1. Introduction
Sepsis is currently defined as a life-threatening organ dysfunction resulting from a dysregulated host response to infection [
1]. The Sepsis-3 consensus operationalizes organ dysfunction as an acute increase of at least two points in the Sequential Organ Failure Assessment (SOFA) score, reflecting clinically significant impairment across multiple physiological systems [
2,
3]. Despite advances in critical care management and antimicrobial stewardship, sepsis continues to represent a substantial global health burden. Among its diverse etiologies, intra-abdominal infections (IAIs) constitute a particularly relevant source in surgical populations, frequently leading to severe systemic complications and increased mortality [
4,
5].
Abdominal sepsis poses distinct diagnostic challenges. The clinical presentation is often heterogeneous and may overlap with postoperative physiological inflammatory responses, particularly in patients undergoing major abdominal procedures. Fever, leukocytosis, tachycardia, and elevated inflammatory markers may occur in both sterile postoperative inflammation and evolving infection, thereby complicating early recognition. Although prompt source control and timely antimicrobial therapy are well-established determinants of outcome, diagnostic uncertainty in the early phase remains a critical obstacle to optimal management.
Biomarkers are therefore widely employed to assist in clinical decision-making. C-reactive protein (CRP) and procalcitonin (PCT) are among the most commonly utilized inflammatory markers in surgical and intensive care settings. While both have demonstrated utility in identifying systemic inflammatory states, their specificity in differentiating bacterial infection from non-infectious inflammatory conditions remains suboptimal, particularly in the immediate postoperative context [
6,
7]. Consequently, there is sustained interest in identifying biomarkers that more directly reflect pathogen-driven immune activation.
Presepsin, also known as soluble CD14 subtype (sCD14-ST), has emerged as a candidate biomarker grounded in innate immune biology. CD14 is a glycoprotein expressed primarily on monocytes and macrophages and plays a central role in the recognition of lipopolysaccharide (LPS) and other pathogen-associated molecular patterns through Toll-like receptor-mediated signaling pathways [
8,
9,
10]. During acute inflammatory activation, enzymatic cleavage of CD14 generates presepsin, a 13 kDa circulating fragment detectable in plasma through chemiluminescent immunoassays [
10,
11]. Because its release is mechanistically linked to microbial recognition rather than nonspecific inflammation, presepsin has been proposed as a biomarker with potentially greater specificity for bacterial infection compared with conventional inflammatory markers [
12,
13,
14,
15].
Over the past decade, an expanding body of literature has explored the diagnostic and prognostic implications of presepsin across various clinical contexts, including emergency medicine, critical care, and surgical populations. However, reported performance characteristics vary substantially across studies, and interpretation is complicated by differences in patient selection, timing of measurement, disease severity, and renal function status. In abdominal sepsis in particular, where diagnostic uncertainty frequently complicates early management, a critical appraisal of the available evidence is warranted.
The present narrative review aims to comprehensively evaluate the diagnostic accuracy, prognostic relevance, and potential clinical utility of presepsin in abdominal sepsis and related surgical settings, contextualizing current findings within the broader framework of contemporary sepsis management.
2. Materials and Methods
This study was conducted as a narrative review aimed at critically synthesizing the available evidence regarding the diagnostic and prognostic role of presepsin in abdominal sepsis and related surgical contexts. Given the heterogeneity of existing studies and the exploratory nature of the topic, a narrative approach was considered more appropriate than a formal systematic review or meta-analysis.
A comprehensive literature search was performed using the MEDLINE database (via PubMed) without temporal restriction up to the date of manuscript preparation. The search strategy incorporated combinations of the following keywords: “presepsin”, “sCD14-ST”, “abdominal sepsis”, “intra-abdominal infection”, “postoperative infectious complications”, and “sepsis biomarkers”. Boolean operators (AND, OR) were applied to refine search combinations and optimize retrieval. In addition, reference lists of relevant articles were manually screened to identify further pertinent studies not captured in the initial search.
