Submitted:
10 March 2026
Posted:
11 March 2026
You are already at the latest version
Abstract
Keywords:
“At the heart of science is an essential balance between two seemingly contradictory attitudes- an openness to new ideas, no matter how bizarre or counterintuitive they may be, and the most ruthless skeptical scrutiny of all ideas, old and new”.C. Sagan, in: The Demon-Haunted World: Science as a candle in the Dark
1. Introduction: Formulating the Nature of the Problem
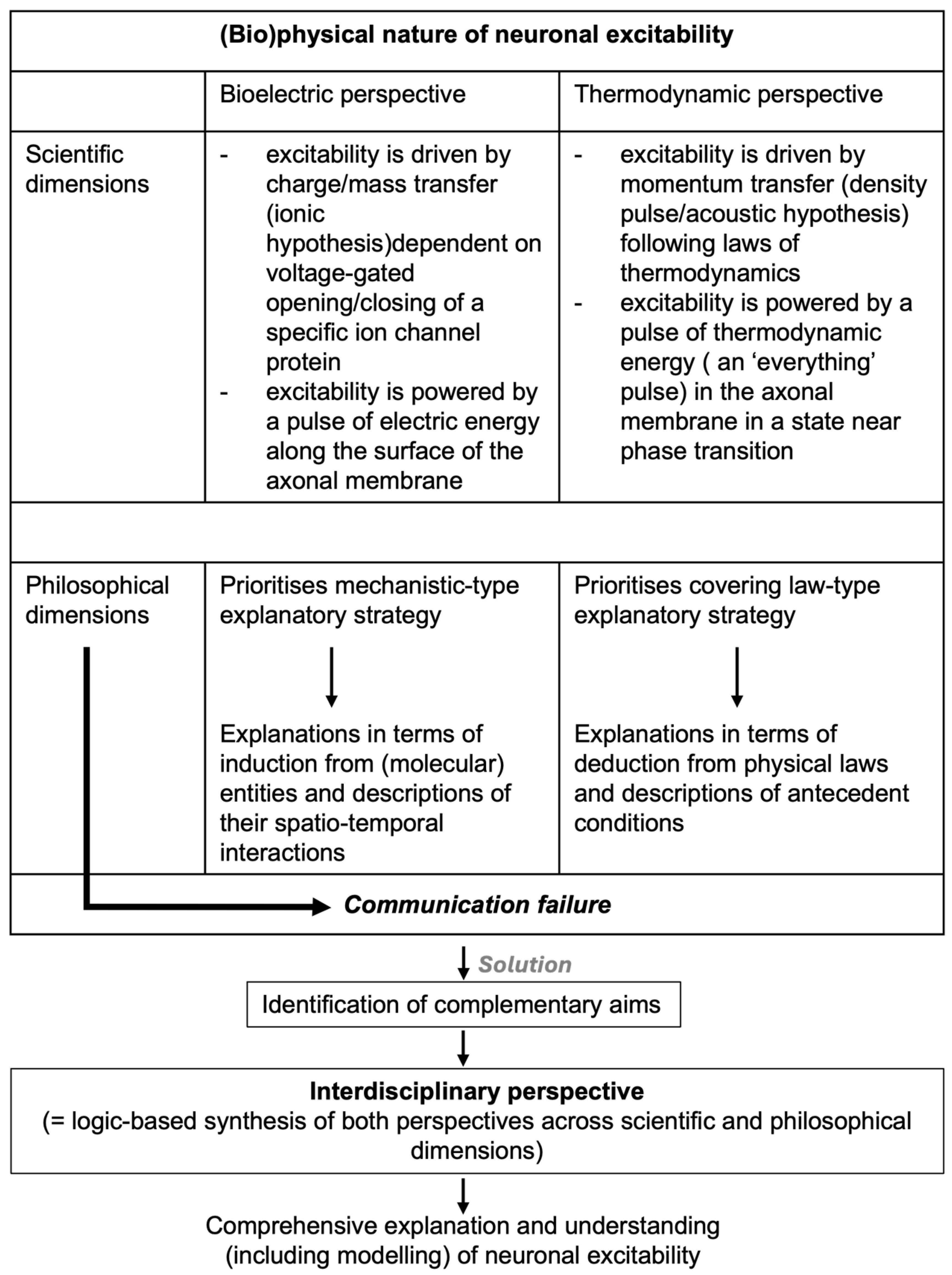
2. Explaining the Physics of Neuronal Excitability: The Bioelectric Perspective
3. Explaining the Physics of Neuronal Excitability: The Thermodynamic Perspective
4. Explaining the Physics of Neuronal Excitability: Towards an Interdisciplinary Perspective
5. Debating the Physical Nature of Neuronal Excitability: Some Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Akkin, T.; Landowne, D.; Sivaprakasam, A. Optical coherence tomography phase measurement of transient changes in squid giant axons during activity. J Membr Biol. 2009, 231, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Alvargonzalez, D. Multidisciplinarity, interdisciplinarity, transdisciplinarity, and the sciences. Int Stud Phil Sci. 2011, 25, 387–403. [Google Scholar] [CrossRef]
- Andersen, O.S. Introduction to biophysics week: what is biophysics? Biophys J 2016, 110, E01–E03. [Google Scholar] [CrossRef]
- Andersen, S.S.L.; Jackson, A.D.; Heimburg, T. Towards a thermodynamic theory of nerve pulse propagation. Progr Neurobiol. 2009, 88, 104–113. [Google Scholar] [CrossRef]
- Baluska, F.; Miller, W.B., Jr.; Reber, A.S. Sentient cells as basic units of tissues, organs and organismal physiology. J Physiol. 2024, 602, 2491–2501. [Google Scholar] [CrossRef]
- Bavi, N.; Cox, C.D.; Nikolaev, Y.A.; et al. Molecular insights into the force-from-lipids gating of mechanosensitive channels. Curr Opin Physiol. 2023, 36, 100706. [Google Scholar] [CrossRef]
- Bean, B.P. The action potential in mammalian central neurons. Nat Rev Neurosci. 2007, 8, 451–465. [Google Scholar] [CrossRef]
- Behrends, J.C. Evolution of the ion channel concept: the historical perspective. Chem Rev. 2012, 112, 6218–6226. [Google Scholar] [CrossRef]
- Bowditch, H.P. What is nerve-force? Science 1886, 8, 196–198. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, N. Models of the nerve impulse. In Routledge Handbook of Philosophy of Scientific Modeling; 2024; pp. 549–560. [Google Scholar] [CrossRef]
- Clay, J.R. Axon excitability revisited. Prog Biophys Mol Biol. 2005, 88, 59–90. [Google Scholar] [CrossRef] [PubMed]
- Catterall, W.A. Voltage gated sodium and calcium channels: discovery, structure, function, and pharmacology. Channels 2023, 17, 2281714. [Google Scholar] [CrossRef]
- Catterall, W.A.; Raman, I.M.; Robinson, H.P.C.; et al. The Hodgkin-Huxley heritage: from channels to circuits. J Neurosci. 2012, 32, 14064–14073. [Google Scholar] [CrossRef]
- Cook, N.D.; Carvalho, G.B.; Damasio, A. From membrane excitability to metazoan psychology. Trends Neurosci. 2014, 37, 698–705. [Google Scholar] [CrossRef]
- Craver, C.F. Physical laws and mechanistic explanation in the Hodgkin and Huxley model of the action potential. Philos Sci. 2008, 75, 1022–1033. [Google Scholar] [CrossRef]
