Submitted:
10 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
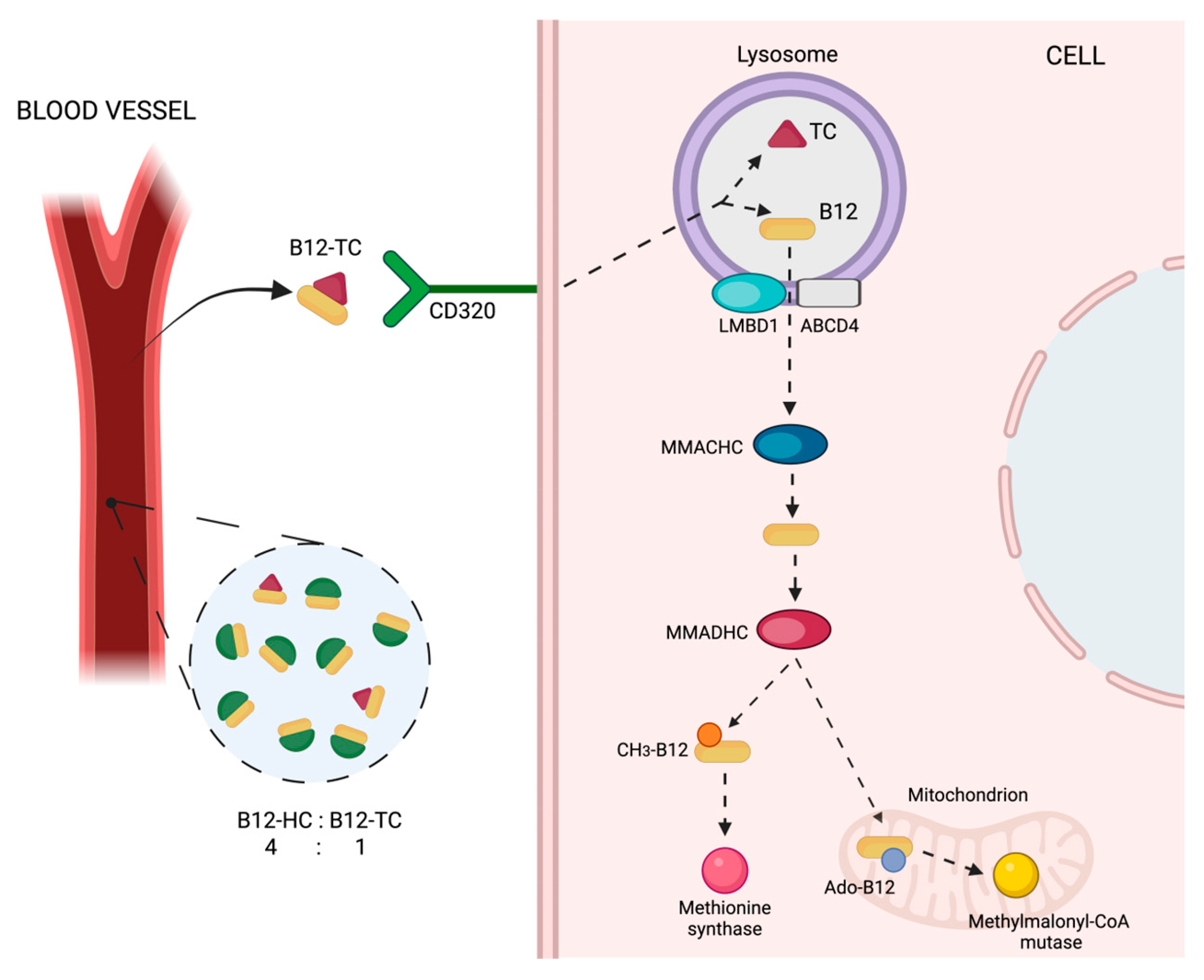
3. Vitamin B12 and Holotranscobalamin (holoTC)
3.1. Biochemical Properties
3.2. Metabolic Aspects and Plasma Levels
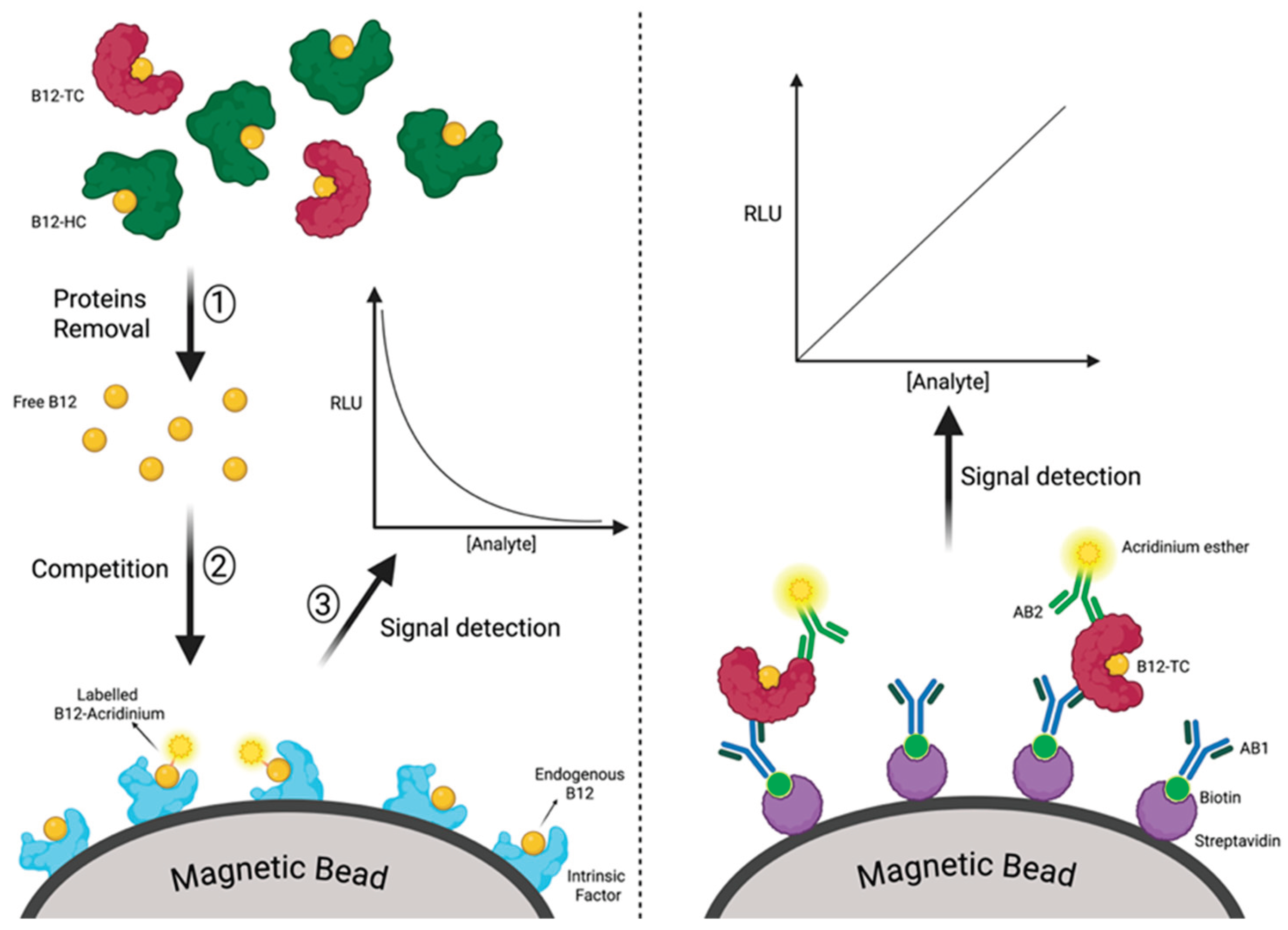
3.3. Analytical Methods
| Analyte | Assay | Manufacturer | Platforms | Method | Assay range | Expected Values |
Standardization |
|---|---|---|---|---|---|---|---|
| Total B12 | Mindray Vitamin B12 Assay [73] |
Mindray Medical International Limited | Mindray CL series | CLIA | 50.0-2000.0 pg/mL |
180.0-916.0 pg/mL |
Against a commercial VB12 test (CLIA) |
| DiaSorin LIAISON Vitamin B12 [74] |
DiaSorin S.p.a. | Liaison/XL | CLIA | 55.0-1500.0 pg/mL |
107.2-653.3 pg/mL |
In-house Standard preparation |
|
|
MAGLUMI Vitamin B12 [75] |
Snibe | MAGLUMI | CLIA | 12.5-2000.0 pg/mL |
200.0-1100.0 pg/mL |
WHO IS 03/178 | |
| TOSOH AIA-PACK B12 [76] |
Tosoh Bioscience | Tosoh AIA | EIA | 50.0-2000.0 pg/mL |
230.0-1050.0 pg/mL |
In-house Standard preparation |
|
| VIDAS Vitamin B12 total [77] |
Biomeriéux | VIDAS | ELFA | 100.0-1200.0 pg/mL |
166.9-582.8 pg/mL |
WHO IS 03/178 | |
| Alinity i B12 [78] |
Abbott Diagnostics | Alinity i | CMIA | 146.0-2000.0 pg/mL |
187.0-883.0 pg/mL |
WHO IS 03/178 | |
| Atellica IM Vitamin B12 (VB12) [79] |
Siemens Healthineers | Atellica IM | CLIA | 45.0-2000.0 pg/mL |
211.0-911.0 pg/mL |
Cyanocobalamin (B12) USP Reference Standard | |
| Elecsys Vitamin B12 II [80] | Roche Diagnostics | Cobas E series | ECLIA | 150.0-2000.0 pg/mL |
232.0-1245.0 pg/mL |
WHO IS 03/178 | |
| Access Vitamin B12 [81] |
Beckman Coulter | Access/UniCel DxI | CLIA | 105.0-2100.0 pg/mL |
180.0-914.0 pg/mL |
EN ISO 17511 | |
| HoloTC | Alinity i Active-B12 (holoTC) [82] |
Abbott Diagnostics |
Alinity i | CMIA | 5.0-128.0 pmol/L |
25.1-165.0 pmol/L |
WHO IS 03/178 |
| Atellica Active-B12 [83] | Siemens Healthineers |
Atellica IM | CLIA | 4.25–146.0 pmol/L |
27.2-169.6 pmol/L |
WHO IS 03/178 | |
| Elecsys Active B12 [84] | Roche Diagnostics |
Cobas E series |
ECLIA | 3.0-150.0 pmol/L |
37.5-188.0 pmol/L |
WHO IS 03/178 | |
| Access Active-B12 (holoTC) [85] |
Beckman Coulter |
Access/UniCel DxI | CLIA | 3.0-160.0 pmol/L |
32.2-152.6 pmol/L |
EN ISO 17511 |
3.3.1. Reference Materials and Assay Alignment
4. Discussion
5. Conclusions
Institutional Review Board
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AB1 | Antibody 1 |
| AB2 | Antibody 2 |
| Ado-Cbl | Adenosyl Cobalamin |
| B6 | Pyridoxine vitamine |
| B12 | Riboflavin vitamine |
| Cbl | Cobalamin |
| CBS | Cystathionine β-synthase |
| CGL | Cystathionine γ-lyase |
| CH3 | Methyl group |
| CH3-Cbl | Methylcobalamin |
| CLIA | Chemiluminescence immunoassay |
| CMIA | Chemiluminescent microparticle immunoassay |
| CoA | Coenzyme A |
| DNA | Deoxyribonucleic Acid |
| ECLIA | Electrochemiluminescence immunoassay |
| EFLM | European Federation of Clinical Chemistry and Laboratory Medicine |
| EIA | Enzyme Immunoassay |
| ELFA | Enzyme-linked Fluorescent Assay |
| ELISA | Enzyme-linked immunosorbent assays |
| EN ISO | European Norm International Organization for Standardization. |
| EQA | external quality assessment |
| f | females |
| HC | Haptocorrin |
| Hcy | Homocysteine |
