Submitted:
06 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
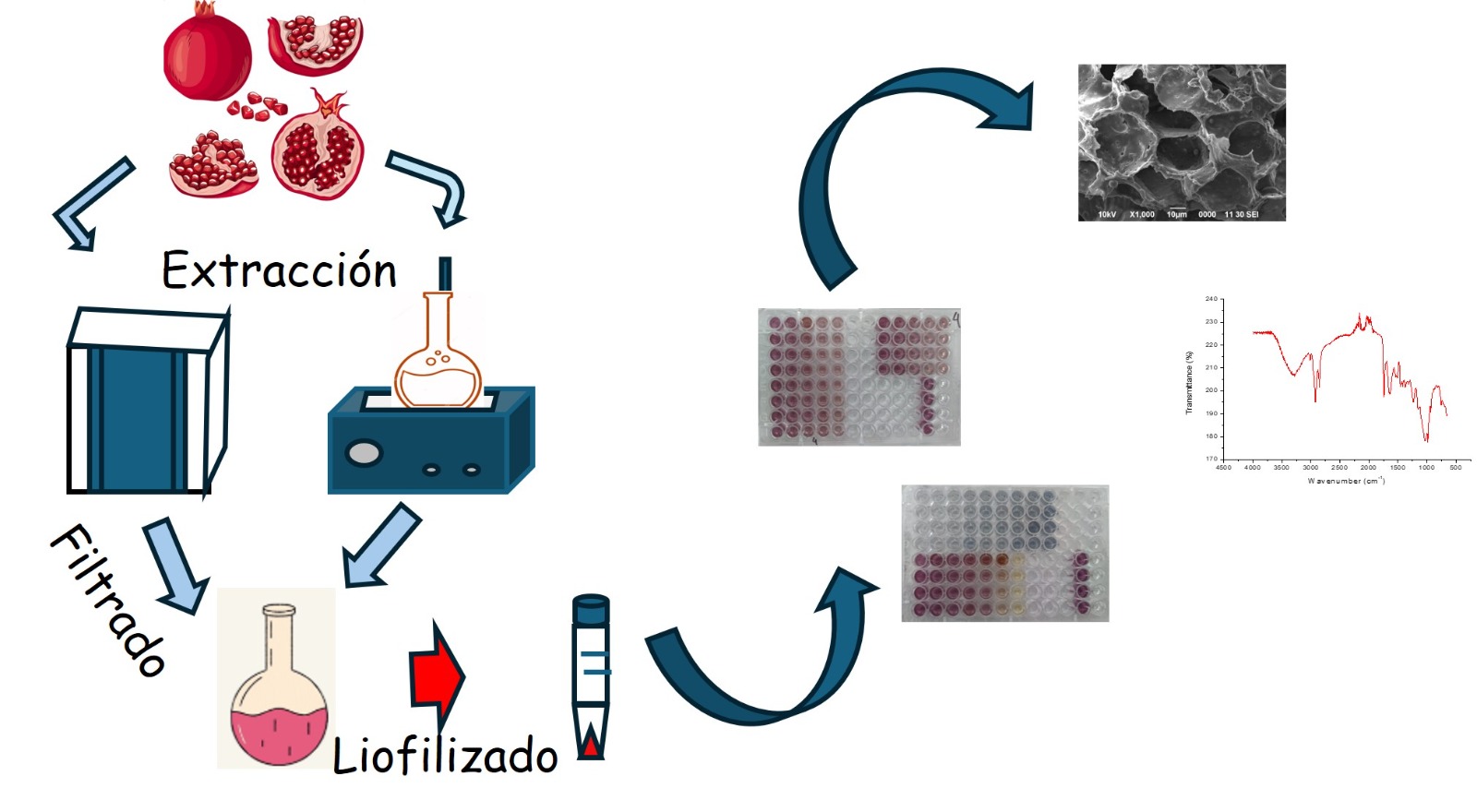
1. Introduction
2. Results and Discussion
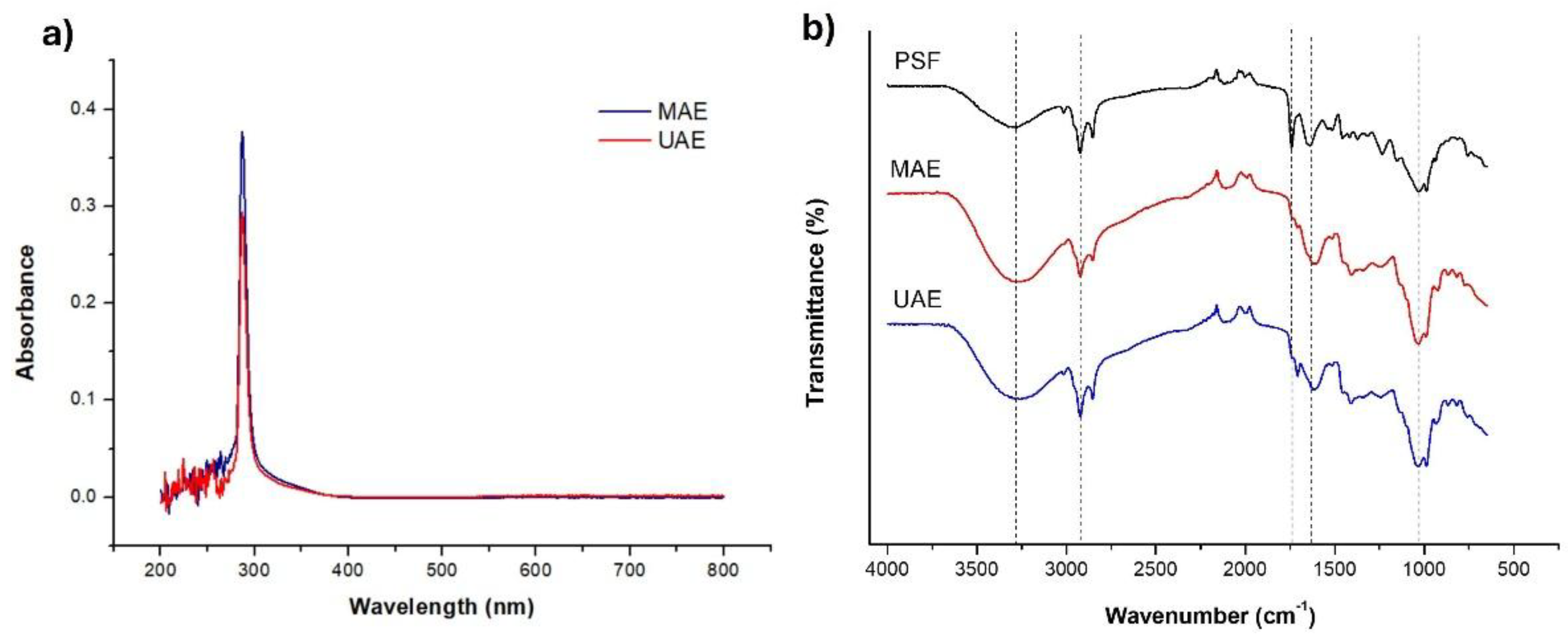
2.1. UV-Vis and Infrared Spectroscopies
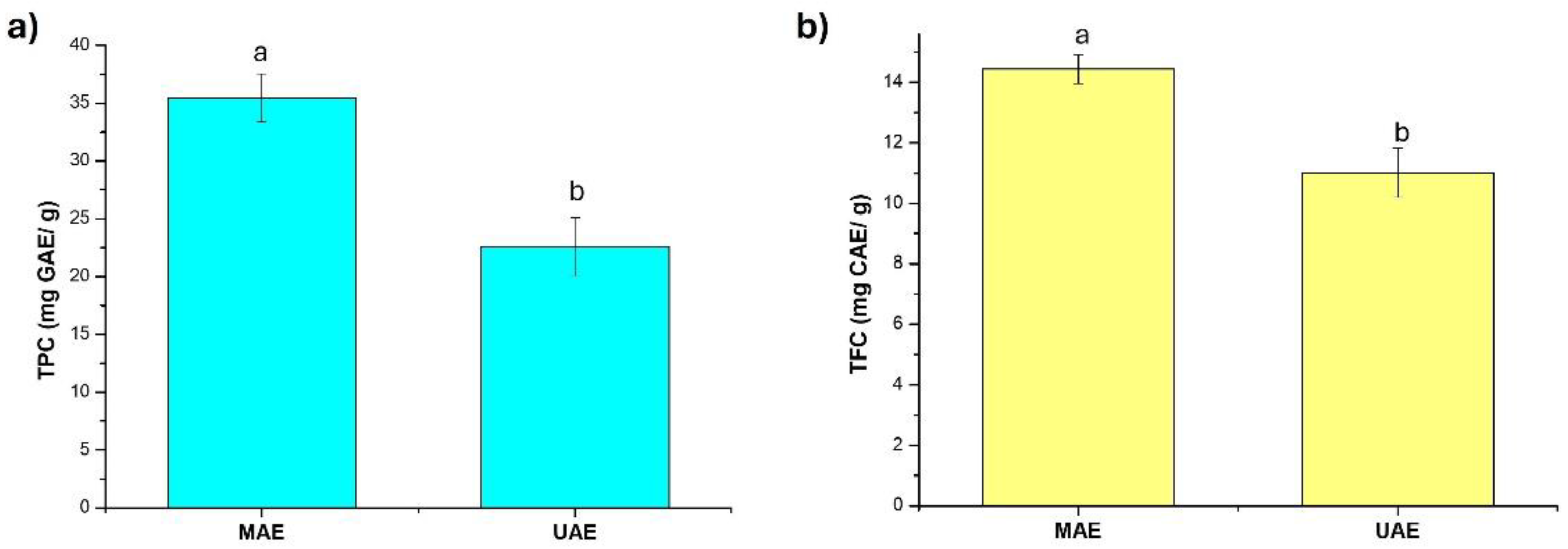
2.2. Total Phenolic Content and Flavonoid Content
2.3. Antioxidant Capacity
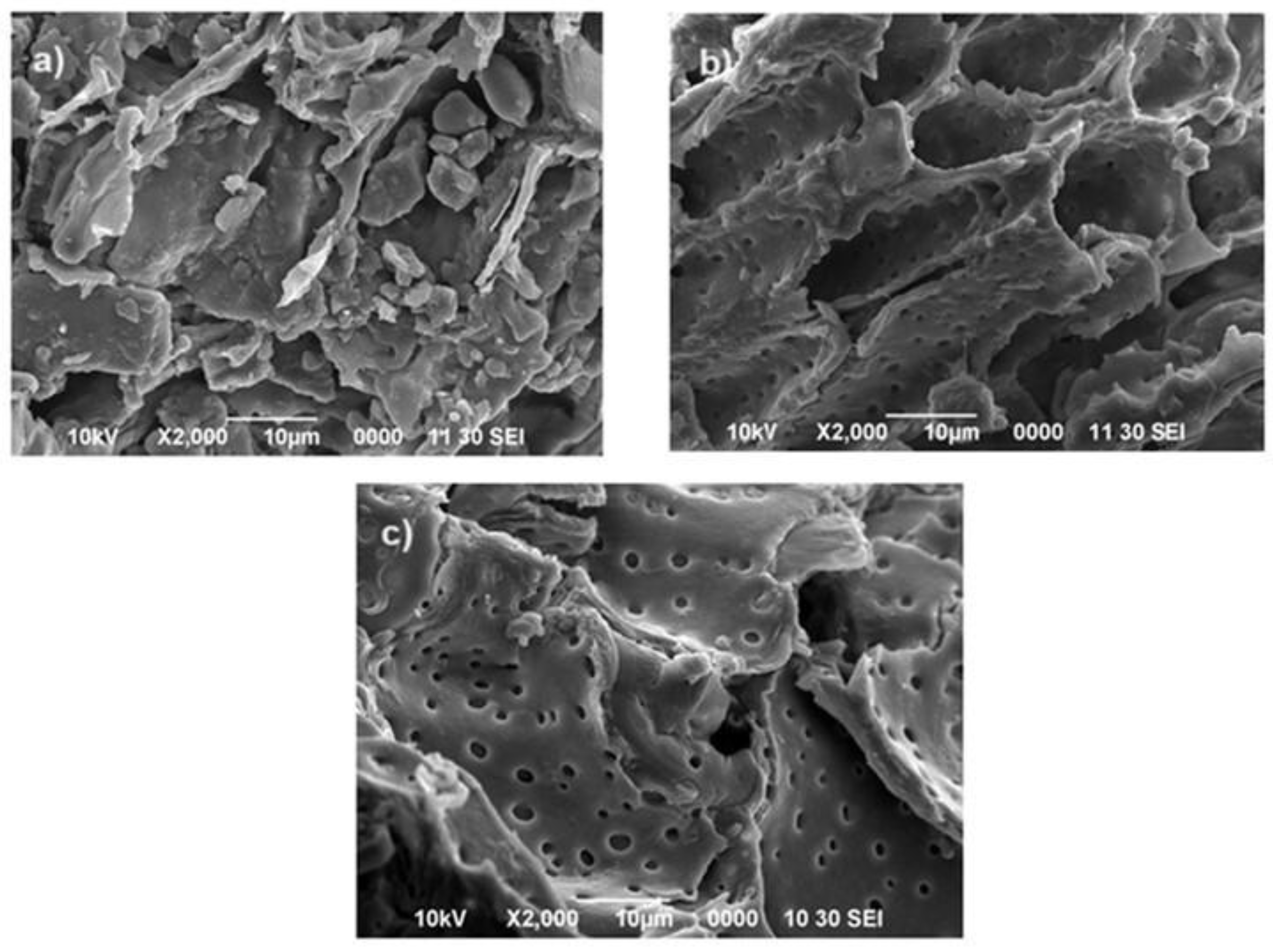
2.4. SEM Microscopy
2.5. HPLC-ESI-MS Compound Identification
4. Materials and Methods
4.1. Materials
4.2. Conditioning of Raw Materials
4.3. Microwave-Assisted Extraction
4.4. Ultrasound-Assisted Extraction
4.5. Total Phenol Content
4.6. Total Flavonoid Content
4.7. FRAP
4.8. DPPH and ABTS
4.9. Scanning Electron Microscopy (SEM)
4.10. UV-Vis and Fourier Transform Infrared (FTIR) Spectroscopy
4.11. HPLC-ESI-MS Analysis
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MAE | Microwave assisted extraction |
| UAE | Ultrasound assisted extraction |
| PSF | Pomegranate seed flour |
| TPC | Total phenolic content |
| TFC | Total flavonoid content |
References
- El Hosry, L.; Bou-Mitri, C.; Dargham, M. B.; Abou Jaoudeh, M.; Farhat, A.; El Hayek, J.; Bou Mosleh, J. M.; Bou-Maroun, E. Phytochemical composition, biological activities and antioxidant potential of pomegranate fruit, juice and molasses: A review. Food Bioscience 2023, 55, 103034. [Google Scholar] [CrossRef]
- SADER. Secretaría de Agricultura y Desarrollo Rural. Que hay detrás de la producción de granada. 2022. Available online: https://www.gob.mx/agricultura/articulos/que-hay-detras-de-la-produccion-de-granada?idiom=es.
- Singh, N.; Kumar, S.; Patle, D. S. Intensification of extraction of bioactive compounds from pomegranate peel using an ultrasound-microwave assisted extraction approach: Parametric optimization, kinetics and thermodynamics. Separation and Purification Technology 2025, 359, 130681. [Google Scholar] [CrossRef]
