Submitted:
08 March 2026
Posted:
10 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Scope, Terminology, and Literature Sampling
2.1. Why a Mechanics Unification Is Needed
2.2. Literature Sampling and Evidence Mapping
3. RV Anatomy, Fiber Architecture, and the Mechanical Consequences of Geometry
4. Mechanobiology of RV Remodeling Under Pressure Overload and Post-MI Contexts
5. Mechanical Characterization: From Pressure-Volume Loops to Ex-Vivo Tissue Testing
6. Constitutive and Electromechanical Models Tailored to the RV
7. Imaging-Derived RV Mechanics: What Is Measured, What Is Inferred, and What Is Comparable
8. Patient-Specific Modeling Pipelines: Geometry, Fibers, Inference, and Uncertainty
9. From Mechanics to Endpoints: What Should Be Predicted and How to Interpret Associations
10. Towards Reference Ranges and Reproducible Reporting for RV Remodeling Mechanics
11. Research Agenda: What to Measure Next
12. Conclusions
References
- Naeije, R.; Manes, A. The right ventricle in pulmonary arterial hypertension. European respiratory review 2014, 23, 476–487. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.J.; Huston, J.; Kutty, S.; Hatton, N.D.; Bowman, L.; Tian, L.; Herr, J.E.; Johri, A.M.; Archer, S.L. Right ventricular adaptation and failure in pulmonary arterial hypertension. Canadian Journal of Cardiology 2015, 31, 391–406. [Google Scholar] [CrossRef]
- Bernardo, R.J.; Haddad, F.; Couture, E.J.; Hansmann, G.; de Jesus Perez, V.A.; Denault, A.Y.; de Man, F.S.; Amsallem, M. Mechanics of right ventricular dysfunction in pulmonary arterial hypertension and heart failure with preserved ejection fraction. Cardiovascular diagnosis and therapy 2020, 10, 1580. [Google Scholar] [CrossRef]
- Dayer, N.; Ltaief, Z.; Liaudet, L.; Lechartier, B.; Aubert, J.-D.; Yerly, P. Pressure overload and right ventricular failure: from pathophysiology to treatment. Journal of clinical medicine 2023, 12, 4722. [Google Scholar] [CrossRef]
- Moceri, P.; Bouvier, P.; Baudouy, D.; Dimopoulos, K.; Cerboni, P.; Wort, S.J.; Doyen, D.; Schouver, E.-D.; Gibelin, P.; Senior, R. Cardiac remodelling amongst adults with various aetiologies of pulmonary arterial hypertension including Eisenmenger syndrome—implications on survival and the role of right ventricular transverse strain. European Heart Journal-Cardiovascular Imaging 2017, 18, 1262–1270. [Google Scholar] [CrossRef]
- Ryo, K.; Goda, A.; Onishi, T.; Delgado-Montero, A.; Tayal, B.; Champion, H.C.; Simon, M.A.; Mathier, M.A.; Gladwin, M.T.; Gorcsan, J., III. Characterization of right ventricular remodeling in pulmonary hypertension associated with patient outcomes by 3-dimensional wall motion tracking echocardiography. Circulation: Cardiovascular Imaging 2015, 8, e003176. [Google Scholar] [CrossRef]
- Fukuda, Y.; Tanaka, H.; Sugiyama, D.; Ryo, K.; Onishi, T.; Fukuya, H.; Nogami, M.; Ohno, Y.; Emoto, N.; Kawai, H. Utility of right ventricular free wall speckle-tracking strain for evaluation of right ventricular performance in patients with pulmonary hypertension. Journal of the American Society of Echocardiography 2011, 24, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Koestenberger, M.; Avian, A.; Chouvarine, P.; Gamillscheg, A.; Cvirn, G.; Schweintzger, S.; Kurath-Koller, S.; Cantinotti, M.; Hohmann, D.; Hansmann, G. Right ventricular end-systolic remodeling index in the assessment of pediatric pulmonary arterial hypertension. The European Pediatric Pulmonary Vascular Disease Network (EPPVDN). Pediatric Research 2020, 88, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Haddad, F.; Guihaire, J.; Skhiri, M.; Denault, A.Y.; Mercier, O.; Al-Halabi, S.; Vrtovec, B.; Fadel, E.; Zamanian, R.T.; Schnittger, I. Septal curvature is marker of hemodynamic, anatomical, and electromechanical ventricular interdependence in patients with pulmonary arterial hypertension. Echocardiography 2014, 31, 699–707. [Google Scholar] [CrossRef]
- Sanz, J.; García-Alvarez, A.; Fernández-Friera, L.; Nair, A.; Mirelis, J.G.; Sawit, S.T.; Pinney, S.; Fuster, V. Right ventriculo-arterial coupling in pulmonary hypertension: a magnetic resonance study. Heart 2012, 98, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Arroyo, J.; Santos-Martinez, L.E.; Aranda, A.; Pulido, T.; Beltran, M.; Muñoz-Castellanos, L.; Dominguez-Cano, E.; Sonnino, C.; Voelkel, N.F.; Sandoval, J. Differences in right ventricular remodeling secondary to pressure overload in patients with pulmonary hypertension. American journal of respiratory and critical care medicine 2014, 189, 603–606. [Google Scholar] [CrossRef]
- Tello, K.; Axmann, J.; Ghofrani, H.A.; Naeije, R.; Narcin, N.; Rieth, A.; Seeger, W.; Gall, H.; Richter, M.J. Relevance of the TAPSE/PASP ratio in pulmonary arterial hypertension. International journal of cardiology 2018, 266, 229–235. [Google Scholar] [CrossRef]
- Breeman, K.; Dufva, M.; Ploegstra, M.; Kheyfets, V.; Willems, T.; Wigger, J.; Hunter, K.; Ivy, D.; Berger, R.; Truong, U. Right ventricular-vascular coupling ratio in pediatric pulmonary arterial hypertension: a comparison between cardiac magnetic resonance and right heart catheterization measurements. International journal of cardiology 2019, 293, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Tello, K.; Dalmer, A.; Husain-Syed, F.; Seeger, W.; Naeije, R.; Ghofrani, H.A.; Gall, H.; Richter, M.J. Multibeat right ventricular–arterial coupling during a positive acute vasoreactivity test. American journal of respiratory and critical care medicine 2019, 199, e41–e42. [Google Scholar] [CrossRef] [PubMed]
- Kusakari, Y.; Urashima, T.; Shimura, D.; Amemiya, E.; Miyasaka, G.; Yokota, S.; Fujimoto, Y.; Akaike, T.; Inoue, T.; Minamisawa, S. Impairment of excitation-contraction coupling in right ventricular hypertrophied muscle with fibrosis induced by pulmonary artery banding. PLoS One 2017, 12, e0169564. [Google Scholar] [CrossRef]
- Rain, S.; Andersen, S.; Najafi, A.; Gammelgaard Schultz, J.; da Silva Gonçalves Bós, D.; Handoko, M.L.; Bogaard, H.-J.; Vonk-Noordegraaf, A.; Andersen, A.; van der Velden, J. Right ventricular myocardial stiffness in experimental pulmonary arterial hypertension: relative contribution of fibrosis and myofibril stiffness. Circulation: Heart Failure 2016, 9, e002636. [Google Scholar] [CrossRef]
- Guazzi, M.; Naeije, R.; Arena, R.; Corrà, U.; Ghio, S.; Forfia, P.; Rossi, A.; Cahalin, L.P.; Bandera, F.; Temporelli, P. Echocardiography of right ventriculoarterial coupling combined with cardiopulmonary exercise testing to predict outcome in heart failure. Chest 2015, 148, 226–234. [Google Scholar] [CrossRef]
- Vanderpool, R.R.; Pinsky, M.R.; Naeije, R.; Deible, C.; Kosaraju, V.; Bunner, C.; Mathier, M.A.; Lacomis, J.; Champion, H.C.; Simon, M.A. RV-pulmonary arterial coupling predicts outcome in patients referred for pulmonary hypertension. Heart 2015, 101, 37–43. [Google Scholar] [CrossRef]
- Meris, A.; Faletra, F.; Conca, C.; Klersy, C.; Regoli, F.; Klimusina, J.; Penco, M.; Pasotti, E.; Pedrazzini, G.B.; Moccetti, T. Timing and magnitude of regional right ventricular function: a speckle tracking-derived strain study of normal subjects and patients with right ventricular dysfunction. Journal of the American Society of Echocardiography 2010, 23, 823–831. [Google Scholar] [CrossRef]
- Vonk-Noordegraaf, A.; Marcus, J.T.; Gan, C.T.; Boonstra, A.; Postmus, P.E. Interventricular mechanical asynchrony due to right ventricular pressure overload in pulmonary hypertension plays an important role in impaired left ventricular filling. Chest 2005, 128, 628S–630S. [Google Scholar] [CrossRef] [PubMed]
- Piao, L.; Fang, Y.-H.; Cadete, V.J.; Wietholt, C.; Urboniene, D.; Toth, P.T.; Marsboom, G.; Zhang, H.J.; Haber, I.; Rehman, J. The inhibition of pyruvate dehydrogenase kinase improves impaired cardiac function and electrical remodeling in two models of right ventricular hypertrophy: resuscitating the hibernating right ventricle. Journal of molecular medicine 2010, 88, 47–60. [Google Scholar] [CrossRef]