| holoTC | holotranscobalamin |
| IF | Intrinsic Factor |
| IS | International Standard |
| kDa | kiloDalton |
| LC-MS/MS | liquid chromatography–tandem mass spectrometry |
| m | males |
| MAT | Methionine Adenosyle Transferase |
| MMA | Methylmalonic acid |
| MRP1 | Multidrug Resistance Protein 1 |
| MTHFR | Methylenetetrahydrofolate reductase |
| NG239 | NICE Guideline 239 |
| ng/L | nanograms/liter |
| NH2 | Amino group |
| NIBSC | National Institute for Biological Standards and Control |
| NICE | National Institute for Health and Care Excellence |
| OH | Hydroxyl group |
| PCC | Propionyl-CoA Carboxylase |
| pg/mL | picograms/milliliter |
| POCT | Point-Of-Care Testing |
| pmol/L | picomoles/liter |
| RI | Reference Intervals |
| RIA | radioimmunoassays |
| RLU | Relative Lights Units |
| SAH | S-adenosyl-homocysteine |
| SAHH | S-adenosyl-homocysteine hydrolase |
| SAM | S-adenosyl-methionine |
| TC | Transcobalamin |
| TCN2 | Transcobalamin 2 |
| THF | Tetrahydrofolate |
| USP | Unites States Pharmacopeia |
| WHO | World Health Organization |
| y.o | years old |
| ug | micrograms |
References
- Lu, T.; Paterson, A.D. Estimating effects of serum vitamin B12 levels on psychiatric disorders and cognitive impairment: A Mendelian randomization study. Commun Med 2025, 5, 316. [Google Scholar] [CrossRef]
- Green, R.; Allen, L.H.; Bjorke-Monsen, A.L.; Brito, A.; Gueant, J.L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.H.; et al. Vitamin B(12) deficiency. Nat Rev Dis Primers 2017, 3, 17040. [Google Scholar] [CrossRef] [PubMed]
- Hariz, A.; Bhattacharya, P.T. Megaloblastic Anemia; StatPearls: Treasure Island, FL, USA, 2025. [Google Scholar]
- Calderon-Ospina, C.A.; Nava-Mesa, M.O. B Vitamins in the nervous system: Current knowledge of the biochemical modes of action and synergies of thiamine, pyridoxine, and cobalamin. CNS Neurosci Ther 2020, 26, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Venkatramanan, S.; Armata, I.E.; Strupp, B.J.; Finkelstein, J.L. Vitamin B-12 and Cognition in Children. Adv Nutr 2016, 7, 879–888. [Google Scholar] [CrossRef]
- Zaric, B.L.; Obradovic, M.; Bajic, V.; Haidara, M.A.; Jovanovic, M.; Isenovic, E.R. Homocysteine and Hyperhomocysteinaemia. Curr Med Chem 2019, 26, 2948–2961. [Google Scholar] [CrossRef] [PubMed]
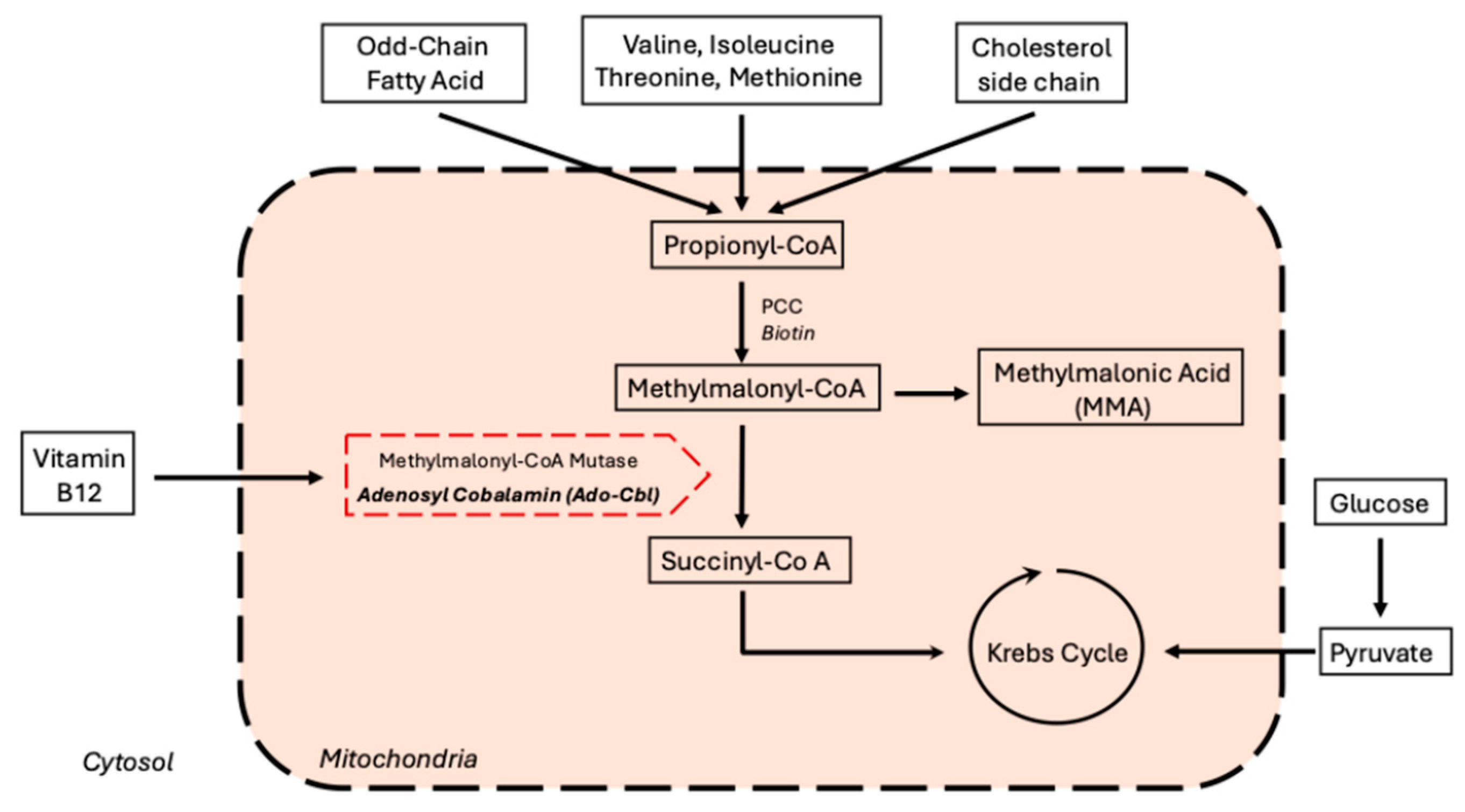
- Takahashi-Iniguez, T.; Garcia-Hernandez, E.; Arreguin-Espinosa, R.; Flores, M.E. Role of vitamin B12 on methylmalonyl-CoA mutase activity. J Zhejiang Univ Sci B 2012, 13, 423–437. [Google Scholar] [CrossRef]
- Yadav, D.K.; Shrestha, S.; Lillycrop, K.A.; Joglekar, C.V.; Pan, H.; Holbrook, J.D.; Fall, C.H.; Yajnik, C.S.; Chandak, G.R. Vitamin B(12) supplementation influences methylation of genes associated with Type 2 diabetes and its intermediate traits. Epigenomics 2018, 10, 71–90. [Google Scholar] [CrossRef]
- Moore, E.; Mander, A.; Ames, D.; Carne, R.; Sanders, K.; Watters, D. Cognitive impairment and vitamin B12: A review. Int Psychogeriatr 2012, 24, 541–556. [Google Scholar] [CrossRef]
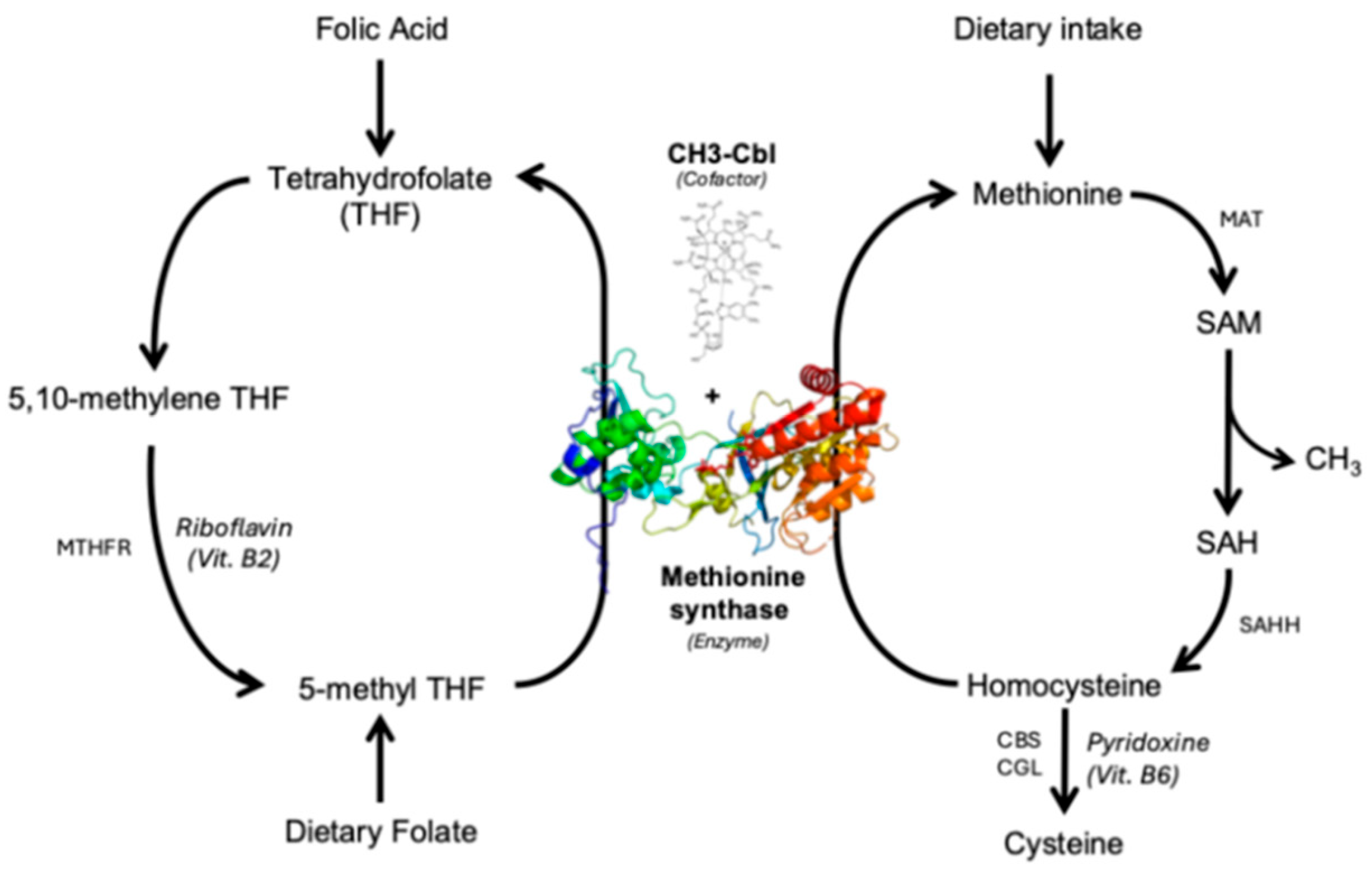
- Froese, D.S.; Fowler, B.; Baumgartner, M.R. Vitamin B(12), folate, and the methionine remethylation cycle-biochemistry, pathways, and regulation. J Inherit Metab Dis 2019, 42, 673–685. [Google Scholar] [CrossRef]
