Submitted:
09 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
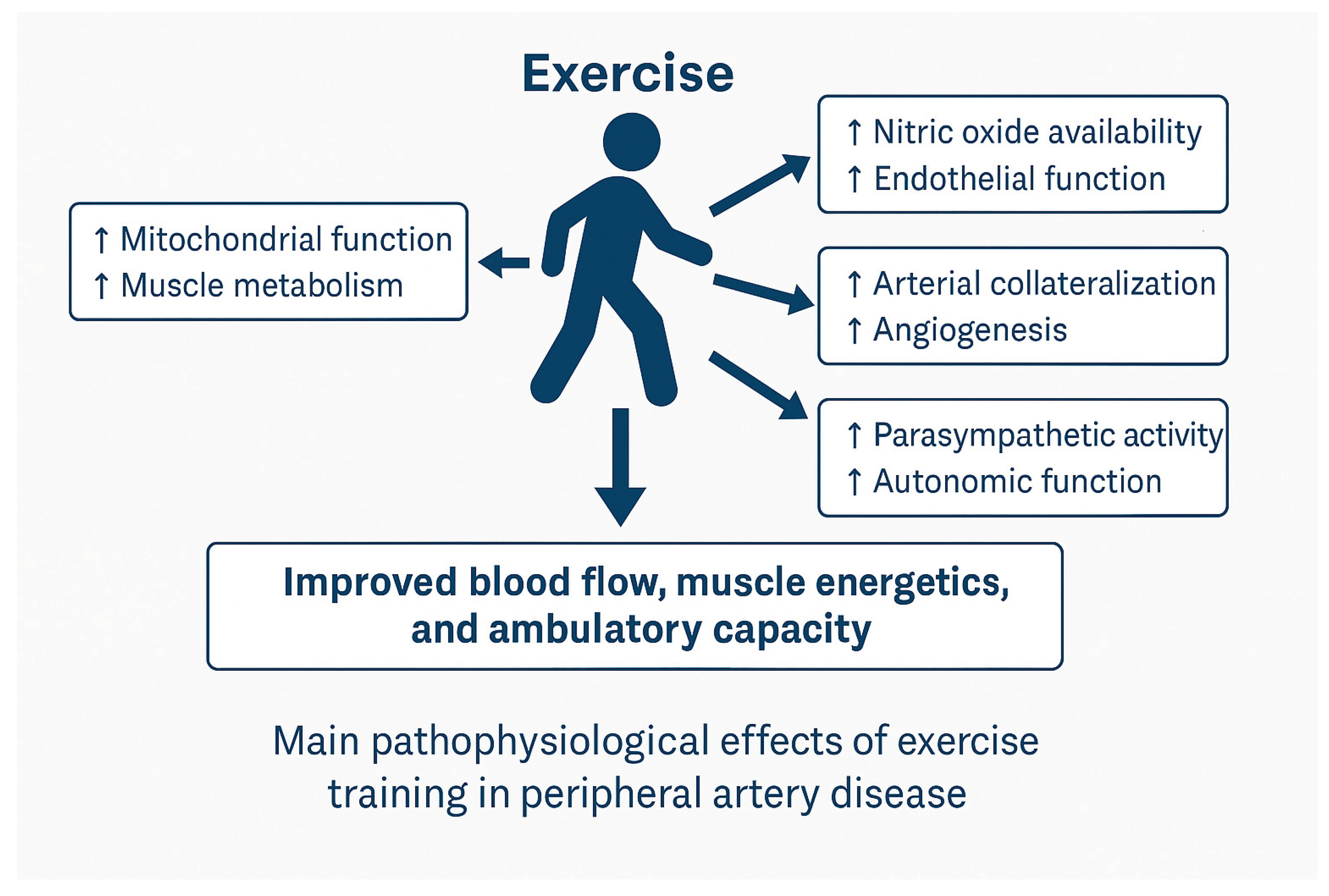
2. Beneficial Effects of Exercise Training in PAD: Mechanistic Overview
2.1. Endothelial Function and Nitric Oxide (NO)
2.2. Angiogenesis and Collateralization
2.3. Skeletal Muscle Metabolism and Mitochondria
2.4. Autonomic Balance and Baroreflex
2.5. Inflammation and Oxidative Stress
3. Supervised Exercise Therapy (SET): The Gold Standard
3.1. Protocol and Implementation
3.2. Effectiveness
3.3. Endovascular Therapy Plus SET vs SET Alone
3.4. HIIT and Alternative Intensities Within SET
4. Home-Based and Hybrid Exercise Models
4.1. Why We Need Them
4.2. Safety of Home-Based Programs
4.3. What Works at Home
4.4. Digital and Hybrid Designs
4.5. Real-World Outcomes and Program Completion
5. Alternative and Adjunct Exercise Modalities
5.1. Resistance Training
5.2. Arm Ergometry (Arm-Crank)
5.3. Cycling
5.4. Hydrotherapy
6. Clinical Outcomes
6.1. Functional Performance
6.2. Endothelial Function
6.3. Quality of Life (QoL)
6.4. ABI and Hemodynamics
6.5. Mortality and Hospitalizations
7. Prehabilitation and Post-Revascularization Rehabilitation
8. Safety, Risk Stratification, and Contraindications
9. Implementation and Policy
10. Practical Exercise Prescription in PAD (SET or Structured Home Programs)
11. Special Populations
12. Clinicl Pathway for PAD management in CR
12. Unresolved Questions and Research Priorities
13. Conclusions
References
- Gornik, H.L.; Aronow, H.D.; Goodney, P.P.; Arya, S.; Brewster, L.P.; Byrd, L.; Chandra, V.; Drachman, D.E.; Eaves, J.M.; Ehrman, J.K.; et al. ACC/AHA/AACVPR/APMA/ABC/SCAI/SVM/SVN/SVS/SIR/VESS Guideline for the Management of Lower Extremity Peripheral Artery Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2024, 149, e1313–e1410. [Google Scholar] [CrossRef]
- GBD 2019 Peripheral Artery Disease Collaborators Global Burden of Peripheral Artery Disease and Its Risk Factors, 1990-2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet Glob Health 2023, 11, e1553–e1565. [CrossRef]
- Liu, W.; Yang, C.; Chen, Z.; Lei, F.; Qin, J.-J.; Liu, H.; Ji, Y.-X.; Zhang, P.; Cai, J.; Liu, Y.-M.; et al. Global Death Burden and Attributable Risk Factors of Peripheral Artery Disease by Age, Sex, SDI Regions, and Countries from 1990 to 2030: Results from the Global Burden of Disease Study 2019. Atherosclerosis 2022, 347, 17–27. [Google Scholar] [CrossRef]
- Sprague, B.; Chesler, N.C.; Magness, R.R. Shear Stress Regulation of Nitric Oxide Production in Uterine and Placental Artery Endothelial Cells: Experimental Studies and Hemodynamic Models of Shear Stresses on Endothelial Cells. Int J Dev Biol 2010, 54, 331–339. [Google Scholar] [CrossRef]
- Sallam, N.; Laher, I. Exercise Modulates Oxidative Stress and Inflammation in Aging and Cardiovascular Diseases. Oxid Med Cell Longev 2016, 2016, 7239639. [Google Scholar] [CrossRef]
