Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodological Approach: Evidence Sourcing and Synthesis
2.1. Review Design and Reporting
2.2. Scope and Eligibility
2.3. Information Sources and Search Strategy
2.4. Study Selection, Synthesis, and Appraisal
2.5. Review Positioning and Rationale
3. Redox Signaling in Exercise: The Mechanistic Scaffold
3.1. Sources and Kinetics of Reactive Species During Exercise
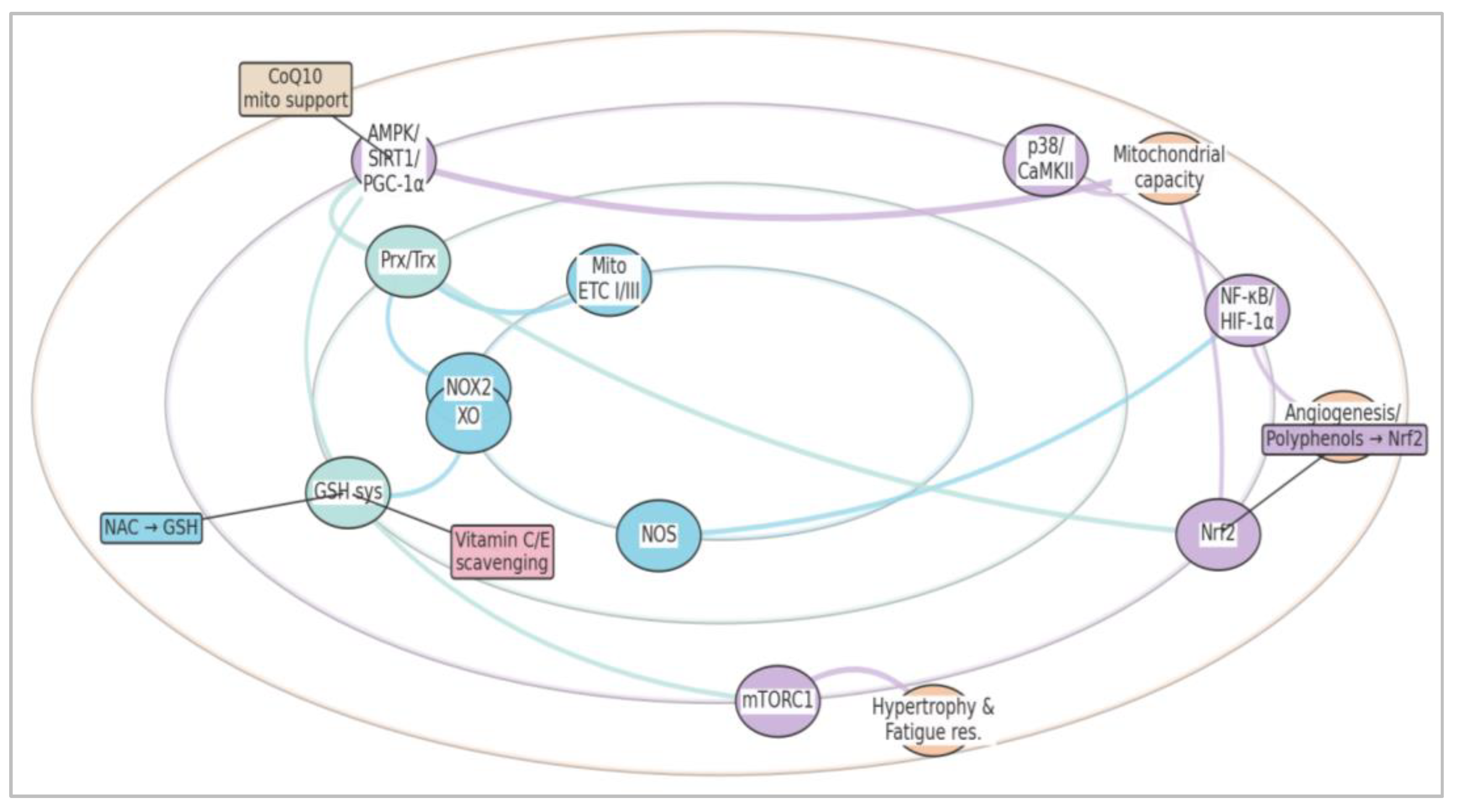
3.2. Redox-Sensitive Nodes That Couple Work to Adaptation
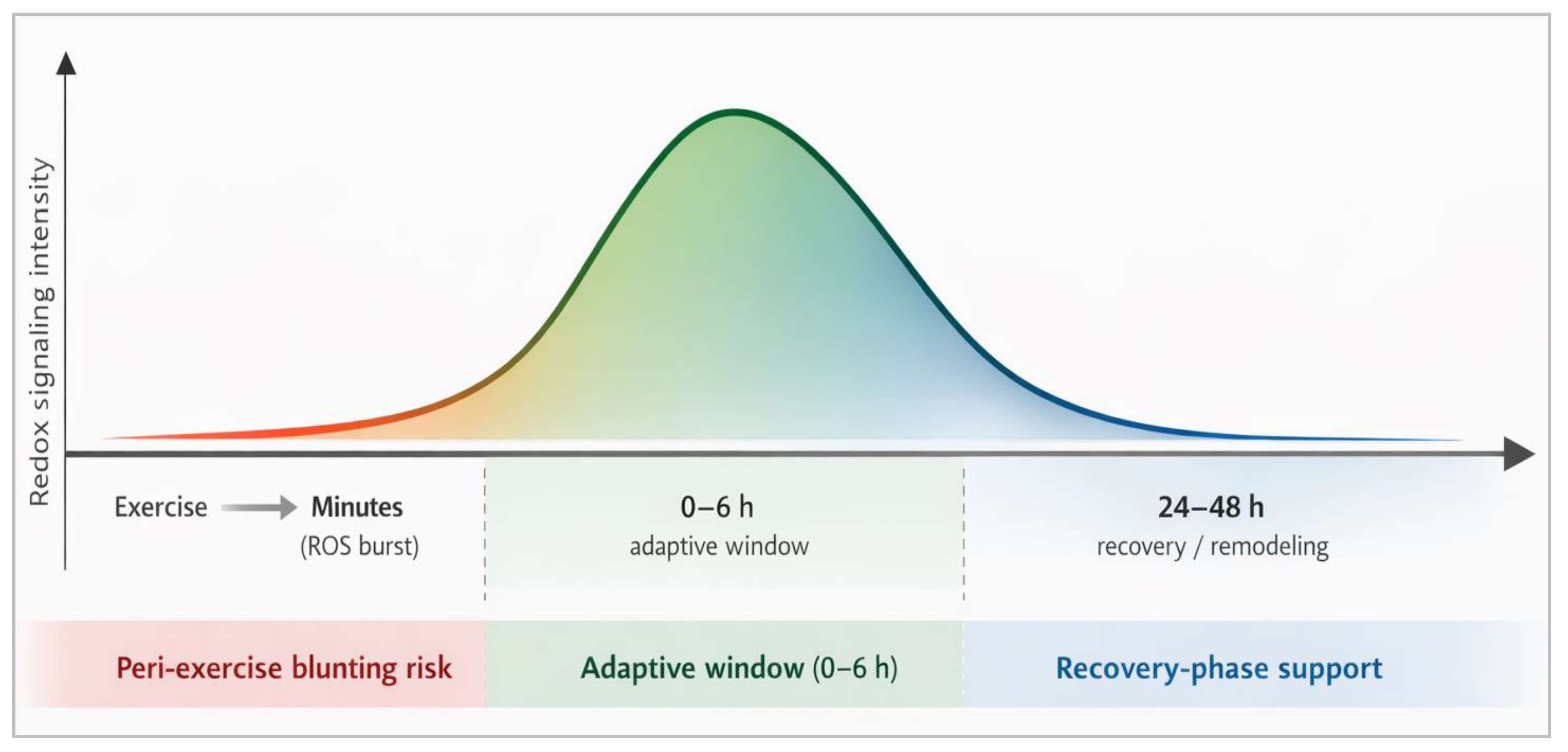
3.3. Hormesis and the “Redox Window”
3.4. Practical Implication
4. Antioxidant Taxonomy and Practical Pharmacology
4.1. Mechanistic Classes and Where they Interface with Redox Signaling
4.2. Polyphenols and Other Indirect Redox Modulators
4.3. Practical Pharmacology: Formulation, Safety, and Compliance
| Class / compound |
Primary action(s) |
Typical trial dose/form | Timing patterns studied |
Bio availability notes |
Common AEs | Interaction flags |
Practice note |
|---|---|---|---|---|---|---|---|
| Vitamin C | Aqueous radical scavenger; regenerates E |
500–1000 mg/day; 0.5–1 g pre/post |
Chronic near training; acute pre-exercise |
SVCT- mediated, saturable |
GI upset at high doses |
Enhances non-heme iron absorption; high-dose use may increase kidney-stone risk | Avoid chronic peri-exercise in adaptation blocks unless correcting deficiency |
|
Vitamin E (α-tocopherol) |
Lipid chain -breaking scavenger |
200–800 IU/day | Chronic | Requires fat for absorption |
GI, rare bleeding risk at high doses | Anticoagulants | Similar caution as vitamin C; avoid high doses peri-exercise |
| NAC | Cys donor that supports glutathione (GSH); direct scavenging | 600–1200 mg acute; short courses 2–7 d |
60–90 min pre-exercise; short performance blocks | Oral bioavailability moderate; IV research-only |
GI upset, sulfur taste |
Caution with nitroglycerin (headache/ hypotension) |
Consider for repeated-bout performance or heat/hypoxia; trial in training |
|
CoQ10 (ubiquinone/ ubiquinol) |
ETC carrier; membrane antioxidant | 100–300 mg/day | Chronic | Better with fat; ubiquinol increases exposure |
GI | Warfarin interaction reported |
Mixed effects; safe/neutral in athletes |
| ALA | Mitochondrial redox cycling |
150–600 mg/day | Chronic | R-ALA more active |
GI, hypo- glycemia in sensitive |
Diabetes meds |
Sport evidence limited; small RCT suggests improved recovery during intensive microcycles; long-term training effects unclear. |
|
Anthocyanin -rich foods (tart cherry, berries) |
Nrf2 activation; anti-inflammatory; sleep support (tart cherry) | Juice /concentrate; 2×30–60 mL/day or equivalent |
3–10 d pre-competition and through competition/recovery | Food matrix matters; microbiome metabolism |
Rare GI |
Minimal | Useful for DOMS/recovery in competition weeks |
|
Pomegranate polyphenols |
Endothelial/ Anti - inflammatory |
250–500 mL juice or equivalent extract |
2–7 d and acute pre-exercise |
Polyphenol metabolites drive effects |
GI in large volumes |
None major |
May aid blood flow in endurance events |
|
Cocoa flavanols |
NO bio- availability; endothelial |
300–600 mg CF/day |
1–7 d and acute |
Processed chocolate varies widely |
Caffeine in chocolate |
None major |
Consider for submax/endurance support |
|
Curcumin (phytosome) |
NF-κB modulation; soreness |
500–1000 mg/day (std extract); lower if phytosome | 1–3 d pre-damaging work and 2–3 d post-damaging work | Low oral bioavailability; phytosome increases |
GI; piperine interactions |
Anti- coagulants |
Recovery support; avoid peri-strength if concerned about signaling (data limited) |
| Resveratrol | SIRT1/AMPK activator | 150–500 mg/day | Chronic | Low bioavailability |
GI, headaches |
CYP interactions |
Potential to blunt adaptations in older men at high doses—use cautiously |
| Astaxanthin | Membrane antioxidant | 4–12 mg/day |
Chronic | Lipid carriers aid |
Well tolerated |
— | Neutral /mixed for performance |
| Melatonin | Indirect antioxidant; sleep |
1–5 mg pre-sleep |
Nights before competition/ travel |
Chronobiotic; timing critical | Next-day sleepiness | Sedatives | Use for jet-lag/sleep with circadian guidance |
5. Effects on Training Adaptations (Chronic Outcomes)
5.1. Endurance Adaptations
| Antioxidant (Class) | Population (Status; n) [Ref] |
Dose & Timing |
Training Protocol (Duration; Modality) |
Primary Adaptation Outcomes |
Direction | Key Moderators |
RoB |
|---|---|---|---|---|---|---|---|
|
Vitamin C (direct scavenger) |
Trained men (n=14) [108] |
1 g/day; chronic; peri-exercise |
8 wk endurance training |
Mitochondrial biogenesis markers; endurance performance |
Decrease mitochondrial markers; performance: mixed |
High dose; proximity to sessions |
Some concerns |
|
Vitamins C/E (direct scavengers) |
Young men & women (n=54) [109] |
1000 mg C + 235 mg E/day; chronic |
11 wk endurance training |
Cellular adaptation markers; VO₂max |
Decrease cellular adaptation; VO₂max: no clear change/mixed |
Sex; training status |
Low |
| Vitamins C/E | Healthy adults [110] |
Chronic supplementation during training |
Exercise training intervention |
Metabolic/ health adaptations |
Attenuate exercise-induced metabolic benefits |
Baseline metabolic status |
Low |
|
Resveratrol (polyphenol bioactive) |
Aged men (n=27) [111] |
Chronic; during training |
Exercise training program |
VO₂max; vascular adaptations |
Attenuate training response |
Older age; AMPK/SIR 1 modulation |
Low |
|
MitoQ (mitochondria -targeted) |
Untrained middle-aged men [112] |
20 mg/day; chronic | 3 wk HIIT |
VO₂peak; peak power; CS activity |
VO₂peak: no clear change; peak power: increase |
Short intervention; untrained |