- Hunt, A.; Harrington, D.; Robinson, S. Vitamin B12 deficiency. BMJ 2014, 349, g5226. [Google Scholar] [CrossRef]
- Guillerme, J.; Feugray, G.; Girot, H.; Brunel, V.; Muraine, M.Q. Preliminary evaluation of the diagnostic performance of Roche Elecsys(R) active vitamin B12 versus total vitamin B12 for vitamin B12 deficiency screening. Ann Clin Biochem 2024, 61, 55–62. [Google Scholar] [CrossRef]
- Obeid, R.; Andres, E.; Ceska, R.; Hooshmand, B.; Gueant-Rodriguez, R.M.; Prada, G.I.; Slawek, J.; Traykov, L.; Ta Van, B.; Varkonyi, T.; et al. Diagnosis, Treatment and Long-Term Management of Vitamin B12 Deficiency in Adults: A Delphi Expert Consensus. J Clin Med 2024, 13. [Google Scholar] [CrossRef]
- Andres, E.; Serraj, K.; Zhu, J.; Vermorken, A.J. The pathophysiology of elevated vitamin B12 in clinical practice. QJM 2013, 106, 505–515. [Google Scholar] [CrossRef]
- Vollbracht, C.; McGregor, G.P.; Kraft, K. Supraphysiological vitamin B12 serum concentrations without supplementation: The pitfalls of interpretation. QJM 2020, 113, 619–620. [Google Scholar] [CrossRef] [PubMed]
- Hurkmans, E.G.E.; Deneer, R.; Nijziel, M.R.; Scharnhorst, V. Unexplained Elevated Vitamin B12: Consider Macro-B12. Int J Lab Hematol 2026. [Google Scholar] [CrossRef] [PubMed]
- Vitamin B12 Deficiency in over 16s: Diagnosis and Management; National Institute for Health and Care Excellence: Clinical Guidelines: London, 2024.
- Leleu, D.; Denimal, D. A case of functional vitamin B12 deficiency after recreational nitrous oxide use. Biochem Med 2024, 34, 010801. [Google Scholar] [CrossRef] [PubMed]
- Nexo, E.; Parkner, T. Vitamin B12-Related Biomarkers. Food Nutr Bull 2024, 45, S28–S33. [Google Scholar] [CrossRef]
- Devalia, V.; Hamilton, M.S.; Molloy, A.M.; British Committee for Standards in Haematology. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol 2014, 166, 496–513. [Google Scholar] [CrossRef]
- Fedosov, S.N.; Brito, A.; Miller, J.W.; Green, R.; Allen, L.H. Combined indicator of vitamin B12 status: Dodification for missing biomarkers and folate status and recommendations for revised cut-points. Clin Chem Lab Med 2015, 53, 1215–1225. [Google Scholar] [CrossRef]
- Smith, A.D.; Warren, M.J.; Refsum, H. Vitamin B(12). Adv Food Nutr Res 2018, 83, 215–279. [Google Scholar] [CrossRef]
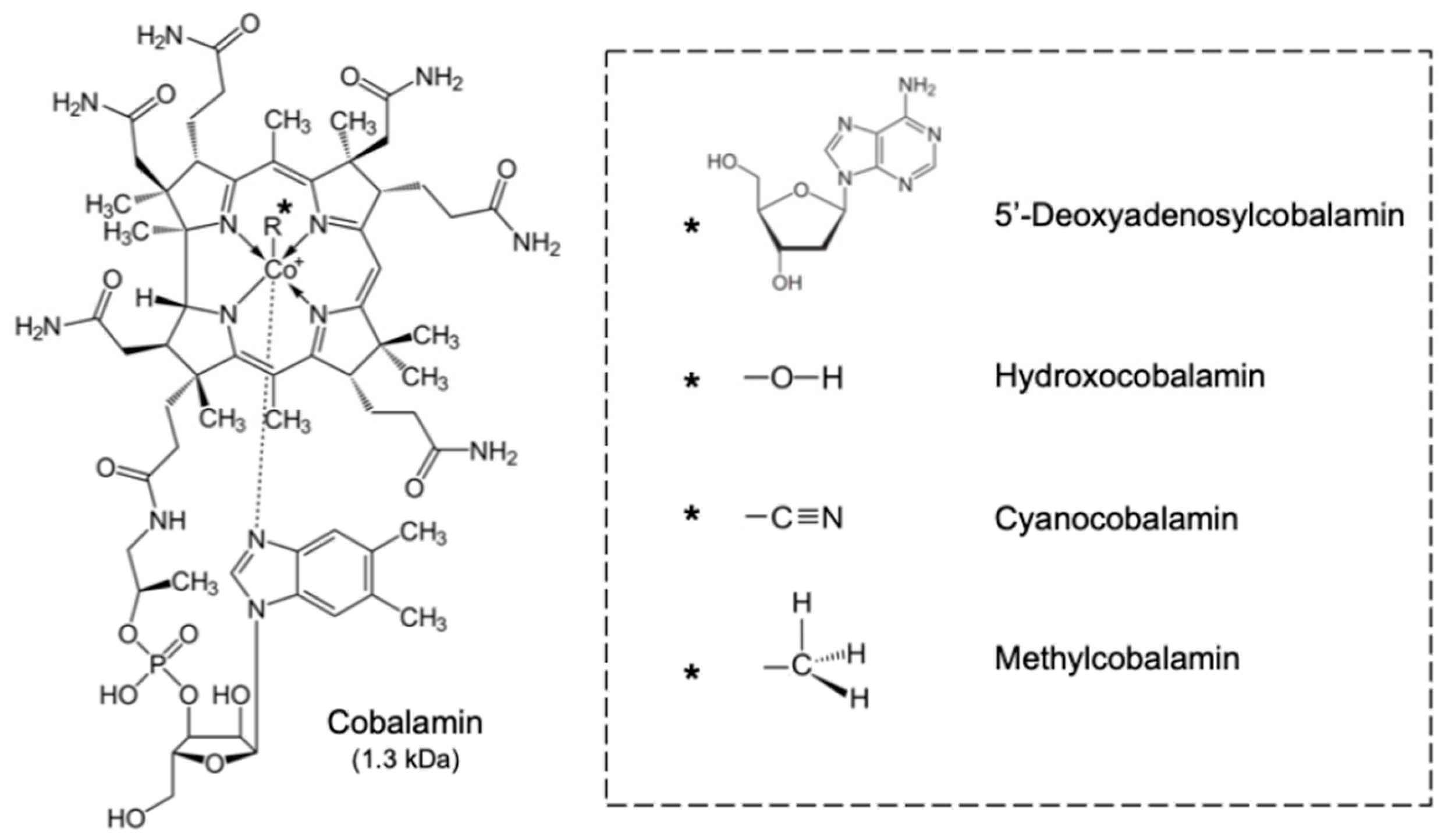
- Randaccio, L.; Geremia, S.; Demitri, N.; Wuerges, J. Vitamin B12: Unique metalorganic compounds and the most complex vitamins. Molecules 2010, 15, 3228–3259. [Google Scholar] [CrossRef]
- Bloch, J.S.; Ruetz, M.; Krautler, B.; Locher, K.P. Structure of the human transcobalamin beta domain in four distinct states. PLoS ONE 2017, 12, e0184932. [Google Scholar] [CrossRef]
- Obeid, R.; Fedosov, S.N.; Nexo, E. Cobalamin coenzyme forms are not likely to be superior to cyano- and hydroxyl-cobalamin in prevention or treatment of cobalamin deficiency. Mol Nutr Food Res 2015, 59, 1364–1372. [Google Scholar] [CrossRef] [PubMed]
- Mucha, P.; Kus, F.; Cysewski, D.; Smolenski, R.T.; Tomczyk, M. Vitamin B(12) Metabolism: A Network of Multi-Protein Mediated Processes. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- Quadros, E.V.; Rothenberg, S.P.; Pan, Y.C.; Stein, S. Purification and molecular characterization of human transcobalamin II. J Biol Chem 1986, 261, 15455–15460. [Google Scholar] [CrossRef] [PubMed]
