Submitted:
07 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction: Mechanics as a Unifying Language for Post-MI Remodeling
2. Literature Search and Scope
3. The Remodeling Timeline: Linking Biological Phases to Mechanical Transitions
4. Microstructure-to-Mechanics in Infarct, Border Zone, and Remote Myocardium
5. Constitutive Models for Post-MI Ventricular Mechanics: Region Specificity and Model-Form Uncertainty
6. Growth and Remodeling Frameworks for Healing and Chronic Remodeling
7. Multiscale Coupling: Strategies for Bridging Microstructure and Organ Function
8. Imaging-Informed Personalization Pipelines: From Data to a Calibrated Model
9. Parameter Identifiability in Post-MI Models: What Can Be Learned from Clinical Data?
| Pitfall | Mechanistic cause | Observed symptom | Practical remedy |
|---|---|---|---|
| Diastolic stiffness–pressure confounding | Similar PV behavior can be reproduced by different stiffness–pressure pairs | Wide range of fitted stiffness values with similar error | Treat pressure as uncertain; include filling dynamics; use physiologic priors and sensitivity tests |
| Scar stiffness–border contractility confounding | Passive and active contributions trade off in regional motion | Non-unique regional parameter maps | Use multi-time strain; incorporate viability/perfusion priors; constrain scar properties by maturation stage |
| Uncertain fiber architecture | Stress and strain depend strongly on fiber directions | Parameters change markedly under different fiber fields | Propagate fiber uncertainty; use diffusion MRI when feasible; ensemble testing |
| Boundary condition ambiguity | Basal constraints and pericardial effects alter deformation | Good fit but unrealistic twist or basal motion | Calibrate basal constraints; validate torsion; include simplified pericardial contact models |
| Overparameterized constitutive law | Too many degrees of freedom relative to data | Optimization instability; large posterior uncertainty | Reduce parameter count; perform model selection; constrain with microstructure priors |
| Data package | Typical measurements | Mechanics constraint | Key limitation | Decision-grade outputs most defensible |
|---|---|---|---|---|
| Package A: Routine, non-invasive | Cine CMR (geometry and LV volume curves); LGE-CMR (scar localization/transmurality); brachial blood pressure (bounded proxy for systolic loading); multi-phase regional strain (feature tracking or speckle-tracking echocardiography, STE) | Constrains global kinematics and relative regional dysfunction; provides scar geometry priors; supports coarse region-wise differences (infarct vs. remote) | Passive stiffness–pressure confounding (no LV pressure/filling pressure); passive–active trade-off (contractility vs. stiffness); boundary-condition uncertainty; strain method bias/variability | Qualitative or probabilistic risk ranking with wide uncertainty; relative indicators of remodeling risk rather than absolute stress-based thresholds |
| Package B: Enhanced, still non-invasive | Package A plus T1 mapping/ECV (diffuse fibrosis prior); higher-fidelity strain (DENSE/tagging where feasible); Doppler/echo hemodynamics to bound diastolic filling pressures | Narrower bounds on passive stiffness using tissue characterization priors; improved regional calibration; reduced posterior uncertainty; better separation of infarct/border/remote behavior | Residual uncertainty in absolute LV pressure and boundary conditions; remaining passive–active confounding if activation is weakly constrained; inter-method variability in strain and mapping | More credible probabilistic forecasts of remodeling trajectory; improved patient selection for escalation/surveillance; preliminary stress-based metrics with uncertainty bands (not point estimates) |
| Package C: Reference standard for parameter separation | Packages A/B plus invasive LV pressure waveform (or well-validated pressure estimation); ideally paired pressure–volume data synchronized with strain | Substantially improved separation of passive stiffness vs. loading and passive vs. active contributions; stronger identifiability of region-wise stiffness/contractility scaling; more credible stress/strain field inference | Model-form uncertainty (constitutive and activation choices); spatial heterogeneity remains under-determined if data are sparse; numerical/interface assumptions can affect stress gradients | Decision-relevant stress-based metrics with quantified uncertainty; counterfactual therapy simulations (e.g., infarct reinforcement) with defensible credibility claims; validation against longitudinal endpoints |
10. Uncertainty Quantification and Credibility: Verification, Validation, and Decision Relevance
11. What to Measure Next: Building Datasets That Make Prediction Possible
11.1. Illustrative Clinical Use Cases for Decision-Relevant Modeling
12. Translational Outlook: From Mechanistic Models to Clinical Tools
13. Conclusions
References
- Sutton, M. G. S. J.; Sharpe, N. Left ventricular remodeling after myocardial infarction: pathophysiology and therapy. Circulation 2000, vol. 101(no. 25), 2981–2988. [Google Scholar] [CrossRef]
- Holmes, J. W.; Borg, T. K.; Covell, J. W. Structure and mechanics of healing myocardial infarcts. Annu. Rev. Biomed. Eng. 2005, vol. 7(no. 1), 223–253. [Google Scholar] [CrossRef]
- Weisman, H. F.; Healy, B. Myocardial infarct expansion, infarct extension, and reinfarction: pathophysiologic concepts. Progress in cardiovascular diseases 1987, vol. 30(no. 2), 73–110. [Google Scholar] [CrossRef]
- Frangogiannis, N. G. The inflammatory response in myocardial injury, repair, and remodelling. Nature Reviews Cardiology 2014, vol. 11(no. 5), 255–265. [Google Scholar] [CrossRef]
- Prabhu, S. D.; Frangogiannis, N. G. The biological basis for cardiac repair after myocardial infarction: from inflammation to fibrosis. Circulation research 2016, vol. 119(no. 1), 91–112. [Google Scholar] [CrossRef]
- Talman, V.; Ruskoaho, H. Cardiac fibrosis in myocardial infarction—from repair and remodeling to regeneration. Cell and tissue research 2016, vol. 365(no. 3), 563–581. [Google Scholar] [CrossRef]
- Omens, J. H.; Fung, Y.-C. Residual strain in rat left ventricle. Circulation research 1990, vol. 66(no. 1), 37–45. [Google Scholar] [CrossRef] [PubMed]
- Richardson, W. J.; Clarke, S. A.; Quinn, T. A.; Holmes, J. W. Physiological implications of myocardial scar structure. Comprehensive Physiology 2015, vol. 5(no. 4), 1877–1909. [Google Scholar] [CrossRef]
- Tomasek, J. J.; Gabbiani, G.; Hinz, B.; Chaponnier, C.; Brown, R. A. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nature reviews Molecular cell biology 2002, vol. 3(no. 5), 349–363. [Google Scholar] [CrossRef]
- Hinz, B. The myofibroblast: paradigm for a mechanically active cell. Journal of biomechanics 2010, vol. 43(no. 1), 146–155. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Halade, G. V.; Lindsey, M. L. Extracellular matrix and fibroblast communication following myocardial infarction. Journal of cardiovascular translational research 2012, vol. 5(no. 6), 848–857. [Google Scholar] [CrossRef]
- Clarke, S. A.; Richardson, W. J.; Holmes, J. W. Modifying the mechanics of healing infarcts: Is better the enemy of good? Journal of molecular and cellular cardiology 2016, vol. 93, 115–124. [Google Scholar] [CrossRef]
- Richardson, W. J.; Holmes, J. W. Emergence of collagen orientation heterogeneity in healing infarcts and an agent-based model. Biophysical journal 2016, vol. 110(no. 10), 2266–2277. [Google Scholar] [CrossRef]
- Heineke, J.; Molkentin, J. D. Regulation of cardiac hypertrophy by intracellular signalling pathways. Nature reviews Molecular cell biology 2006, vol. 7(no. 8), 589–600. [Google Scholar] [CrossRef] [PubMed]
- Díez, J. Mechanisms of cardiac fibrosis in hypertension. The Journal of Clinical Hypertension 2007, vol. 9(no. 7), 546–550. [Google Scholar] [CrossRef]
- González; Ravassa, S.; Beaumont, J.; López, B.; Díez, J. New targets to treat the structural remodeling of the myocardium. Journal of the American College of Cardiology 2011, vol. 58(no. 18), 1833–1843. [Google Scholar] [CrossRef] [PubMed]
- Spinale, F. G. Myocardial matrix remodeling and the matrix metalloproteinases: influence on cardiac form and function. Physiological reviews 2007, vol. 87(no. 4), 1285–1342. [Google Scholar] [CrossRef]
- Otsuji, Y.; Handschumacher, M. D.; Schwammenthal, E.; Jiang, L.; Song, J.-K.; Guerrero, J. L.; Vlahakes, G. J.; Levine, R. A. Insights from three-dimensional echocardiography into the mechanism of functional mitral regurgitation: direct in vivo demonstration of altered leaflet tethering geometry. Circulation 1997, vol. 96(no. 6), 1999–2008. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, M. A.; Braunwald, E. Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation 1990, vol. 81(no. 4), 1161–1172. [Google Scholar] [CrossRef]
- Bursi, F.; Enriquez-Sarano, M.; Nkomo, V. T.; Jacobsen, S. J.; Weston, S. A.; Meverden, R. A.; Roger, V. L. Heart failure and death after myocardial infarction in the community: the emerging role of mitral regurgitation. Circulation 2005, vol. 111(no. 3), 295–301. [Google Scholar] [CrossRef]
