Submitted:
11 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
1. Basic Knowledge of Insulin Secretory Oscillations
1.1. Introduction
1.2. Description and Definitions of Voltage Pulsatility
1.3. Which Oscillating β-Cell Process Is Responsible for Insulin Secretion Oscillations?
2. Insulin Secretory Oscillations Are Produced by Glycolytic Oscillations in the β-Cell
2.1. Preliminary Data
2.2. Mechanism of Glycolytic Oscillations
2.3. Catalytic Regulation of Recombinant Human PFK-M
2.4. Catalytic Regulation of PFK in Pancreatic Islets and Insulinoma Cells
3. Are Insulin Secretory Oscillations Determined by Metabolic or Plasma Membrane Ionic Channels Oscillations?
4. Does the Experimental Strategy to Suppress [Ca2+]cyt Oscillations with Diazoxide and High KCl Concentrations Exerts off Target Effects on β-Cell Glucose Metabolism?
5. Are Insulin Secretory Oscillations Regulated by the Antagonistic Roles of Two β-Cell Plasma Membrane Channels?
5.1. Proposal of the Two Channels´s Hypothesis
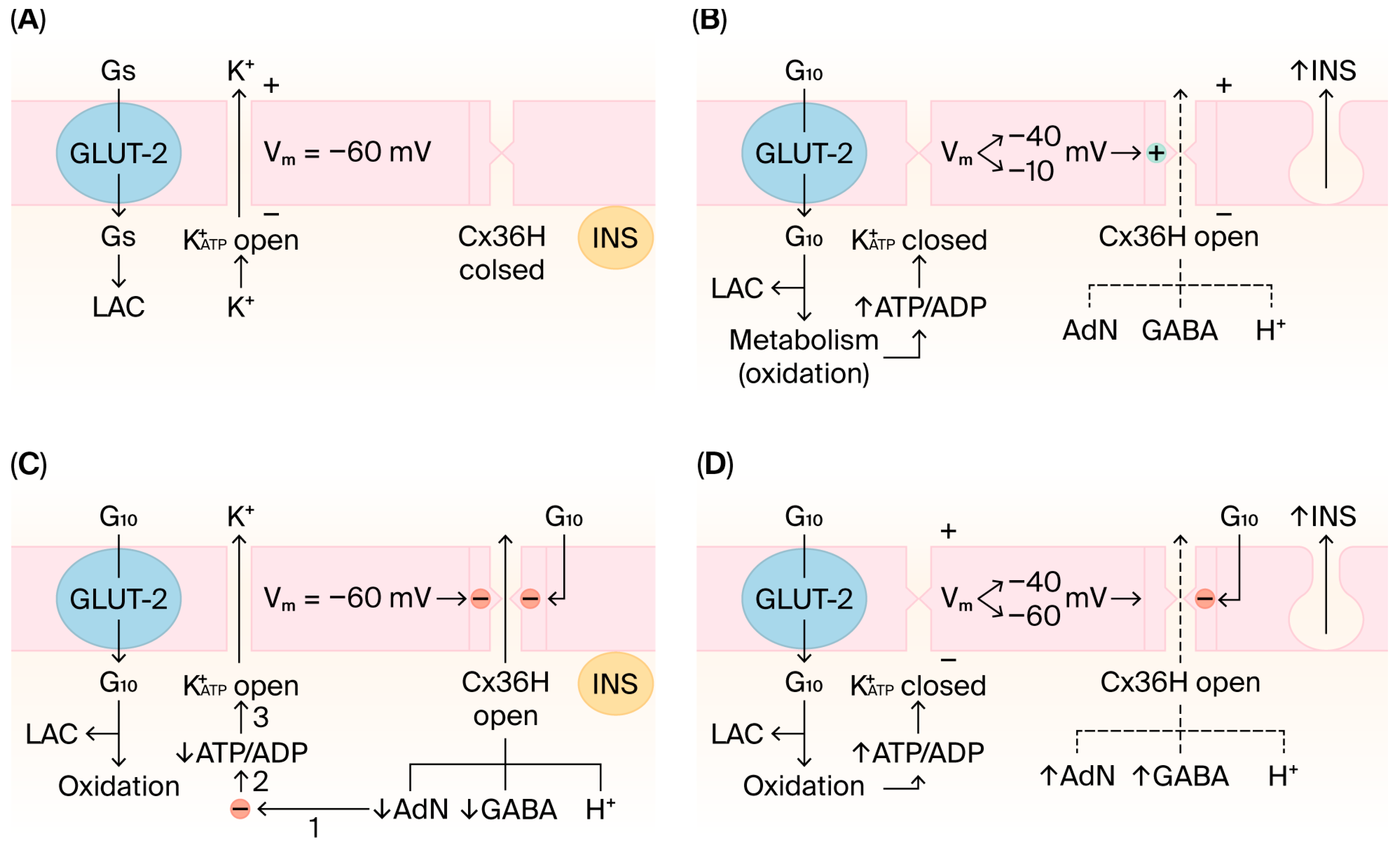
5.2. Contradiction Between the Dual Channel Hypothesis and Those Relying on a Direct Driving of K+ATP Regulation by β-Cell Glucose Metabolism (Glycolysis or Oxphos)
Funding
Acknowledgments
References
- Rorsman, P.; Ashcroft, F.M. Pancreatic β-Cell Electrical Activity and Insulin Secretion: Of Mice and Men. Physiol. Rev. 2018, 98, 117–214. [Google Scholar] [CrossRef]
- Dean, P.M.; Matthew, E.K. Glucose-induced electrical activity in pancreatic islet cells. J. Physiol. 1970, 210, 255–264. [Google Scholar] [CrossRef]
- Tornheim, K. Are Metabolic Oscillations Responsible for Normal Oscillatory Insulin Secretion? Diabetes 1997, 46, 1375–1380. [Google Scholar] [CrossRef]
- Hellman, B. Pulsatility of insulin release – a clinically important phenomenon. Upsala J. Med Sci. 2009, 114, 193–205. [Google Scholar] [CrossRef]
- Pralong, W.; Bartley, C.; Wollheim, C. Single islet beta-cell stimulation by nutrients: relationship between pyridine nucleotides, cytosolic Ca2+ and secretion. EMBO J. 1990, 9, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Longo, E.; Tornheim, K.; Deeney, J.; Varnum, B.; Tillotson, D.; Prentki, M.; Corkey, B. Oscillations in cytosolic free Ca2+, oxygen consumption, and insulin secretion in glucose-stimulated rat pancreatic islets. J. Biol. Chem. 1997, 266, 9314–9319. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, T.; Schultz, V.; Berggren, B.E.; Tornheim, K. Temporal patterns of changes in ATP/ADP ratio, glucose 6-phosphate and cytoplasmic free Ca2+ in glucose-stimulated pancreatic b-cells. Biochem. J. 1996, 314, 91–94. [Google Scholar] [CrossRef]