- Wuerges, J.; Garau, G.; Geremia, S.; Fedosov, S.N.; Petersen, T.E.; Randaccio, L. Structural basis for mammalian vitamin B12 transport by transcobalamin. Proc Natl Acad Sci USA 2006, 103, 4386–4391. [Google Scholar] [CrossRef]
- Obeid, R. High Plasma Vitamin B12 and Cancer in Human Studies: A Scoping Review to Judge Causality and Alternative Explanations. Nutrients 2022, 14. [Google Scholar] [CrossRef]
- Moravcova, M.; Siatka, T.; Krcmova, L.K.; Matousova, K.; Mladenka, P.; Oemonom. Biological properties of vitamin B(12). Nutr Res Rev 2025, 38, 338–370. [Google Scholar] [CrossRef]
- Watanabe, F.; Bito, T. Vitamin B(12) sources and microbial interaction. Exp Biol Med 2018, 243, 148–158. [Google Scholar] [CrossRef]
- Wan, Z.; Zheng, J.; Zhu, Z.; Sang, L.; Zhu, J.; Luo, S.; Zhao, Y.; Wang, R.; Zhang, Y.; Hao, K.; et al. Intermediate role of gut microbiota in vitamin B nutrition and its influences on human health. Front Nutr 2022, 9, 1031502. [Google Scholar] [CrossRef]
- Basalamah, M.A.; Ibrahim, M.O.; Qutob, M.S.; Jazar, A.S.; Bakr, E.H.; Alazzeh, A.Y.; Al-Slaihat, A.H.; Azzeh, F.S. Vitamin B12 status among asymptomatic young adult females and its association with some anthropometric and biochemical parameters: A cross-sectional study from Makkah (cobalamin deficiency in young adult females). Medicine 2023, 102, e35838. [Google Scholar] [CrossRef] [PubMed]
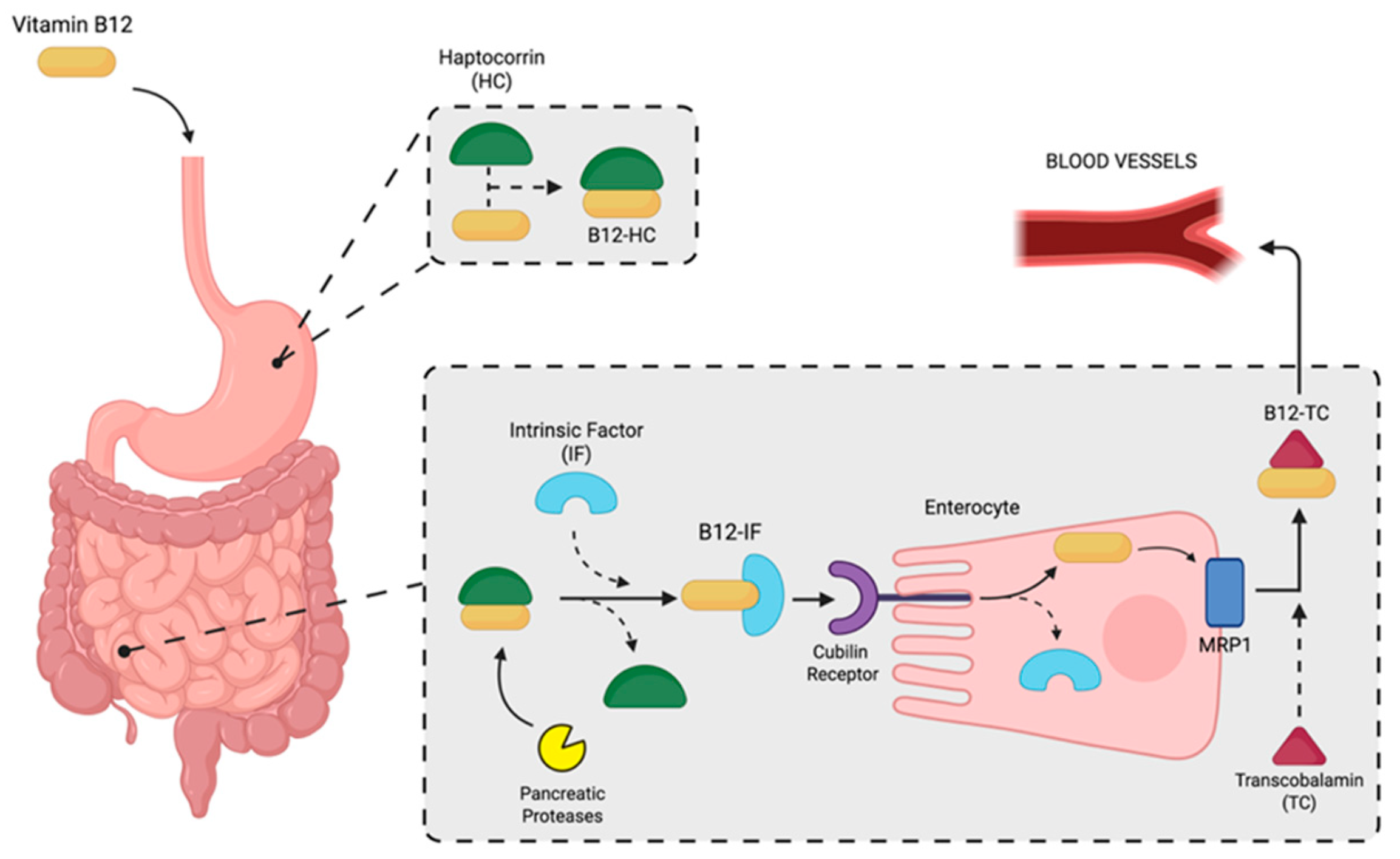
- Fedosov, S.N. Physiological and molecular aspects of cobalamin transport. Subcell Biochem 2012, 56, 347–367. [Google Scholar] [CrossRef] [PubMed]
- Gueant, J.L.; Gueant-Rodriguez, R.M.; Alpers, D.H. Vitamin B12 absorption and malabsorption. Vitam Horm 2022, 119, 241–274. [Google Scholar] [CrossRef] [PubMed]
- Gick, G.G.; Arora, K.; Sequeira, J.M.; Nakayama, Y.; Lai, S.C.; Quadros, E.V. Cellular uptake of vitamin B(12): Role and fate of TCblR/CD320, the transcobalamin receptor. Exp Cell Res 2020, 396, 112256. [Google Scholar] [CrossRef]
- Deme, J.C.; Hancock, M.A.; Xia, X.; Shintre, C.A.; Plesa, M.; Kim, J.C.; Carpenter, E.P.; Rosenblatt, D.S.; Coulton, J.W. Purification and interaction analyses of two human lysosomal vitamin B12 transporters: LMBD1 and ABCD4. Mol Membr Biol 2014, 31, 250–261. [Google Scholar] [CrossRef]
- Nexo, E.; Hoffmann-Lucke, E. Holotranscobalamin, a marker of vitamin B-12 status: Analytical aspects and clinical utility. Am J Clin Nutr 2011, 94, 359S–365S. [Google Scholar] [CrossRef]
- Pardo-Cabello, A.J.; Manzano-Gamero, V.; Puche-Canas, E. Vitamin B12: For more than just the treatment of megaloblastic anemia? Rev Clin Esp 2023, 223, 114–119. [Google Scholar] [CrossRef]
- Damayanti, D.; Jaceldo-Siegl, K.; Beeson, W.L.; Fraser, G.; Oda, K.; Haddad, E.H. Foods and Supplements Associated with Vitamin B(12) Biomarkers among Vegetarian and Non-Vegetarian Participants of the Adventist Health Study-2 (AHS-2) Calibration Study. Nutrients 2018, 10. [Google Scholar] [CrossRef]
- Denissen, K.F.M.; Heil, S.G.; Eussen, S.; Heeskens, J.P.J.; Thijs, C.; Mommers, M.; Smits, L.J.M.; van Dongen, M.; Dagnelie, P.C. Intakes of Vitamin B-12 from Dairy Food, Meat, and Fish and Shellfish Are Independently and Positively Associated with Vitamin B-12 Biomarker Status in Pregnant Dutch Women. J Nutr 2019, 149, 131–138. [Google Scholar] [CrossRef]
