Submitted:
10 March 2026
Posted:
11 March 2026
You are already at the latest version
Abstract
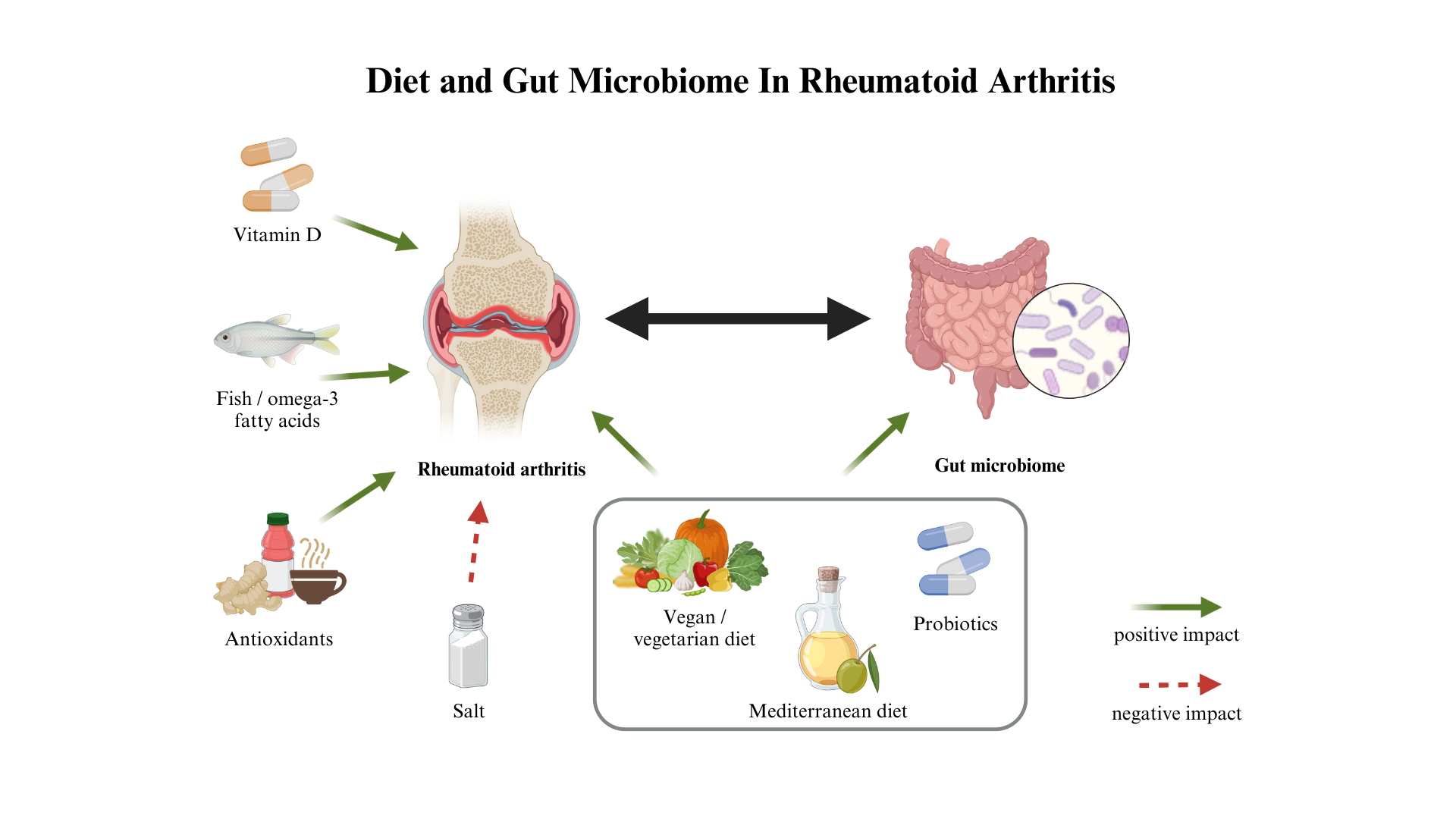
Keywords:
1. Introduction
2. Dietary Regimens and Supplements in Prophylaxis and Treatment of Rheumatoid Arthritis
2.1. Vitamin D
2.2. Mediterranean Diet
2.3. Fish and Omega-3 Fatty Acids
2.4. Vegan Diet, Gluten-Free Diet and Fasting
2.5. Antioxidants
2.6. Salt
3. Gut Microbiome in Rheumatoid Arthritis
3.1. Segmented Filamentous Bacteria
3.2. Prevotella
3.3. Subdoligranulum
3.4. Fusobacterium nucleatum
4. Probiotics in Rheumatoid Arthritis
5. Prebiotics and Synbiotics in Rheumatoid Arthritis
6. Diet and Gut Microbiome in Rheumatoid Arthritis
6.1. Mediterranean Diet
6.2. Vegan and Vegetarian Diets
6.3. Gluten-Free Diet
6.4. Dietary Timing
7. Conclusions
Abbreviations.
| AA ACPA ACR AID CAT CCP CDAI CRP DAS28 DHA DMARD EMS EPA ESR GPx HAQ HAQ-DI hs-CRP IFNα IL-1 IL-1β IL-6 IL-9 IL-12 IL-17 IL-23 LF-HC MD MDA mHAQ NSAID |
arachidonic acid anti-citrullinated protein autoantibody American College of Rheumatology response criteria anti-inflammatory diet catalase cyclic citrullinated peptide Clinical Disease Activity Indice C-reactive protein 28 joint count Disease Activity Score docosahexaenoic acid disease-modifying anti-rheumatic drugs early morning stiffness eicosapentaenoic acid erythrocyte sedimentation rate glutathione peroxidase Health Assessment Questionnaire Health Assessment Questionnaire Disability Index high-sensitivity C-reactive protein interferon α interleukin 1 interleukin 1β interleukin 6 interleukin 9 interleukin 12 interleukin 17 interleukin 23 low-fat high-carbohydrate Mediterranean diet malonedialdehyde modified Health Assessment Questionnaire nonsteroidal anti-inflammatory drugs |
| PCBs RA RAI RAI1 RF SDAI SF-36 SOD |
polychlorinated biphenyls rheumatoid arthritis Ritchie’s articular index relative activity index rheumatoid factor Simplified Disease Activity Indice short form-36 health survey superoxide dismutase |
| TGFβ1 Th17 t.i.d. TNF-α Treg |
transforming growth factor β1 T helper 17 three times a day tumor necrosis factor α regulatory T cells |
| VAS vdHS WBC |
visual analog scale van der Heijde-Sharp white blood cells |
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Scott, DL; Wolfe, F; Huizinga, TWJ. Rheumatoid arthritis. Lancet 2010, 376, 1094–108. [Google Scholar] [CrossRef]
- Van Der Woude, D; Houwing-Duistermaat, JJ; Toes, REM; et al. Quantitative heritability of anti-citrullinated protein antibody-positive and anti-citrullinated protein antibody-negative rheumatoid arthritis. Arthritis Rheum. 2009, 60, 916–923. [Google Scholar] [CrossRef]
- Sokka, T; Hakkinen, A; Kautiainen, H; et al. Physical inactivity in patients with rheumatoid arthritis: data from twenty-one countries in a cross-sectional, international study. Arthritis Rheum. 2008, 59, 42–50. [Google Scholar] [CrossRef]
- Ajeganova, S; Andersson, ML; Hafstrom, I. Association of obesity with worse disease severity in rheumatoid arthritis as well as with comorbidities: a long-term followup from disease onset. Arthritis Care Res. (Hoboken) 2013, 65, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Kupczyk, D; Bilski, R; Szeleszczuk, Ł; et al. The Role of Diet in Modulating Inflammation and Oxidative Stress in Rheumatoid Arthritis, Ankylosing Spondylitis, and Psoriatic Arthritis. Nutrients 2025, 17(9), 1603. [Google Scholar] [CrossRef]
- Arnson, Y; Amital, H; Shoenfeld, Y. Vitamin D and autoimmunity: new aetiological and therapeutic considerations. Ann. Rheum. Dis. 2007, 66, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- Song, GG; Bae, S-C; Lee, YH. Association between vitamin D intake and the risk of rheumatoid arthritis: a meta-analysis. Clin. Rheumatol. 2012, 31, 1733–1739. [Google Scholar] [CrossRef] [PubMed]
- Lee, YH; Bae, S-C. Vitamin D level in rheumatoid arthritis and its correlation with the disease activity: a meta-analysis. Clin. Exp. Rheumatol. 2016, 34, 827–833. [Google Scholar]
- Lin, J; Liu, J; Davies, ML; et al. Serum Vitamin D Level and Rheumatoid Arthritis Disease Activity: Review and Meta-Analysis. PLoS One 2016, 11(1), e0146351. [Google Scholar] [CrossRef]
- Craig, SM; Yu, F; Curtis, JR; et al. Vitamin D status and its associations with disease activity and severity in African Americans with recent-onset rheumatoid arthritis. J. Rheumatol. 2010, 37, 275–281. [Google Scholar] [CrossRef]
- Baker, JF; Baker, DG; Toedter, G; et al. Associations between vitamin D, disease activity, and clinical response to therapy in rheumatoid arthritis. Clin. Exp. Rheumatol. 2012, 30, 658–664. [Google Scholar]
- Matsumoto, Y; Sugioka, Y; Tada, M; et al. Relationships between serum 25-hydroxycalciferol, vitamin D intake and disease activity in patients with rheumatoid arthritis--TOMORROW study. Mod. Rheumatol. 2015, 25, 246–250. [Google Scholar] [CrossRef]
- Polasik, K; Piotrowska, E; Lipinska, B; et al. Vitamin D status in patients with rheumatoid arthritis: a correlation analysis with disease activity and progression, as well as serum IL-6 levels. Acta Biochim. Pol. 2017, 64, 667–670. [Google Scholar] [CrossRef] [PubMed]
- Jelsness-Jørgensen, LP; Grøvle, L; Haugen, AJ. Association between Vitamin D and fatigue in patients with rheumatoid arthritis: A cross-sectional study. BMJ Open 2020, 10, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekara, S; Patted, A. Role of vitamin D supplementation in improving disease activity in rheumatoid arthritis: An exploratory study. Int. J. Rheum. Dis. 2017, 20, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Buondonno, I; Rovera, G; Sassi, F; et al. Vitamin D and immunomodulation in early rheumatoid arthritis: A randomized doubleblind placebo-controlled study. PLoS One 2017, 12, 1–18. [Google Scholar] [CrossRef]
- El-Banna, HS; Gado, SE. Vitamin D: does it help Tregs in active rheumatoid arthritis patients. Expert Rev. Clin. Immunol. 2020, 16, 847–853. [Google Scholar] [CrossRef]
- Sofi, F; Macchi, C; Abbate, R; et al. Mediterranean diet and health. Biofactors 2013, 39, 335–342. [Google Scholar] [CrossRef]
- Sköldstam, L; Hagfors, L; Johansson, G. An experimental study of a Mediterranean diet intervention for patients with rheumatoid arthritis. Ann. Rheum. Dis. 2003, 62, 208–214. [Google Scholar] [CrossRef]
- McKellar, G; Morrison, E; McEntegart, A; et al. A pilot study of a Mediterranean-type diet intervention in female patients with rheumatoid arthritis living in areas of social deprivation in Glasgow. Ann. Rheum. Dis. 2007, 66, 1239–1243. [Google Scholar] [CrossRef] [PubMed]
