Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
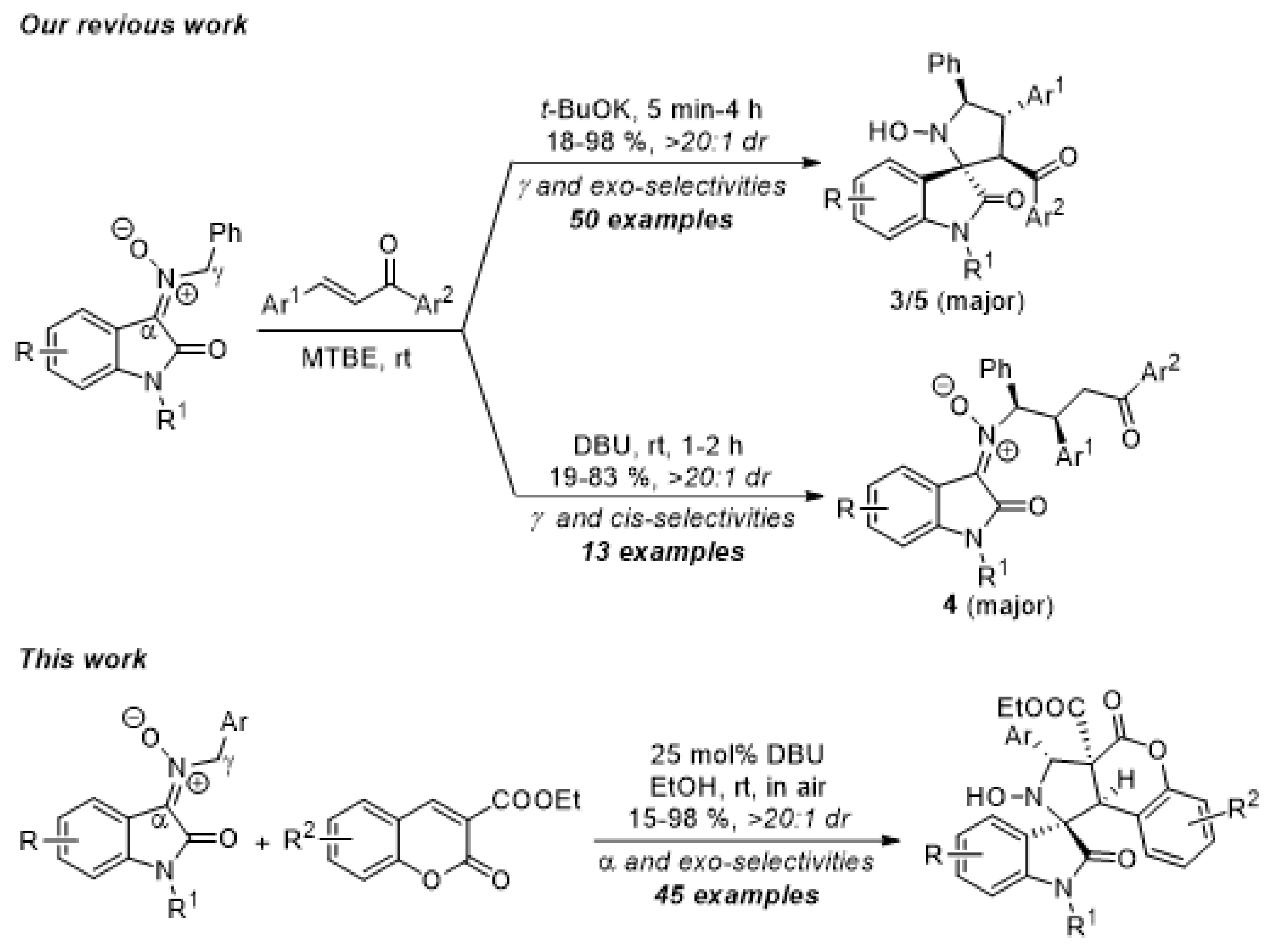
1. Introduction
2. Results and Discussion
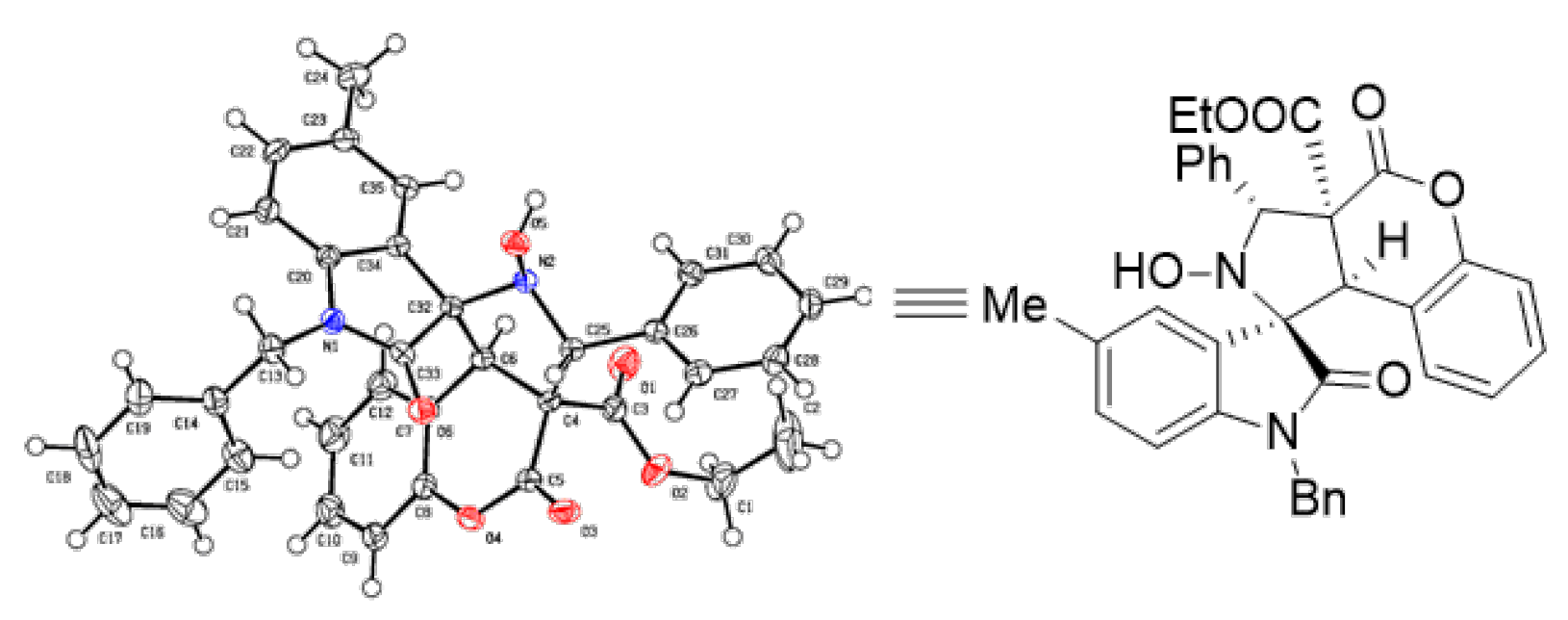
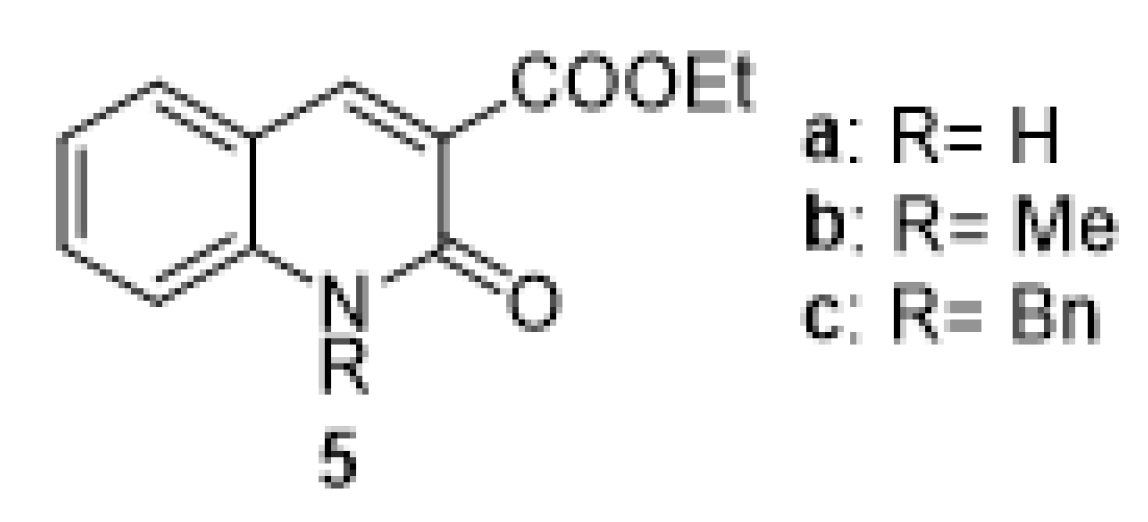
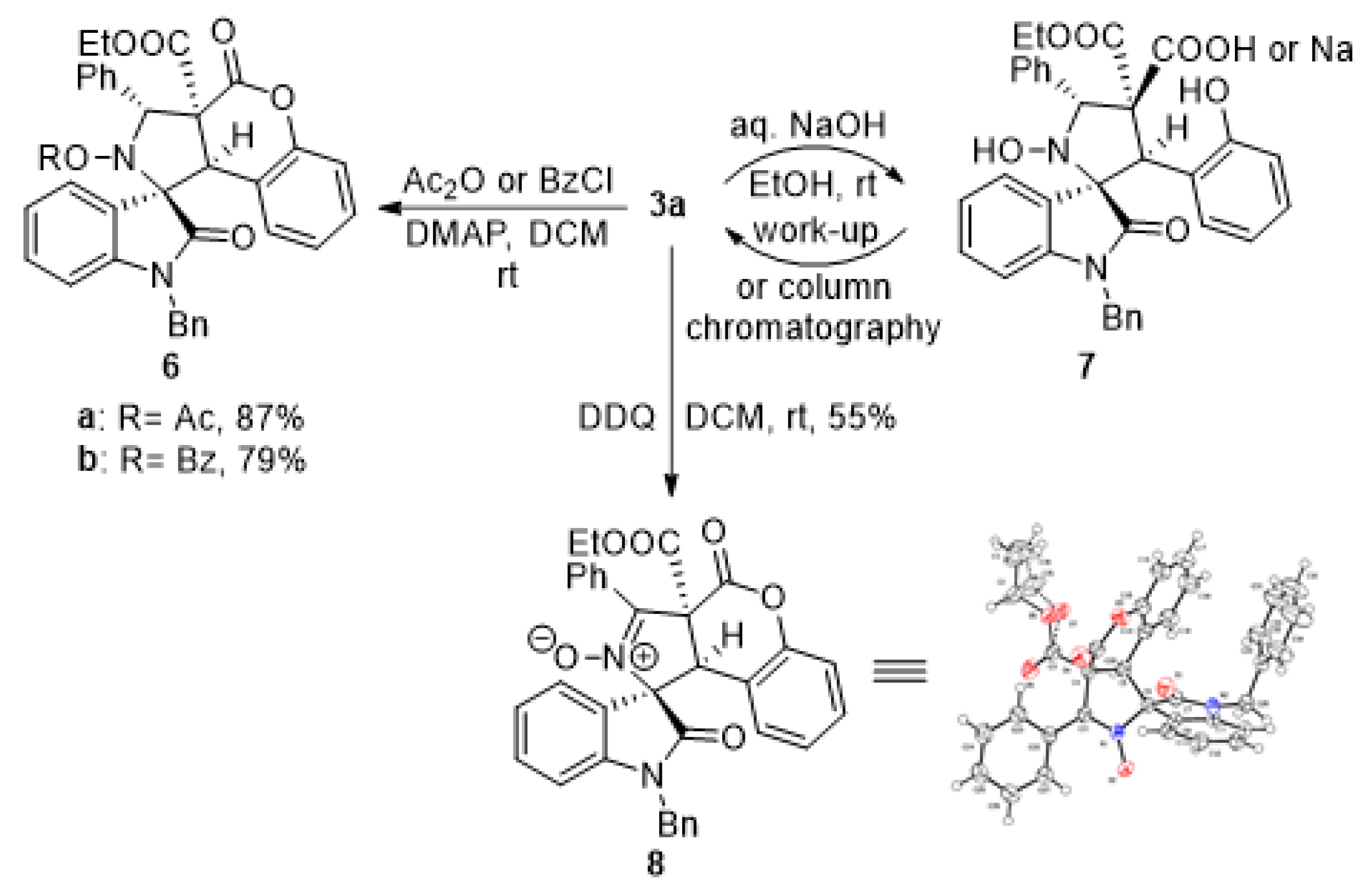
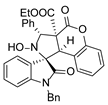
3. Materials and Methods
3.1. General Methods
3.2. Preparation of Intermediates
3.3. General Procedure for Condition Optimization
3.4. General Procedure for Typical Procedure for Cycloaddition
3.5. The Phenomenon of the Reaction and TLC
3.6. Deriverziation of 3a
3.7. Data for All New Compounds
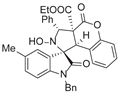
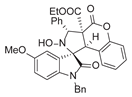
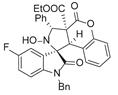
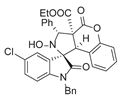
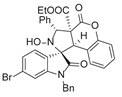
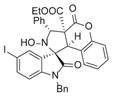
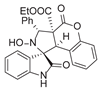
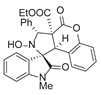
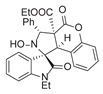
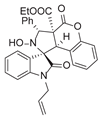
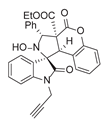
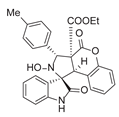
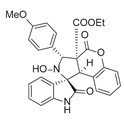
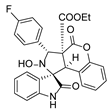
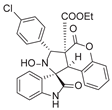
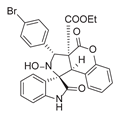
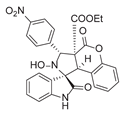
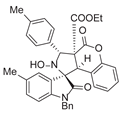
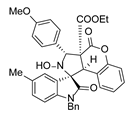
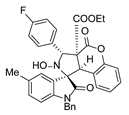
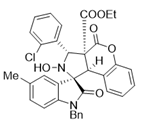
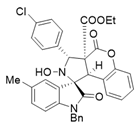
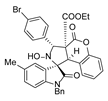
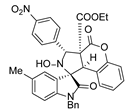
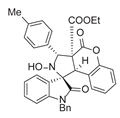
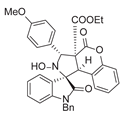
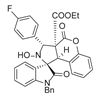
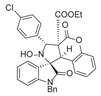
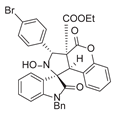
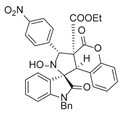
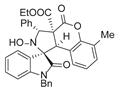
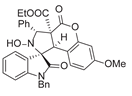
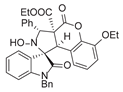
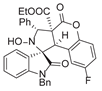
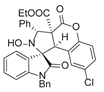
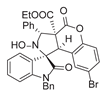
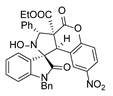
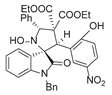
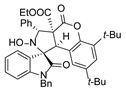
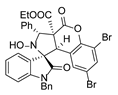
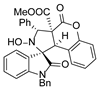
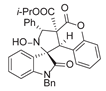
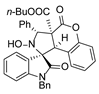
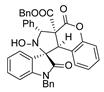
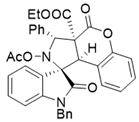
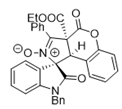
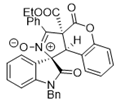
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sardari, S.; Nishibe, S.; Daneshtalab, M. Coumarins, the bioactive structures with antifungal property. In Stud. Nat. Prod. Chem.; Attaur, R., Ed.; Elsevier, 2000; vol. 23, pp. 335–393. [Google Scholar]
- Estévez-Braun, A.; González, A. G. Coumarins. Nat. Prod. Rep. 1997, 14, 465–475. [Google Scholar] [CrossRef]
- Malikov, V. M.; Saidkhodzhaev, A. I.; Aripov, K. N. Coumarins: Plants, structure, properties. Chem. Nat. Compd. 1998, 34, 202–264. [Google Scholar] [CrossRef]
- Malikov, V. M.; Saidkhodzhaev, A. I. Coumarins. Plants, structure, properties. Chem. Nat. Compd. 1998, 34, 345–409. [Google Scholar] [CrossRef]
- Malikov, V. M.; Saidkhodzhaev, A. I. Coumarins: Plants, structures, properties. Chem. Nat. Compd. 1998, 34, 517–548. [Google Scholar]
- Kong, L. Coumarin chemistry. In Natural Product Chemistry Series; (in Chinese). Chemical Industry Press, 2007. [Google Scholar]
- Nan, Z.-D.; Shang, Y.; Zhu, Y.-D.; Zhang, H.; Sun, R.-R.; Tian, J.-J.; Jiang, Z.-B.; Ma, X.-L.; Bai, C. Systematic review of natural coumarins in plants (2019–2024): chemical structures and pharmacological activities. Phytochemistry 2025, 235, 114480. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Su, M.; Yuan, Z.; Deng, S.; Yu, L.; Wang, H. Research progress on the application of chemically synthesized coumarin derivatives. Chem. Res. Appl. (in Chinese). 2026, 38, 1–10. [Google Scholar]
- Barot, K. P.; Jain, S. V.; Kremer, L.; Singh, S.; Ghate, M. D. Recent advances and therapeutic journey of coumarins: current status and perspectives. Med. Chem. Res. 2015, 24, 2771–2798. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Cruz-Martins, N.; López-Jornet, P.; Lopez, E. P.-F.; Harun, N.; Yeskaliyeva, B.; Beyatli, A.; Sytar, O.; Shaheen, S.; Sharopov, F.; Taheri, Y.; Docea, A. O.; Calina, D.; Cho, W. C. Natural coumarins exploring the pharmacological. Oxid. Med. Cell. Longev. 2021, 2021, 6492346. [Google Scholar] [CrossRef]
- Croce, M.; Gallo, N.; Arienzo, V.; Salvatore, A.; Antonini, G. Coumarin-induced hepatotoxicity: a narrative review. Molecules 2022, 27, 9063. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Sharma, A.; Wadje, B. N.; Bharate, S. B. Benzopyrone, a privileged scaffold in drug discovery: an overview of FDA-approved drugs and clinical candidates. Med. Chem. Res. 2024, 44, 2035–2077. [Google Scholar] [CrossRef]
- Hussain, M. K.; Khatoon, S.; Khan, M. F.; Akhtar, M. S.; Ahamad, S.; Saquib, M. Coumarins as versatile therapeutic phytomolecules: a systematic review. Phytomedicine 2024, 134, 155972. [Google Scholar] [CrossRef]
- Zeki, N. M.; Mustafa, Y. F. 6,7-Coumarin-heterocyclic hybrids: A comprehensive review of their natural sources, synthetic approaches, and bioactivity. J. Mol. Struct. 2024, 1303, 137601. [Google Scholar] [CrossRef]
- Aqib, M.; Khatoon, S.; Ali, M.; Sajid, S.; Assiri, M. A.; Ahamad, S.; Saquib, M.; Hussain, M. K. Exploring the anticancer potential and mechanisms of action of natural coumarins and isocoumarins. Eur. J. of Med. Chem. 2025, 282, 117088. [Google Scholar] [CrossRef] [PubMed]
- Tariq, H.; Khan, S.; Miyan, K.; Qidwai, S. N.; Ahamad, S.; Saquib, M.; Hussain, M. K. Exploring natural coumarins in antiprotozoal drug discovery: a comprehensive review. Chem. Biodiv. 2025, 22, e01964. [Google Scholar] [CrossRef]
- Liu, W.; Wu, L.; Liu, W.; Tian, L.; Chen, H.; Wu, Z.; Wang, N.; Liu, X.; Qiu, J.; Feng, X.; Xu, Z.; Jiang, X.; Zhao, Q. Design, synthesis and biological evaluation of novel coumarin derivatives as multifunctional ligands for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2022, 242, 114689. [Google Scholar] [CrossRef]
- Dang, T.; Zhou, W.; Zhou, Y.; Meng, D.; Xu, Q.; Chen, L.; Lin, G.; Qing, B.; Sun, D.; Hou, Y.; Li, Y.N. Sesquiterpene coumarins isolated from Ferula bungeana and their anti-neuroinflammatory activities. Bioorg. Chem. 2022, 128, 106102. [Google Scholar]
- Zhang, Y.; Xu, Z.; Dou, M.; Xu, Y.; Fu, X.; Zhu, F.; Ye, H.; Zhang, J.; Feng, G. Design, synthesis, and bioactivity of novel coumarin-3-carboxylic acid derivatives containing a thioether quinoline moiety. ACS Omega 2024, 9, 50695–50704. [Google Scholar] [CrossRef]
- Szwaczko, K.; Strzyga-Łach, P.; Struga, M.; Kiernozek-Kalińska, E.; Szafrański, K.; Skiba, A.; Płazińska, A.; Skalicka-Woźniak, K.; Bielenica, A. Design, synthesis, structure–activity relationships, and preliminary anticancer properties of menthol-modified coumarin esters and 3,4-dihydrocoumarin derivatives. ACS Omega 2025, 10, 46418–46434. [Google Scholar] [CrossRef] [PubMed]
