Submitted:
07 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
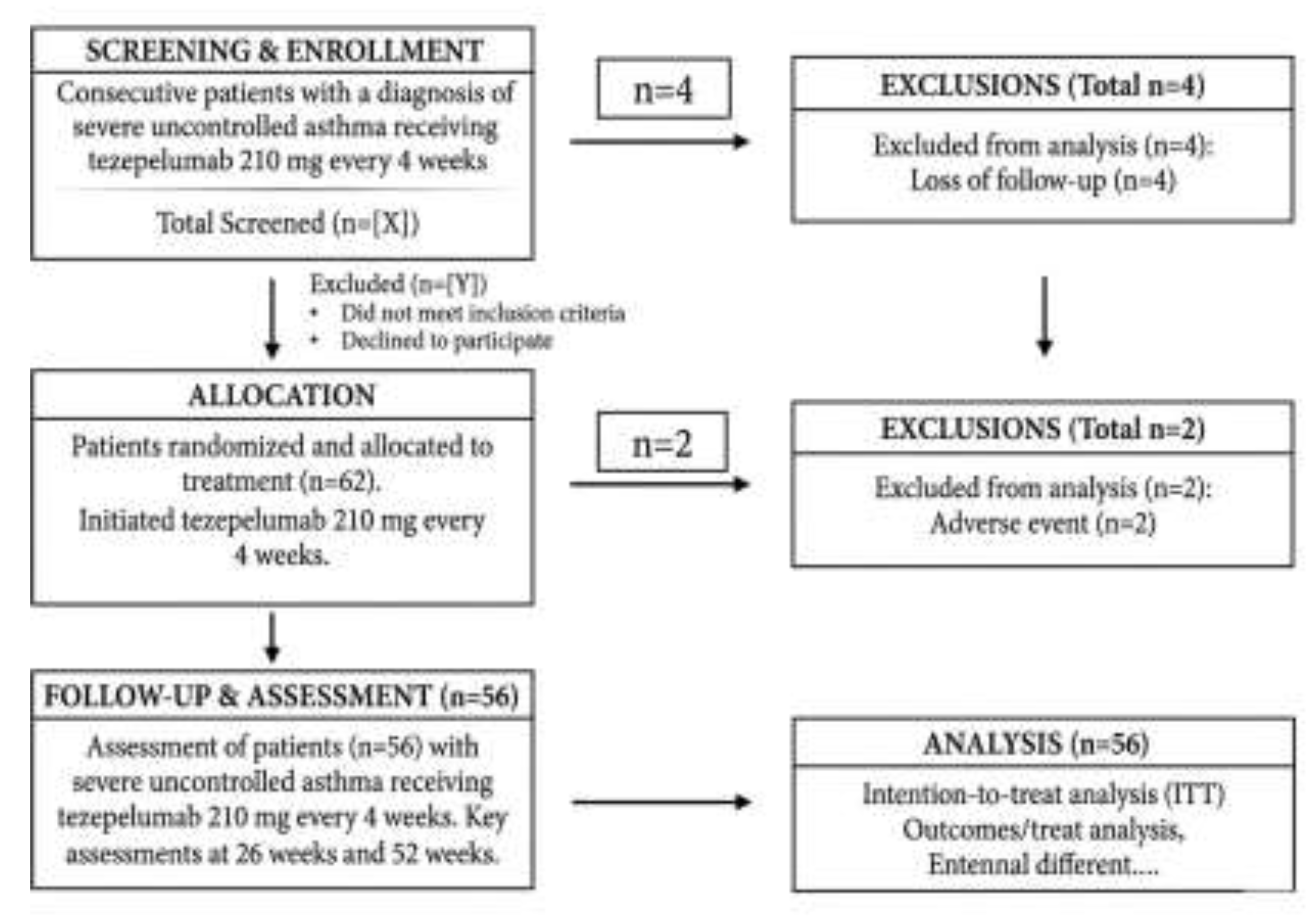
2.1. Study Design and Setting
2.2. Participants
2.3. Data Collection and Clinical Outcomes
2.4. Classification of Inflammatory Endotypes
2.5. Laboratory and Serological Assessments
2.6. Skin Prick Test
2.7. Definition of Treatment Response
2.8. Statistical Analysis