Eligible publications included prospective and retrospective observational studies, randomized controlled trials, pilot studies, systematic reviews, and meta-analyses evaluating the diagnostic accuracy, prognostic value, or clinical applicability of presepsin in adult patients with abdominal infections, postoperative infectious complications, sepsis, or septic shock. Studies were included irrespective of clinical setting, encompassing surgical wards, intensive care units, and emergency departments. Only articles published in peer-reviewed journals and available in English were considered. Case reports, conference abstracts without full-text availability, and studies lacking quantitative assessment of presepsin were excluded.
For each included study, relevant data were extracted, including year of publication, study design, sample size, patient population, sampling time points, reported diagnostic performance metrics (sensitivity, specificity, positive and negative predictive values, and, when available, area under the receiver operating characteristic curve), associations with severity scores (e.g., SOFA, APACHE II), and reported mortality outcomes. Where appropriate, comparisons with established biomarkers such as CRP, PCT, interleukin-6, or other inflammatory markers were documented to contextualize relative performance.
Given the methodological heterogeneity across studies—including differences in inclusion criteria, sepsis definitions (Sepsis-2 versus Sepsis-3), timing of measurement, and reported cut-off thresholds—a quantitative pooled analysis was not undertaken. Instead, findings were qualitatively synthesized and structured according to three principal domains: diagnostic discrimination, prognostic stratification, and longitudinal monitoring. This framework informed the organization of
Table 1,
Table 2 and
Table 3 and guided the interpretative discussion.
No new patient data were generated or analyzed. Accordingly, institutional review board approval and informed consent were not required for the present study.
3. Results
The main findings of the included studies are summarized in
Table 1 and
Table 2. In accordance with the methodological framework adopted for this review, the available evidence is presented across three complementary domains: diagnostic discrimination, prognostic stratification, and longitudinal monitoring
3.1. Diagnostic Performance
Across heterogeneous surgical and critical care populations, presepsin demonstrated moderate-to-high diagnostic accuracy for identifying sepsis and septic shock. In postoperative abdominal surgery cohorts, early sampling frequently yielded clinically relevant discriminatory performance. In a study of 298 patients undergoing major abdominal procedures, sensitivity for sepsis reached 69.8% at baseline (T0) and increased to 76.9% at 24 hours, while specificity reached 82% at 48 hours. For septic shock, sensitivity was 83.8% at T0, with specificity up to 89.8% at 48 hours [
16]. In a prospective emergency surgical cohort, sensitivity and specificity were reported at 70% and 90%, respectively, with positive predictive value of 90% [
18].
Meta-analytic data further support diagnostic performance in the postoperative setting. In pooled analyses of postoperative infectious complications (n = 984), sensitivity and specificity were 76% and 83%, respectively [
19]. Similarly, in cirrhotic populations evaluated for bacterial infection, pooled sensitivity and specificity were 75% and 80% [
30]. These findings indicate consistent discriminatory capacity across selected high-risk populations. In critically ill patients, performance characteristics varied according to disease severity and timing of measurement. In ICU cohorts, sensitivity values ranged from 79% [
20] to 82–83% at 24–72 hours [
29], with specificity ranging from 63% [
20] to 89.74% [
29]. In febrile patients evaluated for early sepsis, sensitivity reached 89.5%, with a positive predictive value of 64.6% [
33]. Conversely, in a large retrospective cohort of 2225 patients, specificity for septic shock reached 92.2%, although sensitivity was lower (39.6%) [
39], reflecting stronger rule-in performance in advanced disease states.
Collectively, these data demonstrate that reported sensitivity typically ranges between approximately 60% and 90%, while specificity may reach values exceeding 90% in selected contexts (
Table 2). However, variability in thresholds, sampling strategies, and comparator biomarkers contributes to inter-study heterogeneity.
3.2. Prognostic Associations
Beyond diagnostic discrimination, several studies reported significant associations between presepsin concentrations and indices of disease severity and mortality. Correlations with SOFA and APACHE II scores were consistently documented across multiple cohorts [
20,
21,
26,
39]. In the multicenter ALBIOS trial (n = 997), baseline concentrations increased proportionally with organ dysfunction severity, and early rises were associated with adverse clinical outcomes and increased 90-day mortality [
21]. Similarly, observational data demonstrated higher baseline values in non-survivors compared with survivors [
35], and a cut-off value exceeding 1.47 ng/mL was associated with mortality in septic shock patients [
37]. In abdominal sepsis complicated by enterocutaneous fistula, concentrations greater than 726 pg/mL were associated with greater disease severity [
26].
