Submitted:
09 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Search Methods
3. NK Cells in BD
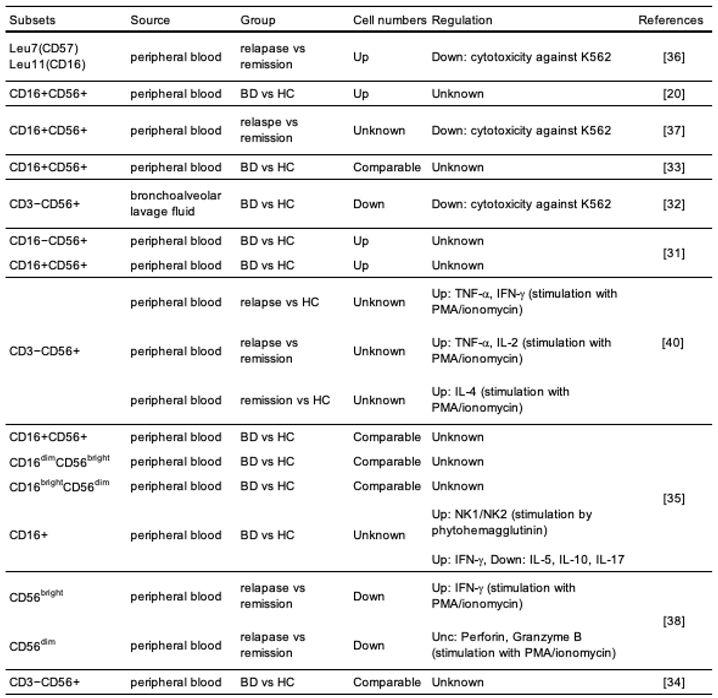
3.1. Numbers, Cytokine Production, and Cytotoxicity
3.2. NK Cell Receptors and Their Ligands
3.2.1. HLA-B and KIR
3.2.2. MICA and KLRK1(NKG2D)
3.2.3. HLA-E and KLRD1/KLRC1 (CD94/NKG2A)
3.3. Expression of NK Receptors
3.4. Other Genetic Factors
4. Regulation of NK Receptor Genes
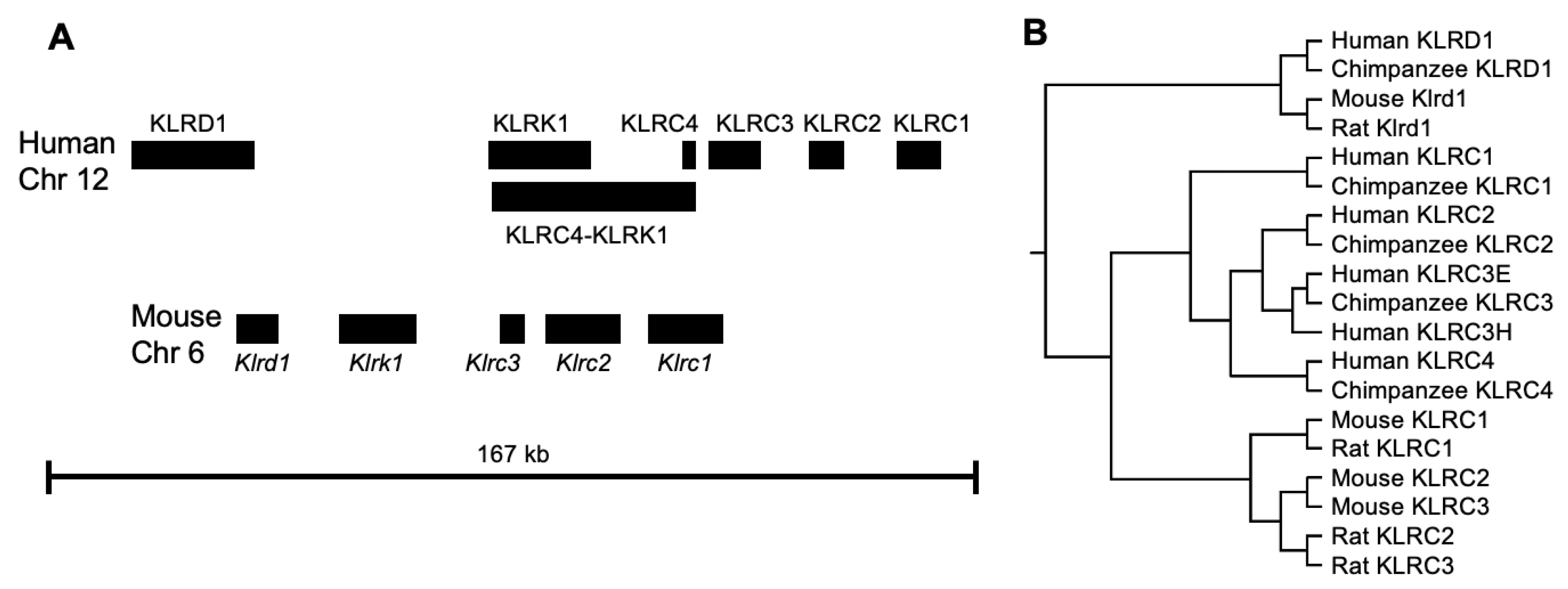
4.1. Genomic Organization and Gene Expression of the NK Complex
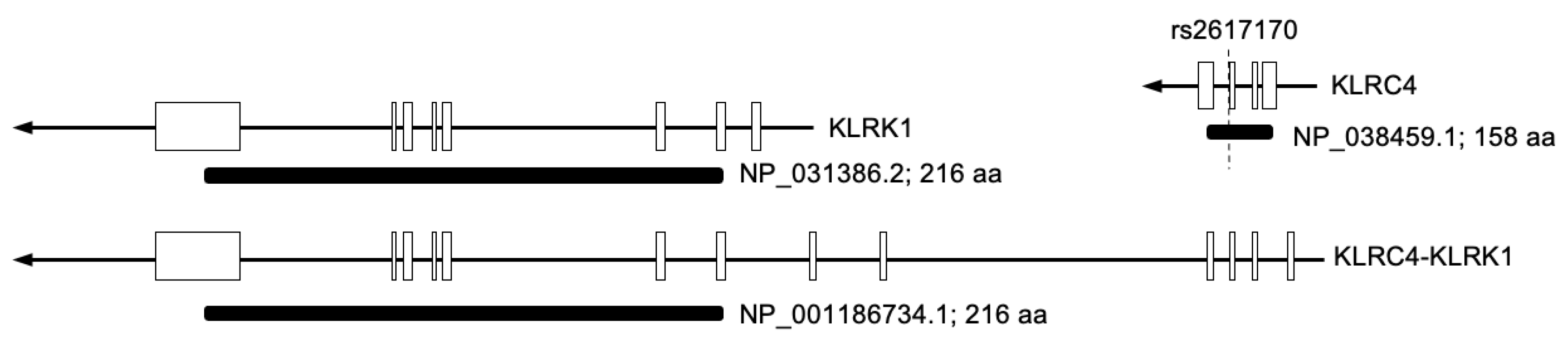
4.2. Splicing of KLRK1 (NKG2D) and Its Function
4.3. KLRK1-KLRC4 Readthrough Gene
4.4. Dysregulation of KLRC4 in BD
4.5. Fucosylation of CD16a
5. Distinct NK Subsets
5.1. Circulating and Tissue Resident NK Cells
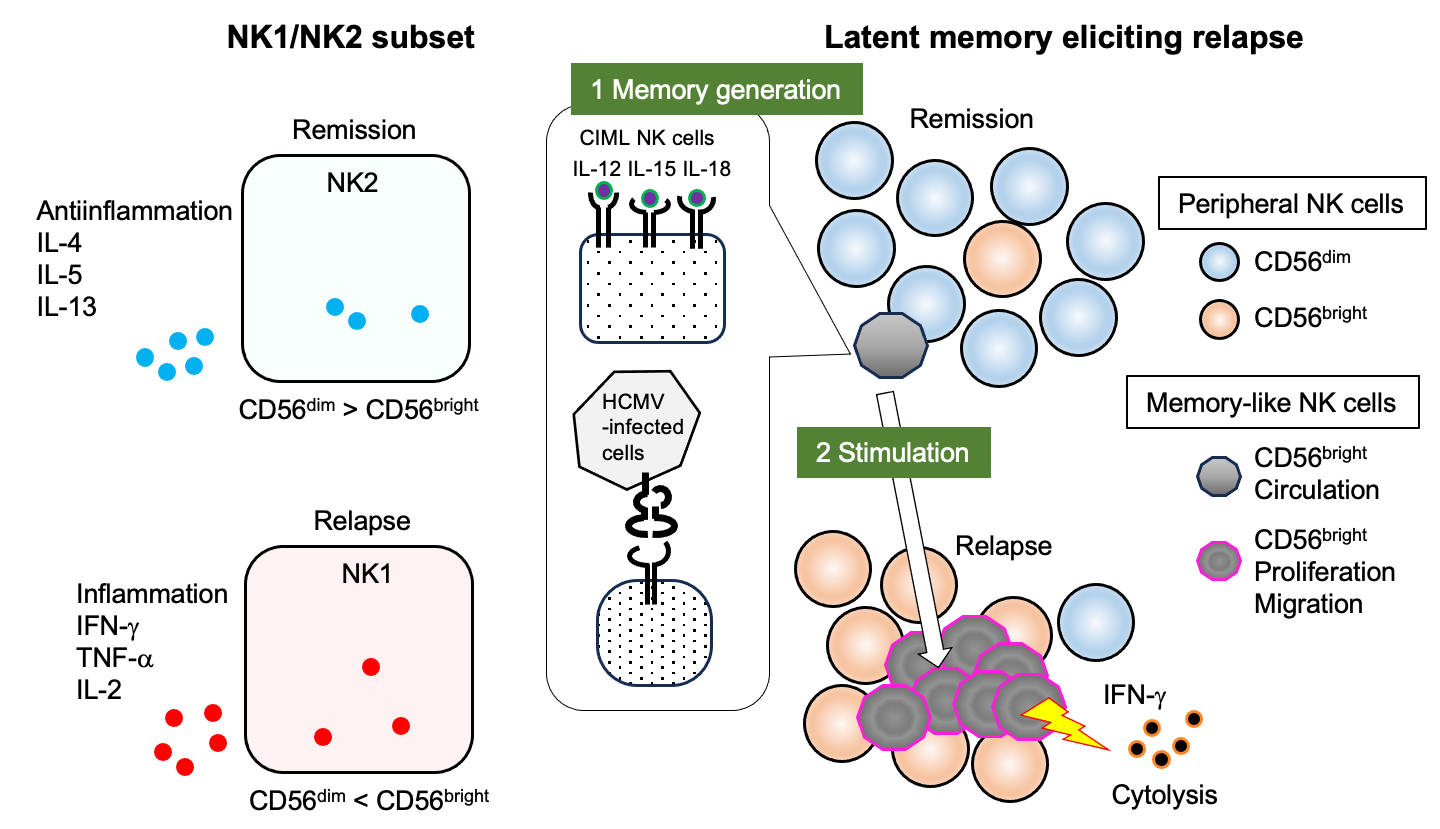
5.2. NK Cell Memory
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moghoofei, M; Pajavand, H; Shahbazi, R; Rezaei, M; Taki, E. The Role of Viral and Bacterial Infections in the Etiology of Behçet’s Disease. J Clin Lab Anal. 2026, 40, e70133. [Google Scholar] [CrossRef] [PubMed]
- Isogai, E; Ohno, S; Kotake, S; Isogai, H; Tsurumizu, T; Fujii, N; Yokota, K; Syuto, B; Yamaguchi, M; Matsuda, H; et al. Chemiluminescence of neutrophils from patients with Behcet’s disease and its correlation with an increased proportion of uncommon serotypes of Streptococcus sanguis in the oral flora. Arch Oral Biol. 1990, 35, 43–48. [Google Scholar] [CrossRef]
- Ohno, S; Ohguchi, M; Hirose, S; Matsuda, H; Wakisaka, A; Aizawa, M. Close association of HLA-Bw51 with Behcet’s disease. Arch Ophthalmol. 1982, 100, 1455–1458. [Google Scholar] [CrossRef]
- Mizuki, N; Ohno, S; Tanaka, H; Sugimura, K; Seki, T; Mizuki, N; Kera, J; Inaba, G; Tsuji, K; Inoko, H. Association of HLA-B51 and lack of association of class II alleles with Behcet’s disease. Tissue Antigens. 1992, 40, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Remmers, EF; Cosan, F; Kirino, Y; Ombrello, MJ; Abaci, N; Satorius, C; Le, JM; Yang, B; Korman, BD; Cakiris, A; Aglar, O; Emrence, Z; Azakli, H; Ustek, D; Tugal-Tutkun, I; Akman-Demir, G; Chen, W; Amos, CI; Dizon, MB; Kose, AA; Azizlerli, G; Erer, B; Brand, OJ; Kaklamani, VG; Kaklamanis, P; Ben-Chetrit, E; Stanford, M; Fortune, F; Ghabra, M; Ollier, WE; Cho, YH; Bang, D; O’Shea, J; Wallace, GR; Gadina, M; Kastner, DL; Gül, A. Genome-wide association study identifies variants in the MHC class I, IL10, and IL23R-IL12RB2 regions associated with Behçet’s disease. Nat Genet. 2010, 42, 698–702. [Google Scholar] [CrossRef]
- Hughes, T; Coit, P; Adler, A; Yilmaz, V; Aksu, K; Düzgün, N; Keser, G; Cefle, A; Yazici, A; Ergen, A; Alpsoy, E; Salvarani, C; Casali, B; Kötter, I; Gutierrez-Achury, J; Wijmenga, C; Direskeneli, H; Saruhan-Direskeneli, G; Sawalha, AH. Identification of multiple independent susceptibility loci in the HLA region in Behçet’s disease. Nat Genet. 2013, 45, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Ombrello, MJ; Kirino, Y; de Bakker, PI; Gül, A; Kastner, DL; Remmers, EF. Behçet disease-associated MHC class I residues implicate antigen binding and regulation of cell-mediated cytotoxicity. Proc Natl Acad Sci U S A. 2014, 111, 8867–8872. [Google Scholar] [CrossRef]
- McGonagle, D; Aydin, SZ; Gül, A; Mahr, A; Direskeneli, H. ‘MHC-I-opathy’-unified concept for spondyloarthritis and Behcet disease. Nat Rev Rheumatol. 2015, 11, 731–740. [Google Scholar] [CrossRef]
- Ucar-Comlekoglu, D; Fox, A; Sen, HN. Gender Differences in Behçet’s Disease Associated Uveitis. J Ophthalmol. 2014, 2014, 820710. [Google Scholar] [CrossRef] [PubMed]
- Jo, YG; Ortiz-Fernández, L; Coit, P; Yilmaz, V; Yentür, SP; Alibaz-Oner, F; Aksu, K; Erken, E; Düzgün, N; Keser, G; Cefle, A; Yazici, A; Ergen, A; Alpsoy, E; Salvarani, C; Kısacık, B; Kötter, I; Henes, J; Çınar, M; Schaefer, A; Nohutcu, RM; Takeuchi, F; Harihara, S; Kaburaki, T; Messedi, M; Song, YW; Kaşifoğlu, T; Martin, J; González Escribano, MF; Saruhan-Direskeneli, G; Direskeneli, H; Sawalha, AH. Sex-specific analysis in Behcet’s disease reveals higher genetic risk in male patients. J Autoimmun. 2022, 132, 102882. [Google Scholar] [CrossRef]
