Submitted:
04 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
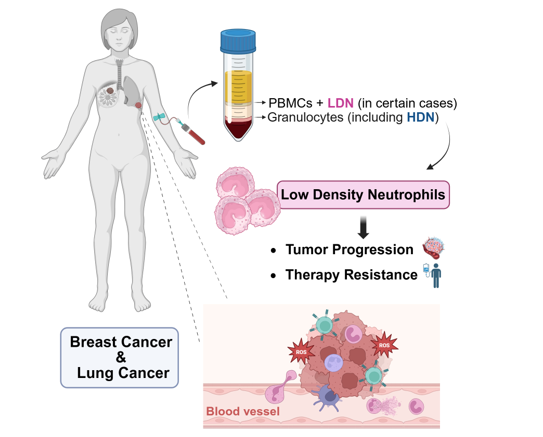
Keywords:
Summary Sentence
Key Concepts
- Historically regarded as bystanders, neutrophils are now well-established key players in cancer
- In cancer, LDNs exhibit activated, immunosuppressive phenotypes that promote tumor progression.
- In breast cancer, elevated LDNs correlate with metastatic disease, poor therapeutic response, farter disease progression and worse prognosis.
- In lung cancer, increased LDN levels are also associated with advanced disease stage and poorer clinical outcomes.
- LDNs are emerging as accessible biomarkers and promissing therapeutic targets in precision oncology.
Open Questions
- Which molecular pathways drive the generation, expansion, and persistence of LDNs in cancer?
- How can LDN subpopulations be consistently defined and standardized across studies?
- Which LDN associated markers most accurately predict prognosis or treatment response?
- How should LDN quantification be incorporated into clinical decision making in breast and lung cancer?
- Through which mechanisms can LDN expansion or function be therapeutically modulated?
Introduction
1. Overview of Neutrophils
1.1. Basic Characteristics and Lifespan
1.2. Effector Mechanisms: Phagocytosis, Degranulation and NETosis
1.2.1. Phagocytosis
1.2.2. Degranulation
1.2.3. NETosis
1.3. Recruitment and Resolution of Inflammation
2. Development of Neutrophils
2.1. Hematopoiesis in the Bone Marrow
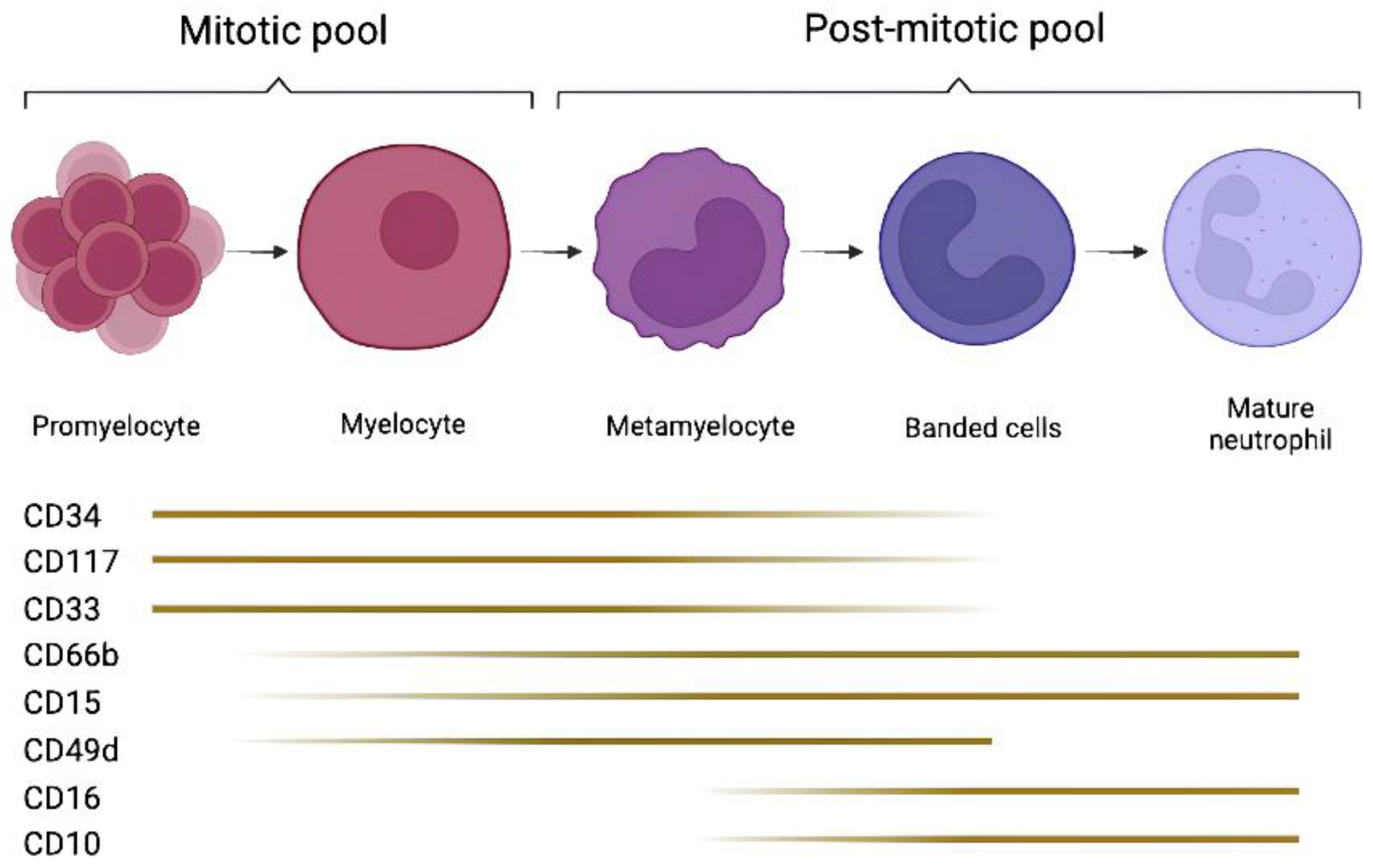
2.2. Maturation Stages and Phenotypic Markers
2.3. Mobilization to Circulation
3. Neutrophils in the Immune System
4. Neutrophils in Cancer
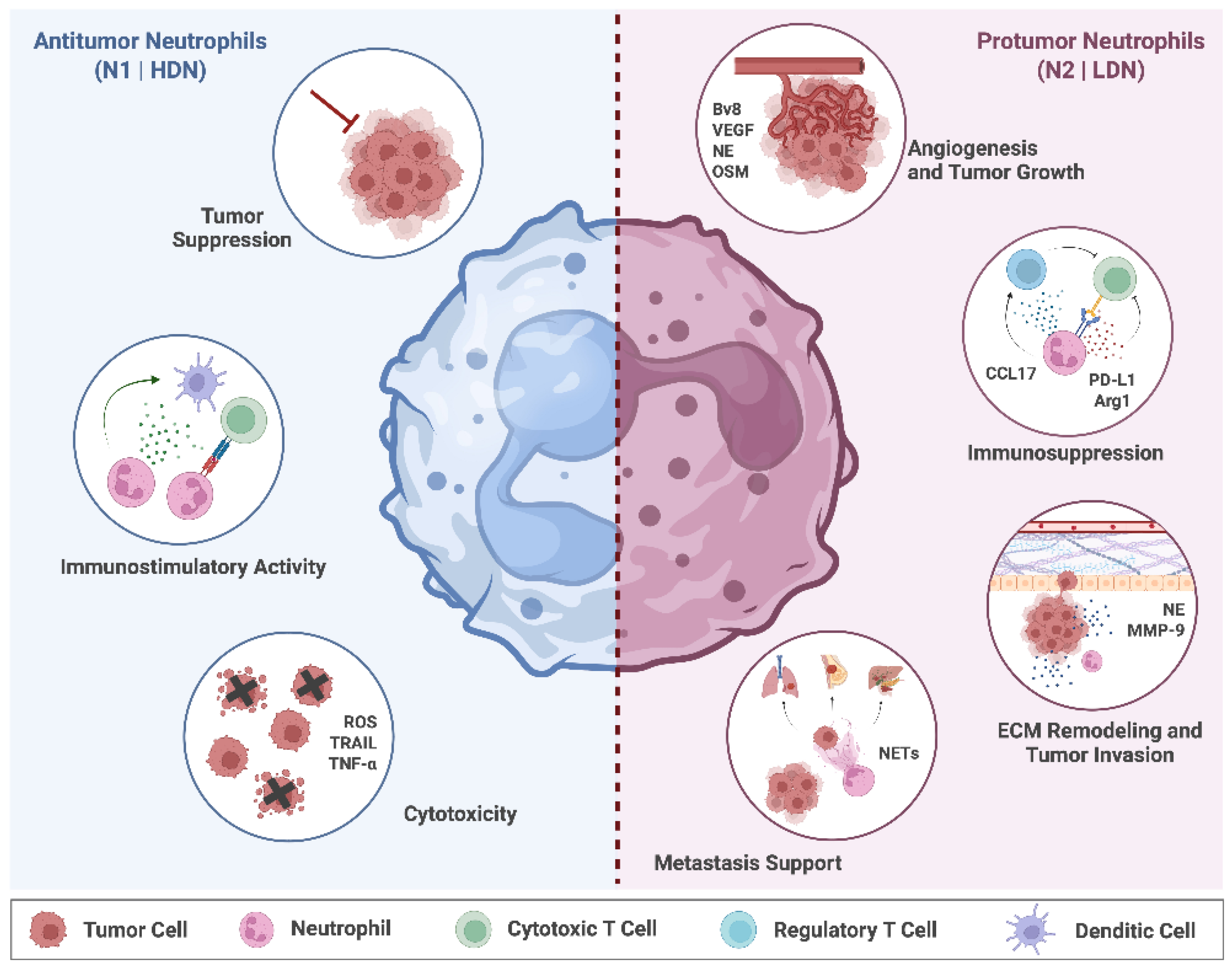
4.1. TANs
4.2. Circulating Neutrophils in Cancer
5. LDNs
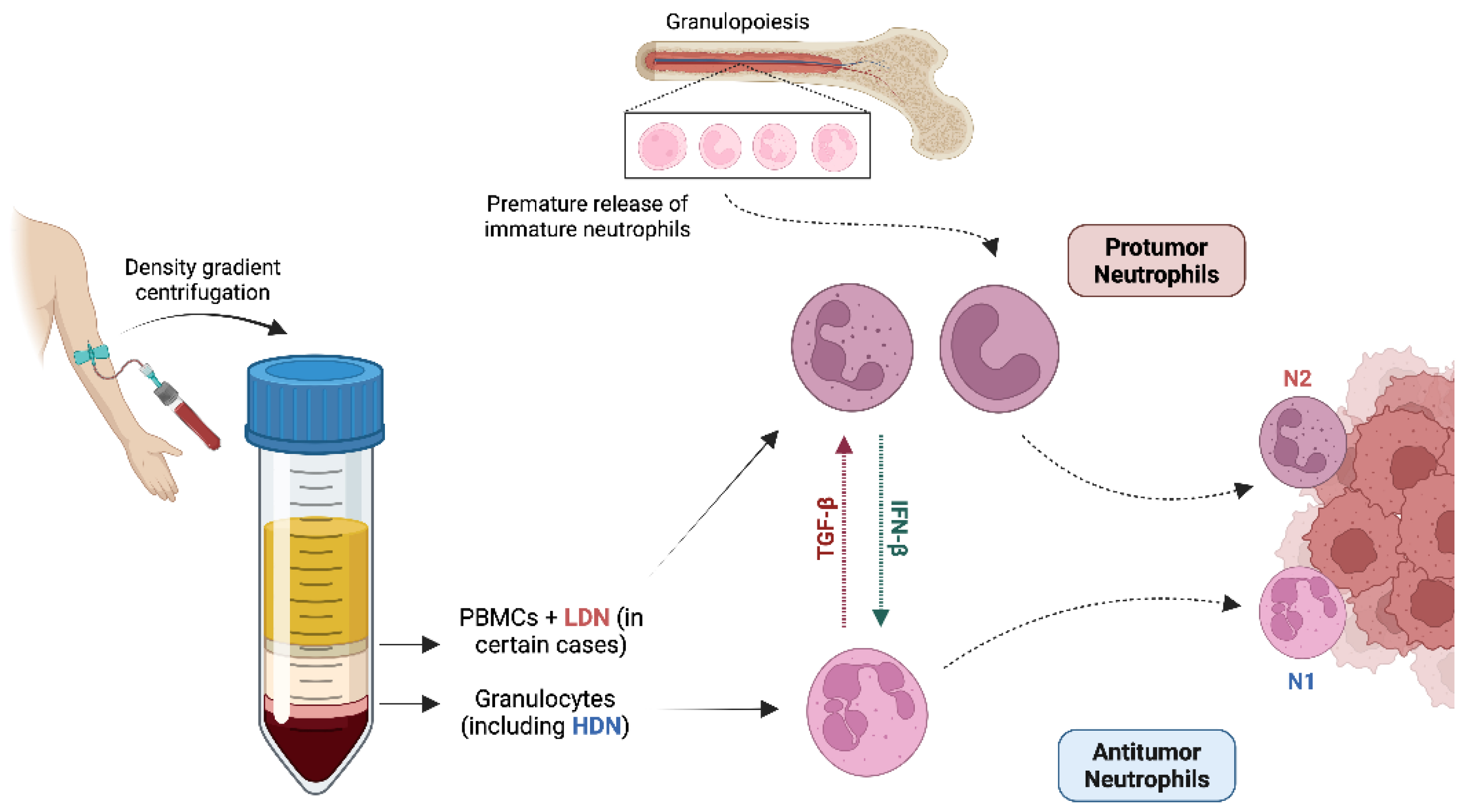
5.1. Definition and Historical Discovery
5.2. Distinctive Density, Phenotype, and Function Compared to HDNs
5.3. Origin, Development, and Heterogeneity of LDNs
5.4. Activation and Metabolic Properties of LDNs
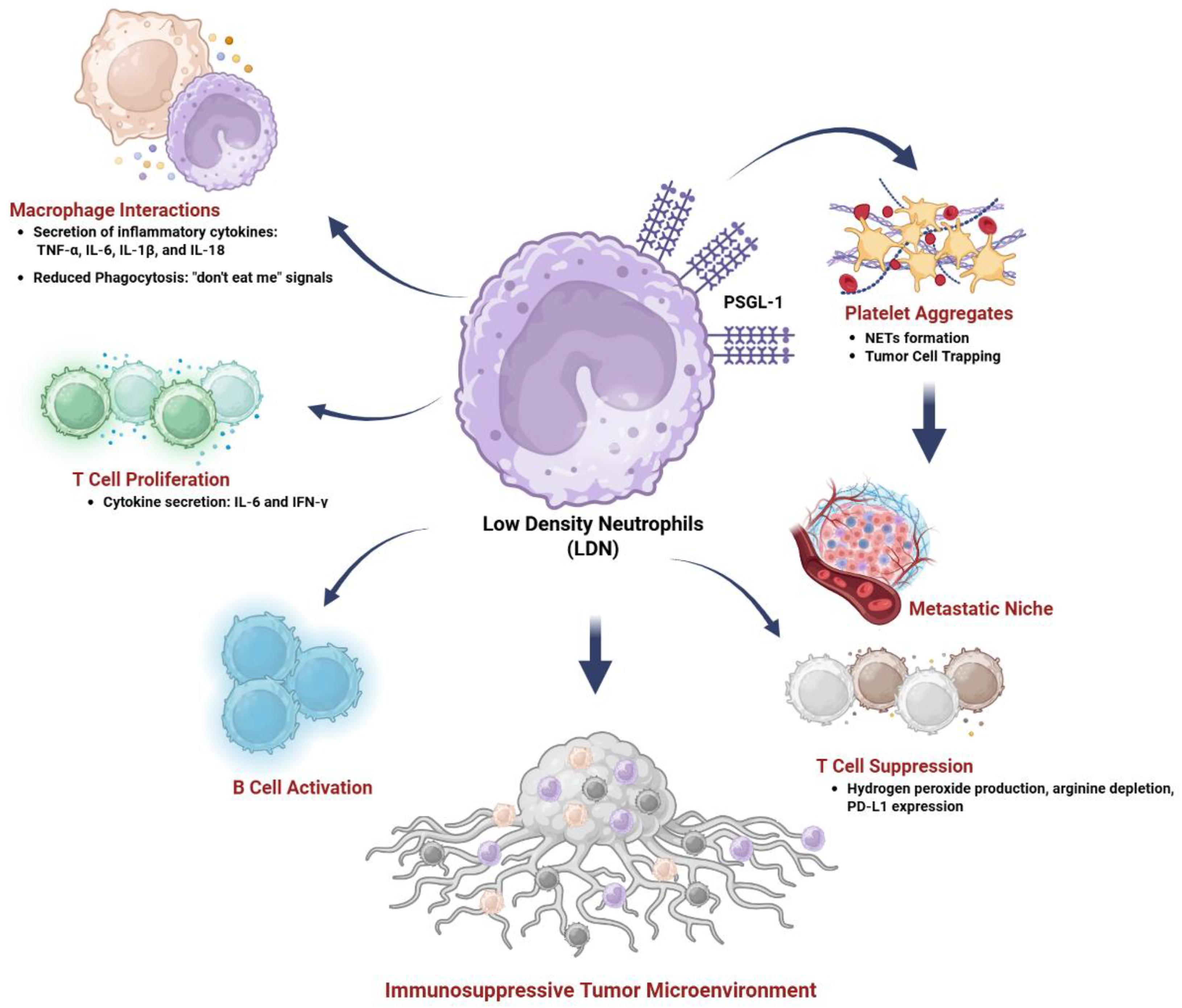
5.5. Interactions of LDNs with Other Cells in Immune Responses and Cancer
6. Dynamic Changes of LDNs During Human Cancer
6.1. Variation in Frequency, Phenotype, and Functional Relevance Across Cancer Stages
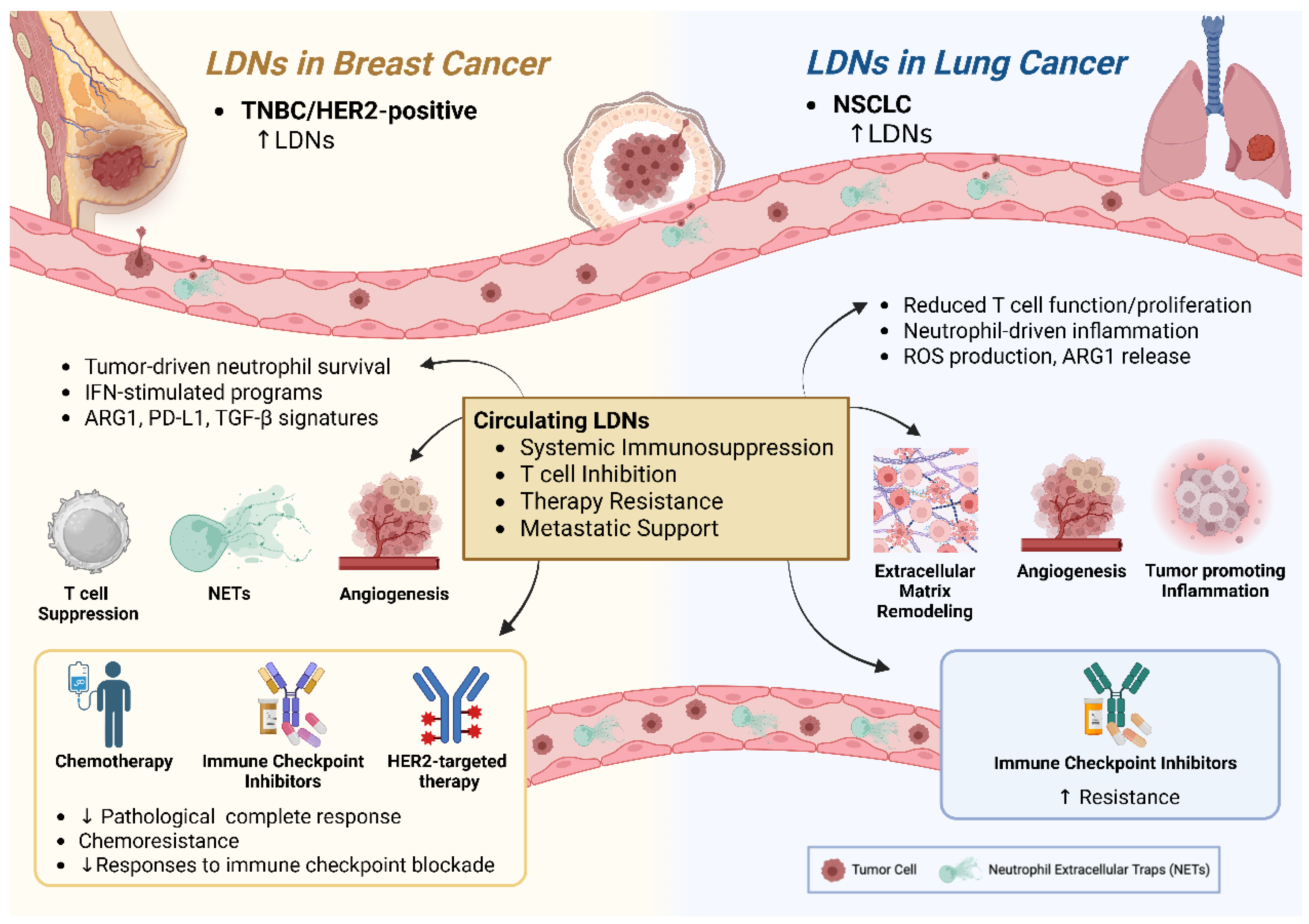
6.2. LDNs in Breast Cancer: From Subtype Associations to Clinical Outcomes
6.3. LDNs in Lung Cancer: Clinical Relevance and Immunoregulatory Functions
7. Therapeutic Potential of Targeting LDNs
- Modulation of neutrophil plasticity – targeting TGF-β or other tumor-derived factors to prevent the conversion of HDNs into pro-tumor LDNs [10].
8. Concluding Remarks
Authorship
Acknowledgments
Conflicts of Interest/ Disclosure
List of abbreviations
| CEBP | CCAAT/Enhancer Binding Protein |
| CTLs | Cytotoxic T lymphocytes |
| CXCL | C-X-C motif chemokine ligand |
| CXCR | C-X-C chemokine receptor |
| ECM | Extracellular matrix |
| G-CSF | Granulocyte colony-stimulating factor |
| GFI-1 | Growth factor independent 1 |
| GMPs | Granulocyte–monocyte progenitors |
| HDNs | High-density neutrophils |
| HSCs | Hematopoietic stem cells |
| IL | Interleukin |
| JAK/STAT | Janus kinase / Signal transducer and activator of transcription |
| LDNs | Low-density neutrophils |
| MAPK | Mitogen-activated protein kinase |
| MHC | Major histocompatibility complex |
| MMPs | Matrix metalloproteinases |
| MPO | Myeloperoxidase |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NE | Neutrophil elastase |
| NETs | Neutrophil extracellular traps |
| NETosis | NET-mediated neutrophil cell death |
| NLR | Neutrophil-to-lymphocyte ratio |
| NOD | Nucleotide-binding oligomerization domain |
| NSCLC | Non-small cell lung carcinoma |
| PAD4 | Protein-arginine deiminase 4 |
| PBMC | Peripheral blood mononuclear cell |
| PD-L1 | Programmed death-ligand 1 |
| PRRs | Pattern recognition receptors |
| PU.1 | PU box-binding protein 1 |
| RNS | Reactive nitrogen species |
| ROS | Reactive oxygen species |
| RUNX1 | Runt-related transcription factor 1 |
| TANs | Tumor-associated neutrophils |
| TGF-β | Transforming growth factor-β |
| TIME | Tumor immune microenvironment |
| TLRs | Toll-like receptors |
| TME | Tumor microenvironment |
| TNF-α | Tumor necrosis factor-α |
| TRAIL | TNF-related apoptosis-inducing ligand |
| VEGF | Vascular endothelial growth factor |
References
- Kraus, R.F.; Gruber, M.A. Neutrophils—From Bone Marrow to First-Line Defense of the Innate Immune System. Front. Immunol. 2021, 12, 767175. [Google Scholar] [CrossRef]
- Kolaczkowska, E.; Kubes, P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 2013, 13, 159–175. [Google Scholar] [CrossRef]
- Mayadas, T.N.; Cullere, X.; Lowell, C.A. The Multifaceted Functions of Neutrophils. Annu. Rev. Pathol. Mech. Dis. 2014, 9, 181–218. [Google Scholar] [CrossRef]
