Submitted:
07 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Descriptive Characteristics of the Analysed Cases
2.1.1. Impact of Anatomical Localization
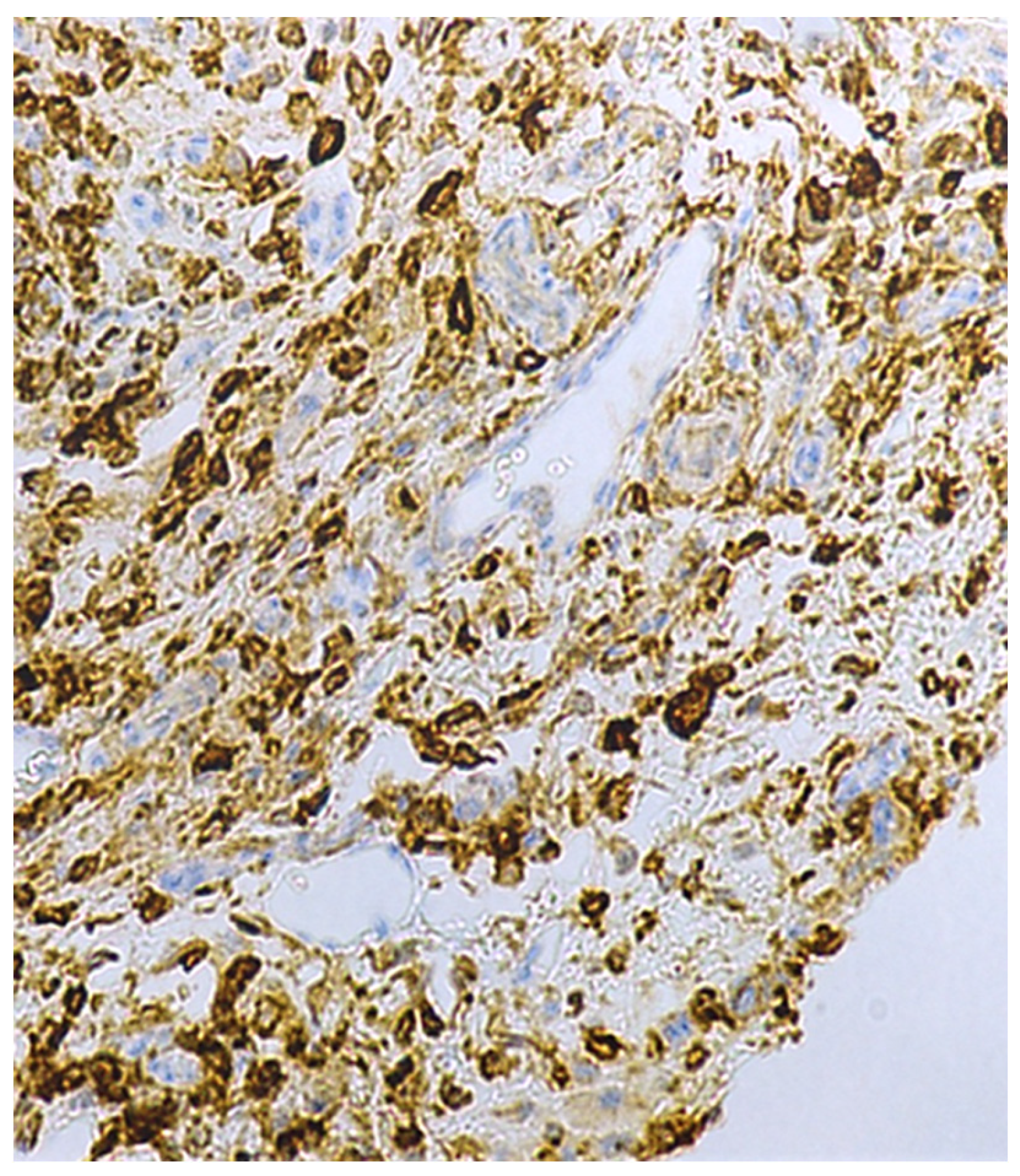
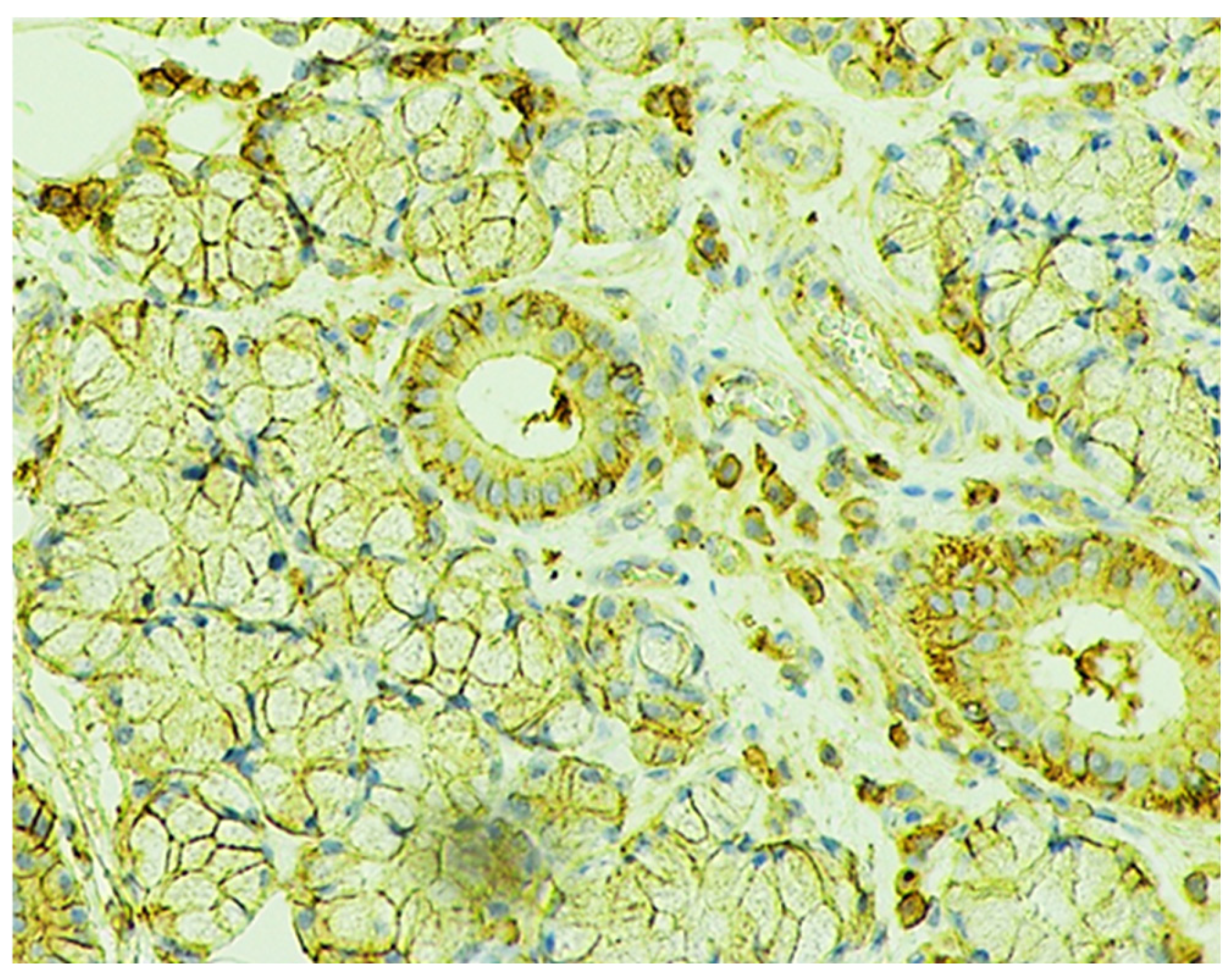
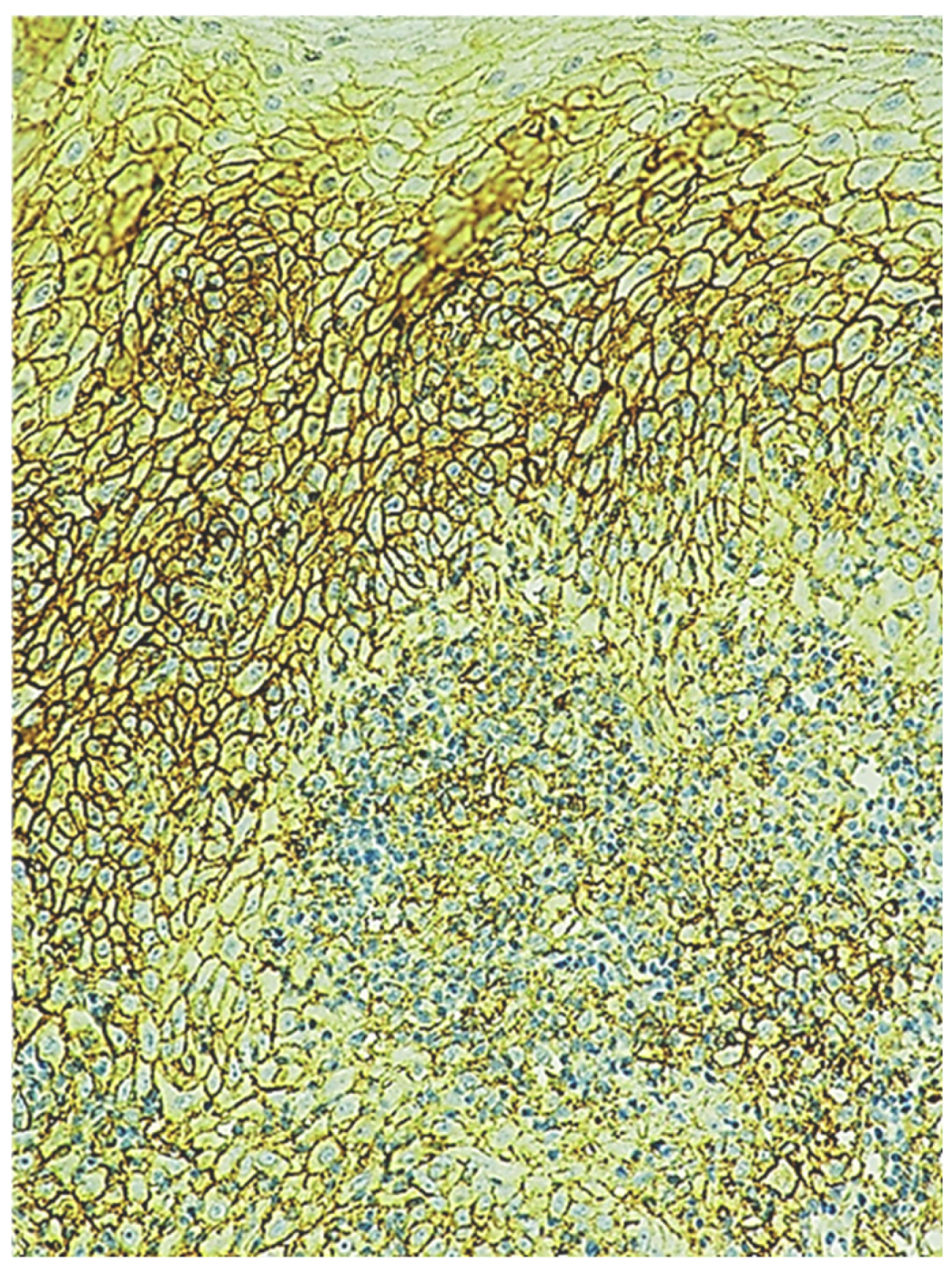
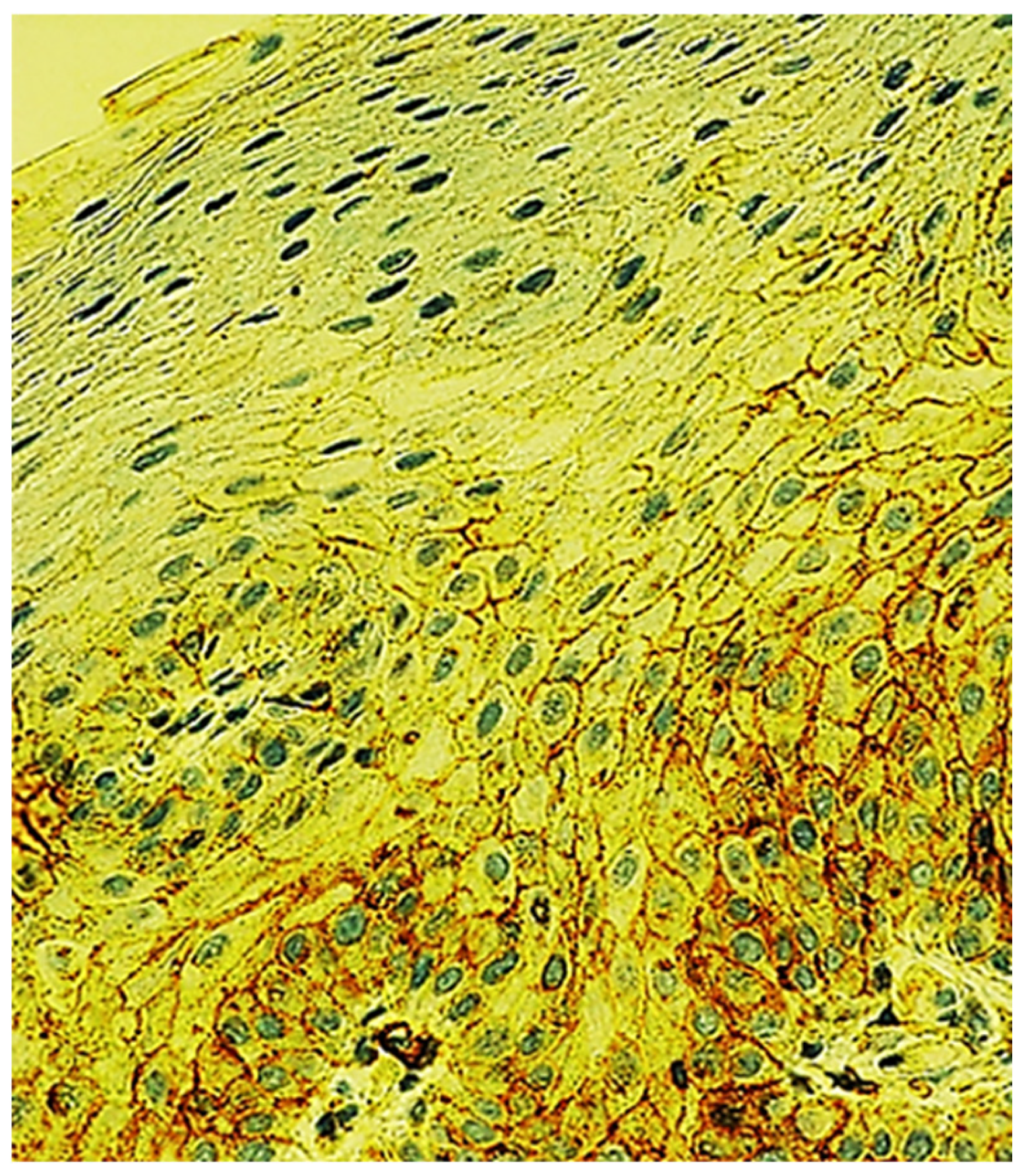
2.2. Immunohistochemical Characteristics of Immune Cells in Oral Leukoplakia
2.3. General Linear Model and Interaction Analysis
2.4. Characteristics of Dysplasia in the Various Clinical Types of Oral Leukoplakia
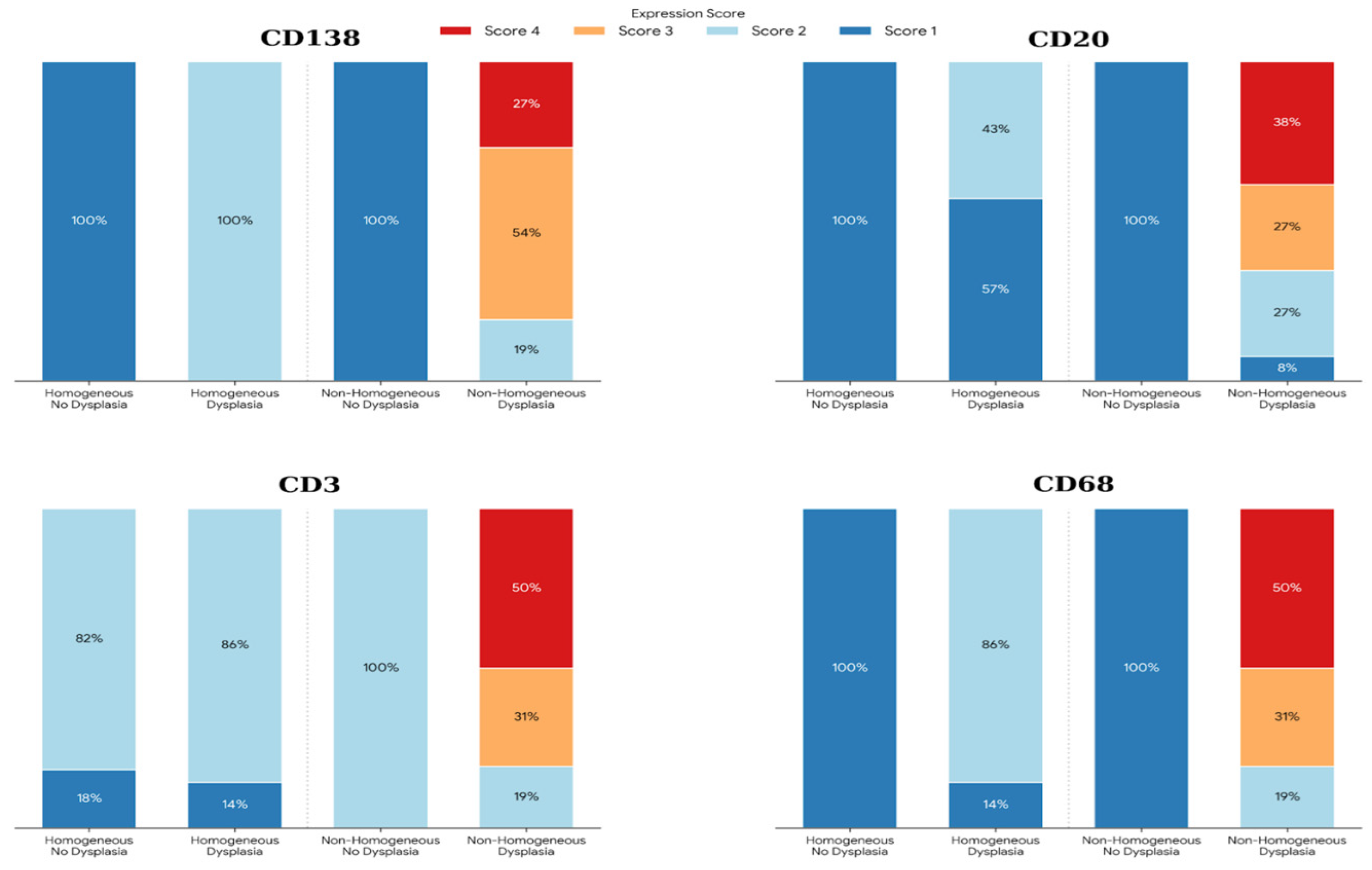
2.5. Distribution of Immunohistochemical Expression Scores in Oral Leukoplakia
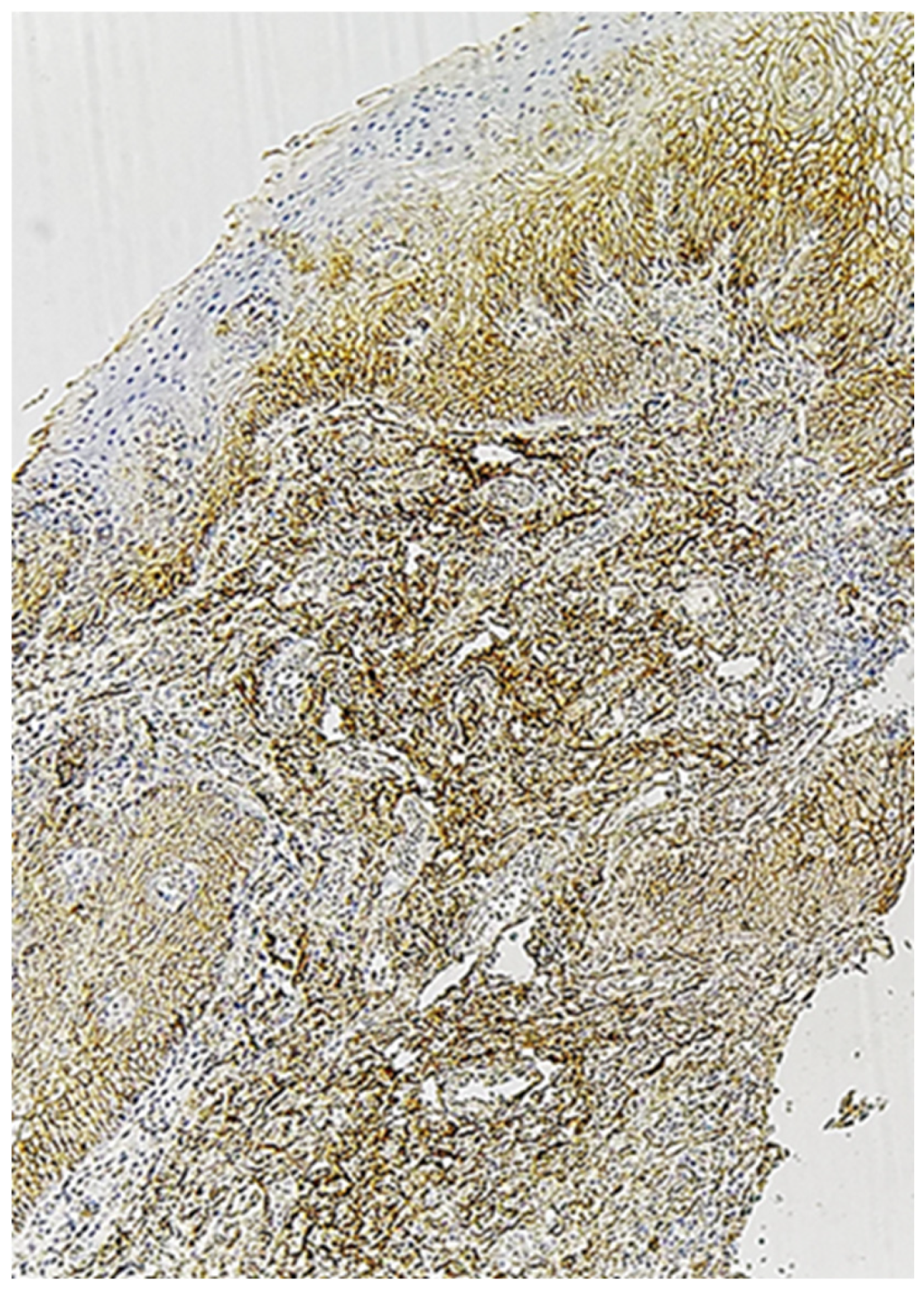
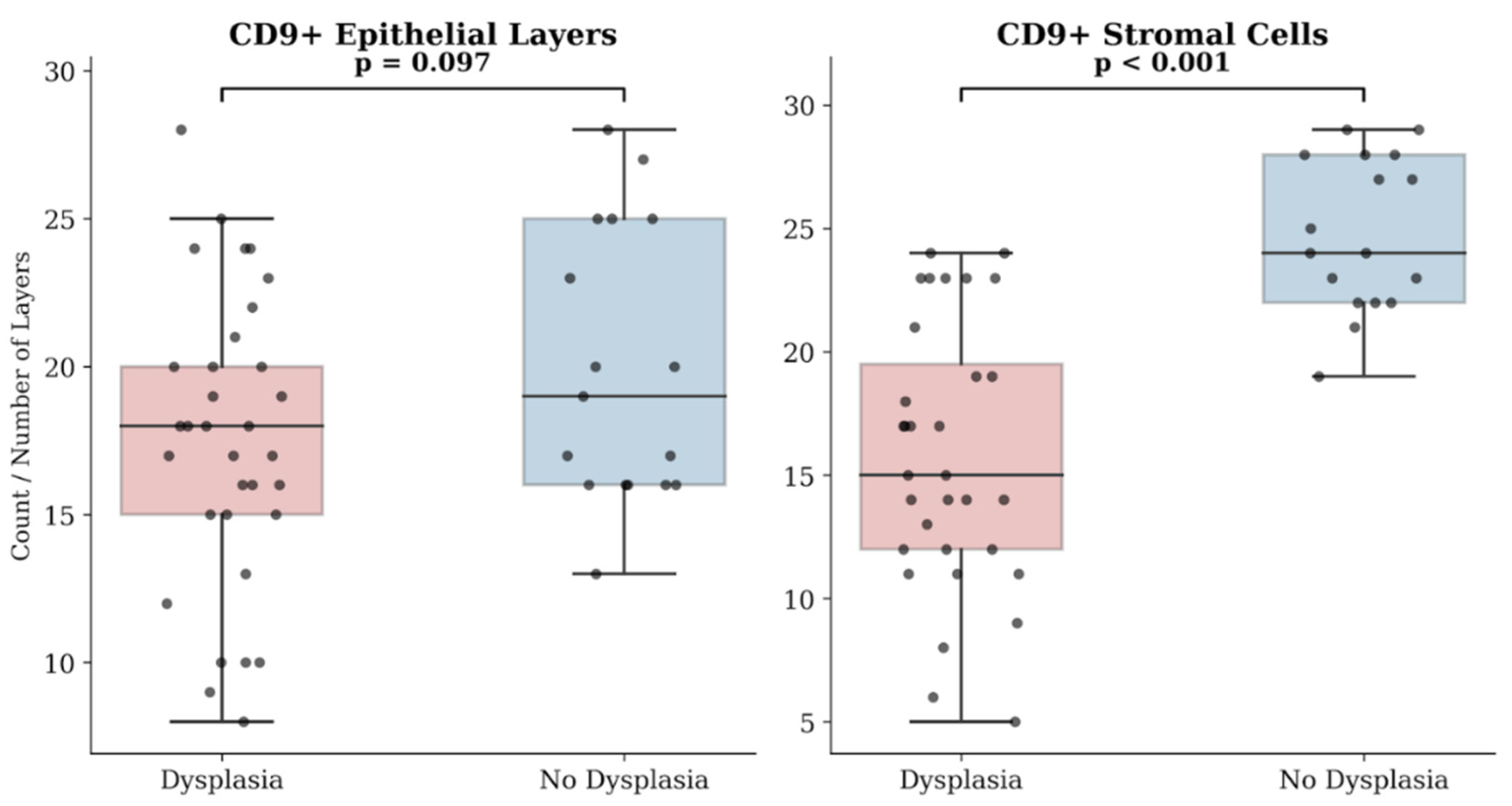
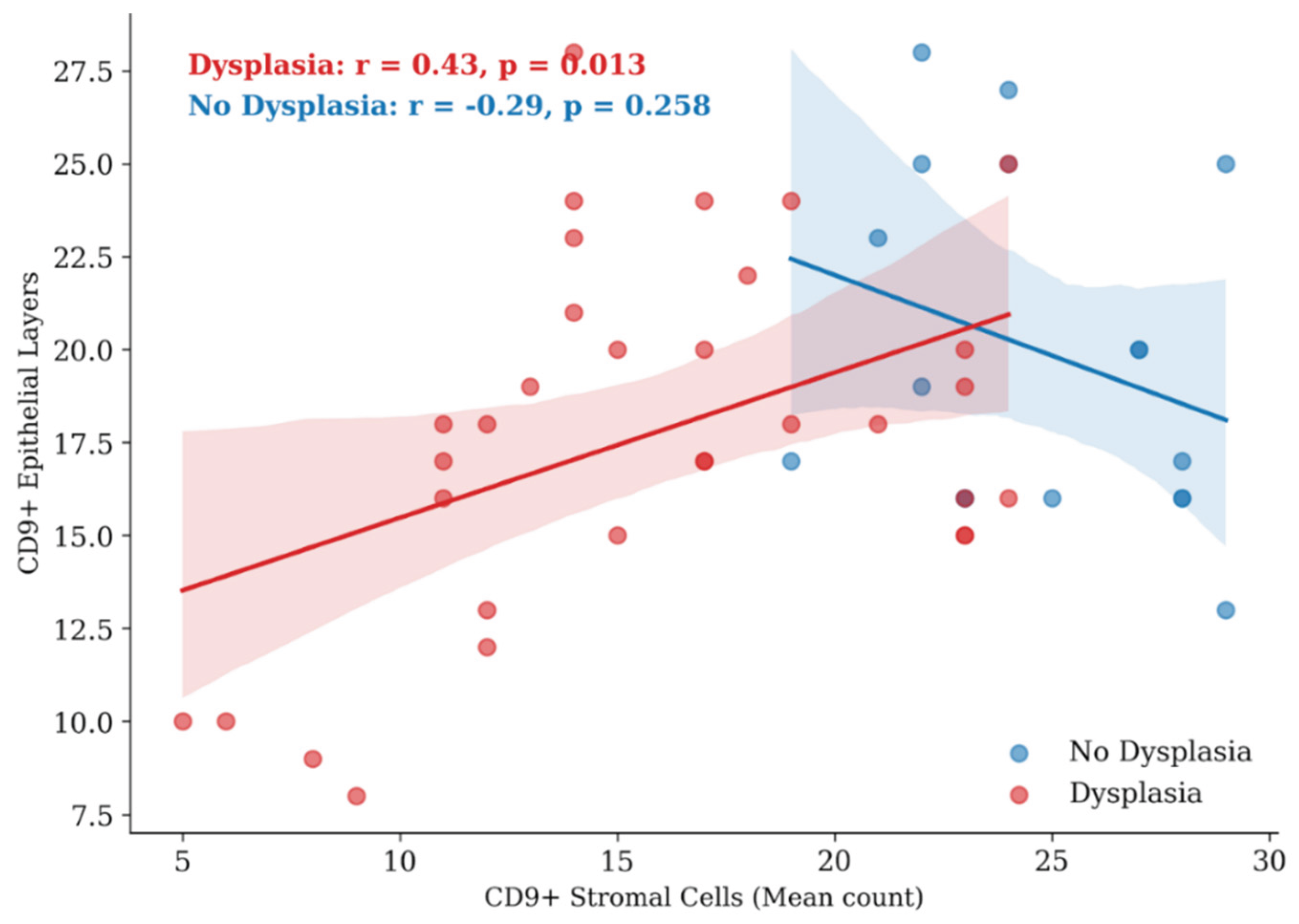