- García-Morales, JM; Lozada-Mellado, M; Hinojosa-Azaola, A; et al. Effect of a Dynamic Exercise Program in Combination With Mediterranean Diet on Quality of Life in Women With Rheumatoid Arthritis. J. Clin. Rheumatol. 2020, 26, S116–S122. [Google Scholar] [CrossRef]
- Papandreou, P; Gioxari, A; Daskalou, E; et al. Mediterranean Diet and Physical Activity Nudges versus Usual Care in Women with Rheumatoid Arthritis: Results from the MADEIRA Randomized Controlled Trial. Nutrients 2023, 15(3), 676. [Google Scholar] [CrossRef]
- Raad, T; George, E; Griffin, A; et al. Effects of a telehealth-delivered Mediterranean diet intervention in adults with Rheumatoid Arthritis (MEDRA): a randomised controlled trial. BMC Musculoskelet. Disord. 2024, 25(1), 631. [Google Scholar] [CrossRef]
- Sadeghi, A; Tabatabaiee, M; Mousavi, MA; et al. Dietary Pattern or Weight Loss: Which One Is More Important to Reduce Disease Activity Score in Patients with Rheumatoid Arthritis? A Randomized Feeding Trial. Int. J. Clin. Pract. 2022, 6004916. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y; Costenbader, KH; Gao, X; et al. Mediterranean diet and incidence of rheumatoid arthritis in women. Arthritis Care Res. (Hoboken) 2015, 67, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Sundstrom, B; Johansson, I; Rantapaa-Dahlqvist, S. Diet and alcohol as risk factors for rheumatoid arthritis: a nested case-control study. Rheumatol. Int. 2015, 35, 533–539. [Google Scholar] [CrossRef]
- Nguyen, Y; Salliot, C; Gelot, A; et al. Mediterranean Diet and Risk of Rheumatoid Arthritis: Findings From the French E3N-EPIC Cohort Study. Arthritis and Rheumatology 2021, 73, 69–77. [Google Scholar] [CrossRef]
- Matsumoto, Y; Sugioka, Y; Tada, M; et al. Monounsaturated fatty acids might be key factors in the Mediterranean diet that suppress rheumatoid arthritis disease activity: The TOMORROW study. Clin. Nutr. 2018, 37, 675–680. [Google Scholar] [CrossRef]
- Barbalace, MC; Talotta, R; Rapisarda, F; et al. Unlocking the Power of the Mediterranean Diet: Two in One—Dual Benefits for Rheumatic and Thyroid Autoimmune Diseases. Nutrients 2025, 17(8), 1383. [Google Scholar] [CrossRef]
- Papanikolaou, Y; Brooks, J; Reider, C; et al. U.S. adults are not meeting recommended levels for fish and omega-3 fatty acid intake: results of an analysis using observational data from NHANES 2003-2008. Nutr. J. 2014, 13, 31. [Google Scholar] [CrossRef] [PubMed]
- Burdge, GC; Calder, PC. Conversion of alpha-linolenic acid to longer-chain polyunsaturated fatty acids in human adults. Reprod. Nutr. Dev. 2005, 45, 581–597. [Google Scholar] [CrossRef]
- Adam, O; Beringer, C; Kless, T; et al. Anti-inflammatory effects of a low arachidonic acid diet and fish oil in patients with rheumatoid arthritis. Rheumatol. Int. 2003, 23, 27–36. [Google Scholar] [CrossRef]
- Nielsen, GL; Faarvang, KL; Thomsen, BS; et al. The effects of dietary supplementation with n-3 polyunsaturated fatty acids in patients with rheumatoid arthritis: a randomized, double blind trial. Eur. J. Clin. Invest. 1992, 22, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Skoldstam, L; Borjesson, O; Kjallman, A; et al. Effect of six months of fish oil supplementation in stable rheumatoid arthritis. A double-blind, controlled study. Scand. J. Rheumatol. 1992, 21, 178–185. [Google Scholar] [CrossRef]
- Lau, CS; Morley, KD; Belch, JJ. Effects of fish oil supplementation on non-steroidal anti-inflammatory drug requirement in patients with mild rheumatoid arthritis--a double-blind placebo controlled study. Br. J. Rheumatol. 1993, 32, 982–989. [Google Scholar] [CrossRef] [PubMed]
- Proudman, SM; James, MJ; Spargo, LD; et al. Fish oil in recent onset rheumatoid arthritis: a randomised, double-blind controlled trial within algorithm-based drug use. Ann. Rheum. Dis. 2015, 74, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, RJ; Katz, J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain 2007, 129, 210–223. [Google Scholar] [CrossRef]
- Lee, Y-H; Bae, S-C; Song, G-G. Omega-3 polyunsaturated fatty acids and the treatment of rheumatoid arthritis: a meta-analysis. Arch. Med. Res. 2012, 43, 356–362. [Google Scholar] [CrossRef]
- Gioxari, A; Kaliora, AC; Marantidou, F; et al. Intake of omega-3 polyunsaturated fatty acids in patients with rheumatoid arthritis: A systematic review and meta-analysis. Nutrition 2018, 45, 114–124.e4. [Google Scholar] [CrossRef]
- Ghaseminasab-Parizi, M; Nazarinia, MA; Akhlaghi, M. The effect of flaxseed with or without anti-inflammatory diet in patients with rheumatoid arthritis, a randomized controlled trial. Eur. J. Nutr. 2022, 61, 1377–1389. [Google Scholar] [CrossRef]
- Tedeschi, SK; Bathon, JM; Giles, JT; et al. Relationship Between Fish Consumption and Disease Activity in Rheumatoid Arthritis. Arthritis Care Res. (Hoboken) 2018, 70, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Hulander, E; Bärebring, L; Winkvist, A; et al. A randomized controlled cross-over trial investigating the acute inflammatory and metabolic response after meals based on red meat, fatty fish, or soy protein: the postprandial inflammation in rheumatoid arthritis (PIRA) trial. Eur. J. Nutr. 2024, 63, 2631–2642. [Google Scholar] [CrossRef]
- Lindqvist, HM; Hulander, E; Bärebring, L; et al. A randomized controlled cross-over trial of differences in acute effects on serum metabolites from isocaloric meals based on red meat, fatty fish, or soy protein. Eur. J. Nutr. 2025, 64(5), 187. [Google Scholar] [CrossRef] [PubMed]
- Di Giuseppe, D; Crippa, A; Orsini, N; et al. Fish consumption and risk of rheumatoid arthritis: a dose-response meta-analysis. Arthritis Res. Ther. 2014, 16, 446. [Google Scholar] [CrossRef]
- Lee, D-H; Steffes, M; Jacobs, DR. Positive associations of serum concentration of polychlorinated biphenyls or organochlorine pesticides with self-reported arthritis, especially rheumatoid type, in women. Environ. Health Perspect. 2007, 115, 883–888. [Google Scholar] [CrossRef]
- Di Giuseppe, D; Wallin, A; Bottai, M; et al. Long-term intake of dietary long-chain n-3 polyunsaturated fatty acids and risk of rheumatoid arthritis: a prospective cohort study of women. Ann. Rheum. Dis. 2014, 73, 1949–1953. [Google Scholar] [CrossRef]
- Shapiro, JA; Koepsell, TD; Voigt, LF; et al. Diet and rheumatoid arthritis in women: a possible protective effect of fish consumption. Epidemiology 1996, 7, 256–263. [Google Scholar] [CrossRef]
- Sparks, JA; O’Reilly, ÉJ; Barbhaiya, M; et al. Association of fish intake and smoking with risk of rheumatoid arthritis and age of onset: a prospective cohort study. BMC Musculoskelet. Disord. 2019, 20, 2. [Google Scholar] [CrossRef]