Some concerns |
|
NAC (thiol donor) |
Recreational men [113] |
Acute responses pre/post training period |
6 wk Sprint -interval training |
Redox markers; fatigue physiology |
Context- dependent (training modifies NAC effect) |
Training status; redox baseline |
Some concerns |
|
CoQ10 (mitochondrial support) |
Mixed training status [114] |
100–300 mg/day; chronic | Various endurance programs |
Performance endpoints | No clear change /mixed |
Dose hetero- geneity |
N/A |
5.2. Resistance/Hypertrophy
| Antioxidant (Class) | Population (Status; n) [Ref] |
Dose & Timing |
Training Protocol (Duration; Modality) |
Primary Adaptation Outcomes |
Direction | Key Moderators |
RoB |
|---|---|---|---|---|---|---|---|
|
Vitamins C/E (direct scavengers) |
Young men & women (n=54) [115] |
1000 mg C + 235 mg E/day; chronic; peri-exercise likely |
10–12 wk heavy-load resistance training |
p70S6K signaling; lean mass; maximal strength |
Decrease signaling; hypertrophy: no clear change; strength: mixed |
High-dose; training status; timing proximity |
Low |
|
Vitamins C/E (direct scavengers) |
Elderly men, 60–81 y (n=34) [116] |
500 mg C + 117.5 mg E before & after training (per session); 12 wk | 12 wk strength training (3 sessions/week) |
Lean mass (DXA); muscle thickness (US); 1RM strength |
Decrease lean-mass gains; strength gains: no clear change | Older age; peri-exercise high-dose |
Some concerns |
|
Vitamin C/E (direct scavengers) |
Healthy men trained & untrained; (n=28) [117] |
Vitamin C 1 g/day + vitamin E 400 IU/day for 11 wk (started 4 wk before pre-testing); taken before breakfast | Eccentric training: 4 wk (2×/wk) + acute eccentric bouts pre- and post-training |
Muscle performance; redox biomarkers; hemolysis; muscle damage indices |
No clear change |
Training status; eccentric model; biomarker selection |
Some concerns |
|
Polyphenol blend (tea extracts; indirect antioxidant) |
Untrained men (n=40) [118] |
2000 mg/day proprietary polyphenol blend; 4-wk preload; continued daily during training | 4 wk supplementation + 6 wk progressive full-body RT (3 d/wk) | Lower-body 1RM strength; systemic TAC/oxidative markers | Strength gains: no clear change; TAC: increase |
Proprietary blend (tea extracts + caffeine); untrained status; industry funding |
Some concerns |
|
Vitamins C/E (direct scavengers) |
Trained men (n=23) [119] |
1000 mg C + 235 mg E/day; mornings; 10 wk |
10 wk RT + ~300 kcal/day surplus (hypertrophy-oriented) |
Fat-free mass (DXA); upper/lower body strength; VAT |
Upper-body hypertrophy /strength: small decrease; most outcomes: no clear change; VAT gain: decrease |
Energy surplus context; trained status; peri-session timing |
Some concerns |
6. Effects on Acute Performance and Recovery
6.1. Performance Within a Session or Within 24 h
6.2. Recovery over 24–72 h
| Antioxidant (Class) | Population (Status; n) | Dose & Timing |
Protocol Type | Primary Functional Outcome |
Direction | Context Sensitivity |
RoB |
|---|---|---|---|---|---|---|---|
|
NAC (thiol donor) |
Trained Men [120] |
IV infusion pre-exercise | Prolonged submax cycling |
Time to fatigue |
Time to fatigue: increase |
Laboratory setting; IV protocol |
Some concerns |
|
NAC (oral) |
Well-trained triathletes (n=10; 8 completed) [121] |
1200 mg/day orally for 9 days (loading) |
Cycle ergometer race simulation during intense training |
Sprint performance during race simulation |
Performance: increase | Small sample; GI tolerance; inter -individual variability |
Some concerns |
|
NAC (systematic review) |
Adult males (healthy/active/athletes; 16 trials) [122] |
Acute to short-term; doses varied |
Controlled trials across multiple performance models |
Performance + laboratory biomarkers (redox, GSH, etc.) |
Mixed; overall trend toward performance improvement |
High heterogeneity; baseline redox status |
Variable/ assessed |
|
Alpha-lipoic acid (ALA) |
Resistance- and Endurance -experienced men (crossover; n=17) [91] |
Single: 150 mg immediately post-session; Short-term: 300 mg/day (150 mg 2 h pre + 150 mg immediately post) during 6 d intensified training |
Acute + short-term intensified training |
Back squat performance; muscle damage (CK, myoglobin); inflammation (IL-6); soreness |
Recovery: improve (modest) |
Timing (pre/post); training load; baseline redox status | Some concerns |
|
Tart cherry (anthocyanins) |
Endurance runners [123] |
~ 8 days preloading |
Marathon/ long-distance run |
Muscle soreness |
Recovery: improve (reduced soreness) |
Running- induced damage |
Low |
|
Tart cherry concentrate |
Semi- professional soccer (n=16) [124] |
30 mL ×2/day; 8 days |
Intermittent sprint activity |
Recovery markers; performance proxies |
Recovery: improve |
Congested match schedule |
Low |
|
Tart cherry powder (Montmorency; anthocyanins) |
Endurance-trained runners/ triathletes (men & women; n=27) [125] |
480 mg/day for ~10 d (7 d pre + day-of + 48 h post endurance challenge) |
Endurance running challenge |
Performance + recovery biomarkers (catabolism, inflammation, redox balance); soreness |
Performance: increase; stress markers: decrease |
Short-term pre-loading; formulation; training status |
Some concerns |
| Pomegranate juice | Resistance-trained men (n=17) [126] |
250 mL twice/day for 15 days + 250 mL immediately post-exercise |
Eccentric damage model (elbow flexors + knee extensors) |
Isometric strength recovery; muscle soreness |
Recovery: improve (elbow flexors); knee extensors: no clear change |
Upper vs lower limb effects |
Some concerns |
| Pomegranate juice | Elite Weightlifters [127] |
750 mL/day for 48 h pre + 500 mL 60 min pre-session |
Weightlifting training session (cross-over vs placebo) |