- Martin, F.; Sánchez-Andrés, J.V.; Soria, B. Slow [Ca2+]i oscillations induced by ketoisocaproate in single mouse pancreatic islets. Diabetes 1995, 44, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Lenzen, S.; Lerch, M.; Peckmann, T.; Tiedge, M. Differential regulation of [Ca2+]i oscillations in mouse pancreatic islets by glucose, α-ketoisocaproic acid, glyceraldehyde and glycolytic intermediates. Biochim. et Biophys. Acta (BBA) - Gen. Subj. 2000, 1523, 65–72. [Google Scholar] [CrossRef]
- Tornheim, K.; Löwenstein, J.M. The purine nucleotide cycle control of phosphofructokinase and glycolytic oscillations in muscle extracts. J. Biol. Chem. 1975, 250, 6304–6314. [Google Scholar] [CrossRef]
- Yaney, G.C.; Schultz, V.; Cunningham, B.A.; Dunaway, G.A.; Corkey, B.E.; Tornheim, K. Phosphofructokinase isozymes in pancreatic islets and clonal beta-cells (INS-1). Diabetes 1995, 44, 1285–1289. [Google Scholar] [CrossRef]
- Fernandes, P.M.; Kinkead, J.; McNae, I.; Michels, P.A.; Walkinshaw, M.D. Biochemical and transcript level differences between the three human phosphofructokinases show optimisation of each isoform for specific metabolic niches. Biochem. J. 2020, 477, 4425–4441. [Google Scholar] [CrossRef]
- Sugden, P.H.; Newsholme, E.A. The effects of amnonium, inorganic phosphate and potassium ions on the activity of phosphofructokinases from muscle and nervous tissues of vertebrates and invertebrates. Biochem. J. 1975, 150, 113–122. [Google Scholar] [CrossRef]
- Tornheim, K.; Lowenstein, J.M. The purine nucleotide cycle. 3. Oscillations in metabolite concentrations during the operation of the cycle in muscle extracts. J. Biol. Chem. 1973, 248, 2670–7. [Google Scholar] [CrossRef]
- Ma, Z.; Ramanadham, S.; Kempe, K.; Hu, Z.; Ladenson, J.; Turk, J. Characterization of expression of phosphofructokinase isoforms in isolated rat pancreatic islets and purified beta cells and cloning and expression of the rat phosphofructokinase-A isoform. Biochim. Biophys. Acta 1996, 1308, 151–163. [Google Scholar] [CrossRef]
- Schuit, F.; Moens, K.; Heimberg, H.; Pipeleers, D. Cellular Origin of Hexokinase in Pancreatic Islets. J. Biol. Chem. 1999, 274, 32803–32809. [Google Scholar] [CrossRef] [PubMed]
- Marinelli, I.; Parekh, V.; Fletcher, P.; Thompson, B.; Ren, J.; Tang, X.; Saunders, T.L.; Ha, J.; Sherman, A.; Bertram, R.; et al. Slow oscillations persist in pancreatic beta cells lacking phosphofructokinase M. Biophys. J. 2022, 121, 692–704. [Google Scholar] [CrossRef]
