Submitted:
09 March 2026
Posted:
12 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Setting and Design
2.2. Data Source and Extraction
2.3. Data Preparation
2.4. Data Analysis
3. Results
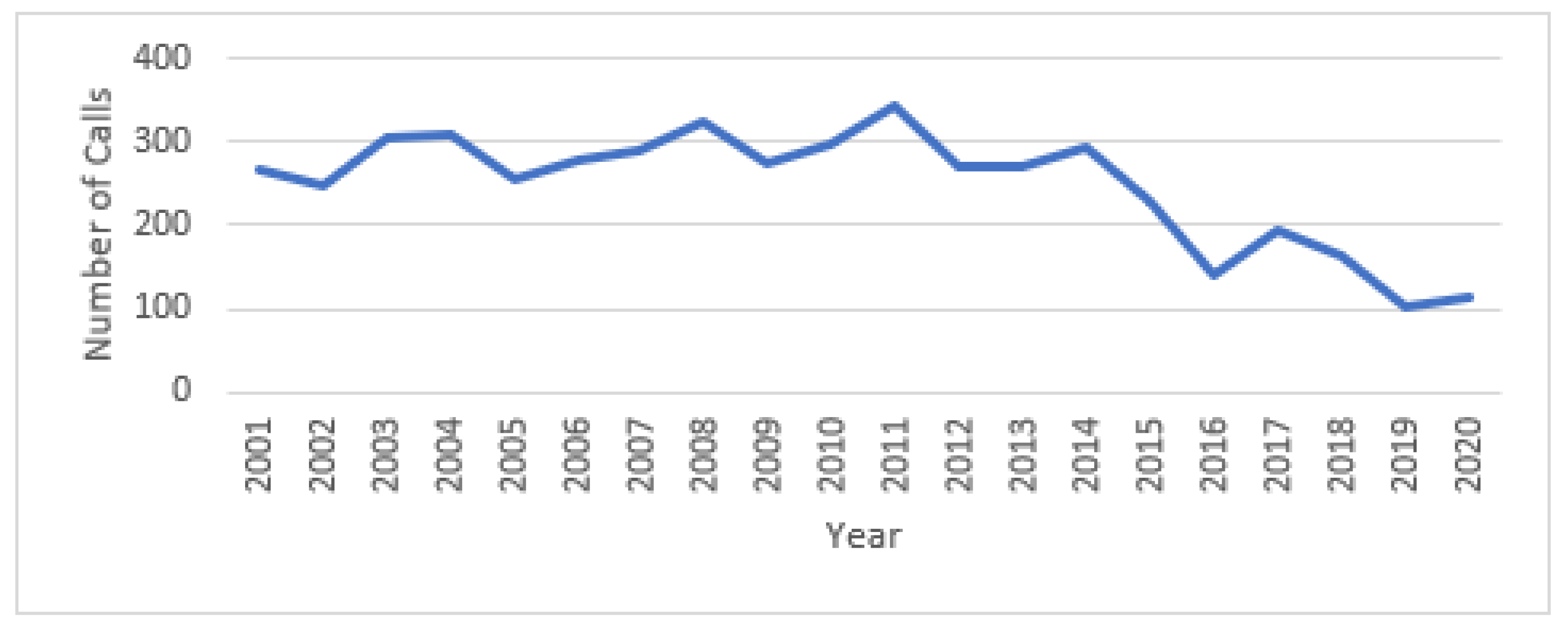
3.1. Call Characteristics
3.2. Characteristics of Callers
3.3. Time Period of Concern: Gestation or Age of Infant
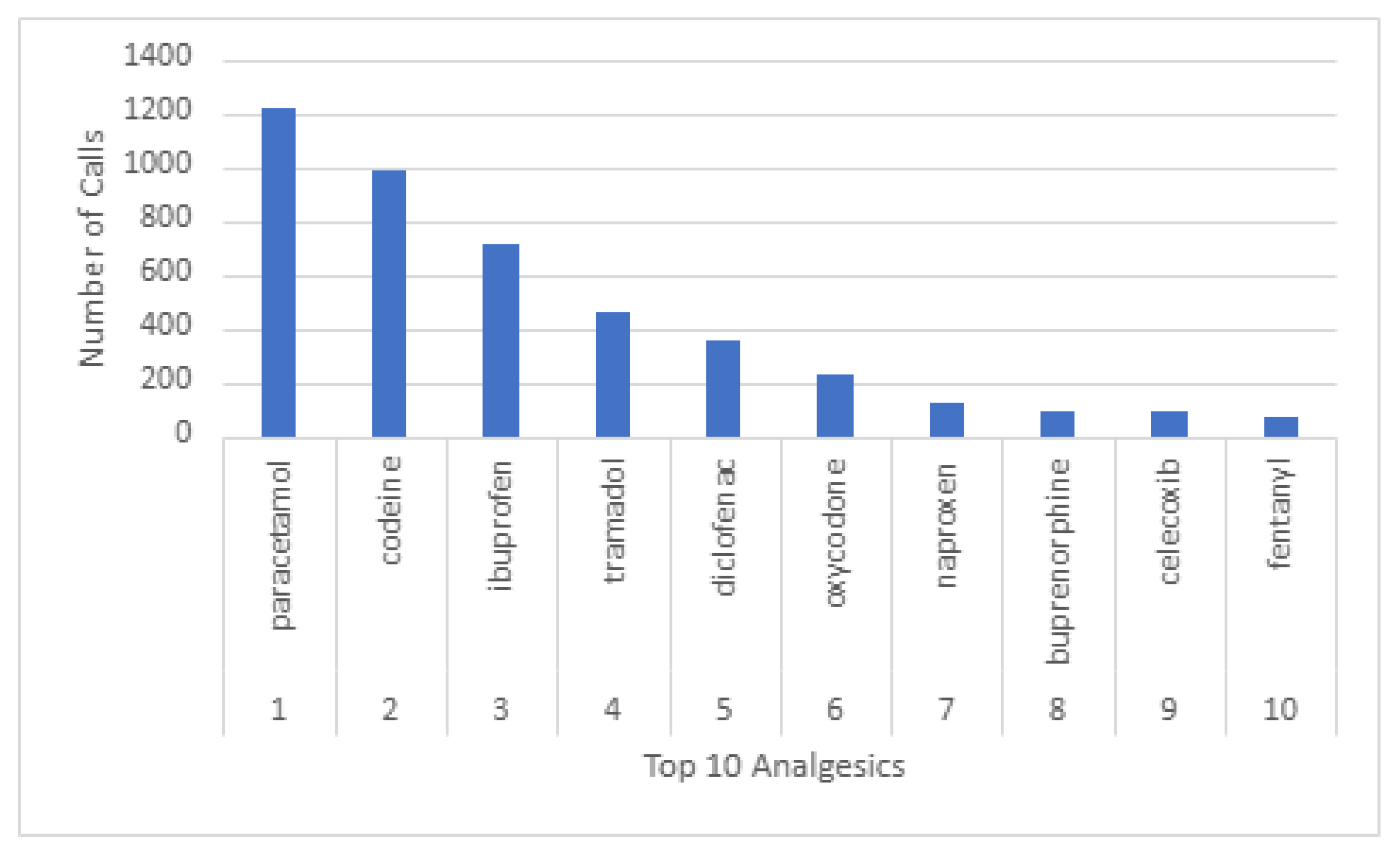
3.4. Analgesic Medication
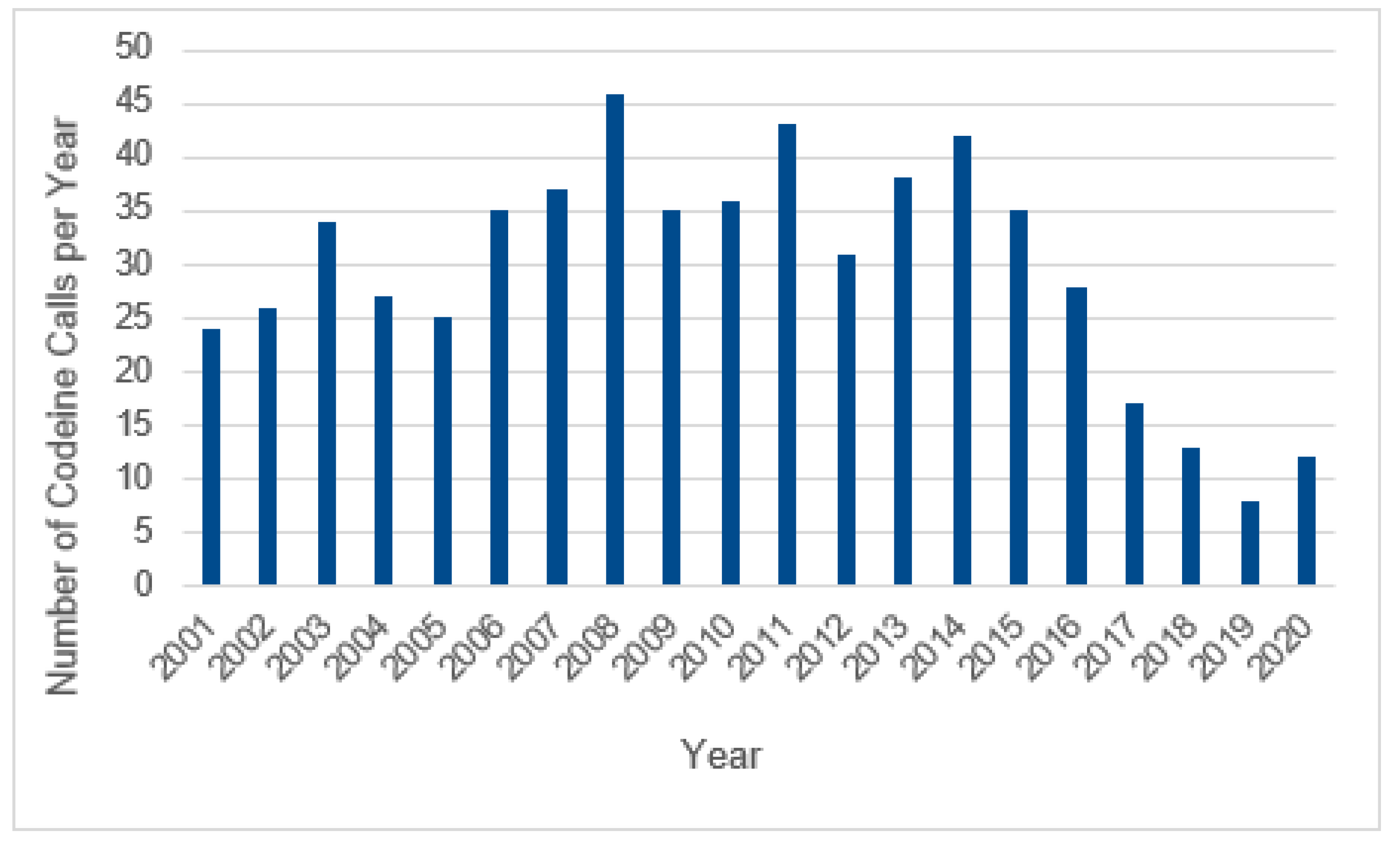
3.3.1. Codeine Use in Breastfeeding
4. Discussion
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| KEMH | King Edward Memorial Hospital |
| OMIS | Obstetric Medicines Information Service |
References
- Henry, A; Crowther, C. Patterns of medication use during and prior to pregnancy: the MAP study. Aust N Z J Obstet Gynaecol. 2000, 40(2), 165–72. [Google Scholar] [CrossRef] [PubMed]
- ACOG Committee Opinion No. 377: Research involving women. Obstet Gynecol. 2007, 110(3), 731–6. [CrossRef]
- Hegedus, E; Oakes, DJ; Hill, M; Ritchie, HE; Kennedy, DS. Calls to a Major Teratogen Information Service Regarding Exposures During Breastfeeding. Breastfeed Med. 2019, 14(9), 674–9. [Google Scholar] [CrossRef]
- Lim, JM; Sullivan, E; Kennedy, D. MotherSafe: review of three years of counselling by an Australian Teratology Information Service. Aust N Z J Obstet Gynaecol. 2009, 49(2), 168–72. [Google Scholar] [CrossRef]
- Bonati, M; Bortolus, R; Marchetti, F; Romero, M; Tognoni, G. Drug use in pregnancy: an overview of epidemiological (drug utilization) studies. Eur J Clin Pharmacol. 1990, 38(4), 325–8. [Google Scholar] [CrossRef]
