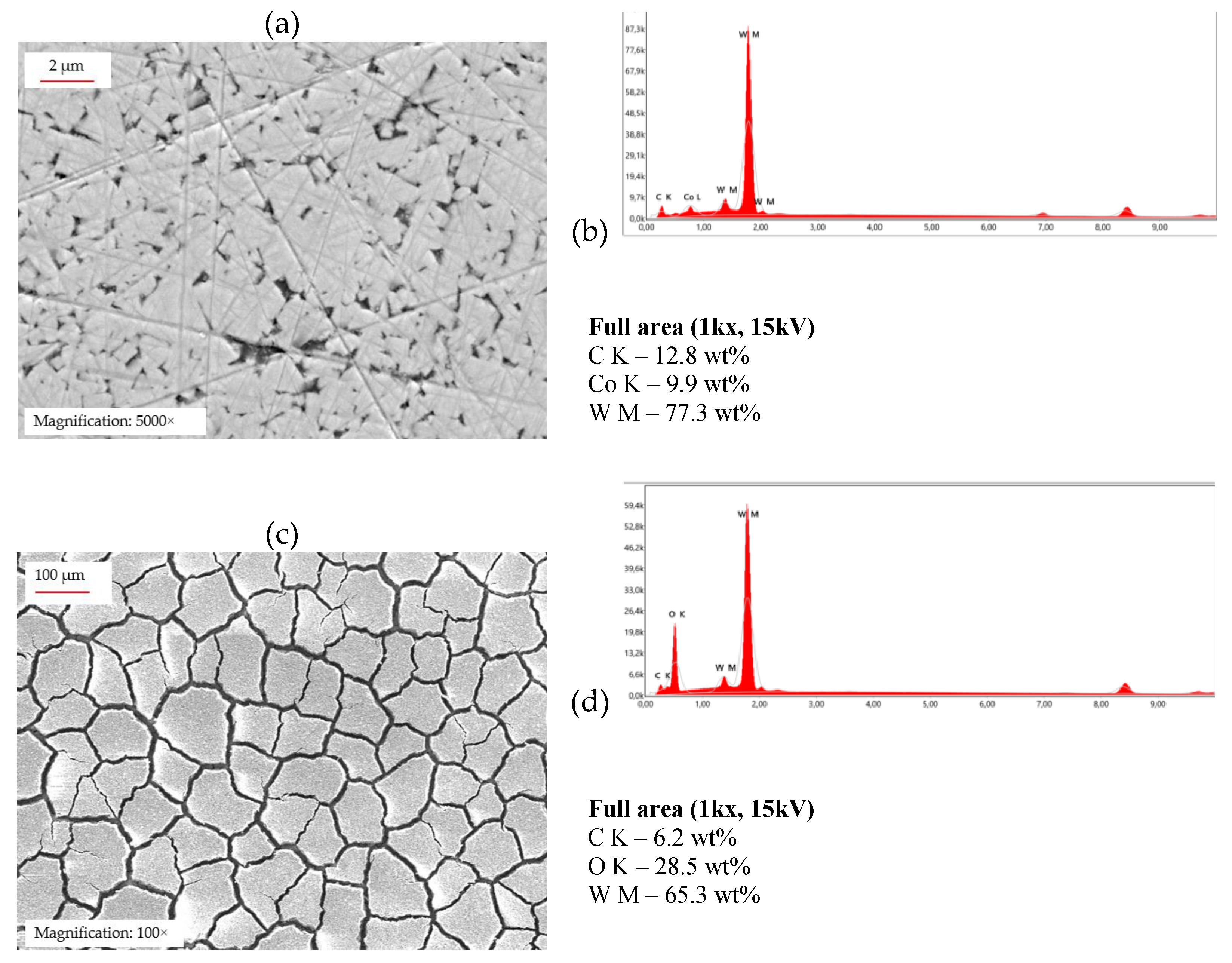
Figure 4 shows the WC-6Co sample after potentiodynamic polarization testing in the acidic electrolyte (pH 2), revealing a separated oxide-layer exposed on the surface. A thick yellowish - grey primary oxide layer is loosely attached to the surface, while a second intact oxide layer is visible beneath it. SEM micrograph at 100x magnification (
Figure 5c) reveal the intact underlaying oxide layer after removal of the fractured layer, showing a porous surface morphology resembling dried, cracked soil. The corresponding EDX spectrum (
Figure 5d) indicates an elevated oxygen and tungsten content, whereas cobalt is not detected. For comparison, SEM and EDX analyses of the untreated surface prior to testing are shown in
Figure 5a and
Figure 5b.