- Wang, Z.; Chesler, N.C. Pulmonary vascular wall stiffness: an important contributor to the increased right ventricular afterload with pulmonary hypertension. Pulmonary circulation 2011, 1, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Guihaire, J.; Haddad, F.; Boulate, D.; Decante, B.; Denault, A.Y.; Wu, J.; Hervé, P.; Humbert, M.; Dartevelle, P.; Verhoye, J.-P. Non-invasive indices of right ventricular function are markers of ventricular–arterial coupling rather than ventricular contractility: insights from a porcine model of chronic pressure overload. European Heart Journal–Cardiovascular Imaging 2013, 14, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.J.; Peters, D.; Ghofrani, H.A.; Naeije, R.; Roller, F.; Sommer, N.; Gall, H.; Grimminger, F.; Seeger, W.; Tello, K. Evaluation and prognostic relevance of right ventricular–arterial coupling in pulmonary hypertension. American journal of respiratory and critical care medicine 2020, 201, 116–119. [Google Scholar] [CrossRef]
- Hsu, S.; Simpson, C.E.; Houston, B.A.; Wand, A.; Sato, T.; Kolb, T.M.; Mathai, S.C.; Kass, D.A.; Hassoun, P.M.; Damico, R.L. Multi-beat right ventricular-arterial coupling predicts clinical worsening in pulmonary arterial hypertension. Journal of the American Heart Association 2020, 9, e016031. [Google Scholar] [CrossRef]
- Bekedam, F.; Goumans, M.; Bogaard, H.; De Man, F.; Llucià-Valldeperas, A. Molecular mechanisms and targets of right ventricular fibrosis in pulmonary hypertension. Pharmacology & Therapeutics 2023, 244, 108389. [Google Scholar] [CrossRef]
- Rako, Z.A.; Kremer, N.; Yogeswaran, A.; Richter, M.J.; Tello, K. Adaptive versus maladaptive right ventricular remodelling. ESC Heart Failure 2023, 10, 762–775. [Google Scholar] [CrossRef] [PubMed]
- Fayyaz, A.U.; Edwards, W.D.; Maleszewski, J.J.; Konik, E.A.; DuBrock, H.M.; Borlaug, B.A.; Frantz, R.P.; Jenkins, S.M.; Redfield, M.M. Global pulmonary vascular remodeling in pulmonary hypertension associated with heart failure and preserved or reduced ejection fraction. Circulation 2018, 137, 1796–1810. [Google Scholar] [CrossRef]
- Amsallem, M.; Boulate, D.; Aymami, M.; Guihaire, J.; Selej, M.; Huo, J.; Denault, A.Y.; McConnell, M.V.; Schnittger, I.; Fadel, E. Load adaptability in patients with pulmonary arterial hypertension. The American Journal of Cardiology 2017, 120, 874–882. [Google Scholar] [CrossRef]
- Attard, M.I.; Dawes, T.J.; de Marvao, A.; Biffi, C.; Shi, W.; Wharton, J.; Rhodes, C.J.; Ghataorhe, P.; Gibbs, J.S.R.; Howard, L.S. Metabolic pathways associated with right ventricular adaptation to pulmonary hypertension: 3D analysis of cardiac magnetic resonance imaging. European Heart Journal-Cardiovascular Imaging 2019, 20, 668–676. [Google Scholar] [CrossRef]
- Trip, P.; Rain, S.; Handoko, M.L.; Van der Bruggen, C.; Bogaard, H.J.; Marcus, J.T.; Boonstra, A.; Westerhof, N.; Vonk-Noordegraaf, A.; de Man, F.S. Clinical relevance of right ventricular diastolic stiffness in pulmonary hypertension. European Respiratory Journal 2015, 45, 1603–1612. [Google Scholar] [CrossRef]
- Barker, N.; Fidock, B.; Johns, C.S.; Kaur, H.; Archer, G.; Rajaram, S.; Hill, C.; Thomas, S.; Karunasaagarar, K.; Capener, D. A systematic review of right ventricular diastolic assessment by 4D flow CMR. BioMed research international 2019, 2019, 6074984. [Google Scholar] [CrossRef]
- Fredriksson, A.G.; Svalbring, E.; Eriksson, J.; Dyverfeldt, P.; Alehagen, U.; Engvall, J.; Ebbers, T.; Carlhäll, C.J. 4D flow MRI can detect subtle right ventricular dysfunction in primary left ventricular disease. Journal of magnetic resonance imaging 2016, 43, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Hussain, I.; Mohammed, S.F.; Forfia, P.R.; Lewis, G.D.; Borlaug, B.A.; Gallup, D.S.; Redfield, M.M. Impaired right ventricular–pulmonary arterial coupling and effect of sildenafil in heart failure with preserved ejection fraction: An ancillary analysis from the phosphodiesterase-5 inhibition to improve clinical status and exercise capacity in diastolic heart failure (RELAX) trial. Circulation: Heart Failure 2016, 9, e002729. [Google Scholar]
- Van Der Bruggen, C.E.; Tedford, R.J.; Handoko, M.L.; Van Der Velden, J.; De Man, F.S. RV pressure overload: from hypertrophy to failure. Cardiovascular research 2017, 113, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Piao, L.; Fang, Y.-H.; Parikh, K.S.; Ryan, J.J.; D'Souza, K.M.; Theccanat, T.; Toth, P.T.; Pogoriler, J.; Paul, J.; Blaxall, B.C. GRK2-mediated inhibition of adrenergic and dopaminergic signaling in right ventricular hypertrophy: therapeutic implications in pulmonary hypertension. Circulation 2012, 126, 2859–2869. [Google Scholar] [CrossRef]
- Ghio, S.; Klersy, C.; Magrini, G.; D'Armini, A.M.; Scelsi, L.; Raineri, C.; Pasotti, M.; Serio, A.; Campana, C.; Viganò, M. Prognostic relevance of the echocardiographic assessment of right ventricular function in patients with idiopathic pulmonary arterial hypertension. International journal of cardiology 2010, 140, 272–278. [Google Scholar] [CrossRef]
- Pirat, B.; McCulloch, M.L.; Zoghbi, W.A. Evaluation of global and regional right ventricular systolic function in patients with pulmonary hypertension using a novel speckle tracking method. The American journal of cardiology 2006, 98, 699–704. [Google Scholar] [CrossRef]
- Haeck, M.L.; Scherptong, R.W.; Marsan, N.A.; Holman, E.R.; Schalij, M.J.; Bax, J.J.; Vliegen, H.W.; Delgado, V. Prognostic value of right ventricular longitudinal peak systolic strain in patients with pulmonary hypertension. Circulation: Cardiovascular Imaging 2012, 5, 628–636. [Google Scholar] [CrossRef]
- Katz, J.; Whang, J.; Boxt, L.M.; Barst, R.J. Estimation of right ventricular mass in normal subjects and in patients with primary pulmonary hypertension by nuclear magnetic resonance imaging. Journal of the American College of Cardiology 1993, 21, 1475–1481. [Google Scholar] [CrossRef] [PubMed]
- Oosterhof, T.; Tulevski, I.I.; Vliegen, H.W.; Spijkerboer, A.M.; Mulder, B.J. Effects of volume and/or pressure overload secondary to congenital heart disease (tetralogy of fallot or pulmonary stenosis) on right ventricular function using cardiovascular magnetic resonance and B-type natriuretic peptide levels. The American journal of cardiology 2006, 97, 1051–1055. [Google Scholar] [CrossRef] [PubMed]
- Usui, S.-i.; Yao, A.; Hatano, M.; Kohmoto, O.; Takahashi, T.; Nagai, R.; Kinugawa, K. Upregulated neurohumoral factors are associated with left ventricular remodeling and poor prognosis in rats with monocrotaline-induced pulmonary arterial hypertension. Circulation Journal 2006, 70, 1208–1215. [Google Scholar] [CrossRef]
- Friedberg, M.K.; Cho, M.-Y.; Li, J.; Assad, R.S.; Sun, M.; Rohailla, S.; Honjo, O.; Apitz, C.; Redington, A.N. Adverse biventricular remodeling in isolated right ventricular hypertension is mediated by increased transforming growth Factor–β1 signaling and is abrogated by angiotensin receptor blockade. American journal of respiratory cell and molecular biology 2013, 49, 1019–1028. [Google Scholar] [CrossRef]
- Guazzi, M.; Bandera, F.; Pelissero, G.; Castelvecchio, S.; Menicanti, L.; Ghio, S.; Temporelli, P.; Arena, R. Tricuspid annular plane systolic excursion and pulmonary arterial systolic pressure relationship in heart failure: an index of right ventricular contractile function and prognosis. American Journal of Physiology-Heart and Circulatory Physiology 2013, 305, H1373–H1381. [Google Scholar] [CrossRef]
- Stevens, G.R.; Garcia-Alvarez, A.; Sahni, S.; Garcia, M.J.; Fuster, V.; Sanz, J. RV dysfunction in pulmonary hypertension is independently related to pulmonary artery stiffness. JACC: Cardiovascular Imaging 2012, 5, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Dambrauskaite, V.; Delcroix, M.; Claus, P.; Herbots, L.; Palecek, T.; D’hooge, J.; Bijnens, B.; Rademakers, F.; Sutherland, G.R. The evaluation of pulmonary hypertension using right ventricular myocardial isovolumic relaxation time. Journal of the American Society of Echocardiography 2005, 18, 1113–1120. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, Y.; Yan, C.; He, J.; Xiong, C.; Zhao, S.; Fang, W. Evaluation of right ventricular volume and ejection fraction by gated 18F-FDG PET in patients with pulmonary hypertension: comparison with cardiac MRI and CT. Journal of Nuclear Cardiology 2013, 20, 242–252. [Google Scholar] [CrossRef]