- Darbellay, F. The gift of interdisciplinarity: towards an ability to think across disciplines. Int J Talent Develop Creativity 2015, 3, 201–211. [Google Scholar]
- Darden, L.; Maull, N. Interfield theories. Philos Sci. 1977, 44, 43–64. [Google Scholar] [CrossRef]
- Debanne, D. Information processing in the axon. Nat Rev Neurosci. 2004, 5, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Debanne, D.; Campanac, E.; Bialowas, A.; et al. Axon physiology. Physiol Rev. 2011, 91, 555–602. [Google Scholar] [CrossRef] [PubMed]
- Drukarch, B.; Holland, H.A.; Velichkov, M.; et al. Thinking about the nerve impulse: a critical analysis of the electricity-centered conception of nerve excitability. Prog Neurobiol. 2018, 169, 172–185. [Google Scholar] [CrossRef] [PubMed]
- Drukarch, B.; Wilhelmus, M.M.M.; Shrivastava, S. The thermodynamic theory of action potential propagation: a sound basis for unification of the physics of nerve impulses. Rev Neurosci. 2022, 33, 285–302. [Google Scholar] [CrossRef]
- Drukarch, B.; Wilhelmus, M.M.M. Thinking about the action potential: the nerve signal as a window to the physical principles guiding neuronal excitability. Front Cell Neurosci. 2023, 17, 1232020. [Google Scholar] [CrossRef] [PubMed]
- Drukarch, B.; Wilhelmus, M.M.M. Understanding the scope of the contemporary controversy about the physical nature and modeling of the action potential: insights from history and philosophy of (neuro)science. OBM Neurobiol. 2025, 9, 269. [Google Scholar] [CrossRef]
- El Hady, A.; Machta, B.B. Mechanical surface waves accompany action potential propagation. Nat Commun. 2015, 6, 6697. [Google Scholar] [CrossRef]
- Engelbrecht, J.; Tamm, K.; Peets, T. Interdisciplinarity in modelling of biophysical processes. Proc Est Acad Sci. 2025, 74, 544–550. [Google Scholar] [CrossRef]
- Fagan, M.B. The joint account of mechanistic explanation. Philos Sci. 2012, 79, 448–472. [Google Scholar] [CrossRef]
- Fagan, M.B. Stem cells and systems models: clashing views of explanation. Synthese 2016, 193, 873–907. [Google Scholar] [CrossRef]
- Fagan, M.B. Explanation, interdisciplinarity, and perspectives. In Understanding perspectivism: scientific challenges and methodological prospects; Massini, M, McCoy, CD, Eds.; Taylor and Francis, 2019; pp. 28–49. [Google Scholar]
- Fedosejevs, C.S.; Schneider, M.F. Sharp, localized phase transitions in single neuronal cells. PNAS USA 2022, 119, e2117521119. [Google Scholar] [CrossRef] [PubMed]
- Fillafer, C.; Paeger, A.; Schneider, M.F. The living state: how cellular excitability is controlled by the thermodynamic state of the membrane. Progr Biophys Mol Biol. 2021, 162, 57–68. [Google Scholar] [CrossRef]
- Finger, S.; Wade, N.J. The neuroscience of Helmholtz and the theories of Johannes Mueller part 1: nerve cell structure, vitalism, and the nerve impulse. J Hist Neurosci. 2002, 11, 136–155. [Google Scholar] [CrossRef] [PubMed]
- Fox, D. The brain, reimagined. Sci Am. 2018, 318, 60–67. [Google Scholar] [CrossRef]
- Green, S.; Fagan, M.; Jaeger, J. Explanatory integration challenges in evolutionary systems biology. Biol Theory 2015, 10, 18–35. [Google Scholar] [CrossRef]