- Lupattelli, A; Spigset, O; Twigg, MJ; Zagorodnikova, K; Mårdby, AC; Moretti, ME; et al. Medication use in pregnancy: a cross-sectional, multinational web-based study. BMJ Open. 2014, 4(2), e004365. [Google Scholar] [CrossRef] [PubMed]
- Eyal, S. Use of Therapeutics in Pregnancy and Lactation. Pharmaceutical Research 2018, 35(5), 107. [Google Scholar] [CrossRef] [PubMed]
- Jayawickrama, HS; Amir, LH; Pirotta, MV. GPs’ decision-making when prescribing medicines for breastfeeding women: Content analysis of a survey. BMC Res Notes 2010, 3, 82. [Google Scholar] [CrossRef]
- Campbell, SC; Kast, TT; Kamyar, M; Robertson, J; Sherwin, CM. Calls to a teratogen information service regarding potential exposures in pregnancy and breastfeeding. BMC Pharmacol Toxicol. 2016, 17(1), 33. [Google Scholar] [CrossRef]
- Adam, MP; Polifka, JE; Friedman, JM. Evolving knowledge of the teratogenicity of medications in human pregnancy. Am J Med Genet C Semin Med Genet. 2011, 157c(3), 175–82. [Google Scholar] [CrossRef]
- Hämeen-Anttila, K; Nordeng, H; Kokki, E; Jyrkkä, J; Lupattelli, A; Vainio, K; et al. Multiple information sources and consequences of conflicting information about medicine use during pregnancy: a multinational Internet-based survey. J Med Internet Res. 2014, 16(2), e60. [Google Scholar] [CrossRef]