These findings indicate that baseline levels and early dynamic changes may reflect the intensity of systemic inflammatory activation and organ dysfunction burden. Nevertheless, heterogeneity in reported thresholds and outcome definitions limits direct comparability across studies.
3.3. Longitudinal Monitoring
A subset of studies evaluated serial measurements and their association with clinical course. In postoperative cohorts, persistent elevation during the early postoperative period was associated with infectious complications and organ dysfunction [
22,
25]. Measurements performed at postoperative days 5–7 demonstrated improved detection of infectious complications compared with conventional inflammatory markers [
27]. In observational ICU studies, persistently elevated concentrations were associated with poorer prognosis, whereas decreasing trends were observed in patients demonstrating clinical improvement [
36].
These observations suggest that temporal trajectories may provide additional contextual information beyond single baseline measurements. However, the optimal timing, frequency, and clinical integration of serial assessment remain insufficiently standardized.
4. Discussion
This narrative review synthesizes the current evidence regarding presepsin (soluble CD14 subtype, sCD14-ST) as a diagnostic, prognostic, and monitoring biomarker in abdominal sepsis. Collectively, the available data indicate that presepsin increases early during bacterial infection, correlates with organ dysfunction severity, and may provide clinically relevant prognostic information. Nonetheless, substantial heterogeneity in study design, patient populations, and diagnostic thresholds necessitates cautious interpretation.
From a mechanistic standpoint, presepsin reflects activation of the innate immune response through CD14-mediated recognition of pathogen-associated molecular patterns, particularly lipopolysaccharide derived from Gram-negative bacteria [
8,
9,