Soreness; RPE; muscle-damage/ inflammation biomarkers |
Recovery: improve |
High muscle-damage load; short pre-load | Some concerns |
|
Pomegranate extract |
Active Adults [128] |
Acute or short-term |
Endurance/ time-trial models |
Performance outcomes |
No clear change/ slight increase |
Vascular modulation; dose |
Some concerns |
|
Curcumin (CurcuWIN®; pre-loading) |
Physically active men & women (n=63) [102] |
CurcuWIN® daily for 8 wk: 250 mg (50 mg curcuminoids) or 1000 mg (200 mg curcuminoids) |
Downhill running (muscle- damaging) |
Isokinetic torque/power; soreness (1–72 h post) |
Recovery: improve (200 mg); no clear change (50 mg) |
Dose- dependent; model (eccentric running) |
Some concerns |
|
Curcumin (bioavailable) |
Physically active adults [129] |
Pre- and post-exercise |
EIMD protocol |
DOMS; functional recovery |
Soreness: decrease; performance: mixed |
Formulation; timing | Some concerns |
|
Turmeric formulation |
Active men [130] |
Acute dosing |
DOMS model |
Muscle pain; CK |
Soreness: decrease |
Bio- availability |
Some concerns |
|
Cocoa flavanols (systematic review) |
Mixed populations & modalities [131] |
Various doses/ durations; heterogeneous |
Multiple Exercise models (performance & recovery) |
Performance, vascular and oxidative stress outcomes (varies by study) |
Mixed | Dose/ composition; co-ingestants (e.g. caffeine); training status |
N/A (review) |
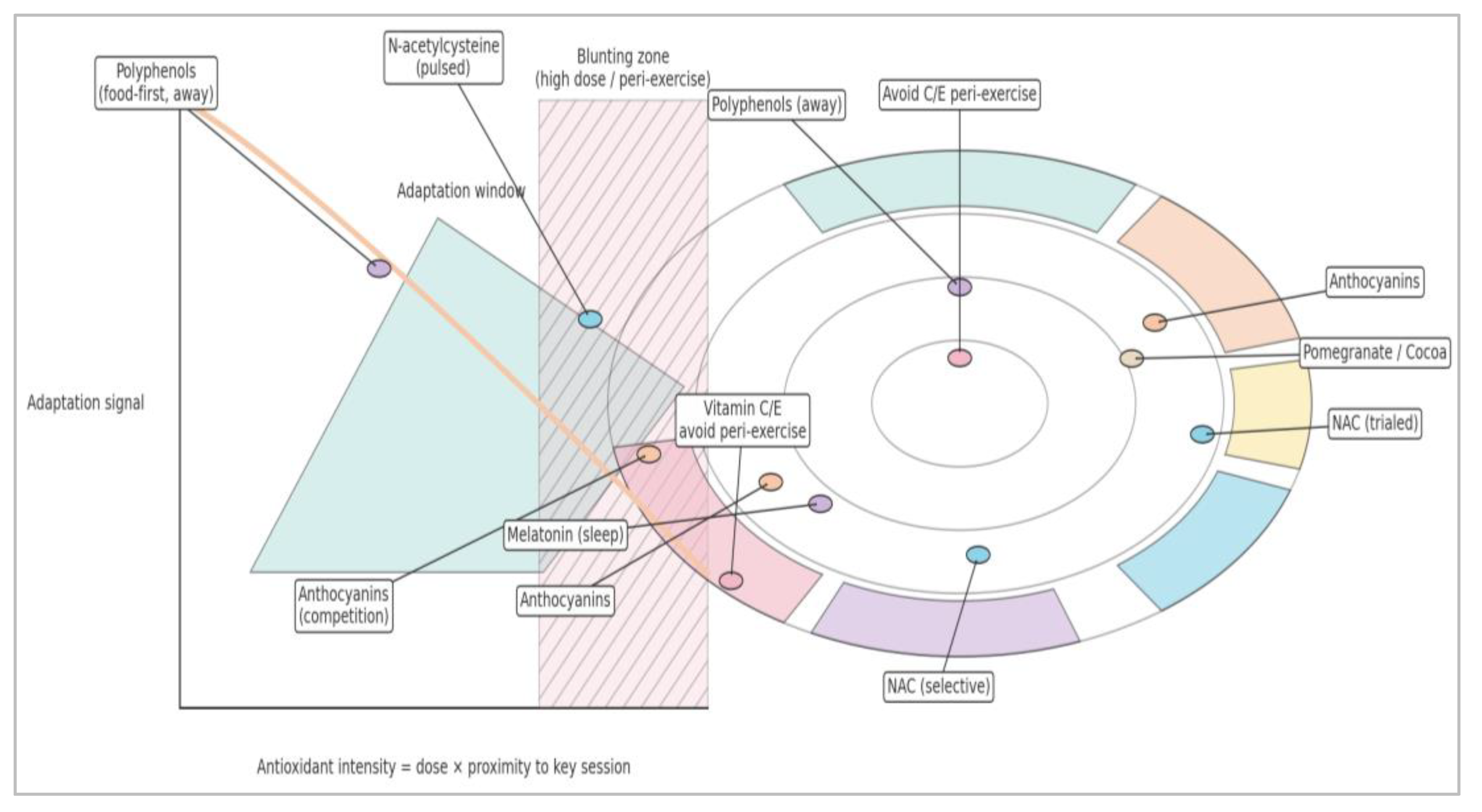
6.3. Timing Relative to Exercise: The Redox Window
6.4. Stressful Environments
7. Moderators and Personalization
7.1. Athlete Biology: Training Status, Sex, and Age
7.2. Nutritional Context: Baseline Diet and Energy Availability
7.3. External Constraints: Environment, Schedule, and Sleep Disruption
8. Biomarkers and Methodology—Measuring What Matters
8.1. Biomarker Quality Matrix
8.2. Sampling Windows and Tissues
8.3. Training-Load and Diet Control
8.4. Statistics for Non-Linear Responses
9. A Redox-Informed Framework for Antioxidant Use in Exercise
9.1. Principles
9.2. Practical Implementation
| Phase / Constraint |
Vitamins C/E (scavengers) |
NAC (thiol donor) |
Polyphenols (food-first) | Anthocyanins (tart cherry/ berries) |
Pome-granate | Curcumin | MitoQ/ CoQ10 |
Mela-tonin |
|---|---|---|---|---|---|---|---|---|
|
Build (adaptation priority) |
Avoid peri; avoid chronic high-dose | Rare | Food-first only | Not routine | Food-first | If needed, away from sessions | Likely neutral; limited data |
Sleep only |
|
Intensified block |
Avoid peri | Consider short pulse | Moderate intake | Optional | Optional | If high muscle damage | Unclear | If sleep disturbed |
|
Taper/ Competition week |
Avoid high-dose |
Trial first; pulsed if responder | Moderate | Pulse (3–10 d) |
Pulse | Short peri-damage window | Individual | If travel/late matches |
|
Heat/ Hypoxia/ Altitude |
Avoid chronic | Consider pulsed | Supportive | Useful | Supportive | Supportive | Uncertain | — |
|
Congested fixtures/ Travel |
Avoid peri | If proven responder | Yes | Yes (recovery focus) |
Yes | Yes (DOMS-heavy) | Optional | Yes |
|
Low energy availability |
Avoid chronic | Cautious | Food-first emphasis |
Supportive | Supportive | Conservative | Neutral | Monitor sleep |
10. Gaps and Priorities for Future Research
11. Conclusions and Key Messages
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fisher-Wellman, K.; Bloomer, R.J. Acute exercise and oxidative stress: A 30 year history. Dyn. Med. 2009, 8, 1. [Google Scholar] [CrossRef]
- Powers, S.K.; Jackson, M.J. Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production. Physiol. Rev. 2008, 88, 1243–1276. [Google Scholar] [CrossRef]
- Clarkson, P.M.; Thompson, H.S. Antioxidants: What role do they play in physical activity and health? Am. J. Clin. Nutr. 2000, 72, 637S–646S. [Google Scholar] [CrossRef]
- Sen, C.K. Oxidants and antioxidants in exercise. J. Appl. Physiol. 1995, 79, 675–686. [Google Scholar] [CrossRef]
- Davies, K.J.A.; Quintanilha, A.T.; Brooks, G.A.; Packer, L. Free radicals and tissue damage produced by exercise. Biochem. Biophys. Res. Commun. 1982, 107, 1198–1205. [Google Scholar] [CrossRef]
- Dröge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P. Redefining oxidative stress. Antioxid. Redox Signal. 2006, 8, 1865–1879. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.G. Cell signaling. H2O2, a necessary evil for cell signaling. Science 2006, 312, 1882–1883. [Google Scholar] [CrossRef]
- Sies, H. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: Oxidative eustress. Redox Biol. 2017, 11, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T. Signal transduction by reactive oxygen species. J. Cell Biol. 2011, 194, 7–15. [Google Scholar] [CrossRef]
- Powers, S.K.; Duarte, J.; Kavazis, A.N.; Talbert, E.E. Reactive oxygen species are signalling molecules for skeletal muscle adaptation. Exp. Physiol. 2010, 95, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Handschin, C.; Spiegelman, B.M. The role of exercise and PGC-1α in inflammation and chronic disease. Nature 2008, 454, 463–469. [Google Scholar] [CrossRef]
- Hood, D.A. Mechanisms of exercise-induced mitochondrial biogenesis in skeletal muscle. Appl. Physiol. Nutr. Metab. 2009, 34, 465–472. [Google Scholar] [CrossRef]
- Egan, B.; Zierath, J.R. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 2013, 17, 162–184. [Google Scholar] [CrossRef]
- Peternelj, T.T.; Coombes, J.S. Antioxidant supplementation during exercise training: Beneficial or detrimental? Sports Med. 2011, 41, 1043–1069. [Google Scholar] [CrossRef]
- Gomez-Cabrera, M.C.; Domenech, E.; Viña, J. Antioxidant supplements in exercise: Worse than useless? Am. J. Physiol. Endocrinol. Metab. 2012, 302, E476–E477. [Google Scholar] [CrossRef]