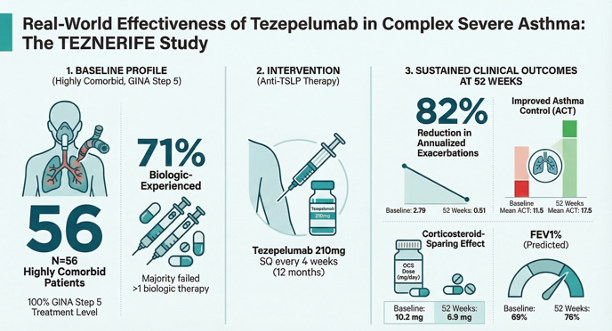
3. Results
3.1. Demographic and Clinical Characteristics of Subjects at Baseline
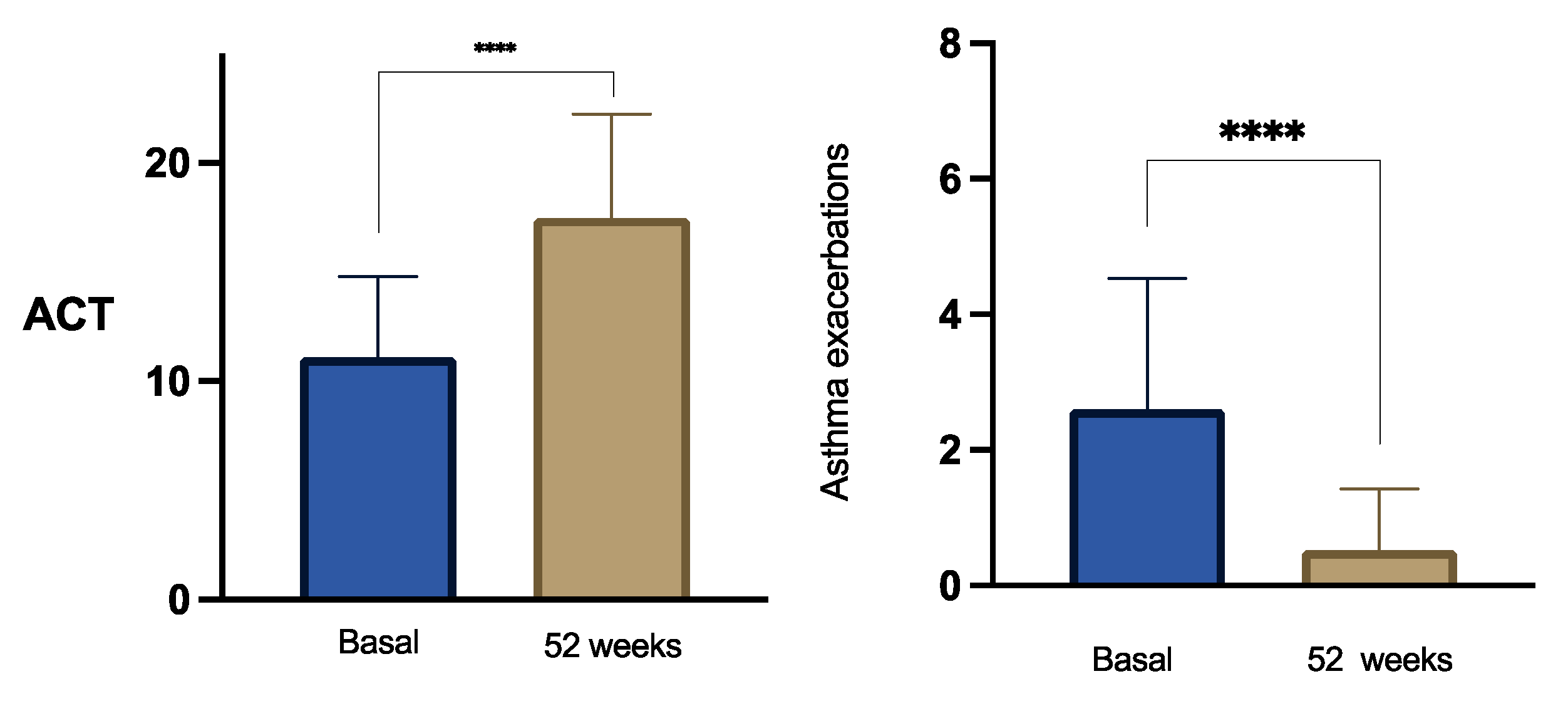
3.2. Asthma Control and Exacerbations
3.3. Oral Corticosteroid Exposure
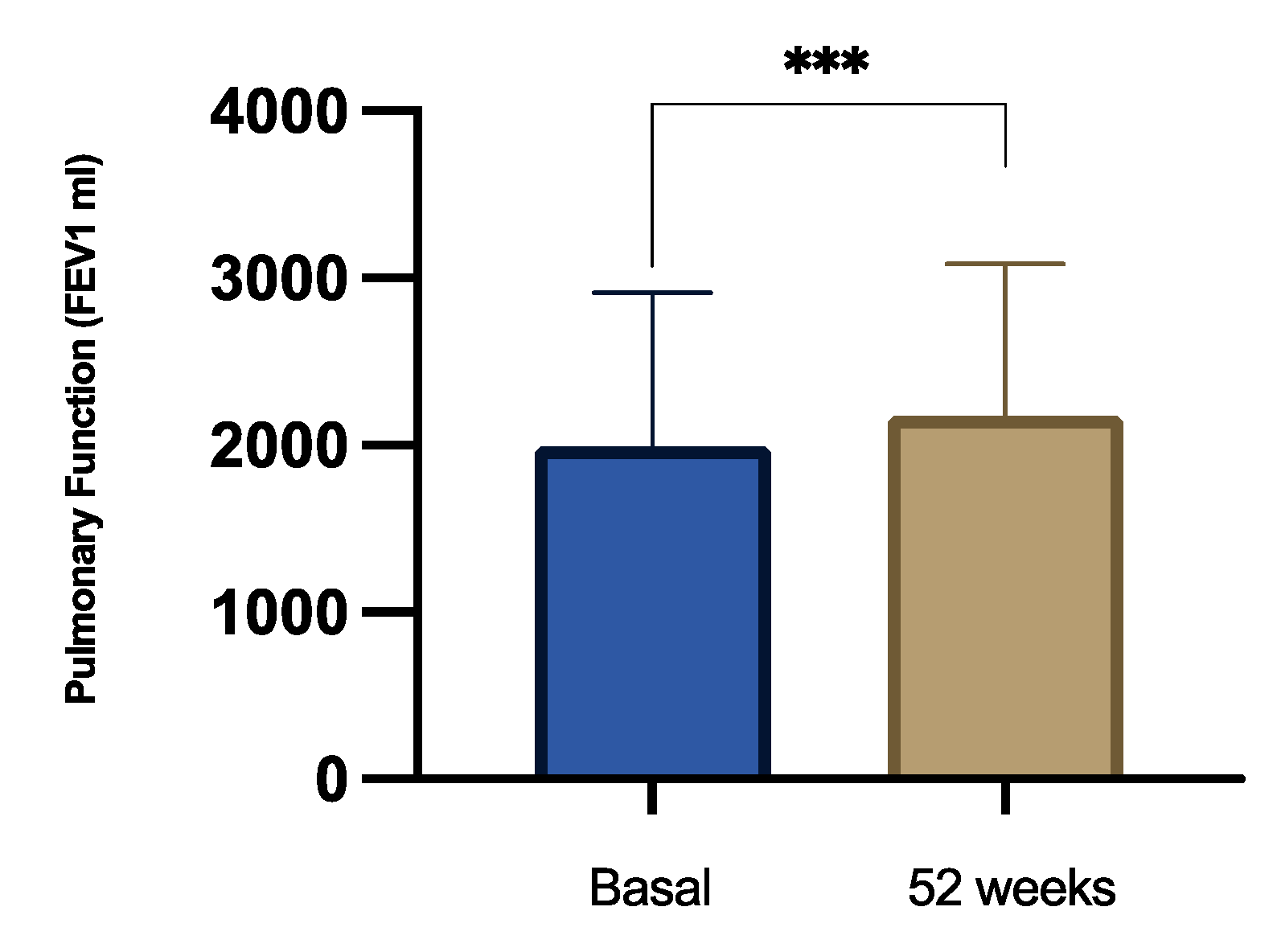
3.4. Lung Function
3.5. Type 2 Inflammatory Biomarkers
3.6. Upper Airway Outcomes and Composite Response
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SA | Severe Asthma |
| TSLP | Thymic stromal lymphopoietin |
| CRSwNP | Chronic Rinosinusitis with Nasal Polyposis |
| BARS | Biologics Asthma Response Score |
| OCS | Oral Corticosteroid |
| FeNO | Fractional exhaled Nitric Oxide |
References
- https://www.asthmaandlung.org.uk/conditions/severe-asthma/what-severe-asthma-how-it-diagnosed Last visited February 20, 2026.