10]. Proteolytic cleavage of CD14 during monocyte–macrophage activation generates circulating sCD14-ST, providing a biologically coherent explanation for its rapid elevation during systemic infection [
10,
11]. This mechanistic specificity distinguishes presepsin from nonspecific inflammatory markers such as CRP, which may increase in sterile inflammatory conditions [
6,
7], and supports its proposed relevance in differentiating infectious from non-infectious postoperative systemic inflammatory responses [
12,
13,
14,
15].
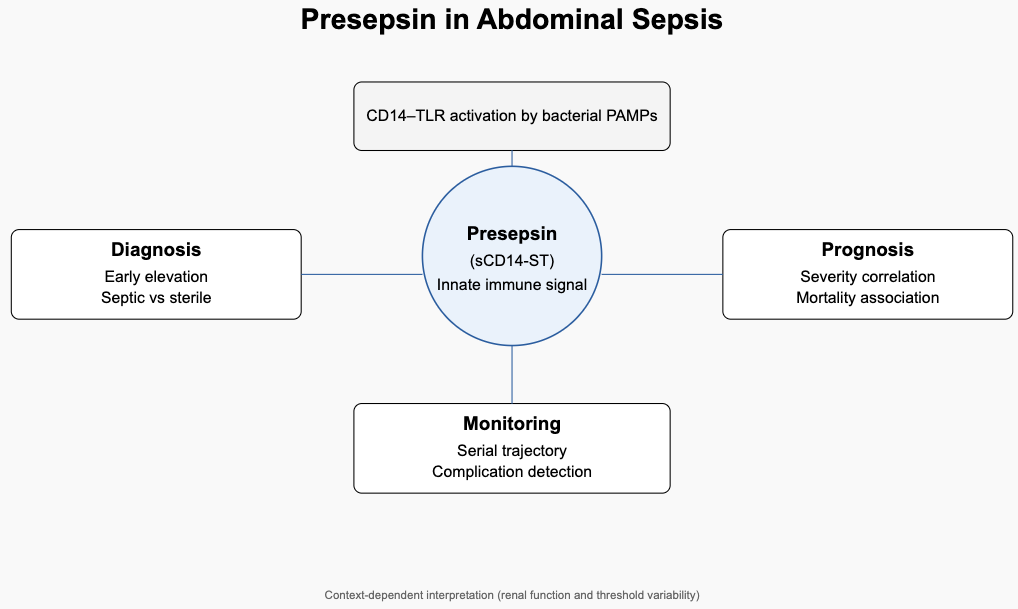
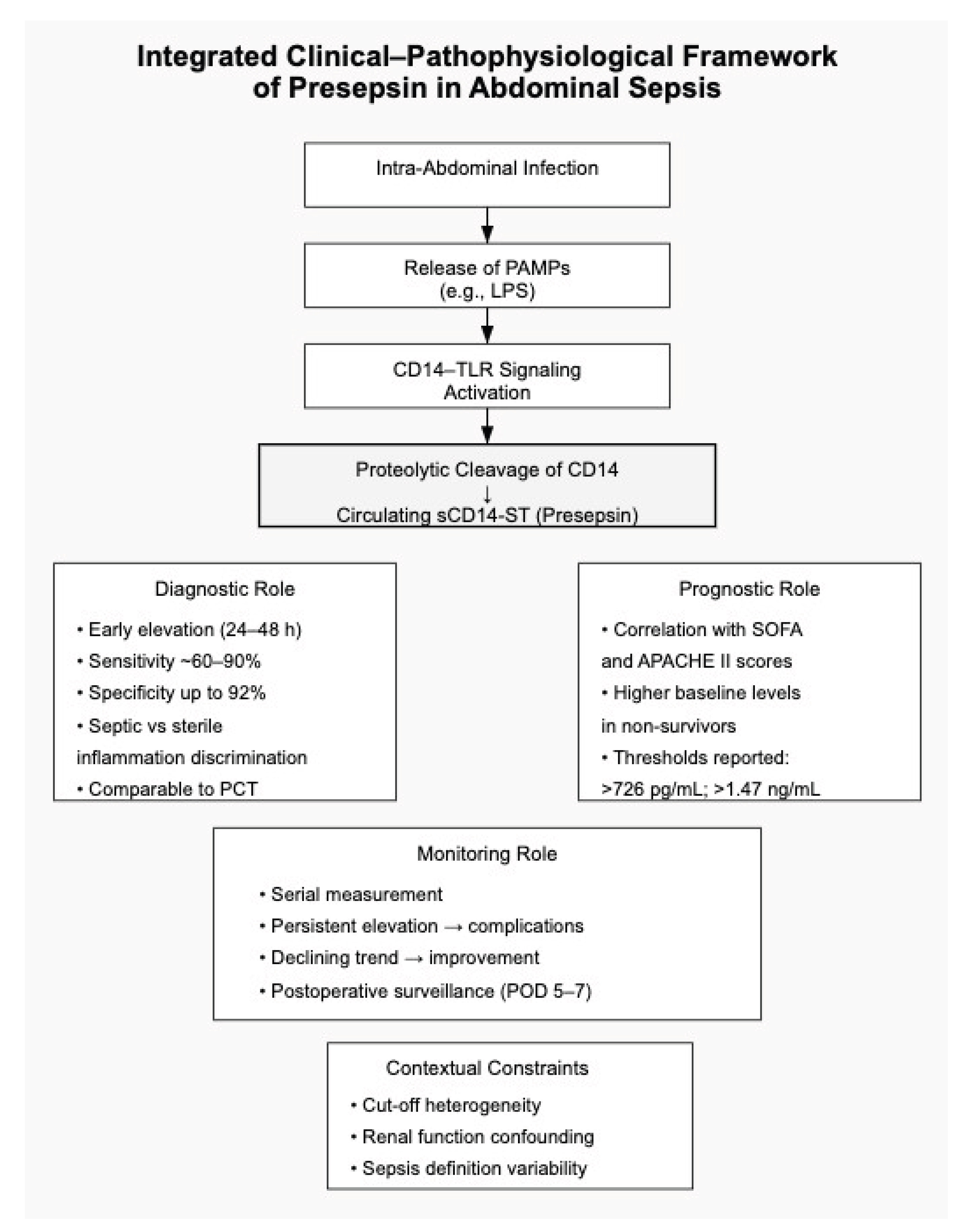
The cumulative evidence may be interpreted across three interrelated domains—diagnostic discrimination, prognostic stratification, and longitudinal monitoring—as summarized in
Table 3. While
Table 3 provides a structured comparative overview of these domains, the mechanistic and clinical interconnections underlying these roles are conceptually integrated in
Figure 1. As illustrated, presepsin occupies a central interface between pathogen recognition and systemic inflammatory amplification. Its early elevation reflects innate immune activation; its magnitude correlates with organ dysfunction burden; and its temporal trajectory may offer additional contextual information regarding disease progression. Importantly, this integrated framework also highlights contextual modifiers—such as renal function and threshold variability—that influence interpretation and limit the applicability of universal cut-off values. As schematically represented, presepsin occupies a central interface between pathogen recognition and measurable systemic response. Its early rise reflects innate immune activation; its magnitude correlates with organ dysfunction burden; and its temporal trajectory may provide additional information regarding disease evolution. Importantly, the framework also emphasizes contextual modifiers, including renal function and disease severity, which influence circulating concentrations and must be considered during interpretation.
