Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
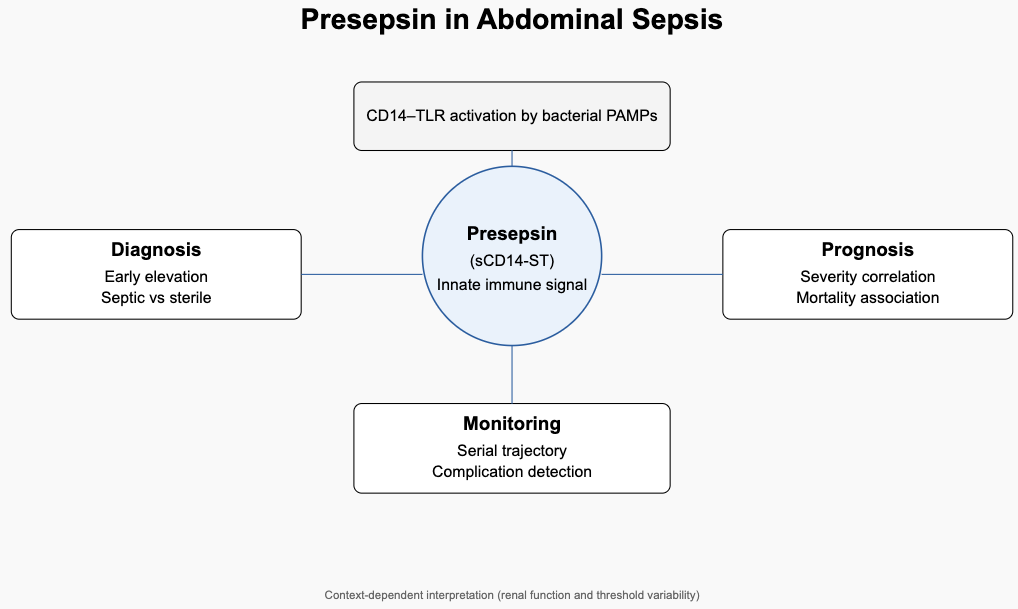
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
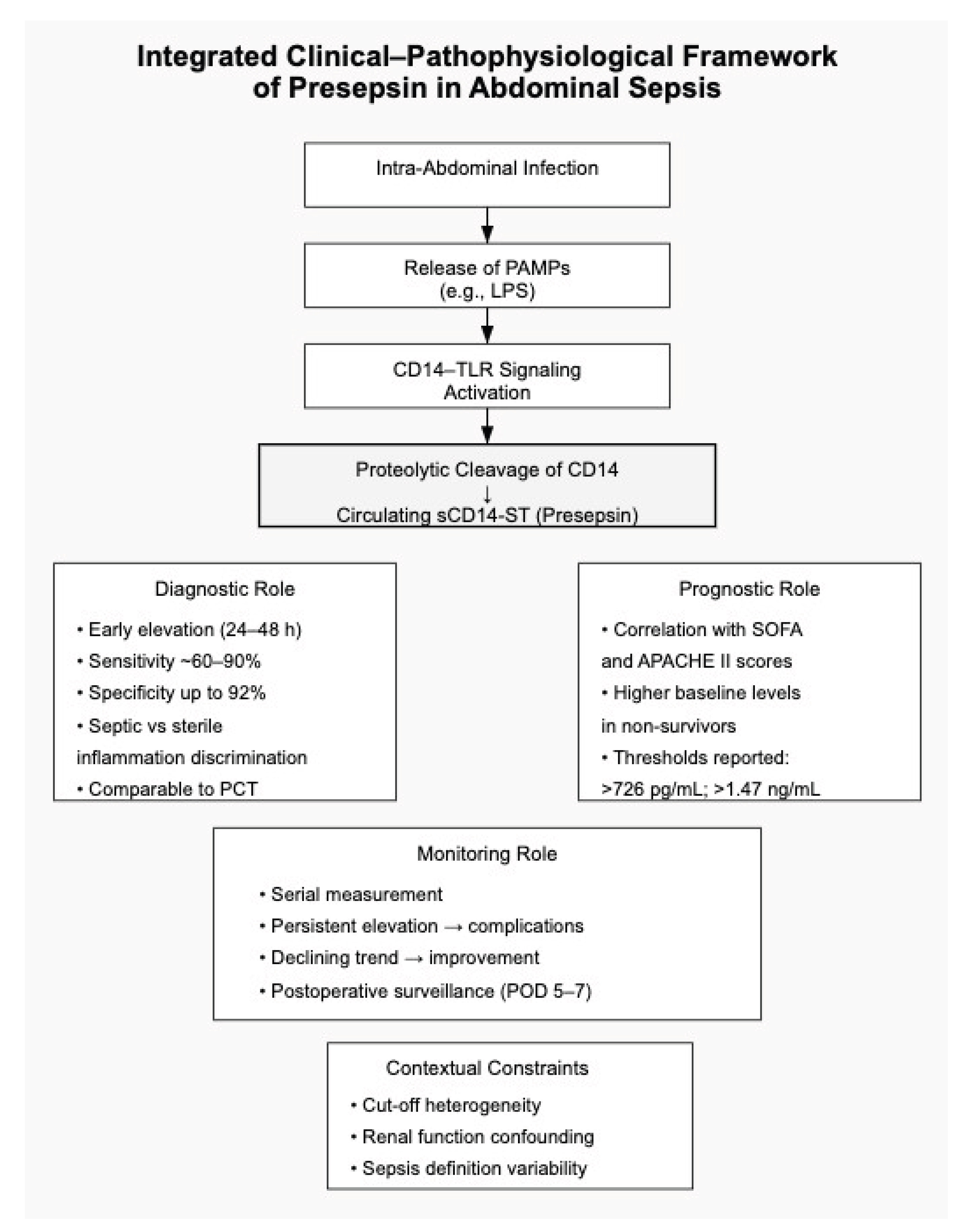
3.1. Diagnostic Performance
3.2. Prognostic Associations
3.3. Longitudinal Monitoring
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- De Backer, D.; et al. Surviving Sepsis Campaign Research Priorities 2023. Crit. Care Med. 2024, 52, 268–296. [CrossRef]
- Lambden, S.; Laterre, P.F.; Levy, M.M.; Francois, B. The SOFA score—Development, utility and challenges of accurate assessment in clinical trials. Crit. Care 2019, 23, 374. [CrossRef]
- Singer, M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [CrossRef]
- Font, M.D.; Thyagarajan, B.; Khanna, A.K. Sepsis and septic shock—Basics of diagnosis, pathophysiology and clinical decision making. Med. Clin. North Am. 2020, 104, 573–585. [CrossRef]
- Sartelli, M.; et al. Global validation of the WSES Sepsis Severity Score for patients with complicated intra-abdominal infections: A prospective multicentre study (WISS Study). World J. Emerg. Surg. 2015, 10, 61. [CrossRef]
- Rogić, D.; Juroš, G.F.; Petrik, J.; Vrančić, A.L. Advances and pitfalls in using laboratory biomarkers for the diagnosis and management of sepsis. EJIFCC 2017, 28, 114–121.
- Póvoa, P.; et al. How to use biomarkers of infection or sepsis at the bedside: Guide to clinicians. Intensive Care Med. 2023, 49, 142–153. [CrossRef]
- Medzhitov, R.; Janeway, C.A. Decoding the patterns of self and nonself by the innate immune system. Science 2002, 296, 298–300. [CrossRef]
- Liu, S.; et al. Expression of CD14 by hepatocytes: Upregulation by cytokines during endotoxemia. Infect. Immun. 1998, 66, 5089–5098.