- McDougall, J; Bruce, B; Spiller, G; et al. Effects of a very low-fat, vegan diet in subjects with rheumatoid arthritis. J. Altern. Complement. Med. 2002, 8, 71–75. [Google Scholar] [CrossRef]
- Walrabenstein, W; Wagenaar, CA; Van Der Leeden, M; et al. A multidisciplinary lifestyle program for rheumatoid arthritis: The ‘Plants for Joints’ randomized controlled trial. Rheumatology 2023, 62, 2683–2691. [Google Scholar] [CrossRef] [PubMed]
- Hanninen; Kaartinen, K; Rauma, AL; et al. Antioxidants in vegan diet and rheumatic disorders. Toxicology 2000, 155, 45–53. [Google Scholar] [CrossRef]
- Rauma, AL; Nenonen, M; Helve, T; et al. Effect of a strict vegan diet on energy and nutrient intakes by Finnish rheumatoid patients. Eur. J. Clin. Nutr. 1993, 47, 747–749. [Google Scholar]
- Hafstrom, I; Ringertz, B; Spangberg, A; et al. A vegan diet free of gluten improves the signs and symptoms of rheumatoid arthritis: the effects on arthritis correlate with a reduction in antibodies to food antigens. Rheumatology (Oxford) 2001, 40, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Elkan, A-C; Sjoberg, B; Kolsrud, B; et al. Gluten-free vegan diet induces decreased LDL and oxidized LDL levels and raised atheroprotective natural antibodies against phosphorylcholine in patients with rheumatoid arthritis: a randomized study. Arthritis Res. Ther. 2008, 10, R34. [Google Scholar] [CrossRef]
- Gregersen, L; Jessen, PD; Lund, HW; et al. Impact of gluten intake on clinical outcomes in patients with chronic inflammatory diseases initiating biologics: Secondary analysis of the prospective multicentre BELIEVE cohort study. Scand. J. Immunol. 2024, 100(6), e13409. [Google Scholar] [CrossRef]
- Khanna, S; Jaiswal, KS; Gupta, B. Managing Rheumatoid Arthritis with Dietary Interventions. Front. Nutr. 2017, 4, 52. [Google Scholar] [CrossRef]
- Kjeldsen-Kragh, J; Haugen, M; Borchgrevink, CF; et al. Controlled trial of fasting and one-year vegetarian diet in rheumatoid arthritis. Lancet 1991, 338, 899–902. [Google Scholar] [CrossRef]
- Fraser, DA; Thoen, J; Djoseland, O; et al. Serum levels of interleukin-6 and dehydroepiandrosterone sulphate in response to either fasting or a ketogenic diet in rheumatoid arthritis patients. Clin. Exp. Rheumatol. 2000, 18, 357–362. [Google Scholar] [PubMed]
- Hartmann, AM; D’Urso, M; Dell’Oro, M; et al. Post Hoc Analysis of a Randomized Controlled Trial on Fasting and Plant-Based Diet in Rheumatoid Arthritis (NutriFast): Nutritional Supply and Impact on Dietary Behavior. Nutrients 2023, 15(4), 851. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, A; Akhgarjand, C; Ansar, H; et al. The effects of intermittent fasting on antioxidant and inflammatory markers and liver enzymes in postmenopausal, overweight and obese women with rheumatoid arthritis: a randomized controlled trial. Sci. Rep. 2025, 15(1), 2357. [Google Scholar] [CrossRef]
- Ranjbar, M; Shab-Bidar, S; Rostamian, A; et al. Effects of intermittent fasting diet in overweight and obese postmenopausal women with rheumatoid arthritis: A randomized controlled clinical trial. Complement. Ther. Med. 2025, 91, 103189. [Google Scholar] [CrossRef]
- Peretz, A; Siderova, V; Neve, J. Selenium supplementation in rheumatoid arthritis investigated in a double blind, placebo-controlled trial. Scand. J. Rheumatol. 2001, 30, 208–212. [Google Scholar] [CrossRef]
- Meki, A-RMA; Hamed, EA; Ezam, KA. Effect of green tea extract and vitamin C on oxidant or antioxidant status of rheumatoid arthritis rat model. Indian J. Clin. Biochem. 2009, 24, 280–287. [Google Scholar] [CrossRef]
- Karlson, EW; Shadick, NA; Cook, NR; et al. Vitamin E in the primary prevention of rheumatoid arthritis: the Women’s Health Study. Arthritis Rheum. 2008, 59, 1589–1595. [Google Scholar] [CrossRef] [PubMed]
- Helmy, M; Shohayeb, M; Helmy, MH; et al. Antioxidants as adjuvant therapy in rheumatoid disease. A preliminary study. Arzneimittelforschung 2001, 51, 293–298. [Google Scholar] [CrossRef] [PubMed]
- van Vugt, RM; Rijken, PJ; Rietveld, AG; et al. Antioxidant intervention in rheumatoid arthritis: results of an open pilot study. Clin. Rheumatol. 2008, 27, 771–775. [Google Scholar] [CrossRef] [PubMed]
- Jalili, M; Kolahi, S; Aref-Hosseini, S-R; et al. Beneficial role of antioxidants on clinical outcomes and erythrocyte antioxidant parameters in rheumatoid arthritis patients. Int. J. Prev. Med. 2014, 5, 835–840. [Google Scholar]
- Thimoteo, NSB; Iryioda, TMV; Alfieri, DF; et al. Cranberry juice decreases disease activity in women with rheumatoid arthritis. Nutrition 2019, 60, 112–117. [Google Scholar] [CrossRef]
- Fatel, ECS; Rosa, FT; Alfieri, DF; et al. Beneficial effects of fish oil and cranberry juice on disease activity and inflammatory biomarkers in people with rheumatoid arthritis. Nutrition 2021, 86, 111183. [Google Scholar] [CrossRef]
- Chandran, B; Goel, A. A randomized, pilot study to assess the efficacy and safety of curcumin in patients with active rheumatoid arthritis. Phytother. Res. 2012, 26, 1719–1725. [Google Scholar] [CrossRef]
- Amalraj, A; Varma, K; Jacob, J; et al. A Novel Highly Bioavailable Curcumin Formulation Improves Symptoms and Diagnostic Indicators in Rheumatoid Arthritis Patients: A Randomized, Double-Blind, Placebo-Controlled, Two-Dose, Three-Arm, and Parallel-Group Study. J. Med. Food 2017, 20, 1022–1030. [Google Scholar] [CrossRef]
- Westerlind, H; Palmqvist, I; Saevarsdottir, S; et al. Is tea consumption associated with reduction of risk of rheumatoid arthritis? A Swedish case-control study. Arthritis Res. Ther. 2021, 23, 1–8. [Google Scholar] [CrossRef]
- Vadell, AKE; Bärebring, L; Hulander, E; et al. Anti-inflammatory Diet in Rheumatoid Arthritis (ADIRA) - A randomized, controlled crossover trial indicating effects on disease activity. American Journal of Clinical Nutrition 2020, 111, 1203–1213. [Google Scholar] [CrossRef]
- Matsumoto, Y; Shivappa, N; Sugioka, Y; et al. Change in dietary inflammatory index score is associated with control of long-term rheumatoid arthritis disease activity in a Japanese cohort: the TOMORROW study. Arthritis Res. Ther. 2021, 23, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kleinewietfeld, M; Manzel, A; Titze, J; et al. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 2013, 496, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Wu, C; Yosef, N; Thalhamer, T; et al. Induction of pathogenic TH17 cells by inducible salt-sensing kinase SGK1. Nature 2013, 496, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Sundstrom, B; Johansson, I; Rantapaa-Dahlqvist, S. Interaction between dietary sodium and smoking increases the risk for rheumatoid arthritis: results from a nested case-control study. Rheumatology (Oxford) 2015, 54, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Salgado, E; Bes-Rastrollo, M; de Irala, J; et al. High Sodium Intake Is Associated With Self-Reported Rheumatoid Arthritis: A Cross Sectional and Case Control Analysis Within the SUN Cohort. Medicine 2015, 94, e924. [Google Scholar] [CrossRef] [PubMed]
