Submitted:
12 March 2026
Posted:
13 March 2026
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
1.1. Clinical Burden and Unmet Needs
1.2. Clinical Definition, Phenotypes, and Diagnostic Landscape of Dpn
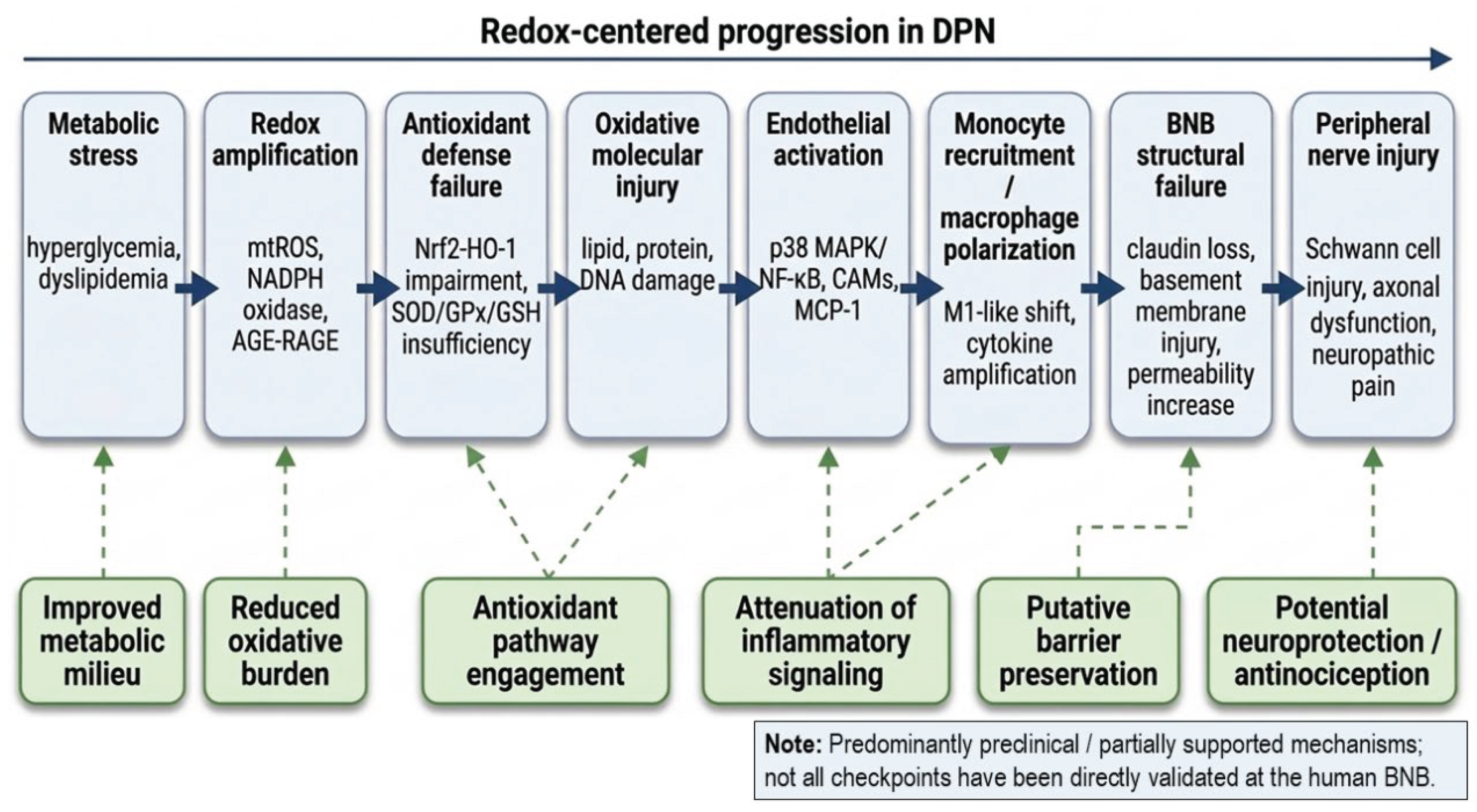
1.3. From Metabolic Stress to Neurovascular-Immune Dysfunction: A Mechanistic Rationale
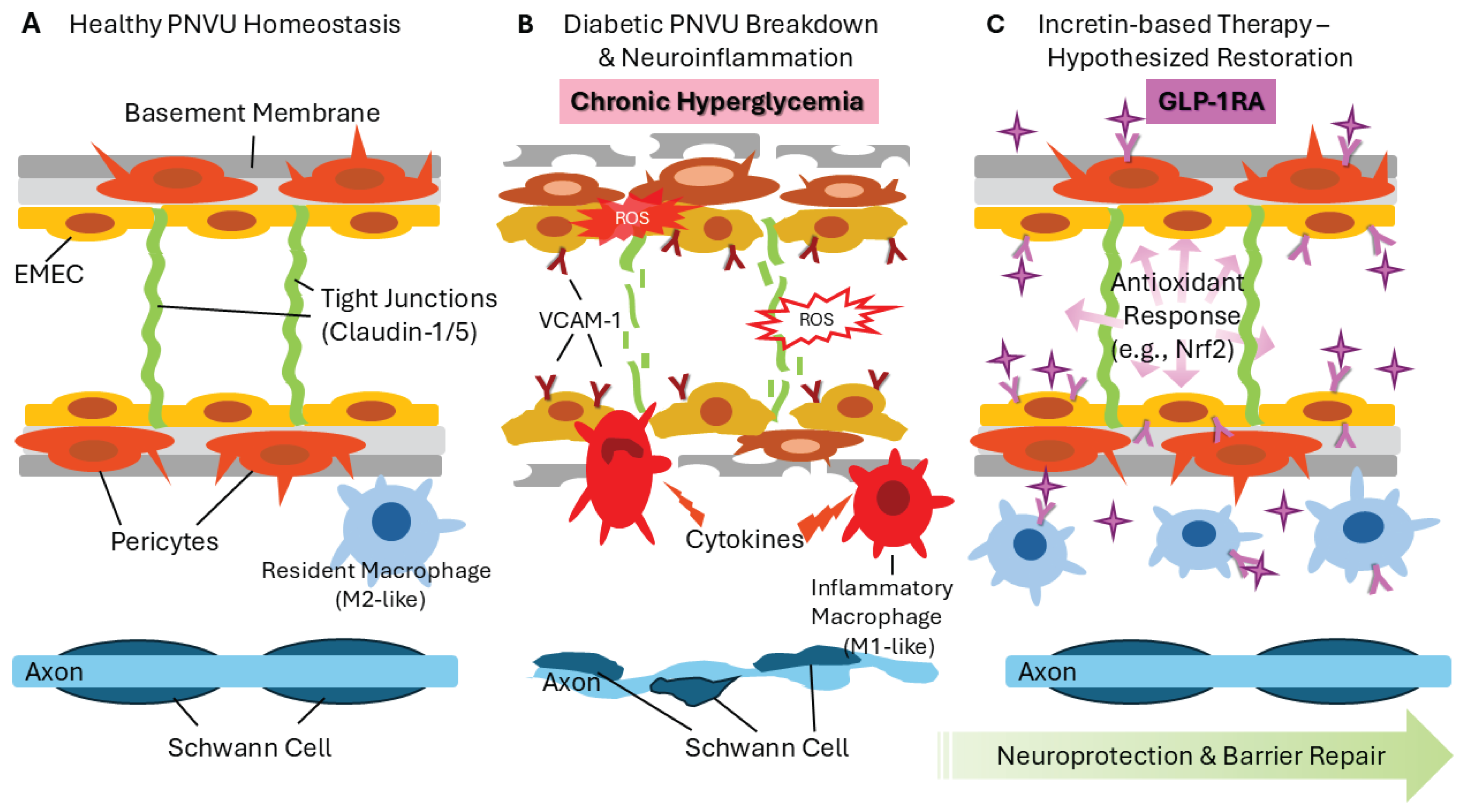
2. The Pnvu: Concept and Overview
2.1. The Pnvu as A Functional Syncytium
2.2. Anatomical and Molecular Architecture of the Bnb
2.3. Comparative Barrier Biology: Bnb Versus Bbb
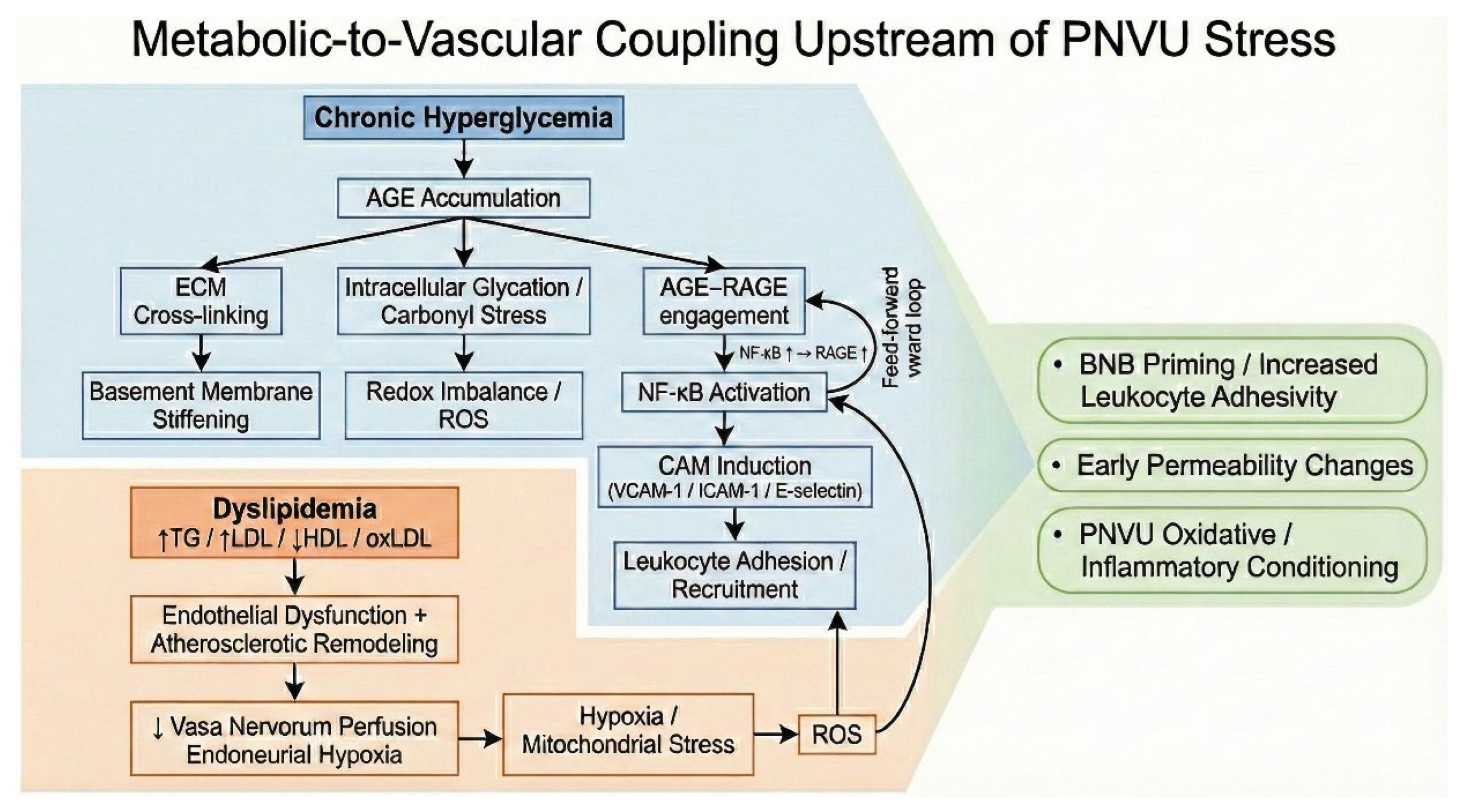
3. Redox Imbalance in the Pnvu: Oxidative Damage, Antioxidant Defense Failure, and Metabolic-to-Vascular Coupling
3.1. The Landscape of Oxidative Damage in the Pnvu
3.2. Metabolic Exhaustion of Endogenous Antioxidant Defenses: Sod, Ho-1, and Impaired Nrf2 Signaling
3.3. Metabolic-to-Vascular Coupling: Age-Rage Signaling and Dyslipidemia as Upstream Drivers of Pnvu Stress
4. Endothelial Activation and the Breach of Immune Privilege
4.1. Endothelial Dysfunction as A Gateway to Bnb Breakdown
4.2. The Nf-Κb Pathway: Translating Metabolic Stress into Inflammation
4.3. The Adhesion Molecule Cascade: Vcam-1, Icam-1, and Selectins
4.4. Mcp-1/ccl2 and the Recruitment of Endoneurial Macrophages