- Tataringa, G.; Tuchilus, C.; Ahmed, M.; Ahmed, S.; Bhat, A. R.; Ben Hadda, T.; Zbancioc, A.-M.; Fahelelbom, K. M. Discovery of new molecular hybrid derivatives with coumarin scaffold bearing pyrazole/oxadiazole moieties: Molecular docking, POM analyses, in silico pharmacokinetics and in vitro antimicrobial evaluation with identification of potent antitumor pharmacophore sites. Bioorg. Chem. 2024, 153, 107761. [Google Scholar]
- Mortel, S. L. R.; Macalino, S. J. Y.; Wang, M.; Tie, J.-K. In silico and in vitro studies of potential novel vitamin K epoxide reductase (VKOR) inhibitors suggest an updated structure–activity relationship. ACS Omega 2025, 10, 26694–26704. [Google Scholar] [CrossRef]
- Zhao, Y.; Peng, Z.; Wang, G. Design, synthesis, kinetic analysis, molecular docking, and mechanistic studies of novel coumarin-oxadiazole derivatives as α-glucosidase and PTP1B inhibitors. Bioorg. Med. Chem. 2025, 129, 118321. [Google Scholar] [CrossRef]
- Khanna, A.; Narang, A.; Thakur, V.; Singh, K.; Kumar, N.; Kaur, R.; Megha; Raj, A.; Devi, M.; Jyoti; Rana, R.; Sharma, A.; Kaur, H.; Singh, P.; Kaur, S.; Bedi, P. M. S. Design, synthesis, antibacterial evaluation, and molecular modelling studies of 1,2,3-triazole-linked coumarin-vanillin hybrids as potential DNA gyrase and topoisomerase IV inhibitors. Bioorg. Chem. 2025, 164, 108815. [Google Scholar] [CrossRef]
- Zakaria, B. S.; Ewes, W. A.; El-Messery, S. M.; Goda, E.F. Synthesis and antiviral activity of new 4-substituted oxycoumarins against SARS and HSV-2 viruses: mechanistic and molecular modeling study. Bioorg. Chem. 2025, 164, 108845. [Google Scholar] [CrossRef]
- Beinema, M.; Brouwers, J. R.; Schalekamp, T.; Wilffert, B. Pharmacogenetic differences between warfarin, acenocoumarol and phenprocoumon. Thromb. Haemost. 2008, 100(6), 1052–1057. [Google Scholar] [CrossRef]
- Winter, Y.; Dodel, R.; Korchounov, A.; Grond, M.; Oertel, W. H.; Back, T. Clinical and pharmacological properties of new oral anticoagulants for the prevention of cerebral thromboembolism: Factor Xa and thrombin inhibitors. World J. Neurosci. 2012, 2, 7–14. [Google Scholar] [CrossRef]
- Pattanayak, S.; Bose, P. Herniarin, a natural coumarin, inhibits mammary carcinogenesis by modulating liver X receptor-α/β-PI3K-Akt-Maf1 Pathway in sprague-dawley rats. Pharmacogn. Mag. 2019, 15, 510. [Google Scholar] [CrossRef]
- Hadjipavlou-Litina, D.; E. B., S; Militsopoulou, M.; Athanassopoulos, C. M.; Papaioannou, D.; Hadjipavlou-Litina, D.; E. B., S; Militsopoulou, M.; Athanassopoulos, C. M.; Papaioannou, D. Trioxsalen derivatives with lipoxygenase inhibitory activity. J. Enzyme Inhib. Med. Chem. 2009, 24, 1351–1356. [Google Scholar] [CrossRef] [PubMed]
- Weng, M.; Deng, Z.; Huang, S.; Lin, X.; Xu, N.; Sun, X.; Wu, W.; Lu, J.; Wang, D. Fraxetin inhibits proliferation and induces apoptosis of bladder cancer through the Akt pathway in vitro and in vivo. J. Biochem. Mol. Toxicol. 2024, 38, e23556. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Lin, P.; Lin, M.; Ye, S.; Papa Akuetteh, P. D.; Zhou, Y. Fraxetin Inhibits the Proliferation and Metastasis of Glioma Cells by Inactivating JAK2/STAT3 Signaling. Evidence-Based Comple. Altern. Med. 2021, 2021, 5540139. [Google Scholar]
- Cui, D.; Zhang, L.; Zhang, J.; Li, W.; Chen, J.; Guo, Z.; Sun, C.; Wang, Y.; Wang, W.; Li, S.; Huang, W.; Zheng, C.; Chen, R. Hybrid local and charge-transfer material with ultralong room temperature phosphorescence for efficient organic afterglow Light-Emitting Diodes. Angew. Chem. Int. Ed. 2024, 63, e202411588. [Google Scholar] [CrossRef]
- Ueda, M.; Uehara, A.; Usui, K.; Hasegawa, M. A folded mechanochromic organic fluorophore based on thianthrene-fused coumarin. Chem. Commun. 2026, 62, 2208–2211. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Jiang, Y.; Wang, Y.; Li, Y.; Tang, H.; Shao, A.; Ni, J. Fluorescence monitoring of STING using a coumarin-based chemigenetic probe. New J. Chem. 2026, 50, 537–543. [Google Scholar] [CrossRef]
- Jiang, L.; Tang, L.; Mei, T.; Xiao, M.; Xiang, K.; Zhou, W.; Cai, Y.; Fan, M.; Jia, T.; Tao, Q.; Ye, L.; Bi, Z.; Zhou, C.; Wang, X.; Chen, J.; Lu, G.; Liu, Y.; Yang, R.; Su, W.; Ma, W.; Fan, Q. Coumarin-based volatile and non-volatile solid additives enable organic solar cells with 20.32% efficiency. Angew. Chem. Int. Ed. 2026, 65, e24002. [Google Scholar] [CrossRef] [PubMed]
- Kouznetsov, V. V.; Puerto-Galvis, C. E.; Villamizar, M. C. O.; Vargas-Mendez, L. Y. Insights into the metal-catalyzed alkyne hydroarylation reactions and related processes for the synthesis of coumarins. Curr. Org. Chem. 2017, 21, 949–963. [Google Scholar] [CrossRef]
- Lončarić, M.; Gašo-Sokač, D.; Jokić, S.; Molnar, M. Recent advances in the synthesis of coumarin derivatives from different starting materials. Biomolecules 2020, 10, 151. [Google Scholar] [CrossRef] [PubMed]
- Moghadam Farid, S.; Seifinoferest, B.; Gholamhosseyni, M.; Larijani, B.; Mahdavi, M. Modern metal-catalyzed and organocatalytic methods for synthesis of coumarin derivatives: a review. Org. Biomol. Chem. 2022, 20, 4846–4883. [Google Scholar] [CrossRef]
- Pitaro, M.; Croce, N.; Gallo, V.; Arienzo, A.; Salvatore, G.; Antonini, G. Coumarin-induced hepatotoxicity: a narrative review. Molecules 2022, 27, 9063. [Google Scholar] [CrossRef]
- Sharma, D.; Dhayalan, V.; Chatterjee, R.; Khatravath, M.; Dandela, R. Recent advances in the synthesis of coumarin and its derivatives by using aryl propiolates. ChemistrySelect 2022, 7, e202104299. [Google Scholar] [CrossRef]
- Patra, S.; Patra, P.; Rout, D.; Adikari, S.; Mahanty, D. S. A short review on the synthesis of oxazolo/thiazolo/imidazolocoumarins and their biological activities. Synth. Commun. 2023, 1–23. [Google Scholar] [CrossRef]
- Habibi, W.; Talbi, S.; Hamri, S.; Hafid, A.; Khouili, M. Coumarin derivatives: microwave synthesis and biological properties—a review. J. Heterocycl. Chem. 2024, 61, 2070–2096. [Google Scholar] [CrossRef]
- Firoz, H.; Ali, R.; Khan, F. A.; Kakkar, P.; Soni, R. K.; Assiri, M. A.; Ahamad, S.; Saquib, M.; Hussain, M. K. Coumarins as versatile scaffolds: innovative synthetic strategies for generating diverse heterocyclic libraries in drug discovery. J. Mol. Struct. 2026, 1352, 144426. [Google Scholar] [CrossRef]
- Abdel-Aziem, A.; Elsharabasy, S. A.; Sayed, M. T. Synthesis, reactions, and biological evaluation potential of coumarin derivatives: review. J. Heterocycl. Chem. 2026, 63, 169–194. [Google Scholar] [CrossRef]
- Nasab, N. H.; Azimian, F.; Kruger, H. G.; Kim, S. J. Acetylcoumarin in cyclic and heterocyclic-containing coumarins: synthesis and biological applications. Tetrahedron 2022, 129, 133158. [Google Scholar] [CrossRef]
- Hooshmand, S. E.; Alavioon, S. I.; Saeb, M.; Brahmachari, G.; Shiri, M. Decarboxylation and cross-coupling reactions of coumarin-3-carboxylic acid: a comprehensive review. Tetrahedron 2024, 167, 134238. [Google Scholar] [CrossRef]
- Langer, P. Adventures in coumarin chemistry. Synlett 2025, 36, 29–43. [Google Scholar] [CrossRef]
- Zuo, X.; Chen, S.; Xu, S.-W.; Chang, S.-Q.; Liu, X.-L.; Zhou, Y.; Yuan, W.-C. Highly efficient, catalyst-free, diastereoselective, diversity-oriented synthesis of dihydrocoumarin–pyrrolidine–spirooxindoles bearing three contiguous stereocenters. Synthesis 2019, 51, 2339–2350. [Google Scholar] [CrossRef]