In surgical populations, particularly following major abdominal procedures, presepsin demonstrated moderate-to-high diagnostic accuracy [
16,
18,
22,
27]. Meta-analytic data reported pooled sensitivity and specificity of 76% and 83%, respectively, for postoperative infectious complications [
19], while pooled estimates of 75% and 80% were observed in cirrhotic patients evaluated for bacterial infections [
30]. These findings suggest clinically meaningful discriminatory capacity in high-risk settings characterized by diagnostic uncertainty.
Beyond diagnosis, several investigations consistently documented significant associations between presepsin concentrations and established severity indices, including SOFA and APACHE II scores [
20,
21,
26,
39]. In the ALBIOS cohort, early increases were associated with adverse outcomes [
21], and higher baseline values were reported among non-survivors in independent cohorts [
35,
37]. These observations indicate that presepsin may reflect the intensity of systemic inflammatory activation and organ dysfunction, extending its relevance beyond binary diagnostic classification.
Nevertheless, comparative performance relative to established biomarkers remains heterogeneous. While certain studies reported comparable or superior diagnostic accuracy compared with procalcitonin [
18,
32], others observed stronger predictive value for bacteremia using PCT in selected contexts [
31]. Such discrepancies likely reflect differences in infection source, sampling timing, disease severity, and applied cut-off thresholds.
Variability in reported diagnostic thresholds represents a major limitation. Cut-off values differ substantially across studies, ranging from lower thresholds in postoperative cohorts [
26] to higher concentrations in septic shock populations [
39]. Additionally, renal function significantly influences presepsin levels due to glomerular filtration and tubular metabolism [
41,
42]. As highlighted in
Figure 1, these contextual modifiers complicate the establishment of universally applicable thresholds and reinforce the need for population-specific interpretation.
Methodologically, most available studies are observational and vary in inclusion criteria, sampling schedules, and comparator biomarkers. Although cumulative evidence supports biological plausibility and clinical potential, the incremental contribution of presepsin within standardized multimodal diagnostic frameworks remains to be definitively established. Within the broader landscape of sepsis biomarker research, integrative approaches that combine pathophysiological specificity with contextual clinical interpretation are increasingly emphasized [
1,
40]. In this setting, presepsin appears most appropriately conceptualized as an adjunctive component within a composite diagnostic strategy rather than as a standalone determinant of clinical decision-making
5. Conclusions
Presepsin is a biologically grounded biomarker reflecting innate immune activation during bacterial infection. The available evidence indicates moderate-to-high diagnostic performance in selected clinical contexts, meaningful correlation with organ dysfunction severity, and potential prognostic relevance in abdominal sepsis. However, variability in diagnostic thresholds, methodological heterogeneity, and renal function confounding limit the generalizability of current findings. Its clinical role appears most appropriate within a multimodal diagnostic framework, pending confirmation through standardized prospective investigations designed to clarify its incremental value over established biomarkers.