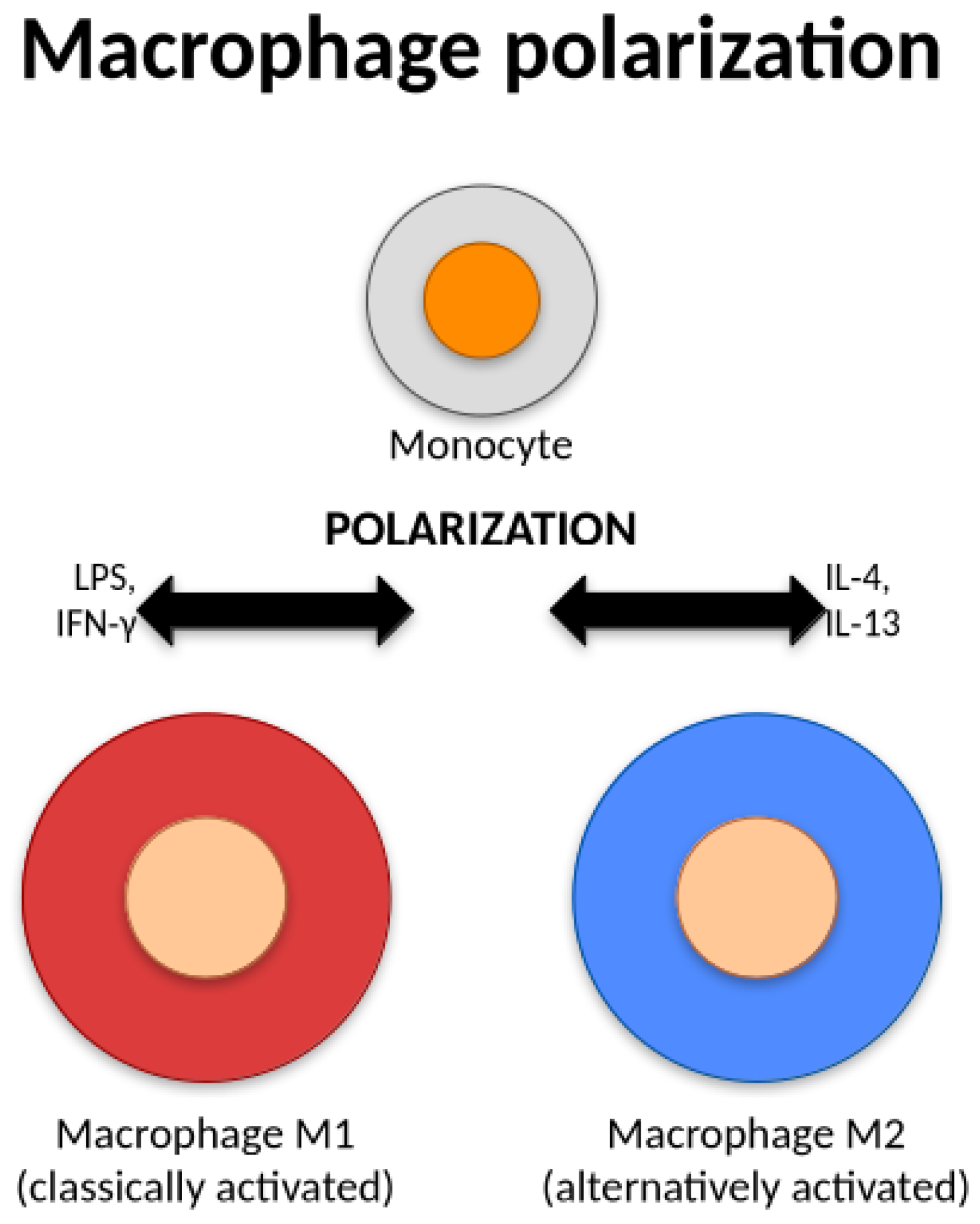
5. Chronic Neuroinflammation and Macrophage Polarization
5.1. Resident Vs. Recruited Macrophages: The Sentinel Shift
5.2. Macrophage Polarization: the M1/m2 Paradigm in the Diabetic Nerve
5.3. The Cytokine Storm and Its Impact on Barrier Integrity
6. Structural Collapse and Junctional Disassembly
6.1. The Molecular Gatekeepers: Claudin-1 and Claudin-5
6.2. Junctional Disassembly and Protein Downregulation
6.3. Assessing Bnb Integrity: A Methodological Toolbox (Structural, Functional, and Cellular Readouts)
7. Incretin-Based Therapies in Dpn and the Pnvu: Class Rationale, Semaglutide, and Translational Opportunities
7.1. Beyond Semaglutide: The Incretin Landscape (Glp-1ras Vs. Dpp-4is; Emerging Multi-Agonists)
7.2. Glp-1 Receptor Agonists, with Semaglutide as A Representative Agent: Pharmacological Properties and Antinociceptive Mechanisms
7.3. Counteracting Metabolic Exhaustion: The Nrf2/antioxidant Axis
7.4. Attenuation of P38 Mapk/nf-Κb Signaling and Neuroinflammation
7.5. Putative Structural Stabilization and Functional Preservation of the Bnb
8. Conclusions
9. Future Perspectives
9.1. Limitations and Unresolved Questions
9.2. Key Research Questions
- Is any neuroprotection independent of metabolic improvement?
