Submitted:
10 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
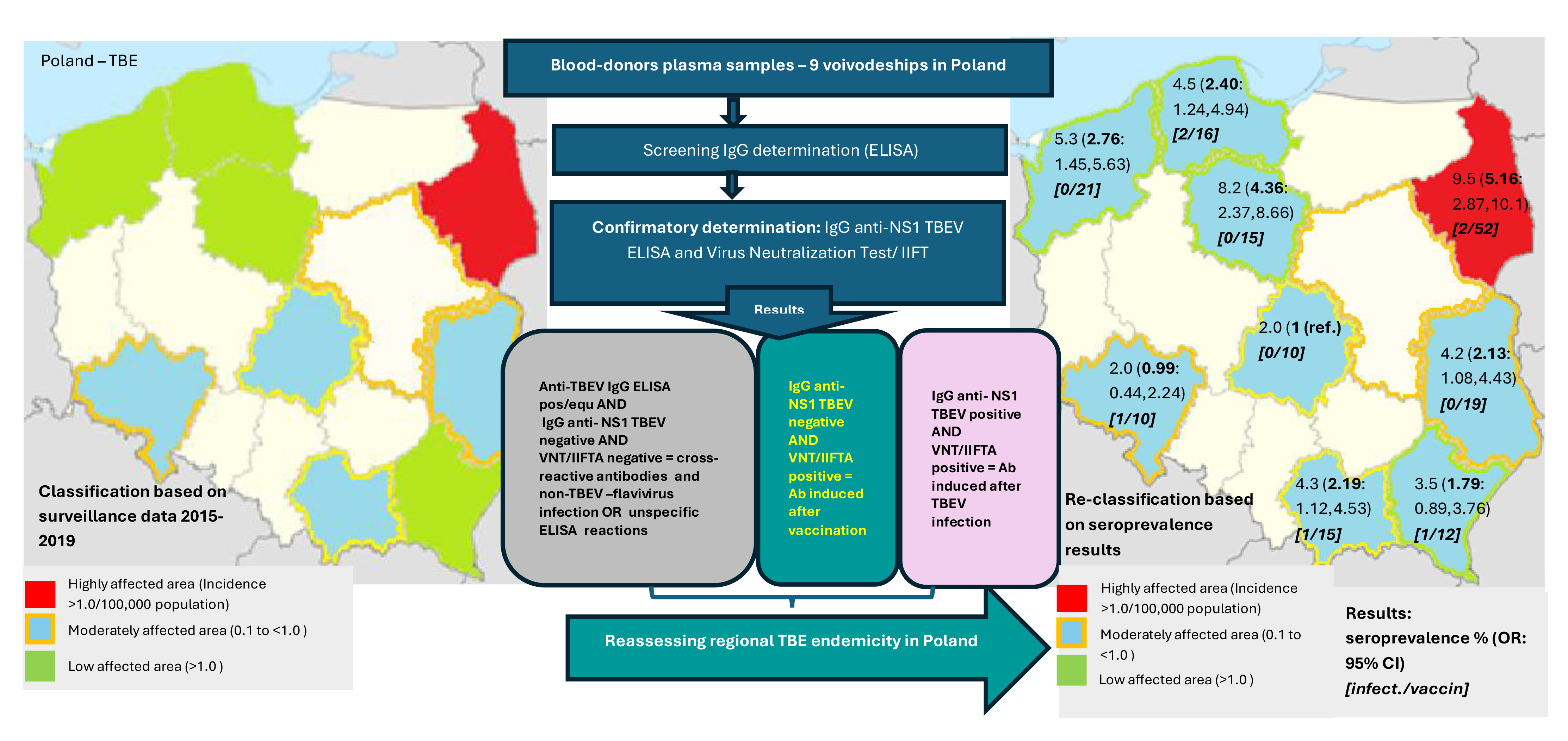
2.1. Selection of Voivodeships
- Highly affected: Podlaskie, Warmińsko-Mazurskie (>1.0 per 100,000 population)
- Moderately affected: Dolnośląskie, Łódzkie, Mazowieckie, Lubelskie, Opolskie, Małopolskie, and Świętokrzyskie. In these regions, with a five-year TBE incidence ranging from sse population, TBE cases are reported annually, and virus has been sporadically detected in environmental studies [7]. One possible explanation is the presence of smaller administrative units within these voivodeships with higher local incidence, despite an overall voivodeship-level incidence above 1 per 100,000.
- Low affected: Zachodniopomorskie, Pomorskie, Kujawsko-Pomorskie, Lubuskie, Podkarpackie, Śląskie, and Wielkopolskie. In these regions, only sporadic TBE cases are reported, with a mean five-year incidence below 0.1 per 100,000 population; many of these cases are likely associated with travel to more highly affected areas.
