Submitted:
09 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Definitions
2.2. National Database
2.3. U.S. Census Geographic Divisions
2.4. Statistics
2.5. Standard Inclusion/Exclusion Criteria
2.6. Artificial Intelligence and Web Searches
2.7. Units
2.8. Ethics
3. Results
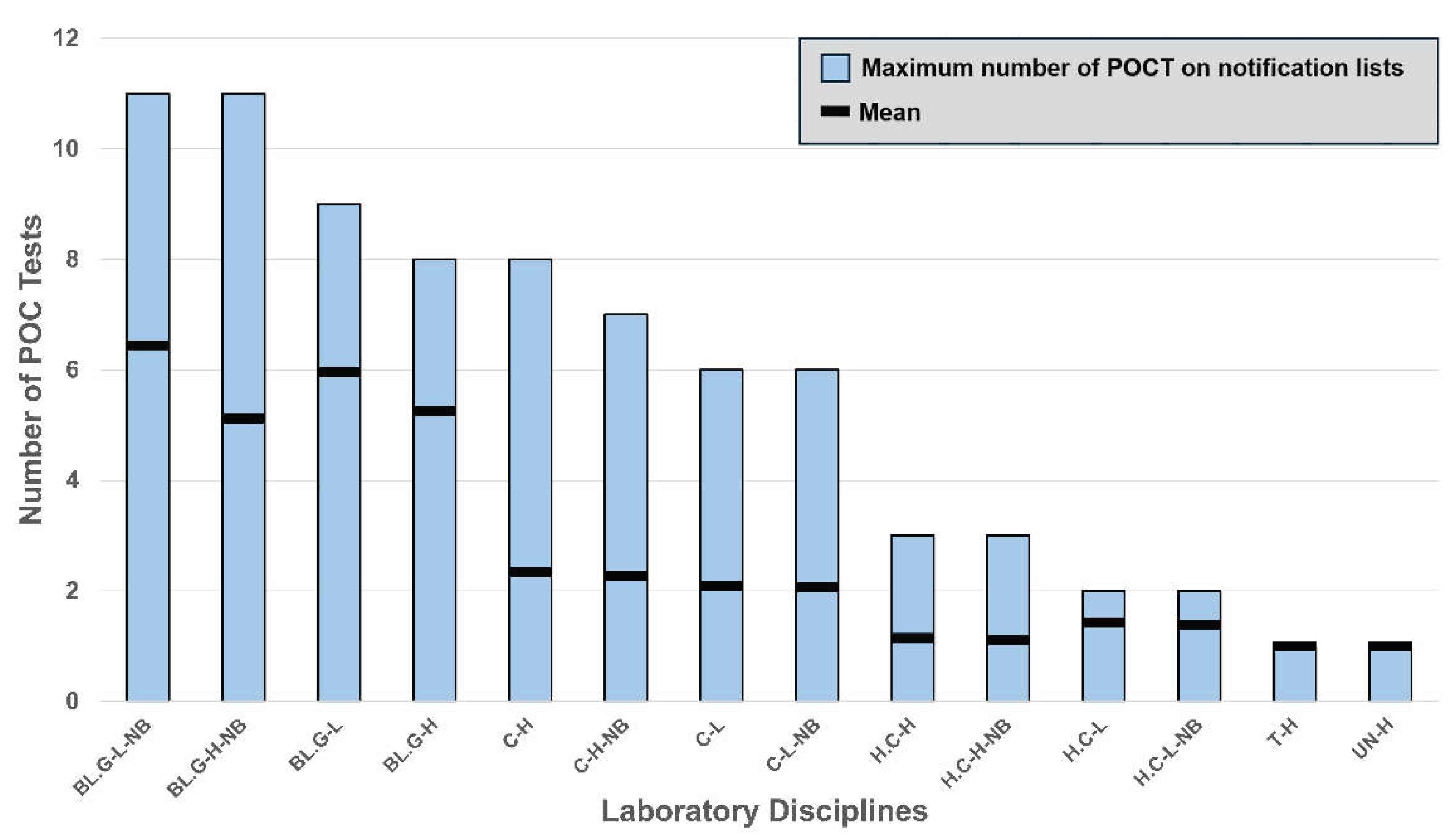
3.1. Number of POC Tests on Notification Lists
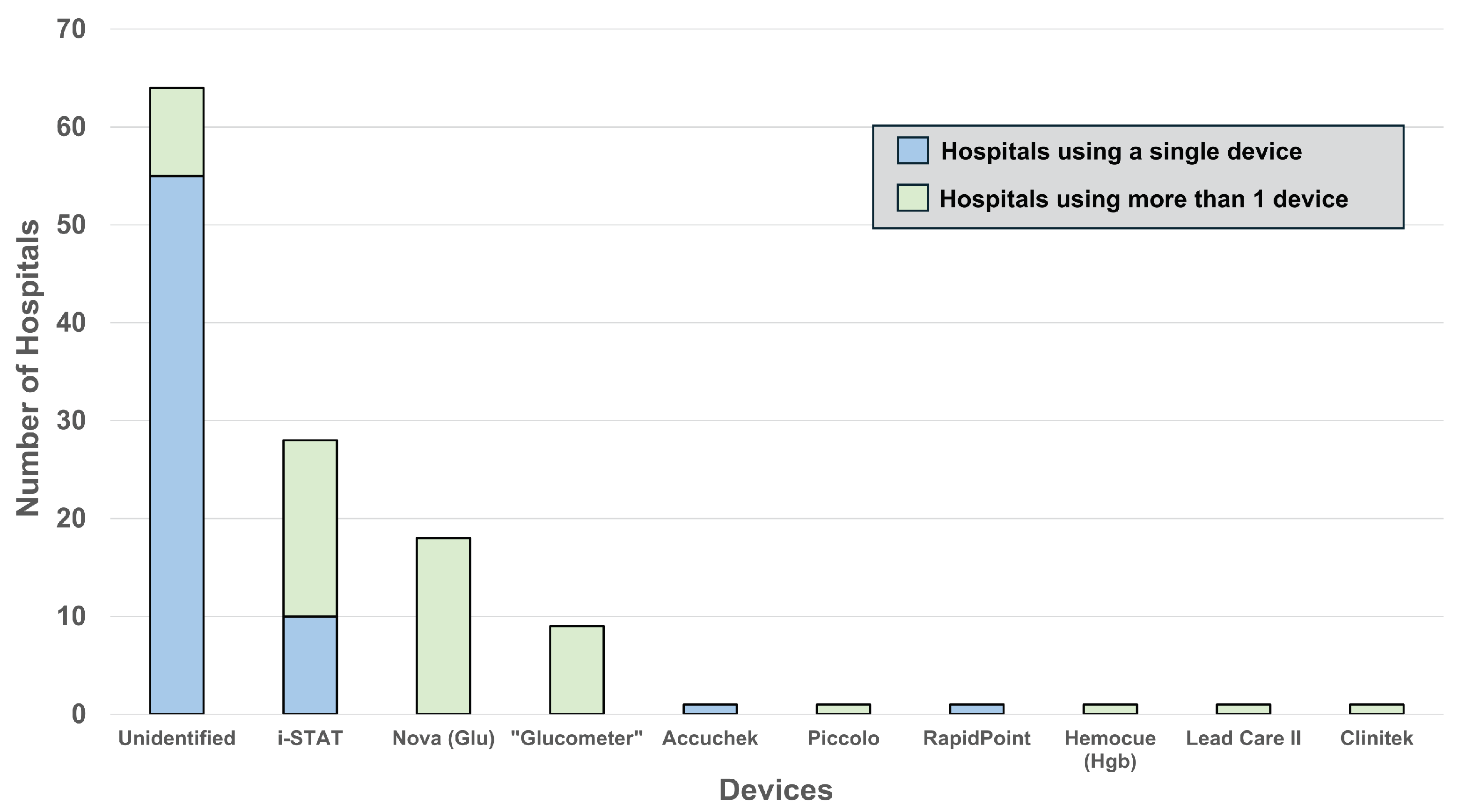
3.2. Instruments
3.3. Chemistry Tests
3.4. Blood Gas and pH Tests
3.5. Hematology and Coagulation Tests
3.6. POC versus Laboratory Matched Pairs
4. Discussion
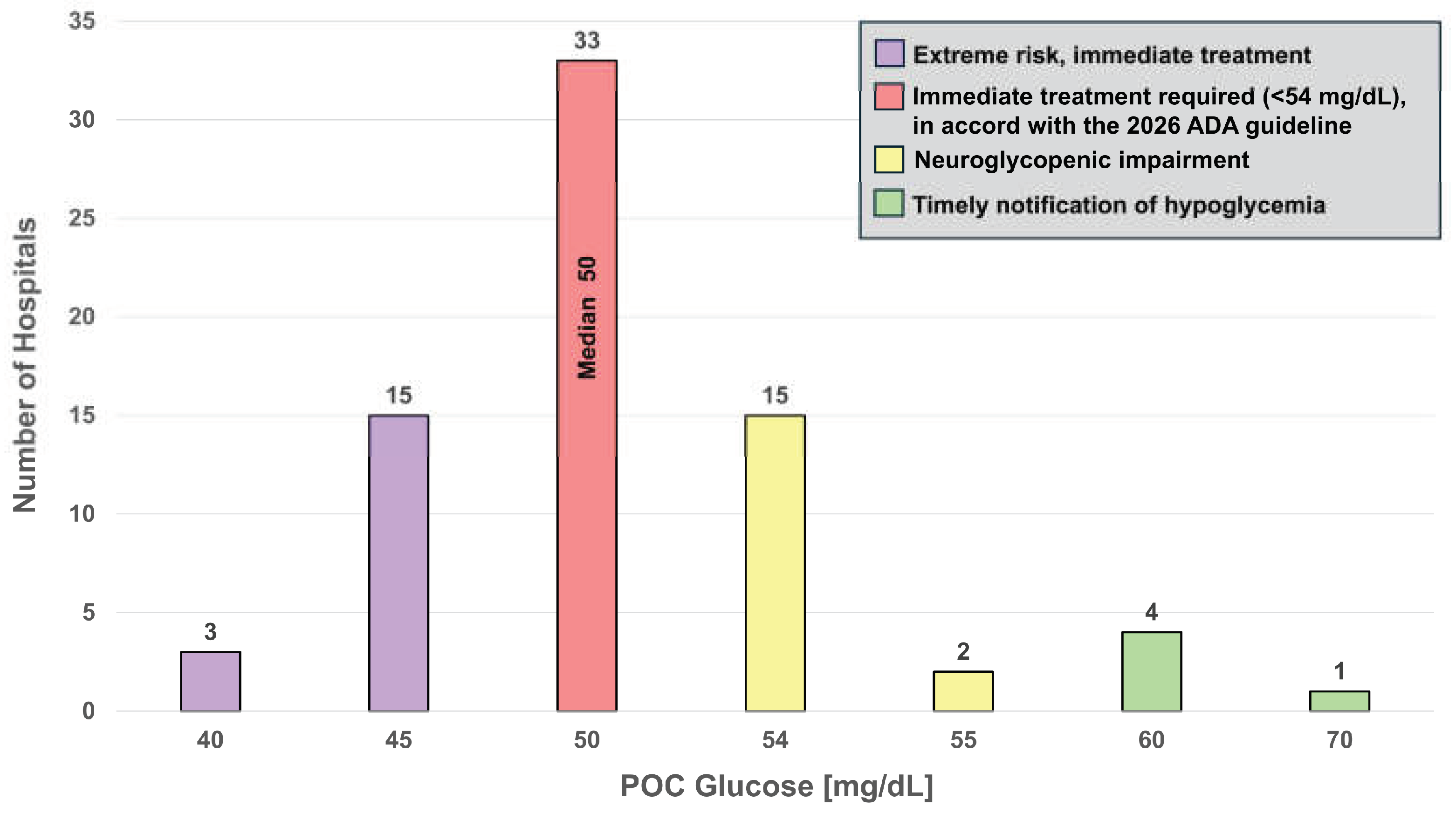
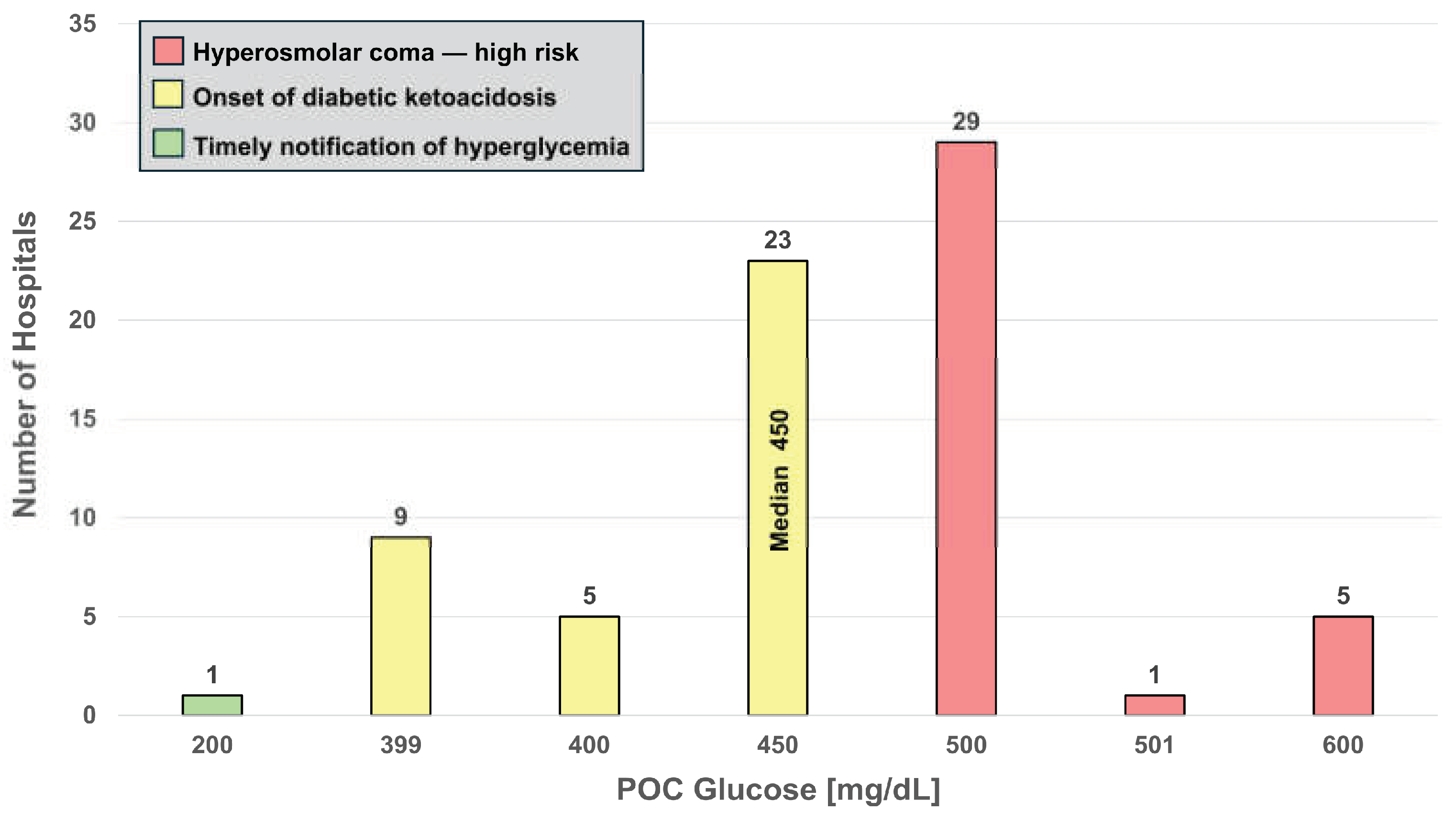
4.1. Glucose
4.2. Troponin
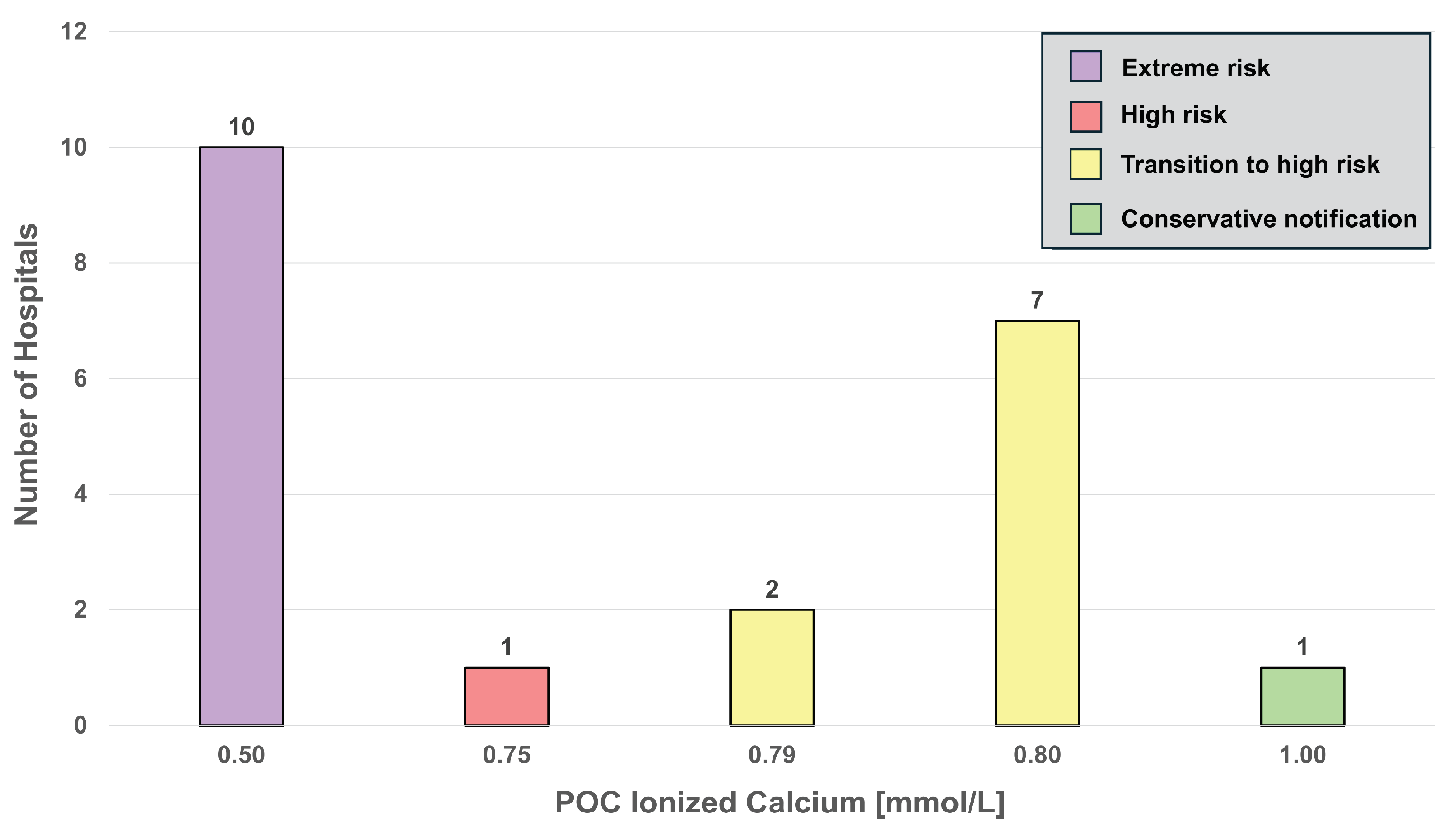
4.3. Ionized Calcium
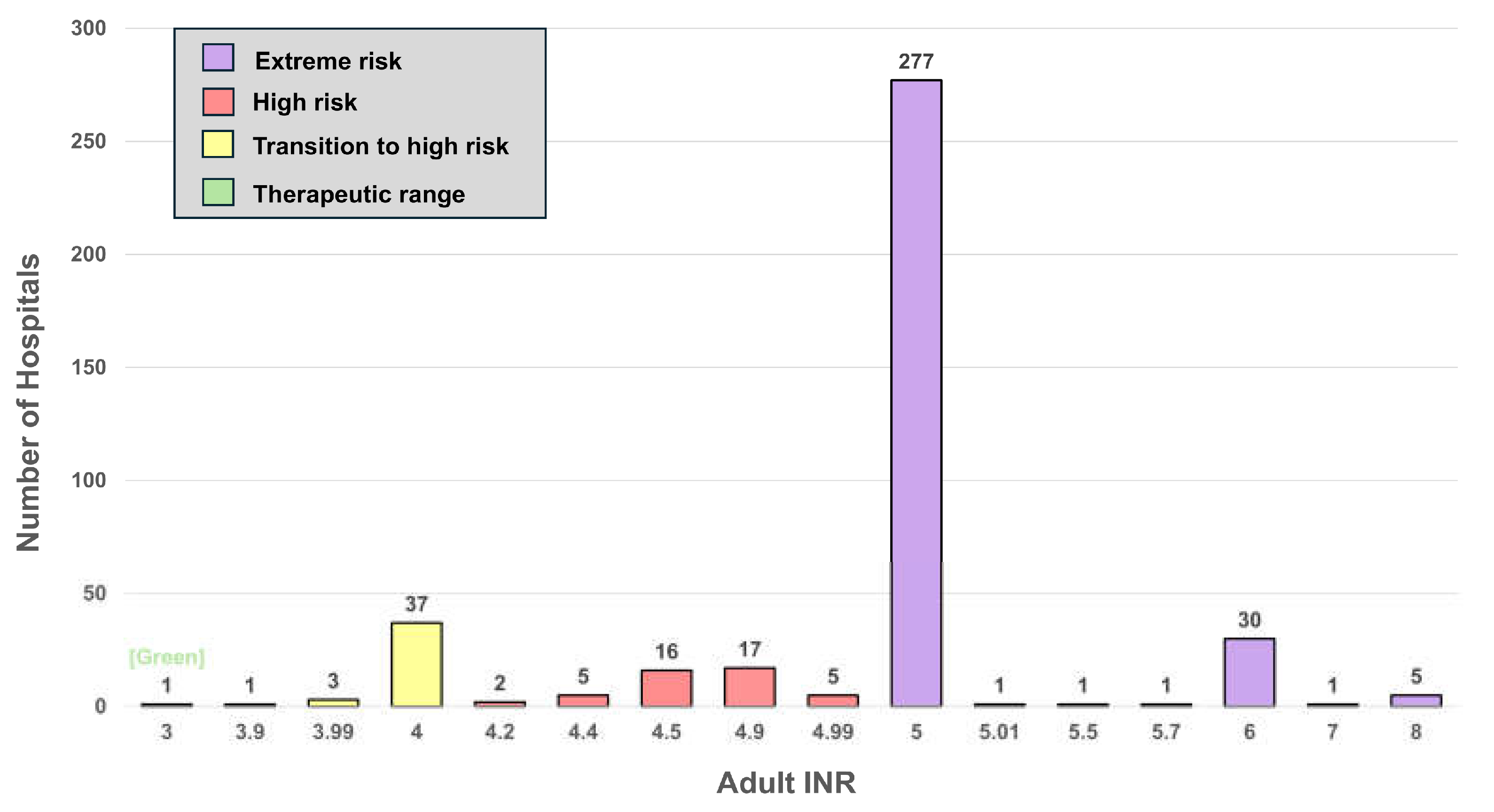
4.4. INR
4.5. Hemoglobin
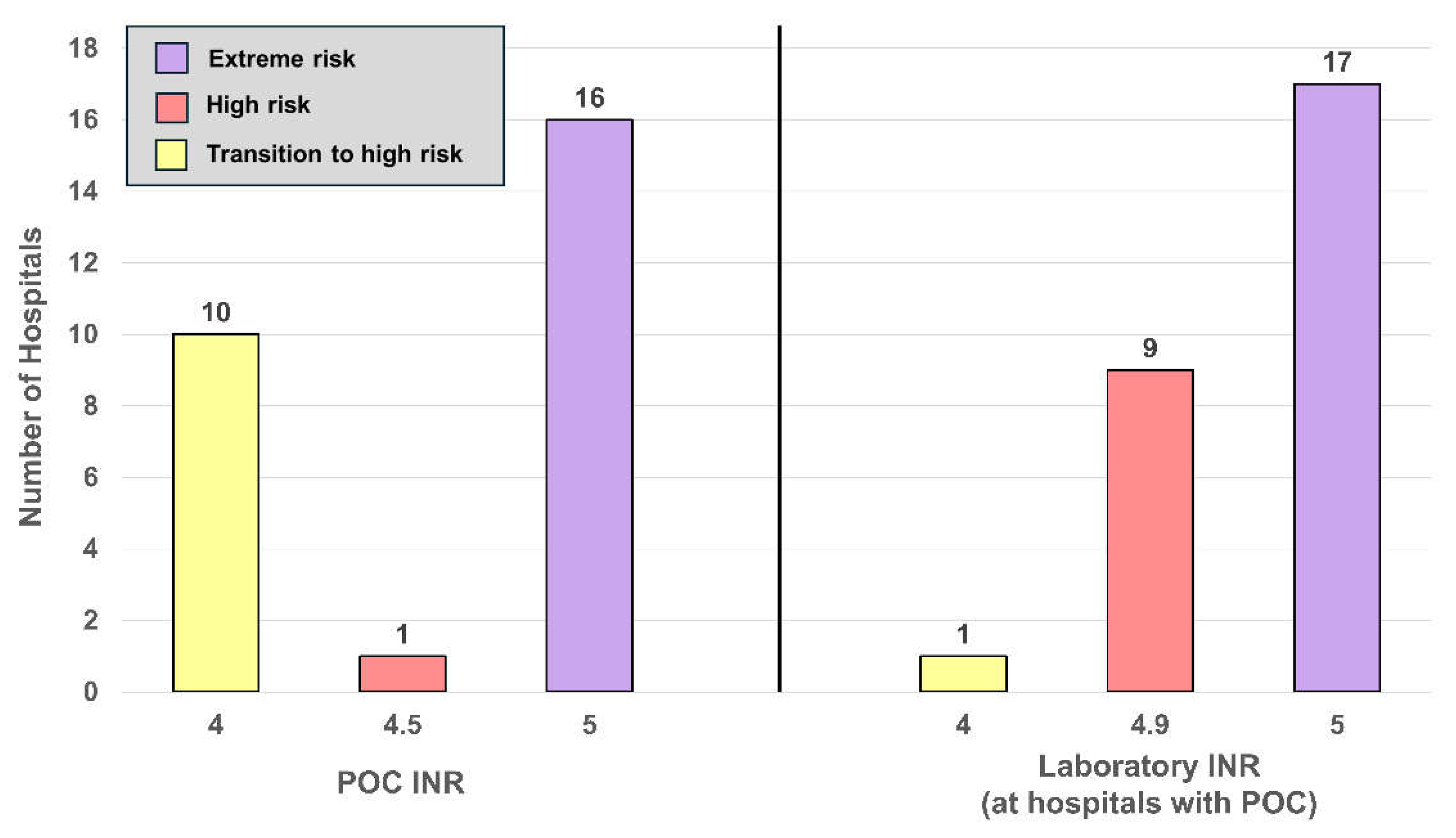
4.6. POC versus Laboratory Matched Pairs
4.7. pH
4.8. POC Adoption
4.9. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACS | Acute coronary syndrome |
