Submitted:
10 March 2026
Posted:
11 March 2026
You are already at the latest version
Abstract
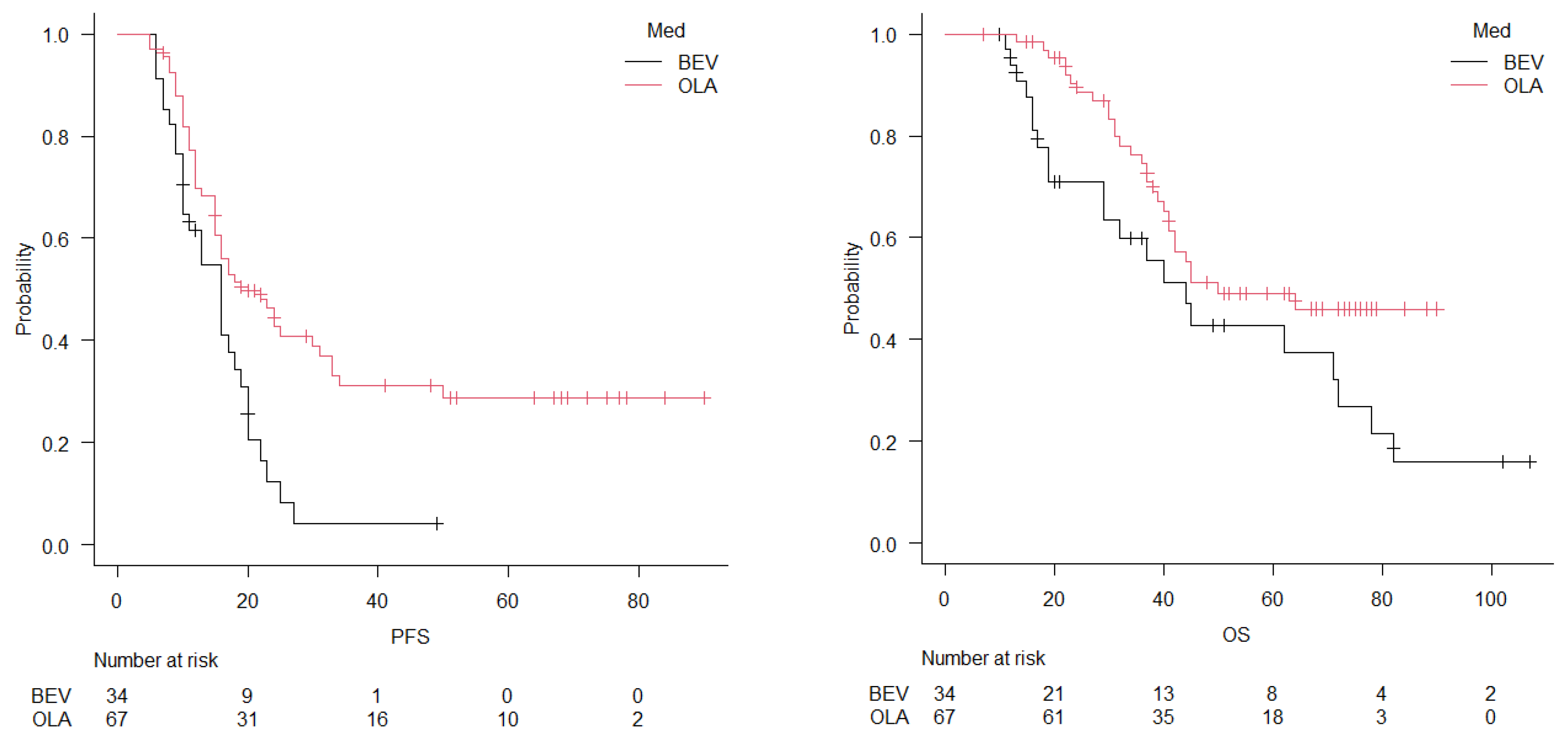
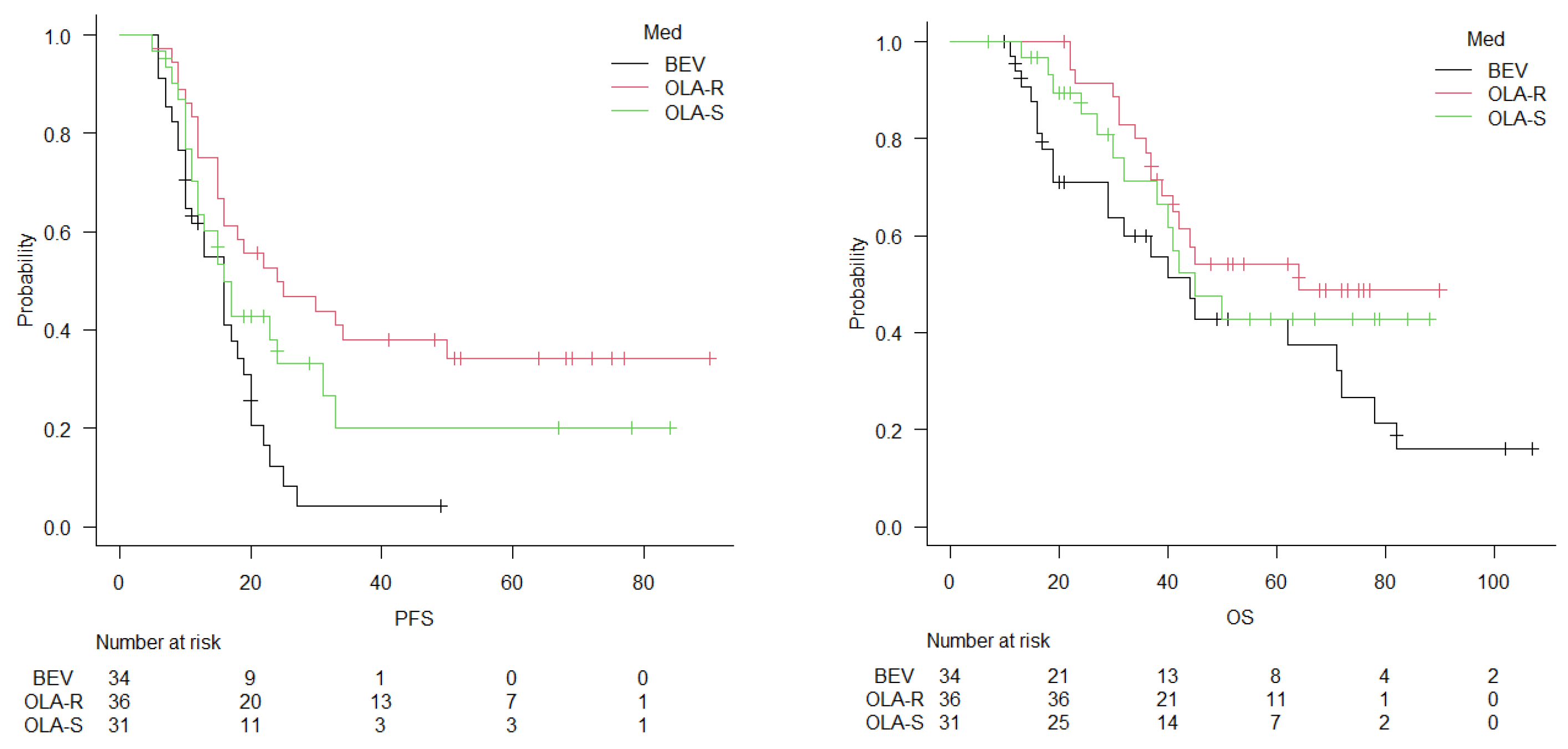
Objective: Bevacizumab (BEV) and Olaparib (OLA) have demonstrated clinical efficacy as maintenance therapies for first platinum-sensitive recurrent ovarian cancer. However, direct comparisons between these agents independent of homologous recombination deficiency (HRD) and BRCA status, remain limited and the clinical validity of OLA dose reduction has not yet been confirmed. This study aimed to compare the efficacy and safety of BEV, standard-dose OLA, and dose-reduced OLA as maintenance therapy and to evaluate the clinical utility of OLA dose reduction. Methods: This retrospective multicenter study included 101 patients with first platinum-sensitive recurrent ovarian, fallopian tube, or primary peritoneal cancer who received maintenance therapy after achieving a response to chemotherapy. Patients were classified into three groups: BEV (n = 34), standard-dose OLA (n = 31), and dose-reduced OLA (n = 36). The primary endpoint was progression-free survival (PFS), and secondary endpoints included overall survival (OS) and adverse events. Survival outcomes were evaluated using Kaplan–Meier analysis and Cox proportional hazards models. Results: Median PFS was 16 months in the BEV group, 16 months in the standard-dose OLA group, and 24 months in the dose-reduced OLA group, with significantly longer PFS in the dose-reduced OLA group (p < 0.001). In the multivariate Cox analysis, treatment remained an independent prognostic factor for PFS (HR 0.67, 95% CI 0.46–0.96, p = 0.030). Median OS was 44, 45, and 64 months, respectively, with no significant differences among groups; PFI ≥12 months was the only independent prognostic factor for OS. Grade ≥3 hematologic toxicities were more frequent in the OLA groups but were manageable. Conclusions: Dose-reduced OLA was associated with prolonged PFS while maintaining manageable toxicity, supporting its clinical validity as a maintenance option independent of HRD and BRCA status.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Treatments
2.3. Endpoints/Variables
2.4. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Survival Analysis
3.3. Adverse Events
| BEV(N=34) | OLA-S(N=31) | OLA-R(N=36) | P value* | ||||
|---|---|---|---|---|---|---|---|
| G3 | G4 | G3 | G4 | G3 | G4 | ||
| Leucopenia | 7 | 1 | 4 | 0 | 8 | 0 | 0.476 |
| Neutropenia | 6 | 4 | 8 | 1 | 12 | 6 | 0.126 |
| Anemia | 4 | 0 | 3 | 0 | 9 | 4 | 0.010 |
| Thrombocytopenia | 1 | 3 | 1 | 0 | 2 | 0 | 0.315 |
| Febrile neutropenia |
1 | 1 | 0 | 0 | 0 | 0 | 0.123 |
| BEV(N=34) | OLA-S(N=31) | OLA-R(N=36) | P value* | ||||
|---|---|---|---|---|---|---|---|
| G3 | G4 | G3 | G4 | G3 | G4 | ||
| Nausea | 2 | 0 | 2 | 0 | 5 | 0 | 0.434 |
| Neuropathy | 4 | 0 | 2 | 0 | 1 | 0 | 0.307 |
| Diarrhea | 0 | 0 | 1 | 0 | 0 | 0 | 0.320 |
| Fatigue | 2 | 0 | 2 | 0 | 2 | 0 | 0.983 |
| Appetite Loss | 1 | 0 | 0 | 0 | 0 | 0 | 0.353 |
| Hypertension | 8 | 0 | 3 | 0 | 0 | 0 | 0.005 |
| Proteinuria | 4 | 0 | 1 | 1 | 0 | 0 | 0.101 |
| CBDCA hypersensitivity |
0 | 0 | 2 | 0 | 0 | 0 | 0.100 |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Siegel, RL.; Miller, KD.; Jemal, A. Cancer statistics, 2020. CA Cancer J Clin. 2020, 70, 7–30.
- Bray, F.; Colombet, M.; Aitken, J.F.; Bardot, A.; Eser, S.; Galceran, J.; Hagenimana, M.; Matsuda, T.; Mery, L.; Piñeros, M.; at al. Cancer Incidence in Five Continents, Vol. XII (IARC CancerBase No. 19). Lyon: International Agency for Research on Cancer.2023.