- Zhang, S.; Tan, T.; Wang, J.; Ma, T.; Li, T. Green extraction of polyphenols from pomegranate seeds by ultrasound-assisted deep eutectic solvent extraction: Optimization and bioactivity. Sustainable Chemistry and Pharmacy 2024, 41, 101710. [Google Scholar] [CrossRef]
- Cheng, J.; Li, J.; Xiong, R. G.; Wu, S. X.; Huang, S. Y.; Zhou, D. D.; Saimaiti, A.; Shang, A.; Feng, Y.; Gan, R.Y.; Li, H. B. Bioactive compounds and health benefits of pomegranate: An updated narrative review. Food Bioscience 2023, 102629. [Google Scholar] [CrossRef]
- Pogorzelska-Nowicka, E.; Hanula, M.; Pogorzelski, G. Extraction of polyphenols and essential oils from herbs with green extraction methods–An insightful review. Food Chemistry 2024, 140456. [Google Scholar] [CrossRef]
- Chan, C. H.; Yusoff, R.; Ngoh, G. C.; Kung, F. W. L. Microwave-assisted extractions of active ingredients from plants. Journal of Chromatography A 2011, 1218(37), 6213–6225. [Google Scholar] [CrossRef]
- Yusoff, I. M.; Taher, Z. M.; Rahmat, Z.; Chua, L. S. A review of ultrasound-assisted extraction for plant bioactive compounds: Phenolics, flavonoids, thymols, saponins and proteins. Food research international 2022, 157, 111268. [Google Scholar] [CrossRef]
- Jha, A. K.; Sit, N. Extraction of bioactive compounds from plant materials using combination of various novel methods: A review. Trends in Food Science & Technology 2022, 119, 579–591. [Google Scholar] [CrossRef]
- Catalin MOT, A.; Silaghi-Dumitrescu, R.; Sarbu, C. Rapid and effective evaluation of the antioxidant capacity of propolis extracts using DPPH bleaching kinetic profiles, FT-IR and UV―vis spectroscopic data. Journal of food composition and analysis 2011, 24(4-5), 516–522. [Google Scholar] [CrossRef]