- Griswold, J.M.; Bonilla-Quintana, M.; Pepper, R.; et al. Membrane mechanics dictate axonal pearls-on-a-string morphology and function. Nat Neurosci. 2025, 28, 49–61. [Google Scholar] [CrossRef]
- Heimburg, T. Mechanical aspects of membrane thermodynamics. Estimation of the mechanical properties of lipid membranes close to the chain melting transition from calorimetry. Biochim Biophys Acta 1998, 1415, 147–162. [Google Scholar] [CrossRef]
- Heimburg, T. The important consequences of the reversible heat production in nerves and the adiabaticity of the action potential. Progr Biophys Mol Biol. 2021, 162, 26–40. [Google Scholar] [CrossRef]
- Heimburg, T. The thermodynamic soliton theory of the nervous impulse and possible medical implications. Progr Biophys Mol Biol. 2022, 173, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Heimburg, T. The mechanical properties of nerves, the size of the action potential, and consequences for the brain. Chem Phys Lip 2025, 267, 105461. [Google Scholar] [CrossRef]
- Heimburg, T.; Jackson, A.D. On soliton propagation in biomembranes and nerves. PNAS USA 2005, 102, 9790–9795. [Google Scholar] [CrossRef] [PubMed]
- Hempel, C.G.; Oppenheim, P. Studies in the logic of explanation. Philos Sci. 1948, 15, 135–175. [Google Scholar] [CrossRef]
- Hein, H. Molecular biology vs. organicism: the enduring dispute between mechanism and vitalism. Synthese 1969, 20, 238–253. [Google Scholar] [CrossRef]
- Hill, A.V. The absence of temperature changes during the transmission of a nervous impulse. J Physiol. 1912, 43, 433–440. [Google Scholar] [CrossRef]
- Hill, A.V. The heat-production and recovery of crustacean nerve. Proc Roy Soc Lond B 1929, 105, 153–176. [Google Scholar] [CrossRef]
- Hill, A.V. The heat production of muscle and nerve 1848-1914. Ann Rev Physiol. 1959, 21, 1–18. [Google Scholar] [CrossRef]
- Hille, B. A brief history of nerve action potentials after 1600. Mol Pharmacol. 2025, 107, 100012. [Google Scholar] [CrossRef]
- Hodgkin, A.L. The ionic basis of electrical activity in nerve and muscle. Biol Rev. 1951, 26, 339–409. [Google Scholar] [CrossRef]
- Hodgkin, A.L. The ionic basis of nervous conduction. Science 1964a, 145, 1148–1154. [Google Scholar] [CrossRef] [PubMed]
- Hodgkin, A.L. The conduction of the nervous impulse; Liverpool University Press: Liverpool, GB, 1964b. [Google Scholar]
- Hodgkin, A.L.; Huxley, A.F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol. 1952, 117, 500–544. [Google Scholar] [CrossRef] [PubMed]
- Hodgkin, A.L.; Katz, B. The effect of sodium ions on the electrical activity of the giant axon of the squid. J Physiol. 1949a, 108, 37–77. [Google Scholar] [CrossRef] [PubMed]
- Hodgkin, A.L.; Katz, B. The effect of temperature on the electrical activity of the giant axon of the squid. J Physiol. 1949b, 109, 240–249. [Google Scholar] [CrossRef]
- Hodgkin, A.L.; Rushton, W.A.H. The electrical constants of a crustacean nerve fibre. Proc R Soc Lond B Biol Sci. 1946, 133, 444–479. [Google Scholar] [CrossRef]