- Tello, K.; Richter, M.J.; Axmann, J.; Buhmann, M.; Seeger, W.; Naeije, R.; Ghofrani, H.A.; Gall, H. More on single-beat estimation of right ventriculoarterial coupling in pulmonary arterial hypertension. American journal of respiratory and critical care medicine 2018, 198, 816–818. [Google Scholar] [CrossRef]
- Ghuysen, A.; Lambermont, B.; Kolh, P.; Tchana-Sato, V.; Magis, D.; Gerard, P.; Mommens, V.; Janssen, N.; Desaive, T.; D'Orio, V. Alteration of right ventricular-pulmonary vascular coupling in a porcine model of progressive pressure overloading. Shock 2008, 29, 197–204. [Google Scholar] [CrossRef]
- Tello, K.; Richter, M.J.; Yogeswaran, A.; Ghofrani, H.A.; Naeije, R.; Vanderpool, R.; Gall, H.; Tedford, R.J.; Seeger, W.; Lahm, T. Sex differences in right ventricular–pulmonary arterial coupling in pulmonary arterial hypertension. American journal of respiratory and critical care medicine 2020, 202, 1042–1046. [Google Scholar] [CrossRef]
- Sachdev, A.; Villarraga, H.R.; Frantz, R.P.; McGoon, M.D.; Hsiao, J.-F.; Maalouf, J.F.; Ammash, N.M.; McCully, R.B.; Miller, F.A.; Pellikka, P.A. Right ventricular strain for prediction of survival in patients with pulmonary arterial hypertension. Chest 2011, 139, 1299–1309. [Google Scholar] [CrossRef]
- Koestenberger, M.; Apitz, C.; Abdul-Khaliq, H.; Hansmann, G. Transthoracic echocardiography for the evaluation of children and adolescents with suspected or confirmed pulmonary hypertension. Expert consensus statement on the diagnosis and treatment of paediatric pulmonary hypertension. The European Paediatric Pulmonary Vascular Disease Network, endorsed by ISHLT and DGPK. Heart 2016, 102, ii14–ii22. [Google Scholar] [CrossRef]
- Badagliacca, R.; Poscia, R.; Pezzuto, B.; Nocioni, M.; Mezzapesa, M.; Francone, M.; Giannetta, E.; Papa, S.; Gambardella, C.; Sciomer, S. Right ventricular remodeling in idiopathic pulmonary arterial hypertension: adaptive versus maladaptive morphology. The Journal of Heart and Lung Transplantation 2015, 34, 395–403. [Google Scholar] [CrossRef]
- Naeije, R. Assessment of right ventricular function in pulmonary hypertension. Current hypertension reports 2015, 17, 35. [Google Scholar] [CrossRef] [PubMed]
- Tello, K.; Seeger, W.; Naeije, R.; Vanderpool, R.; Ghofrani, H.A.; Richter, M.; Tedford, R.J.; Bogaard, H.J. Right heart failure in pulmonary hypertension: diagnosis and new perspectives on vascular and direct right ventricular treatment. British journal of pharmacology 2021, 178, 90–107. [Google Scholar] [CrossRef]
- Philip, J.L.; Chesler, N.C. Know Your Limitations: Assumptions in the Single-Beat Method for Estimating Right Ventricular–Pulmonary Vascular Coupling; American Thoracic Society, 2018; pp. 707–709. [Google Scholar] [CrossRef] [PubMed]
- Murch, S.D.; Gerche, A.L.; Roberts, T.J.; Prior, D.L.; MacIsaac, A.I.; Burns, A.T. Abnormal right ventricular relaxation in pulmonary hypertension. Pulmonary circulation 2015, 5, 370–375. [Google Scholar] [CrossRef]
- French, S.; Amsallem, M.; Ouazani, N.; Li, S.; Kudelko, K.; Zamanian, R.T.; Haddad, F.; Chung, L. Non-invasive right ventricular load adaptability indices in patients with scleroderma-associated pulmonary arterial hypertension. Pulmonary circulation 2018, 8, 2045894018788268. [Google Scholar] [CrossRef] [PubMed]
- Browning, J.; Hertzberg, J.; Schroeder, J.; Fenster, B. 4D Flow Assessment of Vorticity in Right Ventricular Diastolic Dysfunction. Bioengineering 2017, 4, 30. [Google Scholar] [CrossRef]
- Berthelot, E.; Montani, D.; Algalarrondo, V.; Dreyfuss, C.; Rifai, R.; Benmalek, A.; Jais, X.; Bouchachi, A.; Savale, L.; Simonneau, G. A clinical and echocardiographic score to identify pulmonary hypertension due to HFpEF. Journal of Cardiac Failure 2017, 23, 29–35. [Google Scholar] [CrossRef]
- Swift, A.J.; Capener, D.; Johns, C.; Hamilton, N.; Rothman, A.; Elliot, C.; Condliffe, R.; Charalampopoulos, A.; Rajaram, S.; Lawrie, A. Magnetic resonance imaging in the prognostic evaluation of patients with pulmonary arterial hypertension. American journal of respiratory and critical care medicine 2017, 196, 228–239. [Google Scholar] [CrossRef]
- Swift, A.J.; Rajaram, S.; Campbell, M.J.; Hurdman, J.; Thomas, S.; Capener, D.; Elliot, C.; Condliffe, R.; Wild, J.M.; Kiely, D.G. Prognostic value of cardiovascular magnetic resonance imaging measurements corrected for age and sex in idiopathic pulmonary arterial hypertension. Circulation: Cardiovascular Imaging 2014, 7, 100–106. [Google Scholar] [CrossRef]
- Prins, K.W.; Rose, L.; Archer, S.L.; Pritzker, M.; Weir, E.K.; Olson, M.D.; Thenappan, T. Clinical determinants and prognostic implications of right ventricular dysfunction in pulmonary hypertension caused by chronic lung disease. Journal of the American Heart Association 2019, 8, e011464. [Google Scholar] [CrossRef]
- Vanderpool, R.R.; Desai, A.A.; Knapp, S.M.; Simon, M.A.; Abidov, A.; Yuan, J.X.-J.; Garcia, J.G.; Hansen, L.M.; Knoper, S.R.; Naeije, R. How prostacyclin therapy improves right ventricular function in pulmonary arterial hypertension. European Respiratory Journal 2017, 50. [Google Scholar] [CrossRef]
- Vanderpool, R.R.; Rischard, F.; Naeije, R.; Hunter, K.; Simon, M.A. Simple functional imaging of the right ventricle in pulmonary hypertension: can right ventricular ejection fraction be improved? International journal of cardiology 2016, 223, 93–94. [Google Scholar] [CrossRef] [PubMed]
- Brittain, E.L.; Talati, M.; Fessel, J.P.; Zhu, H.; Penner, N.; Calcutt, M.W.; West, J.D.; Funke, M.; Lewis, G.D.; Gerszten, R.E. Fatty acid metabolic defects and right ventricular lipotoxicity in human pulmonary arterial hypertension. Circulation 2016, 133, 1936–1944. [Google Scholar] [CrossRef] [PubMed]
- Brewis, M.J.; Bellofiore, A.; Vanderpool, R.R.; Chesler, N.C.; Johnson, M.K.; Naeije, R.; Peacock, A.J. Imaging right ventricular function to predict outcome in pulmonary arterial hypertension. International journal of cardiology 2016, 218, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.J.; Archer, S.L. The right ventricle in pulmonary arterial hypertension: disorders of metabolism, angiogenesis and adrenergic signaling in right ventricular failure. Circulation research 2014, 115, 176–188. [Google Scholar] [CrossRef]
- Dawes, T.J.; de Marvao, A.; Shi, W.; Fletcher, T.; Watson, G.M.; Wharton, J.; Rhodes, C.J.; Howard, L.S.; Gibbs, J.S.R.; Rueckert, D. Machine learning of three-dimensional right ventricular motion enables outcome prediction in pulmonary hypertension: a cardiac MR imaging study. Radiology 2017, 283, 381–390. [Google Scholar] [CrossRef]
- Simon, M.A.; Pinsky, M.R. Right ventricular dysfunction and failure in chronic pressure overload. Cardiology research and practice 2011, 2011, 568095. [Google Scholar] [CrossRef]
- Piao, L.; Fang, Y.-H.; Parikh, K.; Ryan, J.J.; Toth, P.T.; Archer, S.L. Cardiac glutaminolysis: a maladaptive cancer metabolism pathway in the right ventricle in pulmonary hypertension. Journal of molecular medicine 2013, 91, 1185–1197. [Google Scholar] [CrossRef]
- Addetia, K.; Bhave, N.M.; Tabit, C.E.; Gomberg-Maitland, M.; Freed, B.H.; Dill, K.E.; Lang, R.M.; Mor-Avi, V.; Patel, A.R. Sample size and cost analysis for pulmonary arterial hypertension drug trials using various imaging modalities to assess right ventricular size and function end points. Circulation: Cardiovascular Imaging 2014, 7, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Archer, S.L.; Fang, Y.-H.; Ryan, J.J.; Piao, L. Metabolism and bioenergetics in the right ventricle and pulmonary vasculature in pulmonary hypertension. Pulmonary circulation 2013, 3, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Bogaard, H.J.; Abe, K.; Noordegraaf, A.V.; Voelkel, N.F. The right ventricle under pressure: cellular and molecular mechanisms of right-heart failure in pulmonary hypertension. Chest 2009, 135, 794–804. [Google Scholar] [CrossRef]
- Matthews, J.C.; McLaughlin, V. Acute right ventricular failure in the setting of acute pulmonary embolism or chronic pulmonary hypertension: a detailed review of the pathophysiology, diagnosis, and management. Current cardiology reviews 2008, 4, 49–59. [Google Scholar] [CrossRef]
