Submitted:
09 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Huh7 Cell Culture, Transduction, and Infection Experiments
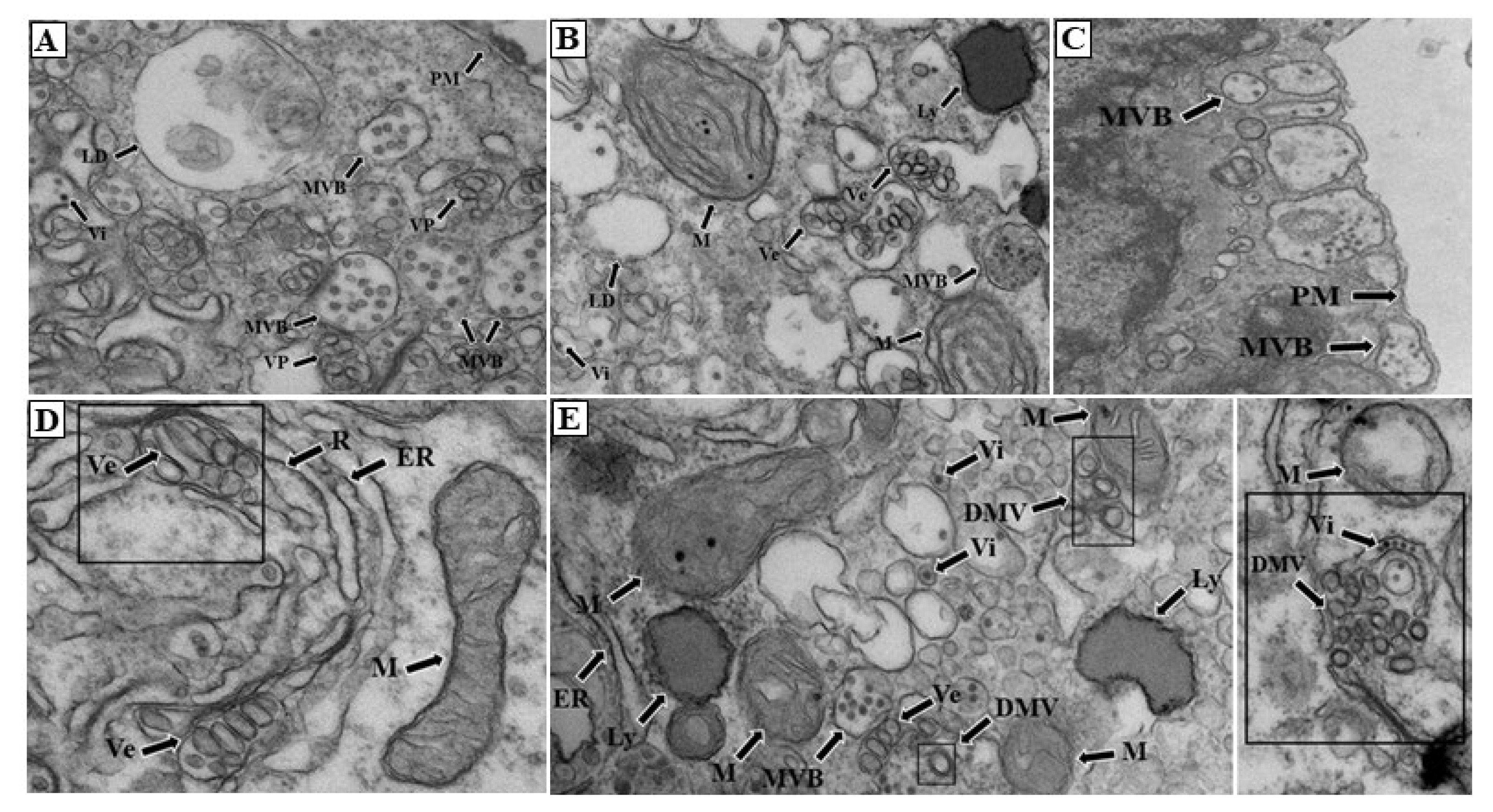
Transmission Electron Microscopy (TEM)
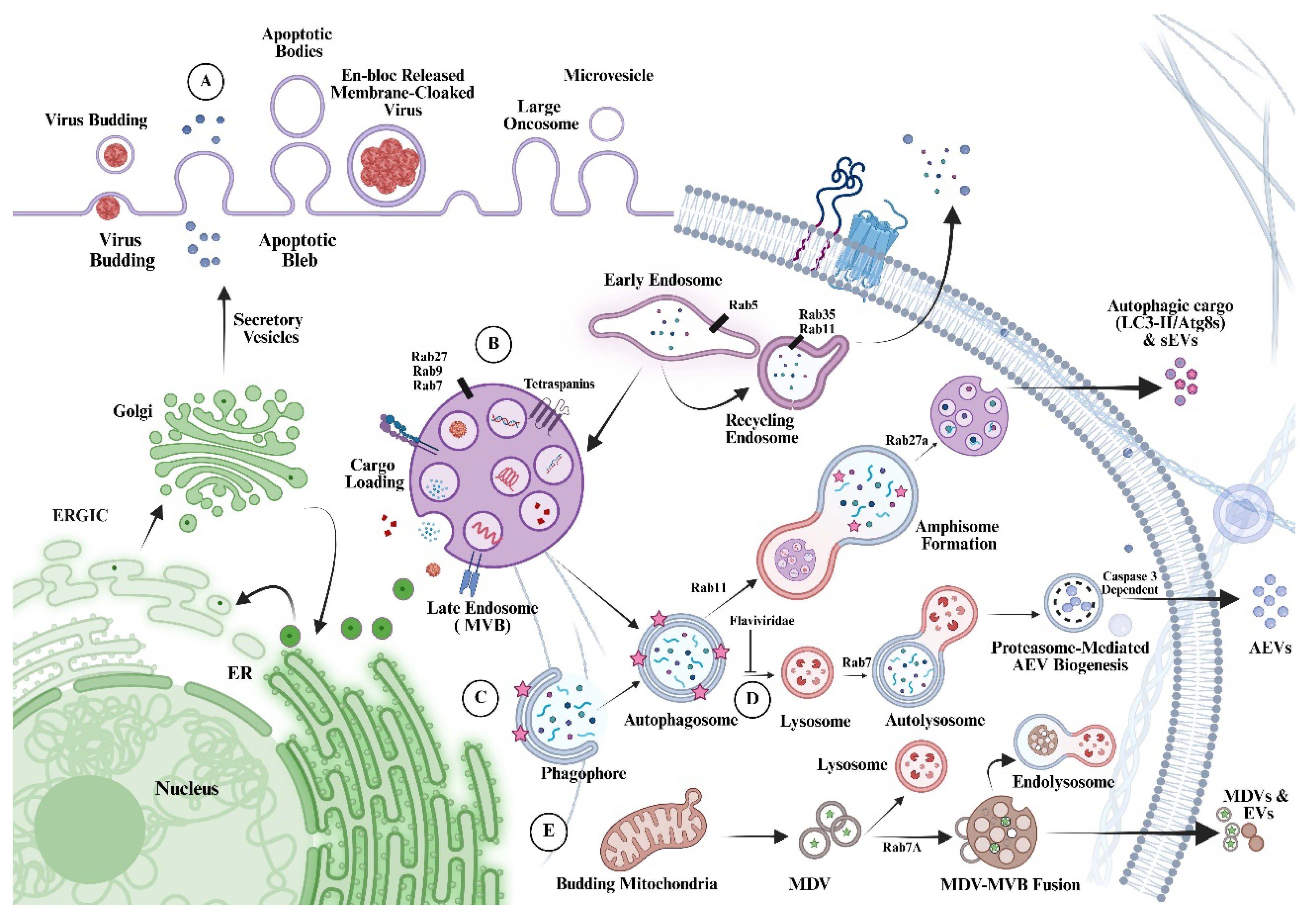
Results and Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Federico, M. The Potential of Extracellular Vesicle-Mediated Spread of Self-Amplifying Rna and a Way to Mitigate It. Int J Mol Sci 2025, 26. [Google Scholar] [CrossRef]
- Kumari, S.; Banerjee, A. Extracellular Vesicles: The Double-Edged Sword in Viral Infections. mBio 2026, 17, e0331625. [Google Scholar] [CrossRef]
- Carvalho Ferraz, L.; Pereira, P.; Ferreira, J. V. Molecular Mechanisms of Extracellular Vesicle Biogenesis and Their Impact on the Design of Custom Evs. Adv Healthc Mater 2025, 14, e2501349. [Google Scholar] [CrossRef]
- Pedrioli, G.; Paganetti, P. Hijacking Endocytosis and Autophagy in Extracellular Vesicle Communication: Where the inside Meets the Outside. Front Cell Dev Biol 2020, 8, 595515. [Google Scholar] [CrossRef] [PubMed]
- Krylova, S. V.; Feng, D. The Machinery of Exosomes: Biogenesis, Release, and Uptake. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Butler, L. R.; Singh, N.; Marnin, L.; Valencia, L. M.; O'Neal, A. J.; Paz, F. E. C.; Shaw, D. K.; Chavez, A. S. O.; Pedra, J. H. F. The Role of Rab27 in Tick Extracellular Vesicle Biogenesis and Pathogen Infection. Parasit Vectors 2024, 17, 57. [Google Scholar] [CrossRef] [PubMed]
- Blanc, L.; Vidal, M. New Insights into the Function of Rab Gtpases in the Context of Exosomal Secretion. Small GTPases 2018, 9, 95–106. [Google Scholar] [CrossRef]
- MacDonald, C.; Buchkovich, N. J.; Stringer, D. K.; Emr, S. D.; Piper, R. C. Cargo Ubiquitination Is Essential for Multivesicular Body Intralumenal Vesicle Formation. EMBO Rep 2012, 13, 331–8. [Google Scholar] [CrossRef]