- Alper, BS; Hand, JA; Elliott, SG; Kinkade, S; Hauan, MJ; Onion, DK; et al. How much effort is needed to keep up with the literature relevant for primary care? J Med Libr Assoc. 2004, 92(4), 429–37. [Google Scholar] [PubMed]
- Lupattelli, A.; Spigset, O.; Twigg, M.J.; Zagorodnikova, K.; Mårdby, A.C.; Moretti, M.E.; Drozd, M.; Panchaud, A.; Hämeen-Anttila, K.; Rieutord, A.; Juraski, R.G. Medication use in pregnancy: a cross-sectional, multinational web-based study. BMJ open 2014, 4(2), e004365. Available online: https://bmjopen.bmj.com/content/bmjopen/4/2/e004365.full.pdf. [CrossRef]
- Werler, MM; Kerr, SM; Ailes, EC; Reefhuis, J; Gilboa, SM; Browne, ML; Kelley, KE; Hernandez-Diaz, S; Smith-Webb, RS; Garcia, MH; Mitchell, AA. Patterns of prescription medication use during the first trimester of pregnancy in the United States, 1997–2018. Clinical Pharmacology & Therapeutics 2023, 114(4), 836–44. Available online: https://ascpt.onlinelibrary.wiley.com/doi/full/10.1002/cpt.2981. [CrossRef]
- Henry, A; Crowther, C. Patterns of medication use during and prior to pregnancy: the MAP study. Australian and New Zealand Journal of Obstetrics and Gynaecology 2000, 40(2), 165–72. Available online: https://obgyn.onlinelibrary.wiley.com/doi/abs/10.1111/j.1479-828X.2000.tb01140.x?sid=nlm%3Apubmed. [CrossRef]
- Anderson, PO. Drug use during breast-feeding. Clin Pharm. 1991, 10(8), 594–624. [Google Scholar] [PubMed]
- Schaefer, C. [Drug safety in pregnancy-a particular challenge]. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2018, 61(9), 1129–38. [Google Scholar] [CrossRef] [PubMed]