- Lehman, H.K.; Segal, B.H. The role of neutrophils in host defense and disease. J. Allergy Clin. Immunol. 2020, 145, 1535–1544. [Google Scholar] [CrossRef]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil Extracellular Traps Kill Bacteria. Science (80- ) 2004, 303, 1532–1535. [Google Scholar] [CrossRef]
- Fuchs, T.A.; Abed, U.; Goosmann, C.; Hurwitz, R.; Schulze, I.; Wahn, V.; Weinrauch, Y.; Brinkmann, V.; Zychlinsky, A. Novel cell death program leads to neutrophil extracellular traps. J. Cell Biol. 2007, 176, 231–241. [Google Scholar] [CrossRef]
- Malech, H.L.; DeLeo, F.R.; Quinn, M.T. The Role of Neutrophils in the Immune System: An Overview. 2014, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Carnevale, S.; Di Ceglie, I.; Grieco, G.; Rigatelli, A.; Bonavita, E.; Jaillon, S. Neutrophil diversity in inflammation and cancer. Front. Immunol. 2023, 14, 1180810. [Google Scholar] [CrossRef]
- Wu, G.; Pan, B.; Shi, H.; Yi, Y.; Zheng, X.; Ma, H.; Zhao, M.; Zhang, Z.; Cheng, L.; Huang, Y.; et al. Neutrophils’ dual role in cancer: from tumor progression to immunotherapeutic potential. Int. Immunopharmacol. 2024, 140, 112788. [Google Scholar] [CrossRef] [PubMed]
- Fridlender, Z.G.; Sun, J.; Kim, S.; Kapoor, V.; Cheng, G.; Ling, L.; Worthen, G.S.; Albelda, S.M. Polarization of Tumor-Associated Neutrophil Phenotype by TGF-β: “N1” versus “N2” TAN. Cancer Cell 2009, 16, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Ohms, M.; Möller, S.; Laskay, T. An Attempt to Polarize Human Neutrophils Toward N1 and N2 Phenotypes in vitro. Front. Immunol. 2020, 11, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, D.P.; Correia, B.F.; Salvador, R.; de Sousa, N.; Jacinto, A.; Braga, S.; Cabral, M.G. Circulating low density neutrophils of breast cancer patients are associated with their worse prognosis due to the impairment of T cell responses. Oncotarget 2021, 12, 2388–2403. [Google Scholar] [CrossRef] [PubMed]
- Sagiv, J.Y.; Michaeli, J.; Assi, S.; Mishalian, I.; Kisos, H.; Levy, L.; Damti, P.; Lumbroso, D.; Polyansky, L.; Sionov, R.V.; et al. Phenotypic Diversity and Plasticity in Circulating Neutrophil Subpopulations in Cancer. Cell Rep. 2015, 10, 562–573. [Google Scholar] [CrossRef] [PubMed]
- Scapini, P.; Marini, O.; Tecchio, C.; Cassatella, M.A. Human neutrophils in the saga of cellular heterogeneity: insights and open questions. Immunol. Rev. 2016, 273, 48–60. [Google Scholar] [CrossRef]
- Vanhaver, C.; Aboubakar Nana, F.; Delhez, N.; Luyckx, M.; Hirsch, T.; Bayard, A.; Houbion, C.; Dauguet, N.; Brochier, A.; van der Bruggen, P.; et al. Immunosuppressive low-density neutrophils in the blood of cancer patients display a mature phenotype. Life Sci. Alliance 2024, 7, e202302332. [Google Scholar] [CrossRef]
- Shaul, M.E.; Eyal, O.; Guglietta, S.; Aloni, P.; Zlotnik, A.; Forkosh, E.; Levy, L.; Weber, L.M.; Levin, Y.; Pomerantz, A.; et al. Circulating neutrophil subsets in advanced lung cancer patients exhibit unique immune signature and relate to prognosis. FASEB J. 2020, 34, 4204–4218. [Google Scholar] [CrossRef]
- Correia, B.F.; Grosa, D.; Salvador, R.; Brites, I.; Martins, T.; Vitorino, M.; Sousa, C.X.; Cristóvão-Ferreira, S.; Braga, S.; Jacinto, A.; et al. Neutrophils matter: new clinical insights on their role in the progression of metastatic breast cancer. Breast Cancer Res. 2025, 27, 223. [Google Scholar] [CrossRef]
- Valadez-Cosmes, P.; Maitz, K.; Kindler, O.; Raftopoulou, S.; Kienzl, M.; Santiso, A.; Mihalic, Z.N.; Brcic, L.; Lindenmann, J.; Fediuk, M.; et al. Identification of Novel Low-Density Neutrophil Markers Through Unbiased High-Dimensional Flow Cytometry Screening in Non-Small Cell Lung Cancer Patients. Front Immunol 2021, 12, 703846. [Google Scholar] [CrossRef]
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2024, GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2022, 74, 229–263. [Google Scholar] [CrossRef]
- Cowland, J.B.; Borregaard, N. Granulopoiesis and granules of human neutrophils. Immunol. Rev. 2016, 273, 11–28. [Google Scholar] [CrossRef]
- Pillay, J.; den Braber, I.; Vrisekoop, N.; Kwast, L.M.; de Boer, R.J.; Borghans, J.A.M.; Tesselaar, K.; Koenderman, L. In vivo labeling with 2H2O reveals a human neutrophil lifespan of 5.4 days. Blood 2010, 116, 625–627. [Google Scholar] [CrossRef]
- Othman, A.; Sekheri, M.; Filep, J.G. Roles of neutrophil granule proteins in orchestrating inflammation and immunity. FEBS J. 2022, 289, 3932–3953. [Google Scholar] [CrossRef]
- Baz, A.A.; Hao, H.; Lan, S.; Li, Z.; Liu, S.; Chen, S.; Chu, Y. Neutrophil extracellular traps in bacterial infections and evasion strategies. Front Immunol 2024, 15, 1357967. [Google Scholar] [CrossRef] [PubMed]
- Sadik, C.D.; Kim, N.D.; Luster, A.D. Neutrophils cascading their way to inflammation. Trends Immunol. 2011, 32, 452–460. [Google Scholar] [CrossRef]
- Hyun, Y.-M.; Hong, C.-W. Deep insight into neutrophil trafficking in various organs. J. Leukoc. Biol. 2017, 102, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Herrero-Cervera, A.; Soehnlein, O.; Kenne, E. Neutrophils in chronic inflammatory diseases. Cell Mol. Immunol. 2022, 19, 177–191. [Google Scholar] [CrossRef]