2.2. Study Population and Design
2.3. Sample Size Calculation
- Highly affected areas: expected seroprevalence of 4%, with an acceptable confidence interval width of 3% (e.g. 2.5%–5.5%), resulting in an estimated sample size of 688.
- Moderately and low-affected areas: expected seroprevalence of 1.5%, with an acceptable confidence interval width of 1% (e.g. 1.0%–2.0%), resulting in an estimated sample size of 2,346.
2.4. Screening Assays
2.5. Confirmatory Assays
2.6. Statistical Analysis
3. Results
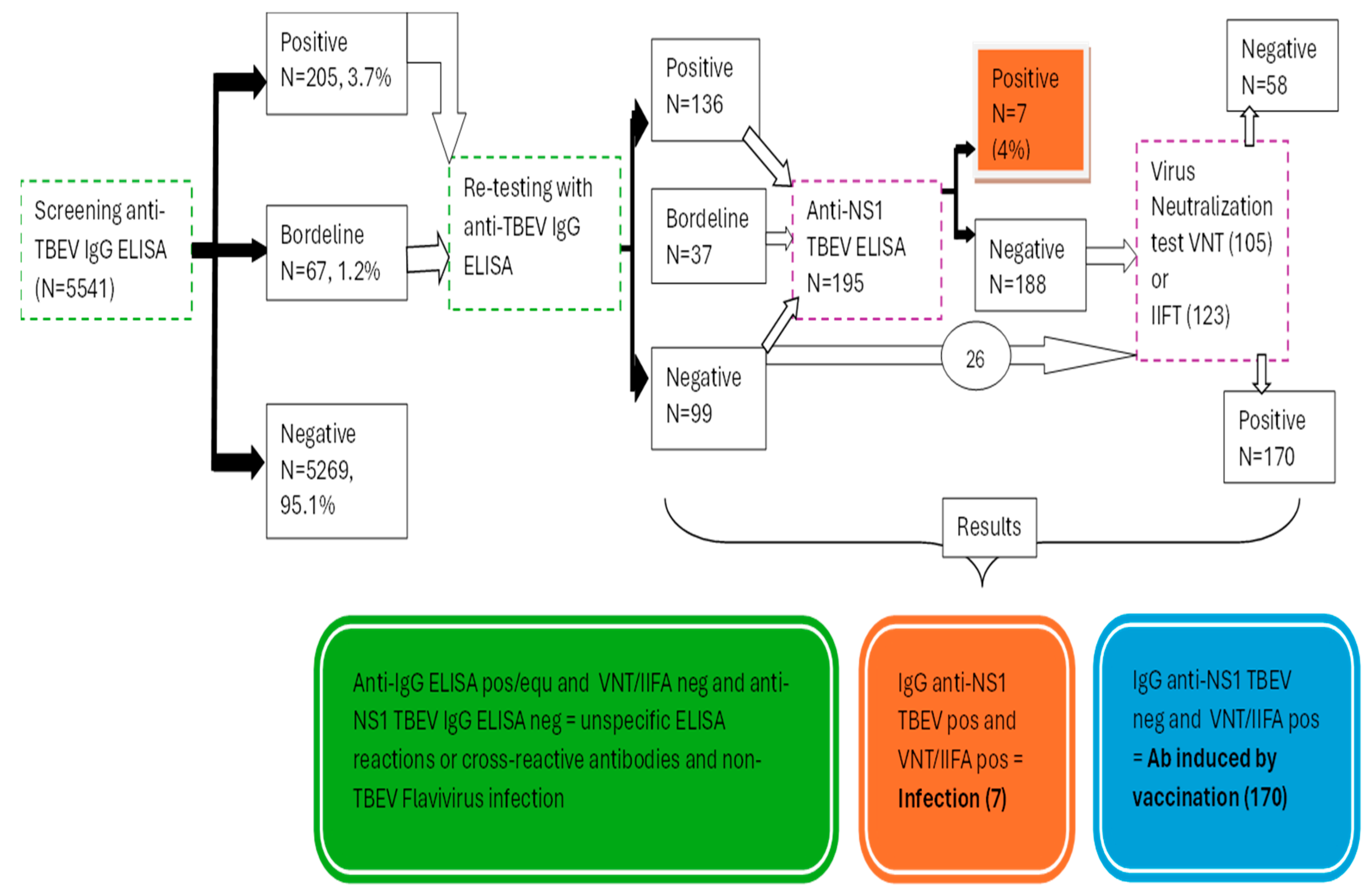
3.1. Screening IgG assays
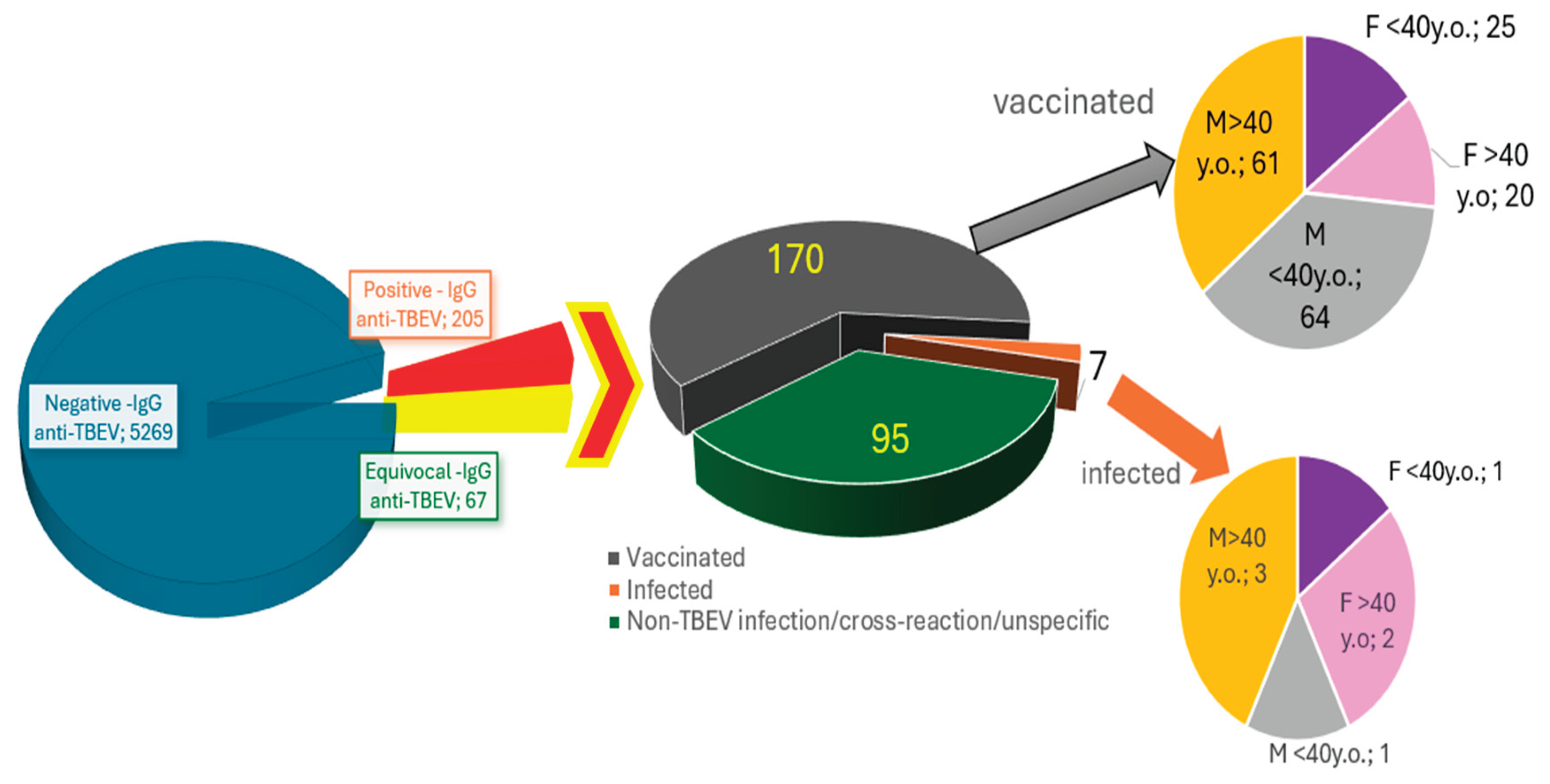
3.2. Differentiation of Post-Vaccination and Post-Infection Immune Responses
3.3. Additional Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| TBEV | Tick-borne encephalitis virus |
| TBE | Tick-borne encephalitis |
| VNT | virus neutralization testing |
| WHO | World Health Organization |
| RBTCs | Regional Blood Transfusion Centers |
| IIF | indirect immunofluorescence |
| ELISAs | enzyme-linked immunosorbent assays |
| IIFT | Indirect immunofluorescence test |
References
- Mrozowska-Nyckowska, K.; Zbrzeźniak, J.; Paradowska-Stankiewicz, I. Meningitis and encephalitis in Poland in 2021. Przegl. Epidemiol. 2023, 77, 387–402. [Google Scholar] [CrossRef]
- Nagańska, A.; Garbacz-Łagożna, E.; Kowalski, J.; Cholewińska-Szymańska, G.; Chylak-Nowosielska, A.; Antosiewicz, M.; Szymański, R. The thick-borne encephalitis caused by alimentary transmission - case series report. Przegl. Epidemiol. 2023, 77, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Król, M.E.; Borawski, B.; Nowicka-Ciełuszecka, A.; Tarasiuk, J.; Zajkowska, J. Outbreak of alimentary tick-borne encephalitis in Podlaskie voivodeship, Poland. Przegl. Epidemiol. 2019, 73, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Lipowski, D.; Popiel, M.; Perlejewski, K.; Nakamura, S.; Bukowska-Osko, I.; Rzadkiewicz, E.; Dzieciatkowski, T.; Milecka, A.; Wenski, W.; Ciszek, M.; et al. Cluster of Fatal Tick-borne Encephalitis Virus Infection in Organ Transplant Setting. J. Infect. Dis. 2017, 215, 896–901. [Google Scholar] [CrossRef]