- Huang, Q.; Fu, J.; Chang, Z.; Gan, W.; Wang, Y.J.; Han, X. Diversity-oriented synthesis of coumarin-fused cyclopentanones via a nucleophilic phosphine controlled cascade reaction. Synlett 2022, 33, 1282–1286. [Google Scholar] [CrossRef]
- Dey, S.; Das, A.; Yadav, R. N.; Boruah, P. J.; Sarkar, K.; Paul, A.; Hossain, M. F. Electron donor-acceptor complex enabled photocascade strategy for synthesis of trans-dihydrofuro [3,2-c]chromen-4-one scaffolds via radical conjugate addition of pyridinium ylide. Chem. Commun. 2024, 60, 14384–14387. [Google Scholar] [CrossRef]
- Chandrasekar, L.; Yun-Zhen, H.; Hao-Tse, C.; Yu-Chen, H.; Liu, W.-M. Synthesis of alkynyl cyclopropa[c]coumarins via propargyl sulfonium salts as C1 synthon. Org. Biomol. Chem. 2024, 22, 9219–9230. [Google Scholar] [CrossRef]
- Li, Y.-J.; Wu, Z.-L.; Gu, Q.-S.; Fan, T.; Duan, M.-H.; Wu, L.; Wang, Y.-T.; Wu, J.-P.; Fu, F.-L.; Sang, F.; Peng, A.-T.; Jiang, Y.; Liu, X.-Y.; Lin, J.-S. Catalytic intermolecular asymmetric [2π+2σ] cycloadditions of bicyclo [1.1.0]butanes: practical synthesis of enantioenriched highly substituted bicyclo [2.1.1]hexanes. J. Am. Chem. Soc. 2024, 146, 34427–34441. [Google Scholar] [CrossRef]
- Voloshkin, V. A.; Villa, M.; Martynova, E. A.; Beliš, M.; Van Hecke, K.; Ceroni, P.; Nolan, S. P. Synthesis of cyclobutane-fused chromanones via gold-mediated photocatalysis. Chem. Sci. 2024, 15, 4571–4580. [Google Scholar] [CrossRef]
- Yi, Z.-Q.; Zhang, W.; Yi, B.; Liu, C.; Xie, Y.; Li, M.; Xiong, Y.; Wu, W.; Tan, J.-P. Stereoselective synthesis of biology-oriented pentacyclic pyrrolo [2,1-a]isoquinoline scaffolds by photoredox-induced radical annulations. Org. Lett. 2025, 27, 2197–2202. [Google Scholar] [CrossRef]
- Long, C.-H.; Zuo, Y.-P.; Kong, C.-C.; Cai, Z.-N.; Qin, H.-B. Visible-light-driven [2+2] cycloaddition of coumarins with diverse olefins using a 4CzIPN photosensitizer: application to the total synthesis of (±)-lindleyanin. Org. Lett. 2025, 27, 11038–11044. [Google Scholar] [CrossRef]
- Nachimuthu, K.; Nallasivam, J. L. Palladium-catalyzed formal [3+2] cycloadditions to access spirocyclopentane-2-oxindole fused chromanones, quinolinones, and thiochromones. Chem. Asian J. 2025, 20, e00550. [Google Scholar] [CrossRef]
- Xiang, M.; Liu, G.-Y.; Liu, H.-M.; Zhou, W.-Y.; Wang, G.; Fei, F.; Jia, Y.-Q.; Shen, L.-W. The [4+2] annulation of o-acylamino-aryl MBH carbonates with coumarins: facile access to tetrahydrochromeno [4,3-b]quinolin-6-ones. New J. Chem. 2025, 49, 683–686. [Google Scholar] [CrossRef]
- Kumari, P.; Tiwari, G.; Narayana, C.; Sagar, R. Stereoselective and regioselective synthesis of chiral dihydrofurocoumarins as glycohybrids. Org. Lett. 2025, 27, 12363–12367. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.; Hasanpour, H. The sequential one-pot reaction of isatylidene malononitriles and 4-hydrazinocoumarin: metal catalyst-free and efficient access to coumarin-fused spiro[diazepin-5,3′-oxindole]. Synthesis 2026. [Google Scholar] [CrossRef]
- Li, N.-B.; Liu, Y.-C.; Liu, L.-H.; Wei, W.-Y.; Zhang, D.; He, W.-M.; Jia, H.-Y.; Bian, W. Visible-light-induced annulation of 4-aminocoumarins to tetrahydropyrimidine-fused coumarins and evaluation of their antitumor activities. Green Chem. 2026, 28, 1601–1606. [Google Scholar] [CrossRef]
- Tripathi, K. N.; Singh, S.; Akhtar, N.; Manna, K.; Singh, R. P. Visible-light-driven site-selective alkylation of the benzo core of coumarins. Chem. Commun. 2022, 58, 9674–9677. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Tripathi, K. N.; Singh, R. P. Redox activated amines in the organophotoinduced alkylation of coumarins. Org. Biomol. Chem. 2022, 20, 5716–5720. [Google Scholar] [CrossRef]
- Sun, B.; Wang, Y.; Wang, J.; Chen, M.; Zhong, Z.; Wang, J.; Jin, C. Photoredox-catalyzed redox-neutral decarboxylative C-H acylations of coumarins with alpha-keto acid. Org. Lett. 2023, 25, 2466–2470. [Google Scholar] [CrossRef]
- Patra, J.; Nair, A. M.; Volla, C. M. R. Expedient radical phosphonylations via ligand to metal charge transfer on bismuth. Chem. Sci. 2024, 15, 7136–7143. [Google Scholar] [CrossRef]
- Mritunjay; Sharma, S.; Singh, P.; Gupta, A.; Singh, V.; Singh, L.; Kumar, A. DBU-catalyzed vinylogous reaction of 3-cyano-4-methylcoumarins with 3-arylsulfonyl-3-indolyloxindoles. ChemistrySelect 2024, 9, e202402133. [Google Scholar] [CrossRef]
- Sharma, P.; Singh, T.; Rawat, N.; Singh, A. Visible-light-mediated, LMCT-enabled C(sp3)-H Bond alkylation of alkanes and silanes via C-4 functionalization of coumarins. J. Org. Chem. 2025, 90, 5574–5577. [Google Scholar] [CrossRef] [PubMed]
- Rawat, N.; Sharma, P.; Nasireddy, S. R.; Vilas, A.; Singh, A. Organophotocatalyzed radical hydrophosphonylation of acrylates, α,β-unsaturated ketones, coumarins, quinoxalinones, and p-quinone methides. J. Org. Chem. 2025, 90, 8703–8711. [Google Scholar] [CrossRef]
- Topolska, A.; Przydacz, A.; Sieroń, L.; Skrzyńska, A.; Fraile, A.; Alemán, J.; Albrecht, Ł. Aminocatalytic 1,6-addition of 2-benzyl-3-furaldehyde to 3-cyano-4-styrylcoumarins: a dearomative approach for the synthesis of furan–coumarin hybrids. J. Org. Chem. 2025, 90, 14931–14942. [Google Scholar] [CrossRef] [PubMed]
- Romaniszyn, M.; Gronowska, K.; Albrecht, Ł. Remote functionalization of 4-(alk-1-en-1-yl)-3-cyanocoumarins via the asymmetric organocatalytic 1,6-addition. Adv. Synth. Catal. 2021, 363, 5116–5121. [Google Scholar] [CrossRef]
- Jia, L.; Zhang, Z. Ru(II)-catalyzed C-H alkylation of indoles at the C-3 position with coumarin-3-carboxylic acids. ChemistrySelect 2025, 10, e00281. [Google Scholar] [CrossRef]
- Hsu, C.-W.; Chen, Y.-J.; Huang, X.-R.; Shi, C.-J.; Lin, T.-I.; Yen, T.-C.; Nagare, Y. K.; Marri, G.; Chen, Y.-R.; Lin, W. Regiodivergent synthesis utilizing alkyl hex-5-en-2-ynoates and 3-homoacyl coumarins: phenol-mediated controllable phosphine-catalyzed michael and 1,7-umpolung addition. Org. Lett. 2025, 27, 12385–12390. [Google Scholar] [CrossRef]
- Cheng, D.; Ishihara, Y.; Tan, B.; Barbas, C. F. Organocatalytic asymmetric assembly reactions: synthesis of spirooxindoles via organocascade strategies. ACS Catal. 2014, 4, 743–762. [Google Scholar] [CrossRef]
- Panda, S. S.; Jones, R. A.; Bachawala, P.; Mohapatra, P. P. Spirooxindoles as potential pharmacophores. Mini-Rev. Med. Chem. 2017, 17, 1515–1536. [Google Scholar] [CrossRef]
- Bariwal, J.; Voskressensky, L. G.; Van der Eycken, E. V. Recent advances in spirocyclization of indole derivatives. Chem. Soc. Rev. 2018, 47, 3831–3848. [Google Scholar] [CrossRef]
- Deepthi, A.; Thomas, N. V.; Sathi, V. Green protocols for the synthesis of 3,3’-spirooxindoles – 2016- mid 2019. Curr. Green Chem. 2019, 6, 210–225. [Google Scholar] [CrossRef]
- Lin, Y.; Du, D. Recent advances in squaramide-catalyzed asymmetric cascade reactions for the synthesis of spirooxindoles. Chin. J. Org. Chem. (in Chinese). 2020, 40, 3214–3236. [Google Scholar] [CrossRef]
- Saeed, R.; Sakla, A. P.; Shankaraiah, N. An update on the progress of cycloaddition reactions of 3-methyleneindolinones in the past decade: versatile approaches to spirooxindoles. Org. Biomol. Chem. 2021, 19, 7768–7791. [Google Scholar] [CrossRef]