- Silvestre-Roig, C.; Braster, Q.; Ortega-Gomez, A.; Soehnlein, O. Neutrophils as regulators of cardiovascular inflammation. Nat. Rev. Cardiol. 2020, 17, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Overbeeke, C.; Tak, T.; Koenderman, L. The journey of neutropoiesis: how complex landscapes in bone marrow guide continuous neutrophil lineage determination. Blood 2022, 139, 2285–2293. [Google Scholar] [CrossRef]
- Hageb, A.; Farjia, M.; Osei-Sarpong, C.; Silvestre-Roig, C. Ontogenetic drivers of neutrophil heterogeneity. Exp. Hematol. 2025, 151, 104863. [Google Scholar] [CrossRef]
- Montaldo, E.; Lusito, E.; Bianchessi, V.; Caronni, N.; Scala, S.; Basso-Ricci, L.; Cantaffa, C.; Masserdotti, A.; Barilaro, M.; Barresi, S.; et al. Cellular and transcriptional dynamics of human neutrophils at steady state and upon stress. Nat. Immunol. 2022, 23, 1470–1483. [Google Scholar] [CrossRef]
- Thind, M.K.; Uhlig, H.H.; Glogauer, M.; Palaniyar, N.; Bourdon, C.; Gwela, A.; Lancioni, C.L.; Berkley, J.A.; Bandsma, R.H.J.; Farooqui, A. A metabolic perspective of the neutrophil life cycle: new avenues in immunometabolism. Front. Immunol. 2024, 14. [Google Scholar] [CrossRef]
- Friedman, A.D. C/EBPα in normal and malignant myelopoiesis. Int. J. Hematol. 2015, 101, 330–341. [Google Scholar] [CrossRef]
- Mehta, H.M.; Corey, S.J. G-CSF, the guardian of granulopoiesis. Semin. Immunol. 2021, 54, 101515. [Google Scholar] [CrossRef]
- Rosales, C. Neutrophil: A Cell with Many Roles in Inflammation or Several Cell Types? Front Physiol 2018, 9, 113. [Google Scholar] [CrossRef] [PubMed]
- McKenna, E.; Mhaonaigh, A.U.; Wubben, R.; Dwivedi, A.; Hurley, T.; Kelly, L.A.; Stevenson, N.J.; Little, M.A.; Molloy, E.J. Neutrophils: Need for Standardized Nomenclature. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef]
- Elghetany, M.T. Surface Antigen Changes during Normal Neutrophilic Development: A Critical Review. Blood Cells, Mol Dis 2002, 28, 260–274. [Google Scholar] [CrossRef]
- Häger, M.; Cowland, J.B.; Borregaard, N. Neutrophil granules in health and disease. J. Intern. Med. 2010, 268, 25–34. [Google Scholar] [CrossRef]
- Zhang, F.; Xia, Y.; Su, J.; Quan, F.; Zhou, H.; Li, Q.; Feng, Q.; Lin, C.; Wang, D.; Jiang, Z. Neutrophil diversity and function in health and disease. Signal Transduct. Target. Ther. 2024, 9, 343. [Google Scholar] [CrossRef]
- Silvestre-Roig, C.; Hidalgo, A.; Soehnlein, O. Neutrophil heterogeneity: implications for homeostasis and pathogenesis. Blood 2016, 127, 2173–2181. [Google Scholar] [CrossRef] [PubMed]
- Tsioumpekou, M.; Krijgsman, D.; Leusen, J.H.W.; Olofsen, P.A. The Role of Cytokines in Neutrophil Development, Tissue Homing, Function and Plasticity in Health and Disease. Cells 2023, 12, 1981. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, W.; Yang, F.; Xu, Y.; Feng, C.; Zhao, Y. The regulatory roles of neutrophils in adaptive immunity. Cell Commun. Signal 2019, 17, 147. [Google Scholar] [CrossRef]
- Mantovani, A.; Cassatella, M.A.; Costantini, C.; Jaillon, S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat. Rev. Immunol. 2011, 11, 519–531. [Google Scholar] [CrossRef]
- Shafqat, A.; Khan, J.A.; Alkachem, A.Y.; Sabur, H.; Alkattan, K.; Yaqinuddin, A.; Sing, G.K. How Neutrophils Shape the Immune Response: Reassessing Their Multifaceted Role in Health and Disease. Int. J. Mol. Sci. 2023, 24, 17583. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef]
- Uribe-Querol, E.; Rosales, C. Neutrophils in Cancer: Two Sides of the Same Coin. J. Immunol. Res. 2015, 2015, 1–21. [Google Scholar] [CrossRef]
- Garcia-Flores, L.A.; Dawid De Vera, M.T.; Pilo, J.; Rego, A.; Gomez-Casado, G.; Arranz-Salas, I.; Hierro Martín, I.; Alcaide, J.; Torres, E.; Ortega-Gomez, A.; et al. Increased neutrophil counts are associated with poor overall survival in patients with colorectal cancer: a five-year retrospective analysis. Front. Immunol. 2024, 15, 1415804. [Google Scholar] [CrossRef]
- Ma, J.; Kuzman, J.; Ray, A.; Lawson, B.O.; Khong, B.; Xuan, S.; Hahn, A.W.; Khong, H.T. Neutrophil-to-lymphocyte Ratio (NLR) as a predictor for recurrence in patients with stage III melanoma. Sci. Rep. 2018, 8, 4044. [Google Scholar] [CrossRef]
- Dirican, A.; Kucukzeybek, B.B.; Alacacioglu, A.; Kucukzeybek, Y.; Erten, C.; Varol, U.; Somali, I.; Demir, L.; Bayoglu, I.V.; Yildiz, Y.; et al. Do the derived neutrophil to lymphocyte ratio and the neutrophil to lymphocyte ratio predict prognosis in breast cancer? Int. J. Clin. Oncol. 2015, 20, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Shen, G.; Zhou, X.; Li, J. Therapeutic potential of tumor-associated neutrophils: dual role and phenotypic plasticity. Signal Transduct. Target. Ther. 2025, 10, 178. [Google Scholar] [CrossRef] [PubMed]
- Grecian, R.; Whyte, M.K.B.; Walmsley, S.R. The role of neutrophils in cancer. Br. Med. Bull. 2018, 128, 5–14. [Google Scholar] [CrossRef]