- Wang, L.; Li, W.; Yang, Y.; Wu, W.; Cai, Q.; Ma, X.; Xiong, C.; He, J.; Fang, W. Quantitative assessment of right ventricular glucose metabolism in idiopathic pulmonary arterial hypertension patients: a longitudinal study. European Heart Journal-Cardiovascular Imaging 2016, 17, 1161–1168. [Google Scholar] [CrossRef]
- Hsu, S.; Houston, B.A.; Tampakakis, E.; Bacher, A.C.; Rhodes, P.S.; Mathai, S.C.; Damico, R.L.; Kolb, T.M.; Hummers, L.K.; Shah, A.A. Right ventricular functional reserve in pulmonary arterial hypertension. Circulation 2016, 133, 2413–2422. [Google Scholar] [CrossRef]
- Hsu, S.; Kokkonen-Simon, K.M.; Kirk, J.A.; Kolb, T.M.; Damico, R.L.; Mathai, S.C.; Mukherjee, M.; Shah, A.A.; Wigley, F.M.; Margulies, K.B. Right ventricular myofilament functional differences in humans with systemic sclerosis–associated versus idiopathic pulmonary arterial hypertension. Circulation 2018, 137, 2360–2370. [Google Scholar] [CrossRef] [PubMed]
- Ohira, H.; deKemp, R.; Pena, E.; Davies, R.A.; Stewart, D.J.; Chandy, G.; Contreras-Dominguez, V.; Dennie, C.; Mc Ardle, B.; Mc Klein, R. Shifts in myocardial fatty acid and glucose metabolism in pulmonary arterial hypertension: a potential mechanism for a maladaptive right ventricular response. European Journal of Echocardiography 2015, 17, 1424–1431. [Google Scholar] [CrossRef]
- Benza, R.; Biederman, R.; Murali, S.; Gupta, H. Role of cardiac magnetic resonance imaging in the management of patients with pulmonary arterial hypertension. Journal of the American College of Cardiology 2008, 52, 1683–1692. [Google Scholar] [CrossRef]
- Grothues, F.; Moon, J.C.; Bellenger, N.G.; Smith, G.S.; Klein, H.U.; Pennell, D.J. Interstudy reproducibility of right ventricular volumes, function, and mass with cardiovascular magnetic resonance. American heart journal 2004, 147, 218–223. [Google Scholar]
- van der Bruggen, C.E.; Happé, C.M.; Dorfmüller, P.; Trip, P.; Spruijt, O.A.; Rol, N.; Hoevenaars, F.P.; Houweling, A.C.; Girerd, B.; Marcus, J.T. Bone morphogenetic protein receptor type 2 mutation in pulmonary arterial hypertension: a view on the right ventricle. Circulation 2016, 133, 1747–1760. [Google Scholar] [CrossRef]
- Fang, Y.-H.; Piao, L.; Hong, Z.; Toth, P.T.; Marsboom, G.; Bache-Wiig, P.; Rehman, J.; Archer, S.L. Therapeutic inhibition of fatty acid oxidation in right ventricular hypertrophy: exploiting Randle’s cycle. Journal of molecular medicine 2012, 90, 31–43. [Google Scholar] [CrossRef]
- Cavasin, M.A.; Demos-Davies, K.; Horn, T.R.; Walker, L.A.; Lemon, D.D.; Birdsey, N.; Weiser-Evans, M.C.; Harral, J.; Irwin, D.C.; Anwar, A. Selective class I histone deacetylase inhibition suppresses hypoxia-induced cardiopulmonary remodeling through an antiproliferative mechanism. Circulation research 2012, 110, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Tual, L.; Morel, O.-E.; Favret, F.; Fouillit, M.; Guernier, C.; Buvry, A.; Germain, L.; Dhonneur, G.; Bernaudin, J.-F.; Richalet, J.-P. Carvedilol inhibits right ventricular hypertrophy induced by chronic hypobaric hypoxia. Pflügers Archiv 2006, 452, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Engelfriet, P.M.; Duffels, M.G.; Møller, T.; Boersma, E.; Tijssen, J.G.; Thaulow, E.; Gatzoulis, M.A.; Mulder, B.J. Pulmonary arterial hypertension in adults born with a heart septal defect: the Euro Heart Survey on adult congenital heart disease. Heart 2007, 93, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Nootens, M.; Kaufmann, E.; Rector, T.; Toher, C.; Judd, D.; Francis, G.S.; Rich, S. Neurohormonal activation in patients with right ventricular failure from pulmonary hypertension: relation to hemodynamic variables and endothelin levels. Journal of the American College of Cardiology 1995, 26, 1581–1585. [Google Scholar] [CrossRef]
- Vogel-Claussen, J.; Skrok, J.; Shehata, M.L.; Singh, S.; Sibley, C.T.; Boyce, D.M.; Lechtzin, N.; Girgis, R.E.; Mathai, S.C.; Goldstein, T.A. Right and left ventricular myocardial perfusion reserves correlate with right ventricular function and pulmonary hemodynamics in patients with pulmonary arterial hypertension. Radiology 2011, 258, 119–127. [Google Scholar] [CrossRef]
- Hopkins, W.E.; Waggoner, A.D. Severe pulmonary hypertension without right ventricular failure: the unique hearts of patients with Eisenmenger syndrome. The American journal of cardiology 2002, 89, 34–38. [Google Scholar] [CrossRef]
- Gómez, A.; Bialostozky, D.; Zajarias, A.; Santos, E.; Palomar, A.; Martínez, M.L.; Sandoval, J. Right ventricular ischemia in patients with primary pulmonary hypertension. Journal of the American College of Cardiology 2001, 38, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- Masithulela, F. Bi-ventricular finite element model of right ventricle overload in the healthy rat heart. Bio-medical materials and engineering 2016, 27, 507–525. [Google Scholar] [CrossRef]
- Dellegrottaglie, S.; Sanz, J.; Poon, M.; Viles-Gonzalez, J.F.; Sulica, R.; Goyenechea, M.; Macaluso, F.; Fuster, V.; Rajagopalan, S. Pulmonary hypertension: accuracy of detection with left ventricular septal-to–free wall curvature ratio measured at cardiac MR. Radiology 2007, 243, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Puwanant, S.; Priester, T.C.; Mookadam, F.; Bruce, C.J.; Redfield, M.M.; Chandrasekaran, K. Right ventricular function in patients with preserved and reduced ejection fraction heart failure. European Journal of Echocardiography 2009, 10, 733–737. [Google Scholar] [CrossRef]
- Kaufman, B.D.; Desai, M.; Reddy, S.; Osorio, J.C.; Chen, J.M.; Mosca, R.S.; Ferrante, A.W.; Mital, S. Genomic profiling of left and right ventricular hypertrophy in congenital heart disease. Journal of cardiac failure 2008, 14, 760–767. [Google Scholar] [CrossRef]
- Reddy, S.; Zhao, M.; Hu, D.-Q.; Fajardo, G.; Hu, S.; Ghosh, Z.; Rajagopalan, V.; Wu, J.C.; Bernstein, D. Dynamic microRNA expression during the transition from right ventricular hypertrophy to failure. Physiological genomics 2012, 44, 562–575. [Google Scholar] [CrossRef]
- Paulin, R.; Sutendra, G.; Gurtu, V.; Dromparis, P.; Haromy, A.; Provencher, S.; Bonnet, S.; Michelakis, E.D. A miR-208–Mef2 axis drives the decompensation of right ventricular function in pulmonary hypertension. Circulation research 2015, 116, 56–69. [Google Scholar] [CrossRef]
- Rajagopalan, V.; Zhao, M.; Reddy, S.; Fajardo, G.; Wang, X.; Dewey, S.; Gomes, A.V.; Bernstein, D. Altered ubiquitin-proteasome signaling in right ventricular hypertrophy and failure. American Journal of Physiology-Heart and Circulatory Physiology 2013, 305, H551–H562. [Google Scholar] [CrossRef]
- Hoeper, M.M.; Barberà, J.A.; Channick, R.N.; Hassoun, P.M.; Lang, I.M.; Manes, A.; Martinez, F.J.; Naeije, R.; Olschewski, H.; Pepke-Zaba, J. Diagnosis, assessment, and treatment of non-pulmonary arterial hypertension pulmonary hypertension. Journal of the American College of Cardiology 2009, 54, S85–S96. [Google Scholar] [CrossRef] [PubMed]
- Hoeper, M.M.; Bogaard, H.J.; Condliffe, R.; Frantz, R.; Khanna, D.; Kurzyna, M.; Langleben, D.; Manes, A.; Satoh, T.; Torres, F. Definitions and diagnosis of pulmonary hypertension. Journal of the American College of Cardiology 2013, 62, D42–D50. [Google Scholar] [CrossRef] [PubMed]
- Hoeper, M.M.; Humbert, M.; Souza, R.; Idrees, M.; Kawut, S.M.; Sliwa-Hahnle, K.; Jing, Z.-C.; Gibbs, J.S.R. A global view of pulmonary hypertension. The Lancet Respiratory Medicine 2016, 4, 306–322. [Google Scholar] [CrossRef]
- Bellofiore, A.; Chesler, N.C. Methods for measuring right ventricular function and hemodynamic coupling with the pulmonary vasculature. Annals of biomedical engineering 2013, 41, 1384–1398. [Google Scholar] [CrossRef]
- Fine, N.M.; Chen, L.; Bastiansen, P.M.; Frantz, R.P.; Pellikka, P.A.; Oh, J.K.; Kane, G.C. Outcome prediction by quantitative right ventricular function assessment in 575 subjects evaluated for pulmonary hypertension. Circulation: Cardiovascular Imaging 2013, 6, 711–721. [Google Scholar] [CrossRef]
- Singh, A.; Huang, X.; Dai, L.; Wyler, D.; Alfirevic, A.; Blackstone, E.H.; Pettersson, G.B.; Duncan, A.E. Right ventricular function is reduced during cardiac surgery independent of procedural characteristics, reoperative status, or pericardiotomy. The Journal of Thoracic and Cardiovascular Surgery 2020, 159, 1430–1438. e1434. [Google Scholar] [CrossRef]