- Cabasag, CJ.; Fagan, PJ.; Ferlay, J.; Vignat, J.; Laversanne, M.; Liu, L.; van der Aa, M.A.; Bray, F.; Soerjomataram, I. Ovarian cancer today and tomorrow: A global assessment by world region and Human Development Index using GLOBOCAN 2020. Int J Cancer. 2022, 151, 1535–1541. [CrossRef]
- McGuire, WP.; Hoskins, WJ.; Brady, MF.; Kucera, PR.; Partridge, EE.; Look, KY.; Clarke-Pearson, DL.; Davidson, M. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N. Engl. J. Med. 1996, 334, 1-6. [CrossRef]
- Siegel, RL.; Giaquinto, AN.; Jemal, A. Cancer statistics, 2024. CA Cancer J Clin. 2024, 74, 12–49.
- Siegel, RL.; Miller, KD.; Fuchs, HE.; Jemal, A. Cancer statistics, 2022. CA Cancer J Clin. 2023, 72, 7–33.
- Torre LA.; Trabert B.; DeSantis CE.; Miller KD.; Samimi G.; Runowicz CD.; Gaudet MM.; Jemal A.; Sigel RL. Ovarian cancer statistics, 2018. CA Cancer J Clin. 2018, 68, 284–296. [CrossRef]
- Sung H.; Ferlay J.; Siegel RL.; Laversanne M.; Soerjomataram I.; Jemal A.; Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries.CA Cancer J. Clin. 2021, 71, 209–249. [CrossRef]
- Piccart MJ.; Bertelsen K.; James K.; Cassidy J.; Mangioni C.; Simonsen E.; Stuart G.; Kaye S.; Vergote I.; Blom R.; Grimshaw R.; Atkinson R.; Swenerton K.; Trope C.; Parmar MKB. Randomized intergroup trial of cisplatin–paclitaxel versus cisplatin–cyclophosphamide in advanced epithelial ovarian cancer. J. Natl. Cancer Inst. 2000, 92, 699–708.
- Ozols RF.; Bundy BN.; Greer BE.; Fowler JM.; Clarke-Pearson D.; Burger, RA.; Mannel RS.; DeGeest K.; Hartenbach, EM.; Baergen R.; Gynecologic Oncology Group. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in optimally resected stage III ovarian cancer. J. Clin. Oncol. 2003, 21, 3194–3200.
- du Bois A.; Luck HJ.; Meier W.; Adams HP.; Möbus V.; Costa S.; Bauknecht T.; Richter B.; Warm M.; Schröder W.; Olbricht S.; Pfisterer J. Cisplatin/paclitaxel versus carboplatin/paclitaxel as first-line treatment of ovarian cancer. J. Natl. Cancer Inst. 2003, 95, 1320–1329. [CrossRef]
- Thigpen JT., Blessing JA., Ball H., Hummel SJ., Barrett RJ. Phase II trial of paclitaxel in patients with progressive ovarian carcinoma after platinum-based chemotherapy: A Gynecologic Oncology Group study. J. Clin. Oncol. 1994, 12, 1748–1753. [CrossRef]
- Hanker LC.; Loibl S.; Burchardi N.; Pfisterer J.; Meier W.; Pujade-Lauraine E.; Ray-Coquard I.; Sehouli J.; Harter P.; du Bois A. The impact of later-line therapy on survival of relapsed ovarian cancer. Ann. Oncol. 2012, 23, 2605–2612. [CrossRef]
- NCCN Clinical Practice Guidelines in Oncology: Ovarian Cancer Including Fallopian Tube Cancer and Primary Peritoneal Cancer. Version 1.2023; NCCN: Plymouth Meeting, PA, USA, 2023.
- Tokunaga H.; Mikami M.; Nagase S.; Kobayashi Y.; Tabata T.; Kaneuchi M.; Satoh T.; Hirashima Y.; Matsumura N.; Yokoyama Y. The 2020 Japan Society of Gynecologic Oncology guidelines for the treatment of ovarian cancer, fallopian tube cancer, and primary peritoneal cancer. J. Gynecol. Oncol. 2021, 32, e49. [CrossRef]
- Coleman RL.; Brady MF.; Herzog TJ.; Sabbatini P.; Armstrong DK.; Walker JL.; Kim BG.; Fujiwara K.; Tewari KS.; O’Malley DM.; Davidson SA.; Benigno B.; Cohn DE.; Lankes HA.; Aghajanian C. Bevacizumab and chemotherapy in recurrent ovarian cancer (GOG-0213). N. Engl. J. Med. 2017, 377, 1695–1706.