| ADA | American Diabetes Association |
| AI | Artificial intelligence |
| CLSI | Clinical and Laboratory Standards Institute |
| COVID-19 | Coronavirus infectious disease-2019 |
| CTR | Center for Teaching and Research |
| ICH | Intracranial hemorrhage |
| INR | International normalized ratio |
| JASP | Jeffrey’s Amazing Statistics Program |
| MICU | Medical Intensive Care Unit |
| POC | Point-of-care |
| POCT | Point-of-care testing |
| TnI | Troponin I |
References
- The Joint Commission. National Patient Safety Goals Effective January 2026 for the Laboratory Program. Goal 2. NPSG.02.03.01 Report critical results of tests and diagnostic procedures on a timely basis. Available online: https://digitalassets.jointcommission.org/api/public/content/3c7a110c215943bc80d9ce87e9d9ee9d?v=aef80e14 (accessed on 17 December 2025).
- Augustine, S.; Venkadesh, A.; Kaushal, S.; Lee, E.; Ajaj, M.; Lee, N.E. Point-of-Care Testing: The Convergence of Innovation and Accessibility in Diagnostics. Anal. Chem. 2025, 97, 9569–9599. [Google Scholar] [CrossRef] [PubMed]
- Kost, G.J. Critical limits for urgent clinician notification at US medical centers. JAMA. 1990, 263, 704–707. [Google Scholar] [CrossRef] [PubMed]
- Parl, F.F.; O’Leary, M.F.; Kaiser, A.B.; Paulett, J.M.; Statnikova, K.; Shultz, E.K. Implementation of a closed-loop reporting system for critical values and clinical communication in compliance with goals of the joint commission. Clin Chem. 2010, 56, 417–423. [Google Scholar] [CrossRef] [PubMed]
- JASP Team. JASP (Version 0.19.3) Computer software. 2024. Available online: https://jasp-stats.org/ (accessed on 28 September 2025).
- American Diabetes Association Professional Practice Committee for Diabetes*. 6. Glycemic Goals, Hypoglycemia, and Hyperglycemic Crises: Standards of Care in Diabetes—2026. Diabetes Care 2026, 49 (Supplement_1), S132–S149. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association Professional Practice Committee for Diabetes*; 16. Diabetes Care in the Hospital: Standards of Care in Diabetes—2026. Diabetes Care 2026, 49 (Supplement_1), S339–S355. [Google Scholar] [CrossRef] [PubMed]
- Chapman, A.R.; Shah, A.S.V.; Lee, K.K.; Anand, A.; Francis, O.; Adamson, P.; McAllister, D.A.; Strachan, F.E.; Newby, D.E.; Mills, N.L. Long-Term Outcomes in Patients With Type 2 Myocardial Infarction and Myocardial Injury. Circulation. 2018, 137, 1236–1245. [Google Scholar] [CrossRef] [PubMed]
- Collet, J.P.; Thiele, H.; Barbato, E.; Barthélémy, O.; Bauersachs, J.; Bhatt, D.L.; Dendale, P.; Dorobantu, M.; Edvardsen, T.; Folliguet, T.; et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021, 42, 1289–1367. [Google Scholar] [CrossRef] [PubMed]
- Apple, F.S.; Smith, S.W.; Greenslade, J.H.; Sandoval, Y.; Parsonage, W.; Ranasinghe, I.; Gaikwad, N.; Schulz, K.; Stephensen, L.; Schmidt, C.W.; et al. Single High-Sensitivity Point-of-Care Whole-Blood Cardiac Troponin I Measurement to Rule Out Acute Myocardial Infarction at Low Risk. Circulation. 2022, 146, 1918–1929. [Google Scholar] [CrossRef] [PubMed]
- Pickering, J.W.; Young, J.M.; George, P.M.; Watson, A.S.; Aldous, S.J.; Troughton, R.W.; Pemberton, C.J.; Richards, A.M.; Cullen, L.A.; Than, M.P. Validity of a Novel Point-of-Care Troponin Assay for Single-Test Rule-Out of Acute Myocardial Infarction. JAMA Cardiol. 2018, 3, 1108–1112. [Google Scholar] [CrossRef] [PubMed]
- Kost, G.J.; Jammal, M.A.; Ward, R.E.; Safwat, A.M. Monitoring of ionized calcium during human hepatic transplantation. Critical values and their relevance to cardiac and hemodynamic management. Am J Clin Pathol. 1986, 86, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Farkas, J. Hypocalcemia. EMCrit Project, Internet Book of Critical Care (IBCC). 5 November 2025. Available online: https://emcrit.org/ibcc/hypocalcemia/ (accessed on 24 December 2025).