- Morrison, D.; Hughes, J.; Della Gatta, P.A.; Mason, S.; Lamon, S.; Russell, A.P.; Wadley, G.D. Vitamin C and E supplementation prevents some of the cellular adaptations to endurance-training in humans. Free Radic. Biol. Med. 2015, 89, 852–862. [Google Scholar] [CrossRef] [PubMed]
- Yfanti, C.; Nielsen, A.R.; Akerström, T.; Nielsen, S.; Rose, A.J.; Richter, E.A.; Lykkesfeldt, J.; Fischer, C.P.; Pedersen, B.K. Effect of antioxidant supplementation on insulin sensitivity in response to endurance exercise training. Am. J. Physiol. Endocrinol. Metab. 2011, 300, E761–E770. [Google Scholar] [CrossRef] [PubMed]
- Cumming, K.T.; Raastad, T.; Holden, G.; Bastani, N.E.; Schneeberger, D.; Paronetto, M.P.; Mercatelli, N.; Østgaard, H.N.; Ugelstad, I.; Caporossi, D.; et al. Effects of Vitamin C and E Supplementation on Endogenous Antioxidant Systems and Heat Shock Proteins in Response to Endurance Training. Physiol. Rep. 2014, 2, e12142. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Wu, C.; Ma, S. Application of Exercise/Training Models to Evaluate Food Functionality with Special Focus on Preventing Inflammation and Oxidative Stress and Enhancing Exercise Performance. Foods 2025, 14, 4025. [Google Scholar] [CrossRef]
- Dutra, M.T.; Alex, S.; Mota, M.R.; Sales, N.B.; Brown, L.E.; Bottaro, M. Effect of strength training combined with antioxidant supplementation on muscular performance. Appl. Physiol. Nutr. Metab. 2018, 43, 775–781. [Google Scholar] [CrossRef]
- Merry, T.L.; Ristow, M. Do antioxidant supplements interfere with skeletal muscle adaptation to exercise training? J. Physiol. 2016, 594, 5135–5147. [Google Scholar] [CrossRef] [PubMed]
- Taherkhani, S.; Valaei, K.; Arazi, H.; Suzuki, K. An Overview of Physical Exercise and Antioxidant Supplementation Influences on Skeletal Muscle Oxidative Stress. Antioxidants 2021, 10, 1528. [Google Scholar] [CrossRef]
- Traber, M.G.; Stevens, J.F. Vitamins C and E: Beneficial effects from a mechanistic perspective. Free Radic. Biol. Med. 2011, 51, 1000–1013. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Davies, K.J.A.; Ursini, F. How do nutritional antioxidants really work: Nucleophilic tone and para-hormesis versus free radical scavenging in vivo. Free Radic. Biol. Med. 2014, 66, 24–35. [Google Scholar] [CrossRef]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q. Role of Nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef]
- Vargas-Mendoza, N.; Morales-González, Á.; Madrigal-Santillán, E.O.; Madrigal-Bujaidar, E.; Álvarez-González, I.; García-Melo, L.F.; Anguiano-Robledo, L.; Fregoso-Aguilar, T.; Morales-Gonzalez, J.A. Antioxidant and adaptative response mediated by Nrf2 during physical exercise. Antioxidants 2019, 8, 196. [Google Scholar] [CrossRef]
- Hardie, D.G. AMP-activated protein kinase: An energy sensor that regulates all aspects of cell function. Genes Dev. 2011, 25, 1895–1908. [Google Scholar] [CrossRef]
- Cantó, C.; Auwerx, J. PGC-1α, SIRT1 and AMPK, an energy sensing network that controls energy expenditure. Curr. Opin. Lipidol. 2009, 20, 98–105. [Google Scholar] [CrossRef]
- Halliwell, B.; Whiteman, M. Measuring reactive species and oxidative damage in vivo and in cell culture: How should you do it and what do the results mean? Br. J. Pharmacol. 2004, 142, 231–255. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.J., II; Morrow, J.D. Products of the isoprostane pathway: Unique bioactive compounds and markers of lipid peroxidation. Cell Mol. Life Sci. 2002, 59, 808–820. [Google Scholar] [CrossRef] [PubMed]
- Montuschi, P.; Barnes, P.J.; Roberts, L.J., II. Isoprostanes: Markers and mediators of oxidative stress. FASEB J. 2004, 18, 1791–1800. [Google Scholar] [CrossRef] [PubMed]
- Comporti, M.; Signorini, C.; Arezzini, B.; Vecchio, D.; Monaco, B.; Gardi, C. F2-isoprostanes are not just markers of oxidative stress. Free Radic. Biol. Med. 2008, 44, 247–256. [Google Scholar] [CrossRef]
- Janero, D.R. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med. 1990, 9, 515–540. [Google Scholar] [CrossRef]
- Dalle-Donne, I.; Rossi, R.; Giustarini, D.; Milzani, A.; Colombo, R. Protein carbonyl groups as biomarkers of oxidative stress. Clin. Chim. Acta 2003, 329, 23–38. [Google Scholar] [CrossRef]
- Michailidis, Y.; Jamurtas, A.Z.; Nikolaidis, M.G.; Fatouros, I.G.; Koutedakis, Y.; Papassotiriou, I.; Kouretas, D. Sampling time is crucial for measurement of aerobic exercise-induced oxidative stress. Med. Sci. Sports Exerc. 2007, 39, 1107–1113. [Google Scholar] [CrossRef]
- Radak, Z.; Chung, H.Y.; Goto, S. Exercise and hormesis: Oxidative stress-related adaptation and aging. Ageing Res. Rev. 2008, 7, 34–42. [Google Scholar] [CrossRef]
- Spanidis, Y.; et al. Exercise-induced reductive stress is a protective mechanism against oxidative stress in peripheral blood mononuclear cells. Oxid. Med. Cell. Longev. 2018, 2018, 3053704. [Google Scholar] [CrossRef]
- Grant, M.J.; Booth, A. A typology of reviews: An analysis of 14 review types and associated methodologies. Health Inf. Libr. J. 2009, 26, 91–108. [Google Scholar] [CrossRef]
- Campbell, M.; McKenzie, J.E.; Sowden, A.; Katikireddi, S.V.; Brennan, S.E.; Ellis, S.; Hartmann-Boyce, J.; Ryan, R.; Shepperd, S.; Thomas, J.; Welch, V.; Thomson, H. Synthesis without meta-analysis (SWiM) in systematic reviews: Reporting guideline. BMJ 2020, 368, l6890. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; John Wiley & Sons: Chichester, UK, 2019. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef] [PubMed]
- Steinbacher, P.; Eckl, P. Impact of Oxidative Stress on Exercising Skeletal Muscle. Biomolecules 2015, 5, 356–377. [Google Scholar] [CrossRef]