- Marouen, S; du Cailar, G; Audo, R; et al. Sodium excretion is higher in patients with rheumatoid arthritis than in matched controls. PLoS One 2017, 12, e0186157. [Google Scholar] [CrossRef]
- Scrivo, R; Massaro, L; Barbati, C; et al. The role of dietary sodium intake on the modulation of T helper 17 cells and regulatory T cells in patients with rheumatoid arthritis and systemic lupus erythematosus. PLoS One 2017, 12, e0184449. [Google Scholar] [CrossRef]
- Mortier, C; Govindarajan, S; Venken, K; et al. It Takes ‘Guts’ to Cause Joint Inflammation: Role of Innate-Like T Cells. Front. Immunol. 2018, 9, 1489. [Google Scholar] [CrossRef]
- Guerreiro, CS; Calado, A; Sousa, J; et al. Diet, Microbiota, and Gut Permeability-The Unknown Triad in Rheumatoid Arthritis. Front. Med. (Lausanne) 2018, 5, 349. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M; Raes, J; Pelletier, E; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef]
- Wu, GD; Chen, J; Hoffmann, C; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef]
- Gorvitovskaia, A; Holmes, SP; Huse, SM. Interpreting Prevotella and Bacteroides as biomarkers of diet and lifestyle. Microbiome 2016, 4, 15. [Google Scholar] [CrossRef]
- Zhang, X; Zhang, D; Jia, H; et al. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 2015, 21, 895–905. [Google Scholar] [CrossRef]
- Vaahtovuo, J; Munukka, E; Korkeamaki, M; et al. Fecal microbiota in early rheumatoid arthritis. J. Rheumatol. 2008, 35, 1500–1505. [Google Scholar] [PubMed]
- Chen, J; Wright, K; Davis, JM; et al. An expansion of rare lineage intestinal microbes characterizes rheumatoid arthritis. Genome Med. 2016, 8, 43. [Google Scholar] [CrossRef]
- Ruiz-Limón, P; Mena-Vázquez, N; Moreno-Indias, I; et al. Collinsella is associated with cumulative inflammatory burden in an established rheumatoid arthritis cohort. Biomedicine and Pharmacotherapy 2022, 153, 113518. [Google Scholar] [CrossRef] [PubMed]
- Li, Y; Zhang, SX; Yin, XF; et al. The Gut Microbiota and Its Relevance to Peripheral Lymphocyte Subpopulations and Cytokines in Patients with Rheumatoid Arthritis. J. Immunol. Res. 2021. [Google Scholar] [CrossRef]
- Kitamura, K; Shionoya, H; Suzuki, S; et al. Oral and Intestinal Bacterial Substances Associated with Disease Activities in Patients with Rheumatoid Arthritis: A Cross-Sectional Clinical Study. J. Immunol. Res. 2022. [Google Scholar] [CrossRef]
- Koh, JH; Lee, EH; Cha, KH; et al. Factors associated with the composition of the gut microbiome in patients with established rheumatoid arthritis and its value for predicting treatment responses. Arthritis Res. Ther. 2023, 25(1), 32. [Google Scholar] [CrossRef]
- Danckert, NP; Freidin, MB; Granville Smith, I; et al. Treatment response in rheumatoid arthritis is predicted by the microbiome: a large observational study in UK DMARD-naive patients. Rheumatology 2024, 63, 3486–3495. [Google Scholar] [CrossRef]
- Wang, Q; Li, CL; Yu, SY; et al. A predictive model based on the gut microbiota improves the diagnostic effect in patients with rheumatoid arthritis. Rheumatology 2025, 64, 4014–4021. [Google Scholar] [CrossRef]
- Zeng, L; Yang, Q; Luo, Y; et al. The Gut Microbiota: Emerging Evidence in Autoimmune and Inflammatory Diseases. Research 2025, 9, 1097. [Google Scholar] [CrossRef] [PubMed]
- Yu, D; Du, J; Pu, X; et al. The Gut Microbiome and Metabolites Are Altered and Interrelated in Patients With Rheumatoid Arthritis. Front. Cell. Infect. Microbiol. 2022, 11, 763507. [Google Scholar] [CrossRef]
- Klaasen, HL; Koopman, JP; Van den Brink, ME; et al. Intestinal, segmented, filamentous bacteria in a wide range of vertebrate species. Lab. Anim. 1993, 27, 141–150. [Google Scholar] [CrossRef]
- Ivanov, II; Atarashi, K; Manel, N; et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 2009, 139, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Wu, H-J; Ivanov, II; Darce, J; et al. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 2010, 32, 815–827. [Google Scholar] [CrossRef] [PubMed]
- Teng, F; Klinger, CN; Felix, KM; et al. Gut Microbiota Drive Autoimmune Arthritis by Promoting Differentiation and Migration of Peyer’s Patch T Follicular Helper Cells. Immunity 2016, 44, 875–888. [Google Scholar] [CrossRef]
- Larsen, JM. The immune response to Prevotella bacteria in chronic inflammatory disease. Immunology 2017, 151, 363–374. [Google Scholar] [CrossRef]
- Scher, JU; Sczesnak, A; Longman, RS; et al. Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. Elife 2013, 2, e01202. [Google Scholar] [CrossRef]
- Alpizar-Rodriguez, D; Lesker, TR; Gronow, A; et al. Prevotella copri in individuals at risk for rheumatoid arthritis. Ann. Rheum. Dis. 2019, 78, 590–593. [Google Scholar] [CrossRef]
- Pianta, A; Arvikar, S; Strle, K; et al. Evidence of the Immune Relevance of Prevotella copri, a Gut Microbe, in Patients With Rheumatoid Arthritis. Arthritis Rheumatol. 2017, 69, 964–975. [Google Scholar] [CrossRef]
- Marietta, E V; Murray, JA; Luckey, DH; et al. Suppression of Inflammatory Arthritis by Human Gut-Derived Prevotella histicola in Humanized Mice. Arthritis Rheumatol. 2016, 68, 2878–2888. [Google Scholar] [CrossRef]
- Chriswell, ME; Lefferts, AR; Clay, MR; et al. Clonal IgA and IgG autoantibodies from individuals at risk for rheumatoid arthritis identify an arthritogenic strain of Subdoligranulum. Sci. Transl. Med. 2022, 14(668), eabn5166. [Google Scholar] [CrossRef] [PubMed]
- Li, H; Dai, J; Zhao, C; et al. Gut Subdoligranulum variabile ameliorates rheumatoid arthritis by promoting TSG-6 synthesis from joint cells. Front. Immunol. 2024, 15, 1418717. [Google Scholar] [CrossRef] [PubMed]
- Hong, M; Li, Z; Liu, H; et al. Fusobacterium nucleatum aggravates rheumatoid arthritis through FadA-containing outer membrane vesicles. Cell Host Microbe 2023, 31, 798–810.e7. [Google Scholar] [CrossRef] [PubMed]
- Hatakka, K; Martio, J; Korpela, M; et al. Effects of probiotic therapy on the activity and activation of mild rheumatoid arthritis--a pilot study. Scand. J. Rheumatol. 2003, 32, 211–215. [Google Scholar] [CrossRef]