- Anastasopoulou, C. Calcium, Ionized; Medscape. July 2025. Available online: https://emedicine.medscape.com/article/2087469.
- Füzéry, A.K.; Kost, G.J.; Tran, N.K. Advances in Point-Of-Care Testing for Emergency Response, Infectious Diseases, and Critical Care — Novel Technologies, Practice Standards, and Improved Outcomes. In Encyclopedia of Analytical Chemistry; Meyers, R.A., Ed.; 2022. [Google Scholar] [CrossRef]
- Terecoasa, E.; Tiu, V.; Huertas, N.; Alonso de Leciñana, M. Oral anticoagulation related intracerebral hemorrhage: more questions than answers. Romanian Journal of Neurology. 2012, 11, 13–23. [Google Scholar] [CrossRef]
- Morotti, A.; Goldstein, J.N. Anticoagulant-associated intracerebral hemorrhage. Brain Hemorrhages 2020, 1, 89–94. [Google Scholar] [CrossRef]
- Shikdar, S.; Vashisht, R.; Zubair, M.; Bhattacharya, P.T. International Normalized Ratio: Assessment, Monitoring, and Clinical Implications. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, Florida, 2025; Available online: https://www.ncbi.nlm.nih.gov/books/NBK507707/.
- Cabral, K.P.; Fraser, G.L.; Duprey, J.; Gibbons, B.A.; Hayes, T.; Florman, J.E.; Seder, D.B. Prothrombin complex concentrates to reverse warfarin-induced coagulopathy in patients with intracranial bleeding. Clin Neurol Neurosurg. 2013, 115, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Montoro, M.; Cucala, M.; Lanas, Á.; Villanueva, C.; Hervás, A.J.; Alcedo, J.; Gisbert, J.P.; Aisa, Á.P.; Bujanda, L.; Calvet, X.; et al. Indications and hemoglobin thresholds for red blood cell transfusion and iron replacement in adults with gastrointestinal bleeding: An algorithm proposed by gastroenterologists and patient blood management experts. Front Med (Lausanne). 2022, 9. [Google Scholar] [CrossRef] [PubMed]
- Birlie Chekol, W.; Teshome, M.; Nigatu, Y.A.; Melesse, D.Y. Hemoglobin threshold and clinical predictors for perioperative blood transfusion in elective surgery: Systemic review. Trends in Anaesthesia and Critical Care. 2020, 31, 8–15. [Google Scholar] [CrossRef]
- Badireddy, M.; Baradhi, K.M. Chronic Anemia. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, Florida, 2023; Available online: https://www.ncbi.nlm.nih.gov/books/NBK534803/.
- Crowther, M.; Podolak-Dawidziak, M. Anemia: General Considerations. In McMaster Textbook of Internal Medicine; Medycyna Praktyczna: Kraków, Poland, 2021; Available online: https://empendium.com/mcmtextbook/chapter/B31.II.15.1 (accessed on 1 January 2026).
- Nichols, J.H.; Alter, D.; Chen, Y.; Isbell, T.S.; Jacobs, E.; Moore, N.; Shajani-Yi, Z. AACC Guidance Document on Management of Point-of-Care Testing. The Journal of Applied Laboratory Medicine. 2020, 5, 762–787. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.W.; Thomas, B.W.; Christmas, A.B.; Cunningham, K.W.; Sing, R.F. Returning from the acidotic abyss: Mortality in trauma patients with a pH <7.0. Am J Surg. 2017, 214, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Rosival, V. Dangers of very low blood pH. Indian J Crit Care Med. 2011, 15, 194. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, D.A. Interpretation of Arterial Blood Gases (ABGs). American Thoracic Society. 2024. Available online: https://www.thoracic.org/professionals/clinical-resources/critical-care/clinical-education/abgs.php (accessed on 25 December 2025).
- Chaisirin, W.; Wongkrajang, P.; Thoesam, T.; Praphruetkit, N.; Nakornchai, T.; Riyapan, S.; Ruangsomboon, O.; Laiwejpithaya, S.; Rattanathummawat, K.; Pavichai, R.; et al. Role of Point-of-Care Testing in Reducing Time to Treatment Decision-Making in Urgency Patients: A Randomized Controlled Trial. West J Emerg Med. 2020, 21, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Clinical Laboratory and Standards (CLSI). Measurement Procedure Comparison and Bias Estimation Using Patient Samples. In CLSI guideline EP09, 3rd ed.; Clinical and Laboratory Standards Institute: Wayne, PA, 2018; Available online: https://clsi.org/shop/standards/ep09/ (accessed on 22 December 2025).
