Submitted:
09 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
Background
Methods
Statistical Analysis
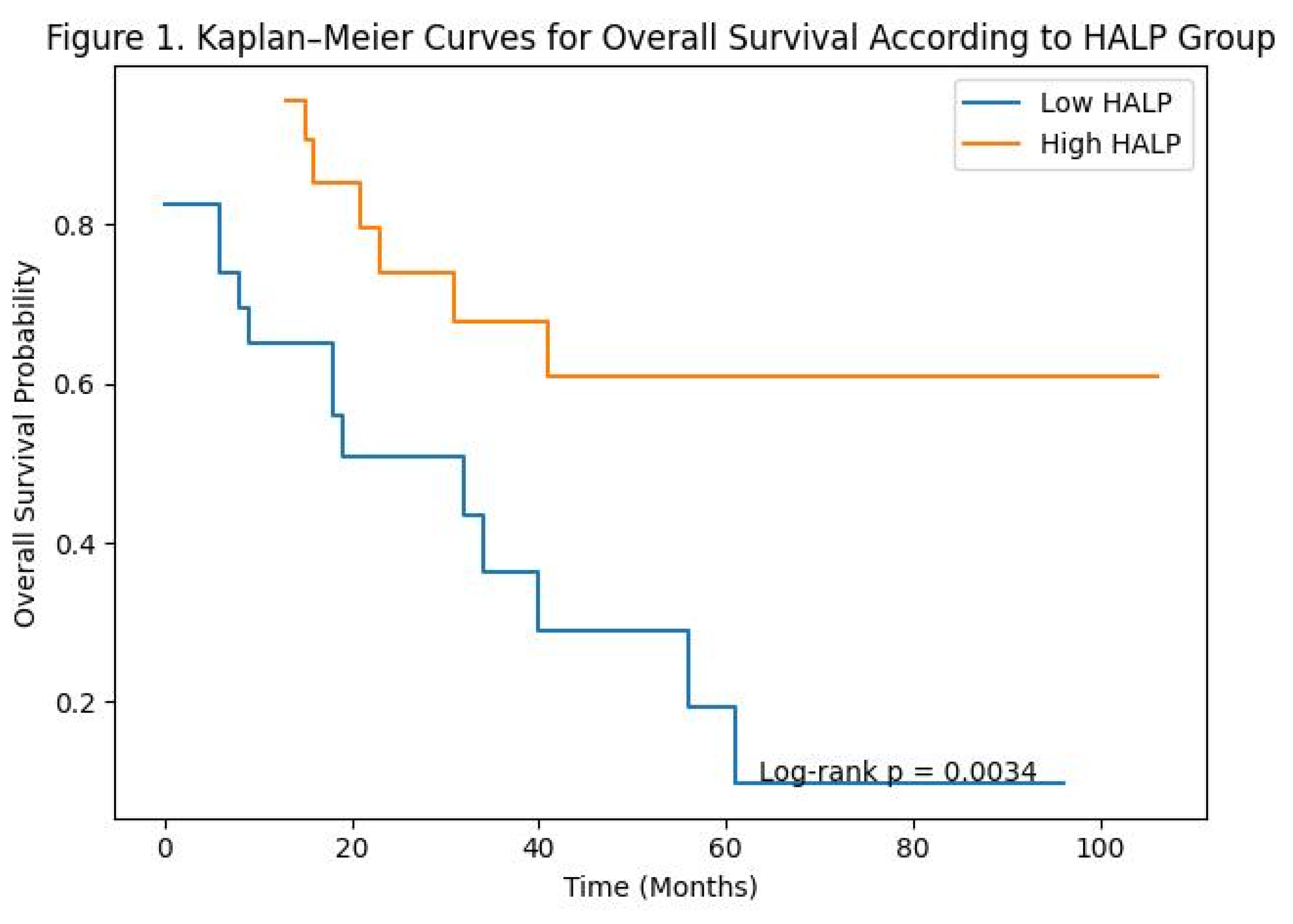
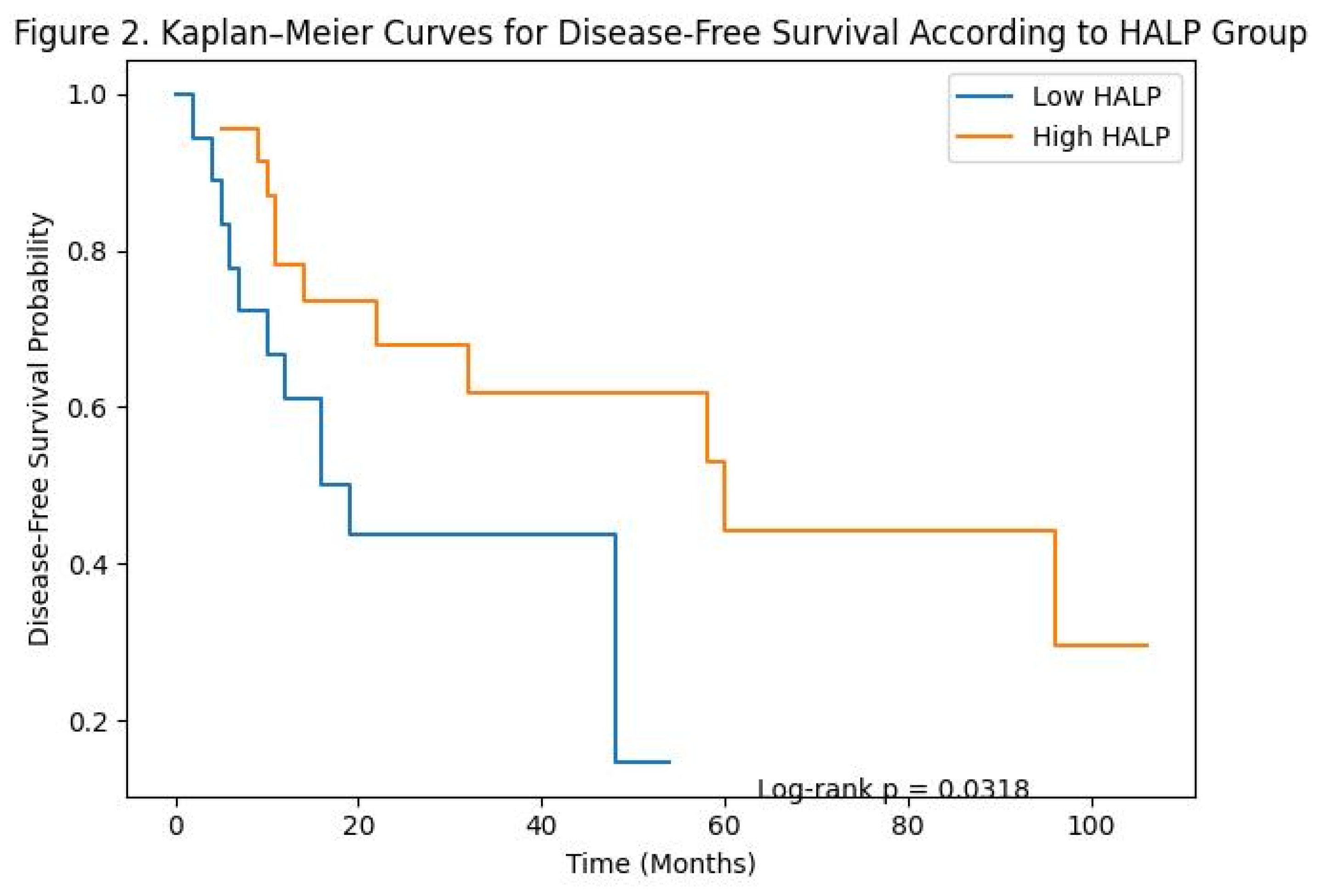
Results
OAS
DFS
Discussion
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Matsui, Y.; et al. The clinical significance of perioperative inflammatory index as a prognostic factor for patients with retroperitoneal soft tissue sarcoma. International Journal of Clinical Oncology 2022, 27(6), 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Stojadinovic, A.; et al. Analysis of the prognostic significance of microscopic margins in 2,084 localized primary adult soft tissue sarcomas. Annals of surgery 2002, 235(3), 424–434. [Google Scholar] [CrossRef] [PubMed]
- Kattan, M.W.; Leung, D.H.; Brennan, M.F. Postoperative nomogram for 12-year sarcoma-specific death. Journal of clinical oncology 2002, 20(3), 791–796. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420(6917), 860–867. [Google Scholar] [PubMed]
- Lorente, D.; et al. Baseline neutrophil–lymphocyte ratio (NLR) is associated with survival and response to treatment with second-line chemotherapy for advanced prostate cancer independent of baseline steroid use. Annals of Oncology 2015, 26(4), 750–755. [Google Scholar] [CrossRef] [PubMed]
- Zhai, B.; et al. Predictive value of the hemoglobin, albumin, lymphocyte, and platelet (HALP) score and lymphocyte-to-monocyte ratio (LMR) in patients with non-small cell lung cancer after radical lung cancer surgery. Annals of Translational Medicine 2021, 9(12), p. 976. [Google Scholar] [CrossRef] [PubMed]
- Yazici, H.; Yegen, S.C.; Yegen, C. Is systemic inflammatory response index (SIRI) a reliable tool for prognosis of gastric cancer patients without neoadjuvant therapy? Cureus 2023, 15(3). [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-L.; et al. Prognostic significance of the combination of preoperative hemoglobin, albumin, lymphocyte and platelet in patients with gastric carcinoma: a retrospective cohort study. Oncotarget 2015, 6(38), p. 41370. [Google Scholar] [CrossRef] [PubMed]