- Masek, A.; Chrzescijanska, E.; Kosmalska, A.; Zaborski, M. Antioxidant activity determination in Sencha and Gun Powder green tea extracts with the application of voltammetry and UV-VIS spectrophotometry. Comptes Rendus. Chimie 2017, 15(5), 424–427. [Google Scholar] [CrossRef]
- Jha, A. K.; Sit, N. Effect of ultrasound, microwave, and enzymatically pre-treated Terminalia chebula pulp on extraction of bioactive compounds using supercritical CO2. Sustainable Chemistry and Pharmacy 2023, 33, 101098. [Google Scholar] [CrossRef]
- Al-Ansari, M. M.; Al-Dahmash, N. D.; Jhanani, G. K. Anti-Candida, antioxidant and antidiabetic potential of ethyl acetate extract fraction-7a from Cymodocea serrulata and its bioactive compound characterization through FTIR and NMR. Environmental Research 2023, 229, 115985. [Google Scholar] [CrossRef]
- Rashid, R.; Wani, S. M.; Manzoor, S.; Masoodi, F. A.; Dar, M. M. Green extraction of bioactive compounds from apple pomace by ultrasound assisted natural deep eutectic solvent extraction: Optimisation, comparison and bioactivity. Food Chemistry 2023, 398, 133871. [Google Scholar] [CrossRef]
- Cheng, M.; He, J.; Li, C.; Wu, G.; Zhu, K.; Chen, X.; Zhang, Y.; Tan, L. Comparison of microwave, ultrasound and ultrasound-microwave assisted solvent extraction methods on phenolic profile and antioxidant activity of extracts from jackfruit (Artocarpus heterophyllus Lam.) pulp. Lwt 2023, 173, 114395. [Google Scholar] [CrossRef]