- Khan, A.I.; Vasquez, Y.; Gray, J.; Wians, F.H., Jr.; Kroll, M.H. The variability of results between point-of-care testing glucose meters and the central laboratory analyzer. Arch Pathol Lab Med. 2006, 130, 1527–1532. [Google Scholar] [CrossRef] [PubMed]
| Measurand | Number of Hospitals (%) |
Units | Low Mean (SD) |
Low Median (Range) |
High Mean (SD) |
High Median (Range) |
|---|---|---|---|---|---|---|
| 1.A. Adults | ||||||
| Glucose |
73 (17.5) |
mg/dL | 50.3 (4.9) |
50 (40–70) |
467.7 (60.5) |
450 (200-600) |
| mmol/L | 2.8 (0.3) |
2.8 (2.2-3.9) |
26 (3.4) |
25 (11.1-33.3) |
||
| Ionized calcium | 21 (5) |
mmol/L | 0.66 (0.17) |
0.75 (0.50-1.00) |
1.65 (0.10) |
1.60 (1.40-1.75) |
| Potassium | 20 (4.8) |
mmol/L | 2.9 (0.2) |
3 (2.5-3) |
6.1 (0.2) |
6 (6-6.5) |
| Sodium | 18 (4.3) |
mmol/L | 123.6 (3) |
126 (120-126) |
156.7 (2.4) |
155 (155-160) |
| CO2 content | 17 (4.1) |
mmol/L | 10.7 (0.6) |
11 (10-12) |
40 (1.9) |
39 (39-45) |
| Lactic Acid | 14 (3.4) |
mmol/L | … | … | 3.6 (0.6) |
3.9 (2-4) |
|
Calcium |
11 (2.6) |
mg/dL | 6.5 (0.2) |
6.6 (6-6.6) |
13 (0.3) |
12.9 (12.9-14) |
| mmol/L | 1.64 (0.05) |
1.65 (1.5-1.65) |
3.25 (0.08) |
3.23 (3.23-3.5) |
||
| Venous Lactic Acid | 10 (2.4) |
mmol/L | … | … | 3.9 (0) |
3.9 (3.9-3.9) |
| 1.B. Newborns | ||||||
| Glucose |
62 (14.9) |
mg/dL | 44.7 (5) |
45 (40-54) |
227.7 (87) |
200 (125-500) |
| mmol/L | 2.5 (0.3) |
2.5 (2.2-3) |
12.6 (4.8) |
11.1 (6.9-27.8) |
||
| Ionized calcium | 18 (4.3) |
mmol/L | 0.86 (0.05) |
0.90 (0.80-0.90) |
1.41 (0.14) |
1.30 (1.30-1.60) |
| Potassium | 15 (3.6) |
mmol/L | 3 (0.1) |
3 (2.8-3) |
6.8 (0.5) |
7.1 (6-7.1) |
| Sodium | 14 (3.4) |
mmol/L | 127.5 (4.3) |
130 (120-130) |
151.1 (2.9) |
150 (150-160) |
| CO2 content | 12 (2.9) |
mmol/L | 11 (0.4) |
11 (10-12) |
39.2 (0.4) |
39 (39-40) |
|
Calcium |
11 (2.6) |
mg/dL | 6.9 (0.3) |
7 (6-7) |
13 (0.3) |
12.9 (12.9-14) |
| mmol/L | 1.73 (0.08) |
1.75 (1.5-1.75) |
3.25 (0.08) |
3.23 (3.23-3.5) |
||
| Bilirubin | 10 (2.4) |
mg/dL | … | … | 17.9 (0) |
17.9 (17.9-17.9) |
| µmol/L | … | … | 306.1 (0) |
306.1 (306.1-306.1) |
||
| Measurand | Number of Hospitals (%) | Units | Low Mean (SD) |
Low Median (Range) |
High Mean (SD) |
High Median (Range) |
|---|---|---|---|---|---|---|
| 2.A. Adults | ||||||
| pH | Low: 29 (7) High: 28 (6.7) |
pH Units | 7.20 (0.06) |
7.20 (7.00-7.30) |
7.59 (0.04) |
7.60 (7.50-7.65) |
| Arterial pO2 | Low: 27 (6.5) High: 1 (0.2) |
mm Hg | 45 (5.9) |
40 (40-55) |
200 (…) |
200 (200-200) |
| kPa | 6 (0.8) |
5.3 (5.3-7.3) |
26.7 (…) |
26.7 (26.7-26.7) |
||
| Arterial pCO2 | Low: 20 (4.8) High: 21 (5) |
mm Hg | 20.3 (1.1) |
20 (20-25) |
67.1 (5.1) |
70 (50-70) |
| kPa | 2.7 (0.1) |
2.7 (2.7-3.3) |
8.9 (0.7) |
9.3 (6.7-9.3) |
||
| Carboxyhe-moglobin | 21 (5) |
% | … | … | 12.6 (4.4) |
10 (10-20) |
| Methemo-globin | 20 (4.8) |
% | … | … | 4.5 (1.8) |
4 (3-10) |
| Venous pH | 20 (4.8) |
pH Units | 7.21 (0.02) |
7.20 (7.20-7.30) |
7.58 (0.05) |
7.60 (7.50-7.65) |
| Venous pCO2 | Low: 15 (3.6) High: 17 (4.1) |
mm Hg | 30 (7.3) |
35 (20-35) |
68.5 (4.1) |
71 (56-71) |
| kPa | 4 (1) |
4.7 (2.7-4.7) |
9.1 (0.5) |
9.5 (7.5-9.5) |
||
| Arterial O2 Saturation | 11 (2.6) |
% | 76.8 (10.6) |
80 (45-80) |
… | … |
| Venous O2 Saturation | 10 (2.4) |
% | 60 (0) |
60 (60-60) |
… | … |
| Arterial O2 Hemoglobin | 10 (2.4) |
% | 79.5 (4) |
82 (75-85) |
… | … |
| Venous pO2 | Low: 10 (2.4) High: 1 (0.2) |
mm Hg | 32 (0) |
32 (32-32) |
200 (…) |
200 (200-200) |
| kPa | 4.3 (0) |
4.3 (4.3-4.3) |
26.7 (…) |
26.7 (26.7-26.7) |
||
| 2.B. Newborns | ||||||
| pH | Low: 25 (6) High: 20 (4.8) |
pH Units | 7.19 (0.04) |
7.20 (7.00-7.27) |
7.59 (0.05) |
7.60 (7.45-7.65) |
| Arterial pO2 | Low: 23 (5.5) High: 1 (0.2) |
mm Hg | 45 (9.2) |
40 (30-60) |
200 (…) |
200 (200-200) |
| kPa | 6 (1.2) |
5.3 (4-8) |
26.7 (…) |
26.7 (26.7-26.7) |
||
| Arterial pCO2 | Low: 17 (4.1) High: 22 (5.3) |
mm Hg | 23.8 (3.8) |
25 (20-35) |
70.9 (7.2) |
70 (50.1-80) |
| kPa | 3.2 (0.5) |
3.3 (2.7-4.7) |
9.5 (1) |
9.3 (6.7-10.7) |
||
| Carboxyhe-moglobin | 20 (4.8) |
% | … | … | 11.5 (5.1) |
9 (8-20) |
| Venous pH | 19 (4.6) |