- Liu, A; Su, Y; Zhu, J; Li, YY. Disease association study of Autoimmune and autoinflammatory diseases by integrating multi-modal data and hierarchical ontologies. Front Immunol. 2025, 16, 1575490. [Google Scholar] [CrossRef]
- Davatchi, F; Assaad-Khalil, S; Calamia, KT; Crook, JE; Sadeghi-Abdollahi, B; Schirmer, M; Tzellos, T; Zouboulis, CC; Akhlagi, M; Al-Dalaan, A; Alekberova, ZS; Ali, AA; Altenburg, A; Arromdee, E; Baltaci, M; Bastos, M; Benamour, S; Ben Ghorbel, I; Boyvat, A; Carvalho, L; Chen, W; Ben-Chetrit, E; Chams-Davatchi, C; Correia, JA; Crespo, J; Dias, C; Dong, Y; Paix~ao-Duarte, F; Elmuntaser, K; Elonakov, AV; Gra~na Gil, J; Haghdoost, AA; Hayani, RM; Houman, H; Isayeva, AR; Jamshidi, AR; Kaklamanis, P; Kumar, A; Kyrgidis, A; Madanat, W; Nadji, A; Namba, K; Ohno, S; Olivieri, I; Vaz Patto, J; Pipitone, N; de Queiroz, MV; Ramos, F; Resende, C; Rosa, CM; Salvarani, C; Serra, MJ; Shahram, F; Shams, H; Sharquie, KE; Sliti-Khanfir, M; Tribolet de Abreu, T; Vasconcelos, C; Vedes, J; Wechsler, B; Cheng, YK; Zhang, Z; Ziaei, N. The International Criteria for Behcet’s Disease (ICBD): a collaborative study of 27 countries on the sensitivity and specificity of the new criteria. J Eur Acad Dermatol Venereol. 2014, 28, 338–347. [Google Scholar]
- Zhan, H; Cheng, L; Chen, H; Liu, Y; Feng, X; Li, H; Li, Z; Li, Y. Evaluation of inflammatory-thrombosis panel as a diagnostic tool for vascular Behcet’s disease. Clin Rheumatol. 2025, 44, 1279–1291. [Google Scholar] [CrossRef] [PubMed]
- Sahin, S; Akoğlu, T; Direskeneli, H; Sen, LS; Lawrence, R. Neutrophil adhesion to endothelial cells and factors affecting adhesion in patients with Behçet’s disease. Ann Rheum Dis. 1996, 55, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Ergun, T; Gürbüz, O; Harvell, J; Jorizzo, J; White, W. The histopathology of pathergy: a chronologic study of skin hyperreactivity in Behçet’s disease. Int J Dermatol 1998, 37, 929–933. [Google Scholar] [CrossRef]
- Le Joncour, A; Martos, R; Loyau, S; Lelay, N; Dossier, A; Cazes, A; Fouret, P; Domont, F; Papo, T; Jandrot-Perrus, M; Bouton, MC; Cacoub, P; Ajzenberg, N; Saadoun, D; Boulaftali, Y. Critical role of neutrophil extracellular traps (NETs) in patients with Behcet’s disease. Ann Rheum Dis. 2019, 78, 1274–1282. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y; Ning, K; Huang, Z; Chen, B; Chen, J; Wen, Y; Bu, J; Hong, H; Chen, Q; Zhang, Z; Jia, R; Su, W. NETs-CD44-IL-17A Feedback Loop Drives Th17-Mediated Inflammation in Behcet’s Uveitis. Adv Sci (Weinh). 2025, 12, e2411524. [Google Scholar] [CrossRef]
- Hirahara, L; Takase-Minegishi, K; Kirino, Y; Iizuka-Iribe, Y; Soejima, Y; Yoshimi, R; Nakajima, H. The Roles of Monocytes and Macrophages in Behçet’s Disease With Focus on M1 and M2 Polarization. Front Immunol. 2022, 13, 852297. [Google Scholar] [CrossRef]
- Nakano, H; Kirino, Y; Takeno, M; Higashitani, K; Nagai, H; Yoshimi, R; Yamaguchi, Y; Kato, I; Aoki, I; Nakajima, H. GWAS-identified CCR1 and IL10 loci contribute to M1 macrophage-predominant inflammation in Behcet’s disease. Arthritis Res Ther. 2018, 20, 124. [Google Scholar] [CrossRef]
- Suzuki, Y; Hoshi, K; Matsuda, T; Mizushima, Y. Increased peripheral blood gamma delta+ T cells and natural killer cells in Behçet’s disease. J Rheumatol. 1992, 19, 588–592. [Google Scholar]
- Hasan, A; Fortune, F; Wilson, A; Warr, K; Shinnick, T; Mizushima, Y; van der Zee, R; Stanford, MR; Sanderson, J; Lehner, T. Role of gamma delta T cells in pathogenesis and diagnosis of Behcet’s disease. Lancet. 1996, 347, 789–794. [Google Scholar] [CrossRef]
- Raziuddin, S; al-Dalaan, A; Bahabri, S; Siraj, AK; al-Sedairy, S. Divergent cytokine production profile in Behçet’s disease. Altered Th1/Th2 cell cytokine pattern. J Rheumatol. 1998, 25, 329–333. [Google Scholar]
- Hasan, MS; Bergmeier, LA; Petrushkin, H; Fortune, F. Gamma Delta (γδ) T Cells and Their Involvement in Behçet’s Disease. J Immunol Res. 2015, 2015, 705831. [Google Scholar] [CrossRef]
- Poli, A; Michel, T; Thérésine, M; Andrès, E; Hentges, F; Zimmer, J. CD56bright natural killer (NK) cells: an important NK cell subset. Immunology. 2009, 126, 458–465. [Google Scholar] [CrossRef]