- Gronchi, A.; et al. Soft tissue and visceral sarcomas: ESMO–EURACAN–GENTURIS Clinical Practice Guidelines for diagnosis, treatment and follow-up☆. Annals of Oncology 2021, 32(11), 1348–1365. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; et al. Hemoglobin, albumin, lymphocyte, and platelet (HALP) score and cancer prognosis: a systematic review and meta-analysis of 13,110 patients. International immunopharmacology 2023, 114, 109496. [Google Scholar] [CrossRef] [PubMed]
- Farag, C.M.; et al. What is hemoglobin, albumin, lymphocyte, platelet (HALP) score? A comprehensive literature review of HALP’s prognostic ability in different cancer types. Oncotarget 2023, 14, 153. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; et al. Prognostic value of pretreatment anemia in patients with soft tissue sarcoma: A meta-analysis. Medicine 2021, 100(37), e27221. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; et al. Significance of nutritional-inflammatory index as predictors for total neoadjuvant therapy-induced tumor regression in locally advanced rectal cancer patients. Journal of Inflammation Research 2024, 3865–3878. [Google Scholar] [CrossRef] [PubMed]
- Fausti, V.; et al. Systemic inflammatory indices in second-line soft tissue sarcoma patients: focus on lymphocyte/monocyte ratio and trabectedin. Cancers 2023, 15(4), 1080. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, Y.; et al. Role of the prognostic nutritional index in patients with soft-tissue sarcoma. in vivo 2021, 35(4), 2349–2355. [Google Scholar] [CrossRef] [PubMed]
- Díaz Casas, S.E.; et al. Prognostic factors associated with tumor recurrence and overall survival in soft tissue sarcomas of the extremities in a Colombian reference cancer center. Current Oncology 2024, 31(4), 1725–1738. [Google Scholar] [CrossRef] [PubMed]