- Aghajanian C.; Blank SV.; Goff BA.; Judson PL.; Teneriello MG.; Husain A.; Sovak MA.; Yi J.; Nycum LR. OCEANS: A randomized, double-blind, placebo-controlled phase III trial of chemotherapy with or without bevacizumab in platinum-sensitive recurrent epithelial ovarian, primary peritoneal, or fallopian tube cancer. J. Clin. Oncol. 2012, 30, 2039–2045. [CrossRef]
- Pignata S.; Lorusso D.; Joly F.; Gallo C.; Colombo N.; Sessa C.; Bamias A.; Sini V.; Gallo A.; Di Maio M.; De Placido S.; du Bois A. Chemotherapy plus or minus bevacizumab for platinum-sensitive ovarian cancer patients recurring after a bevacizumab-containing first-line treatment (MITO16B-MaNGO OV2B-ENGOT OV17). J. Clin. Oncol. 2021, 39, 2737–2748. [CrossRef]
- Pujade-Lauraine E.; Ledermann JA.; Selle F.; Gebski V.; Penson RT.; Oza AM.; Korach J.; Huzarski T.; Poveda A.; Pignata S.; Friedlander M.; Colombo N.; Vergote I.; Glasspool RM.; Guo W.; Scott C.; Patel S.; Matos-Pita AS.; Tonkin K.; Embleton A.; Banerjee S. Olaparib tablets as maintenance therapy in patients with platinum-sensitive relapsed ovarian cancer (SOLO2). Lancet Oncol. 2017, 18, 1274–1284.
- Poveda A.; Floquet A.; Ledermann JA.; Asher R.; Penson RT.; Oza AM.; Korach J.; Huzarski T.; Pignata S.; Friedlander M.; Selle F.; Glasspool RM.; Guo W.; Scott C.; Patel S.; Matos-Pita AS.; Embleton A.; Banerjee S.; Pujade-Lauraine E. Final overall survival analysis of SOLO2/ENGOT-Ov21. Lancet Oncol. 2021, 22, 620–631.
- Ledermann J.; Harter P.; Gourley C.; Friedlander M.; Vergote I.; Rustin G.; Scott C.; Meier W.; Shapira-Frommer R.; Safra T.; Matei D.; Macpherson E.; Watkins C.; Carmichael J.; Matulonis U. Olaparib maintenance therapy in platinum-sensitive relapsed ovarian cancer. N. Engl. J. Med. 2012, 366, 1382–1392. [CrossRef]
- Kim SI.; Lee M.; Kim JW.; Lee JW.; Lee YY.; Kang S.; Park SY.; Korean Gynecologic Oncology Group. Comparisons of survival outcomes between bevacizumab and olaparib in BRCA-mutated, platinum-sensitive relapsed ovarian cancer (KGOG 3052). Cancer Res. Treat. 2022, 54, 1224–1234.
- Abe M.; Shoji T.; Chiba Y.; Takatori E.; Kaido Y.; Nagasawa T.; Kagabu M.; Takahashi F.; Aida T.; Baba T. Efficacy and Safety of Platinum-based Chemotherapy with Bevacizumab Followed by Bevacizumab Maintenance for Recurrent Ovarian, Fallopian Tube, and Primary Peritoneal Cancer During PARP Inhibitor Therapy: A Multicenter Retrospective Study. Anticancer Res. 2023, 43, 1265–1272. [CrossRef]
- Colombo N.; Sessa C.; du Bois A.; Ledermann J.; McCluggage WG.; McNeish I.; Morice P.; Pignata S.; Ray-Coquard I.; Vergote I.; Baert T.; Belaroussi I.; Dashora A.; Gourley C.; Harter P.; Houghton R.; Høgdall C.; Lorusso D.; Marth C.; O’Donnell D.; Querleu D.; Raspagliesi F.; Reed N.; Salvesen HB.; Sehouli J.; Soumerai T.; Thomassin-Naggara I.; Trillsch F.; van Altena AM.; Zola, P. ESMO–ESGO consensus recommendations on ovarian cancer. Int. J. Gynecol. Cancer 2019, 29, 728–760.