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020 Oct 17;396(10258):1204-1222. Erratum in: Lancet. 2020 Nov 14;396(10262):1562. doi: 10.1016/S0140-6736(20)32226-1. [CrossRef]
- https://ginasthma.org/ Last visited February 4, 2026.
- Haldar, P; Pavord, ID; Shaw, DE; Berry, MA; Thomas, M; Brightling, CE; et al. Cluster analysis and clinical asthma phenotypes. Am J Respir Crit Care Med. 2008, 178(3), 218-24. [Google Scholar] [CrossRef]
- Moore, WC; Meyers, DA; Wenzel, SE; Teague, WG; Li, H; Li, X; et al. Identification of asthma phenotypes using cluster analysis in the Severe Asthma Research Program. Am J Respir Crit Care Med. 2010, 181(4), 315-23. [Google Scholar] [CrossRef]
- Holguin, F; Cardet, JC; Chung, KF; Diver, S; Ferreira, DS; Fitzpatrick, A; et al. Management of severe asthma: a European Respiratory Society/American Thoracic Society guideline. Eur Respir J 2020, 55(1), 1900588. [Google Scholar] [CrossRef] [PubMed]
- Gyawali, B; Georas, SN; Khurana, S. Biologics in severe asthma: a state-of-the-art review. Eur Respir Rev. 2025, 34(175), 240088. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Couillard, S; Jackson, DJ; Pavord, ID; Wechsler, ME. Choosing the Right Biologic for the Right Patient With Severe Asthma. Chest 2025, 167(2), 330–342. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Al-Shaikhly, T; Norris, MR; Dennis, EH; Liu, G; Craig, TJ. Comparative Impact of Asthma Biologics: A Nationwide US Claim-Based Analysis. J Allergy Clin Immunol Pract. 2024, 12(6), 1558–1567. [Google Scholar] [CrossRef] [PubMed]
- Lipworth, BJ; Greig, R; Chan, R; Kuo, CR; Jackson, C. Head-To-Head Comparison of Biologic Efficacy in Asthma: What Have We Learned? Allergy 2025, 80(5), 1226–1241. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, Y; Wang, W; Lv, Z; Li, Y; Chen, Y; Huang, K; et al. Elevated Expression of IL-33 and TSLP in the Airways of Human Asthmatics In Vivo: A Potential Biomarker of Severe Refractory Disease. J Immunol. 2018, 200(7), 2253–2262. [Google Scholar] [CrossRef] [PubMed]
- Duchesne, M; Okoye, I; Lacy, P. Epithelial cell alarmin cytokines: Frontline mediators of the asthma inflammatory response. Front Immunol. 2022, 13, 975914. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Diver S, Khalfaoui L, Emson C, Wenzel SE, Menzies-pow A, Wechsler ME, et al. Effect of tezepelumab on airway inflammatory cells, remodelling, and hyperresponsiveness in patients with moderate-to-severe uncontrolled asthma (CASCADE): a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet Respir Med. 2021 Nov;9(11):1299-1312. Epub 2021 Jul 10. Erratum in: Lancet Respir Med. 2021 Nov;9(11):e106. doi: 10.1016/S2213-2600(21)00446-X. [CrossRef] [PubMed]
- Menzies-Gow A, Wechsler ME, Brightling CE, Korn S, Corren J, Israel E, et al. Long-term safety and efficacy of tezepelumab in people with severe, uncontrolled asthma (DESTINATION): a randomised, placebocontrolled extension study. Lancet Respir Med. 2023 May;11(5):425-438. Epub 2023 Jan 23. Erratum in: Lancet Respir Med. 2023 Mar;11(3):e25. doi: 10.1016/S2213-2600(23)00048- 6. [CrossRef]