- SenGupta, S.; Hein, L.E.; Parent, C.A. The Recruitment of Neutrophils to the Tumor Microenvironment Is Regulated by Multiple Mediators. Front. Immunol. 2021, 12, 734188. [Google Scholar] [CrossRef]
- Shaul, M.E.; Eyal, O.; Guglietta, S.; Aloni, P.; Zlotnik, A.; Forkosh, E.; Levy, L.; Weber, L.M.; Levin, Y.; Pomerantz, A.; et al. Circulating neutrophil subsets in advanced lung cancer patients exhibit unique immune signature and relate to prognosis. FASEB J. 2020, 34, 4204–4218. [Google Scholar] [CrossRef]
- Arasanz, H.; Bocanegra, A.I.; Morilla, I.; Fernández-Irigoyen, J.; Martínez-Aguillo, M.; Teijeira, L.; Garnica, M.; Blanco, E.; Chocarro, L.; Ausin, K.; et al. Circulating Low Density Neutrophils Are Associated with Resistance to First Line Anti-PD1/PDL1 Immunotherapy in Non-Small Cell Lung Cancer. Cancers 2022, 14, 3846. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, R.; Lo Russo, G.; Signorelli, D.; Proto, C.; Prelaj, A.; Galli, G.; De Toma, A.; Viscardi, G.; Lobefaro, R.; Brambilla, M.; et al. Circulating and tumor-associated neutrophil subtypes discriminate hyperprogressive disease (HPD) from conventional progression (PD) upon immune checkpoint inhibitors (ICI) in advanced non-small cell lung cancer (NSCLC) patients (pts) and in vivo models. J. Clin. Oncol. 2020, 38, 9547–9547. [Google Scholar] [CrossRef]
- Arrazubi, V.; Goñi, S.; González-Borja, I.; Hernandez-Garcia, I.; Arasanz, H.; Pérez-Sanz, J.; Bocanegra, A.I.; Kochan, G.; Escors, D.; Ruiz de Azúa, Y.; et al. Circulating low density neutrophils are associated with resistance to anti-PD1 immunotherapy in squamous head and neck cancer. Head. Neck 2023, 45, 3075–3085. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, R.; Zhang, D.; Chen, R.; Ren, Z.; Zhang, L. Circulating Neutrophils Predict Poor Survival for HCC and Promote HCC Progression Through p53 and STAT3 Signaling Pathway. J. Cancer 2020, 11, 3736–3744. [Google Scholar] [CrossRef]
- Fernandes, P.C.; Garcia, C.B.; Micheli, D.C.; Cunha, F.Q.; Murta, E.F.C.; Tavares-Murta, B.M. Circulating neutrophils may play a role in the host response in cervical cancer. Int. J. Gynecol. Cancer 2007, 17, 1068–1074. [Google Scholar] [CrossRef] [PubMed]
- Cerezo-Wallis, D.; Ballesteros, I. Neutrophils in cancer, a love–hate affair. FEBS J. 2022, 289, 3692–3703. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Ikenaga, N.; Nakata, K.; Higashijima, N.; Zhong, P.; Kubo, A.; Wu, C.; Tsutsumi, C.; Shimada, Y.; Hayashi, M.; et al. Tumor-associated neutrophils upregulate Nectin2 expression, creating the immunosuppressive microenvironment in pancreatic ductal adenocarcinoma. J. Exp. Clin. Cancer Res. 2024, 43, 258. [Google Scholar] [CrossRef]
- Wang, Z.; Hu, H.; Bao, Y.; Pang, L.; Yang, C. Neutrophils in cancer: from immune defense to tumor promotion. Cancer Biol. Med. 2025, 1–20. [Google Scholar] [CrossRef]
- Obeagu, E.I.; Ezeala, C.C. Neutrophils as key regulators of tumor microenvironment in breast cancer: a focus on N1 and N2 polarization. Ann. Med. Surg. 2025, 87, 3509–3522. [Google Scholar] [CrossRef]
- Toghraie, F.S.; Bayat, M.; Hosseini, M.S.; Ramezani, A. Tumor-infiltrating myeloid cells; mechanisms, functional significance, and targeting in cancer therapy. Cell Oncol. 2025, 48, 559–590. [Google Scholar] [CrossRef]
- Shaul, M.E.; Levy, L.; Sun, J.; Mishalian, I.; Singhal, S.; Kapoor, V.; Horng, W.; Fridlender, G.; Albelda, S.M.; Fridlender, Z.G. Tumor-associated neutrophils display a distinct N1 profile following TGFβ modulation: A transcriptomics analysis of pro- vs. antitumor TANs. Oncoimmunology 2016, 5, e1232221. [Google Scholar] [CrossRef]
- Fang, Q.; Stehr, A.M.; Naschberger, E.; Knopf, J.; Herrmann, M.; Stürzl, M. No NETs no TIME: Crosstalk between neutrophil extracellular traps and the tumor immune microenvironment. Front. Immunol. 2022, 13, 1075260. [Google Scholar] [CrossRef] [PubMed]
- Cools-Lartigue, J.; Spicer, J.; McDonald, B.; Gowing, S.; Chow, S.; Giannias, B.; Bourdeau, F.; Kubes, P.; Ferri, L. Neutrophil extracellular traps sequester circulating tumor cells and promote metastasis. J. Clin. Invest. 2013, 123, 3446–3458. [Google Scholar] [CrossRef]
- Koenderman, L.; Vrisekoop, N. Neutrophils in cancer: from biology to therapy. Cell Mol. Immunol. 2024, 22, 4–23. [Google Scholar] [CrossRef]
- Howard, R.; Kanetsky, P.A.; Egan, K.M. Exploring the prognostic value of the neutrophil-to-lymphocyte ratio in cancer. Sci. Rep. 2019, 9, 19673. [Google Scholar] [CrossRef]