- Ungvari, Z.; Kaley, G.; de Cabo, R.; Sonntag, W.E.; Csiszar, A. Mechanisms of Vascular Aging: New Perspectives. J Gerontol A Biol Sci Med Sci 2010, 65, 1028–1041. [Google Scholar] [CrossRef]
- Thijssen, D.H.J.; Bruno, R.M.; Van Mil, A.C.C.M.; Holder, S.M.; Faita, F.; Greyling, A.; Zock, P.L.; Taddei, S.; Deanfield, J.E.; Luscher, T.; et al. Expert Consensus and Evidence-Based Recommendations for the Assessment of Flow-Mediated Dilation in Humans. European Heart Journal 2019, 40, 2534–2547. [Google Scholar] [CrossRef] [PubMed]
- Ras, R.T.; Streppel, M.T.; Draijer, R.; Zock, P.L. Flow-Mediated Dilation and Cardiovascular Risk Prediction: A Systematic Review with Meta-Analysis. Int J Cardiol 2013, 168, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Zarezadehmehrizi, A.; LaVoy, E.C.; Markofski, M.M.; Park, Y. Exercise Training Improves Brachial Artery Endothelial Function, but Does Not Alter Inflammatory Biomarkers in Patients with Peripheral Artery Disease: A Systematic Review and Meta-Analysis. J Cardiovasc Transl Res 2024, 17, 585–597. [Google Scholar] [CrossRef]
- Tao, X.; Chen, Y.; Zhen, K.; Ren, S.; Lv, Y.; Yu, L. Effect of Continuous Aerobic Exercise on Endothelial Function: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Front Physiol 2023, 14, 1043108. [Google Scholar] [CrossRef] [PubMed]
- Tryfonos, A.; Tzanis, G.; Pitsolis, T.; Karatzanos, E.; Koutsilieris, M.; Nanas, S.; Philippou, A. Exercise Training Enhances Angiogenesis-Related Gene Responses in Skeletal Muscle of Patients with Chronic Heart Failure. Cells 2021, 10, 1915. [Google Scholar] [CrossRef]
- Fan, Z.; Turiel, G.; Ardicoglu, R.; Ghobrial, M.; Masschelein, E.; Kocijan, T.; Zhang, J.; Tan, G.; Fitzgerald, G.; Gorski, T.; et al. Exercise-Induced Angiogenesis Is Dependent on Metabolically Primed ATF3/4+ Endothelial Cells. Cell Metab 2021, 33, 1793–1807.e9. [Google Scholar] [CrossRef]
- Mazzolai, L.; Belch, J.; Venermo, M.; Aboyans, V.; Brodmann, M.; Bura-Rivière, A.; Debus, S.; Espinola-Klein, C.; Harwood, A.E.; Hawley, J.A.; et al. Exercise Therapy for Chronic Symptomatic Peripheral Artery Disease: A Clinical Consensus Document of the European Society of Cardiology Working Group on Aorta and Peripheral Vascular Diseases in Collaboration With the European Society of Vascular Medicine and the European Society for Vascular Surgery. Eur J Vasc Endovasc Surg 2024, S1078-5884(24)00011-X. [Google Scholar] [CrossRef]
- Peñín-Grandes, S.; López-Ortiz, S.; Maroto-Izquierdo, S.; Menéndez, H.; Pinto-Fraga, J.; Martín-Hernández, J.; Lista, S.; Lucia, A.; Santos-Lozano, A. Winners Do What They Fear: Exercise and Peripheral Arterial Disease-an Umbrella Review. Eur J Prev Cardiol 2024, 31, 380–388. [Google Scholar] [CrossRef]
- Brenner, I.K.M.; Brown, C.A.; Hains, S.J.M.; Tranmer, J.; Zelt, D.T.; Brown, P.M. Low-Intensity Exercise Training Increases Heart Rate Variability in Patients With Peripheral Artery Disease. Biol Res Nurs 2020, 22, 24–33. [Google Scholar] [CrossRef]
- Sorriento, D.; Iaccarino, G. Inflammation and Cardiovascular Diseases: The Most Recent Findings. Int J Mol Sci 2019, 20, 3879. [Google Scholar] [CrossRef]