- Demir, N.; et al. HALP score as a prognostic marker for overall survival in advanced pancreatic cancer. Frontiers in Oncology 2025, 15, 1542463. [Google Scholar] [CrossRef] [PubMed]
- Tomčová, Z.; et al. Prognostic value of hemoglobin, albumin, lymphocyte, platelet (HALP) score in patients with metastatic renal cell carcinoma treated with nivolumab. Biomedicines 2025, 13(2), 484. [Google Scholar] [CrossRef] [PubMed]
| Variable | Overall (n=46) |
|---|---|
| Age, median (IQR) | 58 (45–67) |
| Male sex, n (%) | 27 (58.7) |
| Tumor size (mm), median (IQR) | 115 (85–180) |
| Grade 1, n (%) | 8 (17.4) |
| Grade 2, n (%) | 17 (37.0) |
| Grade 3, n (%) | 21 (45.6) |
| R0 resection, n (%) | 24 (52.2) |
| Non-R0 resection, n (%) | 22 (47.8) |
| Major complication (≥3), n (%) | 8 (17.4) |
| Recurrence, n (%) | 23 (50.0) |
| Death, n (%) | 23 (50.0) |
| Variable | Low HALP (n=23) | High HALP (n=23) | p |
|---|---|---|---|
| Tumor size (mm), median (IQR) | 140 (112–210) | 90 (75–130) | 0.011 |
| Non-R0 resection, n (%) | 16 (69.6) | 6 (26.1) | 0.012 |
| Major complication ≥3, n (%) | 8 (34.8) | 0 (0) | 0.004 |
| Recurrence, n (%) | 15 (65.2) | 8 (34.8) | 0.048 |
| Death, n (%) | 16 (69.6) | 7 (30.4) | 0.017 |
| Variable | OS Univariable HR (95% CI) | p | OS Multivariable HR (95% CI) | p | DFS Univariable HR (95% CI) | p | DFS Multivariable HR (95% CI) | p |
|---|---|---|---|---|---|---|---|---|
| High vs. Low HALP | 0.29 (0.12–0.70) | 0.006 | 0.82 (0.23–2.87) | 0.785 | 0.39 (0.16–0.96) | 0.040 | 0.74 (0.28–1.95) | 0.559 |
| Grade | 2.73 (1.50–4.98) | 0.001 | 2.23 (1.18–4.21) | 0.014 | 2.28 (1.27–4.10) | 0.006 | 1.94 (1.05–3.59) | 0.033 |
| Non-R0 resection | 4.04 (1.58–10.32) | 0.003 | 2.76 (0.89–8.56) | 0.083 | 7.58 (2.54–22.64) | <0.001 | 7.22 (2.19–23.77) | 0.002 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