- Wahlberg, P.; Saikku, P.; Brummer-Korvenkontio, M. Tick-borne viral encephalitis in Finland. The clinical features of Kumlinge disease during 1959-1987. J. Intern. Med. 1989, 225, 173–177. [Google Scholar] [CrossRef]
- Mathys, S.; Gültekin, N.; Stanga, Z.; Ülgür, I.; Schlagenhauf, P. Seroprevalence of tick-borne infections in blood donors in Europe: a systematic review. New Microbes New Infect. 2025, 10, 65:101597. [Google Scholar] [CrossRef]
- 7. Pancer, K. TBE in Poland. In The TBE Book, 7th ed.; Dobler, G., Erber, W., Bröker, M., Chitimia-Dobler, L., Schmitt, H.J., Eds.; Global Health Press: Singapore, 2024; p. Chapter 13. [Google Scholar] [CrossRef]
- Stefanoff, P.; Pfeffer, M.; Hellenbrand, W.; Rogalska, J.; Rühe, F.; Makówka, A.; Michalik, J.; Wodecka, B.; Rymaszewska, A.; Kiewra, D.; et al. Virus detection in questing ticks is not a sensitive indicator for risk assessment of tick-borne encephalitis in humans. Zoonoses Public Health 2013, 60, 215–226. [Google Scholar] [CrossRef]
- Stefanoff, P.; Zielicka-Hardy, A.; Hlebowicz, M.; Konior, R.; Lipowski, D.; Szenborn, L.; Siennicka, J.; Orlikova, H. TBE enhanced surveillance working group. New endemic foci of tick-borne encephalitis (TBE) identified in districts where testing for TBE was not available before 2009 in Poland. Parasit. Vectors 2013, 6, 180. [Google Scholar] [CrossRef]
- Stefanoff, P.; Rubikowska, B.; Bratkowski, J.; Ustrnul, Z.; Vanwambeke, S.O.; Rosinska, M. A Predictive Model Has Identified Tick-Borne Encephalitis High-Risk Areas in Regions Where No Cases Were Reported Previously, Poland, 1999-2012. Int. J. Environ. Res. Public Health 2018, 15, 677. [Google Scholar] [CrossRef] [PubMed]