- Grayburn, P. A.; Sannino, A.; Packer, M. Proportionate and disproportionate functional mitral regurgitation: a new conceptual framework that reconciles the results of the MITRA-FR and COAPT trials. JACC: Cardiovascular Imaging 2019, vol. 12(no. 2), 353–362. [Google Scholar]
- Lee, L.; Kassab, G.; Guccione, J. Mathematical modeling of cardiac growth and remodeling. Wiley Interdisciplinary Reviews: Systems Biology and Medicine 2016, vol. 8(no. 3), 211–226. [Google Scholar] [CrossRef]
- Marcos-Garcés, V.; Bertolín-Boronat, C.; Merenciano-González, H.; Mas, M. L. M.; Alberola, J. I. C.; López-Bueno, L.; Rubio, A. P.; Pérez-Solé, N.; Ríos-Navarro, C.; de Dios, E. Left Ventricular Remodeling After Myocardial Infarction—Pathophysiology, Diagnostic Approach and Management During Cardiac Rehabilitation. International Journal of Molecular Sciences vol. 26(no. 22), pp. 10964, 2025. [CrossRef]
- Leancă, S. A.; Crișu, D.; Petriș, A. O.; Afrăsânie, I.; Genes, A.; Costache, A. D.; Tesloianu, D. N.; Costache, I. I. Left ventricular remodeling after myocardial infarction: from physiopathology to treatment. Life 2022, vol. 12(no. 8), pp. 1111. [Google Scholar] [CrossRef] [PubMed]
- Frantz, S.; Hundertmark, M. J.; Schulz-Menger, J.; Bengel, F. M.; Bauersachs, J. Left ventricular remodelling post-myocardial infarction: pathophysiology, imaging, and novel therapies. European heart journal 2022, vol. 43(no. 27), 2549–2561. [Google Scholar] [CrossRef]
- Gaudron, P.; Eilles, C.; Kugler, I.; Ertl, G. Progressive left ventricular dysfunction and remodeling after myocardial infarction. Potential mechanisms and early predictors. Circulation 1993, vol. 87(no. 3), 755–763. [Google Scholar] [CrossRef] [PubMed]
- Cohn, J. N.; Ferrari, R.; Sharpe, N.; Remodeling, a. I. F. o. C. Cardiac remodeling—concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. Journal of the American College of Cardiology 2000, vol. 35(no. 3), 569–582. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, M. A.; Pfeffer, J. M.; Fishbein, M. C.; Fletcher, P. J.; Spadaro, J.; Kloner, R. A.; Braunwald, E. Myocardial infarct size and ventricular function in rats. Circulation research 1979, vol. 44(no. 4), 503–512. [Google Scholar] [CrossRef]
- Weber, K. T. Cardiac interstitium in health and disease: the fibrillar collagen network. Journal of the American College of Cardiology 1989, vol. 13(no. 7), 1637–1652. [Google Scholar] [CrossRef]
- Weber, K. T.; Janicki, J. S.; Pick, R.; Abrahams, C.; Shroff, S. G.; Bashey, R. I.; Chen, R. M. Collagen in the hypertrophied, pressure-overloaded myocardium. Circulation 1987, vol. 75, I40–I47. [Google Scholar]
- Cleutjens, J. P.; Creemers, E. E. Integration of concepts: cardiac extracellular matrix remodeling after myocardial infarction. Journal of cardiac failure 2002, vol. 8(no. 6), S344–S348. [Google Scholar] [CrossRef] [PubMed]
- Creemers, E. E.; Pinto, Y. M. Molecular mechanisms that control interstitial fibrosis in the pressure-overloaded heart. Cardiovascular research 2011, vol. 89(no. 2), 265–272. [Google Scholar] [CrossRef] [PubMed]
- Bogen, D. K.; Rabinowitz, S. A.; Needleman, A.; McMahon, T. A.; Abelmann, W. H. An analysis of the mechanical disadvantage of myocardial infarction in the canine left ventricle. Circulation research 1980, vol. 47(no. 5), 728–741. [Google Scholar] [CrossRef]
- Fomovsky, G. M.; Holmes, J. W. Evolution of scar structure, mechanics, and ventricular function after myocardial infarction in the rat. American Journal of Physiology-Heart and Circulatory Physiology 2010, vol. 298(no. 1), H221–H228. [Google Scholar] [CrossRef]
- Guccione, J. M.; McCulloch, A. D.; Waldman, L. Passive material properties of intact ventricular myocardium determined from a cylindrical model; 1991. [Google Scholar]
- Holzapfel, G. A.; Ogden, R. W. Constitutive modelling of passive myocardium: a structurally based framework for material characterization. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences 2009, vol. 367(no. 1902), 3445–3475. [Google Scholar] [CrossRef] [PubMed]
- Holzapfel, G. A.; Humphrey, J. D.; Ogden, R. W. Biomechanics of soft biological tissues and organs, mechanobiology, homeostasis and modelling. Journal of the Royal Society Interface vol. 22(no. 222), pp. 20240361, 2025. [CrossRef]
- Nemavhola, F. Biaxial quantification of passive porcine myocardium elastic properties by region. In Engineering Solid Mechanics; 2017. [Google Scholar]
- Ngwangwa, H. M.; Nemavhola, F. Evaluating computational performances of hyperelastic models on supraspinatus tendon uniaxial tensile test data. Journal of Computational Applied Mechanics 2021, vol. 52(no. 1), 27–43. [Google Scholar]
- Nemavhola, F.; Ngwangwa, H.; Davies, N.; Franz, T. Passive biaxial tensile dataset of three main rat heart myocardia: left ventricle, mid-wall and right ventricle. 2021. [Google Scholar] [CrossRef]
- Pandelani, T.; Semakane, L.; Msibi, M.; Kuchumov, A. G.; Nemavhola, F. Passive biaxial mechanical properties of sheep myocardium. Frontiers in Bioengineering and Biotechnology vol. 13, pp. 1549829, 2025. [CrossRef]
- Nemavhola, F.; Ngwangwa, H. M.; Pandelani, T. An investigation of uniaxial mechanical properties of excised sheep heart muscle fibre–fitting of different hyperelastic constitutive models. 2021. [Google Scholar]
- Nemavhola, F.; Ngwangwa, H.; Pandelani, T.; Davies, N.; Franz, T. Understanding regional mechanics of rat myocardia by fitting hyperelatsic models. 2021. [Google Scholar] [CrossRef]
- Ashikaga, H.; Mickelsen, S. R.; Ennis, D. B.; Rodriguez, I.; Kellman, P.; Wen, H.; McVeigh, E. R. Electromechanical analysis of infarct border zone in chronic myocardial infarction. American Journal of Physiology-Heart and Circulatory Physiology 2005, vol. 289(no. 3), H1099–H1105. [Google Scholar] [CrossRef] [PubMed]
- Wenk, J. F.; Sun, K.; Zhang, Z.; Soleimani, M.; Ge, L.; Saloner, D.; Wallace, A. W.; Ratcliffe, M. B.; Guccione, J. M. Regional left ventricular myocardial contractility and stress in a finite element model of posterobasal myocardial infarction. J Biomech Eng 2011, vol. 133(no. 4), pp. 044501. [Google Scholar] [CrossRef]
- Shimkunas, R.; Zhang, Z.; Wenk, J. F.; Soleimani, M.; Khazalpour, M.; Acevedo-Bolton, G.; Wang, G.; Saloner, D.; Mishra, R.; Wallace, A. W. Left ventricular myocardial contractility is depressed in the borderzone after posterolateral myocardial infarction. The Annals of thoracic surgery 2013, vol. 95(no. 5), 1619–1625. [Google Scholar] [CrossRef] [PubMed]
- Martin, T. P.; MacDonald, E. A.; Bradley, A.; Watson, H.; Saxena, P.; Rog-Zielinska, E. A.; Raheem, A.; Fisher, S.; Elbassioni, A. A. M.; Almuzaini, O. Ribonucleicacid interference or small molecule inhibition of Runx 1 in the border zone prevents cardiac contractile dysfunction following myocardial infarction. Cardiovascular Research 2023, vol. 119(no. 16), 2663–2671. [Google Scholar] [CrossRef]
- Ma, J.; Chen, Q.; Lin, D.; Ma, S. The Role of Infarct Border Zone Remodelling in Ventricular Arrhythmias: Bridging Basic Research and Clinical Applications. Journal of Cellular and Molecular Medicine vol. 29(no. 7), pp. e70526, 2025. [CrossRef]
- Song, T.; Hui, W.; Huang, M.; Guo, Y.; Yu, M.; Yang, X.; Liu, Y.; Chen, X. Dynamic changes in ion channels during myocardial infarction and therapeutic challenges. International Journal of Molecular Sciences 2024, vol. 25(no. 12), pp. 6467. [Google Scholar] [CrossRef]
- Sayers, J. R.; Martinez-Navarro, H.; Sun, X.; de Villiers, C.; Sigal, S.; Weinberger, M.; Rodriguez, C. C.; Riebel, L. L.; Berg, L. A.; Camps, J. Cardiac conduction system regeneration prevents arrhythmias after myocardial infarction. Nature Cardiovascular Research 2025, vol. 4(no. 2), 163–179. [Google Scholar] [CrossRef]
- Menzel; Kuhl, E. Frontiers in growth and remodeling. Mechanics research communications 2012, vol. 42, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Segmentation, H. A. W. G. o. M.; Imaging, R. f. C.; Cerqueira, M. D.; Weissman, N. J.; Dilsizian, V.; Jacobs, A. K.; Kaul, S.; Laskey, W. K.; Pennell, D. J.; Rumberger, J. A.; Ryan, T. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: a statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 2002, vol. 105(no. 4), 539–542. [Google Scholar]
- Kerckhoffs, R. C.; Neal, M. L.; Gu, Q.; Bassingthwaighte, J. B.; Omens, J. H.; McCulloch, A. D. Coupling of a 3D finite element model of cardiac ventricular mechanics to lumped systems models of the systemic and pulmonic circulation. Annals of biomedical engineering 2007, vol. 35(no. 1), 1–18. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, J. D.; Rajagopal, K. A constrained mixture model for growth and remodeling of soft tissues. Mathematical models and methods in applied sciences 2002, vol. 12(no. 03), 407–430. [Google Scholar] [CrossRef]
- Lazarus; Dalton, D.; Husmeier, D.; Gao, H. Sensitivity analysis and inverse uncertainty quantification for the left ventricular passive mechanics. Biomechanics and Modeling in Mechanobiology 2022, vol. 21(no. 3), 953–982. [Google Scholar] [CrossRef]
- Tarantola. Inverse problem theory and methods for model parameter estimation: SIAM 2005.