- Wang, Y.; Cobo, A. A.; Franz, A. K. Recent advances in organocatalytic asymmetric multicomponent cascade reactions for enantioselective synthesis of spirooxindoles. Org. Chem. Front. 2021, 8, 4315–4348. [Google Scholar] [CrossRef]
- Ganesh, M.; Suraj, S. Expeditious entry into carbocyclic and heterocyclic spirooxindoles. Org. Biomol. Chem. 2022, 20, 5651–5693. [Google Scholar] [CrossRef]
- Jana, M.; Mal, S. Recent update on synthesis of spiro-heterocycles in alcohol using malononitrile as a building block. Arkivoc 2022, 2022, 361–385. [Google Scholar] [CrossRef]
- Panda, S. S.; Aziz, M. N.; Stawinski, J.; Girgis, A. S. Azomethine ylides—versatile synthons for pyrrolidinyl-heterocyclic compounds. Molecules 2023, 28, 618. [Google Scholar] [CrossRef] [PubMed]
- Liandi, A. R.; Cahyana, A. H.; Alfariza, D. N.; Nuraini, R.; Sari, R. W.; Wendari, T. P. Spirooxindoles: recent report of green synthesis approach. Green Synth. Catal. 2023, 5, 1–13. [Google Scholar] [CrossRef]
- Asif, M.; Azaz, T.; Tiwari, B.; Nasibullah, M. Propagative isatin in organic synthesis of spirooxindoles through catalysis. Tetrahedron 2023, 134, 133308. [Google Scholar] [CrossRef]
- Pan, Y.-M.; He, M.-X.; Wang, Q.; Liu, H.-F.; Ren, S.-Y. Research advances in electrochemical synthesis of spirocyclic skeleton compounds. Synthesis 2023, 55, 2873–2895. [Google Scholar] [CrossRef]
- Borah, B.; Veeranagaiah, N. S.; Sharma, S.; Patat, M.; Prasad, M. S.; Pallepogu, R.; Chowhan, L. R. Stereoselective synthesis of CF3-containing spirocyclic-oxindoles using N-2,2,2-trifluoroethylisatin ketimines: an update. RSC Adv. 2023, 13, 7063–7075. [Google Scholar] [CrossRef] [PubMed]
- Helal, M. H.; Owda, M. E.; Mogharbel, A. T.; Hamzah Alessa, A.; Omer, N.; Abdelaziz, M. A.; Ibrahim, I.; Eliwa, E. M. C3-Spirooxindoles: divergent chemical synthesis and bioactivities (2018–2023). Bioorg. Chem. 2024, 143, 107091. [Google Scholar] [CrossRef]
- Borah, B.; Patat, M.; Chowhan, L. R. Organocatalytic enantioselective assembly of dispiro-bisoxindoles with vicinal stereocenters. Org. Biomol. Chem. 2024, 22, 8365–8373. [Google Scholar] [CrossRef]
- Feng, J.; Wang, Y.; Li, E. Q.; Loh, T. P. Recent developments in copper-catalyzed annulations for synthesis of spirooxindoles. Chem. Rec. 2024, 24, e202400126. [Google Scholar] [CrossRef]
- Spirooxindole: chemistry, synthesis, characterization and biological significance; Patel, G., Shah, V. R., Nguyen, T. A., Deshmukh, K., Eds.; Elsevier, 2024. [Google Scholar]
- Zhao, K.; Zhai, M.; Zhang, L.; Meng, X. Recent advances in the reaction of isatin-derived MBH carbonates, synthesis of spiro-oxindoles. Org. Biomol. Chem. 2025, 23, 1292–1308. [Google Scholar] [CrossRef]
- Hari, D. P.; Singha, T. Visible-light-mediated strain-release radical spirocyclizations: access to functionalized spirocyclobutanes. Synlett 2025, 36, 445–451. [Google Scholar] [CrossRef]
- Liu, Y.-Q.; Wu, Y.; Li, B.; Tang, X.; Chen, C. Recent advances in the organocatalytic synthesis of chiral C3-spiro-cyclopentaneoxindoles. Org. Biomol. Chem. 2025, 23, 757–773. [Google Scholar] [CrossRef] [PubMed]
- Fatima, H. N.; Ahmad, M.; Nazir, M. S.; Akram, S.; Aslam, S. Recent synthetic methodologies for pyrrolidine derivatives through multicomponent reactions. Synth. Commun. 2025, 55, 1201–1227. [Google Scholar] [CrossRef]
- Padavi, S. K.; Vasave, S. P.; Patil, D. B. Eco-friendly strategies for synthesizing spirooxindoles and pyranopyrazoles: a comprehensive review. ChemistrySelect 2026, 11, e04793. [Google Scholar] [CrossRef]
- Chen, R.; Zhang, L.; Zhao, X.; Fang, Z.; Zhang, L.; Zhang, Q.; Zhang, C.; Zhu, Y. Discovery, bioactivities and biosynthesis of spirooxindole alkaloids. Nat. Prod. Rep. 2026. [Google Scholar] [CrossRef] [PubMed]
- Kaur, A.; Singh, B.; Vyas, B.; Silakari, O. Synthesis and biological activity of 4-aryl-3-benzoyl-5-phenylspiro[pyrrolidine-2.3′-indolin]-2′-one derivatives as novel potent inhibitors of advanced glycation end product. Eur. J. Med. Chem. 2014, 79, 282–289. [Google Scholar] [CrossRef]
- Teja, C.; Babu, S. N.; Noor, A.; Daniel, J. A.; Devi, S. A.; Nawaz Khan, F. R. Cu/TEMPO catalyzed dehydrogenative 1,3-dipolar cycloaddition in the synthesis of spirooxindoles as potential antidiabetic agents. RSC Adv. 2020, 10, 12262–12271. [Google Scholar] [CrossRef]
- Yang, C.; Fang, Y.; Luo, X.; Teng, D.; Liu, Z.; Zhou, Y.; Liao, G. Discovery of natural product-like spirooxindole derivatives as highly potent and selective LSD1/KDM1A inhibitors for AML treatment. Bioorg. Chem. 2022, 120, 105596. [Google Scholar] [CrossRef]
- Lotfy, G.; Said, M. M.; El Ashry, E. S. H.; El Tamany, E. S. H.; Al-Dhfyan, A.; Abdel Aziz, Y. M.; Barakat, A. Synthesis of new spirooxindole-pyrrolothiazole derivatives: Anti-cancer activity and molecular docking. Bioorg. Med. Chem. 2017, 25, 1514–1523. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Xu, H. C. Electrochemical rearrangement of indoles to spirooxindoles in continuous flow. Eur. J. Org. Chem. 2022, 26, e202200987. [Google Scholar] [CrossRef]
- Liu, J.; Mallick, S.; Xie, Y.; Grassin, C.; Lucas, B.; Scholermann, B.; Pahl, A.; Scheel, R.; Strohmann, C.; Protzel, C.; Berg, T.; Merten, C.; Ziegler, S.; Waldmann, H. Morphological profiling identifies the motor protein Eg5 as cellular target of spirooxindoles. Angew. Chem. Int. Ed. 2023, 62, e202301955. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tian, X.; Zhang, Y.; Ren, W. Synthesis of functionalized 3,2′-pyrrolidinyl spirooxindoles via domino 1,6-addition/annulation reactions of para-quinone methides and 3-chlorooxindoles. Org. Chem. Front. 2022, 9, 615–626. [Google Scholar]
- Li, T.-H.; Du, D.-M. Asymmetric synthesis of isoxazole and trifluoromethyl-containing 3,2’-pyrrolidinyl dispirooxindoles via squaramide-catalysed [3+2] cycloaddition reactions. Org. Biomol. Chem. 2022, 20, 817–823. [Google Scholar] [CrossRef]
- Li, X.-T.; Tian, H.-Z.; Sun, X.-W. Organocatalytic aza-Michael/Mannich cascade reaction: synthesis of enantioenriched 3,3′-spirooxindole γ-lactams. J. Org. Chem. 2023, 88, 7839–7843. [Google Scholar] [CrossRef]
- Leena, S. S.; Akhir, A.; Saxena, D.; Maitra, R.; Chopra, S.; Deepthi, A. Synthesis of tryptanthrin appended dispiropyrrolidine oxindoles and their antibacterial evaluation. RSC Med. Chem. 2023, 14, 1165–1171. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Mei, H.; Escorihuela, J.; Han, J. Electrochemical multicomponent cascade radical process enabling synthesis of iodomethyl spiropyrrolidinyl-oxindoles. Chin. J. Chem. 2024, 42, 1691–1698. [Google Scholar] [CrossRef]
- Jia, B.; Sun, Z.; Miao, X.; Ma, S.; Dong, Y.; Dang, G.; Zhang, X.; Ma, Y. Copper-catalyzed enantioselective synthesis of spirohydroindoles by ethoxyformylmethylene oxindole and iminoester 1,3-dipole cycloaddition: an examination of associated biological activities. ACS Omega 2024, 9, 24406–24414. [Google Scholar] [CrossRef] [PubMed]
- Wei, F.; Zhang, Y. Palladium-catalyzed cascade distal C–H methylation and cyclization for the construction of spirooxindole skeletons. Org. Lett. 2024, 26, 9221–9226. [Google Scholar] [CrossRef]
- Li, C.; Wang, Y.; Hu, D.; Zhang, B.; Yin, H.; Li, Y.-L. A Base promoted [3+2] cycloaddition of benzothiazolium salts with isoindigos to synthesis N, S-polyheterocyclic 3, 3′-bispirooxindoles. Asian J. Org. Chem. 2024, 13, e202400478. [Google Scholar] [CrossRef]