- Nayak, B.; Dahmoune, F.; Moussi, K.; Remini, H.; Dairi, S.; Aoun, O.; Khodir, M. Comparison of microwave, ultrasound and accelerated-assisted solvent extraction for recovery of polyphenols from Citrus sinensis peels. Food chemistry 2015, 187, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Alzagameem, A.; Khaldi-Hansen, B. E.; Büchner, D.; Larkins, M.; Kamm, B.; Witzleben, S.; Schulze, M. Lignocellulosic biomass as source for lignin-based environmentally benign antioxidants. Molecules 2018, 23(10), 2664. [Google Scholar] [CrossRef]
- Kalaycıoğlu, Z.; Erim, F. B. Total phenolic contents, antioxidant activities, and bioactive ingredients of juices from pomegranate cultivars worldwide. Food chemistry 2017, 221, 496–507. [Google Scholar] [CrossRef]
- Manye, SJ; Saleh, JS; Ishaya, HB; Chiroma, SM; Attah, MOO; Dibal, NI. Análisis fitoquímico y actividad antioxidante in vitro de extractos acuosos y metanólicos de aloe vera. Investigación farmacológica - Medicina China Moderna 2023, 8, 100291. [Google Scholar] [CrossRef]
- Pourshoaib, S. J.; Ghatrami, E. R.; Shamekhi, M. A. Comparing ultrasonic-and microwave-assisted methods for extraction of phenolic compounds from Kabkab date seed (Phoenix dactylifera L.) and stepwise regression analysis of extracts antioxidant activity. Sustainable Chemistry and Pharmacy 2022, 30, 100871. [Google Scholar] [CrossRef]
- More, P. R.; Arya, S. S. Intensification of bio-actives extraction from pomegranate peel via microwave irradiation: Effect of factors, optimization, kinetics, and bioactive profiling. Chemical Engineering and Processing-Process Intensification 2024, 202, 109839. [Google Scholar] [CrossRef]
- Che, H.; Zhang, R.; Wang, X.; Yu, H.; Shi, X.; Yi, J.; Li, J.; Qi, Q.; Dong, R.; Li, Q. Ultrasound-assisted extraction of polyphenols from Phyllanthi Fructus: Comprehensive insights from extraction optimization and antioxidant activity. Ultrasonics Sonochemistry 2024, 111, 107083. [Google Scholar] [CrossRef]