- Reefman, E; Kay, JG; Wood, SM; Offenhäuser, C; Brown, DL; Roy, S; Stanley, AC; Low, PC; Manderson, AP; Stow, JL. Cytokine secretion is distinct from secretion of cytotoxic granules in NK cells. J Immunol. 2010, 184, 4852–4862. [Google Scholar] [CrossRef]
- Anfossi, N; André, P; Guia, S; Falk, CS; Roetynck, S; Stewart, CA; Breso, V; Frassati, C; Reviron, D; Middleton, D; Romagné, F; Ugolini, S; Vivier, E. Human NK cell education by inhibitory receptors for MHC class I. Immunity. 2006, 25, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, WM; Kim, S. Licensing of natural killer cells by self-major histocompatibility complex class I. Immunol Rev. 2006, 214, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Kärre, K. Natural killer cell recognition of missing self. Nat Immunol. 2008, 9, 477–480. [Google Scholar] [CrossRef]
- Fukuda, M. Lysosomal membrane glycoproteins. Structure, biosynthesis, and intracellular trafficking. J Biol Chem. 1991, 266, 21327–21330. [Google Scholar] [CrossRef] [PubMed]
- Alter, G; Malenfant, JM; Altfeld, M. CD107a as a functional marker for the identification of natural killer cell activity. J Immunol Methods. 2004, 294, 15–22. [Google Scholar] [CrossRef]
- Sakly, K; Lahmar, R; Nefzi, F; Hammami, S; Harzallah, O; Sakly, N; Sakly, W; Hassine, M; Mahjoub, S; Ghedira, I; Feki, S. Phenotypic abnormalities of peripheral blood mononuclear cells in patients with Behçet’s disease and association with HLA-B51 expression. Immunol Invest. 2014, 43, 463–478. [Google Scholar] [CrossRef]
- Hamzaoui, K; Berraies, A; Kaabachi, W; Ammar, J; Hamzaoui, A. Pulmonary manifestations in Behcet disease: impaired natural killer cells activity. Multidiscip Respir Med. 2013, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Saruhan-Direskeneli, G; Uyar, FA; Cefle, A; Onder, SC; Eksioglu-Demiralp, E; Kamali, S; Inanç, M; Ocal, L; Gül, A. Expression of KIR and C-type lectin receptors in Behcet’s disease. Rheumatology (Oxford). 2004, 43, 423–427. [Google Scholar] [CrossRef]
- Bonacini, M; Soriano, A; Zerbini, A; Calò, E; Cimino, L; Muratore, F; Fontana, L; Braglia, L; Parmeggiani, M; Salvarani, C; Croci, S. Higher Frequencies of Lymphocytes Expressing the Natural Killer Group 2D Receptor in Patients With Behcet Disease. Front Immunol. 2018, 9, 2157. [Google Scholar] [CrossRef]
- Cosan, F; Aktas Cetin, E; Akdeniz, N; Emrence, Z; Cefle, A; Deniz, G. Natural Killer Cell Subsets and Their Functional Activity in Behcet’s Disease. Immunol Invest. 2017, 46, 419–432. [Google Scholar] [CrossRef]
- Kaneko, F; Takahashi, Y; Muramatsu, R; Adachi, K; Miura, Y; Nakane, A; Minagawa, T. Natural killer cell numbers and function in peripheral lymphoid cells in Behcet’s disease. Br J Dermatol. 1985, 113, 313–318. [Google Scholar] [CrossRef]
- Onder, M; Bozkurt, M; Gürer, MA; Gülekon, A; Sezgin, P; Imir, T. Natural cellular cytotoxicity in Behcet’s disease. J Dermatol. 1994, 21, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Hasan, MS; Ryan, PL; Bergmeier, LA; Fortune, F. Circulating NK cells and their subsets in Behçet’s disease. Clin Exp Immunol. 2017, 188, 311–322. [Google Scholar] [CrossRef]
- Deniz, G; Akdis, M; Aktas, E; Blaser, K; Akdis, CA. Human NK1 and NK2 subsets determined by purification of IFN-gamma-secreting and IFN-gamma-nonsecreting NK cells. Eur J Immunol. 2002, 32, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Kucuksezer, UC; Aktas-Cetin, E; Bilgic-Gazioglu, S; Tugal-Tutkun, I; Gül, A; Deniz, G. Natural killer cells dominate a Th-1 polarized response in Behcet’s disease patients with uveitis. Clin Exp Rheumatol. 2015, 33, S24–S29. [Google Scholar]
- Rebuffet, L; Melsen, JE; Escalière, B; Basurto-Lozada, D; Bhandoola, A; Björkström, NK; Bryceson, YT; Castriconi, R; Cichocki, F; Colonna, M; Davis, DM; Diefenbach, A; Ding, Y; Haniffa, M; Horowitz, A; Lanier, LL; Malmberg, KJ; Miller, JS; Moretta, L; Narni-Mancinelli, E; O’Neill, LAJ; Romagnani, C; Ryan, DG; Sivori, S; Sun, D; Vagne, C; Vivier, E. High-dimensional single-cell analysis of human natural killer cell heterogeneity. Nat Immunol. 2024, 25, 1474–1488. [Google Scholar] [CrossRef]
- Sallakci, N; Tahrali, I; Kucuksezer, UC; Cetin, EA; Gul, A; Deniz, G. Effect of different cytokines in combination with IL-15 on the expression of activating receptors in NK cells of patients with Behcet’s disease. Immunol Res. 2022, 70, 654–666. [Google Scholar] [CrossRef] [PubMed]