- Kunze, M.; Banović, P; Bogovič, P.; Briciu, V; Čivljak, R.; Dobler, G.; Hristea, A.; Kerlik, J.; Kuivanen, S.; Kynčl, J.; et al. Recommendations to Improve Tick-Borne Encephalitis Surveillance and Vaccine Uptake in Europe. Microorganisms 2022, 10, 1283. [Google Scholar] [CrossRef]
- Zielicka-Hardy, A.; Rosińska, M.; Kondrusik, M.; Hlebowicz, M.; Konior, R.; Stefanoff, P. Predictors for diagnosis of tick-borne encephalitis infection in Poland, 2009-2010. Infect. Dis. 2015, 47, 604–610. [Google Scholar] [CrossRef]
- Zajkowska, J.; Waluk, E.; Świerzbińska, R.; Dunaj, J.; Zajkowska, O.; Wawrzuta, D.; Niścigorska-Olsen, J.; Matukiewicz, M.; Oczko-Grzesik, B.; Veltze, D. The impact of serological testing implementation on tick-borne encephalitis detection in Poland. PLOS ONE 2025, 20, e0323022. [Google Scholar] [CrossRef] [PubMed]
- Caputo, M.; Stumpe, V.; Rübsamen, N.; Mikolajczyk, R.T.; Karch, A. Implementation of preventive measures against tick-borne infections in a non-endemic area for tick-borne encephalitis-Results from a population-based survey in Lower Saxony, Germany. Ticks Tick Borne Dis. 2019, 10, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Zavadska, D.; Anca, I.; André, F.; Bakir, M.; Chlibek, R.; Cižman, M.; Ivaskeviciene, I.; Mangarov, A.; Mészner, Z.; Pokorn, M.; et al. Recommendations for tick-borne encephalitis vaccination from the Central European Vaccination Awareness Group (CEVAG). Hum. Vaccin. Immunother. 2013, 9, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Hertzell, K.B.; Pauksens, K.; Rombo, L.; Knight, A.; Vene, S.; Askling, H.H. Tick-borne encephalitis (TBE) vaccine to medically immunosuppressed patients with rheumatoid arthritis: A prospective, open-label, multi-centre study. Vaccine 2016, 34, 650–655. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Tick-borne encephalitis. Immunization offers the most effective protection [Internet]. Available from: https://www.who.int/teams/immunization-vaccines-and-biologicals/diseases/tick-borne-encephalitis (accessed on 10 February 2026).
