Submitted:
06 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
2.1. Study Subjects
2.2. Genotyping, Imputation, and Quality Controls
2.3. Genome-Wide Genetic Score Construction and Analysis
2.4. Statistical Analysis
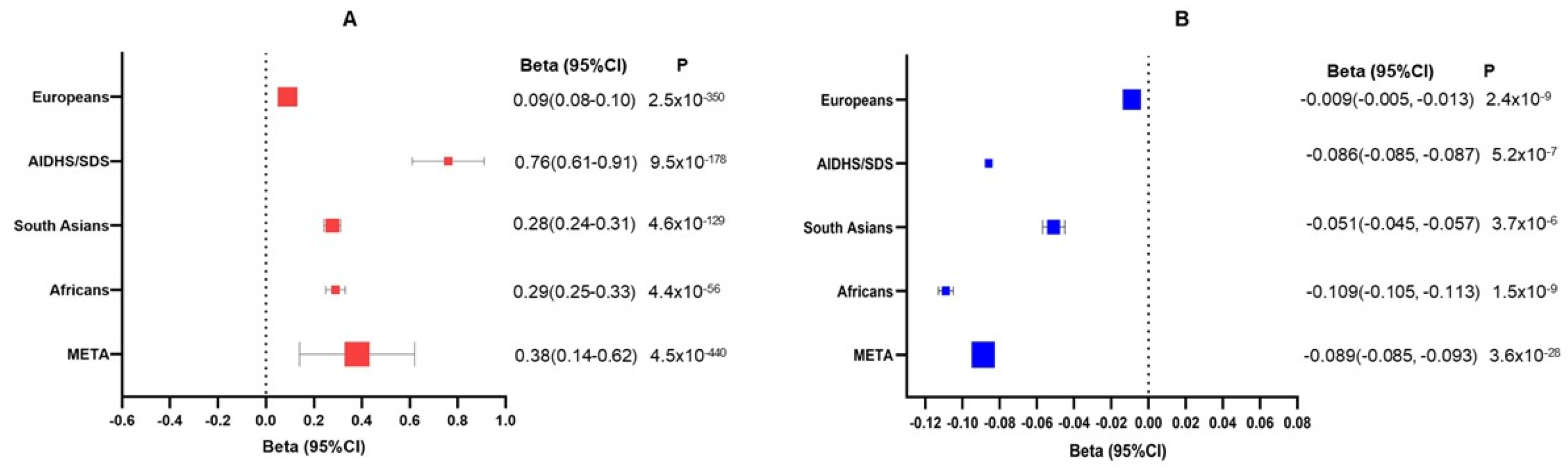
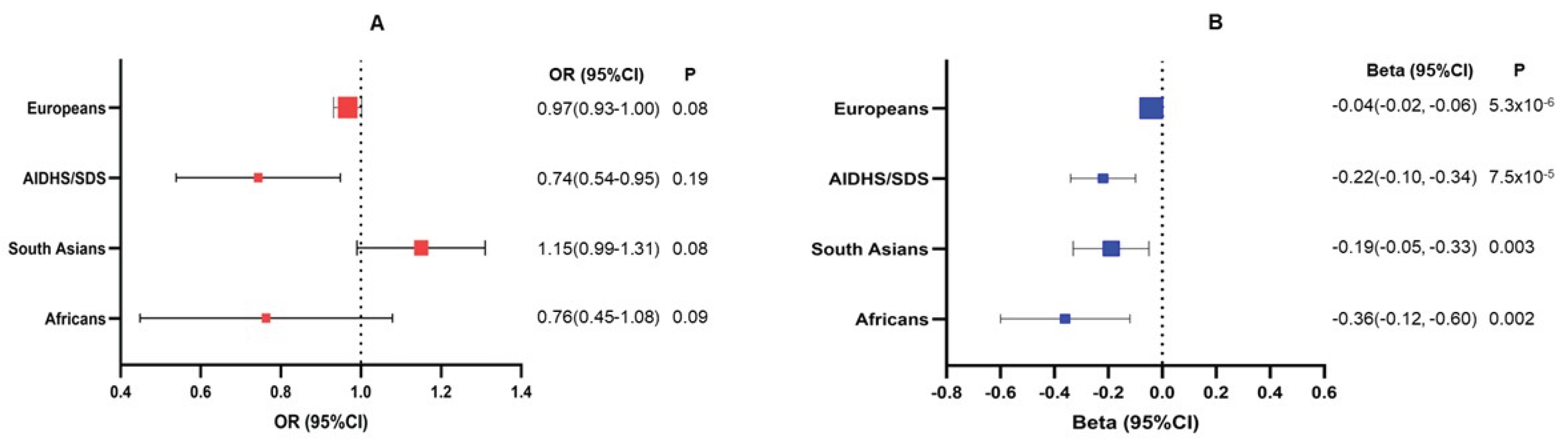
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
- AIS: Acute Ischemic Stroke
- AIDHS/SDS: Asian Indian Diabetic Heart Study/Sikh Diabetes Study
- AF: Africans
- BG: Blood glucose
- BMI: Body mass index
- BP: Blood pressure
- CABG: Coronary artery bypass graft
- CAD: Coronary artery disease
- CVD : Cardiovascular disease
- DBP: Diastolic blood pressure
- EU: European
- GSA: Global Screening Arrays
- GWAS: Genome-wide association studies
- HDL-C: High-density lipoprotein cholesterol
- HWE: Hardy-Weinberg equilibrium
- LD: Linkage disequilibrium
- LDL-C: Low-density lipoprotein cholesterol
- MAF: Minor allele frequency
- MR: Mendelian randomization
- PC: Principal components
- PGS: Polygenic score
- PRS: Polygenic risk score
- SA: South Asians
- SBP: Systolic blood pressure
- T2D: Type 2 diabetes
- TC: Total cholesterol
- TG: Triglycerides
- UKBB: UK Biobank
- Vitamin D: 25(OH)D
- WHR: Waist-to-hip ratio
References
- Holick, M.F.; Chen, T.C. Vitamin D deficiency: a worldwide problem with health consequences. Am J Clin Nutr 2008, 87, 1080S–1086S. [Google Scholar] [CrossRef] [PubMed]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Kostenberger, M.; Tmava Berisha, A.; et al. Vitamin D deficiency 2.0: an update on the current status worldwide. Eur J Clin Nutr 2020, 74, 1498–1513. [Google Scholar] [CrossRef] [PubMed]
- Revez, J.A.; Lin, T.; Qiao, Z.; Xue, A.; Holtz, Y.; Zhu, Z.; et al. Genome-wide association study identifies 143 loci associated with 25 hydroxyvitamin D concentration. Nat Commun 2020, 11, 1647. [Google Scholar] [CrossRef]
- Bouillon, R.; Lips, P.; Bilezikian, J.P. Vitamin D supplementation and musculoskeletal health. Lancet Diabetes Endocrinol 2019, 7, 85–86. [Google Scholar] [CrossRef]
- Davis, C.D. Vitamin D and cancer: current dilemmas and future research needs. Am J Clin Nutr 2008, 88, 565S–569S. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Staten, M.A.; Knowler, W.C.; Nelson, J.; Vickery, E.M.; LeBlanc, E.S.; et al. Intratrial Exposure to Vitamin D and New-Onset Diabetes Among Adults With Prediabetes: A Secondary Analysis From the Vitamin D and Type 2 Diabetes (D2d) Study. Diabetes Care 2020, 43, 2916–2922. [Google Scholar] [CrossRef]