- Cifelli, C.J.; Agarwal, S.; Fulgoni, V.L., III. Association between Intake of Total Dairy and Individual Dairy Foods and Markers of Folate, Vitamin B(6) and Vitamin B(12) Status in the U.S. Population. Nutrients 2022, 14. [Google Scholar] [CrossRef]
- Huang, H.H.; Cohen, A.A.; Gaudreau, P.; Auray-Blais, C.; Allard, D.; Boutin, M.; Reid, I.; Turcot, V.; Presse, N. Vitamin B-12 Intake from Dairy but Not Meat Is Associated with Decreased Risk of Low Vitamin B-12 Status and Deficiency in Older Adults from Quebec, Canada. J Nutr 2022, 152, 2483–2492. [Google Scholar] [CrossRef] [PubMed]
- Risch, M.; Meier, D.W.; Sakem, B.; Medina Escobar, P.; Risch, C.; Nydegger, U.; Risch, L. Vitamin B12 and folate levels in healthy Swiss senior citizens: A prospective study evaluating reference intervals and decision limits. BMC Geriatr 2015, 15, 82. [Google Scholar] [CrossRef] [PubMed]
- Hinds, H.E.; Johnson, A.A.; Webb, M.C.; Graham, A.P. Iron, folate, and vitamin B12 status in the elderly by gender and ethnicity. J Natl Med Assoc 2011, 103, 870–877. [Google Scholar] [CrossRef] [PubMed]
- Sturmer, P.; Strathmann, E.A.; Liedtke, T.P.; Overmohle, C.; Rimbach, G.; Weber, K.S.; Lieb, W. Correlates of serum holo-Transcobalamin in the elderly general population. Eur J Nutr 2025, 64, 270. [Google Scholar] [CrossRef]
- Morkbak, A.L.; Hvas, A.M.; Milman, N.; Nexo, E. Holotranscobalamin remains unchanged during pregnancy. Longitudinal changes of cobalamins and their binding proteins during pregnancy and postpartum. Haematologica 2007, 92, 1711–1712. [Google Scholar] [CrossRef]
- Refsum, H.; Johnston, C.; Guttormsen, A.B.; Nexo, E. Holotranscobalamin and total transcobalamin in human plasma: Determination, determinants, and reference values in healthy adults. Clin Chem 2006, 52, 129–137. [Google Scholar] [CrossRef]
- Surendran, S.; Adaikalakoteswari, A.; Saravanan, P.; Shatwaan, I.A.; Lovegrove, J.A.; Vimaleswaran, K.S. An update on vitamin B12-related gene polymorphisms and B12 status. Genes Nutr 2018, 13, 2. [Google Scholar] [CrossRef]
- Guetterman, H.M.; Huey, S.L.; Knight, R.; Fox, A.M.; Mehta, S.; Finkelstein, J.L. Vitamin B-12 and the Gastrointestinal Microbiome: A Systematic Review. Adv Nutr 2022, 13, 530–558. [Google Scholar] [CrossRef]
- Kelleher, B.P.; Broin, S.D. Microbiological assay for vitamin B12 performed in 96-well microtitre plates. J Clin Pathol 1991, 44, 592–595. [Google Scholar] [CrossRef]
- Stuart, J.; Sklaroff, S.A. Rapid microbiological assay of serum vitamin B 12 by electronic counter. J Clin Pathol 1966, 19, 46–50. [Google Scholar] [CrossRef]
- Davis, R.E.; Moulton, J.; Kelly, A. An automated microbiological method for the measurement of vitamin B12. J Clin Pathol 1973, 26, 494–498. [Google Scholar] [CrossRef]
- Berg, T.M.; de Vries, J.A.; van Gasteren, C.J.; Meeuwissen, H.; Stevens, G.A.; Verbon, F.J. Comparative studies on the microbiological vitamin B12 assay at two laboratories. Appl Environ Microbiol 1976, 31, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, B.P.; Scott, J.M.; O’Broin, S.D. Use of beta-lactamase to hydrolyse interfering antibiotics in vitamin B12 microbiological assay using Lactobacillus leichmannii. Clin Lab Haematol 1990, 12, 87–95. [Google Scholar] [CrossRef]