- Act of 26 June 1974 – Labour Code (consolidated text: Journal of Laws 2023, item 1465, as amended). Republic of Poland. Available online: https://isap.sejm.gov.pl/ (accessed on 10 February 2026).
- National Institute of Public Health NIH – National Research Institute. Program Szczepień Ochronnych dzieci i młodzieży w 2026 roku [Protective Vaccination Program for Children and Adolescents in 2026]. Szczepienia.info. 2026. Available online: https://szczepienia.pzh.gov.pl/kalendarz-szczepien-2026-2/ (accessed on 10 February 2026).
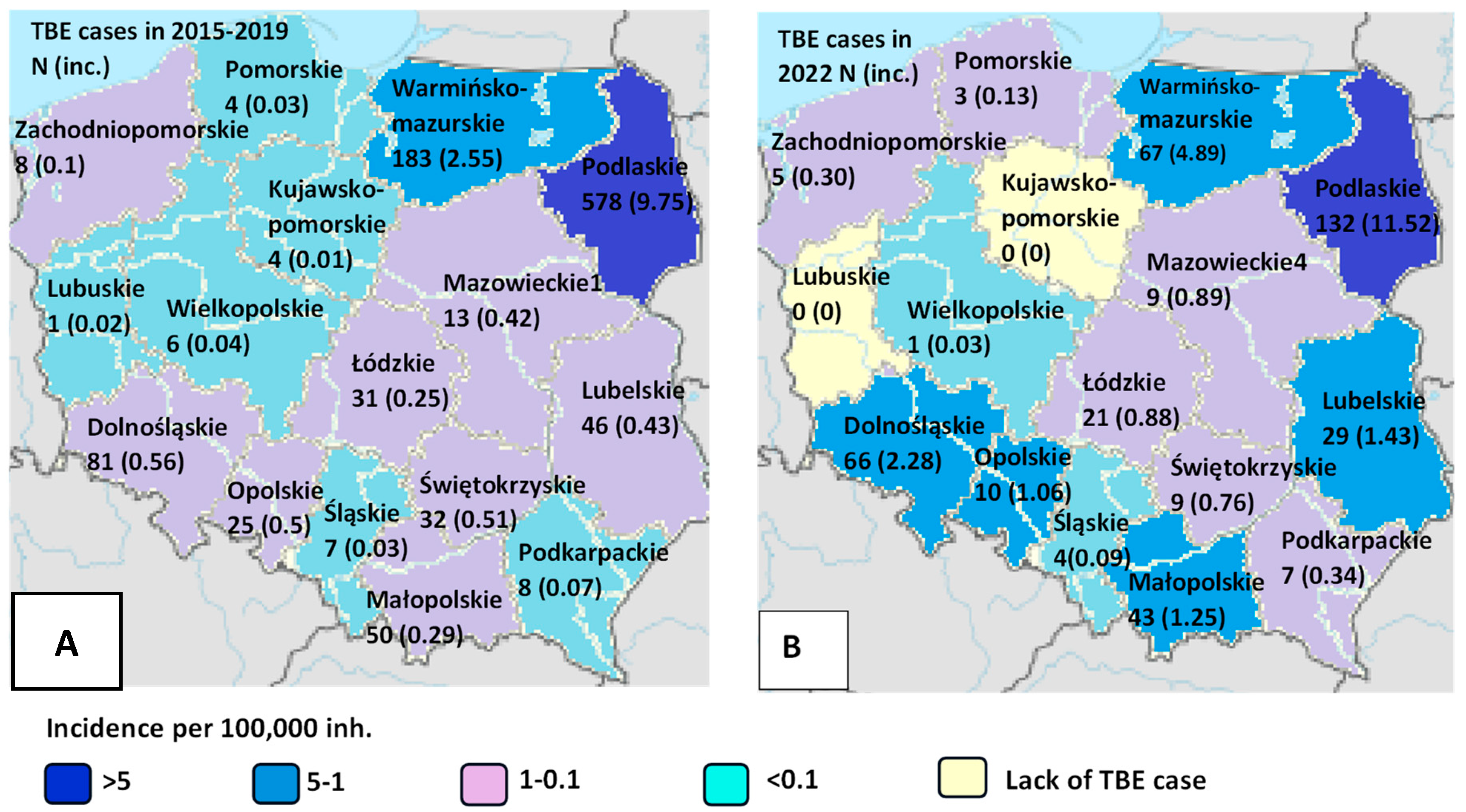
- Paradowska-Stankiewicz, I.; Pancer, K.; Poznańska, A.; Hordowicz, M.; Skibicka, M.; Słowiński, M.; Motak, G.; Falkiewicz, B. Tick-borne encephalitis epidemiology and surveillance in Poland, and comparison with selected European countries before and during the COVID-19 pandemic, 2008 to 2020. Euro Surveill. 2023, 28, 2200452. [Google Scholar] [CrossRef]
- Paradowska-Stankiewicz, I.; Piotrowska, A. Meningitis and encephalitis in Poland in 2015. Przegl. Epidemiol. 2017, 71, 493–500. [Google Scholar] [PubMed]
- Bojkiewicz, E.; Toczylowski, K.; Grygorczuk, S.; Zelazowska-Rutkowska, B.; Dunaj, J.; Zebrowska, A.; Czupryna, P.; Moniuszko-Malinowska, A.; Sulik, A. The Prevalence of Asymptomatic Infections with Tick-Borne Encephalitis Virus and Attitude towards Tick-Borne Encephalitis Vaccine in the Endemic Area of Northeastern Poland. Vaccines 2022, 10, 1294. [Google Scholar] [CrossRef]
- Stefanoff, P.; Siennicka, J.; Kaba, J.; Nowicki, M.; Ferenczi, E.; Gut, W. Identification of new endemic tick-borne encephalitis foci in Poland – a pilot seroprevalence study in selected regions. Int. J. Med. Microbiol. 2008, 298, 102–107. [Google Scholar] [CrossRef]
- Girl, P.; Bestehorn-Willmann, M.; Zange, S.; Borde, J.P.; Dobler, G.; von Buttlar, H. Tick-Borne Encephalitis Virus Nonstructural Protein 1 IgG Enzyme-Linked Immunosorbent Assay for Differentiating Infection versus Vaccination Antibody Responses. J. Clin. Microbiol. 2020, 58, e01783-19. [Google Scholar] [CrossRef]
- Dobler, G.; Euringer, K.; Kaier, K.; Borde, J.P. Serological Protection Rates against TBEV Infection in Blood Donors from a Highly Endemic Region in Southern Germany. Vaccines 2023, 11, 522. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing [Internet]; R Foundation for Statistical Computing: Vienna, Austria, 2023; Available online: https://www.R-project.org/.
- Kelly, P.H.; Kwark, R.; Marick, H.M.; Davis, J.; Stark, J.H.; Madhava, H.; Dobler, G.; Moïsi, J.C. Different environmental factors predict the occurrence of tick-borne encephalitis virus (TBEV) and reveal new potential risk areas across Europe via geospatial models. Int J Health Geogr 2025, 24, 3. [Google Scholar] [CrossRef] [PubMed]