- Ramagopalan, S.V.; Heger, A.; Berlanga, A.J.; Maugeri, N.J.; Lincoln, M.R.; Burrell, A.; et al. A ChIP-seq defined genome-wide map of vitamin D receptor binding: associations with disease and evolution. Genome Res 2010, 20, 1352–1360. [Google Scholar] [CrossRef]
- Afzal, S.; Brondum-Jacobsen, P.; Bojesen, S.E.; Nordestgaard, B.G. Vitamin D concentration, obesity, and risk of diabetes: a mendelian randomisation study. Lancet Diabetes Endocrinol 2014, 2, 298–306. [Google Scholar] [CrossRef]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: a systematic review. Lancet Diabetes Endocrinol 2014, 2, 76–89. [Google Scholar] [CrossRef] [PubMed]
- Braun, T.R.; Been, L.F.; Blackett, P.R.; Sanghera, D.K. Vitamin D Deficiency and Cardio-Metabolic Risk in a North Indian Community with Highly Prevalent Type 2 Diabetes. J Diabetes Metab 2012, 3. [Google Scholar] [CrossRef]
- Gagnon, C.; Lu, Z.X.; Magliano, D.J.; Dunstan, D.W.; Shaw, J.E.; Zimmet, P.Z.; et al. Serum 25-hydroxyvitamin D, calcium intake, and risk of type 2 diabetes after 5 years: results from a national, population-based prospective study (the Australian Diabetes, Obesity and Lifestyle study). Diabetes Care 2011, 34, 1133–1138. [Google Scholar] [CrossRef] [PubMed]
- Scragg, R.; Sowers, M.; Bell, C.; Third National, H.; Nutrition Examination, S. Serum 25-hydroxyvitamin D, diabetes, and ethnicity in the Third National Health and Nutrition Examination Survey. Diabetes Care 2004, 27, 2813–2818. [Google Scholar] [CrossRef]
- Theodoratou, E.; Tzoulaki, I.; Zgaga, L.; Ioannidis, J.P. Vitamin D and multiple health outcomes: umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ 2014, 348, g2035. [Google Scholar] [CrossRef]
- Pittas, A.G.; Dawson-Hughes, B.; Sheehan, P.; Ware, J.H.; Knowler, W.C.; Aroda, V.R.; et al. Vitamin D Supplementation and Prevention of Type 2 Diabetes. N Engl J Med 2019, 381, 520–530. [Google Scholar] [CrossRef]