- Shozushima, T.; et al. Usefulness of presepsin (sCD14-ST) measurements as a marker for the diagnosis and severity of sepsis. J. Infect. Chemother. 2011, 17, 764–769. [CrossRef]
- Velissaris, D.; et al. Presepsin as a diagnostic and prognostic biomarker in sepsis. Cureus 2021, 13, e15019. [CrossRef]
- Claessens, Y.-E.; et al. Plasmatic presepsin (sCD14-ST) concentrations in acute pyelonephritis. Clin. Chim. Acta 2017, 464, 182–188. [CrossRef]
- Uzun, N.; et al. Presepsin is a biomarker that can predict mortality in sepsis patients. Rev. Assoc. Med. Bras. 2025, 71. [CrossRef]
- Henriquez-Camacho, C.; Losa, J. Biomarkers for sepsis. Biomed Res. Int. 2014, 2014, 547818. [CrossRef]
- Vodnik, T.; et al. Presepsin (sCD14-ST) in preoperative diagnosis of abdominal sepsis. Clin. Chem. Lab. Med. 2013, 51, 2053–2062. [CrossRef]
- Jeong, Y.K.; Kim, E.Y. Predictive role of changes in presepsin and early sepsis in ICU patients after abdominal surgery. J. Surg. Res. 2022, 278, 207–215. [CrossRef]
- Paraskevas, T.; et al. Presepsin in the diagnosis of sepsis. Clin. Chim. Acta 2023, 550, 117588. [CrossRef]
- Bösch, F.; et al. The prognostic value of presepsin for sepsis in abdominal surgery: A prospective study. Shock 2020, 54, 56–61. [CrossRef]
- Lu, C.-Y.; et al. Diagnostic efficacy of serum presepsin for postoperative infectious complications: A meta-analysis. Front. Immunol. 2023, 14, 1320683. [CrossRef]
- Drăgoescu, A.N.; et al. Presepsin as a potential prognostic marker for sepsis. J. Pers. Med. 2020, 10, 1–14. [CrossRef]
- Masson, S.; et al. Circulating presepsin as a marker of host response in severe sepsis or septic shock. Intensive Care Med. 2015, 41, 12–20. [CrossRef]
- Amanai, E.; et al. Usefulness of presepsin for early detection of infectious complications after colorectal surgery. Sci. Rep. 2022, 12, 3960. [CrossRef]
- Ozdal, E.; et al. Usability of presepsin, calprotectin, and IL-6 in appendicitis. Biomarkers 2024, 29, 479–484. [CrossRef]
- Sater, M.S.; et al. Potentials of presepsin as a novel sepsis biomarker. Diagnostics 2025, 15, 217. [CrossRef]
- Shakeyev, K.; et al. Presepsin as a risk factor for infectious complications in colorectal surgery. Ann. Coloproctol. 2022, 38, 442–448. [CrossRef]
- Song, X.; et al. Prognostic value of presepsin in enterocutaneous fistula with abdominal sepsis. Int. J. Surg. 2016, 33, 96–101. [CrossRef]
- Takeuchi, M.; et al. Perioperative presepsin as a diagnostic marker after esophagectomy. Esophagus 2020, 17, 399–407. [CrossRef]
- Wang, C.; et al. Early predictive value of presepsin for secondary sepsis in severe acute pancreatitis. Shock 2023, 59, 560–568. [CrossRef]
- Wang, S.; et al. Validity of presepsin for diagnosis and prognosis of sepsis in elderly ICU patients. Minerva Anestesiol. 2020, 86, 123–130. [CrossRef]
- Wejnaruemarn, S.; et al. Procalcitonin and presepsin for detecting bacterial infection and SBP in cirrhosis. World J. Gastroenterol. 2025, 31, 99506. [CrossRef]
- Xiao, H.; et al. Comparison among presepsin, procalcitonin, and CRP in predicting bacteremia. Shock 2024, 61, 387–394. [CrossRef]
- Contenti, J.; et al. Presepsin versus other biomarkers to predict sepsis. Emergencias 2019, 31, 311–317.