- Coroian, M.; Mihalca, A.D.; Dobler, G.; Euringer, K.; Girl, P.; Borșan, S.D.; Kalmár, Z.; Tincuța Briciu, V.; Flonta, M.; Topan, A.; et al. Seroprevalence Rates against West Nile, Usutu, and Tick-Borne Encephalitis Viruses in Blood-Donors from North-Western Romania. Int. J. Environ. Res. Public Health 2022, 19, 8182. [Google Scholar] [CrossRef] [PubMed]
- Panciu, A.M.; Cheran, C.A.; Militaru, E.D.; Rîciu, C.D.; Hristea, A. Serosurvey of Tick-Borne Encephalitis Virus Infection in Romania. Pathogens 2024, 13, 231. [Google Scholar] [CrossRef] [PubMed]
- Fafangel, M.; Cassini, A.; Colzani, E.; Klavs, I.; Grgič Vitek, M.; Učakar, V.; Muehlen, M.; Vudrag, M.; Kraigher, A. Estimating the annual burden of tick-borne encephalitis to inform vaccination policy, Slovenia, 2009 to 2013. Euro Surveill. 2017, 22, 30509. [Google Scholar] [CrossRef] [PubMed]
- Castagna, A.; Schiavon, S.; Mengozzi, G.; Baldovin, T.; Nozza, S.; Canetti, D.; Cocchio, S.; Montuori, E.A.; Iantomasi, R.; Baldo, V. Tick-borne encephalitis seroprevalence in northern Italy: a cross-sectional study on a randomly selected population. IJID Reg. 2024, 12, 100404. [Google Scholar] [CrossRef] [PubMed]
- Kriz, B.; Hubalek, Z.; Marek, M.; Daniel, M.; Strakova, P.; Betasova, L. Results of the Screening of Tick-Borne Encephalitis Virus Antibodies in Human Sera from Eight Districts Collected Two Decades Apart. Vector-Borne Zoonotic Dis. 2015, 15, 489–493. [Google Scholar] [CrossRef]
- Banović, P.; Obregón, D.; Mijatović, D.; Simin, V.; Stankov, S.; Budakov-Obradović, Z.; Bujandrić, N.; Grujić, J.; Sević, S.; Turkulov, V.; et al. Tick-Borne Encephalitis Virus Seropositivity among Tick Infested Individuals in Serbia. Pathogens 2021, 10, 301. [Google Scholar] [CrossRef]
- Euringer, K.; Girl, P.; Kaier, K.; Peilstöcker, J.; Schmidt, M.; Müller-Steinhardt, M.; Rauscher, B.; Bressau, E.; Kern, W.V.; Dobler, G.; et al. Tick-borne encephalitis virus IgG antibody surveillance: vaccination- and infection-induced seroprevalences, south-western Germany, 2021. Euro Surveill. 2023, 28, 2200408. [Google Scholar] [CrossRef]
- Albinsson, B.; Hoffman, T.; Kolstad, L.; Bergström, T.; Bogdanovic, G.; Heydecke, A.; Hägg, M.; Kjerstadius, T; Lindroth, Y; Petersson, A.; et al. Seroprevalence of tick-borne encephalitis virus and vaccination coverage of tick-borne encephalitis, Sweden, 2018 to 2019. Euro Surveill. 2024, 29, 2300221. [Google Scholar] [CrossRef]
- Ackermann-Gäumann, R.; Eyer, C.; Vock, M.; Gowland, P.; Tinguely, C.; Leib, S.L.; Bori, M.; Buser, A.; Fontana, S.; Thierbach, J.; et al. Prevalence of anti-tick-borne encephalitis virus (TBEV) antibodies in Swiss blood donors in 2014-2015. Blood Transfus. 2023, 21, 100–109. [Google Scholar] [CrossRef]