- Tobias, D.K.; Pradhan, A.D.; Duran, E.K.; Li, C.; Song, Y.; Buring, J.E.; et al. Vitamin D supplementation vs. placebo and incident type 2 diabetes in an ancillary study of the randomized Vitamin D and Omega-3 Trial. Nat Commun 2025, 16, 3332. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.; De Lange, M.; Snieder, H.; MacGregor, A.J.; Swaminathan, R.; Thakker, R.V.; et al. Genetic contribution to bone metabolism, calcium excretion, and vitamin D and parathyroid hormone regulation. J Bone Miner Res 2001, 16, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Karohl, C.; Su, S.; Kumari, M.; Tangpricha, V.; Veledar, E.; Vaccarino, V.; et al. Heritability and seasonal variability of vitamin D concentrations in male twins. Am J Clin Nutr 2010, 92, 1393–1398. [Google Scholar] [CrossRef]
- Ahn, J.; Yu, K.; Stolzenberg-Solomon, R.; Simon, K.C.; McCullough, M.L.; Gallicchio, L.; et al. Genome-wide association study of circulating vitamin D levels. Hum Mol Genet 2010, 19, 2739–2745. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; O’Reilly, P.F.; Aschard, H.; Hsu, Y.H.; Richards, J.B.; Dupuis, J.; et al. Genome-wide association study in 79,366 European-ancestry individuals informs the genetic architecture of 25-hydroxyvitamin D levels. Nat Commun 2018, 9, 260. [Google Scholar] [CrossRef]
- Wang, T.J.; Zhang, F.; Richards, J.B.; Kestenbaum, B.; van Meurs, J.B.; Berry, D.; et al. Common genetic determinants of vitamin D insufficiency: a genome-wide association study. Lancet 2010, 376, 180–188. [Google Scholar] [CrossRef]
- Becerra-Cervera, A.; Jimenez-Ortega, R.F.; Aparicio-Bautista, D.I.; Lopez-Perez, T.V.; Patino, N.; Castillejos-Lopez, M.; et al. Genetic variants in vitamin D metabolism-related genes are associated with vitamin D status and adiposity markers. Nutr Res 2025, 136, 105–119. [Google Scholar] [CrossRef]
- Sapkota, B.R.; Hopkins, R.; Bjonnes, A.; Ralhan, S.; Wander, G.S.; Mehra, N.K.; et al. Genome-wide association study of 25(OH) Vitamin D concentrations in Punjabi Sikhs: Results of the Asian Indian diabetic heart study. J Steroid Biochem Mol Biol 2016, 158, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Vasileiou, E.S.; Hu, C.; Bernstein, C.N.; Lublin, F.; Wolinsky, J.S.; Cutter, G.R.; et al. Association of Vitamin D Polygenic Risk Scores and Disease Outcome in People With Multiple Sclerosis. Neurol Neuroimmunol Neuroinflamm 2023, 10. [Google Scholar] [CrossRef]