- Kanchrana, M.; Krishna, G. R.; Dey, B.; Pandey, N.; Guru, S. K.; Sangolkar, A. A.; Pawar, R.; Basavoju, S. Ionic liquid assisted green synthesis of quinoxaline based bisspirooxindoles: anticancer evaluation and molecular dynamics. ChemistrySelect 2024, 9, e202403608. [Google Scholar] [CrossRef]
- Nichinde, C.; Bhati, M.; Girase, A.; Patil, B.; Chaudhari, S.; Gamidi, R. K.; Joshi, K.; Kinage, A. K. A sequential nitro-Michael addition and reductive cyclization cascade reaction for diastereoselective synthesis of multifunctionalized 3,3’-pyrrolidinyl-spirooxindoles. Eur. J. Org. Chem. 2024, e202401121. [Google Scholar] [CrossRef]
- Tan, H.; Xiang, Y.; Huang, J.; Ren, S.; Lin, C.; Xu, Z.; Tang, D.; Chen, Z. Microwave-assisted synthesis of pyrrolidinyl-spirooxindoles via tandem 1,3-dipolar cycloaddition and oxidative dehydrogenation. Tetrahedron 2025, 169, 134379. [Google Scholar] [CrossRef]
- Bharani, S.; Ananda Rao, B.; Chowhan, L. R.; Pallepogu, R.; Prasad, M. S. Asymmetric synthesis of spiro[benzofuran-pyrrolidine]-indolinedione via bifunctional urea catalyzed [3 + 2]-annulation. Org. Biomol. Chem. 2025, 23, 914–919. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, L.; Zi, R.; Niu, Y.; Yang, S.; Hu, D.; Dong, J.-W. Diastereoselective Three-Component 1,3-Dipolar Cycloaddition: Concise Synthesis of Functionalized Tetrahydrocarboline-fused Spirooxindoles. Org. Biomol. Chem. 2025, 23, 2941–2953. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Huang, A.; Jia, A.; Tang, S. Synthesis of 3,2′-pyrrolidinyl spirooxindole derivative via 2,3-rearrangement of ammonium ylide. J. Org. Chem. 2025, 90, 4216–4224. [Google Scholar] [CrossRef]
- Indira, M.; Reddy, P. V. G. Efficient synthesis of spirooxindole-pyrrolo [1,2-a]pyrazin-2-ones via ionic liquid-ultrasonication. ChemistrySelect 2025, 10, e202405095. [Google Scholar] [CrossRef]
- Pronina, Y.; Filatov, A.; Shmakov, S.; Selivanov, S.; Kryukova, M.; Spiridonova, D.; Ponyaev, A.; Stepakov, A.; Boitsov, V. Highly efficient synthesis of spiro [1-azabicyclo [3.2.0]heptane] frameworks via [3+2]-cycloaddition. J. Org. Chem. 2025, 90, 4926–4945. [Google Scholar] [CrossRef]
- Lavanya, M.; Sundararaj, R.; Mani, K. S.; Balu, K.; Durai, M.; Alam, P.; Sd, P.; Durai, M.; Ahn, Y. H. Computational design, synthesis, and biological assessment of some pyrrolo [3,4-c]pyrroles targeting mycobacterium tuberculosis. ChemistrySelect 2025, 10, e202405796. [Google Scholar] [CrossRef]
- Al-Jassas, R. M.; Islam, M. S.; Al-Majid, A. M.; Haukka, M.; Nafie, M. S.; Abu-Serie, M. M.; Teleb, M.; Shaaban, M. M.; Alayyaf, A. M. A.; Domingo, L. R.; Ashraf, S.; Ul-Haq, Z.; Abdel Aziz, Y. M.; Barakat, A. Marine-inspired spirooxindole PIM-1 kinase inhibitors endowed with concomitant TRKA/CDK2 inhibition for multifaceted NSCLC apoptotic induction. ChemMedChem 2025, 20, e202500028. [Google Scholar] [CrossRef] [PubMed]
- Hazra, A.; Maity, D.; Bhosale, S. S.; Hajra, S. Asymmetric synthesis of 3,3′-pyrrolidonyl spirooxindole-5′-carboxylic ester from l-tryptophan. Synthesis 2025, 57, 488–494. [Google Scholar]
- Tan, H.; Xiang, Y.; Huang, J.; Ren, S.; Lin, C.; Xu, Z.; Tang, D.; Chen, Z. Microwave-assisted synthesis of pyrrolidinyl-spirooxindoles via tandem 1,3-dipolar cycloaddition and oxidative dehydrogenation. Tetrahedron 2025, 169, 134379. [Google Scholar] [CrossRef]
- Xuan, T.; Wang, X.; Wang, Y. Asymmetric [3+2] cycloannulation of benzoxazinones for the synthesis of imidazo [5,1-c]oxazinones. Org. Lett. 2025, 27, 3134–3138. [Google Scholar] [CrossRef]
- Gao, H.; Yang, X.; Shi, L. Asymmetric oxidative rearrangement of indoles enabled by dual catalysis with in situ generated acyl hypoiodite and chiral phosphoric acid. Org. Chem. Front. 2025, 12, 3848–3855. [Google Scholar] [CrossRef]
- Zimnitskiy, N. S.; Korotaev, V. Y.; Ulitko, M. V.; Sosnovskikh, V. Y. 1-Styryl-1,3-diketones in the synthesis of spiro[oxindole-3,2′-pyrrolidines] with notable anticancer activity. Mol. Diver. 2025, 29, 5979–5992. [Google Scholar] [CrossRef]
- Cao, X.; Tao, P.; Yu, S.; Yang, S.; Li, S.-W. Asymmetric Michael-Mannich cascade reaction of azomethine ylides with isatin-derived trifluoromethyl acrylates catalyzed by a Cu(I) catalyst. Org. Chem. Front. 2026. [Google Scholar] [CrossRef]
- Lashgari, N.; Ziarani, G. M. Synthesis of heterocyclic compounds based on isatin through 1,3-dipolar cycloaddition reactions. Arkivoc 2012, i, 277–320. [Google Scholar] [CrossRef]
- Fokas, D.; Ryan, W. J.; Casebier, D. S.; Coffen, D. L. Solution-phase synthesis of a spiro[pyrrolidine-2,3’-oxindole] library via a three component 1,3-dipolar cycloaddition reaction. Tetrahedron Lett. 1998, 39, 2235–2238. [Google Scholar] [CrossRef]
- Shanmugam, P.; Viswambharan, B.; Madhavan, S. Synthesis of novel functionalized 3-spiropyrrolizidine and 3-spiropyrrolidine oxindoles from Baylis-Hillman adducts of isatin and heteroaldehydes with azomethine ylides via [3+2]-cycloaddition. Org. Lett. 2007, 9, 4095–4098. [Google Scholar] [CrossRef]
- Mehrdad, M.; Faraji, L.; Jadidi, K.; Eslami, P.; Sureni, H. A regioselective and diastereoselective synthesis of new spiro-isoxazolidines via 1,3-dipolar cycloaddition of stable isatin ketonitrone and various dipolarophiles. Monatsh. Chem. 2011, 142, 917–921. [Google Scholar] [CrossRef]
- Liu, J.; Sun, H.; Liu, X.; Ouyang, L.; Kang, T.; Xie, Y.; Wang, X. Direct construction of novel exo′-selective spiropyrrolidine bisoxindoles via a three-component 1,3-dipolar cycloaddition reaction. Tetrahedron Lett. 2012, 53, 2336–2340. [Google Scholar] [CrossRef]
- Xiao, J.-A.; Liu, Q.; Ren, J.-W.; Liu, J.; Carter, R. G.; Chen, X.-Q.; Yang, H. Highly enantioselective construction of polycyclic spirooxindoles by organocatalytic 1,3-dipolar cycloaddition of 2-cyclohexenone catalyzed by proline-sulfonamide. Eur. J. Org. Chem. 2014, 2014, 5700–5704. [Google Scholar] [CrossRef]
- Revathy, K.; Lalitha, A. An efficient green chemistry protocol for the synthesis of novel spiropyrrolizidine compounds. RSC Adv. 2014, 4, 279–285. [Google Scholar] [CrossRef]
- Pavlovskaya, T. L.; Yaremenko, F. G.; Lipson, V. V.; Shishkina, S. V.; Shishkin, O. V.; Musatov, V. I.; Karpenko, A. S. The regioselective synthesis of spirooxindolo pyrrolidines and pyrrolizidines via three-component reactions of acrylamides and aroylacrylic acids with isatins and alpha-amino acids. Beilstein J. Org. Chem. 2014, 10, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Jiang, X.-L.; Wu, Q.; Shi, F.; Tu, S.-J. Diastereo- and enantioselective construction of 3,3′-pyrrolidinyldispirooxindole framework via catalytic asymmetric 1,3-dipolar cycloadditions. J. Org. Chem. 2015, 80, 5737–5744. [Google Scholar] [CrossRef]
- Haddad, S.; Boudriga, S.; Akhaja, T. N.; Raval, J. P.; Porzio, F.; Soldera, A.; Askri, M.; Knorr, M.; Rousselin, Y.; Kubicki, M. M.; Rajani, D. A strategic approach to the synthesis of functionalized spirooxindole pyrrolidine derivatives: in vitro antibacterial, antifungal, antimalarial and antitubercular studies. New J. Chem. 2015, 39, 520–528. [Google Scholar] [CrossRef]