- Montes-Cano, MA; Conde-Jaldón, M; García-Lozano, JR; Ortiz-Fernández, L; Ortego-Centeno, N; Castillo-Palma, MJ; Espinosa, G; Graña-Gil, G; González-Gay, MA; Barnosi-Marín, AC; Solans, R; Fanlo, P; Camps, T; Castañeda, S; Sánchez-Bursón, J; Núñez-Roldán, A; Martín, J; González-Escribano, MF. HLA and non-HLA genes in Behçet’s disease: a multicentric study in the Spanish population. Arthritis Res Ther. 2013, 15, R145. [Google Scholar] [CrossRef] [PubMed]
- Litwin, V; Gumperz, J; Parham, P; Phillips, JH; Lanier, LL. NKB1: a natural killer cell receptor involved in the recognition of polymorphic HLA-B molecules. J Exp Med. 1994, 180, 537–543. [Google Scholar] [CrossRef]
- Gumperz, JE; Litwin, V; Phillips, JH; Lanier, LL; Parham, P. The Bw4 public epitope of HLA-B molecules confers reactivity with natural killer cell clones that express NKB1, a putative HLA receptor. J Exp Med. 1995, 181, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Sanjanwala, B; Draghi, M; Norman, PJ; Guethlein, LA; Parham, P. Polymorphic sites away from the Bw4 epitope that affect interaction of Bw4+ HLA-B with KIR3DL1. J Immunol. 2008, 181, 6293–6300. [Google Scholar] [CrossRef]
- Erer, B; Takeuchi, M; Ustek, D; Tugal-Tutkun, I; Seyahi, E; Özyazgan, Y; Duymaz-Tozkir, J; Gül, A; Kastner, DL; Remmers, EF; Ombrello, MJ. Evaluation of KIR3DL1/KIR3DS1 polymorphism in Behçet’s disease. Genes Immun. 2016, 17, 396–399. [Google Scholar] [CrossRef]
- Pando, MJ; Gardiner, CM; Gleimer, M; McQueen, KL; Parham, P. The protein made from a common allele of KIR3DL1 (3DL1*004) is poorly expressed at cell surfaces due to substitution at positions 86 in Ig domain 0 and 182 in Ig domain 1. J Immunol. 2003, 171, 6640–6649. [Google Scholar] [CrossRef]
- Castaño-Núñez, Á; Montes-Cano, MA; García-Lozano, JR; Ortego-Centeno, N; García-Hernández, FJ; Espinosa, G; Graña-Gil, G; Sánchez-Bursón, J; Juliá, MR; Solans, R; Blanco, R; Barnosi-Marín, AC; Gómez de la Torre, R; Fanlo, P; Rodríguez-Carballeira, M; Rodríguez-Rodríguez, L; Camps, T; Castañeda, S; Alegre-Sancho, JJ; Martín, J; González-Escribano, MF. Association of Functional Polymorphisms of KIR3DL1/DS1 With Behçet’s Disease. Front Immunol. 2019, 10, 2755. [Google Scholar] [CrossRef]
- Gebreselassie, D; Spiegel, H; Vukmanovic, S. Sampling of major histocompatibility complex class I-associated peptidome suggests relatively looser global association of HLA-B*5101 with peptides. Hum Immunol. 2006, 67, 894–906. [Google Scholar] [CrossRef]
- Kollnberger, S; Bird, L; Sun, MY; Retiere, C; Braud, VM; McMichael, A; Bowness, P. Cell-surface expression and immune receptor recognition of HLA-B27 homodimers. Arthritis Rheum. 2002, 46, 2972–2982. [Google Scholar] [CrossRef]
- Wong-Baeza, I; Ridley, A; Shaw, J; Hatano, H; Rysnik, O; McHugh, K; Piper, C; Brackenbridge, S; Fernandes, R; Chan, A; Bowness, P; Kollnberger, S. KIR3DL2 binds to HLA-B27 dimers and free H chains more strongly than other HLA class I and promotes the expansion of T cells in ankylosing spondylitis. J Immunol. 2013, 190, 3216–3224. [Google Scholar] [CrossRef]
- Faure, M; Long, EO. KIR2DL4 (CD158d), an NK cell-activating receptor with inhibitory potential. J Immunol. 2002, 168, 6208–6214. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi-Maki, A; Catina, TL; Campbell, KS. Cutting edge: KIR2DL4 transduces signals into human NK cells through association with the Fc receptor gamma protein. J Immunol. 2005, 174, 3859–3863. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, S; Fu, J; Long, EO. Cutting edge: induction of IFN-gamma production but not cytotoxicity by the killer cell Ig-like receptor KIR2DL4 (CD158d) in resting NK cells. J Immunol. 2001, 167, 1877–1881. [Google Scholar] [CrossRef]
- Rajagopalan, S; Long, EO. A human histocompatibility leukocyte antigen (HLA)-G-specific receptor expressed on all natural killer cells. J Exp Med. 1999, 189, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, S; Long, EO. KIR2DL4 (CD158d): An activation receptor for HLA-G. Front Immunol. 2012, 3, 258. [Google Scholar] [CrossRef]