- Fourati, M.; Smaoui, S.; Hlima, H. B.; Elhadef, K.; Braïek, O. B.; Ennouri, K.; Chakchouk Mtibaa, A.; Mellouli, L. Bioactive compounds and pharmacological potential of pomegranate (Punica granatum) seeds-a review. Plant Foods for Human Nutrition 2020, 75, 477–486. [Google Scholar] [CrossRef]
- Khan, H.; Ullah, H.; Aschner, M.; Cheang, W. S.; Akkol, E. K. Neuroprotective effects of quercetin in Alzheimer’s disease. Biomolecules 2019, 10(1), 59. [Google Scholar] [CrossRef]
- Li, Y.; Yao, J.; Han, C.; Yang, J.; Chaudhry, M. T.; Wang, S.; Liu, H.; Yin, Y. Quercetin, inflammation and immunity. Nutrients 2016, 8(3), 167. [Google Scholar] [CrossRef]
- Santos-Buelga, C.; Scalbert, A. Proanthocyanidins and tannin-like compounds–nature, occurrence, dietary intake and effects on nutrition and health. Journal of the Science of Food and Agriculture 2000, 80(7), 1094–1117. [Google Scholar] [CrossRef]
- Villamil-Galindo, J. E.; Piagentini, A. Green solvents for the recovery of phenolic compounds from strawberry (Fragaria x ananassa Duch) and apple (Malus domestica) agro-industrial bio-wastes. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina 2024, 56(1), 149–160. [Google Scholar] [CrossRef]
- Farooq, M.; Ahmad, R.; Shahzad, M.; Sajjad, Y.; Hassan, A.; Shah, M. M.; Naz, S.; Khan, S. A. Differential variations in total flavonoid content and antioxidant enzymes activities in pea under different salt and drought stresses. Scientia Horticulturae 2021, 287, 110258. [Google Scholar] [CrossRef]
- Gullon, B.; Eibes, G.; Moreira, M. T.; Davila, I.; Labidi, J.; Gullon, P. Antioxidant and antimicrobial activities of extracts obtained from the refining of autohydrolysis liquors of vine shoots. Industrial Crops and Products 2017, 107, 105–113. [Google Scholar] [CrossRef]