- Which PNVU compartment is the primary target (endothelium, Schwann cells, immune cells)?
- Which outcomes provide the most reliable animal-to-human bridges (NCV, QST, IENFD, MR neurography, circulating endothelial/immune biomarkers)?
- Which BNB/PNVU biomarker panels are feasible, repeatable, and responsive in clinical trials?
- How should redox-focused readouts (e.g., lipid peroxidation, oxidative DNA damage, antioxidant pathway engagement) be incorporated alongside barrier-focused biomarkers?
- Do DPP-4is and emerging multi-agonists (dual/triple agonists) provide additive or distinct benefits versus GLP-1RAs for neuropathy outcomes [127]?
- What duration and dosing are required for structural recovery (e.g., barrier integrity, axonal regeneration)?
- Are there
- patient subgroups (phenotypes) most likely to benefit?
9.3. Translational Roadmap for Clinical Studies
9.4. Closing Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ong, K.L.; Stafford, L.K.; McLaughlin, S.A.; Boyko, E.J.; Vollset, S.E.; Smith, A.E.; Dalton, B.E.; Duprey, J.; Cruz, J.A.; Hagins, H.; et al. Global, regional, and national burden of diabetes from 1990 to 2021, with projections of prevalence to 2050: A systematic analysis for the Global Burden of Disease Study 2021. Lancet 2023, 402, 203–234. [Google Scholar] [CrossRef]
- A Elafros, M.; Andersen, H.; Bennett, D.L.; Savelieff, M.G.; Viswanathan, V.; Callaghan, B.C.; Feldman, E.L. Towards prevention of diabetic peripheral neuropathy: Clinical presentation, pathogenesis, and new treatments. Lancet Neurol. 2022, 21, 922–936. [Google Scholar] [CrossRef]
- Feldman, E.L.; Callaghan, B.C.; Pop-Busui, R.; Zochodne, D.W.; Wright, D.E.; Bennett, D.L.; Bril, V.; Russell, J.W.; Viswanathan, V. Diabetic neuropathy. Nat. Rev. Dis. Prim. 2019, 5, 41. [Google Scholar] [CrossRef]
- Sun, J.; Wang, Y.; Zhang, X.; Zhu, S.; He, H. Prevalence of peripheral neuropathy in patients with diabetes: A systematic review and meta-analysis. Prim. Care Diabetes 2020, 14, 435–444. [Google Scholar] [CrossRef]
- Zhou, P.; Zhou, J.S.; Li, J.J.; Qin, L.; Hu, W.F.; Zhang, X.Y.; Wang, J.X.; Shi, Z. Prevalence and risk factors for painful diabetic peripheral neuropathy: A systematic review and meta-analysis. Front. Neurol. 2025, 16, 1564867. [Google Scholar] [CrossRef] [PubMed]
- Perveen, W.; Ahsan, H.; Fayyaz, S.; Zaif, A.; Paracha, M.A.; Nuhmani, S.; Khan, M.; Alghadir, A.H. Prevalence of peripheral neuropathy, amputation, and quality of life in patients with diabetes mellitus. Sci. Rep. 2024, 14, 14430. [Google Scholar] [CrossRef] [PubMed]
- Petropoulos, I.N.; Ponirakis, G.; Khan, A.; Almuhannadi, H.; Gad, H.; Malik, R.A. Diagnosing Diabetic Neuropathy: Something Old, Something New. Diabetes Metab. J. 2018, 42, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Selvarajah, D.; Kar, D.; Khunti, K.; Davies, M.J.; Scott, A.R.; Walker, J.; Tesfaye, S. Diabetic peripheral neuropathy: Advances in diagnosis and strategies for screening and early intervention. Lancet Diabetes Endocrinol. 2019, 7, 938–948. [Google Scholar] [CrossRef]
- Tomic, D.; Shaw, J.E.; Magliano, D.J. The burden and risks of emerging complications of diabetes mellitus. Nat. Rev. Endocrinol. 2022, 18, 525–539. [Google Scholar] [CrossRef]
- Shy, M.; Frohman, E.; So, Y.; Arezzo, J.; Cornblath, D.; Giuliani, M.; Kincaid, J.; Ochoa, J.; Parry, G.; Weimer, L. Quantitative sensory testing. Neurology 2003, 60, 898–904. [Google Scholar] [CrossRef]
- Reitz, M.-C.; Hrncic, D.; Treede, R.-D.; Caspani, O. A comparative behavioural study of mechanical hypersensitivity in 2 pain models in rats and humans. Pain 2016, 157, 1248–1258. [Google Scholar] [CrossRef]
- Pop-Busui, R.; Boulton, A.J.; Feldman, E.L.; Bril, V.; Freeman, R.; Malik, R.A.; Sosenko, J.M.; Ziegler, D. Diabetic Neuropathy: A Position Statement by the American Diabetes Association. Diabetes Care 2016, 40, 136–154. [Google Scholar] [CrossRef]
- American Diabetes Association Professional Practice Committee; ElSayed, N.A.; McCoy, R.G.; Aleppo, G.; Balapattabi, K.; Beverly, E.A.; Early, K.B.; Bruemmer, D.; Callaghan, B.C.; Echouffo-Tcheugui, J.B.; et al. 12. Retinopathy, Neuropathy, and Foot Care: Standards of Care in Diabetes—2025. Diabetes Care 2024, 48, S252–S265. [Google Scholar] [CrossRef]
- Tesfaye, S.; Boulton, A.J.M.; Dyck, P.J.; Freeman, R.; Horowitz, M.; Kempler, P.; Lauria, G.; Malik, R.A.; Spallone, V.; Vinik, A.; et al. Diabetic Neuropathies: Update on Definitions, Diagnostic Criteria, Estimation of Severity, and Treatments. Diabetes Care 2010, 33, 2285–2293. [Google Scholar] [CrossRef] [PubMed]
- Bril, V.; Tomioka, S.; Buchanan, R.A.; Perkins, B.A. the mTCNS Study Group Reliability and validity of the modified Toronto Clinical Neuropathy Score in diabetic sensorimotor polyneuropathy. Diabet. Med. 2009, 26, 240–246. [Google Scholar] [CrossRef]
- Bril, V.; Perkins, B.A. Validation of the Toronto Clinical Scoring System for Diabetic Polyneuropathy. Diabetes Care 2002, 25, 2048–2052. [Google Scholar] [CrossRef]
- Bouhassira, D.; Attal, N.; Alchaar, H.; Boureau, F.; Brochet, B.; Bruxelle, J.; Cunin, G.; Fermanian, J.; Ginies, P.; Grun-Overdyking, A.; et al. Comparison of pain syndromes associated with nervous or somatic lesions and development of a new neuropathic pain diagnostic questionnaire (DN4). Pain 2005, 114, 29–36. [Google Scholar] [CrossRef]
- Wang, X.; Luo, L.; Xing, J.; Wang, J.; Shi, B.; Li, Y.-M.; Li, Y.-G. Assessment of peripheral neuropathy in type 2 diabetes by diffusion tensor imaging. Quant. Imaging Med. Surg. 2022, 12, 395–405. [Google Scholar] [CrossRef]
- Singh, K.; Gupta, K.; Kaur, S. High resolution ultrasonography of the tibial nerve in diabetic peripheral neuropathy. J. Ultrason. 2017, 17, 246–252. [Google Scholar] [CrossRef] [PubMed]
- England, J.; Gronseth, G.; Franklin, G.; Carter, G.; Kinsella, L.; Cohen, J.; Asbury, A.; Szigeti, K.; Lupski, J.; Latov, N.; et al. Practice Parameter: The Evaluation of Distal Symmetric Polyneuropathy: The Role of Autonomic Testing, Nerve Biopsy, and Skin Biopsy (An Evidence-Based Review). PM&R 2009, 1, 14–22. [Google Scholar] [CrossRef]
- Lauria, G.; Hsieh, S.T.; Johansson, O.; Kennedy, W.R.; Leger, J.M.; Mellgren, S.I.; Nolano, M.; Merkies, I.S.J.; Polydefkis, M.; Smith, A.G.; et al. European Federation of Neurological Societies/Peripheral Nerve Society Guideline on the use of skin biopsy in the diagnosis of small fiber neuropathy. Report of a joint task force of the European Fe-deration of Neurological Societies and the Peripheral Nerve Society. Eur. J. Neurol. 2010, 17, 903-e49. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, M.; Petropoulos, I.N.; Malik, R.A. Corneal Confocal Microscopy to Assess Diabetic Neuropathy: An Eye on the Foot. J. Diabetes Sci. Technol. 2013, 7, 1179–1189. [Google Scholar] [CrossRef]