- Suzuki, K.; Tominaga, T.; Ruhee, R.T.; Ma, S. Characterization and Modulation of Systemic Inflammatory Response to Exhaustive Exercise in Relation to Oxidative Stress. Antioxidants 2020, 9, 401. [Google Scholar] [CrossRef]
- Murphy, M.P. How Mitochondria Produce Reactive Oxygen Species. Biochem. J. 2009, 417, 1–13. [Google Scholar] [CrossRef]
- Meneses-Valdés, R.; Gallero, S.; Henríquez-Olguín, C.; Jensen, T.E. Exploring NADPH Oxidases 2 and 4 in Cardiac and Skeletal Muscle Adaptations—A Cross-Tissue Comparison. Free Radic. Biol. Med. 2024, 223, 296–305. [Google Scholar] [CrossRef]
- Lambeth, J.D. NOX Enzymes and the Biology of Reactive Oxygen. Nat. Rev. Immunol. 2004, 4, 181–189. [Google Scholar] [CrossRef]
- Wadley, G.D.; Nicolas, M.A.; Hiam, D.; McConell, G.K. Xanthine Oxidase Inhibition Attenuates Skeletal Muscle Signaling Following Acute Exercise but Does Not Impair Mitochondrial Adaptations to Endurance Training. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E853–E862. [Google Scholar] [CrossRef]
- Förstermann, U.; Sessa, W.C. Nitric Oxide Synthases: Regulation and Function. Eur. Heart J. 2012, 33, 829–837. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive Oxygen Species (ROS) as Pleiotropic Physiological Signalling Agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef] [PubMed]
- Zelko, I.N.; Mariani, T.J.; Folz, R.J. Superoxide Dismutase Multigene Family: A Comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) Gene Structures, Evolution, and Expression. Free Radic. Biol. Med. 2002, 33, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric Oxide and Peroxynitrite in Health and Disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef]
- Go, Y.-M.; Jones, D.P. Redox Compartmentalization in Eukaryotic Cells. Biochim. Biophys. Acta 2008, 1780, 1273–1290. [Google Scholar] [CrossRef]
- Stretton, C.; Pugh, J.N.; McDonagh, B.; McArdle, A.; Close, G.L.; Jackson, M.J. 2-Cys Peroxiredoxin Oxidation in Response to Hydrogen Peroxide and Contractile Activity in Skeletal Muscle: A Novel Insight into Exercise-Induced Redox Signalling? Free Radic. Biol. Med. 2020, 160, 199–207. [Google Scholar] [CrossRef]
- Stöcker, S.; Van Laer, K.; Mijuskovic, A.; Dick, T.P. The Conundrum of Hydrogen Peroxide Signaling and the Emerging Role of Peroxiredoxins as Redox Relay Hubs. Antioxid. Redox Signal. 2018, 28, 558–573. [Google Scholar] [CrossRef] [PubMed]
- Hawley, J.A.; Hargreaves, M.; Joyner, M.J.; Zierath, J.R. Integrative Biology of Exercise. Cell 2014, 159, 738–749. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Mattson, M.P. Hormesis Provides a Generalized Quantitative Estimate of Biological Plasticity. J. Cell Commun. Signal. 2011, 5, 25–38. [Google Scholar] [CrossRef]
- Holmström, K.M.; Finkel, T. Cellular mechanisms and physiological consequences of redox-dependent signalling. Nat. Rev. Mol. Cell Biol. 2014, 15, 411–421. [Google Scholar] [CrossRef]
- Hardie, D.G.; Ross, F.A.; Hawley, S.A. AMPK: A Nutrient and Energy Sensor that Maintains Energy Homeostasis. Nat. Rev. Mol. Cell Biol. 2012, 13, 251–262. [Google Scholar] [CrossRef]
- Puigserver, P.; Spiegelman, B.M. Peroxisome Proliferator-Activated Receptor-γ Coactivator 1α (PGC-1α): Transcriptional Coactivator and Metabolic Regulator. Endocr. Rev. 2003, 24, 78–90. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Kensler, T.W.; Motohashi, H. The KEAP1-NRF2 System: A Thiol-Based Sensor-Effector Apparatus for Maintaining Redox Homeostasis. Physiol. Rev. 2018, 98, 1169–1203. [Google Scholar] [CrossRef] [PubMed]
- Laplante, M.; Sabatini, D.M. mTOR Signaling in Growth Control and Disease. Cell 2012, 149, 274–293. [Google Scholar] [CrossRef] [PubMed]
- Hayden, M.S.; Ghosh, S. NF-κB, the First Quarter-Century: Remarkable Progress and Outstanding Questions. Genes Dev. 2012, 26, 203–234. [Google Scholar] [CrossRef]
- Semenza, G.L. Hypoxia-Inducible Factors in Physiology and Medicine. Cell 2012, 148, 399–408. [Google Scholar] [CrossRef]
- Paulsen, C.E.; Carroll, K.S. Cysteine-Mediated Redox Signaling: Chemistry, Biology, and Tools for Discovery. Chem. Rev. 2013, 113, 4633–4679. [Google Scholar] [CrossRef]
- Dalle-Donne, I.; Rossi, R.; Colombo, G.; Giustarini, D.; Milzani, A. Protein S-Glutathionylation: A Regulatory Device from Bacteria to Humans. Trends Biochem. Sci. 2009, 34, 85–96. [Google Scholar] [CrossRef]
- Rhee, S.G.; Woo, H.A. Multiple Functions of Peroxiredoxins: Peroxidases, Sensors and Regulators of the Intracellular Messenger H2O2, and Protein Chaperones. Antioxid. Redox Signal. 2011, 15, 781–794. [Google Scholar] [CrossRef]
- Sobotta, M.C.; Liou, W.; Stöcker, S.; Talwar, D.; Oehler, M.; Ruppert, T.; Scharf, A.N.D.; Dick, T.P. Peroxiredoxin-2 and STAT3 Form a Redox Relay for H2O2 Signaling. Nat. Chem. Biol. 2015, 11, 64–70. [Google Scholar] [CrossRef]
- Lu, J.; Holmgren, A. The Thioredoxin Antioxidant System. Free Radic. Biol. Med. 2014, 66, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.C. Regulation of Glutathione Synthesis. Mol. Aspects Med. 2009, 30, 42–59. [Google Scholar] [CrossRef] [PubMed]
- Mason, S.A.; Trewin, A.J.; Parker, L.; Wadley, G.D. Antioxidant Supplements and Endurance Exercise: Current Evidence and Mechanistic Insights. Redox Biol. 2020, 35, 101471. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidis, M.G.; Kerksick, C.M.; Lamprecht, M.; McAnulty, S.R. Does vitamin C and E supplementation impair the favorable adaptations of regular exercise? Oxid. Med. Cell. Longev. 2012, 2012, 707941. [Google Scholar] [CrossRef]
- Vauzour, D.; Rodriguez-Mateos, A.; Corona, G.; Oruna-Concha, M.J.; Spencer, J.P.E. Polyphenols and Human Health: Prevention of Disease and Mechanisms of Action. Nutrients 2010, 2, 1106–1131. [Google Scholar] [CrossRef]