- Hyenne, V.; Labouesse, M.; Goetz, J. G. The Small Gtpase Ral Orchestrates Mvb Biogenesis and Exosome Secretion. Small GTPases 2018, 9, 445–51. [Google Scholar] [CrossRef]
- Li, S. P.; Lin, Z. X.; Jiang, X. Y.; Yu, X. Y. Exosomal Cargo-Loading and Synthetic Exosome-Mimics as Potential Therapeutic Tools. Acta Pharmacol Sin 2018, 39, 542–51. [Google Scholar] [CrossRef]
- Wu, H.; Voeltz, G. K. Reticulon-3 Promotes Endosome Maturation at Er Membrane Contact Sites. Dev Cell 2021, 56, 52–66 e7. [Google Scholar] [CrossRef]
- Bare, Y.; Defourny, K.; Bretou, M.; Van Niel, G.; Nolte-'t Hoen, E.; Gaudin, R. The Endoplasmic Reticulum as a Cradle for Virus and Extracellular Vesicle Secretion. Trends Cell Biol 2025, 35, 282–93. [Google Scholar] [CrossRef]
- Wozniak, A. L.; Adams, A.; King, K. E.; Dunn, W.; Christenson, L. K.; Hung, W. T.; Weinman, S. A. The Rna Binding Protein Fmr1 Controls Selective Exosomal Mirna Cargo Loading during Inflammation. J Cell Biol 2020, 219. [Google Scholar] [CrossRef] [PubMed]
- King, K. E.; Ghosh, P.; Wozniak, A. L. Trim25 Dictates Selective Mirna Loading into Extracellular Vesicles during Inflammation. Sci Rep 2023, 13, 22952. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J. V.; da Rosa Soares, A.; Ramalho, J.; Maximo Carvalho, C.; Cardoso, M. H.; Pintado, P.; Carvalho, A. S.; Beck, H. C.; Matthiesen, R.; Zuzarte, M.; Girao, H.; van Niel, G.; Pereira, P. Lamp2a Regulates the Loading of Proteins into Exosomes. Sci Adv 2022, 8, eabm1140. [Google Scholar] [CrossRef] [PubMed]
- Fader, C. M.; Colombo, M. I. Autophagy and Multivesicular Bodies: Two Closely Related Partners. Cell Death Differ 2009, 16, 70–8. [Google Scholar] [CrossRef]
- Mao, K.; Huo, F.; Wei, H.; Wang, F.; Xiong, S.; Fu, Y. Autophagic Extracellular Vesicles (Aevs) Are Distinct from Exosomes and Play Crucial Roles in Viral Infections. Nat Commun 2025, 17, 1095. [Google Scholar] [CrossRef]