- Menzies-Gow, A; Colice, G; Griffiths, JM; Almqvist, G; Ponnarambil, S; Kaur, P; et al. NAVIGATOR: a phase 3 multicentre, randomized, double-blind, placebo-controlled, parallel-group trial to evaluate the efficacy and safety of tezepelumab in adults and adolescents with severe, uncontrolled asthma. Respir Res. 2020, 21(1), 266. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Menzies-Gow, A; Corren, J; Bourdin, A; et al. Tezepelumab in Adults and Adolescents with Severe, Uncontrolled Asthma. N Engl J Med. 2021c, 384, 1800–1809. [Google Scholar] [CrossRef]
- Corren, J; Garcia Gil, E; Griffiths, JM; et al. Tezepelumab improves patient-reported outcomes in patients with severe, uncontrolled asthma in PATHWAY. Annual Allergy Asthma Immunol. 2021, 126, 187–193. [Google Scholar] [CrossRef]
- Wechsler, ME; Colice, G; Griffiths, JM; Almqvist, G; Skärby, T; Piechowiak, T; et al. SOURCE: a phase 3, multicentre, randomized, double-blind, placebo-controlled, parallel group trial to evaluate the efficacy and safety of tezepelumab in reducing oral corticosteroid use in adults with oral corticosteroid dependent asthma. Respir Res. 2020, 21(1), 264. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Edris, A; De Feyter, S; Maes, T; Joos, G; Lahousse, L. Monoclonal antibodies in type 2 asthma: a systematic review and network meta-analysis. Respir Res. 2019, 20(1), 179. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nagase, H; Suzukawa, M; Oishi, K; Matsunaga, K. Biologics for severe asthma: The real-world evidence, effectiveness of switching, and prediction factors for the efficacy. Allergol Int. 2023, 72(1), 11–23. [Google Scholar] [CrossRef] [PubMed]
- Casas Deza, D; Larrubia Domínguez, C; Pascual Oliver, A; Polo Cuadro, C; Yagüe; et al. Patients Included in Clinical Trials of Biological Drugs for Inflammatory Bowel Disease Do Not Represent the Real-World Population. Am J Gastroenterol 2025. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C; Giacalone, A; Ippolito, G; Pastore, D; Maglio, A; Piazzetta, GL; et al. Difficult-To-Treat and Severe Asthma: Can Real-World Studies On Effectiveness of Biological Treatments Change the Lives of Patients? Pragmat Obs Res 2024, 15, 45–51. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Casas-Maldonado, F; Álvarez-Gutiérrez, FJ; Blanco-Aparicio, M; Domingo-Ribas, C; Cisneros-Serrano, C; Soto-Campos, G; et al. Monoclonal antibody treatment for severe uncontrolled asthma in Spain: analytical map. J Asthma 2022, 59(10), 1997–2007. [Google Scholar] [CrossRef] [PubMed]
- González-Pérez, R; Poza-Guedes, P; Sánchez-Machín, I. Asma en Canarias: derivación centrada en el paciente [Asthma in the Canary Islands: Patient-Centered Derivation Criteria]. Open Respir Arch. 2024, 6(1), 100308, Spanish. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Martin-Gonzalez, E; Perez-Garcia, J; Martin-Almeida, M; Hernández-Pérez, JM; González-Pérez, R; Sardón, O; et al. Genome-wide Association Study of Asthma Exacerbations in the Spanish Population. Arch Bronconeumol 2025, 61(8), 490–494, English, Spanish. [Google Scholar] [CrossRef] [PubMed]