- Liu, H.; Shen, X.; Yu, T.; Wang, Y.; Cai, S.; Jiang, X.; et al. A putative causality of vitamin D in common diseases: A mendelian randomization study. Front Nutr 2022, 9, 938356. [Google Scholar] [CrossRef]
- Ren, Y.; Liu, J.; Li, W.; Zheng, H.; Dai, H.; Qiu, G.; et al. Causal Associations between Vitamin D Levels and Psoriasis, Atopic Dermatitis, and Vitiligo: A Bidirectional Two-Sample Mendelian Randomization Analysis. Nutrients 2022, 14. [Google Scholar] [CrossRef]
- Ye, Z.; Sharp, S.J.; Burgess, S.; Scott, R.A.; Imamura, F.; InterAct, C.; et al. Association between circulating 25-hydroxyvitamin D and incident type 2 diabetes: a mendelian randomisation study. Lancet Diabetes Endocrinol 2015, 3, 35–42. [Google Scholar] [CrossRef]
- Bejar, C.A.; Goyal, S.; Afzal, S.; Mangino, M.; Zhou, A.; van der Most, P.J.; et al. A Bidirectional Mendelian Randomization Study to evaluate the causal role of reduced blood vitamin D levels with type 2 diabetes risk in South Asians and Europeans. Nutr J 2021, 20, 71. [Google Scholar] [CrossRef] [PubMed]
- Rout, M.; Tung, G.K.; Singh, J.R.; Mehra, N.K.; Wander, G.S.; Ralhan, S.; et al. Polygenic Risk Score Assessment for Coronary Artery Disease in Asian Indians. J Cardiovasc Transl Res 2024, 17, 1086–1096. [Google Scholar] [CrossRef] [PubMed]
- Rout, M.; Wander, G.S.; Ralhan, S.; Singh, J.R.; Aston, C.E.; Blackett, P.R.; et al. Assessing the prediction of type 2 diabetes risk using polygenic and clinical risk scores in South Asian study populations. Ther Adv Endocrinol Metab 2023, 14, 20420188231220120. [Google Scholar] [CrossRef]
- Lin, L.Y.; Smeeth, L.; Langan, S.; Warren-Gash, C. Distribution of vitamin D status in the UK: a cross-sectional analysis of UK Biobank. BMJ Open 2021, 11, e038503. [Google Scholar] [CrossRef]
- Sanghera, D.K.; Bhatti, J.S.; Bhatti, G.K.; Ralhan, S.K.; Wander, G.S.; Singh, J.R.; et al. The Khatri Sikh Diabetes Study (SDS): Study design, methodology, sample collection, and initial results. Hum Biol 2006, 78, 43–63. [Google Scholar] [CrossRef]
- Sanghera DK DS. Cardiovascular disease in South Asians; Risk factors, genetics and environment. Medicine Update 2016-1. New Delhi, London, Philadelphia, Panama: The Health Sciences Publishers 2016;2.