- Alipour, B; Homayouni-Rad, A; Vaghef-Mehrabany, E; et al. Effects of Lactobacillus casei supplementation on disease activity and inflammatory cytokines in rheumatoid arthritis patients: a randomized double-blind clinical trial. Int. J. Rheum. Dis. 2014, 17, 519–527. [Google Scholar] [CrossRef]
- Vaghef-Mehrabany, E; Alipour, B; Homayouni-Rad, A; et al. Probiotic supplementation improves inflammatory status in patients with rheumatoid arthritis. Nutrition 2014, 30, 430–435. [Google Scholar] [CrossRef]
- Zamani, B; Golkar, HR; Farshbaf, S; et al. Clinical and metabolic response to probiotic supplementation in patients with rheumatoid arthritis: a randomized, double-blind, placebo-controlled trial. Int. J. Rheum. Dis. 2016, 19, 869–879. [Google Scholar] [CrossRef]
- Pineda M de, LA; Thompson, SF; Summers, K; et al. A randomized, double-blinded, placebo-controlled pilot study of probiotics in active rheumatoid arthritis. Med. Sci. Monit. 2011, 17, CR347–54. [Google Scholar] [CrossRef]
- Cannarella, LAT; Mari, NL; Alcântara, CC; et al. Mixture of probiotics reduces inflammatory biomarkers and improves the oxidative/nitrosative profile in people with rheumatoid arthritis. Nutrition 2021, 89, 111282. [Google Scholar] [CrossRef]
- Barac, B; Lukovic, S; Kojic, T; et al. Therapeutical efficacy of immunobiotics in patients with newly diagnosed rheumatoid arthritis. J. Infect. Dev. Ctries. 2025, 19, 1861–1868. [Google Scholar] [CrossRef]
- Zamani, B; Farshbaf, S; Golkar, HR; et al. Synbiotic supplementation and the effects on clinical and metabolic responses in patients with rheumatoid arthritis: A randomised, double-blind, placebo-controlled trial. British Journal of Nutrition 2017, 117, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Tabatabaeyan, A; Grigorian, A; Salesi, M; et al. Inulin supplementation improves some inflammatory indices, clinical outcomes, and quality of life in rheumatoid arthritis patients. Sci. Rep. 2025, 15(1), 30732. [Google Scholar] [CrossRef]
- Mitsou, EK; Kakali, A; Antonopoulou, S; et al. Adherence to the Mediterranean diet is associated with the gut microbiota pattern and gastrointestinal characteristics in an adult population. Br. J. Nutr. 2017, 117, 1645–1655. [Google Scholar] [CrossRef]
- Zimmer, J; Lange, B; Frick, J-S; et al. A vegan or vegetarian diet substantially alters the human colonic faecal microbiota. Eur. J. Clin. Nutr. 2012, 66, 53–60. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, F; Pellegrini, N; Vannini, L; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- Peltonen, R; Kjeldsen-Kragh, J; Haugen, M; et al. Changes of faecal flora in rheumatoid arthritis during fasting and one-year vegetarian diet. Br. J. Rheumatol. 1994, 33, 638–643. [Google Scholar] [CrossRef]
- Peltonen, R; Nenonen, M; Helve, T; et al. Faecal microbial flora and disease activity in rheumatoid arthritis during a vegan diet. Br. J. Rheumatol. 1997, 36, 64–68. [Google Scholar] [CrossRef] [PubMed]
- De Palma, G; Nadal, I; Collado, MC; et al. Effects of a gluten-free diet on gut microbiota and immune function in healthy adult human subjects. Br. J. Nutr. 2009, 102, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Bonder, MJ; Tigchelaar, EF; Cai, X; et al. The influence of a short-term gluten-free diet on the human gut microbiome. Genome Med. 2016, 8, 45. [Google Scholar] [CrossRef]
- Ma, F; Li, Z; Liu, H; et al. Dietary-timing-induced gut microbiota diurnal oscillations modulate inflammatory rhythms in rheumatoid arthritis. Cell Metab. 2024, 36, 2367–2382.e5. [Google Scholar] [CrossRef] [PubMed]
| Type of study | Participants | Evaluated parameters of disease activity | Results | Reference |
|---|---|---|---|---|
| Meta-analysis of 8 studies (6 cross-sectional studies, and 2 case–control studies) |
2,885 RA patients and 1,084 controls |
DAS28 | Seven of the eight studies showed that vitamin D levels are inversely associated with RA activity parameters, such as DAS28. |
[7] |
| Meta-analysis of 14 studies |
1,143 RA patients and 963 controls |
DAS28 | Serum vitamin D level significantly lower in RA patients. Negative correlation between vitamin D level and DAS28. |
[8] |
| Meta-analysis of 24 studies |
3489 patients | DAS28 CRP |
Negative correlation between vitamin D level and DAS28 or CRP level. |
[9] |
| Cross-sectional study |
266 African Americans with recent-onset RA | DAS28 | No statistically significant association between 25(OH)D concentration and DAS28. |
[10] |
| Cross-sectional study |
499 participants with active RA, age 18–85 |
DAS28-ESR, DAS28-CRP, ESR, CRP, HAQ Score, patient assessment on VAS, evaluator assessment on VAS, pain on VAS, van der Heidje erosion score, vdHS score, joint space narrowing, CCP | No correlation between vitamin D level and DAS28, serum inflammatory markers or van der Heijde-Sharp (vdHS) erosion scores. |
[11] |
| Cross-sectional study |
181 patients with RA and 186 controls | DAS28-ESR | No statistically significant association between 25(OH)D concentration and DAS28-ESR |
[12] |
| Cross-sectional study |
35 female RA patients and 38 age- and gender-matched healthy controls |
DAS28-ESR, joint damage based on the Steinbrocker criteria, serum IL-6 levels |
No statistically significant association between 25(OH)D concentration and DAS28 Positive correlation between 25(OH)D and IL-6 among RA patients |
[13] |
| Cross-sectional study | 169 patients with established RA |
DAS28- CRP, psychological distress, pain and sleep |
No statistically significant association between fatigue and serum concentrations of 25(OH)D. Fatigue was significantly associated with RA disease activity, psychological distress and pain, but not with serum 25(OH)D. |
[14] |
| Factor | Type of study | Study group | Results | Reference |
|---|---|---|---|---|
| Vitamin D | Meta-analysis of 3 cohort studies |
215,757 participants and 874 incident cases of RA |
Significant association between vitamin D intake or supplementation and incidence of RA. |
[7] |
| Mediterranean Diet | Prospective observational study |
83,245 participants from the Nurses’ Health Study (NHS; 1980–2008) and 91,393 participants from NHS II (1991–2009); females only |
No significant correlation. | [25] |
| Prospective observational study |
368 individuals in the Västerbotten Intervention Program (Sweden) who had developed RA and 1,886 controls |
No significant correlation. | [26] | |
| Prospective observational study |
62,629 women, 480 incident cases of RA |
No association between Mediterranean diet and risk of RA. However, adherence to Mediterranean diet diminished the risk of RA among ever-smoking women. |