pH Units | 7.20 (0) |
7.20 (7.20-7.20) |
7.60 (0.04) |
7.60 (7.55-7.65) |
| Venous pO2 | Low: 10 (2.4) High: 1 (0.2) |
mmHg | 32 (0) |
32 (32-32) |
200 (…) |
200 (200-200) |
| kPa | 4.3 (0) |
4.3 (4.3-4.3) |
26.7 (…) |
26.7 (26.7-26.7) |
||
| Arterial O2 Saturation | 10 (2.4) |
% | 80 (0) |
80 (80-80) |
… | … |
| Venous O2 Saturation | 10 (2.4) |
% | 60 (0) |
60 (60-60) |
… | … |
| Methemo-globin | 10 (2.4) |
% | … | … | 6 (1.5) |
6 (5-10) |
| Venous pCO2 | 9 (2.2) |
mmHg | 22.8 (2.6) |
25 (20-25) |
62.2 (2.6) |
60 (60-65) |
| kPa | 3 (0.4) |
3.3 (2.7-3.3) |
8.3 (0.4) |
8 (8-8.7) |
||
| Capillary pH | Low: 9 (2.2) High: 4 (1) |
pH Units | 7.20 (0) |
7.2 (7.20-7.20) |
7.65 (0) |
7.65 (7.65-7.65) |
| Capillary pCO2 | Low: 4 (1) High: 9 (2.2) |
mmHg | 20 (0) |
20 (20-20) |
73.3 (7.9) |
80 (65-80) |
| kPa | 2.7 (0) |
2.7 (2.7-2.7) |
9.8 (1.1) |
10.7 (8.7-10.7) |
||
| Arterial O2 Hemoglobin | 9 (2.2) |
% | 78.9 (3.7) |
82 (75-82) |
… | … |
| Measurand | Number of Hospitals (%) |
Units | Low Mean (SD) |
Low Median (Range) |
High Mean (SD) |
High Median (Range) |
|---|---|---|---|---|---|---|
| 3.A. Adults | ||||||
| INR | 27 (6.5) |
INR | … | … | 4.6 (0.5) |
5 (4-5) |
| Hematocrit | Low: 24 (5.8) High: 12 (2.9) |
% | 19.7 (1.5) |
20 (15-21) |
61.3 (3.1) |
60 (55-65) |
| Hemoglobin | Low: 23 (5.5) High: 6 (1.4) |
g/dL | 6.8 (0.7) |
6.6 (6-8) |
19.1 (0.4) |
18.9 (18.9-20) |
| 3.B. Newborns | ||||||
| Hematocrit | Low: 21 (5) High: 10 (2.4) |
% | 25.3 (5.1) |
25 (18-30) |
68 (6.3) |
65 (60-75) |
| Hemoglobin | Low: 18 (4.3) High: 7 (1.7) |
g/dL | 8.8 (1.5) |
10 (6-10) |
19.6 (1.5) |
18.9 (18.9-23) |
| INR | 12 (2.9) |
INR | … | … | 4.1 (0.3) |
4 (4-5) |
| Measurand, High or Low Critical Limit | Number of Hospitals |
Units | p-value | POC Median (Range) |
Laboratory Median (Range) |
|---|---|---|---|---|---|
| 4.A. Adults | |||||
| INR, High | 27 | INR | 0.003 | 5 (4-5) |
5 (4-5) |
| Hemoglobin, Low | 23 | g/dL | 0.048 | 6.6 (6-8) |
6.6 (6-7) |
| Ionized calcium, Low | 21 | mmol/L | 0.002 | 0.75 (0.50-1.00) |
0.76 (0.75-0.85) |
| Ionized calcium, High | 21 | mmol/L | 0.004 | 1.60 (1.40-1.75) |
1.59 (1.40-1.62) |
| 4.B. Newborns | |||||
| INR, High | 10 | INR | 0.003 | 4 (4-4.5) |
4.9 (4.9-5) |
| Measurand | Number of Hospitals |
POC < Laboratory | POC = Laboratory | POC > Laboratory | |||
|---|---|---|---|---|---|---|---|
| Low | High | Low | High | Low | High | ||
| 5.A. Clinical Chemistry | |||||||
| Adults | |||||||
| Glucose | 73 | 2 | 4 | 64 | 67 | 7 | 2 |
| Ionized calcium | 21 | 13 | 3 | 7 | 8 | 1 | 10 |
| Potassium | 20 | … | … | 20 | … | … | … |
| Sodium | 18 | … | 1 | 18 | 16 | … | 1 |
| CO2 content | Low: 16 High: 15 | … | … | 15 | 15 | 1 | … |
| Lactic Acid | 14 | … | … | … | 13 | … | 1 |
| Calcium | 11 | … | … | 11 | 10 | … | 1 |
| Newborns | |||||||
| Glucose | 62 | 3 | 3 | 52 | 56 | 7 | 3 |
| Ionized calcium | 17 | … | … | 17 | 16 | … | 1 |
| Potassium | 14 | … | 1 | 14 | 13 | … | … |
| Sodium | 12 | … | 1 | 12 | 11 | … | … |
| CO2 content | 12 | … | … | 12 | 11 | … | 1 |
| Calcium | 11 | … | … | 11 | 10 | … | 1 |
| Newborn Bilirubin | 10 | … | … | … | 10 | … | … |
| 5.B. Blood Gas and pH | |||||||
| Adults | |||||||
| pH | Low: 18 High:17 | 1 | 2 | 16 | 15 | 1 | … |
| Arterial pO2 | 16 | … | … | 15 | … | 1 | … |
| Carboxyhemo- globin |
11 | … | … | … | 11 | … | … |
| Arterial pCO2 | 10 | … | 1 | … | 9 | … | … |
| Methemoglobin | 10 | … | … | … | 10 | … | … |
| Venous pH | 10 | … | 1 | 9 | 9 | 1 | … |
| Newborns | |||||||
| pH | 11 | … | … | 11 | … | … | … |
| Arterial pO2 | 11 | … | … | 10 | … | 1 | … |
| Arterial pCO2 | 11 | … | … | 11 | … | … | … |
| Carboxyhemo- globin |
10 | … | … | … | 10 | … | … |
| Methemoglobin | 10 | … | … | … | 10 | … | … |
| 5.C. Hematology and Coagulation | |||||||
| Adults | |||||||
| INR | 27 | … | 10 | … | 17 | … | … |
| Hemoglobin | 23 | … | 18 | … | 5 | … | |
| Hematocrit | 15 | … | … | 15 | … | … | … |
| Newborns | |||||||
| Hemoglobin | 18 | 1 | … | 17 | … | … | … |
| Hematocrit | 13 | 1 | … | 12 | … | … | … |
| INR | 10 | … | 10 | … | … | … | … |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