- Selvarajah, D.; Cash, T.; Davies, J.; Sankar, A.; Rao, G.; Grieg, M.; Pallai, S.; Gandhi, R.; Wilkinson, I.D.; Tesfaye, S. SUDOSCAN: A Simple, Rapid, and Objective Method with Potential for Screening for Diabetic Peripheral Neuropathy. PLoS ONE 2015, 10, e0138224. [Google Scholar] [CrossRef]
- Pham, M.; Oikonomou, D.; Hornung, B.; Weiler, M.; Heiland, S.; Bäumer, P.; Kollmer, J.; Nawroth, P.P.; Bendszus, M. Magnetic resonance neurography detects diabetic neuropathy early and with Proximal Predominance. Ann. Neurol. 2015, 78, 939–948. [Google Scholar] [CrossRef]
- Rolke, R.; Baron, R.; Maier, C.; Tölle, T.R.; Treede, R.-D.; Beyer, A.; Binder, A.; Birbaumer, N.; Birklein, F.; Bötefür, I.C.; et al. Quantitative sensory testing in the German Research Network on Neuropathic Pain (DFNS): Standardized protocol and reference values. Pain 2006, 123, 231–243. [Google Scholar] [CrossRef]
- Vollert, J.; Attal, N.; Baron, R.; Freynhagen, R.; Haanpää, M.; Hansson, P.; Jensen, T.S.; Rice, A.S.; Segerdahl, M.; Serra, J.; et al. Quantitative sensory testing using DFNS protocol in Europe: An evaluation of heterogeneity across multiple centers in patients with peripheral neuropathic pain and healthy subjects. Pain 2016, 157, 750–758. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Hu, Z.; Luo, Y.; Liu, Y.; Luo, W.; Du, X.; Luo, Z.; Hu, J.; Peng, S. Diabetic peripheral neuropathy: Pathogenetic mechanisms and treatment. Front. Endocrinol. 2024, 14, 1265372. [Google Scholar] [CrossRef]
- Khalid, M.; Petroianu, G.; Adem, A. Advanced Glycation End Products and Diabetes Mellitus: Mechanisms and Perspectives. Biomolecules 2022, 12, 542. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.; Li, K.; Chen, Y.; Xie, J.; Wu, C.; Cui, C.; Deng, B. Oxidative Stress in Diabetic Peripheral Neuropathy: Pathway and Mechanism-Based Treatment. Mol. Neurobiol. 2023, 60, 4574–4594. [Google Scholar] [CrossRef]
- Tavares-Ferreira, D.; Shen, B.Q.; Mwirigi, J.M.; Shiers, S.; Sankaranarayanan, I.; Sreerangapuri, A.; Kotamarti, M.B.; Inturi, N.N.; Mazhar, K.; Ubogu, E.E.; et al. Cell and molecular profiles in peripheral nerves shift toward inflammatory phenotypes in diabetic peripheral neuropathy. J. Clin. Investig. 2025, 135, e184075. [Google Scholar] [CrossRef]
- Richner, M.; Ferreira, N.; Dudele, A.; Jensen, T.S.; Vaegter, C.B.; Gonçalves, N.P. Functional and Structural Changes of the Blood-Nerve-Barrier in Diabetic Neuropathy. Front. Neurosci. 2019, 12, 1038. [Google Scholar] [CrossRef]
- Wu, P.-Z.; Yao, J.; Meng, B.; Qin, Y.-B.; Cao, S. Blood-nerve barrier enhances chronic postsurgical pain via the HIF-1α/ aquaporin-1 signaling axis. BMC Anesthesiol. 2023, 23, 381. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, Y.; Sato, R.; Kanda, T. Blood–Nerve Barrier (BNB) Pathology in Diabetic Peripheral Neuropathy and In Vitro Human BNB Model. Int. J. Mol. Sci. 2020, 22, 62. [Google Scholar] [CrossRef] [PubMed]
- Malong, L.; Napoli, I.; Casal, G.; White, I.J.; Stierli, S.; Vaughan, A.; Cattin, A.-L.; Burden, J.J.; Hng, K.I.; Bossio, A.; et al. Characterization of the structure and control of the blood-nerve barrier identifies avenues for therapeutic delivery. Dev. Cell 2023, 58, 174–191.e8. [Google Scholar] [CrossRef]
- Rusli, N.; Ng, C.F.; Makpol, S.; Wong, Y.P.; Isa, I.L.M.; Remli, R. Antioxidant Effect in Diabetic Peripheral Neuropathy in Rat Model: A Systematic Review. Antioxidants 2024, 13, 1041. [Google Scholar] [CrossRef]
- Negi, G.; Nakkina, V.; Kamble, P.; Sharma, S.S. Heme oxygenase-1, a novel target for the treatment of diabetic complications: Focus on diabetic peripheral neuropathy. Pharmacol. Res. 2015, 102, 158–167. [Google Scholar] [CrossRef]
- Siddiqui, K.; George, T.P.; Mujammami, M.; Isnani, A.; Alfadda, A.A. The association of cell adhesion molecules and selectins (VCAM-1, ICAM-1, E-selectin, L-selectin, and P-selectin) with microvascular complications in patients with type 2 diabetes: A follow-up study. Front. Endocrinol. 2023, 14, 1072288. [Google Scholar] [CrossRef]
- Robles-Osorio, M.L.; Sabath, E. Tight junction disruption and the pathogenesis of the chronic complications of diabetes mellitus: A narrative review. World J. Diabetes 2023, 14, 1013–1026. [Google Scholar] [CrossRef]
- Reinhold, A.K.; Schwabe, J.; Lux, T.J.; Salvador, E.; Rittner, H.L. Quantitative and Microstructural Changes of the Blood-Nerve Barrier in Peripheral Neuropathy. Front. Neurosci. 2018, 12, 936. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, F.; Sano, Y.; Haruki, H.; Kanda, T. Advanced glycation end-products induce basement membrane hypertrophy in endoneurial microvessels and disrupt the blood–nerve barrier by stimulating the release of TGF-β and vascular endothelial growth factor (VEGF) by pericytes. Diabetologia 2011, 54, 1517–1526. [Google Scholar] [CrossRef]
- Trujillo, J.M.; Nuffer, W.; Smith, B.A. GLP-1 receptor agonists: An updated review of head-to-head clinical studies. Ther. Adv. Endocrinol. Metab. 2021, 12, 2042018821997320. [Google Scholar] [CrossRef]
- Davies, M.; Færch, L.; Jeppesen, O.K.; Pakseresht, A.; Pedersen, S.D.; Perreault, L.; Rosenstock, J.; Shimomura, I.; Viljoen, A.; A Wadden, T.; et al. Semaglutide 2·4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): A randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet 2021, 397, 971–984. [Google Scholar] [CrossRef]
- Cosmi, F.; Laini, R.; Nicolucci, A. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2017, 376, 890–892. [Google Scholar] [CrossRef]
- Patti, A.M.; Giglio, R.V.; Allotta, A.; Bruno, A.; Di Bella, T.; Stoian, A.P.; Ciaccio, M.; Rizzo, M. Effect of Semaglutide on Subclinical Atherosclerosis and Cardiometabolic Compensation: A Real-World Study in Patients with Type 2 Diabetes. Biomedicines 2023, 11, 1362. [Google Scholar] [CrossRef]
- Bergmann, N.C.; Davies, M.J.; Lingvay, I.; Knop, F.K. Semaglutide for the treatment of overweight and obesity: A review. Diabetes Obes. Metab. 2022, 25, 18–35. [Google Scholar] [CrossRef]
- Liu, D.-X.; Zhao, C.-S.; Wei, X.-N.; Ma, Y.-P.; Wu, J.-K. Semaglutide Protects against 6-OHDA Toxicity by Enhancing Autophagy and Inhibiting Oxidative Stress. Park. Dis. 2022, 2022, 6813017. [Google Scholar] [CrossRef]