- Heshmat-Ghahdarijani, K.; Sarmadi, V.; Heidari, A.; Falahati Marvasti, A.; Neshat, S.; Raeisi, S. The neutrophil-to-lymphocyte ratio as a new prognostic factor in cancers: a narrative review. Front. Oncol. 2023, 13, 1228076. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.-H.; Tuo, M.; Ye, C.; Wu, X.-F.; Wang, H.-H.; Ren, W.-Z.; Liu, G.; Xiang, T. Prognostic value of neutrophil-to-lymphocyte ratio in gastric cancer patients undergoing neoadjuvant chemotherapy: A systematic review and meta-analysis. World J. Gastrointest. Oncol. 2024, 16, 4477–4488. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Wang, J.; Wang, X.; Gu, L.; Pei, H.; Kuai, S.; Zhang, Y.; Shang, Z. Prognostic value of the neutrophil to lymphocyte ratio in lung cancer: A meta-analysis. Clinics 2015, 70, 524–530. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, X.; Xu, R.; Luo, G.; Wang, X. Neutrophil-to-lymphocyte ratio as a prognostic factor in patients with castration-resistant prostate cancer treated with docetaxel-based chemotherapy: a meta-analysis. BMC Urol. 2025, 25, 17. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Liang, Y.; Tang, X.; Qu, H. Decrease in Blood Neutrophil-to-Lymphocyte Ratio Indicates Better Survival After Neoadjuvant Chemotherapy in Patients With Advanced Gastric Cancer. Front. Surg. 2021, 8, 745748. [Google Scholar] [CrossRef]
- Liu, S.; Wu, W.; Du, Y.; Yin, H.; Chen, Q.; Yu, W.; Wang, W.; Yu, J.; Liu, L.; Lou, W.; et al. The evolution and heterogeneity of neutrophils in cancers: origins, subsets, functions, orchestrations and clinical applications. Mol. Cancer 2023, 22, 148. [Google Scholar] [CrossRef]
- Hacbarth, E.; Kajdacsy-Balla, A. Low density neutrophils in patients with systemic lupus erythematosus, rheumatoid arthritis, and acute rheumatic fever. Arthritis Rheum. 1986, 29, 1334–1342. [Google Scholar] [CrossRef] [PubMed]
- Rankin, A.N.; Hendrix, S.V.; Naik, S.K.; Stallings, C.L. Exploring the Role of Low-Density Neutrophils During Mycobacterium tuberculosis Infection. Front. Cell Infect. Microbiol. 2022, 12. [Google Scholar] [CrossRef]
- Jaillon, S.; Ponzetta, A.; Di Mitri, D.; Santoni, A.; Bonecchi, R.; Mantovani, A. Neutrophil diversity and plasticity in tumour progression and therapy. Nat. Rev. Cancer 2020, 20, 485–503. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hu, Y.; Gu, F.; Liang, J.; Zeng, Y.; Hong, X.; Zhang, K.; Liu, L. Phenotypic and clinical characterization of low density neutrophils in patients with advanced lung adenocarcinoma. Oncotarget 2017, 8, 90969–90978. [Google Scholar] [CrossRef]
- Hsu, B.E.; Tabariès, S.; Johnson, R.M.; Andrzejewski, S.; Senecal, J.; Lehuédé, C.; Annis, M.G.; Ma, E.H.; Völs, S.; Ramsay, L.; et al. Immature Low-Density Neutrophils Exhibit Metabolic Flexibility that Facilitates Breast Cancer Liver Metastasis. Cell Rep. 2019, 27, 3902–3915.e6. [Google Scholar] [CrossRef]
- Futoh, Y.; Kumagai, Y.; Miyato, H.; Ozawa, H.; Kanamaru, R.; Sadatomo, A.; Ohnishi, Y.; Koinuma, K.; Horie, H.; Yamaguchi, H.; et al. Peripheral low-density granulocytes after colorectal cancer surgery in predicting recurrence. BJS Open 2023, 7. [Google Scholar] [CrossRef]
- Krieg, C.; Shaul, M.; Guglietta, S.; Weber, L.; Robinson, M.; Fridlender, Z. Circulating Low Density Neutrophils In Advanced Lung Cancer Patients Exhibit Unique Immune Signatures and affect prognosis. J. Immunol. 2020, 204, 164.11–164.11. [Google Scholar] [CrossRef]
- Fu, Y.; Wen, Z.; Fan, J. Interaction of low-density neutrophils with other immune cells in the mechanism of inflammation. Mol. Med. 2025, 31, 133. [Google Scholar] [CrossRef]
- Wang, X.; Qiu, L.; Li, Z.; Wang, X.-Y.; Yi, H. Understanding the Multifaceted Role of Neutrophils in Cancer and Autoimmune Diseases. Front Immunol 2018, 9, 2456. [Google Scholar] [CrossRef]
- Hassani, M.; Hellebrekers, P.; Chen, N.; van Aalst, C.; Bongers, S.; Hietbrink, F.; Koenderman, L.; Vrisekoop, N. On the origin of low-density neutrophils. J. Leukoc. Biol. 2020, 107, 809–818. [Google Scholar] [CrossRef]
- Ng, M.S.F.; Kwok, I.; Tan, L.; Shi, C.; Cerezo-Wallis, D.; Tan, Y.; Leong, K.; Calvo, G.F.; Yang, K.; Zhang, Y.; et al. Deterministic reprogramming of neutrophils within tumors. Science (80- ) 2024, 383. [Google Scholar] [CrossRef]
- Giese, M.A.; Hind, L.E.; Huttenlocher, A. Neutrophil plasticity in the tumor microenvironment. Blood 2019, 133, 2159–2167. [Google Scholar] [CrossRef]
- Brandau, S.; Moses, K.; Lang, S. The kinship of neutrophils and granulocytic myeloid-derived suppressor cells in cancer: Cousins, siblings or twins? Semin. Cancer Biol. 2013, 23, 171–182. [Google Scholar] [CrossRef]
- Curi, R.; Levada-Pires, A.C.; Silva, E.B.D.; Poma, S.O.; Zambonatto, R.F.; Domenech, P.; Almeida, M.M.; Gritte, R.B.; Souza-Siqueira, T.; Gorjão, R.; et al. The Critical Role of Cell Metabolism for Essential Neutrophil Functions. Cell Physiol. Biochem. 2020, 54, 629–647. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Saxena, S.; Goel, P.; Prajapati, D.R.; Wang, C.; Singh, R.K. Breast Cancer Cell–Neutrophil Interactions Enhance Neutrophil Survival and Pro-Tumorigenic Activities. Cancers 2020, 12, 2884. [Google Scholar] [CrossRef]
- Ettel, P.; Weichhart, T. Not just sugar: metabolic control of neutrophil development and effector functions. J. Leukoc. Biol. 2024, 116, 487–510. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Lu, M.; Shi, J.; Gong, Z.; Hua, L.; Li, Q.; Lim, B.; Zhang, X.H.-F.; Chen, X.; Li, S.; et al. Lung mesenchymal cells elicit lipid storage in neutrophils that fuel breast cancer lung metastasis. Nat. Immunol. 2020, 21, 1444–1455. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Shi, J.; Shen, J.; Fan, X. Metabolic reprogramming of neutrophils in the tumor microenvironment: Emerging therapeutic targets. Cancer Lett. 2025, 612, 217466. [Google Scholar] [CrossRef]