- González-Pérez, R; Poza-Guedes, P; Mederos-Luis, E; Sánchez-Machín, I. Real-Life Performance of Mepolizumab in T2-High Severe Refractory Asthma with the Overlapping Eosinophilic-Allergic Phenotype. Biomedicines 2022, 10(10), 2635. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bourdin, A; Bjermer, L; Brightling, C; Brusselle, GG; Chanez, P; Chung, KF; et al. ERS/EAACI statement on severe exacerbations in asthma in adults: facts, priorities and key research questions. Eur Respir J 2019, 54(3), 1900900. [Google Scholar] [CrossRef] [PubMed]
- Cooper, BG; Stocks, J; Hall, GL; Culver, B; Steenbruggen, I; Carter, KW; et al. The Global Lung Function Initiative (GLI) Network: bringing the world's respiratory reference values together. Breathe (Sheff) 2017, 13(3), e56–e64. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Graham, BL; Steenbruggen, I; Miller, MR; Barjaktarevic, IZ; Cooper, BG; Hall, GL; et al. H Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement. Am J Respir Crit Care Med 2019, 200(8), e70–e88. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sim, S; Choi, Y; Park, HS. Update on Inflammatory Biomarkers for Defining Asthma Phenotype. Allergy Asthma Immunol Res 2024, 16(5), 462–472. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bourdin, A; Brusselle, G; Couillard, S; Fajt, ML; Heaney, LG; Israel, E; et al. Phenotyping of Severe Asthma in the Era of Broad-Acting Anti-Asthma Biologics. J Allergy Clin Immunol Pract. 2024, 12(4), 809–823. [Google Scholar] [CrossRef] [PubMed]
- Heinzerling, L; Mari, A; Bergmann, KC; Bresciani, M; Burbach, G; Darsow, U; et al. The skin prick test - European standards. Clin Transl Allergy 2013, 3(1), 3. [Google Scholar] [CrossRef] [PubMed]
- Milger, K; Korn, S; Feder, C; Fuge, J; Mühle, A; Schütte, W; et al. Criteria for evaluation of response to biologics in severe asthma - the Biologics Asthma Response Score (BARS). Pneumologie 2025, 79(6), 439–451. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Milger, K; Suhling, H; Skowasch, D; Holtdirk, A; Kneidinger, N; Behr, J; et al. Response to Biologics and Clinical Remission in the Adult German Asthma Net Severe Asthma Registry Cohort. J Allergy Clin Immunol Pract. 2023, 11(9), 2701–2712.e2. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C; Greco, M; Iaccino, E; Crimi, C; Biafora, M; Dragone, F; et al. Short-term therapeutic effectiveness of tezepelumab in patients with severe asthma: A real-world study. Int Immunopharmacol. 2025, 162, 115185. [Google Scholar] [CrossRef] [PubMed]
- Miralles-López, JC; Bravo-Gutierrez, FJ; Andújar-Espinosa, R; Castilla-Martínez, M; Díaz-Chantar, C; Ramírez-Hernández, M; et al. RE-ASGRAMUR Group. Real-life effectiveness of tezepelumab in severe asthma. Allergol Immunopathol (Madr) 2025, 53(2), 163–173. [Google Scholar] [CrossRef] [PubMed]
- Caminati, M; Buhl, R; Corren, J; Hanania, NA; Kim, H; Korn, S; et al. Tezepelumab in patients with allergic and eosinophilic asthma. Allergy 2024, 79(5), 1134–1145. [Google Scholar] [CrossRef] [PubMed]