- Endres, D.B.; Painter, K.; Niswender, G.D. A solid-phase radioimmunoassay for vitamin B12 in serum, with use of radioiodinated tyrosine methyl ester of vitamin B12. Clin Chem 1978, 24, 460–465. [Google Scholar] [CrossRef]
- Frenkel, E.P.; White, J.D.; Reisch, J.S.; Sheehan, R.G. Comparison of two methods for radioassay of vitamin B12 in serum. Clin Chem 1973, 19, 1357–1360. [Google Scholar] [CrossRef]
- Loikas, S.; Lopponen, M.; Suominen, P.; Moller, J.; Irjala, K.; Isoaho, R.; Kivela, S.L.; Koskinen, P.; Pelliniemi, T.T. RIA for serum holo-transcobalamin: Method evaluation in the clinical laboratory and reference interval. Clin Chem 2003, 49, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Ulleland, M.; Eilertsen, I.; Quadros, E.V.; Rothenberg, S.P.; Fedosov, S.N.; Sundrehagen, E.; Orning, L. Direct assay for cobalamin bound to transcobalamin (holo-transcobalamin) in serum. Clin Chem 2002, 48, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Selva Kumar, L.S.; Thakur, M.S. Competitive immunoassay for analysis of vitamin B(12). Anal Biochem 2011, 418, 238–246. [Google Scholar] [CrossRef]
- Orning, L.; Rian, A.; Campbell, A.; Brady, J.; Fedosov, S.N.; Bramlage, B.; Thompson, K.; Quadros, E.V. Characterization of a monoclonal antibody with specificity for holo-transcobalamin. Nutr Metab 2006, 3, 3. [Google Scholar] [CrossRef]
- Merrigan, S.D.; Owen, W.E.; Straseski, J.A. Performance characteristics of the ARCHITECT Active-B12 (Holotranscobalamin) assay. Clin Lab 2015, 61, 283–288. [Google Scholar] [CrossRef]
- Heil, S.G.; Bodenburg, P.; Findeisen, P.; Luebcke, S.; Sun, Y.; de Rijke, Y.B. Multicentre evaluation of the Roche Elecsys(R) Active B(12) (holotranscobalamin) electro-chemiluminescence immunoassay. Ann Clin Biochem 2019, 56, 662–667. [Google Scholar] [CrossRef]
- Ispir, E.; Serdar, M.A.; Ozgurtas, T.; Gulbahar, O.; Akin, K.O.; Yesildal, F.; Kurt, I. Comparison of four automated serum vitamin B12 assays. Clin Chem Lab Med 2015, 53, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Ma, X.; Zou, Y.; Qiu, L.; Wang, D.; Tang, Y.; Cao, Y.; Yu, S.; Cheng, X. Total serum vitamin B12 (cobalamin) LC-MS/MS assay as an arbiter of clinically discordant immunoassay results. Clin Chem Lab Med 2023, 61, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Obeid, R.; Geisel, J.; Herrmann, W. Comparison of two methods for measuring methylmalonic acid as a marker for vitamin B12 deficiency. Diagnosis 2015, 2, 67–72. [Google Scholar] [CrossRef]
- Ma, X.; Zou, Y.; Tang, Y.; Wang, D.; Zhou, W.; Yu, S.; Qiu, L. High-throughput analysis of total homocysteine and methylmalonic acid with the efficiency to separate succinic acid in serum and urine via liquid chromatography tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2022, 1193, 123135. [Google Scholar] [CrossRef]
- Harrington, D.J.; Stevenson, E.; Sobczynska-Malefora, A. The application and interpretation of laboratory biomarkers for the evaluation of vitamin B12 status. Ann Clin Biochem 2025, 62, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Trad, F.M.; AlHamad, T.; Younes, N.; Abunasser, S.; Younes, S.; Nizamuddin, P.B.; Chaar, D.E.; Salameh, I.M.; Al-Dewik, N.I.; Laiwattanapaisal, W.; et al. Accre 8 emerging point of care CLIA system for vitamin B12 assessment compared with three established assays. Sci Rep 2025, 15, 13328. [Google Scholar] [CrossRef]