- Theofilis, P.; Sagris, M.; Oikonomou, E.; Antonopoulos, A.S.; Siasos, G.; Tsioufis, C.; Tousoulis, D. Inflammatory Mechanisms Contributing to Endothelial Dysfunction. Biomedicines 2021, 9, 781. [Google Scholar] [CrossRef]
- Alp, N.J.; Channon, K.M. Regulation of Endothelial Nitric Oxide Synthase by Tetrahydrobiopterin in Vascular Disease. Arterioscler Thromb Vasc Biol 2004, 24, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Ulevitch, R.J. Limiting Inflammatory Responses during Activation of Innate Immunity. Nat Immunol 2005, 6, 1198–1205. [Google Scholar] [CrossRef] [PubMed]
- Shankar, A.; Li, J.; Nieto, F.J.; Klein, B.E.K.; Klein, R. Association between C-Reactive Protein Level and Peripheral Arterial Disease among US Adults without Cardiovascular Disease, Diabetes, or Hypertension. Am Heart J 2007, 154, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Januszek, R.; Mika, P.; Konik, A.; Petriczek, T.; Nowobilski, R.; Niżankowski, R. Effect of Treadmill Training on Endothelial Function and Walking Abilities in Patients with Peripheral Arterial Disease. J Cardiol 2014, 64, 145–151. [Google Scholar] [CrossRef]
- Lin, M.-L.; Fu, T.-C.; Hsu, C.-C.; Huang, S.-C.; Lin, Y.-T.; Wang, J.-S. Cycling Exercise Training Enhances Platelet Mitochondrial Bioenergetics in Patients with Peripheral Arterial Disease: A Randomized Controlled Trial. Thromb Haemost 2021, 121, 900–912. [Google Scholar] [CrossRef]
- Murphy, T.P.; Cutlip, D.E.; Regensteiner, J.G.; Mohler, E.R.; Cohen, D.J.; Reynolds, M.R.; Massaro, J.M.; Lewis, B.A.; Cerezo, J.; Oldenburg, N.C.; et al. Supervised Exercise versus Primary Stenting for Claudication Resulting from Aortoiliac Peripheral Artery Disease: Six-Month Outcomes from the Claudication: Exercise versus Endoluminal Revascularization (CLEVER) Study. Circulation 2012, 125, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Lanzi, S. From Bed Rest to Never-Ending Walk: Highlighting the Importance of Exercise Training in Patients with Peripheral Artery Disease. Eur J Prev Cardiol 2024, 31, 378–379. [Google Scholar] [CrossRef] [PubMed]
- Fakhry, F.; Spronk, S.; van der Laan, L.; Wever, J.J.; Teijink, J.A.W.; Hoffmann, W.H.; Smits, T.M.; van Brussel, J.P.; Stultiens, G.N.M.; Derom, A.; et al. Endovascular Revascularization and Supervised Exercise for Peripheral Artery Disease and Intermittent Claudication: A Randomized Clinical Trial. JAMA 2015, 314, 1936–1944. [Google Scholar] [CrossRef] [PubMed]
- Fakhry, F.; Rouwet, E.V.; Spillenaar Bilgen, R.; van der Laan, L.; Wever, J.J.; Teijink, J.A.W.; Hoffmann, W.H.; van Petersen, A.; van Brussel, J.P.; Stultiens, G.N.M.; et al. Endovascular Revascularization Plus Supervised Exercise Versus Supervised Exercise Only for Intermittent Claudication: A Cost-Effectiveness Analysis. Circ Cardiovasc Interv 2021, 14, e010703. [Google Scholar] [CrossRef]