- Aslam, MMS; Senn, R; Bagheri, A; Harwood, H; Troost, JP; Lugogo, NL; Mohan, A. Novel Real-World Insights Into Tezepelumab Effectiveness in Steroid-Dependent Asthma. J Asthma Allergy 2025, 18, 1835–1839. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Biener, L; Mümmler, C; Hinze, CA; Suhling, H; Korn, S; Fisser, C; Biener, A; Pizarro, C; Lenoir, A; Hackl, C; Skowasch, D; Milger, K. Real-World Data on Tezepelumab in Patients With Severe Asthma in Germany. J Allergy Clin Immunol Pract. 2024, 12(9), 2399–2407.e5. [Google Scholar] [CrossRef] [PubMed]
- Gates, J; Haris, F; Cefaloni, F; Khooshemehri, P; Green, L; Fernandes, M; Thomson, L; Roxas, C; Lam, J; d'Ancona, G; Nanzer, AM; Dhariwal, J; Jackson, DJ. Clinical and Biological Remission With Tezepelumab: The Real-World Response in Severe Uncontrolled Asthma. Allergy 2025, 80(6), 1669–1676. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Panettieri, R, Jr.; Lugogo, N; Corren, J; Ambrose, CS. Tezepelumab for Severe Asthma: One Drug Targeting Multiple Disease Pathways and Patient Types. J Asthma Allergy 2024, 17, 219–236. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hirai, K; Uehara, S; Shirai, T; Akamatsu, T; Itoh, K. Tezepelumab Targeting Thymic Stromal Lymphopoietin Enhances Steroid Sensitivity in Patients With Severe Asthma. Allergy 2026, 81(1), 277–280. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pelaia, C; Greco, M; Iaccino, E; Crimi, C; Biafora, M; Dragone, F; et al. Short-term therapeutic effectiveness of tezepelumab in patients with severe asthma: A real-world study. Int Immunopharmacol. 2025, 162, 115185. [Google Scholar] [CrossRef] [PubMed]
- Mailhot-Larouche, S; Celis-Preciado, C; Heaney, LG; Couillard, S. Identifying super-responders: A review of the road to asthma remission. Ann Allergy Asthma Immunol 2025, 134(1), 31–45. [Google Scholar] [CrossRef] [PubMed]
- Shackleford, A; Heaney, LG; Redmond, C; McDowell, PJ; Busby, J. Clinical remission attainment, definitions, and correlates among patients with severe asthma treated with biologics: a systematic review and meta-analysis. Lancet Respir Med. 2025, 13(1), 23–34. [Google Scholar] [CrossRef] [PubMed]
- Furci, F; Scaramozzino, MU; Talesa, GR; Pelaia, C. Connections and Unmet Needs: Severe Asthma Biologics and Osteoporosis. Biomedicines 2025, 13(1), 197. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lupia, C; Battaglia, C; Pastore, D; Lee, Y; Piazzetta, GL; Chiarella, E; et al. Tezepelumab in severe asthma: chest computed tomography assessment of airway remodeling and clinical remission. Front Pharmacol. 2026, 17, 1757754. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Corren, J; Pham, TH; Garcia Gil, E; Sałapa, K; Ren, P; Parnes, JR; et al. Baseline type 2 biomarker levels and response to tezepelumab in severe asthma. Allergy 2022, 77(6), 1786–1796. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wechsler, ME; Brusselle, G; Virchow, JC; Bourdin, A; Kostikas, K; Llanos, JP; et al. Clinical response and on-treatment clinical remission with tezepelumab in a broad population of patients with severe, uncontrolled asthma: results over 2 years from the NAVIGATOR and DESTINATION studies. Eur Respir J 2024, 64(6), 2400316. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Boada-Fernández-Del-Campo, C; García-Sánchez-Colomer, M; Fernández-Quintana, E; Poza-Guedes, P; Rolingson-Landaeta, JL; Sánchez-Machín, I; et al. Real-World Safety Profile of Biologic Drugs for Severe Uncontrolled Asthma: A Descriptive Analysis from the Spanish Pharmacovigilance Database. J Clin Med. 2024, 13(14), 4192. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jappe, U; Bergmann, KC; Brinkmann, F; Faihs, V; Gülsen, A; Klimek, L; et al. Biologics in allergology and clinical immunology: Update on therapies for atopic diseases, urticaria, and angioedema and on safety aspects focusing on hypersensitivity reactions. Allergol Select 2024, 8, 365–406. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
| Variable | Severe Uncontrolled Asthma |
|---|---|
| N (%) | 56 (100) |
| Age (y.o.) mean (SD) | 53.47 (±13.3) |
| <20 y.o. (%) | 3 (5.35) |
| ≥20 y.o. (%) | 53 (94.64) |
| Female Sex (%) | 40 (71.42) |
| BMI mean (SD) | 30.1 (±.53) |
| Duration of Asthma (y) mean (SD) | 29.98 (±15.44) |
| Asthma Onset at Childhood (%) | 26 (46.42) |
| Family History of Atopy (%) | 41 (80.32) |
| Asthma phenotype (T2 High/T2 Low,%) | 83,82/16,08 |
| GINA Step 5 Treatment Level (%) | 56 (100) |
| Daily Oral Corticosteroids (%) | 6 (10.74) |
| Former Use of Severe Asthma Biologics before Tezepelumab (%) | 40 (71.4) |
| None (%) | 16 (28.57) |
| One (%) | 18 (32.14) |
| Two (%) | 14 (25.0) |
| Three (%) | 6 (10.71) |
| Four (%) | 1 (1.78) |
| Five (%) | 1 (1.78) |
| Smoking | |
| Never smoker (%) | 37 (66.07) |
| Former smoker (%) | 16 (28.57) |
| Smoker (%) | 3 (5.35) |
| Bronchiectasis (Chest CT Scanner)(%) | 4 (7.1) |
| SPT+ HDM (%) | 25 (44.64) |
| Allergic Rhinitis (%) | 27 (48.21) |
| Atopic Dermatitis (%) | 4 (7.14) |
| Chronic Rinosinusitis with Nasal Polyposis (%) | 5 (8.09) |
| NERD (%) | 2 (3.57) |
| Chronic Rinosinusitis (%) | 2 (3.57) |
| GERD (%) | 15 (26.78) |
| Depression (%) | 9 (16.07) |
| Obstructive Sleep Apnea Syndrome (%) | 13 (23.21) |
| Variables | Baseline | After 26-weeks of Tezepelumab (n=56) | p | After 52-weeks of Tezepelumab (n=53) | p |
|---|---|---|---|---|---|
| ACT | 11.52±3.67 | 15.89±4.73* | <0.0001 | 17.48±4.73* | <0.0001 |
| Number of annual AEs | 2.79±2.0 | 0.5±0.72* | <0.0001 | 0.51±0.89* | <0.0001 |
| Use of OCS (mg/day) | 10.2±.8.3 | 8,41±6.2 | 0.1831 | 6.9±2.4* | <0.0140 |
| FVC (mL) | 2940±992 | 3289±1234* | 0.0161 | 3214±1148* | 0.0125 |
| FEV1 (mL) | 2061±895 | 2206±923 | 0.0572 | 2316±958* | 0.0384 |
| FEV1% | 69.31±19.2 | 75.32±17.68* | 0.0043 | 76.24±20.63* | 0.0012 |
| FENO (ppb) | 29.17±19.9 | 23.93±15.42 | 0.0889 | 26.04±16.87 | 0.1960 |
| Blood Eosinophils/μL | 234±231 | 146±120* | 0.0012 | 147±110* | 0.0125 |
| Total IgE (IU/mL) | 354±549 | 240±382 | 0.1324 | 234±409 | 0.2356 |
| sIgE D. pteronyssinus (kUA/L) | 21.17±33.0 | N/A | N/A | 17.15±4.32 | 0.1833 |
| sIgE D. farinae (kUA/L) | 17.1±30.21 | N/A | N/A | 13.58±4.7 | 0.1499 |
| SNOT-22 | 59.94±19.36 | 55.67±23.69 | 0.5171 | 46.76±26.8 | 0.373 |
| Combined BARS | - | 0.95±0.53 | - | 1.06±0.55 | 0.0501 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