- Zong, X.; et al. Early diagnostic value of presepsin in sepsis. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 2024, 36, 340–344. [CrossRef]
- Imai, Y.; et al. Usefulness of presepsin in postoperative infectious complications after gastrectomy. Sci. Rep. 2022, 12, 21289. [CrossRef]
- Aliu-Bejta, A.; et al. Ability of presepsin concentrations to predict mortality in adult sepsis. J. Clin. Transl. Sci. 2023, 7, e121. [CrossRef]
- Chen, M.; Zhu, Y. Utility of sTREM-1 and presepsin as diagnostic and prognostic markers of sepsis. Clin. Lab. 2020, 66. [CrossRef]
- Narendra, S.; et al. Presepsin vs procalcitonin as predictors of sepsis outcome. J. Assoc. Physicians India 2022, 70, 38–40.
- Pluta, M.P.; et al. Presepsin does not predict risk of death in sepsis patients. Biomedicines 2024, 12, 2313. [CrossRef]
- Ren, E.; et al. Value of procalcitonin and presepsin in diagnosis and severity stratification of sepsis. World J. Emerg. Med. 2024, 15, 135. [CrossRef]
- Sartelli, M.; et al. Management of intra-abdominal infections: Recommendations for antimicrobial optimization. World J. Emerg. Surg. 2024, 19, 23. [CrossRef]
- Nagata, T.; et al. Clinical impact of kidney function on presepsin levels. PLoS ONE 2015, 10, e0129159. [CrossRef]
- Nakamura, Y.; et al. Usefulness of presepsin in sepsis with or without acute kidney injury. BMC Anesthesiol. 2014, 14, 88. [CrossRef]
- Yang, H.S.; et al. Prognostic value of presepsin in adult sepsis: Systematic review and meta-analysis. PLoS ONE 2018, 13, e0191486. [CrossRef]
- Kim, H.; et al. Multi-marker approach including presepsin for mortality prediction in sepsis. Ann. Intensive Care 2017, 7, 27. [CrossRef]
| Author (Ref.) | Year | Study Design | N | Sampling Time Points | Main Findings |
| Jeong et al. [16] | 2022 | Observational | 298 | T0, 24 h, 48 h, 72 h | Demonstrated diagnostic accuracy for sepsis and septic shock in the acute postoperative phase. |
| Paraskevas et al. [17] | 2023 | Systematic review | — | — | Identified as a promising biomarker for triage and early sepsis diagnosis. |
| Bösch et al. [18] | 2020 | Prospective | 31 | T0 | Highest AUC, sensitivity, and specificity among evaluated markers; strong association with mortality. |
| Lu et al. [19] | 2023 | Meta-analysis | 984 | — | Pooled sensitivity 76% and specificity 83% for postoperative infectious complications. |
| Drăgonescu et al. [20] | 2020 | Prospective observational | 114 | T0 | Higher levels in sepsis and septic shock; significant correlation with SOFA score. |
| Masson et al. [21] | 2015 | Multicenter randomized (ALBIOS) | 997 | T0, 24 h, 48 h, 7 d | Baseline concentrations increased with disease severity; early rise associated with worse outcomes and 90-day mortality. |
| Amanai et al. [22] | 2022 | Prospective observational | 114 | T0, 24 h, 48 h, 72 h, 4 d, 6 d | Postoperative increases at days 4–6 predicted infectious complications. |
| Ozdal et al. [23] | 2024 | Prospective | 90 | — | Elevated in appendicitis compared with controls; not discriminatory for complicated cases. |
| Sater et al. [24] | 2025 | Cross-sectional | 129 | T0 | Strong correlation with septic shock parameters compared with other biomarkers. |
| Shakeyev et al. [25] | 2022 | Pilot study | 36 | T0, 72 h | Higher baseline levels associated with postoperative complications and organ dysfunction. |
| Song et al. [26] | 2016 | Prospective cohort | 71 | T0 | Values >726 pg/mL associated with greater disease severity. |
| Takeuchi et al. [27] | 2020 | Prospective cohort | 30 | T0, 24 h, 48 h, 72 h, 5 d, 7 d | Measurements at postoperative days 5–7 outperformed WBC, CRP, and PCT for infectious complications. |