- Kanner, J. Polyphenols by Generating H2O2, Affect Cell Redox Signaling, Inhibit PTPs and Activate Nrf2 Axis for Adaptation and Cell Surviving: In Vitro, In Vivo and Human Health. Antioxidants 2020, 9, 797. [Google Scholar] [CrossRef]
- Pedre, B.; Barayeu, U.; Ezeriņa, D.; Dick, T.P. The Mechanism of Action of N-Acetylcysteine (NAC): The Emerging Role of H2S and Sulfane Sulfur Species. Pharmacol. Ther. 2021, 228, 107916. [Google Scholar] [CrossRef]
- Hernández-Camacho, J.D.; Bernier, M.; López-Lluch, G.; Navas, P. Coenzyme Q10 Supplementation in Aging and Disease. Front. Physiol. 2018, 9, 44. [Google Scholar] [CrossRef]
- Powers, S.K.; Radak, Z.; Ji, L.L. Exercise-induced oxidative stress: Past, present and future. J. Physiol. 2016, 594, 5081–5092. [Google Scholar] [CrossRef]
- Pingitore, A.; Lima, G.P.P.; Mastorci, F.; Quinones, A.; Iervasi, G.; Vassalle, C. Exercise and oxidative stress: Potential effects of antioxidant dietary strategies in sports. Nutrition 2015, 31, 916–922. [Google Scholar] [CrossRef]
- Kawamura, T.; Muraoka, I. Exercise-induced oxidative stress and the effects of antioxidant supplements. Antioxidants 2018, 7, 119. [Google Scholar] [CrossRef] [PubMed]
- Stoian, M.; Mănescu, D.C. Training–Fuel Coupling (TFC): A Molecular Sports Nutrition Framework for Energy Availability, Chrono-Nutrition, and Performance Optimization. Nutrients 2026, 18, 693. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Traber, M.G. Vitamin E: Function and Metabolism. FASEB J. 1999, 13, 1145–1155. [Google Scholar] [CrossRef]
- Samuni, Y.; Goldstein, S.; Dean, O.M.; Berk, M. The chemistry and biological activities of N-acetylcysteine. Biochim. Biophys. Acta Gen. Subj. 2013, 1830, 4117–4129. [Google Scholar] [CrossRef]
- Holdiness, M.R. Clinical pharmacokinetics of N-acetylcysteine. Clin. Pharmacokinet. 1991, 20, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Shay, K.P.; Moreau, R.F.; Smith, E.J.; Smith, A.R.; Hagen, T.M. Alpha-lipoic acid as a dietary supplement: Molecular mechanisms and therapeutic potential. Biochim. Biophys. Acta Gen. Subj. 2009, 1790, 1149–1160. [Google Scholar] [CrossRef]
- Isenmann, E.; Trittel, L.; Diel, P. The effects of alpha lipoic acid on muscle strength recovery after a single and a short-term chronic supplementation - a study in healthy well-trained individuals after intensive resistance and endurance training. J. Int. Soc. Sports Nutr. 2020, 17, 61. [Google Scholar] [CrossRef]
- Richie, J.P., Jr.; Nichenametla, S.; Neidig, W.; Calcagnotto, A.; Haley, J.S.; Schell, T.D.; Muscat, J.E. Randomized controlled trial of oral glutathione supplementation on body stores of glutathione. Eur. J. Nutr. 2015, 54, 251–263. [Google Scholar] [CrossRef]
- Murphy, M.P.; Smith, R.A.J. Targeting antioxidants to mitochondria by conjugation to lipophilic cations. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 629–656. [Google Scholar] [CrossRef] [PubMed]
- Bhagavan, H.N.; Chopra, R.K. Coenzyme Q10: Absorption, tissue uptake, metabolism and pharmacokinetics. Free Radic. Res. 2006, 40, 445–453. [Google Scholar] [CrossRef]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep. 2009, 26, 1001–1043. [Google Scholar] [CrossRef]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef]
- Mănescu, D.C.; Plăstoi, C.D.; Petre, R.L.; Mărgărit, I.R.; Mănescu, A.M.; Pîrvan, A. Metabolic Overdrive in Elite Sport: A Systems Model of AMPK–mTOR Oscillation, NAD+ Economy, and Epigenetic Drift. Int. J. Mol. Sci. 2026, 27, 1817. [Google Scholar] [CrossRef]
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. (-)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc. Natl. Acad. Sci. USA 2006, 103, 1024–1029. [Google Scholar] [CrossRef]
- Mănescu, D.C. Training Load Oscillation and Epigenetic Plasticity: Molecular Pathways Connecting Energy Metabolism and Athletic Personality. Int. J. Mol. Sci. 2026, 27, 792. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC Consensus Statement: Dietary Supplements and the High-Performance Athlete. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 104–125. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of Curcumin: Problems and Promises. Mol. Pharmaceutics 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Purpura, M.; Kerksick, C.M.; et al. Eight weeks of a high dose of curcumin supplementation may attenuate performance decrements following muscle-damaging exercise. Nutrients 2019, 11, 1692. [Google Scholar] [CrossRef]
- Mănescu, A.M.; Hangu, S.Ș.; Mănescu, D.C. Nutritional Supplements for Muscle Hypertrophy: Mechanisms and Morphology—Focused Evidence. Nutrients 2025, 17, 3603. [Google Scholar] [CrossRef] [PubMed]
- Arendt, J. Melatonin: Characteristics, concerns, and prospects. J. Biol. Rhythms 2005, 20, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.R.; Gough, L.A.; Deb, S.K.; Sparks, S.A.; McNaughton, L.R. Astaxanthin in Exercise Metabolism, Performance and Recovery: A Review. Front. Nutr. 2018, 4, 76. [Google Scholar] [CrossRef]
- Brown, D.R.; et al. The Effect of Astaxanthin Supplementation on Performance and Fat Oxidation during a 40 km Cycling Time Trial. J. Sci. Med. Sport 2021, 24, 92–97. [Google Scholar] [CrossRef]
- Herxheimer, A.; Petrie, K.J. Melatonin for the Prevention and Treatment of Jet Lag. Cochrane Database Syst. Rev. 2002, (2), CD001520. [Google Scholar] [CrossRef]
- Gomez-Cabrera, M.C.; Domenech, E.; Romagnoli, M.; Arduini, A.; Borras, C.; Pallardo, F.V.; Sastre, J.; Viña, J. Oral administration of vitamin C decreases muscle mitochondrial biogenesis and hampers training-induced adaptations in endurance performance. Am. J. Clin. Nutr. 2008, 87, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, G.; Cumming, K.T.; Holden, G.; Hallén, J.; Rønnestad, B.R.; Sveen, O.; Skaug, A.; Paur, I.; Bastani, N.E.; Østgaard, H.N.; et al. Vitamin C and E supplementation hampers cellular adaptation to endurance training in humans: A double-blind randomized controlled trial. J. Physiol. 2014, 592, 1887–1901. [Google Scholar] [CrossRef]