- Hotham, N; Hotham, E. Drugs in breastfeeding. Aust Prescr. 2015, 38(5), 156–9. [Google Scholar] [CrossRef]
- Lee, K; Hoti, K; Hughes, JD; Emmerton, LM. Consumer Use of “Dr Google”: A Survey on Health Information-Seeking Behaviors and Navigational Needs. J Med Internet Res. 2015, 17(12), e288. [Google Scholar] [CrossRef]
- Lebedevs, T; Broderick, C; Teoh, S; Mukadam, N; Frestel, J. Enhancing Medicine Information Services: A Future-Proof, Low-Cost Data Management Strategy ‘. Journal of the Society for Clinical Data Management 2025, 1. [Google Scholar] [CrossRef]
- Stephens, A; Brodribb, W; McGuire, T; Deckx, L. Breastfeeding questions to medicines call centres from the Australian public and health professionals. Aust J Prim Health 2018, 24(5), 409–16. [Google Scholar] [CrossRef]
- Hauck, YL; Fenwick, J; Dhaliwal, SS; Butt, J. A Western Australian survey of breastfeeding initiation, prevalence and early cessation patterns. Matern Child Health J. 2011, 15(2), 260–8. [Google Scholar] [CrossRef] [PubMed]
- Hale, T. Medications and Mother’s Milk; Springer Publishing: New York, U.S, 2019; Available online: https://www-halesmeds-com.kelibresources.health.wa.gov.au/.
- Stephens, A; Brodribb, W; McGuire, T; Deckx, L. Breastfeeding questions to medicines call centres from the Australian public and health professionals. Australian Journal of Primary Health 2018, 24(5), 409–16. [Google Scholar] [CrossRef] [PubMed]
- Alwan, S; Conover, EA; Harris-Sagaribay, L; Lamm, SH; Lavigne, SV; Lusskin, SI; et al. Paracetamol use in pregnancy - caution over causal inference from available data. Nat Rev Endocrinol 2021. [Google Scholar] [CrossRef] [PubMed]
- US Food and Drug Administration. News Release: FDA Responds to Evidence of Possible Association Between Autism and Acetaminophen Use During Pregnancy. 2025. Available online: https://www.fda.gov/news-events/press-announcements/fda-responds-evidence-possible-association-between-autism-and-acetaminophen-use-during-pregnancy.
- Bauer, AZ; Swan, SH; Kriebel, D; Liew, Z; Taylor, HS; Bornehag, CG; et al. Reply to ‘Paracetamol use in pregnancy - caution over causal inference from available data’; ‘Handle with care - interpretation, synthesis and dissemination of data on paracetamol in pregnancy’. Nat Rev Endocrinol 2021. [Google Scholar] [CrossRef]
- Therapeutic Goods Administration. Paracetamol use in pregnancy. Australian Government 2025. February 2026. Available online: https://www.tga.gov.au/news/media-releases/paracetamol-use-pregnancy.
- The Australian and New Zealand College of Anaesthetists. Statement on paracetamol use in pregnancy. 2025. Available online: https://www.anzca.edu.au/news-and-safety-alerts/paracetamol-use-in-pregnancy.
- Royal College of Obstetricians and Gynaecologists. Paracetamol in pregnancy. 2026. Available online: https://www.rcog.org.uk/news/paracetamol-in-pregnancy/.
- Changes to patient access for medicines containing codeine Canberra, Australia2018. February 2022. Available online: https://www.tga.gov.au/codeine-info-hub.
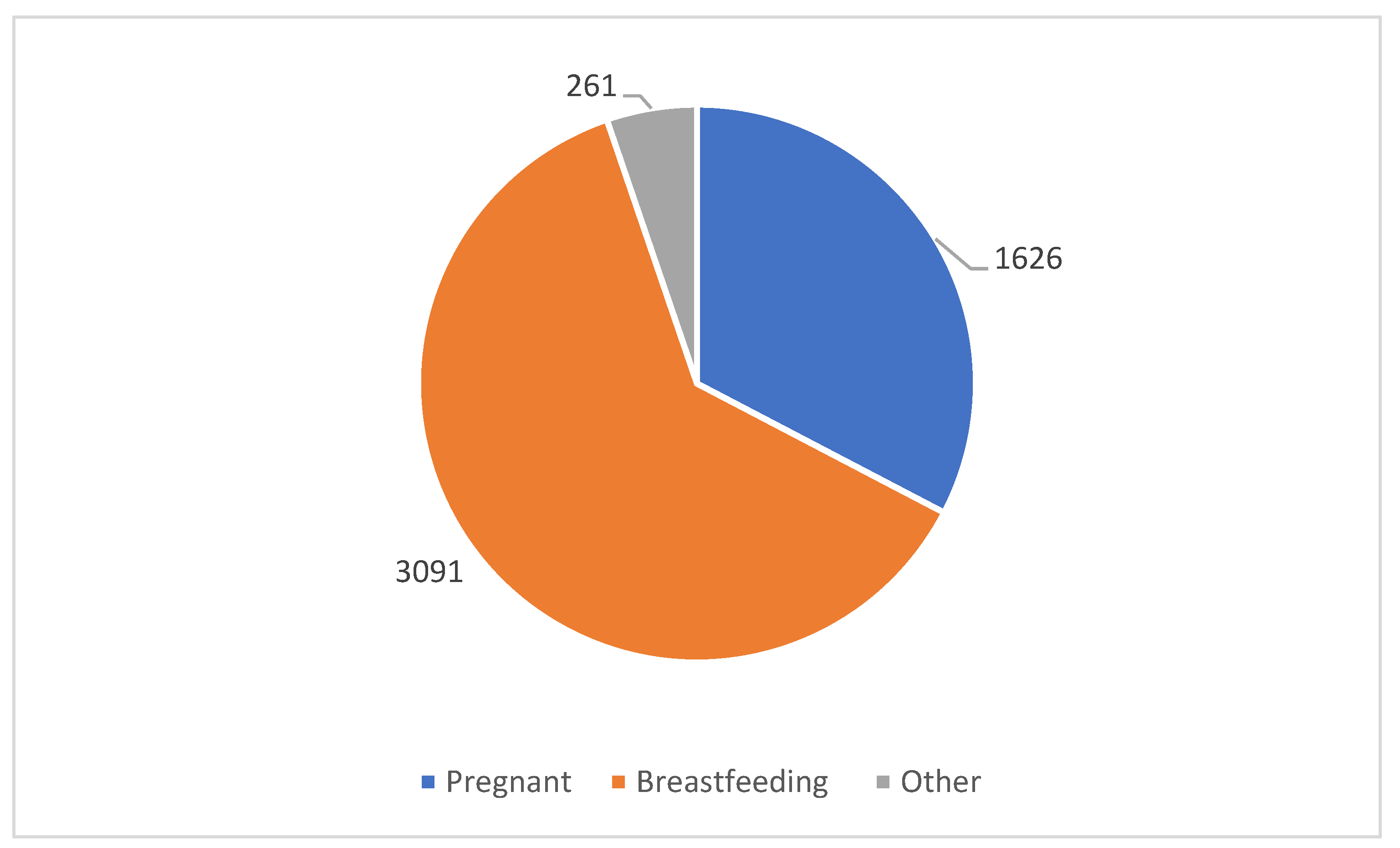
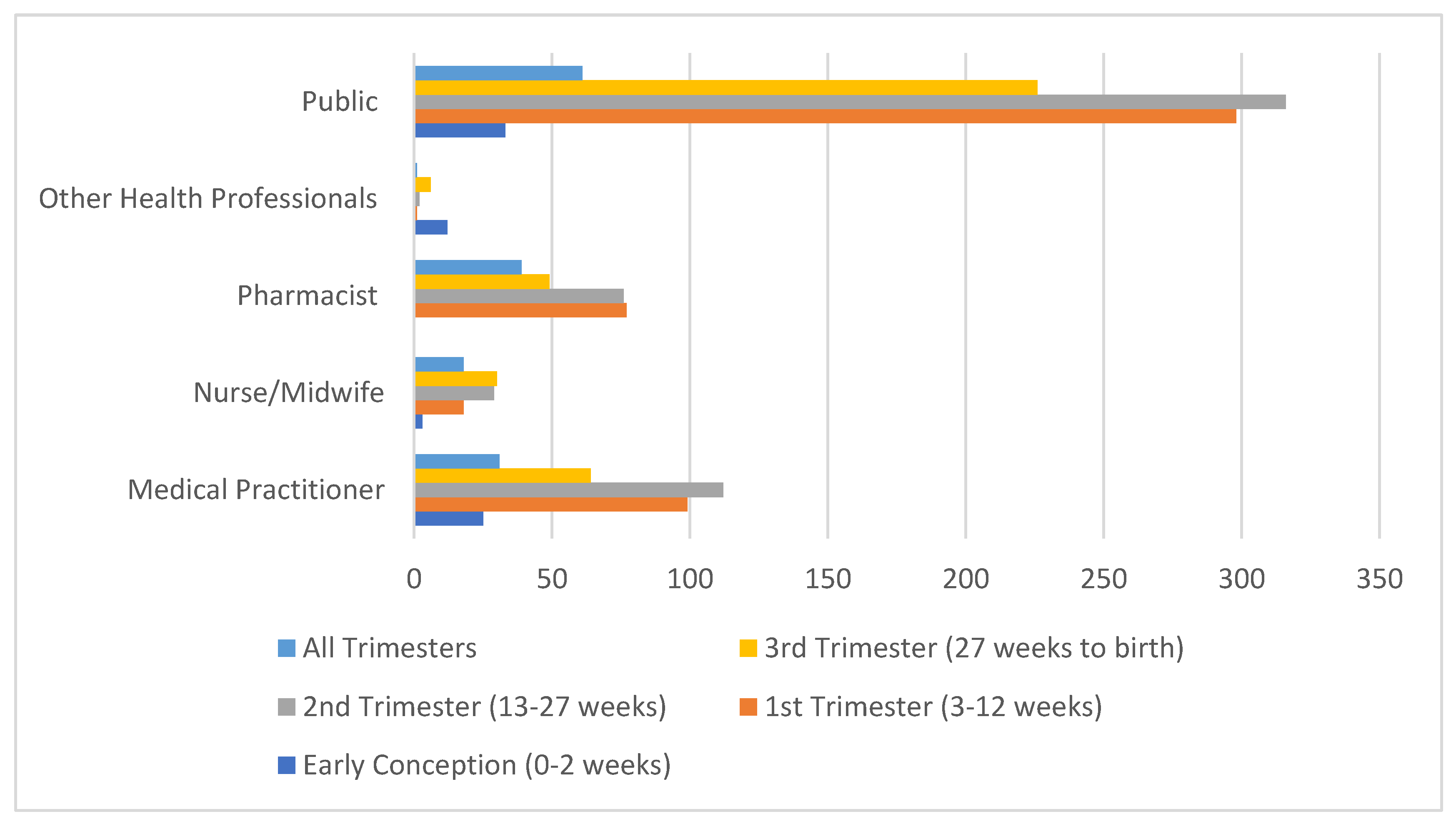
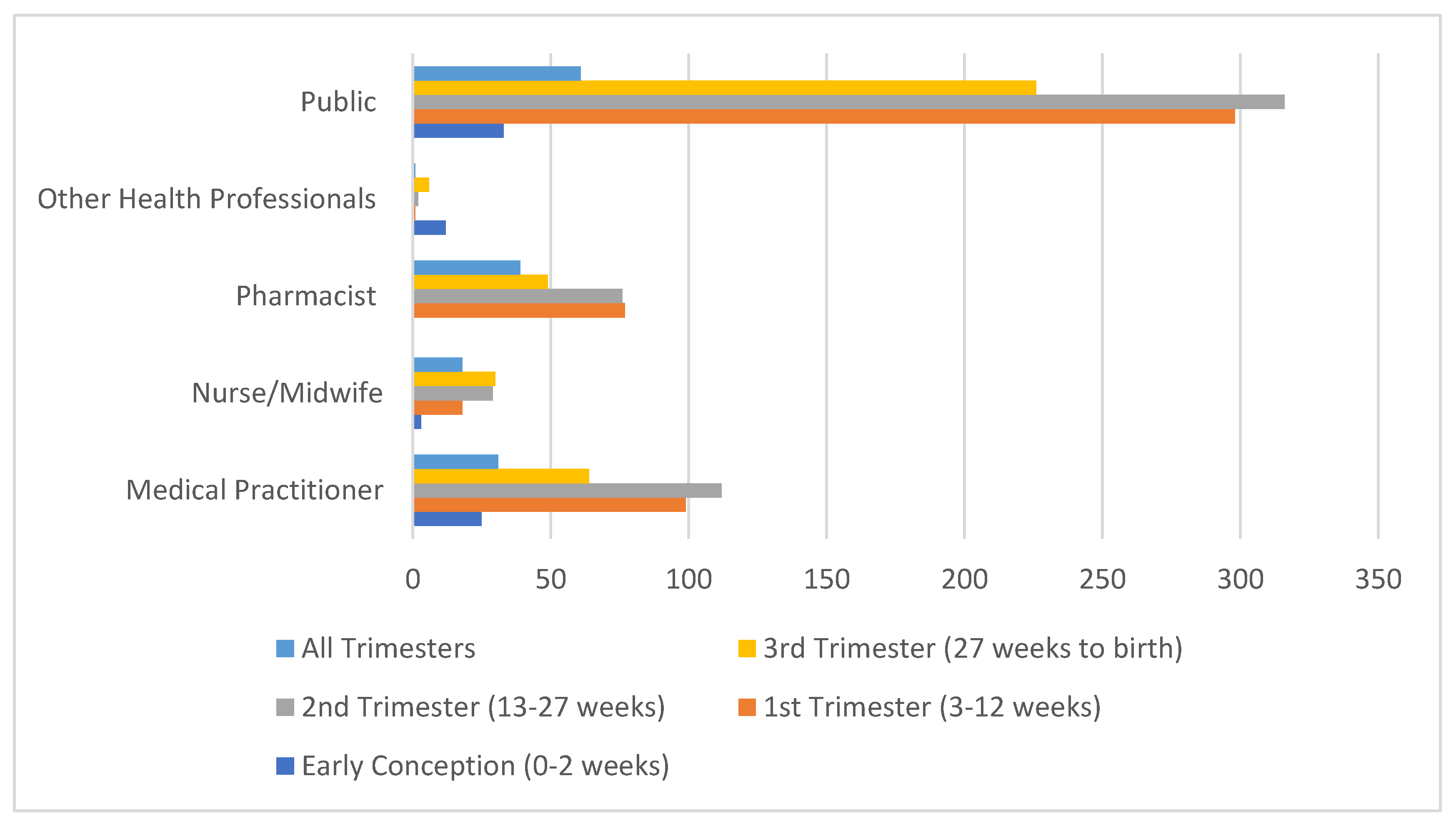
| Type of Caller | Medicines Information in Pregnancy | Medicines Information in Breastfeeding | Other | Total |
| Medical Practitioner | 326 (20.0%) | 365 (11.8%) | 108 (41.4%) | 799 (16.1%) |
| Nurse/Midwife | 91 (5.6%) | 366 (11.8%) | 49 (18.8%) | 506 (10.2%) |
| Pharmacist | 256 (15.7%) | 287 (9.3%) | 74 (28.4%) | 617 (12.4%) |
| Other Health Professionals | 10 (0.6%) | 13 (0.4%) | 2(0.8%) | 25 (0.5%) |
| Public | 942 (57.9%) | 2,0660 (66.6%) | 28 (10.7%) | 3,030 (60.9%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