- Kim, SJ; Lee, S; Park, C; Seo, JS; Kim, JI; Yu, HG. Targeted resequencing of candidate genes reveals novel variants associated with severe Behçet’s uveitis. Exp Mol Med. 2013, 45, e49. [Google Scholar] [CrossRef]
- Steinle, A; Li, P; Morris, DL; Groh, V; Lanier, LL; Strong, RK; Spies, T. Interactions of human NKG2D with its ligands MICA, MICB, and homologs of the mouse RAE-1 protein family. Immunogenetics. 2001, 53, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Bauer, S; Groh, V; Wu, J; Steinle, A; Phillips, JH; Lanier, LL; Spies, T. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science 1999, 285, 727–729. [Google Scholar] [CrossRef]
- Billadeau, DD; Upshaw, JL; Schoon, RA; Dick, CJ; Leibson, PJ. NKG2D-DAP10 triggers human NK cell-mediated killing via a Syk-independent regulatory pathway. Nat Immunol. 2003, 4, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Upshaw, JL; Arneson, LN; Schoon, RA; Dick, CJ; Billadeau, DD; Leibson, PJ. NKG2D-mediated signaling requires a DAP10-bound Grb2-Vav1 intermediate and phosphatidylinositol-3-kinase in human natural killer cells. Nat Immunol. 2006, 7, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Saá, I; Cambra, A; Pallarés, L; Espinosa, G; Juan, A; Pujalte, F; Matamoros, N; Milà, J; Julià, MR. Allelic diversity and affinity variants of MICA are imbalanced in Spanish patients with Behçet’s disease. Scand J Immunol. 2006, 64, 77–82. [Google Scholar] [CrossRef]
- Durmanová, V; Tirpakova, J; Stuchlikova, M; Shawkatova, I; Kuba, D; Sapak, M; Buc, M. Characterization of MICA gene polymorphism of HLA complex in the Slovak population. Ann Hum Biol. 2011, 38, 570–576. [Google Scholar] [CrossRef]
- Hughes, EH; Collins, RW; Kondeatis, E; Wallace, GR; Graham, EM; Vaughan, RW; Stanford, MR. Associations of major histocompatibility complex class I chain-related molecule polymorphisms with Behcet’s disease in Caucasian patients. Tissue Antigens. 2005, 66, 195–199. [Google Scholar] [CrossRef]
- Muñoz-Saá, I; Cambra, A; Pallarés, L; Espinosa, G; Juan, A; Pujalte, F; Matamoros, N; Milà, J; Julià, MR. Allelic diversity and affinity variants of MICA are imbalanced in Spanish patients with Behçet’s disease. Scand J Immunol. 2006, 64, 77–82. [Google Scholar] [CrossRef]
- Mizuki, N; Meguro, A; Tohnai, I; Gül, A; Ohno, S; Mizuki, N. Association of Major Histocompatibility Complex Class I Chain-Related Gene A and HLA-B Alleles with Behçet’s Disease in Turkey. Jpn J Ophthalmol. 2007, 51, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Yabuki, K; Mizuki, N; Ota, M; Katsuyama, Y; Palimeris, G; Stavropoulos, C; Koumantaki, Y; Spyropoulou, M; Giziaki, E; Kaklamani, V; Kaklamani, E; Inoko, H; Ohno, S. Association of MICA gene and HLA-B*5101 with Behçet’s disease in Greece. Invest Ophthalmol Vis Sci. 1999, 40, 1921–1926. [Google Scholar]
- Marin, ML; Savioli, CR; Yamamoto, JH; Kalil, J; Goldberg, AC. MICA polymorphism in a sample of the São Paulo population, Brazil. Eur J Immunogenet. 2004, 31, 63–71. [Google Scholar] [CrossRef]
- Hervier, B; Ribon, M; Tarantino, N; Mussard, J; Breckler, M; Vieillard, V; Amoura, Z; Steinle, A; Klein, R; Kötter, I; Decker, P. Increased Concentrations of Circulating Soluble MHC Class I-Related Chain A (sMICA) and sMICB and Modulation of Plasma Membrane MICA Expression: Potential Mechanisms and Correlation With Natural Killer Cell Activity in Systemic Lupus Erythematosus. Front Immunol. 2021, 12, 633658. [Google Scholar] [CrossRef]
- Lee, N; Llano, M; Carretero, M; Ishitani, A; Navarro, F; López-Botet, M; Geraghty, DE. HLA-E is a major ligand for the natural killer inhibitory receptor CD94/NKG2A. Proc Natl Acad Sci U S A. 1998, 95, 5199–5204. [Google Scholar] [CrossRef]
- Borrego, F; Ulbrecht, M; Weiss, EH; Coligan, JE; Brooks, AG. Recognition of human histocompatibility leukocyte antigen (HLA)-E complexed with HLA class I signal sequence-derived peptides by CD94/NKG2 confers protection from natural killer cell-mediated lysis. J Exp Med. 1998, 187, 813–818. [Google Scholar] [CrossRef] [PubMed]
- Seo, J; Park, JS; Nam, JH; Bang, D; Sohn, S; Lee, ES; Park, KS. Association of CD94/NKG2A, CD94/NKG2C, and its ligand HLA-E polymorphisms with Behcet’s disease. Tissue Antigens. 2007, 70, 307–313. [Google Scholar] [CrossRef]