- Wang, J.; Barr, M. M.; Wehman, A. M. Extracellular Vesicles. Genetics 2024, 227. [Google Scholar]
- Leidal, A. M.; Huang, H. H.; Marsh, T.; Solvik, T.; Zhang, D.; Ye, J.; Kai, F.; Goldsmith, J.; Liu, J. Y.; Huang, Y. H.; Monkkonen, T.; Vlahakis, A.; Huang, E. J.; Goodarzi, H.; Yu, L.; Wiita, A. P.; Debnath, J. The Lc3-Conjugation Machinery Specifies the Loading of Rna-Binding Proteins into Extracellular Vesicles. Nat Cell Biol 2020, 22, 187–99. [Google Scholar] [CrossRef]
- Diosa-Toro, M.; Prasanth, K. R.; Bradrick, S. S.; Garcia Blanco, M. A. Role of Rna-Binding Proteins during the Late Stages of Flavivirus Replication Cycle. Virol J 2020, 17, 60. [Google Scholar] [CrossRef]
- Wilson, A.; McCormick, C. Reticulophagy and Viral Infection. Autophagy 2025, 21, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, Y.; Li, Y.; Chen, X.; Wang, X.; Wang, X.; Yu, S. The Dual Role of Autophagy in the Pathogenesis of Arboviruses with a Focus on Jev and Denv. Rev Med Virol 2025, 35, e70073. [Google Scholar] [CrossRef] [PubMed]
- Randall, G. Lipid Droplet Metabolism during Dengue Virus Infection. Trends Microbiol 2018, 26, 640–42. [Google Scholar] [CrossRef]
- Cloherty, A. P. M.; Olmstead, A. D.; Ribeiro, C. M. S.; Jean, F. Hijacking of Lipid Droplets by Hepatitis C, Dengue and Zika Viruses-from Viral Protein Moonlighting to Extracellular Release. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Fang, D.; Shan, H.; Xiao, B.; Wei, D.; Ouyang, Y.; Huo, L.; Zhang, Z.; Wu, Y.; Zhang, R.; Kang, T.; Gao, Y. The Assembly of Rab22a/Tmem33/Rtn4 Initiates a Secretory Er-Phagy Pathway. Cell Discov 2025, 11, 41. [Google Scholar] [CrossRef]
- Yu, L. A New Route for Ev Biogenesis. Cell Res 2023, 33, 87–88. [Google Scholar] [CrossRef]