- Rice, C.M.; Davies, L.C.; Subleski, J.J.; Maio, N.; Gonzalez-Cotto, M.; Andrews, C.; Patel, N.L.; Palmieri, E.M.; Weiss, J.M.; Lee, J.-M.; et al. Tumour-elicited neutrophils engage mitochondrial metabolism to circumvent nutrient limitations and maintain immune suppression. Nat. Commun. 2018, 9, 5099. [Google Scholar] [CrossRef] [PubMed]
- Bodac, A.; Meylan, E. Neutrophil metabolism in the cancer context. Semin. Immunol. 2021, 57, 101583. [Google Scholar] [CrossRef]
- Arpinati, L.; Kaisar-Iluz, N.; Shaul, M.E.; Groth, C.; Umansky, V.; Fridlender, Z.G. Tumor-Derived Factors Differentially Affect the Recruitment and Plasticity of Neutrophils. Cancers 2021, 13, 5082. [Google Scholar] [CrossRef]
- Wang, X.; Qu, Y.; Xu, Q.; Jiang, Z.; Wang, H.; Lin, B.; Cao, Z.; Pan, Y.; Li, S.; Hu, Y.; et al. NQO1 Triggers Neutrophil Recruitment and NET Formation to Drive Lung Metastasis of Invasive Breast Cancer. Cancer Res. 2024, 84, 3538–3555. [Google Scholar] [CrossRef]
- Lecot, P.; Ardin, M.; Dussurgey, S.; Alcazer, V.; Moudombi, L.; Pereira Abrantes, M.; Hubert, M.; Swalduz, A.; Hernandez-Vargas, H.; Viari, A.; et al. Gene signature of circulating platelet-bound neutrophils is associated with poor prognosis in cancer patients. Int. J. Cancer 2022, 151, 138–152. [Google Scholar] [CrossRef]
- Tay, S.H.; Zharkova, O.; Lee, H.Y.; Toh, M.M.X.; Libau, E.A.; Celhar, T.; Narayanan, S.; Ahl, P.J.; Ong, W.Y.; Joseph, C.; et al. Platelet TLR7 is essential for the formation of platelet–neutrophil complexes and low-density neutrophils in lupus nephritis. Rheumatology 2024, 63, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Hsu, B.E.; Roy, J.; Mouhanna, J.; Rayes, R.F.; Ramsay, L.; Tabariès, S.; Annis, M.G.; Watson, I.R.; Spicer, J.D.; Costantino, S.; et al. C3a elicits unique migratory responses in immature low-density neutrophils. Oncogene 2020, 39, 2612–2623. [Google Scholar] [CrossRef] [PubMed]
- Mezquita, L.; Preeshagul, I.; Auclin, E.; Saravia, D.; Hendriks, L.; Rizvi, H.; Park, W.; Nadal, E.; Martin-Romano, P.; Ruffinelli, J.C.; et al. Predicting immunotherapy outcomes under therapy in patients with advanced NSCLC using dNLR and its early dynamics. Eur. J. Cancer 2021, 151, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Carrión-Barberà, I.; Lood, C. Performance of the neutrophil-to-lymphocyte ratio as a prognostic tool for survival in solid cancers. Front. Oncol. 2025, 15, 1616477. [Google Scholar] [CrossRef]
- Jadoon, S.K.; Soomro, R.; Ahsan, M.N.; Ijaz Khan, R.M.; Iqbal, S.; Yasmin, F.; Najeeb, H.; Saleem, N.; Cho, N.; Resham Shaikh, T.G.; et al. Association of neutrophil-to-lymphocyte ratio with clinical, pathological, radiological, laboratory features and disease outcomes of invasive breast cancer patients: A retrospective observational cohort study. Medicine 2023, 102, e33811. [Google Scholar] [CrossRef] [PubMed]
- Obeagu, E.I.; Obeagu, G.U. Exploring neutrophil functionality in breast cancer progression: A review. Medicine 2024, 103, e37654. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Shen, Y.; Huang, H.; Pan, S.; Jiang, J.; Chen, W.; Zhang, T.; Zhang, C.; Ni, C. A Rosetta Stone for Breast Cancer: Prognostic Value and Dynamic Regulation of Neutrophil in Tumor Microenvironment. Front. Immunol. 2020, 11, 1779. [Google Scholar] [CrossRef]
- Chen, J.; Deng, Q.; Pan, Y.; He, B.; Ying, H.; Sun, H.; Liu, X.; Wang, S. Prognostic value of neutrophil-to-lymphocyte ratio in breast cancer. FEBS Open Bio 2015, 5, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Garner, H.; Martinovic, M.; Liu, N.Q.; Bakker, N.A.M.; Velilla, I.Q.; Hau, C.-S.; Vrijland, K.; Kaldenbach, D.; Kok, M.; de Wit, E.; et al. Understanding and reversing mammary tumor-driven reprogramming of myelopoiesis to reduce metastatic spread. Cancer Cell 2025, 43, 1279–1295.e9. [Google Scholar] [CrossRef]
- Gianni, C.; Palleschi, M.; Schepisi, G.; Casadei, C.; Bleve, S.; Merloni, F.; Sirico, M.; Sarti, S.; Cecconetto, L.; Di Menna, G.; et al. Circulating inflammatory cells in patients with metastatic breast cancer: Implications for treatment. Front. Oncol. 2022, 12, 882896. [Google Scholar] [CrossRef]
- Bakker, N.A.M.; Garner, H.; van Dyk, E.; Champanhet, E.; Klaver, C.; Duijst, M.; Voorwerk, L.; Nederlof, I.; Voorthuis, R.; Liefaard, M.C.; et al. Triple-negative breast cancer modifies the systemic immune landscape and alters neutrophil functionality. npj Breast Cancer 2025, 11, 5. [Google Scholar] [CrossRef]
- Ramessur, A.; Ambasager, B.; Valle Aramburu, I.; Peakman, F.; Gleason, K.; Lehmann, C.; Papayannopoulos, V.; Coombes, R.C.; Malanchi, I. Circulating neutrophils from patients with early breast cancer have distinct subtype-dependent phenotypes. Breast Cancer Res. 2023, 25, 125. [Google Scholar] [CrossRef]