| Wang et al. [29] | 2020 | Prospective | 142 | 24 h, 72 h, 7 d | Elevated levels in septic elderly ICU patients; associated with 30-day mortality. |
| Wejnaruemarn et al. [30] | 2025 | Systematic review/meta-analysis | 1789 | — | Pooled sensitivity 75% and specificity 80% for bacterial infections. |
| Zong et al. [33] | 2024 | Prospective cohort | 149 | T0 | Sensitivity 89.5% and PPV 64.6% for early sepsis detection. |
| Aliu-Bejta et al. [35] | 2023 | Observational | 100 | 0–72 h | Higher baseline levels observed in non-survivors. |
| Chen et al. [36] | 2020 | Observational | 60 | 0 h, 24 h, 4 d, 7 d | Persistent elevation associated with poorer prognosis. |
| Narendra et al. [37] | 2022 | Prospective cohort | 92 | T0, 24 h, 48 h, 72 h | Cut-off >1.47 ng/mL predictive of mortality in septic shock. |
| Pluta et al. [38] | 2024 | Prospective | 86 | — | Correlated with positive blood cultures; not independently predictive of mortality. |
| Ren et al. [39] | 2024 | Retrospective | 2225 | T0, 24 h | Higher concentrations in SOFA >5 and septic shock; high specificity (92.2%). |
| Author (Ref.) | Clinical Setting | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Comparator(s) |
| Jeong et al. [16] | Postoperative abdominal surgery | Sepsis: 69.8 (T0), 76.9 (24 h); Septic shock: 83.8 (T0) | Up to 89.8 (48 h) | — | — | PCT |
| Bösch et al. [18] | Emergency abdominal surgery | 70 | 90 | 90 | 30 | PCT, IL-6, WBC |
| Lu et al. [19] | Postoperative infectious complications (meta-analysis) | 76 | 83 | — | — | PCT, CRP |
| Drăgonescu et al. [20] | ICU patients | 79 | 63 | — | — | — |
| Amanai et al. [22] | Colorectal surgery | Up to 87.9 (72 h) | 43.1–87.8 (time-dependent) | — | — | PCT, CRP, WBC |
| Wang et al. [29] | Elderly ICU patients | 82.05–83.33 (24–72 h) | 66.67–89.74 | — | — | PCT, CRP, IL-6 |
| Wejnaruemarn et al. [30] | Cirrhotic patients (meta-analysis) | 75 | 80 | — | — | PCT |
| Zong et al. [33] | Febrile patients | 89.5 | — | 64.6 | — | WBC, CRP, PCT |
| Chen et al. [36] | Sepsis monitoring | 83 | 85 | — | — | sTREM-1 |
| Pluta et al. [38] | ICU sepsis | 93 | 51 | — | — | IL-6, PCT, CRP |
| Ren et al. [39] | Sepsis and septic shock | 39.6 | 92.2 | — | — | PCT, CRP |
| Clinical Role | Key Evidence (Ref.) | Main Findings | Clinical Implication |
| Diagnostic | [16,18,19,20,22,27,29,30,33,39] | Sensitivity generally ranges from 60–90%; specificity up to 92.2% in septic shock; pooled sensitivity 76% and specificity 83% for postoperative infectious complications; pooled sensitivity 75% and specificity 80% in cirrhotic infections. | May provide adjunctive diagnostic information in selected clinical settings, particularly when interpreted alongside established biomarkers and clinical assessment. |
| Prognostic | [21,26,35,37,39] | Baseline concentrations correlate with SOFA score and disease severity; early increases associated with worse outcomes; cut-off >1.47 ng/mL predictive of mortality; values >726 pg/mL associated with greater clinical severity. | May contribute to risk stratification; however, prognostic performance appears context-dependent and requires validation in standardized prospective cohorts. |
| Monitoring | [22,25,27,36] | Persistent postoperative elevation associated with infectious complications and organ dysfunction; measurements at postoperative days 5–7 improved detection of complications; decreasing trends associated with clinical improvement. | Serial measurements may support clinical monitoring in postoperative and critical care contexts, although optimal timing and thresholds remain to be standardized. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