- Tamborini, G.; Muratori, M.; Brusoni, D.; Celeste, F.; Maffessanti, F.; Caiani, E.G.; Alamanni, F.; Pepi, M. Is right ventricular systolic function reduced after cardiac surgery? A two-and three-dimensional echocardiographic study. European Journal of Echocardiography 2009, 10, 630–634. [Google Scholar] [CrossRef]
- Kind, T.; Mauritz, G.-J.; Marcus, J.T.; van de Veerdonk, M.; Westerhof, N.; Vonk-Noordegraaf, A. Right ventricular ejection fraction is better reflected by transverse rather than longitudinal wall motion in pulmonary hypertension. Journal of Cardiovascular Magnetic Resonance 2010, 12, 35. [Google Scholar] [CrossRef] [PubMed]
- Maughan, W.L.; Shoukas, A.A.; Sagawa, K.; Weisfeldt, M.L. Instantaneous pressure-volume relationship of the canine right ventricle. Circulation research 1979, 44, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Brimioulle, S.; Wauthy, P.; Ewalenko, P.; Rondelet, B.; Vermeulen, F.; Kerbaul, F.; Naeije, R. Single-beat estimation of right ventricular end-systolic pressure-volume relationship. American Journal of Physiology-Heart and Circulatory Physiology 2003, 284, H1625–H1630. [Google Scholar] [CrossRef]
- Burkett, D.A.; Patel, S.S.; Mertens, L.; Friedberg, M.K.; Ivy, D.D. Relationship between left ventricular geometry and invasive hemodynamics in pediatric pulmonary hypertension. Circulation: Cardiovascular Imaging 2020, 13, e009825. [Google Scholar] [CrossRef]
- Burkett, D.A.; Slorach, C.; Patel, S.S.; Redington, A.N.; Ivy, D.D.; Mertens, L.; Younoszai, A.K.; Friedberg, M.K. Left ventricular myocardial function in children with pulmonary hypertension: relation to right ventricular performance and hemodynamics. Circulation: Cardiovascular Imaging 2015, 8, e003260. [Google Scholar] [CrossRef] [PubMed]
- Tedford, R.J. Determinants of right ventricular afterload (2013 Grover Conference series). Pulmonary circulation 2014, 4, 211–219. [Google Scholar] [CrossRef]
- Tedford, R.J.; Mudd, J.O.; Girgis, R.E.; Mathai, S.C.; Zaiman, A.L.; Housten-Harris, T.; Boyce, D.; Kelemen, B.W.; Bacher, A.C.; Shah, A.A. Right ventricular dysfunction in systemic sclerosis–associated pulmonary arterial hypertension. Circulation: Heart Failure 2013, 6, 953–963. [Google Scholar] [CrossRef]
- Bréchot, N.; Gambotti, L.; Lafitte, S.; Roudaut, R. Usefulness of right ventricular isovolumic relaxation time in predicting systolic pulmonary artery pressure. European Journal of Echocardiography 2008, 9, 547–554. [Google Scholar] [CrossRef]
- Humbert, M.; Kovacs, G.; Hoeper, M.M.; Badagliacca, R.; Berger, R.M.; Brida, M.; Carlsen, J.; Coats, A.J.; Escribano-Subias, P.; Ferrari, P. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: Developed by the task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS). Endorsed by the International Society for Heart and Lung Transplantation (ISHLT) and the European Reference Network on rare respiratory diseases (ERN-LUNG). European heart journal 2022, 43, 3618–3731. [Google Scholar]
- Iles, L.; Pfluger, H.; Phrommintikul, A.; Cherayath, J.; Aksit, P.; Gupta, S.N.; Kaye, D.M.; Taylor, A.J. Evaluation of diffuse myocardial fibrosis in heart failure with cardiac magnetic resonance contrast-enhanced T1 mapping. Journal of the American College of Cardiology 2008, 52, 1574–1580. [Google Scholar] [CrossRef]
- Acosta, F.; Sansano, T.; Palenciano, C.; Falcon, L.; Domenech, P.; Robles, R.; Bueno, F.; Ramirez, P.; Parrilla, P. Effects of dobutamine on right ventricular function and pulmonary circulation in pulmonary hypertension during liver transplantation. In Proceedings of the Transplantation proceedings, 2005; pp. 3869–3870. [Google Scholar]
- Verbelen, T.; Godinas, L.; Maleux, G.; Coolen, J.; Claessen, G.; Belge, C.; Meyns, B.; Delcroix, M. Chronic thromboembolic pulmonary hypertension: diagnosis, operability assessment and patient selection for pulmonary endarterectomy. Annals of cardiothoracic surgery 2022, 11, 82. [Google Scholar] [CrossRef]
- Bhullar, S.K.; Dhalla, N.S. Angiotensin II-induced signal transduction mechanisms for cardiac hypertrophy. Cells 2022, 11, 3336. [Google Scholar] [CrossRef]
- Kim, D.; Jang, W.J.; Park, T.K.; Cho, Y.H.; Choi, J.-O.; Jeon, E.-S.; Yang, J.H. Echocardiographic predictors of successful extracorporeal membrane oxygenation weaning after refractory cardiogenic shock. Journal of the American Society of Echocardiography 2021, 34, 414–422. e414. [Google Scholar] [CrossRef]
- Kim, R.J.; Wu, E.; Rafael, A.; Chen, E.-L.; Parker, M.A.; Simonetti, O.; Klocke, F.J.; Bonow, R.O.; Judd, R.M. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. New England Journal of Medicine 2000, 343, 1445–1453. [Google Scholar] [CrossRef] [PubMed]
- Rain, S.; Handoko, M.L.; Trip, P.; Gan, C.T.-J.; Westerhof, N.; Stienen, G.J.; Paulus, W.J.; Ottenheijm, C.A.; Marcus, J.T.; Dorfmüller, P. Right ventricular diastolic impairment in patients with pulmonary arterial hypertension. Circulation 2013, 128, 2016–2025. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.Y.; Ruiter, G.; Lubberink, M.; Raijmakers, P.G.; Knaapen, P.; Marcus, J.T.; Boonstra, A.; Lammertsma, A.A.; Westerhof, N.; van der Laarse, W.J. Right ventricular failure in idiopathic pulmonary arterial hypertension is associated with inefficient myocardial oxygen utilization. Circulation: Heart Failure 2011, 4, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.T.; Pickard, S.S.; Chan, F.P.; Tsao, P.S.; Taylor, C.A.; Feinstein, J.A. Wall shear stress is decreased in the pulmonary arteries of patients with pulmonary arterial hypertension: an image-based, computational fluid dynamics study. Pulmonary circulation 2012, 2, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Ltaief, Z.; Yerly, P.; Liaudet, L. Pulmonary hypertension in left heart diseases: pathophysiology, hemodynamic assessment and therapeutic management. International journal of molecular sciences 2023, 24, 9971. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.P.; Bittner, H.B.; Davis, R.D., Jr.; Van Trigt, P., III. Milrinone improves pulmonary hemodynamics and right ventricular function in chronic pulmonary hypertension. The Annals of thoracic surgery 1997, 63, 814–821. [Google Scholar] [CrossRef]
- Wiklund, A.; Kylhammar, D.; Rådegran, G. Levosimendan attenuates hypoxia-induced pulmonary hypertension in a porcine model. Journal of Cardiovascular Pharmacology 2012, 59, 441–449. [Google Scholar] [CrossRef]
- Gorter, T.M.; Hoendermis, E.S.; van Veldhuisen, D.J.; Voors, A.A.; Lam, C.S.; Geelhoed, B.; Willems, T.P.; van Melle, J.P. Right ventricular dysfunction in heart failure with preserved ejection fraction: a systematic review and meta-analysis. European journal of heart failure 2016, 18, 1472–1487. [Google Scholar] [CrossRef]
- Zakeri, R.; Mohammed, S.F. Epidemiology of right ventricular dysfunction in heart failure with preserved ejection fraction. Current heart failure reports 2015, 12, 295–301. [Google Scholar] [CrossRef]
- Raina, A.; Meeran, T. Right ventricular dysfunction and its contribution to morbidity and mortality in left ventricular heart failure. Current Heart Failure Reports 2018, 15, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Hansmann, G.; Koestenberger, M.; Alastalo, T.-P.; Apitz, C.; Austin, E.D.; Bonnet, D.; Budts, W.; D'Alto, M.; Gatzoulis, M.A.; Hasan, B.S. 2019 updated consensus statement on the diagnosis and treatment of pediatric pulmonary hypertension: The European Pediatric Pulmonary Vascular Disease Network (EPPVDN), endorsed by AEPC, ESPR and ISHLT; Elsevier, 2019; pp. 879–901. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.; Ha, R.; Banerjee, D. Pulmonary artery pulsatility index predicts right ventricular failure after left ventricular assist device implantation. The Journal of Heart and Lung Transplantation 2016, 35, 67–73. [Google Scholar] [CrossRef]
- Masarone, D.; Melillo, E.; Errigo, V.; Martucci, M.L.; Pacileo, R.; Pollesello, P.; Petraio, A.; Pacileo, G. Hemodynamic effects of levosimendan in outpatients with advanced heart failure: an echocardiographic pilot study. Journal of Cardiovascular Pharmacology 2022, 79, e36–e40. [Google Scholar] [CrossRef] [PubMed]