- Carbó, R.; Rodríguez, E. Relevance of Sugar Transport across the Cell Membrane. Int. J. Mol. Sci. 2023, 24, 6085. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.E. Isozymes of mammalian hexokinase: structure, subcellular localization and metabolic function. J. Exp. Biol. 2003, 206, 2049–2057. [Google Scholar] [CrossRef]
- Bedoya, F. J.; Wilson, J. M.; Ghosh, A. K.; Finegold, D.; Matschinsky, F. M. The glucokinase sensor in human pancreatic islet tissue. Diabetes 1986, 35, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Tamarit-Rodriguez, J.; Idahl, L.-Å.; Giné, E.; Alcazar, O.; Sehlin, J. Lactate production by pancreatic islets. Diabetes 1998, 47, 1219–1223. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Parker, J.C.; Najafi, H.; Matschinsky, F.M. Control of Glucose Metabolism in Pancreatic β-Cells by Glucokinase, Hexokinase and Phosphofructokinase: Model Study With Cell Lines Derived From β-Cells. Diabetes 1988, 37, 1524–1530. [Google Scholar] [CrossRef] [PubMed]
- Merrins, M.J.; Bertram, R.; Sherman, A.; Satin, L.S. Phosphofructo-2-kinase/Fructose-2,6-bisphosphatase Modulates Oscillations of Pancreatic Islet Metabolism. PLOS ONE 2012, 7, e34036. [Google Scholar] [CrossRef] [PubMed]
- Chou, H.-F.; Berman, N.; Ipp, E. Oscillations of lactate released from islets of Langerhans: evidence for oscillatory glycolysis in β-cells. Am. J. Physiol. Endocrinol. Metab. 1992, 262, E800–E805. [Google Scholar] [CrossRef]
- Ashcroft, F.M.; Rorsman, P. Electrophysiology of the pancreatic β-cell. Prog. Biophys. Mol. Biol. 1989, 54, 87–143. [Google Scholar] [CrossRef]
- Mazurek, S. Pyruvate kinase type M2: A key regulator of the metabolic budget system in tumor cells. Int. J. Biochem. Cell Biol. 2011, 43, 969–980. [Google Scholar] [CrossRef]
- Merrins, M.J.; Van Dyke, A.R.; Mapp, A.K.; Rizzo, M.A.; Satin, L.S. Direct Measurements of Oscillatory Glycolysis in Pancreatic Islet β-Cells Using Novel Fluorescence Resonance Energy Transfer (FRET) Biosensors for Pyruvate Kinase M2 Activity. J. Biol. Chem. 2013, 288, 33312–33322. [Google Scholar] [CrossRef]
- Gilon, P.; Ravier, M.A.; Jonas, J.-C.; Henquin, J.-C. Control Mechanisms of the Oscillations of Insulin Secretion In Vitro and In Vivo. Diabetes 2002, 51, S144–S151. [Google Scholar] [CrossRef]
- Bertram, R.; Sherman, A.; Satin, L.S. Metabolic and electrical oscillations: partners in controlling pulsatile insulin secretion. Am. J. Physiol. Metab. 2007, 293, E890–E900. [Google Scholar] [CrossRef]
- Larsson, O.; Kindmark, H.; Brandstrom, R.; Fredholm, B.; O Berggren, P. Oscillations in KATP channel activity promote oscillations in cytoplasmic free Ca2+ concentration in the pancreatic beta cell. Proc. Natl. Acad. Sci. 1996, 93, 5161–5165. [Google Scholar] [CrossRef]