- Ferreyra, S.; Bottini, R.; Fontana, A. Assessment of grapevine stems as source of phenolics with antioxidant properties. Revista de la Facultad de Ciencias Agrarias UNCuyo 2019, 51(2), 417–426. Available online: https://revistas.uncu.edu.ar/ojs/index.php/RFCA/article/view/2728.
- Rumpf, J.; Burger, R.; Schulze, M. Statistical evaluation of DPPH, ABTS, FRAP, and Folin-Ciocalteu assays to assess the antioxidant capacity of lignins. International Journal of Biological Macromolecules 2023, 233, 123470. [Google Scholar] [CrossRef]
| Method | FRAP (mmoles TEAC/g) | DPPH IC50 (mmoles TEAC/g) | ABTS (mmoles TEAC/g) |
| MAE | 0.19 ± 0.03 a | 0.17 ± 0.01 a | 0.41 ± 0.02 a |
| UAE | 0.13 ± 0.00 b | 0.15 ± 0.00 b | 0.39 ± 0.07 b |
| Sample | RT (min) | [m-H]+,* | MW& | Formula | Identification |
| 1 | 0.76 | 474.22 | 474 | C9H8O3 | p-Coumaric acid |
| 2 | 14.76 | 518.32 | 518.424 | C24H22O13 | Malonylgenistin |
| 3 | 15.29 | 611.42 | 611.525 | C27H31O16 | Cyanidin |
| 4 | 16.58 | 546.40 | 544.504 | C27H28O12 | Diferuloylquinic acid |
| 5 | 18.57 | 558.46 | 558.617 | C30H38O10 | Secoisolariciresinol-sesquilignan |
| 6 | 24.29 | 654.48 | 652.554 | C29H32O17 | Quercetin |
| Sample | RT (min) | [m-H]+,* | MW& | Formula | Identification |
| 1 | 0.42 | 223.98 | 224.21 | C11H12O5 | Sinapic acid |
| 2 | 0.77 | 474.22 | 474 | C9H8O3 | p-Coumaric acid |
| 3 | 2.38 | 611.16 | 611.527 | C30H27O14 | Cyanidin |
| 4 | 11.86 | 460.49 | 462.36 | C21H18O12 | Kaempferol |
| 5 | 16.24 | 678.48 | 680.564 | C30H32O18 | Chrysoeriol |
| 6 | 17.3 | 705.53 | 706.646 | C36H34O15 | Epicatechin |
| 7 | 23.09 | 636.46 | 638.484 | C27H26O18 | Luteolin |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