- Kalinderi, K.; Papaliagkas, V.; Fidani, L. GLP-1 Receptor Agonists: A New Treatment in Parkinson’s Disease. Int. J. Mol. Sci. 2024, 25, 3812. [Google Scholar] [CrossRef] [PubMed]
- Maskery, M.P.; Holscher, C.; Jones, S.P.; I Price, C.; Strain, W.D.; Watkins, C.L.; Werring, D.J.; Emsley, H.C. Glucagon-like peptide-1 receptor agonists as neuroprotective agents for ischemic stroke: A systematic scoping review. J. Cereb. Blood Flow Metab. 2020, 41, 14–30. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Feng, P.; Zhang, X.; Li, D.; Wang, R.; Ji, C.; Li, G.; Hölscher, C. The diabetes drug semaglutide reduces infarct size, inflammation, and apoptosis, and normalizes neurogenesis in a rat model of stroke. Neuropharmacology 2019, 158, 107748. [Google Scholar] [CrossRef] [PubMed]
- Poupon-Bejuit, L.; Hughes, M.P.; Liu, W.; Geard, A.; Faour-Slika, N.; Whaler, S.; Massaro, G.; Rahim, A.A. A GLP1 receptor agonist diabetes drug ameliorates neurodegeneration in a mouse model of infantile neurometabolic disease. Sci. Rep. 2022, 12, 13825. [Google Scholar] [CrossRef]
- Estato, V.; Obadia, N.; Chateaubriand, P.H.; Figueiredo, V.; Curty, M.; Silva, M.C.; Ferreira, R.G.L.; Santa-Ritta, J.; Baroni, M.C.; Aragão, A.; et al. Semaglutide restores astrocyte–vascular interactions and blood–brain barrier integrity in a model of diet-induced metabolic syndrome. Diabetol. Metab. Syndr. 2025, 17, 2. [Google Scholar] [CrossRef]
- Chen, L.; Xu, H.; Zhang, C.; He, J.; Wang, Y. Semaglutide alleviates early brain injury following subarachnoid hemorrhage by suppressing ferroptosis and neuroinflammation via SIRT1 pathway. Am. J. Transl. Res. 2024, 16, 1102–1117. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-F.; Zhang, D.; Hu, W.-M.; Liu, D.-X.; Li, L. Semaglutide-mediated protection against Aβ correlated with enhancement of autophagy and inhibition of apotosis. J. Clin. Neurosci. 2020, 81, 234–239. [Google Scholar] [CrossRef]
- Lee, S.-O.; Kuthati, Y.; Huang, W.-H.; Wong, C.-S. Semaglutide Ameliorates Diabetic Neuropathic Pain by Inhibiting Neuroinflammation in the Spinal Cord. Cells 2024, 13, 1857. [Google Scholar] [CrossRef]
- Ubogu, E.E. Biology of the human blood-nerve barrier in health and disease. Exp. Neurol. 2020, 328, 113272. [Google Scholar] [CrossRef]
- Nishihara, H.; Maeda, T.; Sano, Y.; Ueno, M.; Okamoto, N.; Takeshita, Y.; Shimizu, F.; Koga, M.; Kanda, T. Fingolimod promotes blood–nerve barrier properties in vitro. Brain Behav. 2018, 8, e00924. [Google Scholar] [CrossRef]
- Lim, T.K.; Shi, X.Q.; Martin, H.C.; Huang, H.; Luheshi, G.; Rivest, S.; Zhang, J. Blood-nerve barrier dysfunction contributes to the generation of neuropathic pain and allows targeting of injured nerves for pain relief. Pain 2014, 155, 954–967. [Google Scholar] [CrossRef]
- Shimizu, F.; Sano, Y.; Saito, K.; Abe, M.-A.; Maeda, T.; Haruki, H.; Kanda, T. Pericyte-derived Glial Cell Line-derived Neurotrophic Factor Increase the Expression of Claudin-5 in the Blood–brain Barrier and the Blood-nerve Barrier. Neurochem. Res. 2011, 37, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Ydens, E.; Amann, L.; Asselbergh, B.; Scott, C.L.; Martens, L.; Sichien, D.; Mossad, O.; Blank, T.; De Prijck, S.; Low, D.; et al. Profiling peripheral nerve macrophages reveals two macrophage subsets with distinct localization, transcriptome and response to injury. Nat. Neurosci. 2020, 23, 676–689. [Google Scholar] [CrossRef]
- Reinhold, A.; Rittner, H. Characteristics of the nerve barrier and the blood dorsal root ganglion barrier in health and disease. Exp. Neurol. 2020, 327, 113244. [Google Scholar] [CrossRef] [PubMed]
- Vincent, A.M.; Russell, J.W.; Low, P.; Feldman, E.L. Oxidative Stress in the Pathogenesis of Diabetic Neuropathy. Endocr. Rev. 2004, 25, 612–628. [Google Scholar] [CrossRef]
- Wada, R.; Yagihashi, S. Role of Advanced Glycation End Products and Their Receptors in Development of Diabetic Neuropathy. Ann. New York Acad. Sci. 2005, 1043, 598–604. [Google Scholar] [CrossRef]
- Eftekharpour, E.; Fernyhough, P. Oxidative Stress and Mitochondrial Dysfunction Associated with Peripheral Neuropathy in Type 1 Diabetes. Antioxid. Redox Signal. 2022, 37, 578–596. [Google Scholar] [CrossRef]
- Negi, G.; Kumar, A.; Joshi, R.P.; Sharma, S.S. Oxidative stress and Nrf2 in the pathophysiology of diabetic neuropathy: Old perspective with a new angle. Biochem. Biophys. Res. Commun. 2011, 408, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Mursal, M.; Hasan, I.; Tiwari, B.; Srivastava, R.K.; Yadav, G.; Fatima, G. Disruptions in nitric oxide homeostasis, lipid peroxidation-derived oxidative stress, and antioxidant defense mechanisms in spinal cord injury: Elucidating biomolecular correlates of disease severity. Mol. Biol. Rep. 2025, 52, 969. [Google Scholar] [CrossRef] [PubMed]
- Boonnoy, P.; Karttunen, M.; Wong-Ekkabut, J. Alpha-tocopherol inhibits pore formation in oxidized bilayers. Phys. Chem. Chem. Phys. 2017, 19, 5699–5704. [Google Scholar] [CrossRef]
- Zou, M.-H.; Cohen, R.A.; Ullrich, V. Peroxynitrite and Vascular Endothelial Dysfunction in Diabetes Mellitus. Endothelium 2004, 11, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Szabo, C. Role of nitrosative stress in the pathogenesis of diabetic vascular dysfunction. Br. J. Pharmacol. 2009, 156, 713–727. [Google Scholar] [CrossRef]
- El-Remessy, A.B.; Tawfik, H.E.; Matragoon, S.; Pillai, B.; Caldwell, R.W. Peroxynitrite Mediates Diabetes-Induced Endothelial Dysfunction: Possible Role of Rho Kinase Activation. Exp. Diabetes Res. 2010, 2010, 247861. [Google Scholar] [CrossRef]
- Al-Shabrawey, M.; Smith, S. Prediction of diabetic retinopathy: Role of oxidative stress and relevance of apoptotic biomarkers. EPMA J. 2010, 1, 56–72. [Google Scholar] [CrossRef]
- Sun, L.-Q.; Zhao, J.; Zhang, T.; Qu, L.; Wang, X.; Xue, B.; Li, X.-J.; Mu, Y.-M.; Lu, J.-M. Protective Effects of Salvianolic Acid B on Schwann Cells Apoptosis Induced by High Glucose. Neurochem. Res. 2012, 37, 996–1010. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Xue, B.; Li, X.; Liu, H. Puerarin prevents high glucose-induced apoptosis of Schwann cells by inhibiting oxidative stress. Neural Regen Res. 2012, 7, 2583–2591. [Google Scholar] [CrossRef] [PubMed]
- Buendia, I.; Michalska, P.; Navarro, E.; Gameiro, I.; Egea, J.; León, R. Nrf2–ARE pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol. Ther. 2016, 157, 84–104. [Google Scholar] [CrossRef]