- Dryselius, S.; Lund, P.; Gylfe, E.; Hellman, B. Variations in ATP-Sensitive K+ Channel Activity Provide Evidence for Inherent Metabolic Oscillations in Pancreatic β-Cells. Biochem. Biophys. Res. Commun. 1994, 205, 880–885. [Google Scholar] [CrossRef]
- Rorsman, P.; Eliasson, L.; Kanno, T.; Zhang, Q.; Gopel, S. Electrophysiology of pancreatic β-cells in intact mouse islets of Langerhans. Prog. Biophys. Mol. Biol. 2011, 107, 224–235. [Google Scholar] [CrossRef]
- Tamarit-Rodriguez, J. A Hypothetical Energy-Dissipating Mechanism Regulated by Glucose in β-Cells Preceding Sustained Insulin Secretion. Cells 2025, 14, 1644. [Google Scholar] [CrossRef] [PubMed]
- Gembal, M.; Detimary, P.; Gilon, P.; Gao, Z.Y.; Henquin, J.C. Mechanisms by which glucose can control insulin release independently from its action on adenosine triphosphate-sensitive K+ channels in mouse B cells. J. Clin. Investig. 1993, 91, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Detimary, P.; Gilon, P.; Henquin, J.-C. Interplay between cytoplasmic Ca2+ and the ATP/ADP ratio: a feedback control mechanism in mouse pancreatic islets. Biochem. J. 1998, 333, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Pizarro-Delgado, J.; Deeney, J.T.; Martín-Del-Río, R.; Corkey, B.E.; Tamarit-Rodriguez, J. KCl -Permeabilized Pancreatic Islets: An Experimental Model to Explore the Messenger Role of ATP in the Mechanism of Insulin Secretion. PLOS ONE 2015, 10, e0140096. [Google Scholar] [CrossRef] [PubMed]
- Kjems, L.L.; Ravier, M.A.; Jonas, J.-C.; Henquin, J.-C. Do Oscillations of Insulin Secretion Occur in the Absence of Cytoplasmic Ca2+ Oscillations in β-Cells? Diabetes 2002, 51, S177–S182. [Google Scholar] [CrossRef]
- Merrins, M.J.; Fendler, B.; Zhang, M.; Sherman, A.; Bertram, R.; Satin, L.S. Metabolic Oscillations in Pancreatic Islets Depend on the Intracellular Ca2+ Level but Not Ca2+ Oscillations. Biophys. J. 2010, 99, 76–84. [Google Scholar] [CrossRef]
- Merrins, M.J.; Corkey, B.E.; Kibbey, R.G.; Prentki, M. Metabolic cycles and signals for insulin secretion. Cell Metab. 2022, 34, 947–968. [Google Scholar] [CrossRef]
- Lewandowski, S.L.; Cardone, R.L.; Foster, H.R.; Ho, T.; Potapenko, E.; Poudel, C.; VanDeusen, H.R.; Sdao, S.M.; Alves, T.C.; Zhao, X.; et al. Pyruvate Kinase Controls Signal Strength in the Insulin Secretory Pathway. Cell Metab. 2020, 32, 736–750.e5. [Google Scholar] [CrossRef]
- Corradi, J.; Thompson, B.; Fletcher, P.A.; Bertram, R.; Sherman, A.S.; Satin, L.S. KATP channel activity and slow oscillations in pancreatic beta cells are regulated by mitochondrial ATP production. J. Physiol. 2023, 601, 5655–5667. [Google Scholar] [CrossRef]