- Raue; Kreutz, C.; Maiwald, T.; Bachmann, J.; Schilling, M.; Klingmüller, U.; Timmer, J. Structural and practical identifiability analysis of partially observed dynamical models by exploiting the profile likelihood. Bioinformatics 2009, vol. 25(no. 15), 1923–1929. [Google Scholar] [CrossRef]
- Bellman, R.; Åström, K. J. On structural identifiability. Mathematical biosciences 1970, vol. 7(no. 3-4), 329–339. [Google Scholar] [CrossRef]
- Kreutz; Raue, A.; Kaschek, D.; Timmer, J. Profile likelihood in systems biology. The FEBS journal 2013, vol. 280(no. 11), 2564–2571. [Google Scholar] [CrossRef]
- Kaipio, J. P.; Somersalo, E. Statistical and computational inverse problems; Springer, 2005. [Google Scholar]
- Borowska; Gao, H.; Lazarus, A.; Husmeier, D. Bayesian optimisation for efficient parameter inference in a cardiac mechanics model of the left ventricle. International Journal for Numerical Methods in Biomedical Engineering 2022, vol. 38(no. 5), pp. e3593. [Google Scholar] [CrossRef]
- Klotz, S.; Hay, I.; Dickstein, M. L.; Yi, G.-H.; Wang, J.; Maurer, M. S.; Kass, D. A.; Burkhoff, D. Single-beat estimation of end-diastolic pressure-volume relationship: a novel method with potential for noninvasive application. American Journal of Physiology-Heart and Circulatory Physiology 2006, vol. 291(no. 1), H403–H412. [Google Scholar] [CrossRef]
- Rumindo, G. K.; Ohayon, J.; Croisille, P.; Clarysse, P. In vivo estimation of normal left ventricular stiffness and contractility based on routine cine MR acquisition. Medical Engineering & Physics 2020, vol. 85, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Nasopoulou; Shetty, A.; Lee, J.; Nordsletten, D.; Rinaldi, C. A.; Lamata, P.; Niederer, S. Improved identifiability of myocardial material parameters by an energy-based cost function. Biomechanics and modeling in mechanobiology 2017, vol. 16(no. 3), 971–988. [Google Scholar] [CrossRef]
- Fernández-Jiménez, R.; García-Prieto, J.; Sánchez-González, J.; Agüero, J.; López-Martín, G. J.; Galán-Arriola, C.; Molina-Iracheta, A.; Doohan, R.; Fuster, V.; Ibáñez, B. Pathophysiology underlying the bimodal edema phenomenon after myocardial ischemia/reperfusion. Journal of the American College of Cardiology 2015, vol. 66(no. 7), 816–828. [Google Scholar] [CrossRef]
- Mavrogeni, S. Evaluation of myocardial iron overload using magnetic resonance imaging. Blood Transfusion 2009, vol. 7(no. 3), pp. 183. [Google Scholar]
- Dobaczewski, M.; Gonzalez-Quesada, C.; Frangogiannis, N. G. The extracellular matrix as a modulator of the inflammatory and reparative response following myocardial infarction. Journal of molecular and cellular cardiology 2010, vol. 48(no. 3), 504–511. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, G. M.; Bulkley, B. H. Infarct expansion versus extension: two different complications of acute myocardial infarction. The American journal of cardiology 1978, vol. 41(no. 7), 1127–1132. [Google Scholar] [CrossRef]
- Figueras, J.; Alcalde, O.; Barrabés, J. A.; Serra, V.; Alguersuari, J.; Cortadellas, J.; Lidón, R.-M. Changes in hospital mortality rates in 425 patients with acute ST-elevation myocardial infarction and cardiac rupture over a 30-year period. Circulation 2008, vol. 118(no. 25), 2783–2789. [Google Scholar] [CrossRef] [PubMed]
- Hamirani, Y. S.; Wong, A.; Kramer, C. M.; Salerno, M. Effect of microvascular obstruction and intramyocardial hemorrhage by CMR on LV remodeling and outcomes after myocardial infarction: a systematic review and meta-analysis. JACC: Cardiovascular Imaging 2014, vol. 7(no. 9), 940–952. [Google Scholar]
- Calvieri; Masselli, G.; Monti, R.; Spreca, M.; Gualdi, G. F.; Fedele, F. Intramyocardial hemorrhage: an enigma for cardiac MRI? BioMed Research International 2015, vol. 2015(no. 1), pp. 859073. [Google Scholar] [CrossRef]
- Biot, M. A. General theory of three-dimensional consolidation. Journal of applied physics 1941, vol. 12(no. 2), 155–164. [Google Scholar] [CrossRef]
- Fung, Y.-c. Fung, Biomechanics: mechanical properties of living tissues; Springer Science & Business Media, 2013. [Google Scholar]
- Cleutjens, J.; Verluyten, M.; Smiths, J.; Daemen, M. Collagen remodeling after myocardial infarction in the rat heart. The American journal of pathology 1995, vol. 147(no. 2), pp. 325. [Google Scholar]
- De Bakker, J.; Van Capelle, F.; Janse, M. J.; Wilde, A.; Coronel, R.; Becker, A. E.; Dingemans, K. P.; Van Hemel, N. M.; Hauer, R. Reentry as a cause of ventricular tachycardia in patients with chronic ischemic heart disease: electrophysiologic and anatomic correlation. Circulation 1988, vol. 77(no. 3), 589–606. [Google Scholar] [CrossRef]
- Stevenson, W. G.; Khan, H.; Sager, P.; Saxon, L.; Middlekauff, H.; Natterson, P.; Wiener, I. Identification of reentry circuit sites during catheter mapping and radiofrequency ablation of ventricular tachycardia late after myocardial infarction. Circulation 1993, vol. 88(no. 4), 1647–1670. [Google Scholar] [CrossRef] [PubMed]
- Fomovsky, G. M.; Rouillard, A. D.; Holmes, J. W. Regional mechanics determine collagen fiber structure in healing myocardial infarcts. Journal of molecular and cellular cardiology 2012, vol. 52(no. 5), 1083–1090. [Google Scholar] [CrossRef]
- Fomovsky, G. M.; Macadangdang, J. R.; Ailawadi, G.; Holmes, J. W. Model-based design of mechanical therapies for myocardial infarction. Journal of cardiovascular translational research 2011, vol. 4(no. 1), 82–91. [Google Scholar] [CrossRef]
- Pitt; Remme, W.; Zannad, F.; Neaton, J.; Martinez, F.; Roniker, B.; Bittman, R.; Hurley, S.; Kleiman, J.; Gatlin, M. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. New England Journal of Medicine 2003, vol. 348(no. 14), 1309–1321. [Google Scholar] [CrossRef]
- Pitt; Zannad, F.; Remme, W. J.; Cody, R.; Castaigne, A.; Perez, A.; Palensky, J.; Wittes, J. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. New England Journal of Medicine 1999, vol. 341(no. 10), 709–717. [Google Scholar] [CrossRef]
- Humphrey, J. D. Cardiovascular solid mechanics: cells, tissues, and organs; Springer Science & Business Media, 2013. [Google Scholar]