2.6. Immunohistochemical Characteristics of Exosome-Associated Antigen CD9 of epi Thelium and Immune Cells in Oral Leukoplakia
3. Discussion
4. Materials and Methods
4.1. Study Group
4.2. Microscopic and Immunohistochemical Examination
Immunohistochemical Examination
4.3. Statistical Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, C.; Li, B.; Zeng, X.; Hu, X.; Hua, H. The global prevalence of oral leukoplakia: a systematic review and meta-analysis from 1996 to 2022. BMC Oral Health 2023, 23, 645. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.Y.; Luo, Y.H.; Lin, Y.Y.; Wu, Z.Y.; Ye, J.Y.; Xie, S.M.; Li, J. Malignant transformation rate of oral leukoplakia in the past 20 years: A systematic review and meta-analysis. J Oral Pathol Med 2023, 52, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Gates, J.C.; Abouyared, M.; Shnayder, Y.; Farwell, D.G.; Day, A.; Alawi, F.; Moore, M.; Holcomb, A.J.; Birkeland, A.; Epstein, J. Clinical Management Update of Oral Leukoplakia: A Review From the American Head and Neck Society Cancer Prevention Service. Head Neck 2025, 47, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Leon, M.E.; Lugo, A.; Boffetta, P.; Gilmore, A.; Ross, H.; Schüz, J.; La Vecchia, C.; Gallus, S. Smokeless tobacco use in Sweden and other 17 European countries. Eur J Public Health 2016, 26, 817–821. [Google Scholar] [CrossRef]
- Tan, Y.; Wang, Z.; Xu, M.; Li, B.; Huang, Z.; Qin, S.; Nice, E.C.; Tang, J.; Huang, C. Oral squamous cell carcinomas: state of the field and emerging directions. Int J Oral Sci 2023, 15, 44. [Google Scholar] [CrossRef]
- Miluna, S.; Melderis, R.; Briuka, L.; Skadins, I.; Broks, R.; Kroica, J.; Rostoka, D. The Correlation of Swedish Snus, Nicotine Pouches and Other Tobacco Products with Oral Mucosal Health and Salivary Biomarkers. Dent J (Basel) 2022, 10. [Google Scholar] [CrossRef]
- Kuribayashi, Y.; Tsushima, F.; Morita, K.I.; Matsumoto, K.; Sakurai, J.; Uesugi, A.; Sato, K.; Oda, S.; Sakamoto, K.; Harada, H. Long-term outcome of non-surgical treatment in patients with oral leukoplakia. Oral Oncol 2015, 51, 1020–1025. [Google Scholar] [CrossRef]
- Iocca, O.; Sollecito, T.P.; Alawi, F.; Weinstein, G.S.; Newman, J.G.; De Virgilio, A.; Di Maio, P.; Spriano, G.; Pardiñas López, S.; Shanti, R.M. Potentially malignant disorders of the oral cavity and oral dysplasia: A systematic review and meta-analysis of malignant transformation rate by subtype. Head Neck 2020, 42, 539–555. [Google Scholar] [CrossRef]
- Maloney, B.D.; Galvin, S.D.; Healy, C.P. Oral leukoplakia: an update for dental practitioners. Journal of the Irish Dental Association 2024. [Google Scholar] [CrossRef]
- Li, W.; Han, Y.; Zhao, Z.; Ji, X.; Wang, X.; Jin, J.; Wang, Q.; Guo, X.; Cheng, Z.; Lu, M.; et al. Oral mucosal mesenchymal stem cell-derived exosomes: A potential therapeutic target in oral premalignant lesions. Int J Oncol 2019, 54, 1567–1578. [Google Scholar] [CrossRef]
- Zou, Y.; Zhou, Y.; Li, G.; Dong, Y.; Hu, S. Clinical applications of extracellular vesicles: recent advances and emerging trends. Front Bioeng Biotechnol 2025, 13, 1671963. [Google Scholar] [CrossRef]
- Andreu, Z.; Yáñez-Mó, M. Tetraspanins in extracellular vesicle formation and function. Front Immunol 2014, 5, 442. [Google Scholar] [CrossRef] [PubMed]