Surface profile measurements obtained using laser microscope reveal a highly rough surface with an average profile depth of approximately 3.4 µm. Upon higher magnification, individual depressions of up to 6 µm in depth were observed. These depressions confirm the highly porous and non-uniform nature of the WO₃ layer, consistent with the cracked and irregular oxide morphology seen in the SEM micrograph in
Figure 5c. To estimate the possible thickness of the WO₃ oxide layer, an additional surface profile measurement was carried out on a WC-6Co sample polarized at a higher scan rate of 5 mV·s⁻¹. In contrast to the polarization curve recorded at 1 mV·s⁻¹, the curve obtained at 5 mV·s⁻¹ did not exhibit a pronounced anodic current peak, indicating that the initially formed oxide layer had not yet detached (data not shown). This behavior can be attributed to the significantly shorter exposure time at higher scan rates, resulting in reduced oxidation–thinning cycles and delayed film fracture. The corresponding surface profile indicates a WO₃ film thickness of approximately 44 µm, demonstrating substantial anodic oxidation of tungsten under these conditions.
The SEM micrograph presented in
Figure 6 further confirms the brittle and porous nature of the oxide layer, revealing extensive cracking and locally detached fragments. These features are consistent with the electrochemical response shown in
Figure 3, where high anodic currents persist despite continuous oxide growth, indicating that the increasingly porous and ion-permeable WO₃ film formed under prolonged anodic polarization is mechanically unstable and prone to cracking.
Since the oxide layer must be removed prior to recoating, geometric change to the substrate must be considered. To assess the depth of uniform corrosion after removal of the oxide layer, the sample was subjected to ultrasonic treatment in ethanol within a beaker. Subsequent surface profile measurements reveal a uniform material loss with an average surface abrasion of approximately 8 µm.
The FIB (Focused-Ion-Beam) cross-section shown in
Figure 7b, obtained from a cemented carbide cutting tool after industrial electrochemical stripping process in an acidic electrolyte (pH 2) supports the laboratory findings. The cross-section reveals loose WC grains and loss of the cobalt binder matrix in the near-surface region. To further corroborate this behaviour, an additional SEM top-view micrograph of the same tool was analysed (
Figure 7a). The surface image clearly shows exposed WC grains and the absence of the cobalt matrix.
4.4. Surface Characterization of WC-6Co After Potentiodynamic Polarisation in Alkaline Electrolyte
Surface profile measurements performed after the potentiodynamic polarization in the alkaline electrolyte (pH 13) revealed neither the formation of a thick oxide layer nor any evidence of oxide-film breakdown. The average material removal from the surface was approx. 0.3 µm. Localized profilometric analysis of smaller areas revealed depressions up to 3 µm in depth (profilometric data not shown). However, pronounced leaching of WC particles resulted in significant surface roughening, which in industrial applications, necessitates subsequent micro-blasting to remove the remaining loose, soft cobalt matrix. This treatment does not increase the actual material removal depth but eliminates the mechanically weakened cobalt matrix until the original carbide structure is reached.
As shown in
Figure 8, the exposed surface exhibited a faint pinkish coloration, which is characteristic of Co(OH)₂ and indicates cobalt passivation under alkaline conditions. This observation is fully consistent with the Pourbaix predictions for the Co–H₂O system [
6], where cobalt stabilizes thermodynamically as Co(OH)₂ in high-pH environments.