- Sáez, P.; Kuhl, E. Computational modeling of acute myocardial infarction. Computer methods in biomechanics and biomedical engineering 2016, vol. 19(no. 10), 1107–1115. [Google Scholar] [CrossRef]
- 40, V. Assessing Credibility of Computational Models Through Verification and Validation: Application to Medical Devices. In Standard; American Society of Mechanical Engineers: Washington, DC, 2018. [Google Scholar]
- Morrison, T. M.; Hariharan, P.; Funkhouser, C. M.; Afshari, P.; Goodin, M.; Horner, M. Assessing computational model credibility using a risk-based framework: application to hemolysis in centrifugal blood pumps. Asaio Journal 2019, vol. 65(no. 4), 349–360. [Google Scholar] [CrossRef]
- Viceconti, M.; Henney, A.; Morley-Fletcher, E. In silico clinical trials: how computer simulation will transform the biomedical industry. International Journal of Clinical Trials 2016, vol. 3(no. 2), 37–46. [Google Scholar] [CrossRef]
- Viceconti, M.; Pappalardo, F.; Rodriguez, B.; Horner, M.; Bischoff, J.; Tshinanu, F. M. In silico trials: Verification, validation and uncertainty quantification of predictive models used in the regulatory evaluation of biomedical products. Methods 2021, vol. 185, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Peirlinck, M.; Costabal, F. S.; Yao, J.; Guccione, J.; Tripathy, S.; Wang, Y.; Ozturk, D.; Segars, P.; Morrison, T.; Levine, S. Precision medicine in human heart modeling: Perspectives, challenges, and opportunities. Biomechanics and modeling in mechanobiology 2021, vol. 20(no. 3), 803–831. [Google Scholar] [CrossRef]
- Gissler, M. C.; Antiochos, P.; Ge, Y.; Heydari, B.; Gräni, C.; Kwong, R. Y. Cardiac magnetic resonance evaluation of LV remodeling post-myocardial infarction: prognosis, monitoring and trial endpoints. Cardiovascular Imaging 2024, vol. 17(no. 11), 1366–1380. [Google Scholar]
- Jiang, Z.; Sorrentino, G.; Simsek, S.; Roelofs, J. J.; Niessen, H. W.; Krijnen, P. A. Increased perivascular fibrosis and pro-fibrotic cellular transition in intramyocardial blood vessels in myocardial infarction patients. Journal of Molecular and Cellular Cardiology Plus 2024, vol. 10, pp. 100275. [Google Scholar] [CrossRef]
- Claridge; Drack, A.; Pinto, A. R.; Greening, D. W. Defining cardiac fibrosis complexity and regulation towards therapeutic development. Clinical and Translational Discovery 2023, vol. 3(no. 1), pp. e163. [Google Scholar] [CrossRef]
- Sharrack, N.; Das, A.; Kelly, C.; Teh, I.; Stoeck, C. T.; Kozerke, S.; Swoboda, P. P.; Greenwood, J. P.; Plein, S.; Schneider, J. E. The relationship between myocardial microstructure and strain in chronic infarction using cardiovascular magnetic resonance diffusion tensor imaging and feature tracking. Journal of Cardiovascular Magnetic Resonance 2022, vol. 24(no. 1), pp. 66. [Google Scholar] [CrossRef] [PubMed]
- Das; Kelly, C.; Teh, I.; Stoeck, C. T.; Kozerke, S.; Sharrack, N.; Swoboda, P. P.; Greenwood, J. P.; Schneider, J. E.; Plein, S. Pathophysiology of LV remodeling following STEMI: a longitudinal diffusion tensor CMR study. Cardiovascular Imaging 2023, vol. 16(no. 2), 159–171. [Google Scholar]
- Dall’Armellina; Ennis, D. B.; Axel, L.; Croisille, P.; Ferreira, P. F.; Gotschy, A.; Lohr, D.; Moulin, K.; Nguyen, C. T.; Nielles-Vallespin, S. Cardiac diffusion-weighted and tensor imaging: A consensus statement from the special interest group of the Society for Cardiovascular Magnetic Resonance. Journal of Cardiovascular Magnetic Resonance vol. 27(no. 1), pp. 101109, 2025. [CrossRef]
- Guan, X. Luo; Gao, H. Constrained mixture models of growth and remodelling in an infarct left ventricle: A modelling study. Journal of the Mechanics and Physics of Solids vol. 200, pp. 106121, 2025. [CrossRef]
- Hoang, P.; Guccione, J. Finite Element Modeling in Left Ventricular Cardiac Biomechanics: From Computational Tool to Clinical Practice. Bioengineering vol. 12(no. 9), pp. 913, 2025. [CrossRef]
- Black, N.; Bradley, J.; Schelbert, E. B.; Bonnett, L. J.; Lewis, G. A.; Lagan, J.; Orsborne, C.; Brown, P. F.; Soltani, F.; Fröjdh, F. Remote myocardial fibrosis predicts adverse outcome in patients with myocardial infarction on clinical cardiovascular magnetic resonance imaging. Journal of Cardiovascular Magnetic Resonance 2024, vol. 26(no. 2), pp. 101064. [Google Scholar] [CrossRef] [PubMed]
- Carberry, J.; Carrick, D.; Haig, C.; Rauhalammi, S. M.; Ahmed, N.; Mordi, I.; McEntegart, M.; Petrie, M. C.; Eteiba, H.; Hood, S. Remote zone extracellular volume and left ventricular remodeling in survivors of ST-elevation myocardial infarction. Hypertension 2016, vol. 68(no. 2), 385–391. [Google Scholar] [CrossRef]
- Biesbroek, P. S.; Amier, R. P.; Teunissen, P. F.; Hofman, M. B.; Robbers, L. F.; van de Ven, P. M.; Beek, A. M.; van Rossum, A. C.; van Royen, N.; Nijveldt, R. Changes in remote myocardial tissue after acute myocardial infarction and its relation to cardiac remodeling: a CMR T1 mapping study. PLoS One 2017, vol. 12(no. 6), pp. e0180115. [Google Scholar] [CrossRef] [PubMed]
- LeWinter, M. M.; Granzier, H. Cardiac titin: a multifunctional giant. Circulation 2010, vol. 121(no. 19), 2137–2145. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, N.; Wu, Y.; Nair, P.; Granzier, H. L. Phosphorylation of titin modulates passive stiffness of cardiac muscle in a titin isoform-dependent manner. The Journal of general physiology 2005, vol. 125(no. 3), 257–271. [Google Scholar] [CrossRef]
- Masithulela. Bi-ventricular finite element model of right ventricle overload in the healthy rat heart. Bio-medical materials and engineering 2016, vol. 27(no. 5), 507–525. [Google Scholar] [CrossRef]
- Masithulela. p. V003T03A005; The effect of over-loaded right ventricle during passive filling in rat heart: A biventricular finite element model.
- Masithulela, F. J. Computational biomechanics in the remodelling rat heart post myocardial infarction; 2016. [Google Scholar]
- Masithulela, F. p. V003T03A004; Analysis of passive filling with fibrotic myocardial infarction.