- Eisenhauer EA.; Therasse P.; Bogaerts J.; Schwartz LH.; Sargent D.; Ford R.; Dancey J.; Arbuck S.; Gwyther S.; Mooney M.; Rubinstein L.; Shankar L.; Dodd L.; Kaplan R.; Lacombe D.; Verweij J. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [CrossRef]
- Japan Clinical Oncology Group. Common Toxicity Criteria for Adverse Events version 5.0 Japanese Translation (CTCAE JCOG). Available at: https://jcog.jp/doctor/tool/ctcaev5/ [Last accessed on September 2, 2025].
- Friedlander M.; Trimble E.; Tinker A.; Alberts D.; Avall-Lundqvist E.; Brady M.; Harter P.; Pignata S.; Pujade-Lauraine E.; Vergote I.; du Bois A. Clinical trials in recurrent ovarian cancer. Int. J. Gynecol. Cancer 2011, 21, 771–775. [CrossRef]
- Kanda Y: Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013, 48, 452-458. [CrossRef]
- Francis KE.; Kim SI.; Friedlander M.; Gebski V.; Ray-Coquard I.; Clamp A.; Penson RT.; Poveda A.; Banerjee S.; Pujade-Lauraine E. The impact of olaparib dose reduction and treatment interruption on treatment outcome in SOLO2/ENGOT-ov21. Ann. Oncol. 2022, 33, 593–601.
- Nakanishi K.; Toyoshima M.; Ueno Y.; Suzuki S. A retrospective study comparing olaparib and bevacizumab as maintenance therapy for platinum-sensitive recurrent ovarian cancer. Cancers 2023, 15, 2869.
- Mirza MR.; Monk BJ.; Herrstedt J.; Oza AM.; Mahner S.; Redondo A.; Fabbro M.; Ledermann JA.; Lorusso D.; Vergote I.; Ben-Baruch NE.; Marth C.; Madry R.; Christensen RD.; Berek JS.; Dorum A.; Tinker AV.; du Bois A.; Gonzalez-Martin A.; Follana P.; Bessette P.; Plante M.; Kaern J.; Tholander B.; Sharma S.; Ueno T.; Devries-Aboud M.; Sun K.; Malinowski W.; Liu J.; Balasubramanian S.; Matulonis UA. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N. Engl. J. Med. 2016, 375, 2154–2164.