- McDermott, M.M.; Spring, B.; Tian, L.; Treat-Jacobson, D.; Ferrucci, L.; Lloyd-Jones, D.; Zhao, L.; Polonsky, T.; Kibbe, M.R.; Bazzano, L.; et al. Effect of Low-Intensity vs High-Intensity Home-Based Walking Exercise on Walk Distance in Patients With Peripheral Artery Disease: The LITE Randomized Clinical Trial. JAMA 2021, 325, 1266–1276. [Google Scholar] [CrossRef]
- Divakaran, S.; Carroll, B.J.; Chen, S.; Shen, C.; Bonaca, M.P.; Secemsky, E.A. Supervised Exercise Therapy for Symptomatic Peripheral Artery Disease Among Medicare Beneficiaries Between 2017 and 2018: Participation Rates and Outcomes. Circ Cardiovasc Qual Outcomes 2021, 14, e007953. [Google Scholar] [CrossRef]
- Cetlin, M.D.; Polonsky, T.; Ho, K.; Zhang, D.; Tian, L.; Zhao, L.; Greenland, P.; Treat-Jacobson, D.; Kibbe, M.R.; Criqui, M.H.; et al. Barriers to Participation in Supervised Exercise Therapy Reported by People with Peripheral Artery Disease. J Vasc Surg 2023, 77, 506–514. [Google Scholar] [CrossRef]
- Waddell, A.; Seed, S.; Broom, D.R.; McGregor, G.; Birkett, S.T.; Harwood, A.E. Safety of Home-Based Exercise for People with Intermittent Claudication: A Systematic Review. Vasc Med 2022, 27, 186–192. [Google Scholar] [CrossRef]
- Thangada, N.D.; Zhang, D.; Zhao, L.; Tian, L.; McDermott, M.M. Safety and Efficacy of Home-Based Walking Exercise for Peripheral Artery Disease. J Vasc Surg 2025, 81, 441–449.e1. [Google Scholar] [CrossRef]
- McDermott, M.M.; Liu, K.; Guralnik, J.M.; Criqui, M.H.; Spring, B.; Tian, L.; Domanchuk, K.; Ferrucci, L.; Lloyd-Jones, D.; Kibbe, M.; et al. Home-Based Walking Exercise Intervention in Peripheral Artery Disease: A Randomized Clinical Trial. JAMA 2013, 310, 57–65. [Google Scholar] [CrossRef]
- McDermott, M.M.; Spring, B.; Berger, J.S.; Treat-Jacobson, D.; Conte, M.S.; Creager, M.A.; Criqui, M.H.; Ferrucci, L.; Gornik, H.L.; Guralnik, J.M.; et al. Effect of a Home-Based Exercise Intervention of Wearable Technology and Telephone Coaching on Walking Performance in Peripheral Artery Disease: The HONOR Randomized Clinical Trial. JAMA 2018, 319, 1665–1676. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Chuo, J.; Zhao, X. Effectiveness of Home-Based Walking Exercise for Patients with Peripheral Artery Disease and Intermittent Claudication: A Systematic Review and Meta-Analysis. BMJ Open 2025, 15, e086013. [Google Scholar] [CrossRef]
- Silva, I.; Moreira, C.S.; Pedras, S.; Oliveira, R.; Veiga, C.; Moreira, L.; Santarém, D.; Guedes, D.; Paredes, H. Effect of a Monitored Home-Based Exercise Program Combined with a Behavior Change Intervention and a Smartphone App on Walking Distances and Quality of Life in Adults with Peripheral Arterial Disease: The WalkingPad Randomized Clinical Trial. Front Cardiovasc Med 2023, 10, 1272897. [Google Scholar] [CrossRef]
- Fanaroff, A.C.; Coratti, S.; Farraday, D.; Norton, L.; Rareshide, C.; Zhu, J.; Levin, M.G.; Damrauer, S.M.; Giri, J.S.; Chokshi, N.P.; et al. Effect of Gamification Plus Automated Coaching to Increase Physical Activity Among Patients With Peripheral Artery Disease: The GAMEPAD Randomized Controlled Trial. J Am Heart Assoc 2025, 14, e038921. [Google Scholar] [CrossRef]