- Ristow, M.; Zarse, K.; Oberbach, A.; Klöting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Blüher, M. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar] [CrossRef]
- Gliemann, L.; Schmidt, J.F.; Olesen, J.; Biensø, R.S.; Peronard, S.L.; Grandjean, S.U.; Mortensen, S.P.; Nyberg, M.; Bangsbo, J.; Pilegaard, H.; et al. Resveratrol blunts the positive effects of exercise training on cardiovascular health in aged men. J. Physiol. 2013, 591, 5047–5059. [Google Scholar] [CrossRef]
- Broome, S.C.; Woodhead, J.S.T.; Merry, T.L. MitoQ supplementation augments acute exercise-induced increases in muscle PGC-1α mRNA and improves training-induced increases in peak power independent of mitochondrial content and function in untrained middle-aged men. Redox Biol. 2022, 53, 102341. [Google Scholar] [CrossRef]
- Lemminger, A.K.; Fiorenza, M.; Eibye, K.; Bangsbo, J.; Hostrup, M. High-intensity exercise training alters the effect of N-acetylcysteine on exercise-related muscle ionic shifts in men. Antioxidants 2023, 12, 53. [Google Scholar] [CrossRef]
- Deng, H.; Song, T.; Yin, M.; Xu, K.; Zhong, Y.; Bin Mohd, N.; Bin Naharudin, M.N.; Yusof, A.; Fan, X. Coenzyme Q10 supplementation increases blood concentrations but shows limited and inconsistent effects on exercise performance: A systematic review and meta-analysis. Br. J. Nutr. 2025, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, G.; Hamarsland, H.; Cumming, K.T.; Johansen, R.E.; Hulmi, J.J.; Børsheim, E.; Wiig, H.; Garthe, I.; Raastad, T. Vitamin C and E supplementation alters protein signalling after a strength training session, but not muscle growth during 10 weeks of training. J. Physiol. 2014, 592, 5391–5408. [Google Scholar] [CrossRef]
- Bjørnsen, T.; Salvesen, S.; Berntsen, S.; Hetlelid, K.J.; Stea, T.H.; Lohne-Seiler, H.; Rohde, G.; Haraldstad, K.; Raastad, T.; Køpp, U.; Haugeberg, G.; Mansoor, M.A.; Bastani, N.E.; Blomhoff, R.; Stølevik, S.B.; Seynnes, O.R.; Paulsen, G. Vitamin C and E supplementation blunts increases in total lean body mass in elderly men after strength training. Scand. J. Med. Sci. Sports 2016, 26, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Theodorou, A.A.; Nikolaidis, M.G.; Paschalis, V.; Kyparos, A.; Dipla, K.; Zafeiridis, A.; Panayiotou, G.; Vrabas, I.S. No effect of antioxidant supplementation on muscle performance and blood redox status adaptations to eccentric training. Am. J. Clin. Nutr. 2011, 93, 1373–1383. [Google Scholar] [CrossRef] [PubMed]
- Beyer, K.S.; Stout, J.R.; Fukuda, D.H.; Jajtner, A.R.; Townsend, J.R.; Church, D.D.; Wang, R.; Riffe, J.J.; Muddle, T.W.D.; Herrlinger, K.A.; et al. Impact of polyphenol supplementation on acute and chronic response to resistance training. J. Strength Cond. Res. 2017, 31, 2945–2954. [Google Scholar] [CrossRef]
- Martínez-Ferrán, M.; Berlanga, L.A.; Barcelo-Guido, O.; Matos-Duarte, M.; Vicente-Campos, D.; Sánchez-Jorge, S.; Romero-Morales, C.; Munguía-Izquierdo, D.; Pareja-Galeano, H. Antioxidant vitamin supplementation on muscle adaptations to resistance training: A double-blind, randomized controlled trial. Nutrition 2023, 105, 111848. [Google Scholar] [CrossRef]
- Medved, I.; Brown, M.J.; Bjorksten, A.R.; Murphy, K.T.; Petersen, A.C.; Sostaric, S.; Gong, X.; McKenna, M.J. N-acetylcysteine enhances muscle cysteine and glutathione availability and attenuates fatigue during prolonged exercise in endurance-trained individuals. J. Appl. Physiol. 2004, 97, 1477–1485. [Google Scholar] [CrossRef]
- Slattery, K.M.; Dascombe, B.; Wallace, L.K.; Bentley, D.J.; Coutts, A.J. Effect of N-acetylcysteine on cycling performance after intensified training. Med. Sci. Sports Exerc. 2014, 46, 1114–1123. [Google Scholar] [CrossRef]
- Fernández-Lázaro, D.; Domínguez-Ortega, C.; Busto, N.; Santamaría-Peláez, M.; Roche, E.; Gutiérrez-Abejón, E.; Mielgo-Ayuso, J. Influence of N-Acetylcysteine Supplementation on Physical Performance and Laboratory Biomarkers in Adult Males: A Systematic Review of Controlled Trials. Nutrients 2023, 15, 2463. [Google Scholar] [CrossRef]
- Kuehl, K.S.; Perrier, E.T.; Elliot, D.L.; Chesnutt, J.C. Efficacy of tart cherry juice in reducing muscle pain during running: A randomized controlled trial. J. Int. Soc. Sports Nutr. 2010, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Bell, P.G.; Stevenson, E.; Davison, G.W.; Howatson, G. The effects of Montmorency tart cherry concentrate supplementation on recovery following prolonged, intermittent exercise. Nutrients 2016, 8, 441. [Google Scholar] [CrossRef]
- Levers, K.; Dalton, R.; Galvan, E.; O’Connor, A.; Goodenough, C.; Simbo, S.; Mertens-Talcott, S.U.; Rasmussen, C.; Greenwood, M.; Riechman, S.; Crouse, S.; Kreider, R.B. Effects of powdered Montmorency tart cherry supplementation on acute endurance exercise performance in aerobically trained individuals. J. Int. Soc. Sports Nutr. 2016, 13, 22. [Google Scholar] [CrossRef] [PubMed]
- Trombold, J.R.; Reinfeld, A.S.; Casler, J.R.; Coyle, E.F. The effect of pomegranate juice supplementation on strength and soreness after eccentric exercise. J. Strength Cond. Res. 2011, 25, 1782–1788. [Google Scholar] [CrossRef]
- Ammar, A.; Turki, M.; Chtourou, H.; Hammouda, O.; Trabelsi, K.; Kallel, C.; Abdelkarim, O.; Hoekelmann, A.; Bouaziz, M.; Ayadi, F. Pomegranate Supplementation Accelerates Recovery of Muscle Damage and Soreness and Inflammatory Markers after a Weightlifting Training Session. PLoS ONE 2016, 11, e0160305. [Google Scholar] [CrossRef]