- Nemavhola, F. Detailed structural assessment of healthy interventricular septum in the presence of remodeling infarct in the free wall–A finite element model. Heliyon 2019, vol. 5(no. 6). [Google Scholar] [CrossRef]
- Nemavhola, F. Mechanics of the septal wall may be affected by the presence of fibrotic infarct in the free wall at end-systole. International Journal of Medical Engineering and Informatics 2019, vol. 11(no. 3), 205–225. [Google Scholar] [CrossRef]
- Nemavhola, F. Study of biaxial mechanical properties of the passive pig heart: material characterisation and categorisation of regional differences. International Journal of Mechanical and Materials Engineering 2021, vol. 16(no. 1), pp. 6. [Google Scholar] [CrossRef]
- Connolly, J.; Bishop, M. J. Computational representations of myocardial infarct scars and implications for arrhythmogenesis. Clinical Medicine Insights: Cardiology 2016, vol. 10, pp. CMC. S39708. [Google Scholar] [CrossRef]
- Sun, K.; Stander, N.; Jhun, C.-S.; Zhang, Z.; Suzuki, T.; Wang, G.-Y.; Saeed, M.; Wallace, A. W.; Tseng, E. E.; Baker, A. J. A computationally efficient formal optimization of regional myocardial contractility in a sheep with left ventricular aneurysm. 2009. [Google Scholar] [CrossRef]
- Factor, S.; Sonnenblick, E.; Kirk, E. The histologic border zone of acute myocardial infarction--islands or peninsulas? The American journal of pathology 1978, vol. 92(no. 1), pp. 111. [Google Scholar]
- Kung, L.; Vaseghi, M.; Gahm, J. K.; Shevtsov, J.; Garfinkel, A.; Shivkumar, K.; Ennis, D. B. Microstructural infarct border zone remodeling in the post-infarct swine heart measured by diffusion tensor MRI. Frontiers in physiology 2018, vol. 9, pp. 826. [Google Scholar] [CrossRef]
- Costa, Mendonca; Plank, G.; Rinaldi, C. A.; Niederer, S. A.; Bishop, M. J. Modeling the electrophysiological properties of the infarct border zone. Frontiers in physiology 2018, vol. 9, pp. 356. [Google Scholar]
- Dang, B.; Guccione, J. M.; Mishell, J. M.; Zhang, P.; Wallace, A. W.; Gorman, R. C.; Gorman, J. H., III; Ratcliffe, M. B. Akinetic myocardial infarcts must contain contracting myocytes: finite-element model study. American Journal of Physiology-Heart and Circulatory Physiology 2005, vol. 288(no. 4), H1844–H1850. [Google Scholar] [CrossRef]
- Pu; Boyden, P. A. Alterations of Na+ currents in myocytes from epicardial border zone of the infarcted heart: a possible ionic mechanism for reduced excitability and postrepolarization refractoriness. Circulation research 1997, vol. 81(no. 1), 110–119. [Google Scholar] [CrossRef]
- Gerber, L.; Rochitte, C. E.; Melin, J. A.; McVeigh, E. R.; Bluemke, D. A.; Wu, K. C.; Becker, L. C.; Lima, J. A. Microvascular obstruction and left ventricular remodeling early after acute myocardial infarction. Circulation 2000, vol. 101(no. 23), 2734–2741. [Google Scholar] [CrossRef] [PubMed]
- Shimkunas, R.; Makwana, O.; Spaulding, K.; Bazargan, M.; Khazalpour, M.; Takaba, K.; Soleimani, M.; Myagmar, B.-E.; Lovett, D. H.; Simpson, P. C. Myofilament dysfunction contributes to impaired myocardial contraction in the infarct border zone. American Journal of Physiology-Heart and Circulatory Physiology 2014, vol. 307(no. 8), H1150–H1158. [Google Scholar] [CrossRef] [PubMed]
- Kayvanpour; Mansi, T.; Sedaghat-Hamedani, F.; Amr, A.; Neumann, D.; Georgescu, B.; Seegerer, P.; Kamen, A.; Haas, J.; Frese, K. S. Towards personalized cardiology: multi-scale modeling of the failing heart. PLoS One 2015, vol. 10(no. 7), pp. e0134869. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.; Liu, J.; Ennis, D. B.; Marsden, A. L. Cardiac mechanics modeling: recent developments and current challenges. ArXiv 2509.07971 v1. 2025, pp. [Google Scholar]
- Galappaththige, S.; Gray, R. A.; Costa, C. M.; Niederer, S.; Pathmanathan, P. Credibility assessment of patient-specific computational modeling using patient-specific cardiac modeling as an exemplar. PLoS computational biology 2022, vol. 18(no. 10), pp. e1010541. [Google Scholar] [CrossRef] [PubMed]
- Gray, R. A.; Pathmanathan, P. Patient-specific cardiovascular computational modeling: diversity of personalization and challenges. Journal of cardiovascular translational research 2018, vol. 11(no. 2), 80–88. [Google Scholar] [CrossRef] [PubMed]
- Sack, L.; Davies, N. H.; Guccione, J. M.; Franz, T. Personalised computational cardiology: patient-specific modelling in cardiac mechanics and biomaterial injection therapies for myocardial infarction. Heart failure reviews 2016, vol. 21(no. 6), 815–826. [Google Scholar] [CrossRef]
- Maniaci, C. Verification Validation and Uncertainty Quantification (V&V/UQ); Sandia National Lab. (SNL-NM): Albuquerque, NM (United States), 2018. [Google Scholar]
- Avazmohammadi, R.; Soares, J. S.; Li, D. S.; Raut, S. S.; Gorman, R. C.; Sacks, M. S. A contemporary look at biomechanical models of myocardium. Annual review of biomedical engineering 2019, vol. 21(no. 1), 417–442. [Google Scholar] [CrossRef] [PubMed]
- Campos; Sundnes, J.; Dos Santos, R.; Rocha, B. Uncertainty quantification and sensitivity analysis of left ventricular function during the full cardiac cycle. Philosophical Transactions of the Royal Society A 2020, vol. 378(no. 2173), pp. 20190381. [Google Scholar] [CrossRef]
- Chabiniok, R.; Wang, V. Y.; Hadjicharalambous, M.; Asner, L.; Lee, J.; Sermesant, M.; Kuhl, E.; Young, A. A.; Moireau, P.; Nash, M. P. Multiphysics and multiscale modelling, data–model fusion and integration of organ physiology in the clinic: ventricular cardiac mechanics. Interface focus 2016, vol. 6(no. 2), pp. 20150083. [Google Scholar] [CrossRef] [PubMed]
- Mirams, R.; Pathmanathan, P.; Gray, R. A.; Challenor, P.; Clayton, R. H. Uncertainty and variability in computational and mathematical models of cardiac physiology. The Journal of physiology 2016, vol. 594(no. 23), 6833–6847. [Google Scholar] [CrossRef]
- Land, S.; Gurev, V.; Arens, S.; Augustin, C. M.; Baron, L.; Blake, R.; Bradley, C.; Castro, S.; Crozier, A.; Favino, M. Verification of cardiac mechanics software: benchmark problems and solutions for testing active and passive material behaviour. Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences 2015, vol. 471(no. 2184), pp. 20150641. [Google Scholar] [CrossRef]
- Taylor, A.; Figueroa, C. Patient-specific modeling of cardiovascular mechanics. Annual review of biomedical engineering 2009, vol. 11(no. 1), 109–134. [Google Scholar] [CrossRef]
- Sermesant; Delingette, H.; Ayache, N. An electromechanical model of the heart for image analysis and simulation. IEEE transactions on medical imaging 2006, vol. 25(no. 5), 612–625. [Google Scholar] [CrossRef]
- Smith; de Vecchi, A.; McCormick, M.; Nordsletten, D.; Camara, O.; Frangi, A. F.; Delingette, H.; Sermesant, M.; Relan, J.; Ayache, N. euHeart: personalized and integrated cardiac care using patient-specific cardiovascular modelling. Interface focus 2011, vol. 1(no. 3), 349–364. [Google Scholar] [CrossRef]
- Niederer, S. A.; Lumens, J.; Trayanova, N. A. Computational models in cardiology. Nature reviews cardiology 2019, vol. 16(no. 2), 100–111. [Google Scholar] [CrossRef]
- Biondi-Zoccai; D’Ascenzo, F.; Giordano, S.; Mirzoyev, U.; Erol, Ç.; Cenciarelli, S.; Leone, P.; Versaci, F. Artificial Intelligence in Cardiology: General Perspectives and Focus on Interventional Cardiology. Anatolian Journal of Cardiology vol. 29(no. 4), pp. 152, 2025. [CrossRef] [PubMed]
- Bragard, J. R.; Camara, O.; Echebarria, B.; Giorda, L. G.; Pueyo, E.; Saiz, J.; Sebastian, R.; Soudah, E.; Vazquez, M. Cardiac computational modelling. Revista Española de Cardiología (English Edition) 2021, vol. 74(no. 1), 65–71. [Google Scholar] [CrossRef]
- Trayanova, A. Whole-heart modeling: applications to cardiac electrophysiology and electromechanics. Circulation research 2011, vol. 108(no. 1), 113–128. [Google Scholar] [CrossRef]
- Milošević, M.; Milićević, B.; Simić, V.; Filipovic, N.; Kojić, M. HEART MODEL FOR ELECTROPHYSIOLOGY, MECHANICS AND BLOOD FLOW. Journal of the Serbian Society for Computational Mechanics 2025, vol. 19(no. 1). [Google Scholar] [CrossRef]
- Yang, C.; Cao, Y.; Xiang, M. Integration of electromechanical feedback in cardiac electrophysiology: A multiphysics approach using finite element analysis. Chaos, Solitons & Fractals vol. 199, pp. 116819, 2025.