- Whipple, M.O.; Burt, M.A.; Pergolski, A.L.; McArthur, P.; Treat-Jacobson, D.; Salisbury, D.L. Uptake and Outcomes of Supervised Exercise Therapy for Peripheral Artery Disease: The Importance of Vascular Medicine Specialists at a Large Midwestern Health Care System during the First 5 Years of CMS Reimbursement. Vasc Med 2024, 29, 112–119. [Google Scholar] [CrossRef]
- Hollings, M.; Mavros, Y.; Freeston, J.; Fiatarone Singh, M. The Effect of Progressive Resistance Training on Aerobic Fitness and Strength in Adults with Coronary Heart Disease: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Eur J Prev Cardiol 2017, 24, 1242–1259. [Google Scholar] [CrossRef] [PubMed]
- Marterer, N.; Mugele, H.; Schäfer, S.K.; Faulhaber, M. Effects of Upper Body Exercise Training on Aerobic Fitness and Performance in Healthy People: A Systematic Review. Biology (Basel) 2023, 12, 355. [Google Scholar] [CrossRef] [PubMed]
- Kapusta, J.; Kapusta, A.; Babicki, M.; Irzmański, R. Evaluation of Peripheral Circulatory Changes Following Hydrotherapy and Controlled Physical Training in Patients with Atherosclerotic Lower Limb Ischemia. Life (Basel) 2024, 14, 1578. [Google Scholar] [CrossRef]
- Cucato, G.; Longano, P.P.; Perren, D.; Ritti-Dias, R.M.; Saxton, J.M. Effects of Additional Exercise Therapy after a Successful Vascular Intervention for People with Symptomatic Peripheral Arterial Disease. Cochrane Database Syst Rev 2024, 5, CD014736. [Google Scholar] [CrossRef]
- Saratzis, A.; Paraskevopoulos, I.; Patel, S.; Donati, T.; Biasi, L.; Diamantopoulos, A.; Zayed, H.; Katsanos, K. Supervised Exercise Therapy and Revascularization for Intermittent Claudication: Network Meta-Analysis of Randomized Controlled Trials. JACC Cardiovasc Interv 2019, 12, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Banerjee, S.; Ngo, C.; Mody, P.; Marso, S.P.; Brilakis, E.S.; Armstrong, E.J.; Giri, J.; Bonaca, M.P.; Pradhan, A.; et al. Comparative Efficacy of Endovascular Revascularization Versus Supervised Exercise Training in Patients With Intermittent Claudication: Meta-Analysis of Randomized Controlled Trials. JACC Cardiovasc Interv 2017, 10, 712–724. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kang, M.; Park, Y. Exercise Training Enhances Brachial Artery Endothelial Function, Possibly via Improved HDL-C, Not LDL-C and TG, in Patients with Coronary Artery Disease: A Systematic Review and Meta-Analysis. Am J Cardiovasc Drugs 2025, 25, 399–410. [Google Scholar] [CrossRef]
- Luk, T.-H.; Dai, Y.-L.; Siu, C.-W.; Yiu, K.-H.; Chan, H.-T.; Lee, S.W.L.; Li, S.-W.; Fong, B.; Wong, W.-K.; Tam, S.; et al. Effect of Exercise Training on Vascular Endothelial Function in Patients with Stable Coronary Artery Disease: A Randomized Controlled Trial. Eur J Prev Cardiol 2012, 19, 830–839. [Google Scholar] [CrossRef]