- Hohol, M.; Furman, M. On explanation in neuroscience: the mechanistic framework. In The concept of explanation; Heller, M, Brozek, B, Hohol, M, Eds.; Copernicus Center Press: Krakow, PL, 2015; pp. 207–235. ISBN 1-13, 9788378862048. [Google Scholar]
- Holland, L.; de Regt, H.W.; Drukarch, B. Two scientific perspectives on nerve signal propagation: how incompatible approaches jointly promote progress in explanatory understanding. Hist Philos Life Sci. 2024, 46, 43. [Google Scholar] [CrossRef]
- Huang, C.-H.; Albeck, J.G.; Devreotes, P.N. Self-organizing and excitable signaling networks in cell biology. Front Cell Dev Biol. 2024, 12, 1430911. [Google Scholar] [CrossRef]
- Iwasa, K.; Tasaki, I. Mechanical changes in squid giant axons associated with production of action potentials. Biochem Biophys Res Commun. 1980, 95, 1328–1331. [Google Scholar] [CrossRef]
- Iwasa, K.; Tasaki, I.; Gibbons, R.C. Swelling of nerve fibers associated with action potentials. Science 1980, 210, 338–339. [Google Scholar] [CrossRef]
- Jerusalem, A.; Al-Rekabi, Z.; Chen Het, a.l. Electrophysiological-mechanical coupling in the neuronal membrane and its role in ultrasound neuromodulation and general anaesthesia. Acta Biomater. 2019, 97, 116–149. [Google Scholar] [CrossRef]
- Joly, E. Hypothesis: could the signalling function of membrane microdomains involve a localized transition of lipids from liquid to solid state? BMC Cell Biol. 2004, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Keener, J.; Sneyd, J. Mathematical physiology I: cellular physiology, 2nd ed.; Springer Verlag: New York, NY, 2009. [Google Scholar] [CrossRef]
- Konsman, J.P.; Reyes, T.M. The lost cause of not being mechanistic enough? A perspective inspired by philosophy of science. Brain Behav Immunity 2020, 84, 1–3. [Google Scholar] [CrossRef]
- Kukita, F.; Yamagishi, S. Slowing of the time course of the excitation of squid giant axons in viscous solutions. J Membr Biol. 1979, 47, 303–325. [Google Scholar] [CrossRef]
- Kumar, J.; Gupta, P.D.; Ghosh, S. Investigating the role of axonal ion channel cooperativity in action potential dynamics: studies on Hodgkin-Huxley model. Biophys Chem. 2024, 311, 107257. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, E.I. (Mis)Understanding scientific disagreement: success versus pursuit-worthiness in theory choice. Stud Hist Phil Sci. 2021, 85, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Ling, G.N. The cellular resting and action potentials: interpretation based on the association-induction hypothesis. Physiol Chem Phys. 1982, 14, 47–96. [Google Scholar]
- Ling, T.; Boyle, K.C.; Zuckerman, V.; et al. High-speed interferometric imaging reveals dynamics of neuronal deformation during the action potential. PNAS USA 2020, 117, 10278–10285. [Google Scholar] [CrossRef] [PubMed]
- Machamer, P.; Darden, L.; Craver, C.F. Thinking about mechanisms. Philos Sci. 2000, 67, 1–25. [Google Scholar] [CrossRef]
- Mosgaard, L.D.; Heimburg, T. Lipid ion channels and the role of proteins. Accounts Chem Res. 2013, 46, 2966–2976. [Google Scholar] [CrossRef]
- Mosgaard, L.D.; Zecchi, K.A.; Heimburg, T.; et al. The effects of the nonlinearity of the response of lipid membranes to voltage perturbations on the interpretation of their electrical properties. A new theoretical description. Membranes 2015, 5, 495–512. [Google Scholar] [CrossRef]