- Lahm, T.; Douglas, I.S.; Archer, S.L.; Bogaard, H.J.; Chesler, N.C.; Haddad, F.; Hemnes, A.R.; Kawut, S.M.; Kline, J.A.; Kolb, T.M. Assessment of right ventricular function in the research setting: knowledge gaps and pathways forward. An Official American Thoracic Society Research Statement. American journal of respiratory and critical care medicine 2018, 198, e15–e43. [Google Scholar] [CrossRef]
- Vanderpool, R.R.; Saul, M.; Nouraie, M.; Gladwin, M.T.; Simon, M.A. Association between hemodynamic markers of pulmonary hypertension and outcomes in heart failure with preserved ejection fraction. JAMA cardiology 2018, 3, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Fenster, B.E.; Browning, J.; Schroeder, J.D.; Schafer, M.; Podgorski, C.A.; Smyser, J.; Silveira, L.J.; Buckner, J.K.; Hertzberg, J.R. Vorticity is a marker of right ventricular diastolic dysfunction. American Journal of Physiology-Heart and Circulatory Physiology 2015, 309, H1087–H1093. [Google Scholar] [CrossRef]
- Legchenko, E.; Chouvarine, P.; Borchert, P.; Fernandez-Gonzalez, A.; Snay, E.; Meier, M.; Maegel, L.; Mitsialis, S.A.; Rog-Zielinska, E.A.; Kourembanas, S. PPARγ agonist pioglitazone reverses pulmonary hypertension and prevents right heart failure via fatty acid oxidation. Science translational medicine 2018, 10, eaao0303. [Google Scholar] [CrossRef]
- Culley, M.K.; Chan, S.Y. Mitochondrial metabolism in pulmonary hypertension: beyond mountains there are mountains. The Journal of clinical investigation 2018, 128, 3704–3715. [Google Scholar] [CrossRef]
- Aschauer, S.; Kammerlander, A.A.; Zotter-Tufaro, C.; Ristl, R.; Pfaffenberger, S.; Bachmann, A.; Duca, F.; Marzluf, B.A.; Bonderman, D.; Mascherbauer, J. The right heart in heart failure with preserved ejection fraction: insights from cardiac magnetic resonance imaging and invasive haemodynamics. European journal of heart failure 2016, 18, 71–80. [Google Scholar] [CrossRef]
- Panagiotou, M.; Vogiatzis, I.; Louvaris, Z.; Jayasekera, G.; MacKenzie, A.; Mcglinchey, N.; Baker, J.S.; Church, A.C.; Peacock, A.J.; Johnson, M.K. Near infrared spectroscopy for the assessment of peripheral tissue oxygenation in pulmonary arterial hypertension. The European Respiratory Journal 2016, 48, 1224–1227. [Google Scholar] [CrossRef]
- Frost, A.; Badesch, D.; Gibbs, J.S.R.; Gopalan, D.; Khanna, D.; Manes, A.; Oudiz, R.; Satoh, T.; Torres, F.; Torbicki, A. Diagnosis of pulmonary hypertension. European Respiratory Journal 2019, 53. [Google Scholar] [CrossRef] [PubMed]
- Tabassian, M.; Sunderji, I.; Erdei, T.; Sanchez-Martinez, S.; Degiovanni, A.; Marino, P.; Fraser, A.G.; D'hooge, J. Diagnosis of heart failure with preserved ejection fraction: machine learning of spatiotemporal variations in left ventricular deformation. Journal of the American society of echocardiography 2018, 31, 1272–1284. e1279. [Google Scholar] [CrossRef]
- Pinsky, M.R. The right ventricle: interaction with the pulmonary circulation. Critical Care 2016, 20, 266. [Google Scholar] [CrossRef] [PubMed]
- Vieillard-Baron, A.; Naeije, R.; Haddad, F.; Bogaard, H.; Bull, T.; Fletcher, N.; Lahm, T.; Magder, S.; Orde, S.; Schmidt, G. Diagnostic workup, etiologies and management of acute right ventricle failure: a state-of-the-art paper. Intensive care medicine 2018, 44, 774–790. [Google Scholar] [CrossRef] [PubMed]
- Lyon, R.C.; Zanella, F.; Omens, J.H.; Sheikh, F. Mechanotransduction in cardiac hypertrophy and failure. Circulation research 2015, 116, 1462–1476. [Google Scholar] [CrossRef]
- Bitker, L.; Sens, F.; Payet, C.; Turquier, S.; Duclos, A.; Cottin, V.; Juillard, L. Presence of kidney disease as an outcome predictor in patients with pulmonary arterial hypertension. American journal of nephrology 2018, 47, 134–143. [Google Scholar] [CrossRef]
- Medvedofsky, D.; Mor-Avi, V.; Amzulescu, M.; Fernandez-Golfin, C.; Hinojar, R.; Monaghan, M.J.; Otani, K.; Reiken, J.; Takeuchi, M.; Tsang, W. Three-dimensional echocardiographic quantification of the left-heart chambers using an automated adaptive analytics algorithm: multicentre validation study. European Heart Journal-Cardiovascular Imaging 2018, 19, 47–58. [Google Scholar] [CrossRef]
- Brugger, N.; Lichtblau, M.; Maeder, M.T.; Müller, H.; Pellaton, C.; Yerly, P. Two-dimensional transthoracic echocardiography at rest for the diagnosis, screening and management of pulmonary hypertension. Swiss medical weekly 2021, 151, w20486. [Google Scholar] [CrossRef]
- Schmeißer, A.; Rauwolf, T.; Groscheck, T.; Fischbach, K.; Kropf, S.; Luani, B.; Tanev, I.; Hansen, M.; Meißler, S.; Schäfer, K. Predictors and prognosis of right ventricular function in pulmonary hypertension due to heart failure with reduced ejection fraction. ESC heart failure 2021, 8, 2968–2981. [Google Scholar] [CrossRef]
- He, Q.; Lin, Y.; Zhu, Y.; Gao, L.; Ji, M.; Zhang, L.; Xie, M.; Li, Y. Clinical usefulness of right ventricle–pulmonary artery coupling in cardiovascular disease. Journal of clinical medicine 2023, 12, 2526. [Google Scholar] [CrossRef] [PubMed]
- Leather, H.A.; Segers, P.; Berends, N.; Vandermeersch, E.; Wouters, P.F. Effects of vasopressin on right ventricular function in an experimental model of acute pulmonary hypertension. Critical care medicine 2002, 30, 2548–2552. [Google Scholar] [CrossRef]
- Vildbrad, M.D.; Andersen, A.; Holmboe, S.; Ringgaard, S.; Nielsen, J.M.; Nielsen-Kudsk, J.E. Acute effects of levosimendan in experimental models of right ventricular hypertrophy and failure. Pulmonary circulation 2014, 4, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Vonk Noordegraaf, A.; Westerhof, B.E.; Westerhof, N. The relationship between the right ventricle and its load in pulmonary hypertension. Journal of the American College of Cardiology 2017, 69, 236–243. [Google Scholar] [CrossRef]
- Kovács, A.; Lakatos, B.; Tokodi, M.; Merkely, B. Right ventricular mechanical pattern in health and disease: beyond longitudinal shortening. Heart failure reviews 2019, 24, 511–520. [Google Scholar] [CrossRef]
- Koestenberger, M.; Friedberg, M.K.; Nestaas, E.; Michel-Behnke, I.; Hansmann, G. Transthoracic echocardiography in the evaluation of pediatric pulmonary hypertension and ventricular dysfunction. Pulmonary circulation 2016, 6, 15–29. [Google Scholar] [CrossRef]
- Amsallem, M.; Sternbach, J.M.; Adigopula, S.; Kobayashi, Y.; Vu, T.A.; Zamanian, R.; Liang, D.; Dhillon, G.; Schnittger, I.; McConnell, M.V. Addressing the controversy of estimating pulmonary arterial pressure by echocardiography. Journal of the American Society of Echocardiography 2016, 29, 93–102. [Google Scholar] [CrossRef]
- Morrison, T.M.; Hariharan, P.; Funkhouser, C.M.; Afshari, P.; Goodin, M.; Horner, M. Assessing computational model credibility using a risk-based framework: application to hemolysis in centrifugal blood pumps. Asaio Journal 2019, 65, 349–360. [Google Scholar] [CrossRef]
- Yang, W.; Marsden, A.L.; Ogawa, M.T.; Sakarovitch, C.; Hall, K.K.; Rabinovitch, M.; Feinstein, J.A. Right ventricular stroke work correlates with outcomes in pediatric pulmonary arterial hypertension. Pulmonary circulation 2018, 8, 2045894018780534. [Google Scholar] [CrossRef]
- Chen, X.; Zhai, Z.; Huang, K.; Xie, W.; Wan, J.; Wang, C. Bosentan therapy for pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension: A systemic review and meta-analysis. The clinical respiratory journal 2018, 12, 2065–2074. [Google Scholar] [CrossRef]
- Guazzi, M.; Dixon, D.; Labate, V.; Beussink-Nelson, L.; Bandera, F.; Cuttica, M.J.; Shah, S.J. RV contractile function and its coupling to pulmonary circulation in heart failure with preserved ejection fraction: stratification of clinical phenotypes and outcomes. JACC: Cardiovascular Imaging 2017, 10, 1211–1221. [Google Scholar] [PubMed]
- Van De Veerdonk, M.C.; Kind, T.; Marcus, J.T.; Mauritz, G.-J.; Heymans, M.W.; Bogaard, H.-J.; Boonstra, A.; Marques, K.M.; Westerhof, N.; Vonk-Noordegraaf, A. Progressive right ventricular dysfunction in patients with pulmonary arterial hypertension responding to therapy. Journal of the American College of Cardiology 2011, 58, 2511–2519. [Google Scholar] [CrossRef] [PubMed]
- Forfia, P.R.; Fisher, M.R.; Mathai, S.C.; Housten-Harris, T.; Hemnes, A.R.; Borlaug, B.A.; Chamera, E.; Corretti, M.C.; Champion, H.C.; Abraham, T.P. Tricuspid annular displacement predicts survival in pulmonary hypertension. American journal of respiratory and critical care medicine 2006, 174, 1034–1041. [Google Scholar] [CrossRef]