| BEV (N=34) |
Standard-dose OLA (N=31) |
Dose-reduced OLA (N=36) |
P value | ||
|---|---|---|---|---|---|
| Age | Median, Range | 62 (33-82) | 59 (46-74) | 63(40-80) | 0.167** |
| Diagnosis | Ovarian | 20 | 18 | 33 | 0.001* |
| Fallopian Tube | 5 | 7 | 2 | ||
| Peritoneal | 9 | 6 | 1 | ||
| Stage | I~II | 8 | 8 | 4 | 0.256* |
| III~IV | 26 | 23 | 32 | ||
| Histology | Serous/Endometrioid | 28 | 27 | 34 | 0.576* |
| Clear/Mucinous | 5 | 3 | 2 | ||
| Other | 1 | 1 | 0 | ||
| HRD status | Positive | 5 | 4 | 0 | 0.119* |
| Negative | 4 | 4 | 2 | ||
| Untested | 25 | 23 | 34 | ||
| BRCA variant | Positive | 4 | 5 | 2 | 0.041* |
| Negative | 12 | 4 | 5 | ||
| Untested | 18 | 22 | 29 | ||
| Prior maintenance therapy |
BEV | 6 | 15 | 15 | 0.014* |
| OLA | 12 | 3 | 3 | ||
| BEV+OLA | 1 | 0 | 0 | ||
| No treatment | 15 | 13 | 18 | ||
| PFI | Median, Range | 17.5(6-140) | 19(6-114) | 21(6-96) | 0.310** |
| Chemotherapy regimen before maintenance | TC(+BEV) | 27 | 22 | 28 | 0.441* |
| TP(+BEV) | 1 | 3 | 0 | ||
| DC(+BEV) | 1 | 1 | 1 | ||
| PLDC(+BEV) | 5 | 5 | 5 | ||
| Other | 0 | 2 | |||
| Anti-tumor response |
CR | 7 | 10 | 11 | 0.515* |
| PR | 27 | 21 | 25 |
| PFS-Cox proportional hazard model (Univariate analysis) | |||||||
| Category | HR | 95%CI | P value | ||||
| Treatment | BEV/OLA-S/OLA-R | 0.62 | 0.47-0.84 | 0.002 | |||
| Age | 1.03 | 1.00-1.05 | 0.028 | ||||
| Stage | I・II / III・IV | 1.39 | 0.76-2.53 | 0.288 | |||
| Histology | HGSC・EM/CCC・M | 1.34 | 0.67-2.70 | 0.409 | |||
| PFI | (<12 / >12 months) | 0.46 | 0.27-0.78 | 0.003 | |||
| Prior maintenance | None/BEV/PARP-i/BEV+PARP-i | 1.32 | 0.94-1.85 | 0.105 | |||
| Anti-tumor response | CR/PR | 1.56 | 0.97-2.48 | 0.063 | |||
| PFS-Cox proportional hazard model (Multivariate analysis) | |||||||
| Category | HR | 95%CI | P value | ||||
| Treatment | BEV/OLA-S/OLA-R | 0.67 | 0.46-0.96 | 0.030 | |||
| Stage | I・II / III・IV | 1.54 | 0.78-2.99 | 0.206 | |||
| PFI | (<12 / >12 months) | 0.58 | 0.33-1.02 | 0.060 | |||
| Prior maintenance | None/BEV/PARP-i/BEV+PARP-i | 1.06 | 0.75-1.51 | 0.732 | |||
| Anti-tumor response | CR/PR | 1.18 | 0.68-2.05 | 0.564 | |||
| OS-Cox proportional hazard model (Univariate analysis) | ||||
| Category | HR | 95%CI | P value | |
| Treatment | BEV/OLA-S/OLA-R | 0.72 | 0.51-1.00 | 0.050 |
| Age | 1.02 | 0.99-1.05 | 0.191 | |
| Stage | I・II / III・IV | 1.40 | 0.68-2.89 | 0.363 |
| Histology | HGSC・EM/CCC・M | 1.35 | 0.57-3.19 | 0.499 |
| PFI | (<12 / >12 months) | 0.38 | 0.21-0.69 | 0.001 |
| Prior maintenance | None/BEV/PARPi/BEV+PARPi | 1.45 | 0.95-2.21 | 0.085 |
| Anti-tumor response | CR/PR | 1.20 | 0.68-2.11 | 0.531 |
| OS-Cox proportional hazard model (Multivariate analysis) | ||||
| Category | HR | 95%CI | P value | |
| Treatment | BEV/OLA-S/OLA-R | 0.73 | 0.49-1.08 | 0.114 |
| Age | 1.12 | 0.50-2.51 | 0.792 | |
| Stage | I・II / III・IV | 0.43 | 0.23-0.81 | 0.009 |
| Histology | HGSC・EM/CCC・M | 1.34 | 0.88-2.04 | 0.174 |
| PFI | (<12 / >12 months) | 0.87 | 0.46-1.64 | 0.666 |
| Prior maintenance | None/BEV/PARPi/BEV+PARPi | 0.73 | 0.49-1.08 | 0.114 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