- Hellman, B.; Sehlin, J.; Idahl, L.A.; Täljedal, I.B. Influence of anoxia on glucose metabolism in pancreatic islets: Lack of correlation between fructose-1,6-diphosphate and apparent glycolytic flux. Diabetologia 1975, 11, 495–500. [Google Scholar] [CrossRef]
- Jensen, M.V.; Joseph, J.W.; Ilkayeva, O.; Burgess, S.; Lu, D.; Ronnebaum, S.M.; Odegaard, M.; Becker, T.C.; Sherry, A.D.; Newgard, C.B. Compensatory Responses to Pyruvate Carboxylase Suppression in Islet β-Cells. Preservation of glucose-stimulated insulin secretion. J. Biol. Chem. 2006, 281, 22342–22351. [Google Scholar] [CrossRef]
- Pizarro-Delgado, J.; Fasciani, I.; Temperan, A.; Romero, M.; González-Nieto, D.; Alonso-Magdalena, P.; Nualart-Marti, A.; Estil´les, E.; Paul, D.L.; Martín-del-Río, R. Inhibition of connexin36 hemichannels by glucose contributes to the stimulation of insulin secretion. Am. J. Physiol. Endocrinol. Metab. 2014, 306, E1354–E1366. [Google Scholar] [CrossRef] [PubMed]
- Haythorne, E.; Rohm, M.; van de Bunt, M.; Brereton, M.F.; Tarasov, A.I.; Blacker, T.S.; Sachse, G.; dos Santos, M.S.; Exposito, R.T.; Davis, S.; et al. Diabetes causes marked inhibition of mitochondrial metabolism in pancreatic β-cells. Nat. Commun. 2019, 10, 2474. [Google Scholar] [CrossRef]
- Chareyron, I.; Christen, S.; Moco, S.; Valsesia, A.; Lassueur, S.; Dayon, L.; Wollheim, C.B.; Domingo, J.S.; Wiederkehr, A. Augmented mitochondrial energy metabolism is an early response to chronic glucose stress in human pancreatic beta cells. Diabetologia 2020, 63, 2628–2640. [Google Scholar] [CrossRef]
- Jakovljevic, N.K.; Lukic, L.; Pavlovic, K.; Jotic, A.; Lalic, K.; Stoiljkovic, M.; Milicic, T.; Macesic, M.; Stanarcic, J.; Gajovic, J.S.; Lalic, N.M. Targeting Mitochondria in Diabetes. Int. J. Mol. Sci. 2021, 22, 6642. [Google Scholar] [CrossRef] [PubMed]
- Tamarit-Rodriguez, J. Metabolic Role of GABA in the Secretory Function of Pancreatic β-Cells: Its Hypothetical Implication in β-Cell Degradation in Type 2 Diabetes. Metabolites 2023, 13, 697. [Google Scholar] [CrossRef] [PubMed]
- Kamat, V.; Radtke, J.R.; Hu, Q.; Wang, W.; Sweet, I.R.; Hampe, C.S. Autoantibodies directed against glutamate decarboxylase interfere with glucose-stimulated insulin secretion in dispersed rat islets. Int. J. Exp. Pathol. 2022, 103, 140–148. [Google Scholar] [CrossRef]
- Hampe, C.S.; Kamat, V.; Bryan, C.L.; Pyle, L.; Morton, G.J.; Sweet, I.R. Deleterious Effects of a GAD65 Monoclonal Autoantibody on Islet Function. Diabetes 2025, 74, 2375–2389. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