- Goyal, S.; Sanghera, D.K. Genetic and Non-genetic Determinants of Cardiovascular Disease in South Asians. Curr Diabetes Rev 2021, 17, e011721190373. [Google Scholar] [CrossRef]
- Sanghera, D.K.; Nath, S.K.; Ortega, L.; Gambarelli, M.; Kim-Howard, X.; Singh, J.R.; et al. TCF7L2 polymorphisms are associated with type 2 diabetes in Khatri Sikhs from North India: genetic variation affects lipid levels. Ann Hum Genet 2008, 72, 499–509. [Google Scholar] [CrossRef]
- Saxena, R.; Bjonnes, A.; Prescott, J.; Dib, P.; Natt, P.; Lane, J.; et al. Genome-wide association study identifies variants in casein kinase II (CSNK2A2) to be associated with leukocyte telomere length in a Punjabi Sikh diabetic cohort. Circ Cardiovasc Genet 2014, 7, 287–295. [Google Scholar] [CrossRef]
- Goyal, S.; Tanigawa, Y.; Zhang, W.H.; Chai, J.F.; Almeida, M.; Sim, X.L.; et al. APOC3 genetic variation, serum triglycerides, and risk of coronary artery disease in Asian Indians, Europeans, and other ethnic groups. Lipids in Health and Disease 2021, 20. [Google Scholar] [CrossRef] [PubMed]
- Consultation WHOE. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 2004, 363, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Schierer, A.; Been, L.F.; Ralhan, S.; Wander, G.S.; Aston, C.E.; Sanghera, D.K. Genetic variation in cholesterol ester transfer protein, serum CETP activity, and coronary artery disease risk in Asian Indian diabetic cohort. Pharmacogenet Genomics 2012, 22, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Rout, M.; Lerner, M.; Blackett, P.R.; Peyton, M.D.; Stavrakis, S.; Sidorov, E.; et al. Ethnic differences in ApoC-III concentration and the risk of cardiovascular disease: No evidence for the cardioprotective role of rare/loss of function APOC3 variants in non-Europeans. Am Heart J Plus 2022, 13. [Google Scholar] [CrossRef]
- Sapkota, B.; Subramanian, A.; Priamvada, G.; Finely, H.; Blackett, P.R.; Aston, C.E.; et al. Association of APOE polymorphisms with diabetes and cardiometabolic risk factors and the role of APOE genotypes in response to anti-diabetic therapy: results from the AIDHS/SDS on a South Asian population. J Diabetes Complications 2015, 29, 1191–1197. [Google Scholar] [CrossRef]
- Sanghera, D.K.; Bejar, C.; Sapkota, B.; Wander, G.S.; Ralhan, S. Frequencies of poor metabolizer alleles of 12 pharmacogenomic actionable genes in Punjabi Sikhs of Indian Origin. Sci Rep 2018, 8, 15742. [Google Scholar] [CrossRef]
- Sanghera, D.K.; Hopkins, R.; Malone-Perez, M.W.; Bejar, C.; Tan, C.; Mussa, H.; et al. Targeted sequencing of candidate genes of dyslipidemia in Punjabi Sikhs: Population-specific rare variants in GCKR promote ectopic fat deposition. PloS one 2019, 14, e0211661. [Google Scholar] [CrossRef] [PubMed]
- Rout, M.; Ramu, D.; Mariana, M.; Koshy, T.; Venkatesan, V.; Lopez-Alvarenga, J.C.; et al. Excess of rare noncoding variants in several type 2 diabetes candidate genes among Asian Indian families. Commun Med (Lond) 2025, 5, 47. [Google Scholar] [CrossRef] [PubMed]
- Sudlow, C.; Gallacher, J.; Allen, N.; Beral, V.; Burton, P.; Danesh, J.; et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med 2015, 12, e1001779. [Google Scholar] [CrossRef] [PubMed]
- Saxena, R.; Saleheen, D.; Been, L.F.; Garavito, M.L.; Braun, T.; Bjonnes, A.; et al. Genome-Wide Association Study Identifies a Novel Locus Contributing to Type 2 Diabetes Susceptibility in Sikhs of Punjabi Origin From India. Diabetes 2013, 62, 1746–1755. [Google Scholar] [CrossRef]
- Das, S.; Forer, L.; Schonherr, S.; Sidore, C.; Locke, A.E.; Kwong, A.; et al. Next-generation genotype imputation service and methods. Nat Genet 2016, 48, 1284–1287. [Google Scholar] [CrossRef]