- Soares, M.S.; Vidal, M.; Santos, N.F.; Costa, F.M.; Marques, C.; Pereira, S.O.; Leitao, C. Immunosensing Based on Optical Fiber Technology: Recent Advances. Biosensors 2021, 11. [Google Scholar] [CrossRef]
- Rohilla, R.; Kaur, A.; Rani, S.; Prabhakar, N. Ultrasensitive detection of holoTC for analysis of Vitamin B12 levels using Ag(2)MoO(4) deposited PEDOT sensing platform. Biosens Bioelectron 2025, 267, 116783. [Google Scholar] [CrossRef]
- Bissen, A.; Yunussova, N.; Myrkhiyeva, Z.; Salken, A.; Tosi, D.; Bekmurzayeva, A. Unpacking the packaged optical fiber bio-sensors: Understanding the obstacle for biomedical application. Front Bioeng Biotechnol 2024, 12, 1401613. [Google Scholar] [CrossRef]
- Mindray Vitamin B12 CLIA IFU 2015.
- DiaSorin LIAISON Vitamin B12 (CLIA).
- Snibe MAGLUMI Vitamin B12 (CLIA) IFU 2022.
- TOSOH AIA-PACK B12 IFU 2024.
- VIDAS Vitamin B12 total IFU 2024.
- Abbott Alinity Vitamin B12 IFU 2017.
- Siemens Healthineers. Atellica IM Vitamin B12 (VB12) IFU 2019.
- Elecsys® Vitamin B12 II. Roche Diagnostics: Mannheim, Germany. Available online: https://elabdoc-prod.roche.com/eLD/web/global/en/products/CPS_000536 (accessed on 25 January 2026).
- Access Vitamin B12: Instruction for Use; Beckman Coulter, Inc.: Brea, CA, USA. Document n. D12001.B. Available online: https://www.beckmancoulter.com/products/part?partnumber=33000 (accessed on 25 January 2026).
- Abbott Alinity Active B12 IFU 2025.
- Active-B12 Assay (Holotranscobalamin) — Laboratory Diagnostics. Siemens Healthineers. Available online: https://www.siemens-healthineers.com/sk/laboratory-diagnostics/assays-by-diseases-conditions/anemia/active-b12-assay (accessed on 23 January 2026).
- Diagnostics Roche Elecsys®. Active B12 (holotranscobalamin) assay: Method Sheet; Roche Diagnostics. Available online: https://elabdoc-prod.roche.com/eLD/web/global/en/products/CPS_000430 (accessed on 23 January 2026).
- Coulter, B. Access Active-B12 (holotranscobalamin) assay: Instructions for Use; Document no. D12001; Beckman Coulter: Brea, CA, USA. Available online: https://www.beckmancoulter.com/products/part?partnumber=33000&utm_ (accessed on 23 January 2026).
- Thorpe, S.J.; Rigsby, P.; Roberts, G.; Lee, A.; Hamilton, M.; Craig, D. An International Standard for holotranscobalamin (holoTC): International collaborative study to assign a holoTC value to the International Standard for vitamin B12 and serum folate. Clin Chem Lab Med 2016, 54, 1467–1472. [Google Scholar] [CrossRef] [PubMed]
- Jassam, N.; Bancroft, T.; Luvai, A.; Mbagaya, W.; Barth, J.H. Vitamin B12 reference intervals on Beckman, Roche and Siemens analytical platforms. Ann Clin Biochem 2023, 60, 417–422. [Google Scholar] [CrossRef]
- Harrington, D.J. Laboratory assessment of vitamin B12 status. J Clin Pathol 2017, 70, 168–173. [Google Scholar] [CrossRef]
- EFLM Cobalamin biological variability. Available online: https://biologicalvariation.eu/api/search/by_analyte?format=html&query=Cobalamin (accessed on 28 February 2026).