- Park, KS; Park, JS; Nam, JH; Bang, D; Sohn, S; Lee, ES. HLA-E*0101 and HLA-G*010101 reduce the risk of Behcet’s disease. Tissue Antigens. 2007, 69, 139–144. [Google Scholar] [CrossRef]
- Castaño-Núñez, ÁL; Montes-Cano, MA; García-Lozano, JR; Ortego-Centeno, N; García-Hernández, FJ; Espinosa, G; Graña-Gil, G; Sánchez-Bursón, J; Juliá, MR; Solans, R; Blanco, R; Barnosi-Marín, AC; Gómez de la Torre, R; Fanlo, P; Rodríguez-Carballeira, M; Rodríguez-Rodríguez, L; Camps, T; Castañeda, S; Alegre-Sancho, JJ; Martín, J; González-Escribano, MF. The complex HLA-E-nonapeptide in Behçet disease. Front Immunol. 2023, 14, 1080047. [Google Scholar] [CrossRef]
- Picard, LK; Claus, M; Fasbender, F; Watzl, C. Human NK cells responses are enhanced by CD56 engagement. Eur J Immunol. 2022, 52, 1441–1451. [Google Scholar] [CrossRef]
- Bexte, T; Alzubi, J; Reindl, LM; Wendel, P; Schubert, R; Salzmann-Manrique, E; von Metzler, I; Cathomen, T; Ullrich, E. CRISPR-Cas9 based gene editing of the immune checkpoint NKG2A enhances NK cell mediated cytotoxicity against multiple myeloma. Oncoimmunology 2022, 11, 2081415. [Google Scholar] [CrossRef] [PubMed]
- Mac Donald, A; Guipouy, D; Lemieux, W; Harvey, M; Bordeleau, LJ; Guay, D; Roméro, H; Li, Y; Dion, R; Béland, K; Haddad, E. KLRC1 knockout overcomes HLA-E-mediated inhibition and improves NK cell antitumor activity against solid tumors. Front Immunol. 2023, 14, 1231916. [Google Scholar] [CrossRef]
- Takeno, M; Shimoyama, Y; Kashiwakura, J; Nagafuchi, H; Sakane, T; Suzuki, N. Abnormal killer inhibitory receptor expression on natural killer cells in patients with Behcet’s disease. Rheumatol Int. 2004, 24, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Gelmez, MY; Cinar, S; Cetin, EA; Ozcit-Gürel, G; Babuna-Kobaner, G; Erdugan, M; Gul, A; Akdag-Kose, A; Deniz, G. Inflammatory status might direct ILC and NK cells to IL-17 expressing ILC3 and NK subsets in Behcet’s disease. Immunol Lett. 2021, 235, 1–8. [Google Scholar] [CrossRef]
- Alves, E; McLeish, E; Blancafort, P; Coudert, JD; Gaudieri, S. Manipulating the NKG2D Receptor-Ligand Axis Using CRISPR: Novel Technologies for Improved Host Immunity. Front Immunol. 2021, 12, 712722. [Google Scholar] [CrossRef]
- Saric, T; Chang, SC; Hattori, A; York, IA; Markant, S; Rock, KL; Tsujimoto, M; Goldberg, AL. An IFN-gamma-induced aminopeptidase in the ER, ERAP1, trims precursors to MHC class I-presented peptides. Nat Immunol. 2002, 3, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Kirino, Y; Bertsias, G; Ishigatsubo, Y; Mizuki, N; Tugal-Tutkun, I; Seyahi, E; Ozyazgan, Y; Sacli, FS; Erer, B; Inoko, H; Emrence, Z; Cakar, A; Abaci, N; Ustek, D; Satorius, C; Ueda, A; Takeno, M; Kim, Y; Wood, GM; Ombrello, MJ; Meguro, A; Gül, A; Remmers, EF; Kastner, DL. Genome-wide association analysis identifies new susceptibility loci for Behçet’s disease and epistasis between HLA-B*51 and ERAP1. Nat Genet. 2013, 45, 202–207. [Google Scholar] [CrossRef]
- Chi, W; Zhu, X; Yang, P; Liu, X; Lin, X; Zhou, H; Huang, X; Kijlstra, A. Upregulated IL-23 and IL-17 in Behcet patients with active uveitis. Invest Ophthalmol Vis Sci. 2008, 49, 3058–3064. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, Y; Takahashi, H; Satoh, T; Okazaki, Y; Mizuki, N; Takahashi, K; Ikezawa, Z; Kuwana, M. Natural killer cells control a T-helper 1 response in patients with Behcet’s disease. Arthritis Res Ther. 2010, 12, R80. [Google Scholar] [CrossRef]
- Le Dréan, E; Vély, F; Olcese, L; Cambiaggi, A; Guia, S; Krystal, G; Gervois, N; Moretta, A; Jotereau, F; Vivier, E. Inhibition of antigen-induced T cell response and antibody-induced NK cell cytotoxicity by NKG2A: association of NKG2A with SHP-1 and SHP-2 protein-tyrosine phosphatases. Eur J Immunol. 1998, 28, 264–276. [Google Scholar] [CrossRef]
- Lanier, LL; Corliss, B; Wu, J; Phillips, JH. Association of DAP12 with activating CD94/NKG2C NK cell receptors. Immunity. 1998, 8, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Orbelyan, GA; Tang, F; Sally, B; Solus, J; Meresse, B; Ciszewski, C; Grenier, JC; Barreiro, LB; Lanier, LL; Jabri, B. Human NKG2E is expressed and forms an intracytoplasmic complex with CD94 and DAP12. J Immunol. 2014, 193, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Kim, DK; Kabat, J; Borrego, F; Sanni, TB; You, CH; Coligan, JE. Human NKG2F is expressed and can associate with DAP12. Mol Immunol. 2004, 41, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Shum, BP; Flodin, LR; Muir, DG; Rajalingam, R; Khakoo, SI; Cleland, S; Guethlein, LA; Uhrberg, M; Parham, P. Conservation and variation in human and common chimpanzee CD94 and NKG2 genes. J Immunol. 2002, 168, 240–252. [Google Scholar] [CrossRef]