- Anghel, R.; Adam, C.A.; Mitu, O.; Marcu, D.T.M.; Onofrei, V.; Roca, M.; Costache, A.D.; Miftode, R.S.; Tinica, G.; Mitu, F. Cardiac Rehabilitation and Mortality Risk Reduction in Peripheral Artery Disease at 6-Month Outcome. Diagnostics (Basel) 2022, 12, 1500. [Google Scholar] [CrossRef] [PubMed]
- Yasu, T. Comprehensive Cardiac Rehabilitation Program for Peripheral Arterial Diseases. J Cardiol 2022, 80, 303–305. [Google Scholar] [CrossRef]
- Nordanstig, J.; Taft, C.; Hensäter, M.; Perlander, A.; Österberg, K.; Jivegård, L. Two-Year Results from a Randomized Clinical Trial of Revascularization in Patients with Intermittent Claudication. Br J Surg 2016, 103, 1290–1299. [Google Scholar] [CrossRef]
- Klaphake, S.; Buettner, S.; Ultee, K.H.; van Rijn, M.J.; Hoeks, S.E.; Verhagen, H.J. Combination of Endovascular Revascularization and Supervised Exercise Therapy for Intermittent Claudication: A Systematic Review and Meta-Analysis. J Cardiovasc Surg (Torino) 2018, 59, 150–157. [Google Scholar] [CrossRef]
- Bø, E.; Bergland, A.; Stranden, E.; Jørgensen, J.J.; Sandbaek, G.; Grøtta, O.J.; Hisdal, J. Effects of 12 Weeks of Supervised Exercise After Endovascular Treatment: A Randomized Clinical Trial. Physiother Res Int 2015, 20, 147–157. [Google Scholar] [CrossRef]
- Waddell, A.; Denton, F.; Powell, R.; Broom, D.R.; Birkett, S.T.; McGregor, G.; Harwood, A.E. Home-Based Circuit Training and Community Walking for Intermittent Claudication. Ann Vasc Surg 2024, 105, 38–47. [Google Scholar] [CrossRef]
- Nordanstig, J.; Behrendt, C.-A.; Baumgartner, I.; Belch, J.; Bäck, M.; Fitridge, R.; Hinchliffe, R.; Lejay, A.; Mills, J.L.; Rother, U.; et al. Editor’s Choice -- European Society for Vascular Surgery (ESVS) 2024 Clinical Practice Guidelines on the Management of Asymptomatic Lower Limb Peripheral Arterial Disease and Intermittent Claudication. Eur J Vasc Endovasc Surg 2024, 67, 9–96. [Google Scholar] [CrossRef]
- Giallauria, F.; Di Lorenzo, A.; Venturini, E.; Pacileo, M.; D’Andrea, A.; Garofalo, U.; De Lucia, F.; Testa, C.; Cuomo, G.; Iannuzzo, G.; et al. Frailty in Acute and Chronic Coronary Syndrome Patients Entering Cardiac Rehabilitation. J Clin Med 2021, 10, 1696. [Google Scholar] [CrossRef]
- Qiu, B.; Zhou, Y.; Tao, X.; Hou, X.; Du, L.; Lv, Y.; Yu, L. The Effect of Exercise on Flow-Mediated Dilation in People with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Front Endocrinol (Lausanne) 2024, 15, 1347399. [Google Scholar] [CrossRef] [PubMed]
- Lucà, F.; Abrignani, M.G.; Parrini, I.; Di Fusco, S.A.; Giubilato, S.; Rao, C.M.; Piccioni, L.; Cipolletta, L.; Passaretti, B.; Giallauria, F.; et al. Update on Management of Cardiovascular Diseases in Women. J Clin Med 2022, 11, 1176. [Google Scholar] [CrossRef] [PubMed]
- Marinus, N.; Vigorito, C.; Giallauria, F.; Haenen, L.; Jansegers, T.; Dendale, P.; Feys, P.; Meesen, R.; Timmermans, A.; Spildooren, J.; et al. Frailty Is Highly Prevalent in Specific Cardiovascular Diseases and Females, but Significantly Worsens Prognosis in All Affected Patients: A Systematic Review. Ageing Res Rev 2021, 66, 101233. [Google Scholar] [CrossRef] [PubMed]