[27] | |
| Fish and omega-3 consumption |
Meta-analysis of 7 studies | 174 701 participants | Non-statistically significant decrease of RA risk for up to two servings of fish per week; 3 and 4 portions per week associated with a slightly increased risk of RA. | [44] |
| Population-based prospective study |
32 232 middle-aged and older women from the Swedish Mammography Cohort; 205 RA cases |
Regular intake of > 0,21 g omega-3 per day was associated with a 52% lower risk of RA. Consistent long-term consumption of fish ≥1 serving per week compared with<1 was associated with a 29% decrease in RA risk. |
[46] | |
| Population-based case-control study |
324 incident RA cases, 1245 controls |
Higher intake of omega-3 estimated from Frequency of fish consumption was associated with reduced risk of RA. |
[47] | |
| Prospective cohort study |
166,013 women in two prospective cohorts, the Nurses’ Health Study (NHS, 1984–2014) And NHSII (1991–2015). |
No protective impact of fish or omega-3 fatty acids consumption on RA risk. |
[48] | |
| PCBs | Prospective observational study |
1,721 adults ≥ 20 years of age in the National Health and Nutrition Examination Survey 1999–2002 | Exposure to PCBs was positively Associated with RA risk in women. |
[45] |
| Tea | Population-based case control study |
2237 incident RA cases and 4661 matched controls | The risk of RA was inversely correlated to high tea consumption in comparison to irregular consumption among smokers and in the ACPA-positive subset of RA |
[72] |
| Salt | Nested case-control study | 386 RA patients and 1886 matched controls | No significant association between sodium intake and RA development |
[77] |
| Cross-sectional nested study | 18,555 individuals, 392 developed RA |
Sodium intake was associated with RA risk and the effect was dose-dependent. |
[78] |
| Factor | Type of study | Dosage | Duration | Participants | Evaluated markers of disease activity | Effect | Reference |
|---|---|---|---|---|---|---|---|
| Vitamin D | Exploratory study | 60000 IU/week for 6 weeks, followed by 60000 IU/month | 12 weeks | 59 RA patients with vitamin D deficiency (below 20 ng/mL) |
DAS28-CRP, DAS28-ESR | Amelioration of DAS28-CRP and DAS28-ESR. |
[15] |
| Randomized double- blind placebo-controlled study |
300,000 IU (a single dose) |
3 months | 39 female patients with early RA: 21 patients assigned to treatment, 18 to placebo |
DAS 28, VAS Pain, HAQ, GH, ESR, CRP |
Treatment with vitamin D improved global health, but had no significant effect on specific signs and symptoms of RA, CRP, VAS pain and DAS28. |
[16] | |
| Randomized, non-blinded interventional study |
50000 IU of vitamin D2 weekly |
3 months | 40 RA patients | DAS-28, ESR, CRP, rheumatoid factor, anti-CCP antibodies | Regulatory T cells (Tregs) elevation and significant DAS-28 score reduction. |
[17] | |
| n-3 fatty acids + low arachidonic acid diet |
Randomized-controlled double-blind crossover study | Anti-inflammatory diet (modified lactovegetarian diet) with AA intake reduced to < 90 mg per day + 30 mg of n-3 fatty acids per kg of body weight per day or placebo (corn oil) |
8 months: 3 months of diet, 2-month washout period, crossover of supplementation, another 3 months of diet |
60 patients with RA; 30 in experimental group (anti-inflammatory diet), 30 in control group (western diet) |
Number of tender and swollen joints, patient’s global assessment, physician’s global assessment, patient’s assessment of pain (VAS) |
Greater reduction in the number of tender and swollen joints in the AID group. |
[32] |
| EPA and DHA/ fish oil supplementation |
Randomized, double-blind trial |
2 g EPA and 1,2 g DHA per day | 12 weeks | 51 patients with RA: 27 In intervention group, 24 in control group | grip strength, joint tenderness, joint swelling, morning stiffness, visual pain score, daily activity score, ESR, CRP | Improvement in: - morning stiffness, joint tenderness and CRP in experimental group, - joint tenderness in both groups. |
[33] |
| Double-blind, controlled study | 10 g of fish oil per day | 6 months | 43 patients with RA: 22 in intervention group, 21 in control group |
NSAID intake, global arthritis activity, pain (VAS), functional capacity (Fries index), grip strength, joint tenderness (Ritchie’s index), physician’s global assessment, ESR, CRP | Decreased intake of NSAID at 3 and 6 months; improvement of global arthritic activity at 3 months. |
[34] | |
| Double-blind placebo controlled study |
1,71 g EPA and 1,14 g DHA per day | 15 months (12 months of supplementation/ placebo, 3 months all patients on placebo) |
52 patients with mild RA: 29 in intervention group, 23 in control group | RAI, grip strength, morning stiffness, 100 mm VAS of joint pain severity as assessed by the patient; 100 mm VAS of joint pain severity as assessed by the metrologist, ESR, CRP |
Reduction of NSAID requirement at 3, 6, 9, 12, 15 months. | [35] | |
| Randomized, double-blind controlled trial |
5,5 g EPA and DHA per day (as adjunctive therapy in modern treat-to-target drug treatment) |
12 months | 121 patients with recent onset RA (<12 months’ duration): 75 in intervention group, 46 in control group |
rate of remission, DMARD use, methotrexate dose, mHAQ, DAS28-ESR |
Higher rate of remission and decreased DMARD use. | [36] | |
| Meta-analysis of 17 randomized controlled trials |
At least 2,7g per day of EPA and DHA | 3-4 months | 823 patients; with RA, but also with joint pain secondary to inflammatory bowel disease and dysmenorrhea |
patient assessed pain, physician assessed pain, duration of morning stiffness, number of painful and/or tender joints, RAI, nonselective NSAID consumption |
Improvement in duration of morning stiffness, number of painful and/or tender joints, patient assessed pain and NSAID intake. | [37] | |
| Meta-analysis of 10 randomized controlled trials |
At least 2,7g of omega-3 fatty acids | At least 3 months | 183 RA patients and 187 placebo-treated RA controls |
NSAID intake tender joint count, swollen joint count, morning stiffness and physical function |
Reduction of NSAID dose. | [38] | |
| Meta-analysis of 20 randomized controlled trials |
0.30 to 9.60 g omega-3 fatty acids (either as a supplement or from food sources) | 12 to 72 weeks | 717 patients with RA in the intervention group and 535 RA patients in the control group. | EMS, tender joint count, ESR and pain scale, CRP, IL-6, IL-1 and TNF-α |
Improved early morning stiffness, tender joint count, ESR and pain scale, reduction of leukotriene B4. | [39] | |