- Freed, B.H.; Gomberg-Maitland, M.; Chandra, S.; Mor-Avi, V.; Rich, S.; Archer, S.L.; Jamison, E.B., Jr.; Lang, R.M.; Patel, A.R. Late gadolinium enhancement cardiovascular magnetic resonance predicts clinical worsening in patients with pulmonary hypertension. Journal of Cardiovascular Magnetic Resonance 2012, 14, 14. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.; Zhao, M.; Hu, D.-Q.; Fajardo, G.; Katznelson, E.; Punn, R.; Spin, J.M.; Chan, F.P.; Bernstein, D. Physiologic and molecular characterization of a murine model of right ventricular volume overload. American Journal of Physiology-Heart and Circulatory Physiology 2013, 304, H1314–H1327. [Google Scholar] [CrossRef]
- Oikawa, M.; Kagaya, Y.; Otani, H.; Sakuma, M.; Demachi, J.; Suzuki, J.; Takahashi, T.; Nawata, J.; Ido, T.; Watanabe, J. Increased [18F] fluorodeoxyglucose accumulation in right ventricular free wall in patients with pulmonary hypertension and the effect of epoprostenol. Journal of the American College of Cardiology 2005, 45, 1849–1855. [Google Scholar] [CrossRef]
- Kitahori, K.; He, H.; Kawata, M.; Cowan, D.B.; Friehs, I.; Del Nido, P.J.; McGowan, F.X., Jr. Development of left ventricular diastolic dysfunction with preservation of ejection fraction during progression of infant right ventricular hypertrophy. Circulation: Heart Failure 2009, 2, 599–607. [Google Scholar] [CrossRef]
- Mauritz, G.-J.; Kind, T.; Marcus, J.T.; Bogaard, H.-J.; van de Veerdonk, M.; Postmus, P.E.; Boonstra, A.; Westerhof, N.; Vonk-Noordegraaf, A. Progressive changes in right ventricular geometric shortening and long-term survival in pulmonary arterial hypertension. Chest 2012, 141, 935–943. [Google Scholar] [CrossRef]
- Marrone, G.; Mamone, G.; Luca, A.; Vitulo, P.; Bertani, A.; Pilato, M.; Gridelli, B. The role of 1.5 T cardiac MRI in the diagnosis, prognosis and management of pulmonary arterial hypertension. The international journal of cardiovascular imaging 2010, 26, 665–681. [Google Scholar] [CrossRef]
- Paulus, W.J.; Tschöpe, C. A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. Journal of the American college of cardiology 2013, 62, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Trip, P.; Kind, T.; van de Veerdonk, M.C.; Marcus, J.T.; de Man, F.S.; Westerhof, N.; Vonk-Noordegraaf, A. Accurate assessment of load-independent right ventricular systolic function in patients with pulmonary hypertension. The Journal of Heart and Lung Transplantation 2013, 32, 50–55. [Google Scholar] [CrossRef]
- Leeuwenburgh, B.P.; Steendijk, P.; Helbing, W.A.; Baan, J. Indexes of diastolic RV function: load dependence and changes after chronic RV pressure overload in lambs. American Journal of Physiology-Heart and Circulatory Physiology 2002, 282, H1350–H1358. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, J.M.; Grant, B.J. Effects of Pulmonary Vascular Obstruction on Right Ventricular Afterload1-3. Am Rev Respir Dis 1990, 141, 944–952. [Google Scholar] [CrossRef] [PubMed]
- Nickel, N.P.; Yuan, K.; Dorfmuller, P.; Provencher, S.; Lai, Y.-C.; Bonnet, S.; Austin, E.D.; Koch, C.D.; Morris, A.; Perros, F. Beyond the lungs: systemic manifestations of pulmonary arterial hypertension. American Journal of Respiratory and Critical Care Medicine 2020, 201, 148–157. [Google Scholar] [CrossRef]
- Opotowsky, A.R.; Ojeda, J.; Rogers, F.; Prasanna, V.; Clair, M.; Moko, L.; Vaidya, A.; Afilalo, J.; Forfia, P.R. A simple echocardiographic prediction rule for hemodynamics in pulmonary hypertension. Circulation: Cardiovascular Imaging 2012, 5, 765–775. [Google Scholar] [CrossRef]
- Ruiter, G.; Wong, Y.Y.; de Man, F.S.; Handoko, M.L.; Jaspers, R.T.; Postmus, P.E.; Westerhof, N.; Niessen, H.W.; van der Laarse, W.J.; Vonk-Noordegraaf, A. Right ventricular oxygen supply parameters are decreased in human and experimental pulmonary hypertension. The Journal of Heart and Lung Transplantation 2013, 32, 231–240. [Google Scholar] [CrossRef]
- Rosenkranz, S.; Howard, L.S.; Gomberg-Maitland, M.; Hoeper, M.M. Systemic consequences of pulmonary hypertension and right-sided heart failure. Circulation 2020, 141, 678–693. [Google Scholar] [CrossRef]
- Ghignone, M.; Girling, L.; Prewitt, R. Volume expansion versus norepinephrine in treatment of a low cardiac output complicating an acute increase in right ventricular afterload in dogs. Survey of Anesthesiology 1984, 28, 413. [Google Scholar] [CrossRef]
- Eichhorn, E.J.; Konstam, M.A.; Weiland, D.S.; Roberts, D.J.; Martin, T.T.; Stransky, N.B.; Salem, D.N. Differential effects of milrinone and dobutamine on right ventricular preload, afterload and systolic performance in congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. The American journal of cardiology 1987, 60, 1329–1333. [Google Scholar] [CrossRef] [PubMed]
- Revermann, M.; Schloss, M.; Mieth, A.; Babelova, A.; Schröder, K.; Neofitidou, S.; Buerkl, J.; Kirschning, T.; Schermuly, R.; Hofstetter, C. Levosimendan attenuates pulmonary vascular remodeling. Intensive care medicine 2011, 37, 1368–1377. [Google Scholar] [CrossRef] [PubMed]
- Houston, B.A.; Brittain, E.L.; Tedford, R.J. Right ventricular failure. New England Journal of Medicine 2023, 388, 1111–1125. [Google Scholar] [CrossRef]
- Klinke, A.; Schubert, T.; Müller, M.; Legchenko, E.; Zelt, J.G.; Shimauchi, T.; Napp, L.C.; Rothman, A.M.; Bonnet, S.; Stewart, D.J. Emerging therapies for right ventricular dysfunction and failure. Cardiovascular Diagnosis and Therapy 2020, 10, 1735. [Google Scholar] [CrossRef]
- Noordegraaf, A.V.; Galiè, N. The role of the right ventricle in pulmonary arterial hypertension. European Respiratory Review 2011, 20, 243–253. [Google Scholar] [CrossRef]
- Bosch, L.; Lam, C.S.; Gong, L.; Chan, S.P.; Sim, D.; Yeo, D.; Jaufeerally, F.; Leong, K.T.G.; Ong, H.Y.; Ng, T.P. Right ventricular dysfunction in left-sided heart failure with preserved versus reduced ejection fraction. European journal of heart failure 2017, 19, 1664–1671. [Google Scholar] [CrossRef]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F.; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. European Journal of Echocardiography 2016, 17, 1321–1360. [Google Scholar]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. European Heart Journal-Cardiovascular Imaging 2015, 16, 233–271. [Google Scholar] [CrossRef] [PubMed]
- Weatherald, J.; Boucly, A.; Chemla, D.; Savale, L.; Peng, M.; Jevnikar, M.; Jaïs, X.; Taniguchi, Y.; O’connell, C.; Parent, F. Prognostic value of follow-up hemodynamic variables after initial management in pulmonary arterial hypertension. Circulation 2018, 137, 693–704. [Google Scholar] [CrossRef]
- Madani, M.M. Surgical treatment of chronic thromboembolic pulmonary hypertension: pulmonary thromboendarterectomy. Methodist DeBakey cardiovascular journal 2016, 12, 213. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, J.; Golden, P.J.; Kajiura, L.N.; Murata, L.-A.M.; Uyehara, C.F. Vasopressin Decreases Pulmonary–to–Systemic Vascular Resistance Ratio in a Porcine Model of Severe Hemorrhagic Shock. Shock 2015, 43, 475–482. [Google Scholar] [CrossRef]
- Jiang, R.; Zhao, Q.H.; Wu, W.H.; Zhang, R.; Yuan, P.; Gong, S.G.; He, J.; Luo, C.J.; Qiu, H.L.; Wang, L. Efficacy and safety of a calcium sensitizer, levosimendan, in patients with right heart failure due to pulmonary hypertension. The clinical respiratory journal 2018, 12, 1518–1525. [Google Scholar] [CrossRef]
- Grignola, J.C.; Domingo, E. Acute right ventricular dysfunction in intensive care unit. BioMed Research International 2017, 2017, 8217105. [Google Scholar] [CrossRef]
- Amsallem, M.; Boulate, D.; Kooreman, Z.; Zamanian, R.T.; Fadel, G.; Schnittger, I.; Fadel, E.; McConnell, M.V.; Dhillon, G.; Mercier, O. Investigating the value of right heart echocardiographic metrics for detection of pulmonary hypertension in patients with advanced lung disease. The International Journal of Cardiovascular Imaging 2017, 33, 825–835. [Google Scholar] [CrossRef] [PubMed]