- Perks, J.; Zaccardi, F.; Paterson, C.; Houghton, J.S.M.; Nickinson, A.T.O.; Pepper, C.J.; Rayt, H.; Yates, T.; Sayers, R. Effect of High-Pain versus Low-Pain Structured Exercise on Walking Ability in People with Intermittent Claudication: Meta-Analysis. British Journal of Surgery 2022, 109, 686–694. [Google Scholar] [CrossRef]
| Study (Year) | Population/Setting | Arms/Intervention | Primary Outcome | Key Results | Follow-up | Notes |
| CLEVER (2012; 18-mo analysis 2013) | Aortoiliac PAD with claudication | OMC vs Stent vs SET | Peak walking time (graded treadmill) | SET > Stent at 6 mo for treadmill performance; both SET and Stent > OMC at 18 mo; QoL favored Stent early | 6 and 18 mo | Functional vs QoL trade-offs; importance of maintenance |
| ERASE (JAMA 2015; CEA 2021) | Intermittent claudication (aortoiliac/femoropopliteal) | SET vs Endovascular + SET | Max walking distance | +282 m (99% CI 60–505) for ER+SET vs SET; cost-effective from societal perspective | 12 mo | Supports synergy when anatomy warrants intervention |
| LITE (JAMA 2021; AJC 2025 PROMs) | PAD with diverse symptoms | High-intensity (symptom-eliciting) vs Low-intensity (pain-free) home-based walking vs Control | 6MWD | High-intensity +34–45 m at 12 mo; low-intensity ≈ no benefit vs control; PROMs improved mainly with high-intensity | 12 mo | Coaching weekly; accelerometer-monitored |
| JAMA Netw Open IPD MA (2023) | PAD RCTs (n≈719) | Home-based walking vs Supervised treadmill (vs controls) | 6MWD and treadmill outcomes | Home-based > Supervised for 6MWD (+≈24 m); Supervised > Home for treadmill distance | 6 mo | Task specificity (community vs treadmill) |
| WalkingPad RCT (Front Cardiovasc Med 2023) | IC; single-center | HBET + behavior change ± smartphone app | PFWD/MWD/6MWD; QoL | Both arms improved at 3 mo; MWD advantage with app at 6 mo in sensitivity analyses | 3–6 mo | Digital augmentation potentially helpful |
| HY-PAD feasibility (CJC Open 2025) | PAD; pre–post | 4 wk supervised then 8 wk home with calls | 6MWD; WIQ | +56 m 6MWD; high adherence; few adverse events | 12 wk | Feasibility; needs controlled trials |
| Safety of HBEP (Waddell 2021) | IC; 27 studies; 147,810 patient-hours | Home-based programs (varied) | Complication rate | ≈1 related event/36,953 patient-hours; most without cardiac screening | Varied | Supports safe HBEP with prudent screening |
| BMJ Open 2025 MA (HBET efficacy) | IC; 7 RCTs | Home-based exercise vs control | PFWD/MWD | Significant PFWD benefit; MWD NS overall; heterogeneity high | 6–52 wk | Intensity/adherence likely moderators |
| Endothelial Function MA (PAD/CAD) 2024 | PAD/CAD cohorts | Structured aerobic training | Flow-mediated dilation | Increased brachial FMD following training | Varied | Mechanistic support for vascular benefit |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