| Flaxseed + anti- inflammatory diet |
Randomized controlled trial for healthy eating | Three groups: flax-seed (30 g/day) plus anti-inflammatory diet (AIF group), flaxseed (30 g/day) plus regular diet (RF group), and roasted wheat (30 g/day) plus regular diet (RW group) |
12 weeks | 120 patients with RA (111 women) |
DAS28, HAQ, disability and pain, quality of life, ESR, CRP, rheumatoid factor, anti-CCP |
Flaxseed decreased DAS28 in RF group compared to RW. Pain severity, morning stiffness and disease feeling decreased significantly in AIF and RF groups. HAQ disability and quality of life measurements improved in all 3 groups, with a greater extent in AIF and RF groups compared to RW. Between-group differences were significant for DAS28, pain scores, and physical and mental health variables. ESR, CRP, anti-CCP, and rheumatoid factor were not different between groups. |
[40] |
| Meals based on red meat, fatty fish, or soy protein |
Randomized controlled cross-over trial |
Patients were assigned to eat isocaloric hamburger meals consisting of red meat (60% beef, 40% pork), fatty fish (salmon), or soy protein for breakfast. |
Blood samples were taken before meals and at intervals up to 5 h postprandial. | 25 patients with RA | IL-6 | Postprandial inflammation assessed by IL-6 did not indicate any significant differences between meals. |
[42,43] |
| Fish oil + cranberry juice |
Randomized, single-blind intervention study |
First group: control Second group: 3 g of omega-3 fatty acids daily Third group: 3 g of omega-3 fatty acids and 500 mL of reduced-calorie cranberry juice daily |
90 days | First group: 21 RA patients Second group: 21 RA patients Third group: 20 RA patients |
ESR, CRP, IL-6, DAS28-CRP, adiponectin, RF, leptin |
Decrease in ESR, CRP, DAS28-CRP, adiponectin and IL-6 in the cranberry juice group. Reduction in DAS28- CRP and adiponectin in the fish oil group. |
[69] |
| Double blind, placebo-controlled trial | 200 µg selenium-enriched yeast or a placebo |
90 days | 55 patients with moderate RA |
VAS, the Ritchie index, the number of swollen and painful joints, morning stiffness | No significant differences between groups. |
[62] | |
| Antioxidants | Randomized controlled study |
Group 1: a standard treatment of intramuscular methotrexate (12.5 mg/week), oral sulphasalazine (1g daily in 2 divided doses) and indomethacin (100 mg suppository at bed-time); Group 2: the standard treatment plus a combination of antioxidants (selenium 50 μg, medicinal yeast 105 mg, vitamin A acetate 5.54 mg, ascorbic acid 100 mg and vitamin E 30 mg); Group 3: standard treatment plus high dose of vitamin E (400 mg t.i.d.) |
2 months | 30 RA patients divided into 3 equal groups |
RAI, duration of morning stiffness, rheumatoid factor (Rose Waaler test), ESR, plasma levels of vitamin E and MDA, the activity of GPx |
Improvement in patients condition (RAI, morning stiffness, ESR) was reported after the first month in group 2 and 3. | [65] |
| Open pilot study | 20 g of antioxidant-enriched spread daily: mix of a-tocopherol (400 mg), lycopene (10 mg), palm oil carotenoids (5 mg; mainly α-carotene) and lutein (10 mg). + vitamin C (200 mg daily) as a supplement |
10 weeks + ‘wash-out’ period of 4 weeks. |
8 non-smoking female patients with rheumatoid factor + RA and DAS28 higher than 2.5 | morning stiffness, VAS pain, general health score and DAS28 (including pain, swelling of joints, ESR, general feeling of well-being) |
The number of swollen and painful joints were significantly decreased, and general health significantly increased at t=10 weeks. DAS was significantly decreased (−1.6) at 10 weeks. After the “wash-out period”, the DAS score was increased again by 0.6. |
[66] | |
| Pre-post clinical trial |
Capsules containing 50 μg selenium, 8 mg zinc, 400 μg vitamin A, 125 mg vitamin C, and 40 mg vitamin E | 12 weeks | 39 female patients with RA | DAS28, hs-CRP, number of swollen and painful joints, CAT, GP¬x, SOD, total antioxidant capacity | Significantly decreased DAS28 score and serum hs-CRP. No differences in number of swollen and painful joints. CAT, GP¬¬x, SOD and total antioxidant capacity increased significantly after intervention | [67] | |
| Randomized controlled study |
500 ml of low-calorie cranberry juice |
90 days | 38 women with RA, 20 in intervention group and 18 in control group |
DAS28, ESR, CRP, RF, anti-CCP antibodies | Significant reduction in DAS28 and anti-CCP antibodies levels | [68] | |
| Randomized, single-blinded clinical study | 500 mg curcumin, 50 mg diclofenac sodium or combination of both |
8 weeks | 38 patients with active RA: 14 in curcumin group, 12 in diclofenac sodium group, 12 in curcumin + diclofenac sodium group | DAS28, ESR, VAS, CRP, ACR20, ACR50, ACR70 |
Significant decrease in CRP level only in the curcumin group. Significant decrease in DAS28 and improvement in ACR scores (ACR 20, 50 and 70) in all groups. The results in the curcumin group were significantly better than in the diclofenac sodium group. | [70] | |
| Randomized, Double-Blind, Placebo-Controlled, Two-Dose, Three-Arm, and Parallel-Group Study | Placebo, 250 or 500mg of the curcumin product twice daily |
90 days | 36 RA patients, divided into 3 groups, 12 patients each |
ESR, CRP, RF, VAS, ACR20, tender joint count, swollen joint count, duration of morning stiffness | Significant improvement in DAS28, VAS and ACR scores, significant decrease in ESR, CRP and RF values. |
[71] | |
| Salt | Pre-post clinical trial |
Three weeks of low-sodium intake (< 5 grams of sodium chloride/day) followed by 2 weeks of normal-sodium dietary regimen (5 g of sodium chloride/day) | 5 weeks | 14 RA patients | Th17 and Treg cells , serum levels of IFNα, TGFβ1, IL-23, IL-1β, IL-6, IL-9, IL-12, IL-17, and TNF | Reduction in the percentage of Th17 cells after low-sodium intake period, increase after normal-sodium intake, Treg lymphocytes showed the opposite trend (not statistically significant); reduction of TGFβ and IL-9 |
[80] |
| Diet | Details of intervention | Duration | Participants | Evaluated markers of disease activity |
Effect | Reference |
|---|---|---|---|---|---|---|
| Mediterranean diet | Experimental group: Cretan version of Mediterranean diet Control group: regular Swedish food |
12 weeks | 51 patients with RA, mostly women; 26 in experimental group, 25 in control group | DAS28 HAQ SF-36 NSAID intake |
Improvement in DAS28, HAQ, SF-36. No difference in NSAID intake. | [19] |
| Intervention group: written information about Mediterranean Diet + cooking classes once a week for 6 weeks Control group: written information about healthy eating in general |