- Sandireddy, R.; Yerra, V.G.; Areti, A.; Komirishetty, P.; Kumar, A. Neuroinflammation and Oxidative Stress in Diabetic Neuropathy: Futuristic Strategies Based on These Targets. Int. J. Endocrinol. 2014, 2014, 674987. [Google Scholar] [CrossRef] [PubMed]
- Lv, R.; Zhao, Y.; Wang, X.; He, Y.; Dong, N.; Min, X.; Liu, X.; Yu, Q.; Yuan, K.; Yue, H.; et al. GLP-1 analogue liraglutide attenuates CIH-induced cognitive deficits by inhibiting oxidative stress, neuroinflammation, and apoptosis via the Nrf2/HO-1 and MAPK/NF-κB signaling pathways. Int. Immunopharmacol. 2024, 142, 113222. [Google Scholar] [CrossRef]
- Yang, X.; Yao, W.; Shi, H.; Liu, H.; Li, Y.; Gao, Y.; Liu, R.; Xu, L. Paeoniflorin protects Schwann cells against high glucose induced oxidative injury by activating Nrf2/ARE pathway and inhibiting apoptosis. J. Ethnopharmacol. 2016, 185, 361–369. [Google Scholar] [CrossRef]
- Brownlee, M. The Pathobiology of Diabetic Complicationss: A unifying mechanism. Diabetes 2005, 54, 1615–1625. [Google Scholar] [CrossRef]
- Giacco, F.; Brownlee, M. Oxidative Stress and Diabetic Complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Basta, G.; Lazzerini, G.; Massaro, M.; Simoncini, T.; Tanganelli, P.; Fu, C.; Kislinger, T.; Stern, D.M.; Schmidt, A.M.; De Caterina, R. Advanced Glycation End Products Activate Endothelium Through Signal-Transduction Receptor RAGE: A mechanism for amplification of inflammatory responses. Circulation 2002, 105, 816–822. [Google Scholar] [CrossRef]
- Schmidt, A.M.; Hori, O.; Chen, J.X.; Li, J.F.; Crandall, J.; Zhang, J.; Cao, R.; Yan, S.D.; Brett, J.; Stern, D. Advanced glycation endproducts interacting with their endothelial receptor induce expression of vascular cell adhesion molecule-1 (VCAM-1) in cultured human endothelial cells and in mice. A potential mechanism for the accelerated vasculopathy of diabetes. J. Clin. Investig. 1995, 96, 1395–1403. [Google Scholar] [CrossRef]
- Callaghan, B.C.; Cheng, H.T.; Stables, C.L.; Smith, A.L.; Feldman, E.L. Diabetic neuropathy: Clinical manifestations and current treatments. Lancet Neurol. 2012, 11, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Yang, Y.; Zhang, J. A systematic review and meta-analysis of the serum lipid profile in prediction of diabetic neuropathy. Sci. Rep. 2021, 11, 499. [Google Scholar] [CrossRef]
- Horton, W.B.; Barrett, E.J. Microvascular Dysfunction in Diabetes Mellitus and Cardiometabolic Disease. Endocr. Rev. 2021, 42, 29–55. [Google Scholar] [CrossRef]
- Zmijewski, J.W.; Moellering, D.R.; Le Goffe, C.; Landar, A.; Ramachandran, A.; Darley-Usmar, V.M. Oxidized LDL induces mitochondrially associated reactive oxygen/nitrogen species formation in endothelial cells. Am. J. Physiol. Circ. Physiol. 2005, 289, H852–H861. [Google Scholar] [CrossRef] [PubMed]
- Maiuolo, J.; Gliozzi, M.; Musolino, V.; Carresi, C.; Nucera, S.; Macrì, R.; Scicchitano, M.; Bosco, F.; Scarano, F.; Ruga, S.; et al. The Role of Endothelial Dysfunction in Peripheral Blood Nerve Barrier: Molecular Mechanisms and Pathophysiological Implications. Int. J. Mol. Sci. 2019, 20, 3022. [Google Scholar] [CrossRef]
- Panou, T.; Gouveri, E.; Popovic, D.S.; Papazoglou, D.; Papanas, N. The Role of Inflammation in the Pathogenesis of Diabetic Peripheral Neuropathy: New Lessons from Experimental Studies and Clinical Implications. Diabetes Ther. 2025, 16, 371–411. [Google Scholar] [CrossRef]
- Fenzi, F.; Latronico, N.; Refatti, N.; Rizzuto, N. Enhanced expression of E-selectin on the vascular endothelium of peripheral nerve in critically ill patients with neuromuscular disorders. Acta Neuropathol. 2003, 106, 75–82. [Google Scholar] [CrossRef]
- Jude, E.B.; Abbott, C.A.; Young, M.J.; Anderson, S.G.; Douglas, J.T.; Boulton, A.J.M. The potential role of cell adhesion molecules in the pathogenesis of diabetic neuropathy. Diabetologia 1998, 41, 330–336. [Google Scholar] [CrossRef]
- Chong, Z.Z.; Souayah, N. Crumbling Pathogenesis and Biomarkers for Diabetic Peripheral Neuropathy. Biomedicines 2025, 13, 413. [Google Scholar] [CrossRef]
- Zochodne, D.W. The challenges of diabetic polyneuropathy: A brief update. Curr. Opin. Neurol. 2019, 32, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Myers, R.R.; Campana, W.M.; Shubayev, V.I. The role of neuroinflammation in neuropathic pain: Mechanisms and therapeutic targets. Drug Discov. Today 2006, 11, 8–20. [Google Scholar] [CrossRef]
- Cao, L.; Ding, L.; Xia, Q.; Zhang, Z.; Li, M.; Song, S.; Yin, K.; Li, Z.; Li, X.; Wang, Z.; et al. Macrophage polarization in diabetic vascular complications: Mechanistic insights and therapeutic targets. J. Transl. Med. 2025, 23, 1050. [Google Scholar] [CrossRef]
- Nichols, J.M.; Crelli, C.V.; Liu, L.; Pham, H.V.; Janjic, J.M.; Shepherd, A.J. Tracking macrophages in diabetic neuropathy with two-color nanoemulsions for near-infrared fluorescent imaging and microscopy. J. Neuroinflammation 2021, 18, 299. [Google Scholar] [CrossRef] [PubMed]
- Rochfort, K.D.; Cummins, P.M. The blood–brain barrier endothelium: A target for pro-inflammatory cytokines. Biochem. Soc. Trans. 2015, 43, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Ma, P.; Wu, M.; Liao, H.; Song, X.-J. Role of Matrix Metalloproteinases in Myelin Abnormalities and Mechanical Allodynia in Rodents with Diabetic Neuropathy. Aging Dis. 2021, 12, 1808–1820. [Google Scholar] [CrossRef]
- Weerasuriya, A.; Mizisin, A.P. The Blood-Nerve Barrier: Structure and Functional Significance. In The Blood-Brain and Other Neural Barriers: Reviews and Protocols; Nag, S., Ed.; Humana Press: Totowa, NJ, USA, 2011; pp. 149–173. [Google Scholar]
- Reinhold, A.K.; Rittner, H.L. Barrier function in the peripheral and central nervous system—A review. Pflügers Arch.-Eur. J. Physiol. 2017, 469, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Radu, M.; Chernoff, J. An in vivo Assay to Test Blood Vessel Permeability. J. Vis. Exp. 2013, e50062. [Google Scholar] [CrossRef]
- Seneviratne, K.N. Permeability of blood nerve barriers in the diabetic rat. J. Neurol. Neurosurg. Psychiatry 1972, 35, 156–162. [Google Scholar] [CrossRef]
- Hulström, D.; Malmgren, L.; Gilstring, D.; Olsson, Y. FITC-Dextrans as tracers for macromolecular movements in the nervous system: A freeze-drying method for dextrans of various molecular sizes injected into normal animals. Acta Neuropathol. 1983, 59, 53–62. [Google Scholar] [CrossRef]
- Poduslo, J.F.; Curran, G.L.; Berg, C.T. Macromolecular permeability across the blood-nerve and blood-brain barriers. Proc. Natl. Acad. Sci. USA 1994, 91, 5705–5709. [Google Scholar] [CrossRef]