Author Contributions
Conceptualization, M.F. and G.C.; methodology, M.F.; software, F.M.R.; validation, V.P., P.S. and F.C.; investigation, G.C. and L.S.; data curation, G.C.; writing—original draft preparation, M.F. and G.C.; writing—review and editing, M.F. and G.C.; visualization, M.F.; supervision, F.S. and M.C.P.; funding acquisition, V.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This study did not require ethical approval.
Informed Consent Statement
Not applicable.
Data Availability Statement
This manuscript constitutes a narrative review of previously published literature. No new data were generated or analyzed during the study; consequently, data availability is not applicable.
Acknowledgments
This manuscript constitutes a narrative review of previously published literature. No new data were generated or analyzed during the study; consequently, data availability is not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
References
- De Backer, D.; et al. Surviving Sepsis Campaign Research Priorities 2023. Crit. Care Med. 2024, 52, 268–296. [CrossRef]
- Lambden, S.; Laterre, P.F.; Levy, M.M.; Francois, B. The SOFA score—Development, utility and challenges of accurate assessment in clinical trials. Crit. Care 2019, 23, 374. [CrossRef]
- Singer, M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [CrossRef]
- Font, M.D.; Thyagarajan, B.; Khanna, A.K. Sepsis and septic shock—Basics of diagnosis, pathophysiology and clinical decision making. Med. Clin. North Am. 2020, 104, 573–585. [CrossRef]
- Sartelli, M.; et al. Global validation of the WSES Sepsis Severity Score for patients with complicated intra-abdominal infections: A prospective multicentre study (WISS Study). World J. Emerg. Surg. 2015, 10, 61. [CrossRef]
- Rogić, D.; Juroš, G.F.; Petrik, J.; Vrančić, A.L. Advances and pitfalls in using laboratory biomarkers for the diagnosis and management of sepsis. EJIFCC 2017, 28, 114–121.
- Póvoa, P.; et al. How to use biomarkers of infection or sepsis at the bedside: Guide to clinicians. Intensive Care Med. 2023, 49, 142–153. [CrossRef]
- Medzhitov, R.; Janeway, C.A. Decoding the patterns of self and nonself by the innate immune system. Science 2002, 296, 298–300. [CrossRef]
- Liu, S.; et al. Expression of CD14 by hepatocytes: Upregulation by cytokines during endotoxemia. Infect. Immun. 1998, 66, 5089–5098.
- Shozushima, T.; et al. Usefulness of presepsin (sCD14-ST) measurements as a marker for the diagnosis and severity of sepsis. J. Infect. Chemother. 2011, 17, 764–769. [CrossRef]
- Velissaris, D.; et al. Presepsin as a diagnostic and prognostic biomarker in sepsis. Cureus 2021, 13, e15019. [CrossRef]
- Claessens, Y.-E.; et al. Plasmatic presepsin (sCD14-ST) concentrations in acute pyelonephritis. Clin. Chim. Acta 2017, 464, 182–188. [CrossRef]
- Uzun, N.; et al. Presepsin is a biomarker that can predict mortality in sepsis patients. Rev. Assoc. Med. Bras. 2025, 71. [CrossRef]
- Henriquez-Camacho, C.; Losa, J. Biomarkers for sepsis. Biomed Res. Int. 2014, 2014, 547818. [CrossRef]
- Vodnik, T.; et al. Presepsin (sCD14-ST) in preoperative diagnosis of abdominal sepsis. Clin. Chem. Lab. Med. 2013, 51, 2053–2062. [CrossRef]
- Jeong, Y.K.; Kim, E.Y. Predictive role of changes in presepsin and early sepsis in ICU patients after abdominal surgery. J. Surg. Res. 2022, 278, 207–215. [CrossRef]
- Paraskevas, T.; et al. Presepsin in the diagnosis of sepsis. Clin. Chim. Acta 2023, 550, 117588. [CrossRef]
- Bösch, F.; et al. The prognostic value of presepsin for sepsis in abdominal surgery: A prospective study. Shock 2020, 54, 56–61. [CrossRef]