- Rout, M.; Tung, G.K.; Singh, J.R.; Mehra, N.K.; Wander, G.S.; Ralhan, S.; et al. Polygenic Risk Score Assessment for Coronary Artery Disease in Asian Indians. J Cardiovasc Transl Res. 2024. [Google Scholar] [CrossRef]
- Saxena, R.; Saleheen, D.; Been, L.F.; Garavito, M.L.; Braun, T.; Bjonnes, A.; et al. Genome-wide association study identifies a novel locus contributing to type 2 diabetes susceptibility in Sikhs of Punjabi origin from India. Diabetes 2013, 62, 1746–1755. [Google Scholar] [CrossRef]
- Wang, X.; Hivert, V.; Groot, S.; Wang, Y.; Yengo, L.; McGrath, J.J.; et al. Cross-ancestry analyses identify new genetic loci associated with 25-hydroxyvitamin D. PLoS Genet 2023, 19, e1011033. [Google Scholar] [CrossRef]
- Collister, J.A.; Liu, X.; Clifton, L. Calculating Polygenic Risk Scores (PRS) in UK Biobank: A Practical Guide for Epidemiologists. Front Genet 2022, 13, 818574. [Google Scholar] [CrossRef]
- O’Connor, M.J.; Schroeder, P.; Huerta-Chagoya, A.; Cortes-Sanchez, P.; Bonas-Guarch, S.; Guindo-Martinez, M.; et al. Recessive Genome-Wide Meta-analysis Illuminates Genetic Architecture of Type 2 Diabetes. Diabetes 2022, 71, 554–565. [Google Scholar] [CrossRef]
- Burgess, S.; Scott, R.A.; Timpson, N.J.; Davey Smith, G.; Thompson, S.G.; Consortium, E.-I. Using published data in Mendelian randomization: a blueprint for efficient identification of causal risk factors. Eur J Epidemiol 2015, 30, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Burgess, S.; Thompson, S.G. Use of allele scores as instrumental variables for Mendelian randomization. Int J Epidemiol 2013, 42, 1134–1144. [Google Scholar] [CrossRef] [PubMed]
- Rees, J.M.B.; Foley, C.N.; Burgess, S. Factorial Mendelian randomization: using genetic variants to assess interactions. Int J Epidemiol 2020, 49, 1147–1158. [Google Scholar] [CrossRef]
- Sanderson, E.; Glymour, M.M.; Holmes, M.V.; Kang, H.; Morrison, J.; Munafo, M.R.; et al. Mendelian randomization. Nat Rev Methods Primers 2022, 2. [Google Scholar] [CrossRef]
- Weeks, J.P. plink: An R Package for Linking Mixed-Format Tests Using IRT-Based Methods. J Stat Softw 2010, 35, 1–33. [Google Scholar] [CrossRef]
- Deleskog, A.; Hilding, A.; Brismar, K.; Hamsten, A.; Efendic, S.; Ostenson, C.G. Low serum 25-hydroxyvitamin D level predicts progression to type 2 diabetes in individuals with prediabetes but not with normal glucose tolerance. Diabetologia 2012, 55, 1668–1678. [Google Scholar] [CrossRef]
- Vacek, J.L.; Vanga, S.R.; Good, M.; Lai, S.M.; Lakkireddy, D.; Howard, P.A. Vitamin D deficiency and supplementation and relation to cardiovascular health. Am J Cardiol 2012, 109, 359–363. [Google Scholar] [CrossRef]
- Sanghera, D.K.; Sapkota, B.R.; Aston, C.E.; Blackett, P.R. Vitamin D Status, Gender Differences, and Cardiometabolic Health Disparities. Ann Nutr Metab 2017, 70, 79–87. [Google Scholar] [CrossRef]
- Sun, K.; Chen, Y.; Lam, H.; Nelson, E.A.S.; Kong, A.P.; Au Yeung, S.L.; et al. Association between vitamin D level and risk of type 2 diabetes: a systematic review of Mendelian Randomization studies. Crit Rev Food Sci Nutr 2025, 65, 7301–7310. [Google Scholar] [CrossRef]
- Zheng, J.S.; Luan, J.; Sofianopoulou, E.; Sharp, S.J.; Day, F.R.; Imamura, F.; et al. The association between circulating 25-hydroxyvitamin D metabolites and type 2 diabetes in European populations: A meta-analysis and Mendelian randomisation analysis. PLoS Med 2020, 17, e1003394. [Google Scholar] [CrossRef] [PubMed]