- Paradowska-Stankiewicz, I.; Zbrzeźniak, J. Kleszczowe zapalenie mózgu w Polsce i na świecie. Ocena sytuacji epidemiologicznej KZM w Polsce w latach 2015–2019 w oparciu o dane pochodzące z nadzoru epidemiologicznego [Tick-borne encephalitis in Poland and worldwide. Assessment of the epidemiological situation of TBE in Poland in 2015–2019 based on epidemiological surveillance data]. National Institute of Public Health NIH—National Research Institute: Warsaw, Poland, 2021. Available online: https://www.pzh.gov.pl/wp-content/uploads/2021/03/KleszczoweZapalenieMoz (accessed on 10 February 2026).
- Girl, P.; Schindler, A.; Martin, S.; Dobler, G.; Borde, J.P. Surveillance of Tick-Borne Encephalitis Virus-Comparison of Vaccination- and Infection-Induced Seroprevalences in Lower Bavaria, Germany. J. Med. Virol. 2025, 97, e70514. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.F.; Gynthersen, R.M.M.; Ocias, L.F.; Sørensen, C.A.; Jensen, B.A.; Erikstrup, C.; Holm, D.K.; Sækmose, S.G.; Harritshøj, L.H.; Kolstad, L.; et al. A serosurvey examining exposure to Borrelia burgdorferi sensu lato and tick-borne encephalitis virus in Danish blood donors, August 2022. IJID Reg. 2024, 12, 100414. [Google Scholar] [CrossRef]
- Costantini, M.; Callegaro, A.; Beran, J.; Berlaimont, V.; Galgani, I. Predicted long-term antibody persistence for a tick-borne encephalitis vaccine: results from a modeling study beyond 10 years after a booster dose following different primary vaccination schedules. Hum. Vaccin. Immunother. 2020, 16, 2274–2279. [Google Scholar] [CrossRef] [PubMed]
- Beran, J.; Lattanzi, M.; Costantini, M.; Pammolli, A.; Galgani, I. Sustained antibody persistence for at least 15 years after a booster vaccination against tick-borne encephalitis following different primary vaccination schedules: Third 5-year follow-up. Vaccine 2023, 41, 3518–3524. [Google Scholar] [CrossRef]
- Freimane, Z.; Dobler, G.; Karelis, G.; Chitimia-Dobler, L.; Savicka, O.; Zavadska, D. Seroprevalence of Tick-Borne Encephalitis Virus in Latvia Using Standard and Subtype-Specific NS1-Based ELISA Assays. Pathogens 2025, 14, 1115. [Google Scholar] [CrossRef]
- Chan, K.R.; Ismail, A.A.; Thergarajan, G.; Raju, C.S.; Yam, H.C.; Rishya, M.; Sekaran, S.D. Serological cross-reactivity among common flaviviruses. Front. Cell. Infect. Microbiol. 2022, 12, 975398. [Google Scholar] [CrossRef]
- Waldeck, M.; Winqvist, N.; Henriksson, G.; Dyrdak, R.; Settergren, B.; Lindgren, P.E. Surveillance of tick-borne encephalitis in emerging risk areas in southern Sweden: a retrospective case finding study. Eur. J. Clin. Microbiol. Infect. Dis. 2023, 42, 13–22. [Google Scholar] [CrossRef]