- Mueller, J.K.; Tyler, W.J. A quantitative overview of biophysical forces impinging on neural function. Phys. Biol. 2014, 11, 051001. [Google Scholar] [CrossRef] [PubMed]
- Mussel, M. Information propagated by longitudinal pulses near a van der Waals phase transition. Phys Rev E 2023, 108, 034209. [Google Scholar] [CrossRef]
- Mussel, M.; Fillafer, C.; Ben-Porath, G.; et al. Surface deformation during an action potential in pearled cells. Phys Rev E 2017, 96, 052406. [Google Scholar] [CrossRef]
- Mussel, M.; Schneider, M.F. Sound pulses in lipid membranes and their potential function in biology. Progr Biophys Mol Biol. 2021, 162, 101–110. [Google Scholar] [CrossRef]
- Naundorf, B.; Wolf, F.; Volgushev, M. Unique features of action potential initiation in cortical neurons. Nature 2006, 440, 1060–1063. [Google Scholar] [CrossRef]
- Nigro, C. The brain electric: a history of neuroscientific ideas about how we change. UCSF electronic theses and dissertations, San Francisco, CA, 2020. Available online: https://scholarship.org/uc/item/3fw5n2rd.
- O’Brien, J.E. The identity of nerve force and electricity. In JAMA; Annals of 53th annual meeting of American Medical Association, 1900; pp. 630–635. [Google Scholar] [CrossRef]
- Oh, S.; Fang-Yen, C.; Choi, W.; et al. Label-free imaging of membrane potential using membrane electromotility. Biophys J. 2012, 103, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Parker, D. Kuhnian revolutions in neuroscience: the role of tool development. Biol Philos. 2018, 33, 17. [Google Scholar] [CrossRef]
- Pezzulo, G.; Levin, M. Top-down models in biology: explanation and control of complex living systems above the molecular level. J R Soc Interface 2016, 13, 20160555. [Google Scholar] [CrossRef]
- Piccolino, M. Animal electricity and the birth of electrophysiology: the legacy of Luigi Galvani. Brain Res Bull. 1998, 46, 381–407. [Google Scholar] [CrossRef]
- Rvachev, M.M. On axoplasmic pressure waves and their possible role in nerve impulse propagation. Biophys Rev Lett. 2010, 5, 73–88. [Google Scholar] [CrossRef]
- Rvachev, M.M.; Drukarch, B. Surface waves and axoplasmic pressure waves in action potential propagation: fundamentally different physics or two sides of the same coin? Biophys Rev Lett. 2025, 20, 283–289. [Google Scholar] [CrossRef]
- Salmon, W.C. Four decades of scientific explanation; University of Minnesota Press: Minneapolis Minn, 1989. [Google Scholar]
- Schneider, M.F. This is not about the molecules. On the violation of momentum conservation in biology. arXiv 2020, 2004.10307. [Google Scholar] [CrossRef]
- Schneider, M.F. Living systems approached from physical principles. Progr Biophys Mol Biol. 2021, 162, 2–25. [Google Scholar] [CrossRef]
- Schoffeniels, E.; Margineanu, D. Molecular basis and thermodynamics of bioelectrogenesis, 1nd ed.; Kluwer Academic Publishers: Dordrecht, the Netherlands, 1990; p. 125. [Google Scholar] [CrossRef]
- Shrivastava, S. Shock and detonation waves at an interface and the collision of action potentials. Progr Biophys Mol Biol. 2012, 162, 111–121. [Google Scholar] [CrossRef]