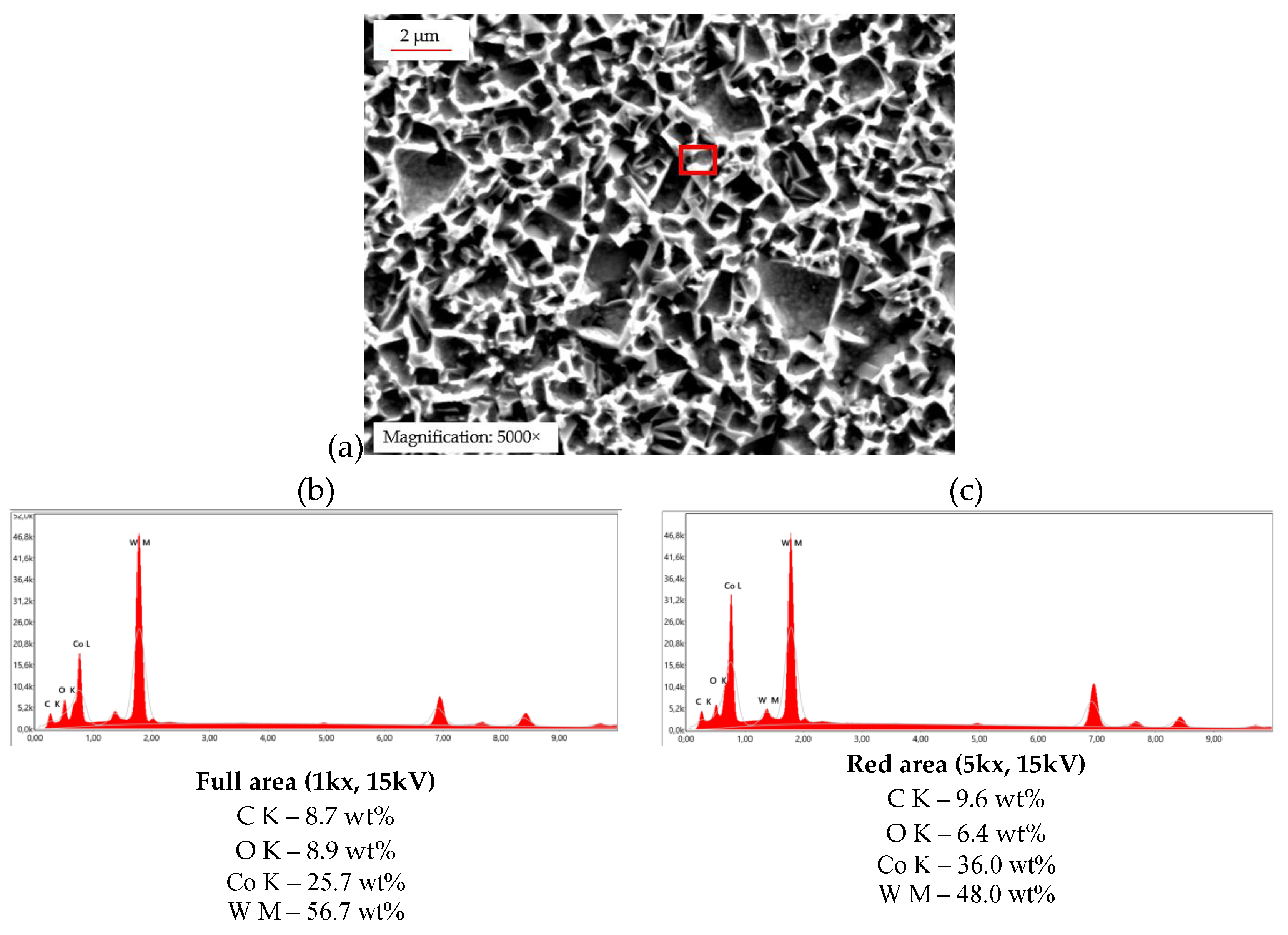
SEM micrograph and EDX spectrum analyses (
Figure 9) clearly demonstrate leached WC grains and an oxidized Co-matrix (highlighted in red), which aligns with the predictions from the Pourbaix diagrams [
6].
It should be noted that the parameters of the potentiodynamic polarization process differ significantly from those used during industrial electrochemical stripping, where much higher voltages are applied for only a few seconds or minutes. In contrast, during the laboratory polarisation measurement, the samples were exposed to lower potentials but for significantly longer duration, which resulted in more pronounced surface damage. Nonetheless, this measurement provides valuable insights into the corrosion mechanisms occurring during the stripping process.