- Uma Rani, G.; Vivek Kumar, S.; Bharkavi, C.; Menéndez, J. C.; Perumal, S. One-pot access to a library of dispiro oxindole-pyrrolidine/pyrrolothiazole-thiochromane hybrids via three-component 1,3-dipolar cycloaddition reactions. ACS Comb. Sci. 2016, 18, 337–342. [Google Scholar] [CrossRef]
- Du, Y.; Yu, A.; Jia, J.; Zhang, Y.; Meng, X. Direct N-H/alpha,alpha,beta,beta-C(sp3)-H functionalization of piperidine via an azomethine ylide route: synthesis of spirooxindoles bearing 3-substituted oxindoles. Chem. Commun. 2017, 53, 1684–1687. [Google Scholar] [CrossRef]
- Lin, W.; Zhan, G.; Shi, M.; Du, W.; Chen, Y. [3 + 3] Formal cycloadditions of nitrones from isatins and azaoxyallyl cations for construction of spirooxindoles. Chin. J. Chem. 2017, 35, 857–860. [Google Scholar] [CrossRef]
- Huang, W.-J.; Chen, Q.; Lin, N.; Long, X.-W.; Pan, W.-G.; Xiong, Y.-S.; Weng, J.; Lu, G. Asymmetric synthesis of trifluoromethyl-substituted 3,3′-pyrrolidinyl-dispirooxindoles through organocatalytic 1,3-dipolar cycloaddition reactions. Org. Chem. Front. 2017, 4, 472–482. [Google Scholar] [CrossRef]
- Wu, P.; Gao, H.; Sun, J.; Yan, C.-G. 1,3-Dipolar cycloaddition reaction for diastereoselective synthesis of functionalized dihydrospiro[indoline-3,2′-pyrroles]. Chin. Chem. Lett. 2017, 28, 329–332. [Google Scholar] [CrossRef]
- Filatov, A. S.; Knyazev, N. A.; Molchanov, A. P.; Panikorovsky, T. L.; Kostikov, R. R.; Larina, A. G.; Boitsov, V. M.; Stepakov, A. V. Synthesis of functionalized 3-spiro[cyclopropa[a]pyrrolizine]- and 3-spiro [3-azabicyclo [3.1.0]hexane]oxindoles from cyclopropenes and azomethine ylides via [3+2]-cycloaddition. J. Org. Chem. 2017, 82, 959–975. [Google Scholar] [CrossRef]
- Lin, B.; Zhang, W.-H.; Wang, D.-D.; Gong, Y.; Wei, Q.-D.; Liu, X.-L.; Feng, T.-T.; Zhou, Y.; Yuan, W.-C. 3-Methyl-4-nitro-5-isatylidenyl-isoxazoles as 1,3-dipolarophiles for synthesis of polycyclic 3,3′-pyrrolidinyl-dispirooxindoles and their biological evaluation for anticancer activities. Tetrahedron 2017, 73, 5176–5188. [Google Scholar] [CrossRef]
- Yue, G.; Wu, Y.; Dou, Z.; Chen, H.; Yin, Z.; Song, X.; He, C.; Wang, X.; Feng, J.; Zhang, Z.; Zou, P.; Lu, C. Synthesis of spiropyrrolidine oxindoles via Ag-catalyzed stereo- and regioselective 1,3-diploar cycloaddition of indole-based azomethine ylides with chalcones. New J. Chem. 2018, 42, 20024–20031. [Google Scholar] [CrossRef]
- Ryu, H.; Seo, J.; Ko, H. M. Synthesis of spiro[oxindole-3,2′-pyrrolidine] derivatives from benzynes and azomethine ylides through 1,3-dipolar cycloaddition reactions. J. Org. Chem. 2018, 83, 14102–14109. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.-W.; Wang, L.-R.; Guo, J.-M.; Ding, W.-Q.; Song, X.-Q.; Wu, H.-H.; Tang, Z.; Fan, X.-Z.; Bi, X.-F. Formal [5+3] cycloaddition of vinylethylene carbonates with isatin-based α-(trifluoromethyl)imines for diastereoselective synthesis of medium-heterocycle-fused spirooxindoles. Adv. Synth. Catal. 2019, 361, 4761–4771. [Google Scholar] [CrossRef]
- Wu, W.-T.; Han, Y.; Sun, J.; Yan, C.-G. Efficient construction of pyrrolo [1′,2′:1,2]azocino [4,5-c]quinolines via cascade cycloaddition and annulation reaction. Org. Chem. Front. 2019, 6, 3530–3534. [Google Scholar] [CrossRef]
- Niu, B.; Wu, X. Y.; Wei, Y.; Shi, M. Palladium-catalyzed diastereoselective formal [5+3] cycloaddition for the construction of spirooxindoles fused with an eight-membered ring. Org. Lett. 2019, 21, 4859–4863. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Li, S.; Wang, Q.; Chen, H.; Yang, C.; Yin, Z.; Song, X.; Zhang, L.; Lu, C.; Yue, G. K2CO3-Promoted formal [3+3]-cycloaddition of N-unsubstituted isatin N,N’-cyclic azomethine imine 1,3-dipoles with Knoevenagel adducts. Molecules 2023, 28, 1034. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yang, P.; Zhang, Y.; Tang, C.-Z.; Tian, F.; Peng, L.; Wang, L.-X. Isatin N,N’-cyclic azomethine imine 1,3-dipole and abnormal [3 + 2]-cycloaddition with maleimide in the presence of 1,4-diazabicyclo [2.2.2]octane. Org. Lett. 2017, 19, 646–649. [Google Scholar] [CrossRef]
- Wang, X.; Wu, L.; Yang, P.; Song, X.-J.; Ren, H.-X.; Peng, L.; Wang, L.-X. Isatin N,N’-cyclic azomethine imine 1,3-dipole and base catalyzed Michael addition with β-nitrostyrene via C3 umpolung of oxindole. Org. Lett. 2017, 19, 3051–3054. [Google Scholar] [CrossRef]
- Jin, Q.; Zhang, J.; Jiang, C.; Zhang, D.; Gao, M.; Hu, S. Self [3+4] cycloadditions of isatin N,N’-cyclic azomethine imine 1,3-dipole with N-(o-chloromethyl)aryl amides. J. Org. Chem. 2018, 83, 8410–8416. [Google Scholar] [CrossRef]
- Hu, S.; Zhang, J.; Jin, Q. DMAP-catalyzed alkylation of isatin N,N’-cyclic azomethine imine 1,3-dipoles with Morita-Baylis-Hillman carbonates. New J. Chem. 2018, 42, 7025–7029. [Google Scholar] [CrossRef]
- Moghaddam, F. M.; Eslami, m.; Siahpoosh, A.; Hoda, G. S. A diastereoselective construction of functionalized dihydro-pyridazine based spirooxindole scaffold via C-3 umpolung of isatin N,N’-cyclic azomethine imine. New J. Chem. 2019, 43, 10318–10323. [Google Scholar] [CrossRef]
- Wang, Q.-H.; Zhu, Z.-X.; Huang, T.; Wu, M.-S. Base catalyzed unexpected rearrangement of isatin-derived N,N’-cyclic azomethine imines and Michael addition to hindered vinylidene bisphosphonates: access to 3,3-disubstituted oxindole-fused pyrazolidin-3-one derivatives containing bisphosphonates. Tetrahedron 2019, 75, 416–421. [Google Scholar] [CrossRef]
- Yue, G.; Dou, Z.; Zhou, Z.; Zhang, L.; Feng, J.; Chen, H.; Yin, Z.; Song, X.; Liang, X.; Wang, X.; Rao, H.; Lu, C. Rapid abnormal [3+2]-cycloaddition of isatin N,N’-cyclic azomethine imine 1,3-dipoles with chalcones. New J. Chem. 2020, 44, 8813–8817. [Google Scholar] [CrossRef]
- Meerakrishna, R. S.; Suresh, S. S.; Athira, M.; Choutipalli, V. S. K.; Shanmugam, P. Diverse reactivity of isatin based N,N’-cyclic azomethine imine dipoles with arynes: synthesis of 1’-methyl-2’-oxospiro [indene-1,3’-indolines] and 3-aryl-3-pyrazol-2-oxindoles. New J. Chem. 2020, 44, 11593–11601. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, G.; Chen, Z.; Wu, M.; Wang, J.; Trigoura, L.; Guo, H.; Xing, Y.; Sun, S. Synthesis of spiro(indoline-3,1-pyrazolo [1,2-a ]pyrazoles) by 1,3-dipolar cycloadditions of isatin N,N’-cyclic azomethine imines with alkynes. J. Heterocycl. Chem. 2020, 57, 2044–2047. [Google Scholar] [CrossRef]
- Jin, Q.; Zhang, D.; Zhang, J. A [3 + 2] cycloaddition/C-arylation of isatin N,N’-cyclic azomethine imine 1,3-dipole with arynes. RSC Adv. 2020, 10, 30620–30623. [Google Scholar] [CrossRef]
- Wang, Z.-Y.; Yang, T.; Chen, R.; Ma, X.; Liu, H.; Wang, K.-K. 1,3-Dipolar cycloaddition of isatin N,N’-cyclic azomethine imines with α,β-unsaturated aldehydes catalyzed by DBU in water. RSC Adv. 2020, 10, 24288–24292. [Google Scholar] [CrossRef]
- Kartikey, K. D. D.; Reddy, M. S.; Chowhan, L. R. Isatin N,N’-cyclic azomethine imine 1,3-dipole mediated regio and diastereoselective synthesis of isoxazole-containing spirooxindoles by an abnormal [3+2] cycloaddition. Tetrahedron Lett. 2020, 61, 152664. [Google Scholar] [CrossRef]
- Fang, Q.-Y.; Jin, H.-S.; Wang, R.-B.; Zhao, L.-M. A role for isatin azomethine imines as a dipolarophile in cycloaddition reactions. Org. Lett. 2020, 22, 7358–7362. [Google Scholar] [CrossRef]