- Torregrosa-García, A.; et al. Pomegranate extract supplementation improves endurance and oxidative stress markers after aerobic exercise. Nutrients 2019, 11, 721. [Google Scholar] [CrossRef]
- Nicol, L.M.; Rowlands, D.S.; Fazakerly, R.; Kellett, J. Curcumin supplementation likely attenuates delayed onset muscle soreness and decreases muscle damage after eccentric exercise. Eur. J. Appl. Physiol. 2015, 115, 1769–1777. [Google Scholar] [CrossRef]
- Amalraj, A.; Divya, C.; Gopi, S. The effects of bioavailable curcumin (Cureit) on delayed onset muscle soreness induced by eccentric continuous exercise: A randomized, placebo-controlled, double-blind clinical study. J. Med. Food 2020, 23, 545–553. [Google Scholar] [CrossRef]
- Decroix, L.; Tonoli, C.; Soares, D.D.; Meeusen, R. Cocoa flavanols and exercise performance: A systematic review. Sports Med. 2018, 48, 867–892. [Google Scholar] [CrossRef] [PubMed]
- de Melo-Marins, D.; Farinha, J.B.; Rodrigues-Krause, J.; Laitano, O.; Reischak-Oliveira, A. Redox balance during exercise in the heat in healthy adults: A systematic review. J. Therm. Biol. 2021, 99, 102943. [Google Scholar] [CrossRef]
- Debevec, T.; Millet, G.P.; Pialoux, V. Hypoxia-Induced Oxidative Stress Modulation with Physical Activity. Front. Physiol. 2017, 8, 84. [Google Scholar] [CrossRef]
- Giorgini, P.; Rubenfire, M.; Bard, R.L.; Jackson, E.A.; Ferri, C.; Brook, R.D. Air Pollution and Exercise: A Review of the Cardiovascular Implications for Health Care Professionals. J. Cardiopulm. Rehabil. Prev. 2016, 36, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Stellingwerff, T.; Peeling, P.; Garvican-Lewis, L.A.; Hall, R.; Koivisto, A.E.; Heikura, I.A.; Burke, L.M. Nutrition and Altitude: Strategies to Enhance Adaptation, Improve Performance and Maintain Health: A Narrative Review. Sports Med. 2019, 49, 169–184. [Google Scholar] [CrossRef]
- Sims, S.T.; Kerksick, C.M.; Smith-Ryan, A.E.; Janse de Jonge, X.A.K.; Hirsch, K.R.; Arent, S.M.; Hewlings, S.J.; Kleiner, S.M.; Bustillo, E.; Tartar, J.L.; et al. International Society of Sports Nutrition Position Stand: Nutritional Concerns of the Female Athlete. J. Int. Soc. Sports Nutr. 2023, 20, 2204066. [Google Scholar] [CrossRef]
- Peeling, P.; McKay, A. Iron regulation and absorption in athletes: Contemporary thinking and recommendations. Curr. Opin. Clin. Nutr. Metab. Care 2023, 26, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Jeppesen, J.S.; Caldwell, H.G.; Lossius, L.O.; Melin, A.K.; Gliemann, L.; Bangsbo, J.; Hellsten, Y. Low energy availability increases immune cell formation of reactive oxygen species and impairs exercise performance in female endurance athletes. Redox Biol. 2024, 75, 103250. [Google Scholar] [CrossRef] [PubMed]
- Walsh, N.P. Nutrition and Athlete Immune Health: New Perspectives on an Old Paradigm. Sports Med. 2019, 49, 153–168. [Google Scholar] [CrossRef]
- Kölling, S.; Duffield, R.; Erlacher, D.; Venter, R.; Halson, S.L. Sleep-Related Issues for Recovery and Performance in Athletes. Int. J. Sports Physiol. Perform. 2019, 14, 144–148. [Google Scholar] [CrossRef]
- Cobley, J.N.; Close, G.L.; Bailey, D.M.; Davison, G.W. Exercise redox biochemistry: Conceptual, methodological and technical recommendations. Redox Biol. 2017, 12, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Impellizzeri, F.M.; Marcora, S.M.; Coutts, A.J. Internal and External Training Load: 15 Years On. Int. J. Sports Physiol. Perform. 2019, 14, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Hecksteden, A.; Kraushaar, J.; Scharhag-Rosenberger, F.; Theisen, D.; Senn, S.; Meyer, T. Individual response to exercise training—A statistical perspective. J. Appl. Physiol. 2015, 118, 1450–1459. [Google Scholar] [CrossRef] [PubMed]
- Swinton, P.A.; Hemingway, B.S.; Saunders, B.; Gualano, B.; Dolan, E. A Statistical Framework to Interpret Individual Response to Intervention: Paving the Way for Personalized Nutrition and Exercise Prescription. Front. Nutr. 2018, 5, 41. [Google Scholar] [CrossRef] [PubMed]
| Domain | Preferred measures (higher validity) |
Use with caution / pitfalls |
Timing & interpretation notes |
|---|---|---|---|
|
Lipid peroxidation |
F₂-isoprostanes (plasma/urine) | TBARS alone; MDA without validated methods |
Urine integrates over hours; pair with sampling windows and hydration control. |
|
DNA oxidation |
8-oxo-dG (urine/plasma); comet assay with oxidative enzymes |
Single time points without baseline; assay drift |
Interpret with baseline and workload; muscle-local events may not appear in blood. |
|
Protein oxidation/nitration |
Protein carbonyls; nitrotyrosine (with validated immunoassays) |
Non-specific antibody panels; lack of standards |
Best paired with functional outcomes; avoid over-interpreting small deltas. |
|
Redox couples & thiol status |
GSH/GSSG ratio; cysteine/cystine; oxidized/reduced peroxiredoxin states |
Hemolysis; delayed processing; unreported storage |
Requires strict processing and rapid quenching; biopsies improve specificity. |
|
Antioxidant capacity / enzymes |
SOD, GPx, catalase activity (standardized); targeted redox proteomics (if available) |
TAC/FRAP as sole endpoint; non-standard units |
Enzyme activity adapts with training; interpret as adaptation markers, not damage. |
|
Damage & inflammation (EIMD context) |
CK + myoglobin (with responder handling); IL-6/IL-10 panels; soreness + function tests |
CK alone (high variability); untimed cytokines |
Align with 24–72 h recovery window; report training load and prior activity. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