- Lu, C.-Y.; et al. Diagnostic efficacy of serum presepsin for postoperative infectious complications: A meta-analysis. Front. Immunol. 2023, 14, 1320683. [CrossRef]
- Drăgoescu, A.N.; et al. Presepsin as a potential prognostic marker for sepsis. J. Pers. Med. 2020, 10, 1–14. [CrossRef]
- Masson, S.; et al. Circulating presepsin as a marker of host response in severe sepsis or septic shock. Intensive Care Med. 2015, 41, 12–20. [CrossRef]
- Amanai, E.; et al. Usefulness of presepsin for early detection of infectious complications after colorectal surgery. Sci. Rep. 2022, 12, 3960. [CrossRef]
- Ozdal, E.; et al. Usability of presepsin, calprotectin, and IL-6 in appendicitis. Biomarkers 2024, 29, 479–484. [CrossRef]
- Sater, M.S.; et al. Potentials of presepsin as a novel sepsis biomarker. Diagnostics 2025, 15, 217. [CrossRef]
- Shakeyev, K.; et al. Presepsin as a risk factor for infectious complications in colorectal surgery. Ann. Coloproctol. 2022, 38, 442–448. [CrossRef]
- Song, X.; et al. Prognostic value of presepsin in enterocutaneous fistula with abdominal sepsis. Int. J. Surg. 2016, 33, 96–101. [CrossRef]
- Takeuchi, M.; et al. Perioperative presepsin as a diagnostic marker after esophagectomy. Esophagus 2020, 17, 399–407. [CrossRef]
- Wang, C.; et al. Early predictive value of presepsin for secondary sepsis in severe acute pancreatitis. Shock 2023, 59, 560–568. [CrossRef]
- Wang, S.; et al. Validity of presepsin for diagnosis and prognosis of sepsis in elderly ICU patients. Minerva Anestesiol. 2020, 86, 123–130. [CrossRef]
- Wejnaruemarn, S.; et al. Procalcitonin and presepsin for detecting bacterial infection and SBP in cirrhosis. World J. Gastroenterol. 2025, 31, 99506. [CrossRef]
- Xiao, H.; et al. Comparison among presepsin, procalcitonin, and CRP in predicting bacteremia. Shock 2024, 61, 387–394. [CrossRef]
- Contenti, J.; et al. Presepsin versus other biomarkers to predict sepsis. Emergencias 2019, 31, 311–317.
- Zong, X.; et al. Early diagnostic value of presepsin in sepsis. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 2024, 36, 340–344. [CrossRef]
- Imai, Y.; et al. Usefulness of presepsin in postoperative infectious complications after gastrectomy. Sci. Rep. 2022, 12, 21289. [CrossRef]
- Aliu-Bejta, A.; et al. Ability of presepsin concentrations to predict mortality in adult sepsis. J. Clin. Transl. Sci. 2023, 7, e121. [CrossRef]
- Chen, M.; Zhu, Y. Utility of sTREM-1 and presepsin as diagnostic and prognostic markers of sepsis. Clin. Lab. 2020, 66. [CrossRef]
- Narendra, S.; et al. Presepsin vs procalcitonin as predictors of sepsis outcome. J. Assoc. Physicians India 2022, 70, 38–40.
- Pluta, M.P.; et al. Presepsin does not predict risk of death in sepsis patients. Biomedicines 2024, 12, 2313. [CrossRef]
- Ren, E.; et al. Value of procalcitonin and presepsin in diagnosis and severity stratification of sepsis. World J. Emerg. Med. 2024, 15, 135. [CrossRef]
- Sartelli, M.; et al. Management of intra-abdominal infections: Recommendations for antimicrobial optimization. World J. Emerg. Surg. 2024, 19, 23. [CrossRef]
- Nagata, T.; et al. Clinical impact of kidney function on presepsin levels. PLoS ONE 2015, 10, e0129159. [CrossRef]
- Nakamura, Y.; et al. Usefulness of presepsin in sepsis with or without acute kidney injury. BMC Anesthesiol. 2014, 14, 88. [CrossRef]
- Yang, H.S.; et al. Prognostic value of presepsin in adult sepsis: Systematic review and meta-analysis. PLoS ONE 2018, 13, e0191486. [CrossRef]
- Kim, H.; et al. Multi-marker approach including presepsin for mortality prediction in sepsis. Ann. Intensive Care 2017, 7, 27. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |