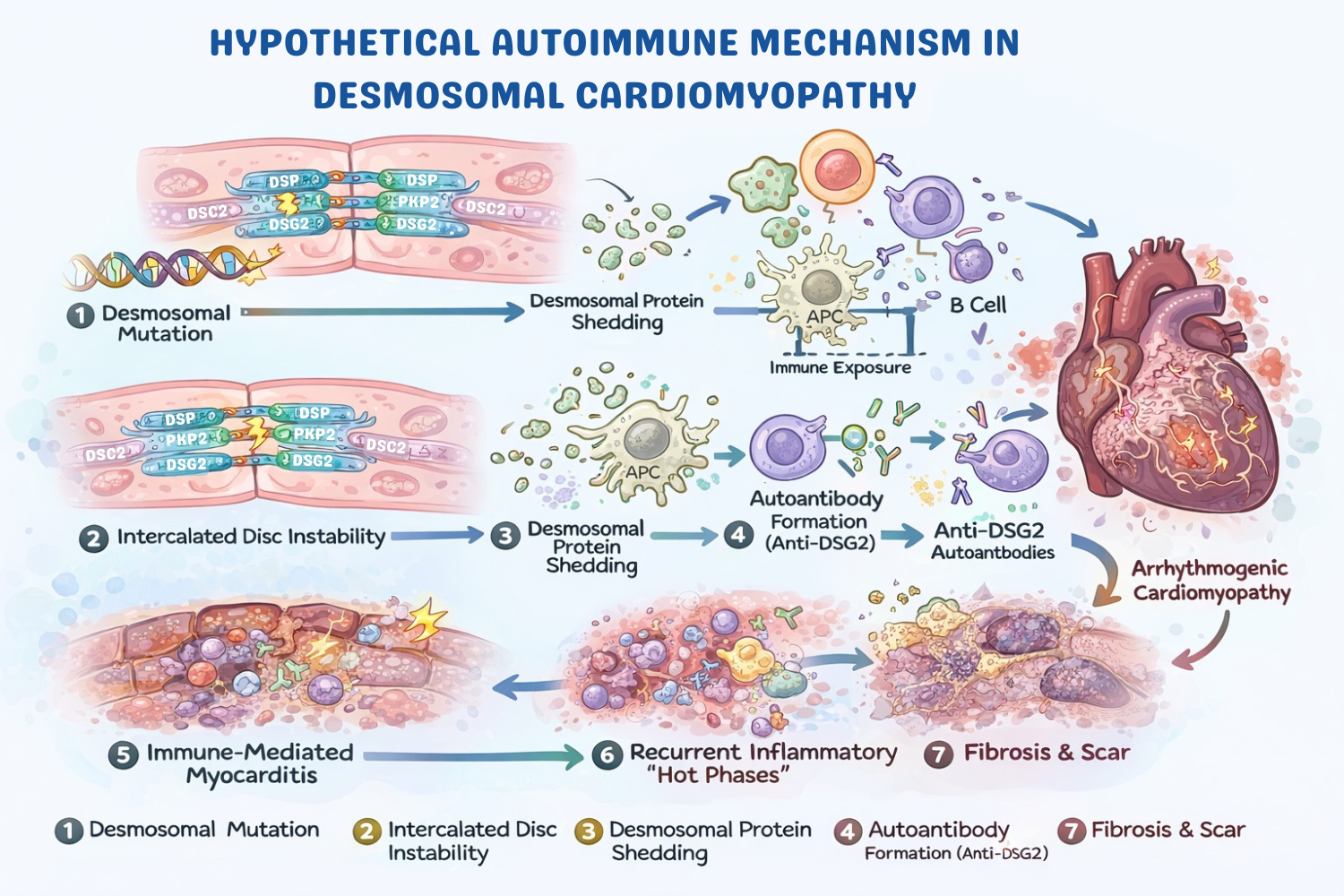
Acute myocarditis has traditionally been regarded as an acquired inflammatory disorder of the myocardium, most commonly triggered by viral infection or immune-mediated injury. However, growing evidence suggests that in a subset of patients, myocarditis may represent the initial clinical manifestation of an underlying genetic cardiomyopathy rather than a purely inflammatory disease. Recent advances in molecular genetics, cardiac magnetic resonance imaging, and translational pathology have revealed a significant overlap between myocarditis and inherited cardiomyopathies, particularly those related to desmosomal dysfunction. Desmosomal gene variants—most frequently involving desmoplakin, plakophilin-2, and desmoglein-2—have been increasingly identified in patients presenting with myocarditis-like syndromes characterized by chest pain, troponin elevation, and imaging findings fulfilling Lake Louise criteria. Importantly, these presentations often demonstrate recurrent inflammatory episodes, ventricular arrhythmias, and progressive myocardial fibrosis that ultimately evolve into the structural phenotype of arrhythmogenic cardiomyopathy. Emerging mechanistic data further suggest that structural instability of the intercalated disc may trigger autoimmune responses through exposure of desmosomal antigens and generation of disease-specific autoantibodies such as anti-DSG2. This review explores the evolving concept of myocarditis as an inflammatory manifestation of genetically mediated cardiomyopathy and highlights the diagnostic and mechanistic implications of this gene–immune interaction.