- Unanua, N.C.; Fontecha, O.; Sanchez, L.T.; Martínez-Aguillo, M.; Ruiz, I.M.; Labiano, I.; Huerta-Hernández, A.E.; Lecumberri, A.; Caseda, I.; Maqueda, M.A.; et al. 6P Circulating low-density neutrophils (LDNs) are associated with resistance to immunotherapy as frontline treatment for non-small cell lung cancer (NSCLC): Updated results and proteomic characterization. Ann. Oncol. 2024, 35, S217. [Google Scholar] [CrossRef]
- Ohashi, K.; Nishito, Y.; Fukuda, H.; Sadahiro, R.; Yoshida, Y.; Watanabe, S.; Motoi, N.; Sonobe, Y.; Mizuno, H.; Tsunoda, H.; et al. Neutrophil-to-lymphocyte ratio is a prognostic factor reflecting immune condition of tumor microenvironment in squamous cell lung cancer. Sci. Rep. 2024, 14, 429. [Google Scholar] [CrossRef]
- Zhang, C.; Huo, Y.; Shang, X.; Zhang, T.; Tang, N.; Wang, H. Tumor mutational burden adjusted by neutrophil-to-lymphocyte ratio serves as a potential biomarker for atezolizumab-treated patients with extensive stage small cell lung cancer. Respir. Res. 2024, 25, 253. [Google Scholar] [CrossRef]
- Mariean, C.R.; Tiucă, O.M.; Mariean, A.; Szekely, T.-B.; Niculescu, R.; Sabau, A.H.; Al-Akel, C.F.; Cotoi, O.S. The Impact of the Histologic Types of Lung Cancer on CBC-Derived Inflammatory Markers—Current Knowledge and Future Perspectives. J. Clin. Med. 2025, 14, 3038. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, H.; Jiang, S.; Wang, W. Role of tumor-associated neutrophils in lung cancer (Review). Oncol. Lett. 2022, 25, 2. [Google Scholar] [CrossRef]
- Yamazaki, N.; Kiyohara, Y.; Uhara, H.; Iizuka, H.; Uehara, J.; Otsuka, F.; Fujisawa, Y.; Takenouchi, T.; Isei, T.; Iwatsuki, K.; et al. Cytokine biomarkers to predict antitumor responses to nivolumab suggested in a phase 2 study for advanced melanoma. Cancer Sci. 2017, 108, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- Laino, A.S.; Woods, D.; Vassallo, M.; Qian, X.; Tang, H.; Wind-Rotolo, M.; Weber, J. Serum interleukin-6 and C-reactive protein are associated with survival in melanoma patients receiving immune checkpoint inhibition. J. Immunother. Cancer 2020, 8, e000842. [Google Scholar] [CrossRef] [PubMed]
- Tazzyman, S.; Barry, S.T.; Ashton, S.; Wood, P.; Blakey, D.; Lewis, C.E.; Murdoch, C. Inhibition of neutrophil infiltration into A549 lung tumors in vitro and in vivo using a CXCR2-specific antagonist is associated with reduced tumor growth. Int. J. Cancer 2011, 129, 847–858. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, M.H.; Rayes, R.F.; Cools-Lartigue, J.; Spicer, J.D. Neutrophil extracellular traps in cancer. Nat. Rev. Cancer 2025. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; He, Y.; Liang, L. Dysregulated arginine metabolism is associated with pro-tumor neutrophil polarization in liver cancer. Front Immunol 2025, 16, 1673665. [Google Scholar] [CrossRef]
- Lynch, C.; Pitroda, S.P.; Weichselbaum, R.R. Radiotherapy, immunity, and immune checkpoint inhibitors. Lancet Oncol. 2024, 25, e352–e362. [Google Scholar] [CrossRef]
- Principe, D.R.; Kamath, S.D.; Korc, M.; Munshi, H.G. The immune modifying effects of chemotherapy and advances in chemo-immunotherapy. Pharmacol. Ther. 2022, 236, 108111. [Google Scholar] [CrossRef]
- Ning, X.; Wang, W.-M.; Jin, H.-Z. Low-Density Granulocytes in Immune-Mediated Inflammatory Diseases. J. Immunol. Res. 2022, 2022, 1–11. [Google Scholar] [CrossRef]
- Carmona-Rivera, C.; Khaznadar, S.S.; Shwin, K.W.; Irizarry-Caro, J.A.; O’Neil, L.J.; Liu, Y.; Jacobson, K.A.; Ombrello, A.K.; Stone, D.L.; Tsai, W.L.; et al. Deficiency of adenosine deaminase 2 triggers adenosine-mediated NETosis and TNF production in patients with DADA2. Blood 2019, 134, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.W.; Houghton, A.M. Targeting neutrophils for cancer therapy. Nat. Rev. Drug Discov. 2025, 24, 666–684. [Google Scholar] [CrossRef]
- Eruslanov, E.; Nefedova, Y.; Gabrilovich, D.I. The heterogeneity of neutrophils in cancer and its implication for therapeutic targeting. Nat. Immunol. 2025, 26, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Chen, X.; Lu, Y.; Sun, N.; Weisgerber, K.E.; Xu, M.; Bai, R.-Y. Neutrophil Dynamics in Response to Cancer Therapies. Cancers 2025, 17, 2593. [Google Scholar] [CrossRef]
- Koh, C.-H.; Bhoo-Pathy, N.; Ng, K.-L.; Jabir, R.S.; Tan, G.-H.; See, M.-H.; Jamaris, S.; Taib, N.A. Utility of pre-treatment neutrophil–lymphocyte ratio and platelet–lymphocyte ratio as prognostic factors in breast cancer. Br. J. Cancer 2015, 113, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Wu, J.; Jia, H.; Yang, Y.; Zhang, X.; Chen, K.; Su, F. The Peripheral Blood Neutrophil-To-Lymphocyte Ratio Is Superior to the Lymphocyte-To-Monocyte Ratio for Predicting the Long-Term Survival of Triple-Negative Breast Cancer Patients. PLoS One 2015, 10, e0143061. [Google Scholar] [CrossRef]