- Chitiboi, T.; Axel, L. Magnetic resonance imaging of myocardial strain: a review of current approaches. Journal of Magnetic Resonance Imaging 2017, vol. 46(no. 5), 1263–1280. [Google Scholar] [CrossRef]
- Nash, M. P.; Hunter, P. J. Computational mechanics of the heart. Journal of elasticity and the physical science of solids 2000, vol. 61(no. 1), 113–141. [Google Scholar]
- Ngwangwa; Nemavhola, F.; Pandelani, T.; Msibi, M.; Mabuda, I.; Davies, N.; Franz, T. Determination of cross-directional and cross-wall variations of passive biaxial mechanical properties of rat myocardia. Processes 2022, vol. 10(no. 4), pp. 629. [Google Scholar] [CrossRef]
- Costa, K. D.; Holmes, J. W.; McCulloch, A. D. Modelling cardiac mechanical properties in three dimensions. Philosophical transactions of the Royal Society of London. Series A: Mathematical, physical and engineering sciences 2001, vol. 359(no. 1783), 1233–1250. [Google Scholar] [CrossRef]
- Dokos, S.; Smaill, B. H.; Young, A. A.; LeGrice, I. J. Shear properties of passive ventricular myocardium. American Journal of Physiology-Heart and Circulatory Physiology 2002, vol. 283(no. 6), H2650–H2659. [Google Scholar] [CrossRef]
- Mirsky; Parmley, W. W. Assessment of passive elastic stiffness for isolated heart muscle and the intact heart. Circulation research 1973, vol. 33(no. 2), 233–243. [Google Scholar] [CrossRef]
- Young, A.; Kramer, C. M.; Ferrari, V. A.; Axel, L.; Reichek, N. Three-dimensional left ventricular deformation in hypertrophic cardiomyopathy. Circulation 1994, vol. 90(no. 2), 854–867. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Singh, A.; Nazir, S. A.; Kanagala, P.; Gershlick, A. H.; McCann, G. P. Comparison of cardiovascular magnetic resonance feature tracking and tagging for the assessment of left ventricular systolic strain in acute myocardial infarction. European journal of radiology 2015, vol. 84(no. 5), 840–848. [Google Scholar] [CrossRef] [PubMed]
- Bogaert; Bosmans, H.; Maes, A.; Suetens, P.; Marchal, G.; Rademakers, F. E. Remote myocardial dysfunction after acute anterior myocardial infarction: impact of left ventricular shape on regional function: a magnetic resonance myocardial tagging study. Journal of the American College of Cardiology 2000, vol. 35(no. 6), 1525–1534. [Google Scholar] [CrossRef] [PubMed]
- Kim, R. J.; Fieno, D. S.; Parrish, T. B.; Harris, K.; Chen, E.-L.; Simonetti, O.; Bundy, J.; Finn, J. P.; Klocke, F. J.; Judd, R. M. Relationship of MRI delayed contrast enhancement to irreversible injury, infarct age, and contractile function. Circulation 1999, vol. 100(no. 19), 1992–2002. [Google Scholar] [CrossRef]
- Wu, C.; Zerhouni, E. A.; Judd, R. M.; Lugo-Olivieri, C. H.; Barouch, L. A.; Schulman, S. P.; Blumenthal, R. S.; Lima, J. A. Prognostic significance of microvascular obstruction by magnetic resonance imaging in patients with acute myocardial infarction. Circulation 1998, vol. 97(no. 8), 765–772. [Google Scholar] [CrossRef]
- Mor-Avi, V.; Lang, R. M.; Badano, L. P.; Belohlavek, M.; Cardim, N. M.; Derumeaux, G.; Galderisi, M.; Marwick, T.; Nagueh, S. F.; Sengupta, P. P. Current and evolving echocardiographic techniques for the quantitative evaluation of cardiac mechanics: ASE/EAE consensus statement on methodology and indications endorsed by the Japanese Society of Echocardiography. European Journal of Echocardiography 2011, vol. 12(no. 3), 167–205. [Google Scholar] [CrossRef]
- Beck, J. V.; Arnold, K. J. Parameter estimation in engineering and science; : James Beck, 1977. [Google Scholar]
- Godfrey, K. Identification of parametric models from experimental data [book review]. IEEE Transactions on Automatic Control 1999, vol. 44(no. 12), 2321–2322. [Google Scholar] [CrossRef]
- Kennedy, C.; O’Hagan, A. Bayesian calibration of computer models. Journal of the Royal Statistical Society: Series B (Statistical Methodology) 2001, vol. 63(no. 3), 425–464. [Google Scholar] [CrossRef]
- Sobol, M. Global sensitivity indices for nonlinear mathematical models and their Monte Carlo estimates. Mathematics and computers in simulation 2001, vol. 55(no. 1-3), 271–280. [Google Scholar] [CrossRef]
- Saltelli; Ratto, M.; Andres, T.; Campolongo, F.; Cariboni, J.; Gatelli, D.; Saisana, M.; Tarantola, S. Global sensitivity analysis: the primer; John Wiley & Sons, 2008. [Google Scholar]
- Axel, L.; Dougherty, L. MR imaging of motion with spatial modulation of magnetization. Radiology 1989, vol. 171(no. 3), 841–845. [Google Scholar] [CrossRef]
- Muthupillai, R.; Lomas, D. J.; Rossman, P. J.; Greenleaf, J. F.; Manduca, A.; Ehman, R. L. Magnetic resonance elastography by direct visualization of propagating acoustic strain waves. science 1995, vol. 269(no. 5232), 1854–1857. [Google Scholar] [CrossRef]
- Zerhouni, A.; Parish, D. M.; Rogers, W. J.; Yang, A.; Shapiro, E. P. Human heart: tagging with MR imaging--a method for noninvasive assessment of myocardial motion. Radiology 1988, vol. 169(no. 1), 59–63. [Google Scholar] [CrossRef]