- Abdelmouna, K.; Laghchioua, F.; Paz, F. A. A.; Mendes, R. F.; Moura, N. M. M.; Faustino, M. A. F.; Cavaleiro, J. A. S.; Kandri Rodi, Y.; Rakib, E. M.; Neves, M. G. P. M. S. Development of catalyst-free approach to synthesize novel spiro[indoline-3,1′-pyrazolo [1,2-a]pyrazoles] via 1,3-dipolar cycloaddition. J. Mol. Struct. 2023, 1272, 134170. [Google Scholar] [CrossRef]
- Li, X.; Li, W.-S.; Fu, X.; Wan, W.-J.; Wang, L.-X. A novel [4 + 3] annulation between isatin N,N’-cyclic azomethine 1,3-dipole and in situ generated nitrosoalkene for direct preparation of seven-membered heterocyclic spirooxindoles. Tetrahedron Lett. 2024, 146, 155183. [Google Scholar] [CrossRef]
- Wang, Y.; Wei, X.; Qu, J.; Wang, B. Palladium-catalyzed umpolung allylation of isatin-derived azomethine imines with vinylcyclopropanes. Eur. J. Org. Chem. 2024, 27, e202400743. [Google Scholar] [CrossRef]
- Mehrdad, M.; Faraji, L.; Jadidi, K.; Eslami, P.; Sureni, H. A regioselective and diastereoselective synthesis of new spiro-isoxazolidines via 1,3-dipolar cycloaddition of stable isatin ketonitrone and various dipolarophiles. Monatsh. Chem. 2011, 142, 917–921. [Google Scholar] [CrossRef]
- Yang, H.-B.; Wei, Y.; Shi, M. Construction of spiro[indoline]oxindoles through one-pot thermal-induced [3+2] cycloaddition/silica gel-promoted fragmentation sequence between isatin ketonitrones and electron-deficient alkynes. Tetrahedron 2013, 69, 4088–4097. [Google Scholar] [CrossRef]
- Chen, Y. R.; Zhan, G.; Du, W.; Chen, Y. C. Regioselective asymmetric formal (3+2) cycloadditions of nitrone ylides from isatins and enals. Adv. Synth. Catal. 2016, 358, 3759–3764. [Google Scholar] [CrossRef]
- Zhan, G.; Shi, M. L.; Lin, W. J.; Ouyang, Q.; Du, W.; Chen, Y. C. Direct asymmetric aza-vinylogous-type Michael additions of nitrones from isatins to nitroalkenes. Chem.–Eur. J. 2017, 23, 6286–6289. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Zhan, G.; Shi, M.; Du, W.; Chen, Y. 3 + 3] Formal cycloadditions of nitrones from isatins and azaoxyallyl cations for construction of spirooxindoles. Chin. J. Chem. 2017, 35, 857–860. [Google Scholar] [CrossRef]
- Shi, M.; Zhan, G.; Du, W.; Chen, Y. Direct asymmetric aza-vinylogous Mannich reaction of nitrones from isatins and ketimines. Acta Chim. Sinica (in Chinese). 2017, 75, 998–1002. [Google Scholar] [CrossRef]
- Maiuolo, L.; Merino, P.; Algieri, V.; Nardi, M.; Di Gioia, M. L.; Russo, B.; Delso, I.; Tallarida, M. A.; De Nino, A. Nitrones and nucleobase-containing spiro-isoxazolidines derived from isatin and indanone: solvent-free microwave-assisted stereoselective synthesis and theoretical calculations. RSC Adv. 2017, 7, 48980–48988. [Google Scholar] [CrossRef]
- Wu, S.-Y.; Chen, W.-L.; Ma, X.-P.; Cui Liang, G.-F. S.; Mo, D.-L. Copper-Catalyzed [3+2] cycloaddition and interrupted Fischer indolization to prepare polycyclic furo [2,3-b]indolines from N-aryl isatin nitrones and methylenecyclopropanes. Adv. Synth. Catal. 2019, 361, 965–970. [Google Scholar] [CrossRef]
- Cheng, X.; Fei, W.; Luo, Z.; Li, J.; Wang, Z.; Yao, W. DABCO-catalyzed α-regio- and diastereoselective (3+2) cycloadditions of nitrone ylides from isatins and activated alkenes. Synthesis 2020, 52, 3632–3639. [Google Scholar]
- Yuan, H.; Lu, D.-L.; Liang, C.; Mo, D.-L. Synthesis of spirooxindole-benzo[d]oxazoles and dihydrobenzofurans through cycloaddition and rearrangement of N-vinyl nitrones and arynes. Adv. Synth. Catal. 2022, 364, 1409–1414. [Google Scholar] [CrossRef]
- Luo, Z.; Wang, Z.; Yao, W. DABCO-catalyzed [3+2] cycloaddition of isatin-derived nitrones and electron-deficient dienes via a 1,6-addition reaction. Synthesis 2022, 54, 4339–4346. [Google Scholar]
- Luo, M.-L.; Hou, Q.; Liu, S.-J.; Zhao, Q.; Qin, R.; Peng, C.; Han, B.; Zhan, G. One-step synthesis of hydropyrrolo [3,2-b]indoles via cascade reactions of oxindole-derived nitrones with allenoates. Org. Lett. 2022, 24, 8493–8497. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Wu, Y.-Z.; Fang, Y.-H.; Chen, C.-H.; Liang, C.; Mo, D.-L. Synthesis of spirooxindole-1,2-oxazinan-5-ones through 2,2,2-trifluoroethanol promoted [3 + 3] cycloaddition of N-Vinyl oxindole nitrones and oxyallyl cations. J. Org. Chem. 2023, 88, 16155–16166. [Google Scholar] [CrossRef]
- Behera, K.; Mohapatra, S.; Nayak, S.; Mohapatra, S.; Parida, S. P.; Bhattacharya, D.; Rout, U. K.; Sahoo, C. R. Transition metal- and base-free [3+2] cycloaddition: design, synthesis, and antibacterial activity of isoxazolidine derivatives. ChemistrySelect 2024, 9, e202404850. [Google Scholar] [CrossRef]
- Zou, N.; Wu, Y.-Z.; Zhong, X.-Y.; Wei, C.; Liao, L.-M.; Mo, D.-L.; Zhou, W.-J. Asymmetric [3+3] cycloaddition of N-vinyl oxindole nitrones with 2-indolylmethanols to prepare spirooxindole [1,2]oxazines. Adv. Synth. Catal. 2024, 366, 4960–4965. [Google Scholar] [CrossRef]
- Hamer, J.; Macaluso, A. Nitrones. Chem. Rev. 1964, 64, 473–495. [Google Scholar] [CrossRef]
- Delpierre, G. R.; Lamchen, M. Nitrones. Quart. Rev., Chem. Soc. 1965, 19, 329–348. [Google Scholar] [CrossRef]
- Black, D. S. C.; Crozier, R. F.; Davis, V. C. 1,3-Dipolar cycloaddition reactions of nitrones. Synthesis 1975, 205–221. [Google Scholar] [CrossRef]
- Confalone, P. N.; Huie, E. M. The [3+2] nitrone-olefin cycloaddition reaction. In Organic Reactions; John Wiley & Sons, Inc., 1988; vol. 36, p. 74. [Google Scholar]
- Zhao, B. Application of nitrones in total synthesis of natural products. Prog. Chem. (in Chinese). 2000, 12, 77–88. [Google Scholar]
- Osborn, H. M. I.; Gemmell, N.; Harwood, L. M. 1,3-dipolar cycloaddition reactions of carbohydrate derived nitrones and oximes. J. Chem. Soc., Perkin Trans. 1 2002, 2419–2438. [Google Scholar] [CrossRef]
- Hu, X.-F.; Feng, Y.-Q.; Li, X.-F. Chiral Lewis acid-catalyzed 1,3-dipolar cycloadditions between nitrones and alkenes. Chin. J. Org. Chem. (in Chinese). 2005, 25, 1–7. [Google Scholar]
- Rück-Braun, K.; Freysoldt, T. H. E.; Wierschem, F. 1,3-Dipolar cycloaddition on solid supports: nitrone approach towards isoxazolidines and isoxazolines and subsequent transformations. Chem. Soc. Rev. 2005, 34, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Yang, J. Recent developments in nitrone chemistry: some novel transformations. Synlett 2012, 23, 2293–2297. [Google Scholar] [CrossRef]
- Murahashi, S.-I.; Imada, Y. Synthesis and transformations of nitrones for organic synthesis. Chem. Rev. 2019, 119, 4684–4716. [Google Scholar] [CrossRef] [PubMed]
- Yue, G.; Liu, B. Research progress on [3+n] (n≥3) cycloaddition of 1,3-dipoles. Chin. J. Org. Chem. (in Chinese). 2020, 40, 3132–3153. [Google Scholar] [CrossRef]
- Thakur, S.; Das, A.; Das, T. 1,3-Dipolar cycloaddition of nitrones: synthesis of multisubstituted, diverse range of heterocyclic compounds. New J. Chem. 2021, 45, 11420–11456. [Google Scholar] [CrossRef]
- Akulov, A. A.; Varaksin, M. V.; Mampuys, P.; Charushin, V.; Chupakhin, O. N.; Maes, B. U. W. C(sp2)–H functionalization in non-aromatic azomethine-based heterocycles. Org. Biomol. Chem. 2021, 19, 297–312. [Google Scholar] [CrossRef]
- Oudeyer, S.; Levacher, V.; Beucher, H.; Brière, J.-F. Recent advances in catalystic and technology-driven radic al addtion to N,N-disubsititued iminum species. Molecules 2023, 28, 1071. [Google Scholar] [CrossRef]