- Sobczynska-Malefora, A.; Delvin, E.; McCaddon, A.; Ahmadi, K.R.; Harrington, D.J. Vitamin B(12) status in health and disease: A critical review. Diagnosis of deficiency and insufficiency - clinical and laboratory pitfalls. Crit Rev Clin Lab Sci 2021, 58, 399–429. [Google Scholar] [CrossRef]
- Wolffenbuttel, B.H.R.; Muller Kobold, A.C.; Sobczynska-Malefora, A.; Harrington, D.J. Macro-B(12) masking B(12) deficiency. BMJ Case Rep 2022, 15. [Google Scholar] [CrossRef]
- Andersen, S.L.; Hansen, A.B.; Hindersson, P.; Andersen, L.; Christensen, P.A. Vitamin B12 reference intervals. Dan Med J 2023, 70. [Google Scholar]
- Jiang, W.; Men, S.; Wen, X.; Yuan, X.; Pu, D.; Liu, X.; Jia, X.; Wang, C. A preliminary study for the establishment of a reference interval for vitamin B12 in China after performance verification of a second-generation ECLIA kit. J Clin Lab Anal 2020, 34, e23165. [Google Scholar] [CrossRef]
- Sole-Enrech, G.; San-Jose, P.; Aliste-Fernandez, M.; Cano-Corres, R.; Claver-Belver, N.; Palencia-Dominguez, J.; Teodoro-Marin, V.; Serrano-Fuentes, R.; Ustrell-Abanco, R.; Campos-Barreda, F.; et al. Vitamin B12 and folate levels in a healthy population: Establishing reference intervals. Clin Chem Lab Med 2019, 57, e173–e175. [Google Scholar] [CrossRef]
- Sobczynska-Malefora, A.; Katayev, A.; Steed, D.; O’Logbon, J.; Crook, M.; Harrington, D.J. Age- and ethnicity-related reference intervals for serum vitamin B(12). Clin Biochem 2023, 111, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Alpdemir, M.; Alpdemir, M.F. Determination of Reference Range for B12 and Folate Levels According to Laboratory Data in an Adult Population. Bagcilar Med. Bull. 2020, 5, 160–165. [Google Scholar] [CrossRef]
- Gavars, D.; et al. Extraction of B12 Reference Intervals from a Large Amount of General Patient Data. Proc. Latv. Acad. Sci. Sect. B Nat. Exact Appl. Sci. 2022, 76, 333–337. [Google Scholar] [CrossRef]
- Abildgaard, A.; Knudsen, C.S.; Hoejskov, C.S.; Greibe, E.; Parkner, T. Reference intervals for plasma vitamin B12 and plasma/serum methylmalonic acid in Danish children, adults and elderly. Clin Chim Acta 2022, 525, 62–68. [Google Scholar] [CrossRef]
- Bianciardi, S.; Tesi, G.; Cerutti, H.; Cartocci, A.; Guerranti, R.; Silvestrini, C.; Gori, S.; Bandini, T.; Brogi, A.; Leoncini, R. Validation, performance, and reliability of two automated tests for vitamin B12 and folate assay. J Public Health Res 2023, 12, 22799036231217803. [Google Scholar] [CrossRef]
- Plebani, M.; Lippi, G. Standardization and harmonization in laboratory medicine: Not only for clinical chemistry measurands. Clin Chem Lab Med 2023, 61, 185–187. [Google Scholar] [CrossRef]
- Cesana, B.M.; da Molin, S.; Jassam, N.; Barth, J.H.; Buoro, S.; Tosi, M.; Zuccotti, G.; Marcovina, S.; Ferraro, S. Assessing the harmonization of current total vitamin B12 measurement methods: Relevance and implications. Clin Chem Lab Med 2025, 63, 2433–2441. [Google Scholar] [CrossRef]
- Tufail, N.; Kataria, M.; Chaudhary, A.J.; Dhillon, R.A.; Asif Naveed, M.; Mohsin, S. Comparing Holotranscobalamin and Total Vitamin B12 in Diagnosing Vitamin B12 Deficiency in Megaloblastic Anemia Patients. Cureus 2024, 16, e71278. [Google Scholar] [CrossRef]
- Niklewicz, A.; Hannibal, L.; Warren, M.; Ahmadi, K.R. A systematic review and meta-analysis of functional vitamin B12 status among adult vegans. Nutr Bull 2024, 49, 463–479. [Google Scholar] [CrossRef]
- Ozdemir, S.; Demirtas, S. Are vitamin B-12 measurements adequate for evaluating its deficiency in individuals? Asia Pac J Clin Nutr 2025, 34, 232–239. [Google Scholar] [CrossRef]
| Authors/Year | Individuals | Population | Platform | Literature RI | Manufacturers RI |
|---|---|---|---|---|---|
| Andersen SL et al., 2023 [92] |
41,091 Healthy donors 129 (60 m; 69 f) |
Danish Healthy adults |
Abbott Alinity Roche Cobas 6000 Siemens Atellica IM |
Healthy donors 168-553 pmol/L 202-641 pmol/L 211-551 pmol/L General population 133-541 pmol/L 172-619 pmol/L 182-553 pmol/L |
138-652 pmol/L 145-569 pmol/L 156-672 pmol/L |
| Jiang W et al., 2020 [93] |
389 (198 m; 191 f) |
Chinese Healthy adults (21-80 y.o.) |
Roche Cobas E602 | 250.8-957.1 pg/mL | 197-771 pg/mL |
| Solé-Enrech G et al., 2019 [94] |
123 (59 m; 67 f) |
Spanish Healthy adults |
Roche Cobas 8000 e801 | 150-695 pg/mL (111-513 pmol/L) | 197-771 pg/mL (145-569 pmol/L) |
| Sobczyńska-Malefora A et al., 2023 [95] |
35,988 (11,642 m; 24,346 f) |
White 18,508 Black/British Black: 10,182 Asian/British Asian: 2595 |
ARCHITECT Abbott | 0-1 years: 159-1025 pmol/L 2-5 years 276-1102 pmol/L 6-9 years 245-798 pmol/L 10-13 years 187-643 pmol/L >13 years, Black 166-805 pmol/L > 13 years, white/asian 134-511 pmol/L |
138-652 pmol/L |
| Alpdemir M. et al., 2020 [96] |
36,284 (10,795 m; 25,489 f) |
Turkish | Abbott ARCHITECT i2000sr | 97-397 pmol/L |
138-654 pmol/L |
| Gavars. et al., 2022 [97] |
132,379 | Latvian | Elecsys 2010 (2004-2012) Roche Cobas e 2010b (2012-2017) Roche Cobas e 801 (2017-2022) |
196–942 pg/mL | 180–900 pg/mL (133–664 pmol/L) 191–663 pg/mL (141–489 pmol/L) 197-771 pg/mL (146-569 pmol/L) |
| Abildgaard A. et al., 2022 [98] |
310 (158 m;152 f) (147 adults 18<65) 163 elderly >=65) |
Danish | Siemens ADVIA Centaur XPT | 0–<1 y.o.: 180–1400 pmol/L 1–<12 y.o.: 260–1200 pmol/L 12–<18 y.o.: 200–800 pmol/L 18–<65 y.o.: 200–600 pmol/L ≥ 65 y.o.: 200–600 pmol/L |
/ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