- Aletras, H.; Ding, S.; Balaban, R. S.; Wen, H. DENSE: displacement encoding with stimulated echoes in cardiac functional MRI. Journal of magnetic resonance (San Diego, Calif.: 1997) 1999, vol. 137(no. 1), pp. 247. [Google Scholar] [CrossRef]
- Kim, R. J.; Wu, E.; Rafael, A.; Chen, E.-L.; Parker, M. A.; Simonetti, O.; Klocke, F. J.; Bonow, R. O.; Judd, R. M. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. New England Journal of Medicine 2000, vol. 343(no. 20), 1445–1453. [Google Scholar] [CrossRef]
- Osman, F.; Prince, J. L. Visualizing myocardial function using HARP MRI. Physics in Medicine & Biology 2000, vol. 45(no. 6), pp. 1665. [Google Scholar]
- Osman, F.; Sampath, S.; Atalar, E.; Prince, J. L. Imaging longitudinal cardiac strain on short-axis images using strain-encoded MRI. Magnetic Resonance in Medicine: An Official Journal of the International Society for Magnetic Resonance in Medicine 2001, vol. 46(no. 2), 324–334. [Google Scholar] [CrossRef] [PubMed]
- Scollan, D. F.; Holmes, A.; Winslow, R.; Forder, J. Histological validation of myocardial microstructure obtained from diffusion tensor magnetic resonance imaging. American Journal of Physiology-Heart and Circulatory Physiology 1998, vol. 275(no. 6), H2308–H2318. [Google Scholar] [CrossRef] [PubMed]
- Kuhl; Steinmann, P. Theory and numerics of geometrically non-linear open system mechanics. International Journal for Numerical Methods in Engineering 2003, vol. 58(no. 11), 1593–1615. [Google Scholar] [CrossRef]
- Gherbesi, E.; Gianstefani, S.; Angeli, F.; Ryabenko, K.; Bergamaschi, L.; Armillotta, M.; Guerra, E.; Tuttolomondo, D.; Gaibazzi, N.; Squeri, A. Myocardial strain of the left ventricle by speckle tracking echocardiography: From physics to clinical practice. Echocardiography 2024, vol. 41(no. 1), pp. e15753. [Google Scholar] [CrossRef]
- Canton, L.; Suma, N.; Amicone, S.; Impellizzeri, A.; Bodega, F.; Marinelli, V.; Ciarlantini, M.; Casuso, M.; Bavuso, L.; Belà, R. Clinical impact of multimodality assessment of myocardial viability. Echocardiography 2024, vol. 41(no. 7), pp. e15854. [Google Scholar] [CrossRef]
- Sagawa. The end-systolic pressure-volume relation of the ventricle: definition, modifications and clinical use. Circulation 1981, vol. 63(no. 6), 1223–1227. [Google Scholar] [CrossRef]
- Mendiola, E. A.; Neelakantan, S.; Xiang, Q.; Merchant, S.; Li, K.; Hsu, E. W.; Dixon, R. A.; Vanderslice, P.; Avazmohammadi, R. Contractile adaptation of the left ventricle post-myocardial infarction: predictions by rodent-specific computational modeling. Annals of biomedical engineering 2023, vol. 51(no. 4), 846–863. [Google Scholar] [CrossRef]
- Moon, C.; Messroghli, D. R.; Kellman, P.; Piechnik, S. K.; Robson, M. D.; Ugander, M.; Gatehouse, P. D.; Arai, A. E.; Friedrich, M. G.; Neubauer, S. Myocardial T1 mapping and extracellular volume quantification: a Society for Cardiovascular Magnetic Resonance (SCMR) and CMR Working Group of the European Society of Cardiology consensus statement. Journal of Cardiovascular Magnetic Resonance 2013, vol. 15(no. 1), pp. 92. [Google Scholar] [CrossRef] [PubMed]
- Schelbert, E. B.; Hsu, L.-Y.; Anderson, S. A.; Mohanty, B. D.; Karim, S. M.; Kellman, P.; Aletras, A. H.; Arai, A. E. Late gadolinium-enhancement cardiac magnetic resonance identifies postinfarction myocardial fibrosis and the border zone at the near cellular level in ex vivo rat heart. Circulation: Cardiovascular Imaging 2010, vol. 3(no. 6), 743–752. [Google Scholar] [CrossRef] [PubMed]
- Flett, S.; Hayward, M. P.; Ashworth, M. T.; Hansen, M. S.; Taylor, A. M.; Elliott, P. M.; McGregor, C.; Moon, J. C. Equilibrium contrast cardiovascular magnetic resonance for the measurement of diffuse myocardial fibrosis: preliminary validation in humans. Circulation 2010, vol. 122(no. 2), 138–144. [Google Scholar] [CrossRef] [PubMed]
- Gelman; Carlin, J. B.; Stern, H. S.; Rubin, D. B. Bayesian data analysis; Chapman and Hall/CRC, 1995. [Google Scholar]
- Smith, R. C. Uncertainty quantification: theory, implementation, and applications; : SIAM, 2024. [Google Scholar]
- Gutenkunst, R. N.; Waterfall, J. J.; Casey, F. P.; Brown, K. S.; Myers, C. R.; Sethna, J. P. Universally sloppy parameter sensitivities in systems biology models. PLoS computational biology 2007, vol. 3(no. 10), pp. e189. [Google Scholar] [CrossRef]
- Transtrum, K.; Qiu, P. Model reduction by manifold boundaries. Physical review letters 2014, vol. 113(no. 9), pp. 098701. [Google Scholar] [CrossRef]
- Erdemir; Guess, T. M.; Halloran, J.; Tadepalli, S. C.; Morrison, T. M. Considerations for reporting finite element analysis studies in biomechanics. Journal of biomechanics 2012, vol. 45(no. 4), 625–633. [Google Scholar] [CrossRef]
- Box, E.; Draper, N. R. Empirical model-building and response surfaces; John Wiley & Sons, 1987. [Google Scholar]
- Williams, K.; Rasmussen, C. E. Gaussian processes for machine learning; MIT press Cambridge, MA, 2006. [Google Scholar]
- Bishop, M.; Nasrabadi, N. M. Pattern recognition and machine learning; Springer, 2006. [Google Scholar]