- Averdam, A; Kuhl, H; Sontag, M; Becker, T; Hughes, AL; Reinhardt, R; Walter, L. Genomics and diversity of the common marmoset monkey NK complex. J Immunol. 2007, 178, 7151–7161. [Google Scholar] [CrossRef]
- Gilfillan, S; Ho, EL; Cella, M; Yokoyama, WM; Colonna, M. NKG2D recruits two distinct adapters to trigger NK cell activation and costimulation. Nat Immunol. 2002, 3, 1150–1155. [Google Scholar] [CrossRef]
- Diefenbach, A; Tomasello, E; Lucas, M; Jamieson, AM; Hsia, JK; Vivier, E; Raulet, DH. Selective associations with signaling proteins determine stimulatory versus costimulatory activity of NKG2D. Nat Immunol. 2002, 3, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Zompi, S; Hamerman, JA; Ogasawara, K; Schweighoffer, E; Tybulewicz, VL; Di Santo, JP; Lanier, LL; Colucci, F. NKG2D triggers cytotoxicity in mouse NK cells lacking DAP12 or Syk family kinases. Nat Immunol. 2003, 4, 565–572. [Google Scholar] [CrossRef]
- Wu, J; Cherwinski, H; Spies, T; Phillips, JH; Lanier, LL. DAP10 and DAP12 form distinct, but functionally cooperative, receptor complexes in natural killer cells. J Exp Med. 2000, 192, 1059–1068. [Google Scholar] [CrossRef]
- André, P; Castriconi, R; Espéli, M; Anfossi, N; Juarez, T; Hue, S; Conway, H; Romagné, F; Dondero, A; Nanni, M; Caillat-Zucman, S; Raulet, DH; Bottino, C; Vivier, E; Moretta, A; Paul, P. Comparative analysis of human NK cell activation induced by NKG2D and natural cytotoxicity receptors. Eur J Immunol. 2004, 34, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Lanier, LL; Corliss, BC; Wu, J; Leong, C; Phillips, JH. Immunoreceptor DAP12 bearing a tyrosine-based activation motif is involved in activating NK cells. Nature. 1998, 391, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Karimi, MA; Aguilar, O; Zou, B; Bachmann, MH; Carlyle, JR; Baldwin, CL; Kambayashi, T. A truncated human NKG2D splice isoform negatively regulates NKG2D-mediated function. J Immunol. 2014, 193, 2764–2771. [Google Scholar] [CrossRef]
- Caldas, P; Luz, M; Baseggio, S; Andrade, R; Sobral, D; Grosso, AR. Transcription readthrough is prevalent in healthy human tissues and associated with inherent genomic features. Commun Biol. 2024, 7, 100. [Google Scholar] [CrossRef]
- Padula, MC; Leccese, P; Lascaro, N; Padula, AA; Carbone, T; Martelli, G; D’Angelo, S. A First Step for the Molecular Characterization of Neurological Involvement of Behcet Syndrome: an Italian Pivotal Study. J Mol Neurosci. 2021, 71, 1284–1289. [Google Scholar] [CrossRef]
- Oğuz, AK; Yılmaz, ST; Oygür, ÇŞ; Çandar, T; Sayın, I; Kılıçoğlu, SS; Ergün, İ; Ateş, A; Özdağ, H; Akar, N. Behçet’s: A Disease or a Syndrome? Answer from an Expression Profiling Study. PLoS One. 2016, 11, e0149052. [Google Scholar] [CrossRef]
- Yang, Y; Tan, H; Deng, B; Yu, H; Su, G; Hu, J; Cao, Q; Yuan, G; Kijlstra, A; Yang, P. Genetic polymorphisms of C-type lectin receptors in Behcet’s disease in a Chinese Han population. Sci Rep. 2017, 7, 5348. [Google Scholar] [CrossRef] [PubMed]
- Parmar, AS; Alakulppi, N; Paavola-Sakki, P; Kurppa, K; Halme, L; Färkkilä, M; Turunen, U; Lappalainen, M; Kontula, K; Kaukinen, K; Mäki, M; Lindfors, K; Partanen, J; Sistonen, P; Mättö, J; Wacklin, P; Saavalainen, P; Einarsdottir, E. Association study of FUT2 (rs601338) with celiac disease and inflammatory bowel disease in the Finnish population. Tissue Antigens 2012, 80, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Omata, Y; Aoki, R; Aoki-Yoshida, A; Hiemori, K; Toyoda, A; Tateno, H; Suzuki, C; Takayama, Y. Reduced fucosylation in the distal intestinal epithelium of mice subjected to chronic social defeat stress. Sci Rep. 2018, 8, 13199. [Google Scholar] [CrossRef] [PubMed]