- Kubicka-Russel, D.; Kopacz, A.; Sulkowska, E.; Łętowska, M.; Grabarczyk, P. Demographic changes in the Polish blood donors eligible for blood donation and screened for transfusion-transmitted infections (2005–2018). J. Transfus. Med. Hemost. 2023, 16, 39–64. [Google Scholar] [CrossRef]
| Voivodeship | Classification based on 5years TBE incidence/ 100 000 inhabitants |
Screening anti-TBEV IgG | OR1 | 95% CI1 | p-value2 | Gender % Pos+borderline |
Age % Pos+borderline |
Status3 N |
|||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Number of pos+borderline/N examined samples | % pos+ borderline |
Female % |
Male % |
<40 y.o. % |
>40 y.o. % |
Inf/vac /other | |||||
| Dolnośląskie | Moderately affect | 12/605 | 2.0 | 0.99 | 0.44, 2.24 | 0.977 | 1.8 | 2.1 | 1.9 | 2.1 | 1/10/1 |
| Kujawsko-pomorskie | Less affected | 49/600 | 8.2 | 4.36 | 2.37, 8.66 | <0.001 | 8.4 | 8.1 | 9.9 | 2.6 | 0/15/34 |
| Lubelskie | Moderately affect | 25/600 | 4.2 | 2.13 | 1.08, 4.43 | 0.034 | 2.6 | 5.5 | 5.4 | 2.8 | 0/19/6 |
| Łódzkie | Moderately affect | 12/600 | 2.0 | 14 | - | 0 | 3.92 | 2.3 | 1.6 | 0/10/2 | |
| Małopolskie | Moderately affect | 26/609 | 4.3 | 2.19 | 1.12, 4.53 | 0.027 | 2.3 | 6.32 | 4.5 | 3.1 | 1/15/10 |
| Podkarpackie | Less affected | 22/624 | 3.5 | 1.79 | 0.89, 3.76 | 0.110 | 1.6 | 5.42 | 3.5 | 3.8 | 1/12/9 |
| Podlaskie | Highly affected | 67/703 | 9.5 | 5.16 | 2.87, 10.1 | <0.001 | 8.3 | 10.5 | 7.3 | 12.2 | 2/52/13 |
| Pomorskie | Less affected | 27/600 | 4.5 | 2.40 | 1.24, 4.94 | 0.012 | 2.8 | 5.2 | 5.0 | 4.0 | 2/16/9 |
| Zachodniopomorskie | Less affected | 32/600 | 5.3 | 2.76 | 1.45, 5.63 | 0.003 | 5.0 | 5.7 | 7 | 3.7 | 0/21/11 |
| In total | 272/5541 | 4.9 | 3.5 | 6.12 | 5.2 | 4.5 | 7/170/95 | ||||
| N= 5541, OR1=1.86; 95% CI1 (1.43, 2.43), p<0.001 |
N= 5541, OR1=0.86; 95% CI1 (0.67, 1.09), p>0.05 |
N=272, p<0.001 | |||||||||
| N positive+bordeline / N examined samples | Inf/vac/other1 | Seroprevalence (95% CI2) | Seroprevalece excl other (95% CI) | Seroprevalence, infection (95% CI) | OR3 vs Less affected (seroprev excl other) | OR vs Less affected (seroprev, inf) | |
|---|---|---|---|---|---|---|---|
| Highly affected | 67/703 | 2/52/13 | 9.5% (7.5% - 11.9%) | 7.7% (5.8%-9.9%) | 0.3% (0.03% - 1.0%) | 2.8 (1.9-4.1), p<0.001 | 2.3 (0.2-20.1) p=>0.05 |
| Moderately affect | 75/2414 | 2/54/19 | 3.1% (2.4%-3.9%) | 2.3% (1.8%-3.0%) | 0.08% (0.001%-0.3%) | 0.84 (0.58-1.22), p>0.05 | 0.67 (0.06-5.85) p=>0.05 |
| Less affected | 130/2424 | 3/64/63 | 5.4% (4.5%-6.3%) | 2.8% (2.1%-3.5%) | 0.1% (0.003% - 0.4%) | 1 (ref.) | 1 (ref.) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