- Tamura, O. Exploration and development of nitrone chemistry. Chem. Pharm. Bull. 2024, 72, 731–746. [Google Scholar] [CrossRef]
- Tanaka, K. Recent advances in the direct N–C(sp2) nitrone synthesis from oxime. Eur. J. Org. Chem. 2024, 27, e202400202. [Google Scholar]
- Abdulla, M.; Hussain, M. K.; Ahamad, S. Trifluoromethylnitrone: a versatile building block for synthesizing trifluoromethyl-containing heterocyclic compounds. Org. Biomol. Chem. 2024, 22, 5242–5256. [Google Scholar] [CrossRef]
- Kobus, M.; Friedrich, T.; Zorn, E.; Burmeister, N.; Maison, W. Medicinal chemistry of drugs with N-oxide functionalities. J. Med. Chem. 2024, 67, 5168–5184. [Google Scholar] [CrossRef]
- Rozpara, I.; Marco-Contelles, J.; Piotrowska, D. G.; Głowacka, I. E. Phosphorylated nitrones—synthesis and applications. Molecules 2025, 30, 1333. [Google Scholar] [CrossRef]
- Yokoshima, S. Intramolecular cycloaddition of nitrones in total synthesis of natural products. Nat. Prod. Rep. 2025, 42, 1071–1090. [Google Scholar] [CrossRef] [PubMed]
- Stańska, B.; Dahyia, A.; Loska, R. Nitrones as directing groups in transition metal-catalysed C-H activation. Org. Biomol. Chem. 2026, 24, 11–37. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, S.; Prusty, K.; Bhol, S.; Panigrahi, G.; Nayak, S. Nitrone chemistry: a versatile gateway to diverse heterocycles. RSC Adv. 2026, 16, 292–331. [Google Scholar] [CrossRef]
- Zhu, Z.-Y.; Tian, P.; Chang, J.; Li, H.; Kang, Y.; Xie, P.; Yao, T.; Zhang, X. Photocatalytic (3+3) cycloaddition of arylaminocyclopropanes with nitrones: highly diastereoselective synthesis of 1,2-oxazinan-6-amine derivatives. Chem. Commun. 2025, 61, 18428–18431. [Google Scholar] [CrossRef]
- Zhang, M.; Kong, L.; Li, S.; Gao, S.; Li, X. Diastereoselective synthesis of 5,5-difluoroisoxazolidine via [3 + 2] cycloaddition between nitrones and difluoroacrylates. Tetrahedron Lett. 2025, 167, 155647. [Google Scholar] [CrossRef]
- Zaika, Y. O.; Vashchenko, B. V.; Sosunovych, B. S.; Khutorianskyi, A. V.; Yasman, P.; Ogurok, V. M.; Brovarets, V. S.; Grygorenko, O. O. [3+2] Cycloadditions of α,β-unsaturated sultams. Eur. J. Org. Chem. 2025, 28, e202500093. [Google Scholar] [CrossRef]
- Xu, C.; Wang, Y.; Xiao, M.; Tian, R.; Duan, Z. A tandem route toward isoxazolidine fused phospholene skeleton involving dearomative [3+2] cycloaddition of P-heteroarenes and nitrones. Chin. J. Chem. 2025, 43, 1161–1165. [Google Scholar] [CrossRef]
- Wang, S.; Xie, W.; Jiang, C.; Guo, C.; Chang, J.; Zhu, B. Highly regio- and diastereoselective 1,3-DC reaction of BDAs and nitrones: synthesis of the spiro[benzofuran-2,5′-isoxazolidine] framework. J. Org. Chem. 2025, 90, 8531–8541. [Google Scholar] [CrossRef]
- Wang, J.; Luo, S.; Yuan, X.; Cheng, J.; Yang, Z.; Huang, Z. Enantioselective Zn-catalyzed hydrophosphinylation of nitrones: an efficient approach for constructing chiral α-hydroxyamino-phosphine oxides. Chem. Sci. 2025, 16, 7051–7056. [Google Scholar]
- Salih, R. N.; Algso, M.; Mohammad-Salim, H. Mechanistic study of N-t-butyl nitrone and nitroethene (3 + 2) cycloaddition: a combined DFT, docking, and ADMET approach. J. Heterocycl. Chem. 2025, 62, 1328–1343. [Google Scholar] [CrossRef]
- Li, G.-X.; Xu, Z.-J.; Che, C.-M. Fe-BPsalan complex-catalyzed asymmetric 1,3-dipolar [3+2] cycloaddition of nitrones with α,β-unsaturated acyl imidazoles. J. Org. Chem. 2026, 91, 911–924. [Google Scholar] [CrossRef]
- Stanska, B.; Ostrowska, S.; Loska, R. Rhodium-catalyzed C–H annulation of aldonitrones with allyl methyl carbonate. Synlett 2026. [Google Scholar] [CrossRef]
- Toda, Y.; Soma, Y.; Horiba, N.; Koyama, M.; Iwakuma, F.; Matsumoto, S.; Sukegawa, K.; Kikuchi, A.; Suga, H. Chiral amine-urea mediated asymmetric inverse-electron-demand cycloadditions of cyclic nitrones with o-hydroxystyrenes. Org. Biomol. Chem. 2026, 24, 1006–1010. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Ma, L.; Yue, C.; Du, B.; Lu, C.; Long, C.; Wu, W.; Li, S.; He, Q.; Yue, G. Base-controlled diastereoselective [3+2]-cycloaddition and Michael addition of isatin ketonitrones and chalcones to construct spiropyrrolidine oxoindoles and γ-substituted isatin ketonitrones. New J. Chem. 2025, 49, 16978–16988. [Google Scholar] [CrossRef]
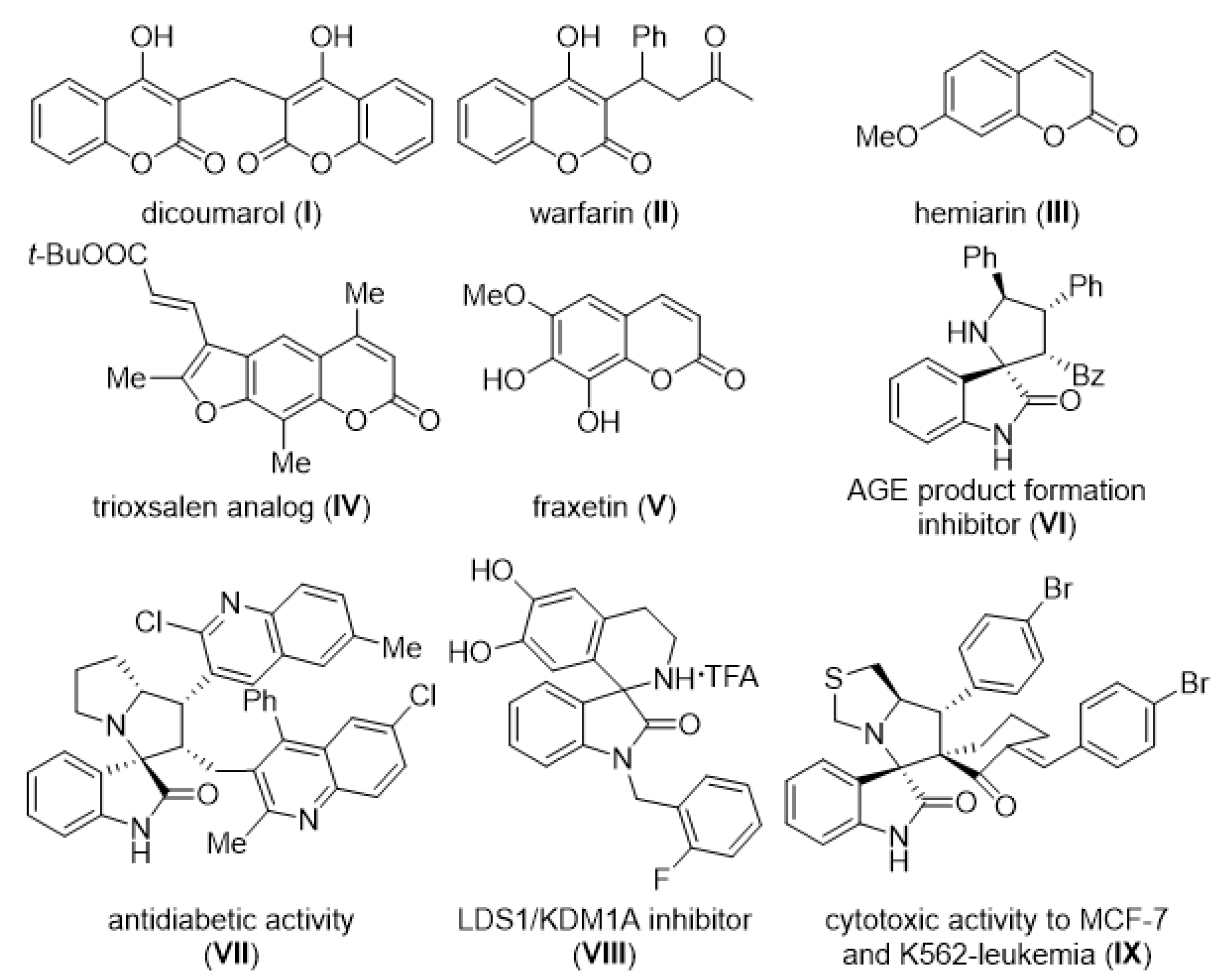

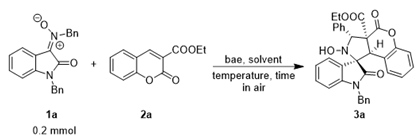
| Entry | Reaction Condition | Yield (%)b,c |
|---|---|---|
| 1 | DBU, 2a, DCM, 24 h | -d |
| 2 | DBU, 2a, DMF, 24 h | complex |
| 3 | DBU, 2a, MeOH, 4 h | 58 |
| 4 | DBU, 2a, EtOH, 5 h | 70 |
| 5 | DBU, 2a, IPA, 11 h | 60 |
| 6 | DBU, 2a, nBuOH, 9 h | 63 |
| 7 | DBU, 2a, tBuOH, 4 h | 54 |
| 8 | DABCO, 2a, EtOH, 4 h | 20e |
| 9 | DMAP, 2a, EtOH, 8 h | 24e |
| 10 | DBU, 2a, dry EtOH, 6 h | 62 |
| 11 | DBU, 2.1 eq 2a, EtOH, 14 h | 71 |
| 12 | 50 mol% DBU, 2a, EtOH, 2.5 h | 51 |
| 13 | 100 mol% DBU, 2a, EtOH, 2.5 h | 36 |
| 14 | DBU, 2a, 1.0 mL, EtOH, 5 h | 74 |
| 15 | DBU, 2a, 4.0 mL, EtOH, 9 h | 69 |
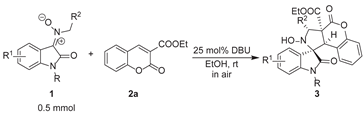
| Entry | Compound | R | R1 | R2 | Yield (%)b |
|---|---|---|---|---|---|
| 1 | 3a | Bn | H | Ph | 74 (74)c |
| 2 | 3b | Bn | 5-Me | Ph | 98 |
| 3 | 3c | Bn | 5-OMe | Ph | 82 |
| 4 | 3d | Bn | 5-F | Ph | 82 |
| 5 | 3e | Bn | 5-Cl | Ph | 74 |
| 6 | 3f | Bn | 6-Br | Ph | 58 |
| 7 | 3g | Bn | 5-I | Ph | 51 |
| 8 | 3h | Bn | 5-NO2 | Ph | -d |
| 9 | 3i | Bn | 7-CF3 | Ph | -d |
| 10 | 3j | H | H | Ph | 76 |
| 11 | 3k | Me | H | Ph | 80 |
| 12 | 3l | Et | H | Ph | 78 |
| 13 | 3m | allyl | H | Ph | 85 |
| 14 | 3n | propargyl | H | Ph | 59 |
| 15 | 3o | H | H | H | -e |
| 16 | 3p | H | H | 4-MeC6H4 | 66 |
| 17 | 3q | H | H | 4-OMeC6H4 | 54 |
| 18 | 3r | H | H | 4-FC6H4 | 78 |
| 19 | 3s | H | H | 4-ClC6H4 | 86 |
| 20 | 3t | H | H | 4-BrC6H4 | 82 |
| 21 | 3u | H | H | 4-NO2C6H4 | 47 |
| 22 | 3v | Bn | 5-Me | H | -d |
| 23 | 3w | Bn | 5-Me | 4-MeC6H4 | 77 |
| 24 | 3x | Bn | 5-Me | 4-OMeC6H4 | 50 |
| 25 | 3y | Bn | 5-Me | 4-FC6H4 | 69 |
| 26 | 3z | Bn | 5-Me | 2-ClC6H4 | 48 |
| 27 | 3aa | Bn | 5-Me | 4-ClC6H4 | 85 |
| 28 | 3ab | Bn | 5-Me | 4-BrC6H4 | 66 |
| 29 | 3ac | Bn | 5-Me | 4-NO2C6H4 | 52 |
| 30 | 3ad | Bn | H | 4-MeC6H4 | 46 |
| 31 | 3ae | Bn | H | 4-OMeC6H4 | 38 |
| 32 | 3af | Bn | H | 4-FC6H4 | 63f |
| 33 | 3ag | Bn | H | 4-ClC6H4 | 76 |
| 34 | 3ah | Bn | H | 4-BrC6H4 | 68 |
| 35 | 3ai | Bn | H | 4-NO2C6H4 | 60 |
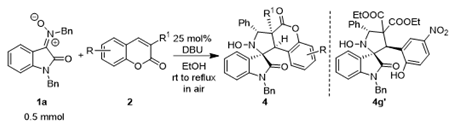
| Entry | Compound | R | R1 | Yield (%)b |
|---|---|---|---|---|
| 1 | 4a | 8-Me | COOEt | 65 |
| 2 | 4b | 7-OMe | COOEt | 22 |
| 3 | 4c | 8-OEt | COOEt | 86 |
| 4 | 4d | 6-F | COOEt | 82 |
| 5 | 4e | 6-Cl | COOEt | 67 |
| 6 | 4f | 6-Br | COOEt | 67 |
| 7 | 4g (4g’) | 6-NO2 | COOEt | 75 (24)c |
| 8 | 4h | 6,8-t-Bu2 | COOEt | 73 |
| 9 | 4i | 7-Et2N | COOEt | -d |
| 10 | 4j | 6,8-Br2 | COOEt | 30 |
| 11 | 4k | H | COOMe | 30e |
| 12 | 4l | H | COOi-Pr | 67 |
| 13 | 4m | H | COOn-Bu | 67 |
| 14 | 4n | H | COOBn | 89e |
| 15 | 4o | H | H | -d |
| 16 | 4p | H | Cl | -d |
| 17 | 4q | H | Br | -d |
| 18 | 4r | H | Bz | -d |
| 19 | 4s | H | COOH | -d |
| 20 | 4t | H | CN | -d |
| 21 | 4u | H | Ac | -d |
| 22 | 4v | H | CONHn-Bu | -d |
| 23 | 4w | H | CONn-Bu2 | -d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