- Horbay, R.; Syrvatka, V.; Bedzay, A.; van der Merwe, M.; Burger, D.; Beug, S. T. From Mitochondria to Immunity: The Emerging Roles of Mitochondria-Derived Vesicles and Small Extracellular Vesicles in Cellular Communication and Disease. J Extracell Vesicles 2025, 14, e70192. [Google Scholar] [CrossRef]
- Wolff, G.; Melia, C. E.; Snijder, E. J.; Barcena, M. Double-Membrane Vesicles as Platforms for Viral Replication. Trends Microbiol 2020, 28, 1022–33. [Google Scholar] [CrossRef]
- Bordanaba-Florit, G.; Royo, F.; Kruglik, S. G.; Falcon-Perez, J. M. Using Single-Vesicle Technologies to Unravel the Heterogeneity of Extracellular Vesicles. Nat Protoc 2021, 16, 3163–85. [Google Scholar] [CrossRef]
- Tran, H. L.; Zheng, W.; Issadore, D. A.; Im, H.; Cho, Y. K.; Zhang, Y.; Liu, D.; Liu, Y.; Li, B.; Liu, F.; Wong, D. T. W.; Sun, J.; Qian, K.; He, M.; Wan, M.; Zeng, Y.; Cheng, K.; Huang, T. J.; Chiu, D. T.; Lee, L. P.; Zheng, L.; Godwin, A. K.; Kalluri, R.; Soper, S. A.; Hu, T. Y. Extracellular Vesicles for Clinical Diagnostics: From Bulk Measurements to Single-Vesicle Analysis. ACS Nano 2025, 19, 28021–109. [Google Scholar] [CrossRef]
- Li, J.; Abosmaha, E.; Coffin, C. S.; Labonte, P.; Bukong, T. N. Reticulon-3 Modulates the Incorporation of Replication Competent Hepatitis C Virus Molecules for Release inside Infectious Exosomes. PLoS One 2020, 15, e0239153. [Google Scholar] [CrossRef]
- Bitazar, R.; Asaba, C. N.; Shegefti, S.; Noumi, T.; Van Grevenynghe, J.; Islam, S. T.; Labonte, P.; Bukong, T. N. Integrative Mechanistic Studies Identify Reticulon-3 as a Critical Modulator of Infectious Exosome-Driven Dengue Pathogenesis. Viruses 2025, 17. [Google Scholar] [CrossRef]
- Joshi, B. S.; de Beer, M. A.; Giepmans, B. N. G.; Zuhorn, I. S. Endocytosis of Extracellular Vesicles and Release of Their Cargo from Endosomes. ACS Nano 2020, 14, 4444–55. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