- Xavier, JM; Shahram, F; Sousa, I; Davatchi, F; Matos, M; Abdollahi, BS; Sobral, J; Nadji, A; Oliveira, M; Ghaderibarim, F; Shafiee, NM; Oliveira, SA. FUT2: filling the gap between genes and environment in Behçet’s disease? Ann Rheum Dis. 2015, 74, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Shields, RL; Lai, J; Keck, R; O’Connell, LY; Hong, K; Meng, YG; Weikert, SH; Presta, LG. Lack of fucose on human IgG1 N-linked oligosaccharide improves binding to human Fcgamma RIII and antibody-dependent cellular toxicity. J Biol Chem. 2002, 277, 26733–26740. [Google Scholar] [CrossRef]
- Dogra, P; Rancan, C; Ma, W; Toth, M; Senda, T; Carpenter, DJ; Kubota, M; Matsumoto, R; Thapa, P; Szabo, PA; Li Poon, MM; Li, J; Arakawa-Hoyt, J; Shen, Y; Fong, L; Lanier, LL; Farber, DL. Tissue Determinants of Human NK Cell Development, Function, and Residence. Cell. 2020, 180, 749–763.e13. [Google Scholar] [CrossRef]
- Charteris, DG; Barton, K; McCartney, AC; Lightman, SL. CD4+ lymphocyte involvement in ocular Behcet’s disease. Autoimmunity. 1992, 12, 201–206. [Google Scholar] [CrossRef]
- Torcellan, T; Friedrich, C; Doucet-Ladevèze, R; Ossner, T; Solé, VV; Riedmann, S; Ugur, M; Imdahl, F; Rosshart, SP; Arnold, SJ; Gomez de Agüero, M; Gagliani, N; Flavell, RA; Backes, S; Kastenmüller, W; Gasteiger, G. Circulating NK cells establish tissue residency upon acute infection of skin and mediate accelerated effector responses to secondary infection. Immunity 2024, 57, 124–140.e7. [Google Scholar] [CrossRef]
- Schuster, IS; Sng, XYX; Lau, CM; Powell, DR; Weizman, OE; Fleming, P; Neate, GEG; Voigt, V; Sheppard, S; Maraskovsky, AI; Daly, S; Koyama, M; Hill, GR; Turner, SJ; O’Sullivan, TE; Sun, JC; Andoniou, CE; Degli-Esposti, MA. Infection induces tissue-resident memory NK cells that safeguard tissue health. Immunity. 2023, 56, 531–546.e6. [Google Scholar] [CrossRef]
- Terrén, I; Orrantia, A; Astarloa-Pando, G; Amarilla-Irusta, A; Zenarruzabeitia, O; Borrego, F. Cytokine-Induced Memory-Like NK Cells: From the Basics to Clinical Applications. Front Immunol. 2022, 13, 884648. [Google Scholar] [CrossRef]
- Cooper, MA; Elliott, JM; Keyel, PA; Yang, L; Carrero, JA; Yokoyama, WM. Cytokine-induced memory-like natural killer cells. Proc Natl Acad Sci U S A. 2009, 106, 1915–1919. [Google Scholar] [CrossRef]
- Romee, R; Schneider, SE; Leong, JW; Chase, JM; Keppel, CR; Sullivan, RP; Cooper, MA; Fehniger, TA. Cytokine activation induces human memory-like NK cells. Blood 2012, 120, 4751–4760. [Google Scholar] [CrossRef] [PubMed]
- Romee, R; Rosario, M; Berrien-Elliott, MM; Wagner, JA; Jewell, BA; Schappe, T; Leong, JW; Abdel-Latif, S; Schneider, SE; Willey, S; Neal, CC; Yu, L; Oh, ST; Lee, YS; Mulder, A; Claas, F; Cooper, MA; Fehniger, TA. Cytokine-induced memory-like natural killer cells exhibit enhanced responses against myeloid leukemia. Sci Transl Med. 2016, 8, 357ra123. [Google Scholar] [CrossRef] [PubMed]
- Ewen, EM; Pahl, JHW; Miller, M; Watzl, C; Cerwenka, A. KIR downregulation by IL-12/15/18 unleashes human NK cells from KIR/HLA-I inhibition and enhances killing of tumor cells. Eur J Immunol. 2018, 48, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, S; Sun, JC. Virus-specific NK cell memory. J Exp Med. 2021, 218, e20201731. [Google Scholar] [CrossRef] [PubMed]
- Foley, B; Cooley, S; Verneris, MR; Curtsinger, J; Luo, X; Waller, EK; Anasetti, C; Weisdorf, D; Miller, JS. Human cytomegalovirus (CMV)-induced memory-like NKG2C(+) NK cells are transplantable and expand in vivo in response to recipient CMV antigen. J Immunol. 2012, 189, 5082–5088. [Google Scholar] [CrossRef]
- Zhang, T; Scott, JM; Hwang, I; Kim, S. Cutting edge: antibody-dependent memory-like NK cells distinguished by FcRγ deficiency. J Immunol. 2013, 190, 1402–1406. [Google Scholar] [CrossRef]
- Lee, J; Zhang, T; Hwang, I; Kim, A; Nitschke, L; Kim, M; Scott, JM; Kamimura, Y; Lanier, LL; Kim, S. Epigenetic modification and antibody-dependent expansion of memory-like NK cells in human cytomegalovirus-infected individuals. Immunity. 2015, 42, 431–442. [Google Scholar] [CrossRef]
- Lee, EB; Kwon, YJ; Shin, KC; Song, YW; Park, CG; Hwang, ES; Cha, CY. Decreased serum level of antibody against human cytomegalovirus in patients with Behçet’s disease. Rheumatol Int. 2005, 25, 33–36. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