- De La Barrera, B.; Manousaki, D. Serum 25-Hydroxyvitamin D Levels and Youth-Onset Type 2 Diabetes: A Two-Sample Mendelian Randomization Study. Nutrients 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Al-Shami, A.S.; Alzumor, M.; Aladhal, A.; Al-Saban, A. Association between vitamin D level and insulin resistance among patients with type 2 diabetes mellitus in Sana’a City. BMC Endocr Disord 2025, 25, 152. [Google Scholar] [CrossRef]
- Ramirez Stieben, L.A.; Dobry, R.; Anca, L.; Gonzalez, A.; Lopez, M.I.; Bayo, S.; et al. Hypovitaminosis D in patients with type 2 diabetes: risk factors and association with glycemic control and established microvascular complications. Rev Fac Cien Med Univ Nac Cordoba 2022, 79, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Dharambir, K.; Sanghera, P.R.B. Vitamin D Status, Genetics, and Diabetes Risk; Academic Press, 2016. [Google Scholar]
- Anderson, R.L.; Ternes, S.B.; Strand, K.A.; Rowling, M.J. Vitamin D homeostasis is compromised due to increased urinary excretion of the 25-hydroxycholecalciferol-vitamin D-binding protein complex in the Zucker diabetic fatty rat. Am J Physiol Endocrinol Metab 2010, 299, E959–E967. [Google Scholar] [CrossRef]
- Yi, B.; Huang, J.; Zhang, W.; Li, A.M.; Yang, S.K.; Sun, J.; et al. Vitamin D Receptor Down-Regulation Is Associated With Severity of Albuminuria in Type 2 Diabetes Patients. J Clin Endocrinol Metab 2016, 101, 4395–4404. [Google Scholar] [CrossRef] [PubMed]
- Parsanathan, R.; Jain, S.K. Glutathione deficiency induces epigenetic alterations of vitamin D metabolism genes in the livers of high-fat diet-fed obese mice. Sci Rep 2019, 9, 14784. [Google Scholar] [CrossRef]
- Tarfeen, N.; Masoodi, S.R.; Nisa, K.U.; Ali, S.; Ahmad, M.B.; Ganai, B.A. VDR downregulation and promoter hypermethylation as one of the causes for triggering type 2 diabetes mellitus: Clinical and molecular studies. J Diabetes Metab Disord 2023, 22, 1443–1451. [Google Scholar] [CrossRef]
- Mirza, I.; Mohamed, A.; Deen, H.; Balaji, S.; Elsabbahi, D.; Munasser, A.; et al. Obesity-Associated Vitamin D Deficiency Correlates with Adipose Tissue DNA Hypomethylation, Inflammation, and Vascular Dysfunction. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- AlSedairy, S.A.; Al-Harbi, L.N.; Binobead, M.A.; Athinarayanan, J.; Arzoo, S.; Al-Tamimi, D.S.; et al. Association of CYP2R1 and CYP27B1 genes with the risk of obesity and vitamin D metabolism in Saudi women. J Genet Eng Biotechnol 2023, 21, 59. [Google Scholar] [CrossRef]
| Trait | Europeans (N =459,143) |
AIDHS/SDS (N = 3486) |
South Asians (N = 9372) |
Africans (N=3346) |
|---|---|---|---|---|
| Males (%) | 46 | 55 | 54 | 51 |
| Age (years) | 56.77+8.03 | 51.98+13.27* | 53.30+8.45^ | 51.00+7.94 |
| BMI (kg/m2) | 27.40+4.77 | 26.56+4.79* | 27.16+4.40^ | 29.68+5.14 |
| Waist (cm) | 90.26+13.51 | 92.23+11.94* | 91.45+11.86^ | 94.16+11.63 |
| Waist-to-hip ratio | 0.87+0.09 | 0.94+0.08* | 0.90+0.09^ | 0.88+0.08 |
| Systolic BP (mmHg) | 137.98+18.64 | 137.07+28.83 | 129.90+29.73^ | 138.66+18.87 |
| Diastolic BP (mmHg) | 82.18+10.12 | 82.51+12.43 | 79.45+17.47^ | 84.92+10.83 |
| Blood glucose (mg/dL) | 80.30+36.89 | 134.78+63.72* | 97.66+33.94^ | 91.99+27.23 |
| Triglycerides (mg/dL) | 147.89+94.52 | 169.87+112.60* | 173.98+103.35 | 107.29+67.55 |
| HDL-C (mg/dL) | 49.03+23.28 | 40.53+14.80* | 48.79+12.48^ | 53.84+13.84 |
| LDL-C (mg/dL) | 131.31+44.17 | 112.79+39.04* | 129.34+32.96^ | 123.96+32.19 |
| Total Cholesterol (mg/dL) | 212.05+61.18 | 184.06+61.74* | 205.38+43.62^ | 198.73+42.34 |
| Vitamin D levels (nmol/L) | 41.23+26.13 | 35.05+26.69* | 21.36+17.81^ | 27.67+19.04 |
| T2D (%) | 5 | 51* | 21^ | 11 |
| CAD (%) | 5 | 19* | 31^ | 6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