- Pfeffer, A.; Braunwald, E.; Moyé, L. A.; Basta, L.; Brown, E. J., Jr.; Cuddy, T. E.; Davis, B. R.; Geltman, E. M.; Goldman, S.; Flaker, G. C. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction: results of the Survival and Ventricular Enlargement Trial. New England journal of medicine 1992, vol. 327(no. 10), 669–677. [Google Scholar] [CrossRef] [PubMed]
- Konstam, A.; Kramer, D. G.; Patel, A. R.; Maron, M. S.; Udelson, J. E. Left ventricular remodeling in heart failure: current concepts in clinical significance and assessment. JACC: Cardiovascular imaging 2011, vol. 4(no. 1), 98–108. [Google Scholar]
- Gjesdal; Hopp, E.; Vartdal, T.; Lunde, K.; Helle-Valle, T.; Aakhus, S.; Smith, H.-J.; Ihlen, H.; Edvardsen, T. Global longitudinal strain measured by two-dimensional speckle tracking echocardiography is closely related to myocardial infarct size in chronic ischaemic heart disease. Clinical science 2007, vol. 113(no. 6), 287–296. [Google Scholar] [CrossRef] [PubMed]
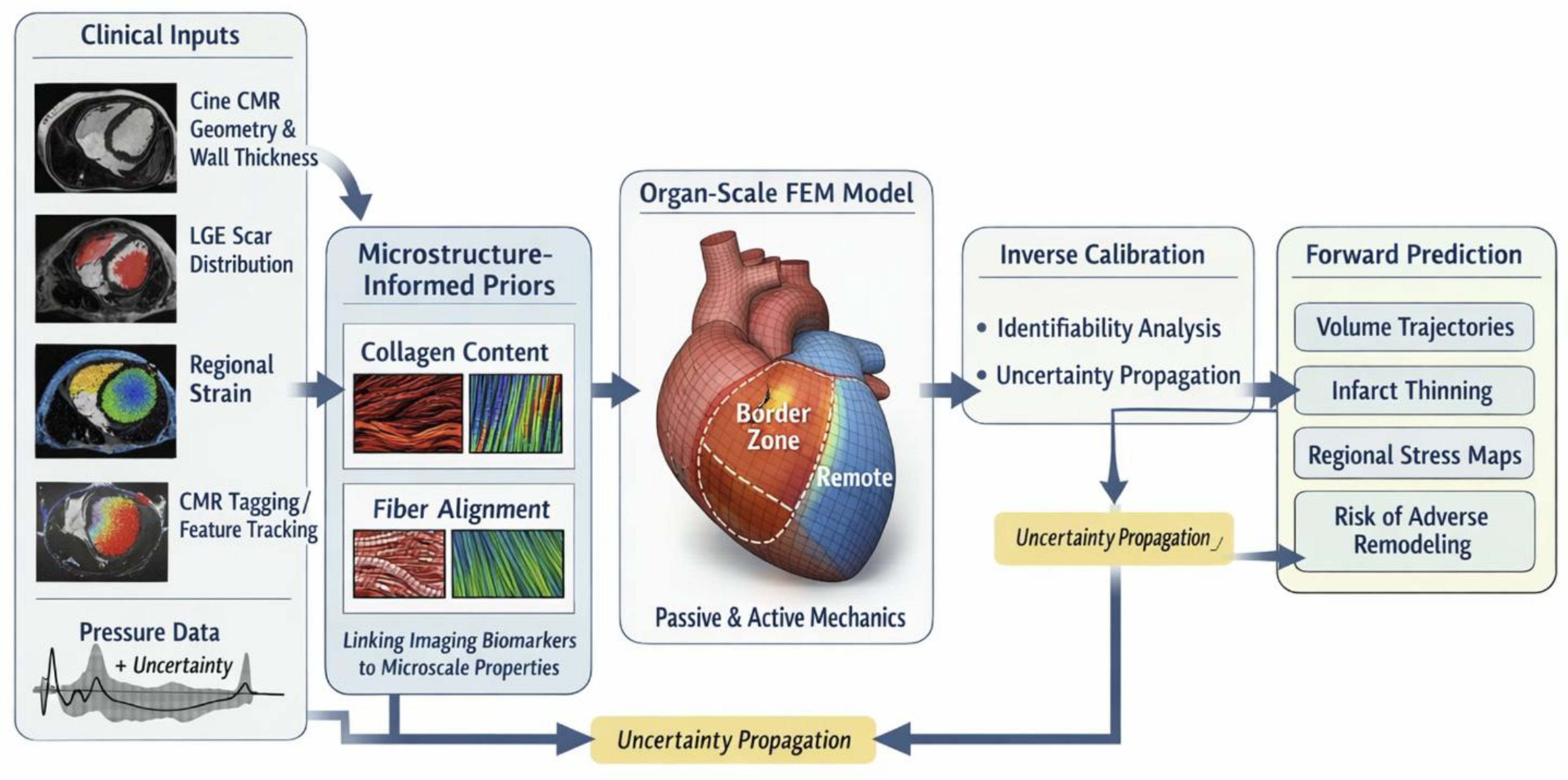
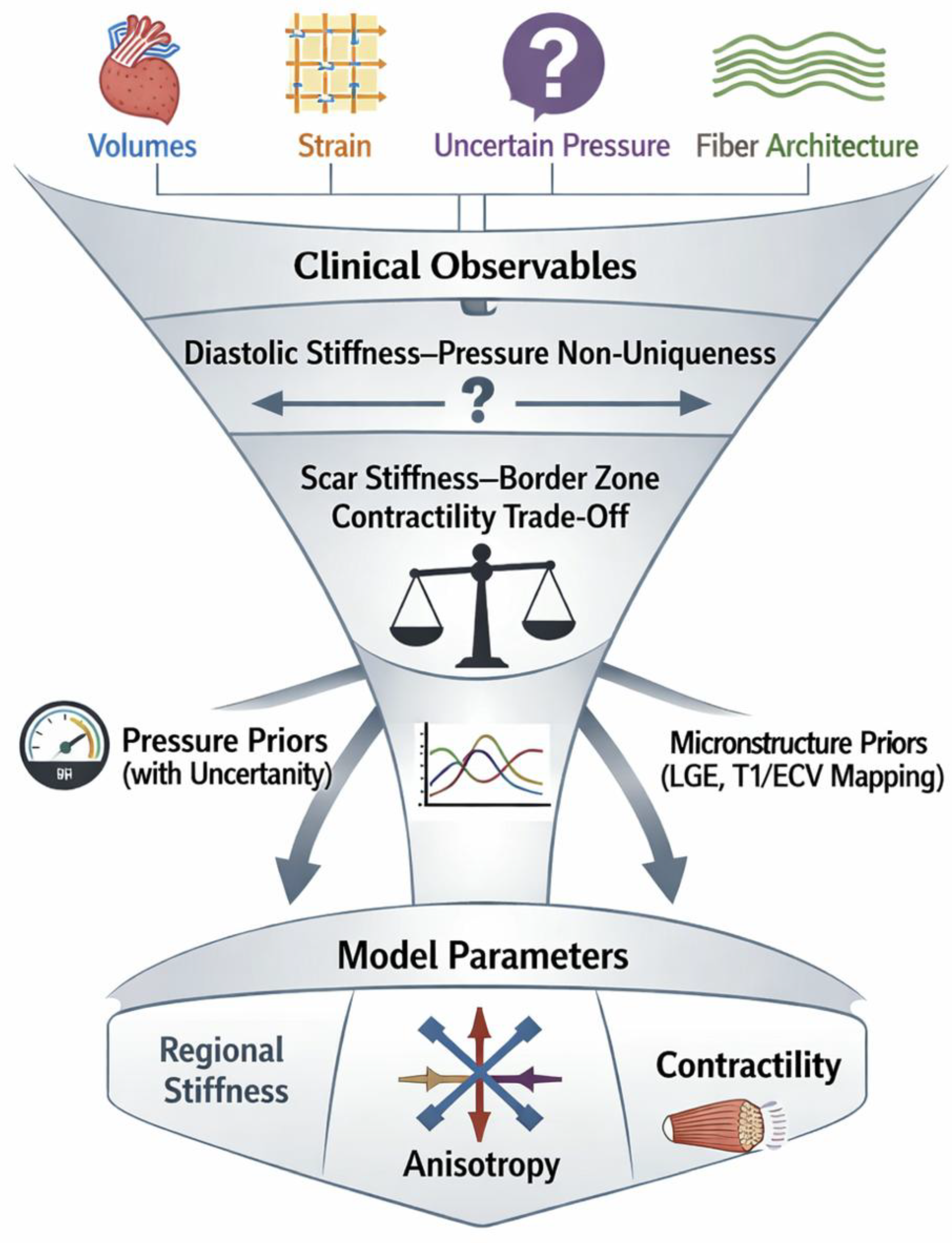
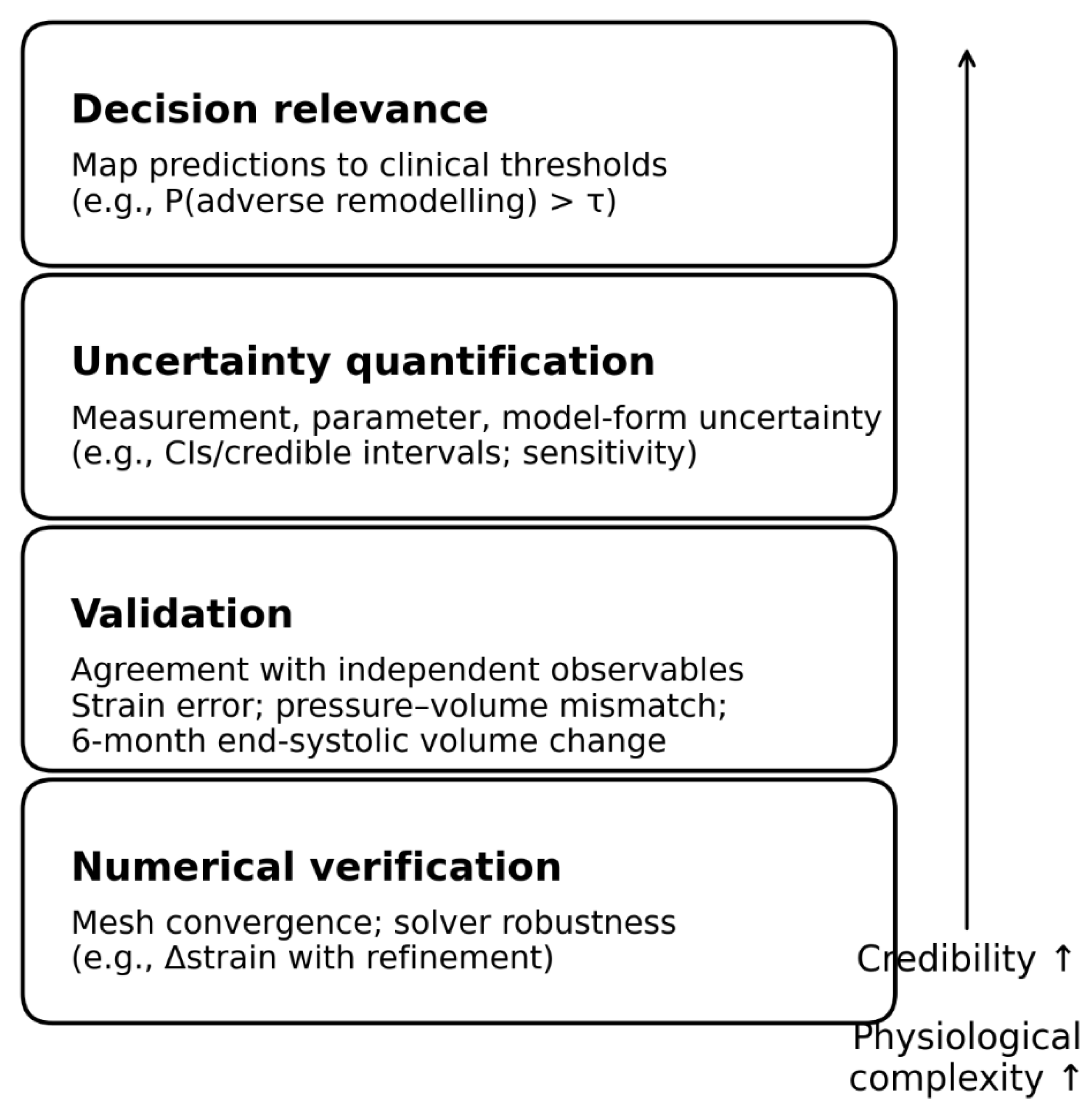
| Phase (approx.) | Infarct core mechanics | Border zone mechanics | Remote myocardium mechanics | Dominant microstructural drivers |
|---|---|---|---|---|
| Acute (hours–days) | Loss of active tension; high compliance; high strain; risk of expansion and rupture | High stress gradients; impaired activation; edema-dependent stiffness variability | Load redistribution; elevated stress; early diastolic changes possible | Myocyte necrosis; edema and hemorrhage; early matrix degradation; inflammatory turnover |
| Proliferative (days–weeks) | Progressive stiffening and strengthening; decreasing expansion tendency | Mixed viable myocytes and fibrosis; heterogeneous anisotropy; high mechanobiological activity | Hypertrophy initiation; onset of diffuse fibrosis in susceptible hearts | Myofibroblast activation; collagen deposition and alignment; early cross-linking; altered titin and cytoskeleton |
| Chronic (weeks–months) | Stiffer, stronger scar; reduced dissipation; stabilized anisotropy pattern | Patchy fibrosis; altered fiber architecture; arrhythmogenic mechanical substrate | Hypertrophy and diffuse fibrosis increase stiffness; dilation may continue | Collagen maturation and cross-linking; fiber reorientation; myocyte hypertrophy; sustained collagen turnover |
| Region | Common passive law | Anisotropy representation | Active behavior | Primary inference risk |
|---|---|---|---|---|
| Remote myocardium | Transversely isotropic or reduced orthotropic hyperelastic | Fiber directions from rule-based assignment or diffusion MRI when available | Active stress/strain with length dependence; often scaled by contractility factor | Passive stiffness and contractility can be confounded without pressure constraints |
| Border zone | Spatially varying hyperelastic parameters or mixture-based formulation | Heterogeneous anisotropy; often simplified to transversely isotropic | Reduced and heterogeneous activation; timing often prescribed | Scar stiffness–border contractility trade-offs produce non-unique fits |
| Infarct scar | Stiffened hyperelastic; time-dependent scaling for healing phase in some studies | Often isotropic; sometimes transversely isotropic aligned with principal strain or assumed fiber field | None | Isotropic assumption can bias stress concentrations and interface mechanics |
| Interface and transition | Smooth interpolation fields or discontinuous partitions with constraints | Gradients or discontinuities in fiber/stiffness | Transition in activation magnitude | Predictions sensitive to smoothing length scale and mesh resolution |
| Data source | Contribution | Mechanics constraint | Key limitation | Dominant uncertainty to propagate |
|---|---|---|---|---|
| CMR cine | Geometry, volumes, wall thickness | Constrains global kinematics and chamber shape | No direct stiffness information | Segmentation error; temporal resolution |
| CMR LGE | Scar map and transmurality | Defines infarct partition; guides heterogeneity priors | Threshold dependence; timing sensitivity | Classification uncertainty and partial volume |
| CMR T1/ECV | Diffuse fibrosis proxy | Prior on remote stiffness and collagen content | Indirect relation to stiffness; confounded by edema | Sequence dependence; calibration uncertainty |
| CMR tagging/DENSE/feature tracking | Regional strain time series | Constrains deformation field for inverse fitting | Noise and tracking bias; limited through-wall detail | Tracking error and smoothing assumptions |
| Echocardiographic strain + Doppler | Regional function and hemodynamics | Constrains timing and filling; accessible longitudinally | Operator dependence; acoustic window limits | Inter-observer variability and signal quality |
| Invasive LV pressure (subset) | True loading | Strongly constrains passive and active inference | Rare in routine follow-up | Timing alignment; measurement drift |
| Elastography (emerging) | Stiffness proxy | Potential direct constraint on regional stiffness | Frequency dependence; anisotropy interpretation | Modeling assumptions and motion artifacts |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