- Shrivastava, S.; Kang, K.H.; Schneider, M.F. Collision and annihilation of nonlinear sound waves and action potentials in interfaces. J R Soc Interface 2018, 15, 20170803. [Google Scholar] [CrossRef]
- Tamagawa, H.; Ikeda, K. Another interpretation of the Goldman-Hodgkin-Katz equation based on Ling’s adsorption theory. Eur Biophys J. 2018, 47, 869–879. [Google Scholar] [CrossRef]
- Tasaki, I. Physiology and electrochemistry of nerve fibers, 1nd ed.; Academic Press: New York, NY, 1982. [Google Scholar] [CrossRef]
- Tasaki, I.; Kusano, K.; Byrne, P.M. Rapid mechanical and thermal changes in the garfish olfactory nerve associated with a propagated impulse. Biophys J. 1989, 55, 1033–1040. [Google Scholar] [CrossRef]
- Tasaki, I.; Byrne, P.M. Heat production associated with a propagated impulse in bullfrog myelinated nerve fibers. Jpn J Physiol. 1992, 42, 805–813. [Google Scholar] [CrossRef]
- Tasaki, I.; Iwasa, K. Rapid pressure changes and surface displacements in the squid giant axon associated with production of action potentials. Jpn J Physiol. 1982, 32, 69–81. [Google Scholar] [CrossRef]
- Tasaki, I.; Singer, I.; Takenaka, T. Effects of internal and external ionic environment on excitability of squid giant axon: a macromolecular approach. J Gen Physiol. 1965a, 48, 1095–1123. [Google Scholar] [CrossRef]
- Tasaki, I.; Singer, I.; Watanabe, A. Excitation of internally perfused squid giant axons in sodium-free media. PNAS USA 1965b, 54, 763–769. [Google Scholar] [CrossRef]
- Tasaki, I.; Watanabe, A.; Singer, I. Excitability of squid giant axon in the absence of univalent cations in the external medium. PNAS USA 1966, 56, 1116–1122. [Google Scholar] [CrossRef]
- Tazawa, M.; Shimmen, T. Artificial control of cytoplasmic pH and its bearing on cytoplasmic streaming, electrogenesis and excitability of Characeae cells. The Botanical Magazine=Shokubutsu-gaku-zasshi 1982, 147–154. [Google Scholar] [CrossRef]
- Terakawa, S; Nakayama, H. Are axoplasmic microtubules necessary for membrane excitation? J Membr Biol. 1985, 85, 65–77. [Google Scholar] [CrossRef]
- Verkhratsky, A.; Krishtal, O.A.; Petersen, O.H. From Galvani to patch clamp: the development of electrophysiology. Pfluegers Arch-Eur J Physiol. 2006, 453, 233–247. [Google Scholar] [CrossRef]
- Wan, K.Y.; Jekely, G. Origins of eukaryotic excitability. Phil Trans R Soc B 2021, 376, 20190758. [Google Scholar] [CrossRef]
- Watanabe, A.; Tasaki, I.; Singer, I.; Lerman, L. Effects of tetrodotoxin on excitability of squid giant axon in sodium-free media. Science 1967, 155, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Wilke, E.; Atzler, E. Experimentelle Beitraege zum Problem der Reizleitung im Nerven. Pflug Arch Gesamte Physiol Menschen Tiere 1912, 146, 430–446. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, X.; Wang, S.; et al. Plasmonic imaging of subcellular electromechanical deformation in mammalian cells. J Biomed Opt. 2019, 24, 066007. [Google Scholar] [CrossRef]
- Zecchi, K.A.; Heimburg, T. Non-linear conductance, rectification, and mechanosensitive channel formation of lipid membranes. Front Cell Dev Biol. 2021, 8, 592520. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