- Kim, I.S.; Gao, Y.; Welte, T.; Wang, H.; Liu, J.; Janghorban, M.; Sheng, K.; Niu, Y.; Goldstein, A.; Zhao, N.; et al. Immuno-subtyping of breast cancer reveals distinct myeloid cell profiles and immunotherapy resistance mechanisms. Nat. Cell Biol. 2019, 21, 1113–1126. [Google Scholar] [CrossRef]
| Cancer Type | Authors, Journal; DOI | Design | Population | Material, Timing | Key Findings |
|---|---|---|---|---|---|
| Breast cancer | Saraiva et al., Oncotarget 2021; [12] doi:10.18632/oncotarget.28135 | Prospective observational | 48 non-metastatic, 12 metastatic;7 healthy donors | Peripheral blood; baseline; fresh biopsies and surgical specimens | LDNs↑ → poor prognosis; ↓CT response; T cell suppression |
| Metastatic breast cancer | Correia et al., Breast Cancer Res. 2025; [17] doi:10.1186/s13058-025-02201-8 | Prospective observational | Patients with metastatic breast cancer | peripheral blood; tissue biopsies 2023-2025 | Neutrophils are associated with tumor progression, increased metastasis, and worse prognosis |
| Breast cancer | Koh et al., British Journal of Cancer 2015; [126] doi:10.1038/bjc.2015.183 | Prospective observational | 1435 patients (stage I-III) | Peripheral blood pre-treatment | High NLR (quintile 5) → worse OS; independent prognostic factor; 5-year survival 51.1% vs 76.4% |
| Breast cancer (TNBC focus) | Jia et al., PLoS One 2015; [127] doi:10.1371/journal.pone.0143061 |
Retrospective cohort | 1570 operable breast cancer patients | Peripheral blood baseline | NLR superior to LMR for prognosis; high NLR → poor DFS/OS in TNBC (HR=2.58 for DFS, HR=3.05 for OS) |
| Breast cancer | Hsu et al., Cell Reports 2019; [78] doi:10.1016/j.celrep.2019.05.091 | Mechanistic (preclinical) | Mouse models of breast cancer liver metastasis | Blood and tissue samples from tumor-bearing mice | iLDNs exhibit metabolic flexibility; promote liver metastasis via NETosis; G-CSF mobilizes iLDNs; HDNs inhibit metastasis |
| Breast cancer | Sagiv et al., Cell Reports 2015; [13] doi:10.1016/j.celrep.2014.12.039 |
Mechanistic (preclinical+clinical samples) | Mouse models + cancer patients | Peripheral blood; density gradient separation | LDNs heterogeneous (immature MDSCs + mature cells); TGF-β-dependent conversion from HDNs; immunosuppressive properties |
| Breast cancer (TNBC) | Kim et al., Nature Cell Biology 2019; [128] doi:10.1038/s41556-019-0373-7 |
Mechanistic (preclinical+clinical datasets) | Multiple murine TNBC models + clinical datasets | Tumor tissue and blood | Neutrophil-enriched subtype resistant to ICI; systemic accumulation of immunosuppressive neutrophils; MES-to-NES conversion mediates acquired ICI resistance |
| Metastatic breast cancer | Bakker et al., NPJ Breast Cancer 2025; [107] doi:10.1038/s41523-025-00721-2 |
Prospective translational | TNBC patients | Peripheral blood; functional assays | Neutrophils showed increased migration, granule protein expression, ROS production, and NETs formation, consistent with pro-tumor LDNs dysfunction |
| Breast cancer | Garner et al., Cancer Cell 2025; [105] doi:10.1016/j.ccell.2025.04.007 |
Preclinical + translational | Mice + human samples | Bone marrow + peripheral blood | Tumor-driven myelopoiesis → dysfunctional LDNs-like and promotes metastasis. Anti-IL-1β reverses these changes and reduces metastatic spread |
| NSCLC | Arasanz et al., Cancers 2022; [53] doi:10.3390/cancers14163846 | Prospective observational | Advanced NSCLC patients (first-line anti-PD1/PDL1) | Peripheral blood; baseline and during treatment | Elevated baseline LDNs predict primary resistance to ICI monotherapy; LDNs mediate humoral immunosuppression |
| Advanced lung cancer | Shaul et al., FASEB Journal 2020; [52] doi:10.1096/fj.201902467R |
Prospective observational + mechanistic |
64 advanced, 35 early-stage patients, 15 healthy, 13 COPD | Peripheral blood; CyTOF analysis | LDNs highly enriched in advanced disease (median 7.0%); elevated LDNs (>10%) → poor prognosis; unique immune signatures (CD66b⁺/CD10⁻/CXCR4⁺/PDL1⁺ subset) |
|
Lung adenocarcinoma |
Liu et al., Oncotarget 2017; [77] doi:10.18632/oncotarget.18771 |
Prospective observational | 52 advanced lung adenocarcinoma patients, 13 healthy controls | Peripheral blood; flow cytometry | LDNs/HDNs ratio ↑ in patients; ratio correlates with disease progression; positive correlation with CD8⁺ T cells; decreased with treatment |
|
NSCLC |
Mezquita et al., European Journal of Cancer 2021; [99] doi:10.1016/j.ejca.2021.03.011 |
Multicenter prospective | NSCLC patients: 1485 ICI; 173 CT | Peripheral blood; baseline and cycle 2 | Persistently high NLR → poor survival (OS=5 months); immature neutrophils correlate with poor PFS/OS; 12-week death rate 49% |
| NSCLC | Valadez-Cosmes et al., Frontiers in Immunology 2021; [18] doi:10.3389/fimmu.2021.703846 |
Mechanistic (clinical samples) | 26 NSCLC patients, 14 healthy | Peripheral blood; high-dimensional flow cytometry screening | NSCLC: LDNs highly enriched (median 20.4%); novel markers identified: CD36↑, CD41↑, CD61↑, CD226↑; 12 markers downregulated, 41 upregulated |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