- Gerges, M.; Gerges, C.; Pistritto, A.-M.; Lang, M.B.; Trip, P.; Jakowitsch, J.; Binder, T.; Lang, I.M. Pulmonary hypertension in heart failure. Epidemiology, right ventricular function, and survival. American journal of respiratory and critical care medicine 2015, 192, 1234–1246. [Google Scholar] [CrossRef] [PubMed]
- Amsallem, M.; Sweatt, A.J.; Aymami, M.C.; Kuznetsova, T.; Selej, M.; Lu, H.; Mercier, O.; Fadel, E.; Schnittger, I.; McConnell, M.V. Right heart end-systolic remodeling index strongly predicts outcomes in pulmonary arterial hypertension: comparison with validated models. Circulation: Cardiovascular Imaging 2017, 10, e005771. [Google Scholar] [CrossRef] [PubMed]
- Masithulela, F. The effect of over-loaded right ventricle during passive filling in rat heart: A biventricular finite element model. In ASME international mechanical engineering congress and exposition; American Society of Mechanical Engineers, 2015. [Google Scholar]
- Masithulela, F. Analysis of passive filling with fibrotic myocardial infarction. In ASME international mechanical engineering congress and exposition; American Society of Mechanical Engineers, 2015. [Google Scholar]
- Masithulela, F.J. Computational biomechanics in the remodelling rat heart post myocardial infarction; 2016. [Google Scholar]
- Nemavhola, F. Detailed structural assessment of healthy interventricular septum in the presence of remodeling infarct in the free wall–A finite element model. Heliyon 2019, 5. [Google Scholar] [CrossRef]
- Nemavhola, F. Fibrotic infarction on the LV free wall may alter the mechanics of healthy septal wall during passive filling. Bio-medical materials and engineering 2017, 28, 579–599. [Google Scholar] [CrossRef] [PubMed]
- Nemavhola, F.; Ngwangwa, H.; Davies, N.; Franz, T. Passive biaxial tensile dataset of three main rat heart myocardia: left ventricle, mid-wall and right ventricle. 2021. [Google Scholar] [CrossRef]
- Nemavhola, F.; Ngwangwa, H.; Pandelani, T.; Davies, N.; Franz, T. Understanding regional mechanics of rat myocardia by fitting hyperelatsic models. 2021. [Google Scholar] [CrossRef]
- Nemavhola, F.; Ngwangwa, H.M.; Pandelani, T. An investigation of uniaxial mechanical properties of excised sheep heart muscle fibre–fitting of different hyperelastic constitutive models. 2021. [Google Scholar]
- Nemavhola, F.; Pandelani, T.; Ngwangwa, H. Fitting of hyperelastic constitutive models in different sheep heart regions based on biaxial mechanical tests. Russian journal of biomechanics 2022, 26, 19–30. [Google Scholar] [CrossRef]
- Ngwangwa, H.; Nemavhola, F.; Pandelani, T.; Msibi, M.; Mabuda, I.; Davies, N.; Franz, T. Determination of cross-directional and cross-wall variations of passive biaxial mechanical properties of rat myocardia. Processes 2022, 10, 629. [Google Scholar] [CrossRef]
- Ngwangwa, H.M.; Nemavhola, F. Evaluating computational performances of hyperelastic models on supraspinatus tendon uniaxial tensile test data. Journal of Computational Applied Mechanics 2021, 52, 27–43. [Google Scholar]
- Nemavhola, F. Mechanics of the septal wall may be affected by the presence of fibrotic infarct in the free wall at end-systole. International Journal of Medical Engineering and Informatics 2019, 11, 205–225. [Google Scholar] [CrossRef]
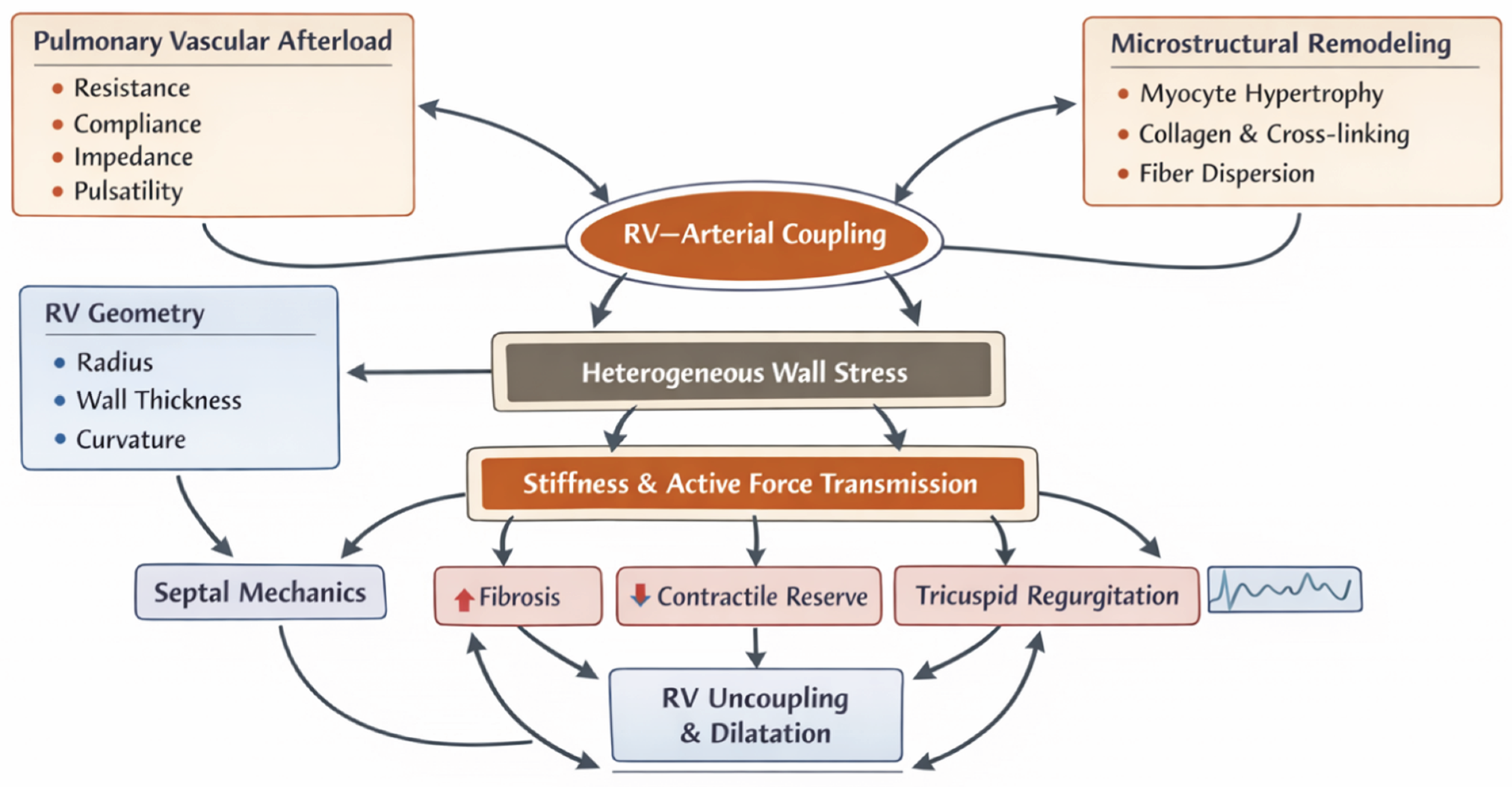
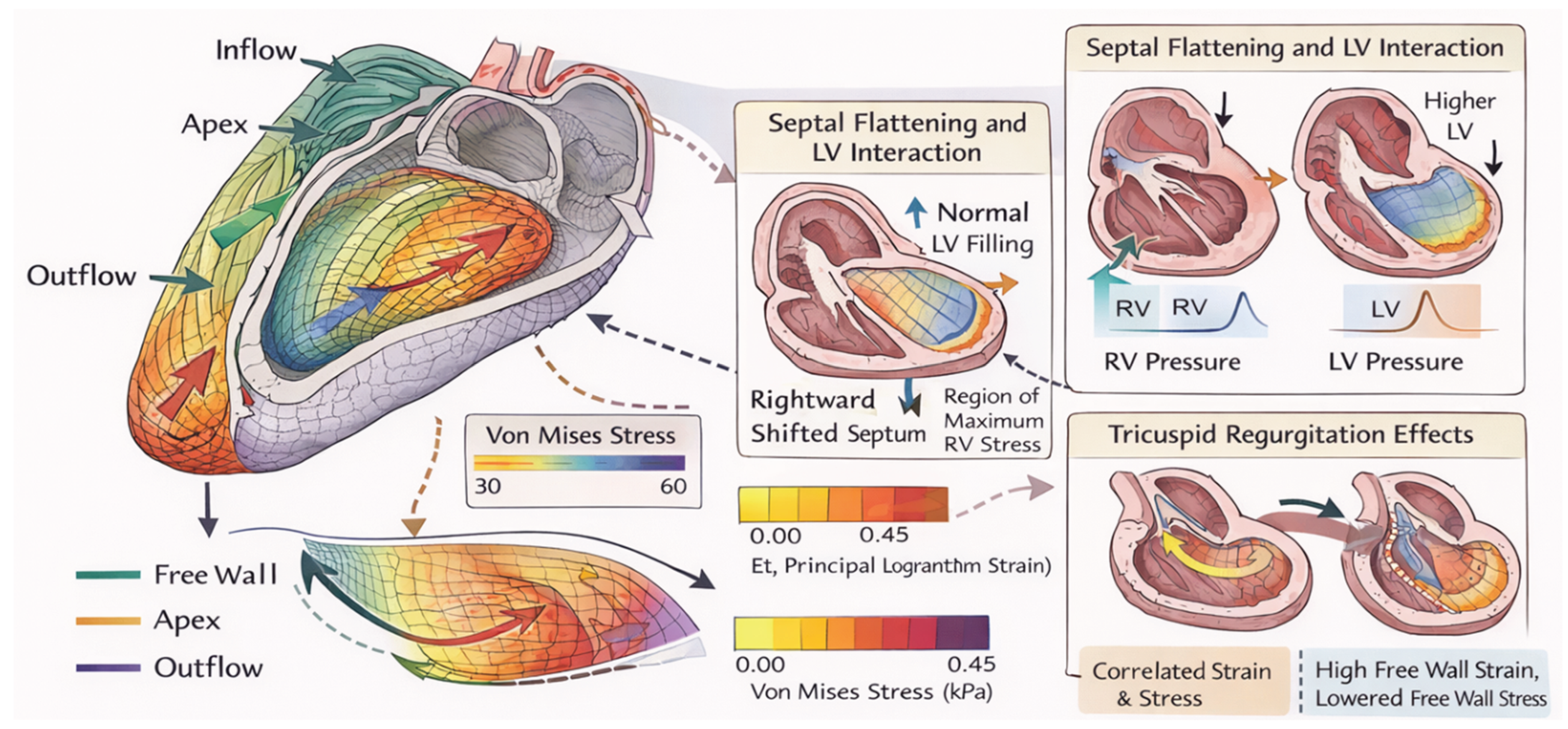
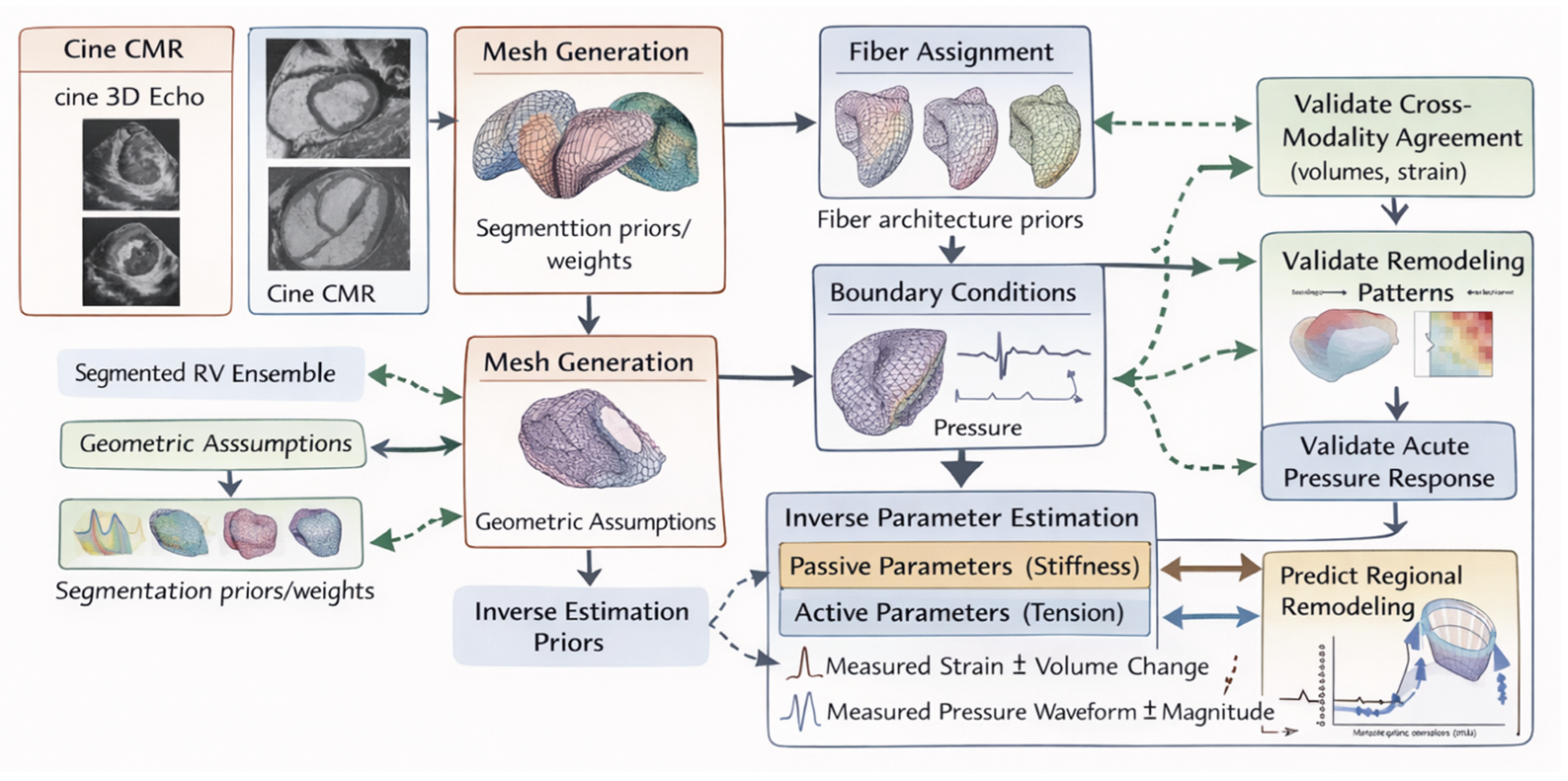
| Scale | Structural / functional feature | Primary mechanical quantity | Typical measurement modality | Interpretation pitfalls | Clinical linkage |
| Organ | RV-arterial coupling and afterload sensitivity | End-systolic elastance, effective arterial elastance, coupling ratio | Pressure-volume analysis; invasive hemodynamics with imaging | Coupling is system-level; surrogates mix load and contractility | Prognosis, therapy response, decompensation risk |
| Chamber | Geometry and wall thickness adaptation | Wall stress proxies; curvature; volume changes | Echo/CMR volumes; wall thickness from imaging | Thin wall makes thickness errors large; trabeculation complicates segmentation | Remodeling stage, dilation risk, tricuspid regurgitation progression |
| Regional | Free-wall versus septal contribution; dyssynchrony | Regional strain, strain rate, timing | Speckle tracking; CMR feature tracking/tagging | Strain depends on load, tethering, algorithm; timing influenced by conduction | Risk stratification; pacing/resynchronization targets |
| Tissue | Passive stiffness and fibrosis | Nonlinear stress-strain behavior; diastolic stiffness indices | Ex-vivo testing; CMR T1/LGE; modeling-inferred stiffness | Imaging fibrosis proxies are indirect; stiffness inference is model-dependent | Diastolic dysfunction; filling limitation; reverse remodeling potential |
| Microstructure | Fiber dispersion; collagen network architecture | Anisotropy parameters; recruitment behavior | Histology; diffusion tensor MRI (research); modeling | Limited in-vivo availability; sampling bias; region definition | Mechanistic insight; model structure selection |
| Model family | Key assumptions | Parameters typically estimated | Data requirements | Strengths | Limitations in RV context |
| Transversely isotropic hyperelastic (exponential forms) | Single preferred fiber direction; nonlinear stiffening | Fiber and matrix stiffness coefficients; dispersion terms | Strain field plus pressure; or tissue test data | Captures basic anisotropy and nonlinearity; widely used | Sensitive to fiber rules; may miss sheet structure; identifiability issues |
| Orthotropic / fiber–sheet models | Distinct fiber, sheet, and normal responses | Multiple directional stiffness parameters | Richer deformation data; often requires ex-vivo calibration | More physiologically faithful; supports transmural effects | High parameter count; difficult to fit from noninvasive data |
| Active stress models (additive along fibers) | Active tension aligned with fibers; time-varying activation | Peak active tension; activation timing; length dependence | Systolic deformation with pressure; activation timing | Separates passive and active components; supports coupling analysis | Activation timing uncertain; load-dependence confounding |
| Viscoelastic extensions | Time-dependent relaxation and hysteresis | Relaxation times; viscosity coefficients | Rate-dependent tests or multi-phase imaging | Explains rate effects and filling dynamics | Data rarely sufficient for fitting; increases complexity |
| Growth and remodeling frameworks | Mass deposition guided by stress/strain stimuli | Homeostatic targets; growth rates; collagen turnover | Longitudinal datasets; biomarker or imaging fibrosis proxies | Links mechanics to progression and reverse remodeling | Many unmeasured drivers; calibration challenging; uncertainty high |
| Metric | What is directly measured | Most defensible mechanical interpretation | Key dependencies | Reporting essentials | Use in modeling |
| Free-wall longitudinal strain (echo/CMR) | Kinematic deformation along long axis | Composite marker of systolic shortening under load | Afterload, preload, tethering, tracking algorithm | Vendor/software, segmentation, region definition, frame convention | Objective function for inverse fitting of active parameters |
| 3D RV volumes and ejection fraction | Chamber volume change | Global pump performance, not contractility | Afterload, heart rate, geometry errors | Acquisition mode, contouring rules, inclusion/exclusion of trabeculae | Boundary condition constraint; calibration of stroke volume |
| TAPSE and S’ velocity | Basal annular motion | Longitudinal shortening surrogate | Angle dependence, loading, conduction | View, alignment, rhythm status, averaging | Low-dimensional constraint; insufficient alone |
| CMR tissue characterization (LGE/T1) | Signal intensity or relaxation proxies | Substrate marker related to fibrosis/injury | Sequence parameters, hematocrit, motion artifacts | Sequence, post-processing, thresholds | Prior on stiffness spatial heterogeneity |
| 4D flow pulmonary metrics | Flow patterns and derived impedance proxies | Afterload characterization beyond pressure | Temporal resolution, segmentation, model assumptions | Acquisition, segmentation, derived formulae | Improves coupling estimation and boundary conditions |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