6 months | 130 female patients with RA; 75 in intervention group, 55 in control group |
Tender and swollen joint count, patient global VAS, pain score, EMS, DAS28, HAQ score, ESR, CRP, IL-6 |
Improvements after 3 months of diet in pain score and HAQ; after 6 months in patient global VAS, pain score, EMS | [20] | |
| Intervention group: personalized Mediterranean Diet plans and lifestyle consultation on improving physical activity levels, Control group: generic dietary and physical activity advice, based on the National Dietary Guidelines |
12 weeks | 40 adult women with RA on remission |
DAS28, anthropometric indices (BodPod), vitamin D concentrations, blood lipid profiles |
Participants in the intervention group exhibited lower DAS28, improvements in body weight and body composition, blood glucose and serum 1,25(OH)2D concentrations |
[22] | |
| Mediterranean dietary (MD) pattern and low-fat high-carbohydrate (LC-HF) diet Control group: regular diet |
12 weeks | 154 overweight and obese patients with rheumatoid arthritis |
DAS28 ESR VAS |
DAS 28 significantly decreased in MD compared to the LF-HC group and controls. Serum ESR level was significantly decreased in MD compared to the LF-HC and control groups. VAS was significantly de- creased in the LF-HC and MD groups compared to the controls; no significant difference between the MD and LF-HC groups |
[24] | |
| Vegan diet | Very low fat (10%), vegan diet No control group |
4 weeks | 24 RA patients | Joint swelling and joint tenderness score, degree of pain (VAS), limitation in ability of function, severity of morning stiffness, and duration of morning stiffness, ESR, CRP, RA factor |
Significant decrease in joint tenderness score, joint swelling score, degree of pain because of RA and improvement in severity of morning stiffness and limitation in ability to function. | [49] |
| Intervention: multidisciplinary lifestyle program based on a whole-food plant-based diet, physical activity, and stress management Control group: usual care |
16 weeks | 83 patients with RA | DAS28, Dutch-Flemish Patient Reported Outcomes Measurement Information System (PROMIS) |
Improvement of DAS28 in intervention group vs control group. All patient-reported outcome measures changed slightly (not statistically significantly) in favour of the intervention group |
[50] | |
| Intervention group: uncooked vegan diet (living food) Control group: omnivorous diet |
3 months | 40 RA patients | RAI1, joint stiffness, pain (VAS) | Significant subjective alleviation of symptoms, improvement in RAI1 | [51] | |
| Vegan gluten-free diet | Intervention group: vegan diet free of gluten Control group: well-balanced non-vegan diet |
At least 9 months (up to 1 year) | 47 RA patients: 22 in intervention group, 25 in control group | ACR20 response criteria, Radiographic assessment (Larsen score, number of erosions, joint count) |
Significant improvement in vegan diet patients in all measures contained within ACR response criteria, except CRP. No significant differences in radiographic progression between two groups. |
[53] |
| Intervention group: vegan diet free of gluten, started with 1-day low-energy fasting Control group: well-balanced non-vegan diet |
At least 3 months (up to 12 months) | 58 patients with active RA: 30 in intervention group, 28 in control group |
DAS28 ACR20 response HAQ score ESR, CRP |
Significantly lower DAS28 and HAQ score after 3 and 12 months, CRP after 12 months of vegan diet | [54] | |
| Gluten intake | Participants were stratified based on their habitual gluten intake: high and low- to- medium gluten intake. | 14-16 weeks | 37 RA patients | clinical response to biological treatment (ACR20), HrQoL, CRP |
No statistically significant differences were found between the groups |
[55] |
| Vegan diet and fasting | Intervention group: 7-10 days of fasting, 3,5 months of gluten-free vegan diet and subsequently lactovegetarian diet for up to a year Control group: ordinary mixed food |
13 months | 34 RA patients | number of tender and swollen joints, RAI, pain, morning stiffness duration, grip strength, HAQ score, ESR, CRP, WBC count | After 4 weeks improvement in vegan group in number of tender and swollen joints, RAI, pain, morning stiffness duration, grip strength, HAQ score, ESR, CRP and WBC count. Effects maintained throughout the year. | [57] |
| Fasting group: 7 days of subtotal fast, 2 weeks of re-feeding with lactovegetarian diet. Ketogenic group: isocaloric ketogenic diet, 2 weeks of re-feeding with lactovegetarian diet. |
21 days | 23 RA patients with active disease: 10 in fasting group, 13 in ketogenic group |
IL-6, CRP, ESR, tender joints |
Significant decrease in serum IL-6 and improvement in ESR, CRP and tender joint count after 7-day-fasting, but not after ketogenic diet. |
[58] | |
| Intervention group: 7-day fast followed by an 11-week whole-food plant-based diet combined with time-restricted eating (16:8) Control group: guideline-based anti-inflammatory diet following official German guidelines (German Nutrition Society, DGE) |
12 weeks | 41 women with RA | DAS28 HAQ-DI CDAI SDAI ACR20, ACR50, ACR70 VAS |
The intervention group achieved a HAQ-DI reduction at day 7 of the fast, which was sustained throughout the whole study period up until week 12 and even to the 6-month follow-up. In the control group a similar functional enhancement was seen from study week 6 on up to 6 months. Both dietary approaches had a positive effect on RA disease activity, no significant differences. |
[59] | |
| Intermittent fasting | Intervention group: 16:8 IF diet Control group: maintaining their usual diet + recommendations for healthy eating |
8 weeks | 44 overweight and obese postmenopausal women with RA |
HAQ-DI morning joint stiffness ESR, hs-CRP, IL-6, DAS28, CDAI |
The intervention diet significantly improved DAS-28, CDAI, and HAQ. No other significant differences. |
[60,61] |
| Anti-inflammatory Diet In Rheumatoid Arthritis (ADIRA) | Intervention diet: containing a portfolio of suggested anti-inflammatory foods Control diet: similar to the general dietary intake in Sweden |
10 weeks. After a 4-month washout period the participants switched diet | 47 RA patients | DAS28-ESR, tender and swollen joints, ESR, and visual analog scale for general health, DAS28-CRP | No significant difference between the intervention and control periods for DAS28-ESR. However, DAS28-ESR was significantly lower after the intervention period than before. |
[73] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