- Denzer, L.; Muranyi, W.; Schroten, H.; Schwerk, C. The role of PLVAP in endothelial cells. Cell Tissue Res. 2023, 392, 393–412. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.J.; Allen, J.E.; Biswas, S.K.; Fisher, E.A.; Gilroy, D.W.; Goerdt, S.; Gordon, S.; Hamilton, J.A.; Ivashkiv, L.B.; Lawrence, T.; et al. Macrophage Activation and Polarization: Nomenclature and Experimental Guidelines. Immunity 2014, 41, 14–20. [Google Scholar] [CrossRef]
- Morrison, H.W.; A Filosa, J. A quantitative spatiotemporal analysis of microglia morphology during ischemic stroke and reperfusion. J. Neuroinflammation 2013, 10, 4. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.M.; Jung, C.H. Effects of Incretin-Based Therapies on Diabetic Microvascular Complications. Endocrinol. Metab. 2017, 32, 316–325. [Google Scholar] [CrossRef] [PubMed]
- Kawanami, D.; Matoba, K.; Sango, K.; Utsunomiya, K. Incretin-Based Therapies for Diabetic Complications: Basic Mechanisms and Clinical Evidence. Int. J. Mol. Sci. 2016, 17, 1223. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, P.E.; Abdelkader, N.F.; El Awdan, S.A.; El-Shabrawy, O.A.; Zaki, H.F. Liraglutide ameliorated peripheral neuropathy in diabetic rats: Involvement of oxidative stress, inflammation and extracellular matrix remodeling. J. Neurochem. 2018, 146, 173–185. [Google Scholar] [CrossRef]
- Avogaro, A.; Fadini, G.P. The Effects of Dipeptidyl Peptidase-4 Inhibition on Microvascular Diabetes Complications. Diabetes Care 2014, 37, 2884–2894. [Google Scholar] [CrossRef]
- Ashish, K.; Akash, S.; Rita, K.; Kunal, K.; Divya, S.; Bharthu, P.; Ashok, S.; Santosh, K.; Samir, G.; Vijay, S.; et al. Sitagliptin, sitagliptin and metformin, or sitagliptin and amitriptyline attenuate streptozotocin-nicotinamide induced diabetic neuropathy in rats. J. Biomed. Res. 2012, 26, 200–210. [Google Scholar] [CrossRef]
- Tsuboi, K.; Mizukami, H.; Inaba, W.; Baba, M.; Yagihashi, S. The dipeptidyl peptidase IV inhibitor vildagliptin suppresses development of neuropathy in diabetic rodents: Effects on peripheral sensory nerve function, structure and molecular changes. J. Neurochem. 2016, 136, 859–870. [Google Scholar] [CrossRef]
- Kuthati, Y.; Rao, V.N.; Huang, W.-H.; Busa, P.; Wong, C.-S. Teneligliptin Co-Infusion Alleviates Morphine Tolerance by Inhibition of Spinal Microglial Cell Activation in Streptozotocin-Induced Diabetic Rats. Antioxidants 2023, 12, 1478. [Google Scholar] [CrossRef]
- Gonçalves, A.; Marques, C.; Leal, E.; Ribeiro, C.F.; Reis, F.; Ambrósio, A.F.; Fernandes, R. Dipeptidyl peptidase-IV inhibition prevents blood–retinal barrier breakdown, inflammation and neuronal cell death in the retina of type 1 diabetic rats. Biochim. Biophys. Acta (BBA)—Mol. Basis Dis. 2014, 1842, 1454–1463. [Google Scholar] [CrossRef] [PubMed]
- Frías, J.P.; Davies, M.J.; Rosenstock, J.; Pérez Manghi, F.C.; Fernández Landó, L.; Bergman, B.K.; Liu, B.; Cui, X.; Brown, K. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N. Engl. J. Med. 2021, 385, 503–515. [Google Scholar] [CrossRef]
- Nauck, M.A.; D‘aLessio, D.A. Tirzepatide, a dual GIP/GLP-1 receptor co-agonist for the treatment of type 2 diabetes with unmatched effectiveness regrading glycaemic control and body weight reduction. Cardiovasc. Diabetol. 2022, 21, 169. [Google Scholar] [CrossRef]
- Jastreboff, A.M.; Kaplan, L.M.; Frías, J.P.; Wu, Q.; Du, Y.; Gurbuz, S.; Coskun, T.; Haupt, A.; Milicevic, Z.; Hartman, M.L. Triple–Hormone-Receptor Agonist Retatrutide for Obesity—A Phase 2 Trial. N. Engl. J. Med. 2023, 389, 514–526. [Google Scholar] [CrossRef]
- Jing, F.; Zeng, Y.; Yu, Q.-L.; Fu, C.-J. The role of GLP-1 receptor in pain disorders and its pharmacological properties. Eur. J. Pharmacol. 2025, 1008, 178345. [Google Scholar] [CrossRef]
- Kuthati, Y.; Davuluri, V.N.G.; Wong, C.-S. Therapeutic Effects of GLP-1 Receptor Agonists and DPP-4 Inhibitors in Neuropathic Pain: Mechanisms and Clinical Implications. Biomolecules 2025, 15, 622. [Google Scholar] [CrossRef]
- Liu, C.; Wu, T.; Ren, N. Glucagon-like peptide-1 receptor agonists for the management of diabetic peripheral neuropathy. Front. Endocrinol. 2024, 14, 1268619. [Google Scholar] [CrossRef]
- Ma, J.; Shi, M.; Zhang, X.; Liu, X.; Chen, J.; Zhang, R.; Wang, X.; Zhang, H. GLP-1R agonists ameliorate peripheral nerve dysfunction and inflammation via p38 MAPK/NF-κB signaling pathways in streptozotocin-induced diabetic rats. Int. J. Mol. Med. 2018, 41, 2977–2985. [Google Scholar] [CrossRef]
- Dhanapalaratnam, R.; Issar, T.; Lee, A.T.K.; Poynten, A.M.; Milner, K.-L.; Kwai, N.C.G.; Krishnan, A.V. Glucagon-like peptide-1 receptor agonists reverse nerve morphological abnormalities in diabetic peripheral neuropathy. Diabetologia 2024, 67, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Jolivalt, C.G.; Fineman, M.; Deacon, C.F.; Carr, R.D.; Calcutt, N.A. GLP-1 signals via ERK in peripheral nerve and prevents nerve dysfunction in diabetic mice. Diabetes Obes. Metab. 2011, 13, 990–1000. [Google Scholar] [CrossRef] [PubMed]
- de Sousa, E.; Sparks, L.; Townsend, K. Incretin Receptors in the Peripheral Nervous System: Implications for Obesity Treatment and Peripheral Neuropathy. Diabetes 2025, 74, 1313–1319. [Google Scholar] [CrossRef] [PubMed]
- Issar, T.; Kwai, N.C.; Poynten, A.M.; Arnold, R.; Milner, K.-L.; Krishnan, A.V. Effect of exenatide on peripheral nerve excitability in type 2 diabetes. Clin. Neurophysiol. 2021, 132, 2532–2539. [Google Scholar] [CrossRef] [PubMed]
- Dhanapalaratnam, R.; Issar, T.; Poynten, A.M.; Milner, K.-L.; Kwai, N.C.G.; Krishnan, A.V. Impact of glucagon-like peptide-1 receptor agonists on axonal function in diabetic peripheral neuropathy. J. Neurophysiol. 2025, 133, 14–21. [Google Scholar] [CrossRef]
- Jaiswal, M.; Martin, C.L.; Brown, M.B.; Callaghan, B.; Albers, J.W.; Feldman, E.L.; Pop-Busui, R. Effects of exenatide on measures of diabetic neuropathy in subjects with type 2 diabetes: Results from an 18-month proof-of-concept open-label randomized study. J. Diabetes Its Complicat. 2015, 29, 1287–1294. [Google Scholar] [CrossRef]
- Fan, S.; Qiu, Y.; Liu, J.; Zhu, T.; Wang, C.; Liu, D.; Yan, L.; Ren, M. Effect of the glucagon-like peptide-1 receptor agonists on diabetic peripheral neuropathy: A meta-analysis. J. Neurochem. 2024, 169, e16242. [Google Scholar] [CrossRef]
- Panou, T.; Gouveri, E.; Popovic, D.S.; Papazoglou, D.; Papanas, N. The Therapeutic Potential of Dipeptidyl Peptidase 4 Inhibitors and Glucagon-Like Peptide-1 Receptor Agonists in Diabetic Peripheral Neuropathy. Diabetes Ther. 2025, 16, 1077–1105. [Google Scholar] [CrossRef] [PubMed]
| Tier | Modality (Examples) | Key Outputs |
|---|---|---|
| Best Use | ||
| Key Limitation | ||
| Screen (bedside) | 10-g monofilament; vibration (128-Hz tuning fork/VPT) | LOPS; VPT. |
| Rapid bedside screening; ulcer risk stratification | ||
| Low sensitivity for early isolated SFN; cutoffs method/device-dependent | ||
| Phenotype/stage | History + neuro exam; ± TCNS/mTCNS [15,16]; ± DN4 (pain) [17] | Symptoms/signs; composite severity; neuropathic pain features. |
| Phenotyping/staging; longitudinal monitoring | ||
| Subjective components; inter-rater variability | ||
| Confirm/stage (large fiber) | NCS/EMG | CV, amplitude, latency (pre-specified nerves) |
| Objective confirmation and staging | ||
| Limited sensitivity for pure SFN; access/discomfort constraints | ||
| Small-fiber function/autonomic | QST (DFNS); sudomotor (QSART, ESC/Sudoscan) | Thermal/mechanical thresholds; sweat/ESC |
| Functional phenotyping; potentially treatment-responsive signals | ||
| Time/cooperation; site/device variability; confounders (skin, meds, temperature) | ||
| Small-fiber structure | Skin biopsy (IENFD); corneal confocal microscopy (CCM) | IENFD (fibers/mm); CNFD/CNFL/CNBD |
| Structural SFN endpoints; early disease and longitudinal monitoring | ||
| Invasive/processing-dependent; CCM access + standardized analysis required | ||
| Imaging (exploratory) | MR neurography (MRN)/DTI [18]; high-resolution ultrasound (HRUS) [19] | Nerve morphology/microstructure (e.g., CSA; diffusion indices) |
| Localization; exploratory biomarkers/morphology | ||
| Cost; harmonization/standardization; clinical utility still evolving | ||
| Abbreviations: CCM, corneal confocal microscopy; CNBD, corneal nerve branch density; CNFD, corneal nerve fiber density; CNFL, corneal nerve fiber length; DFNS, German Research Network on Neuropathic Pain; DPN, diabetic peripheral neuropathy; DTI, diffusion tensor imaging; EMG, electromyography; ESC, electrochemical skin conductance; HRUS, high-resolution ultrasound; IENFD, intraepidermal nerve fiber density; LOPS, loss of protective sensation; MRN, magnetic resonance neurography; NCS, nerve conduction studies; QST, quantitative sensory testing; QSART, quantitative sudomotor axon reflex test; SFN, small-fiber neuropathy; TCNS, Toronto Clinical Neuropathy Score; VPT, vibration perception threshold; CSA, cross-sectional area. | ||
| Domain | Representative Readouts | Biological Significance | Typical Methods |
|---|---|---|---|
| Lipid peroxidation | MDA; 4-HNE | Membrane lipid oxidation; aldehyde-mediated injury | TBARS; HPLC; ELISA; IHC/IF; immunoblotting |
| Protein oxidation/nitration | Nitrotyrosine; protein carbonyls | Protein nitration and oxidative modification | IHC/IF; immunoblotting; DNPH-based assays |
| Oxidative DNA damage | 8-OHdG | Nuclear or mitochondrial DNA oxidation | ELISA; IHC/IF; HPLC; LC-MS |
| Antioxidant response/defense failure | Nrf2 nuclear translocation; HO-1; SOD1/2; catalase; GPx; GSH/GSSG ratio | Antioxidant activation or exhaustion; redox-buffering capacity | Fractionation assays; immunoblotting; RT-qPCR; enzyme activity assays; glutathione assays |
| Mitochondrial stress/dysfunction | mtROS; mitochondrial membrane potential; respiratory enzyme/OXPHOS changes; ATP content | ROS amplification; bioenergetic failure | MitoSOX; JC-1/TMRE/TMRM; respirometry; ATP assays; TEM |
| Abbreviations: 4-HNE, 4-hydroxynonenal; 8-OHdG, 8-hydroxy-2′-deoxyguanosine; DPN, diabetic peripheral neuropathy; GPx, glutathione peroxidase; GSH/GSSG, reduced/oxidized glutathione ratio; HO-1, heme oxygenase-1; IHC/IF, immunohistochemistry/immunofluorescence; LC-MS, liquid chromatography-mass spectrometry; MDA, malondialdehyde; mtROS, mitochondrial reactive oxygen species; Nrf2, nuclear factor erythroid 2-related factor 2; OXPHOS, oxidative phosphorylation; PNVU, peripheral nerve neurovascular unit; SOD, superoxide dismutase; TEM, transmission electron microscopy. | |||
| Class | Representative Agents | Evidence in DPN | Putative PNVU/BNB-Relevant Actions | Key Limitations/Notes |
|---|---|---|---|---|
| GLP-1 receptor agonists (GLP-1RAs) | Liraglutide; semaglutide; exenatide | Preclinical: improved nerve function and reduced oxidative/inflammatory injury in multiple models [107]. | Endothelial anti-oxidative and anti-inflammatory signaling; reduced neuroimmune activation; potential preservation of tight-junction integrity through reduced inflammatory stress. | Human evidence for DPN outcomes is still limited/heterogeneous; class effects vs. agent-specific effects require clarification. |
| DPP-4 inhibitors (DPP-4is) | Sitagliptin; vildagliptin; linagliptin | Preclinical: improved NCV/IENFD and DRG signaling in diabetic rodents [109,110]. Clinical: preliminary data for microvascular benefit [108]. | Augments endogenous GLP-1 and affects non-incretin substrates; may reduce endothelial inflammation and vascular leakage in other barriers (e.g., retina) [112]. | Direct BNB studies are sparse; effects may be partly mediated by improved glycemia; need DPN trials with barrier-relevant endpoints. |
| Dual incretin agonists (GIP/GLP-1) | Tirzepatide | Strong human metabolic efficacy vs. semaglutide; neuropathy-specific endpoints largely untested [113,114]. | Metabolic unloading (glucose, lipids, weight) may reduce upstream PNVU stressors (AGE-RAGE, dyslipidemia/perfusion impairment) and secondarily attenuate endothelial activation. | Translation to DPN/BNB remains hypothesis-driven; microvascular inflammation/perfusion effects need dedicated studies. |
| Multi-agonists (e.g., GLP-1/GIP/glucagon) | Retatrutide (investigational) | Early-phase human trials show substantial weight loss; DPN/BNB endpoints not yet available [115]. | Potential for stronger metabolic and hemodynamic remodeling could translate into reduced neurovascular stress, but mechanisms and safety require validation. | Not approved; long-term microvascular safety data limited; no DPN-focused trials to date. |
| Abbreviations: BNB, blood-nerve barrier; DPN, diabetic peripheral neuropathy; PNVU, peripheral nerve neurovascular unit; GLP-1RA(s), GLP-1 receptor agonist(s); DPP-4i(s), dipeptidyl peptidase-4 inhibitor(s); GIP, glucose-dependent insulinotropic polypeptide; DRG, dorsal root ganglion; NCV, nerve conduction velocity; IENFD, intraepidermal nerve fiber density. | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