- Mamand, R.D.; Gustafsson, O.; Sork, H.; Wiklander, J.R.; Bazaz, S.; Liang, X.; Hou, W.Q.V.; Gupta, D.; Görgens, A.; Nordin, Z.J.; et al. Evaluation of Tetraspanins in Extracellular Vesicle Bioengineering. 2026. [Google Scholar] [CrossRef]
- Nankivell, P.; Williams, H.; McConkey, C.; Webster, K.; High, A.; MacLennan, K.; Senguven, B.; Rabbitts, P.; Mehanna, H. Tetraspanins CD9 and CD151, epidermal growth factor receptor and cyclooxygenase-2 expression predict malignant progression in oral epithelial dysplasia. Br J Cancer 2013, 109, 2864–2874. [Google Scholar] [CrossRef] [PubMed]
- Menck, K.; Sivaloganathan, S.; Bleckmann, A.; Binder, C. Microvesicles in Cancer: Small Size, Large Potential. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Xiao, M.; Zhang, J.; Chen, W. M1-like tumor-associated macrophages activated by exosome-transferred THBS1 promote malignant migration in oral squamous cell carcinoma. J Exp Clin Cancer Res 2018, 37, 143. [Google Scholar] [CrossRef]
- Peng, Q.; Yang, J.Y.; Zhou, G. Emerging functions and clinical applications of exosomes in human oral diseases. Cell Biosci 2020, 10, 68. [Google Scholar] [CrossRef]
- Li, X.; Corbett, A.L.; Taatizadeh, E.; Tasnim, N.; Little, J.P.; Garnis, C.; Daugaard, M.; Guns, E.; Hoorfar, M.; Li, I.T.S. Challenges and opportunities in exosome research-Perspectives from biology, engineering, and cancer therapy. APL Bioeng 2019, 3, 011503. [Google Scholar] [CrossRef]
- Kusukawa, J.; Ryu, F.; Kameyama, T.; Mekada, E. Reduced expression of CD9 in oral squamous cell carcinoma: CD9 expression inversely related to high prevalence of lymph node metastasis. J Oral Pathol Med 2001, 30, 73–79. [Google Scholar] [CrossRef]
- Vered, P.M.; Georgy, D.I.; Hamer, M.S.; Buchner, P.A.; Zlotogorski-hurvitz, D.A. DOWN-EXPRESSION OF TETRASPANIN CD9 IS A SENSITIVE MARKER FOR IDENTIFYING PRE-MALIGNANT CHANGES IN THE ORAL EPITHELIUM. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology 2019, 128, e26. [Google Scholar] [CrossRef]
- Yap, T.; Pruthi, N.; Seers, C.; Belobrov, S.; McCullough, M.; Celentano, A. Extracellular Vesicles in Oral Squamous Cell Carcinoma and Oral Potentially Malignant Disorders: A Systematic Review. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Lorico, A.; Lorico-Rappa, M.; Karbanová, J.; Corbeil, D.; Pizzorno, G. CD9, a tetraspanin target for cancer therapy? Exp Biol Med (Maywood) 2021, 246, 1121–1138. [Google Scholar] [CrossRef] [PubMed]
- Leung, L.L.; Qu, X.; Chen, B.; Chan, J.Y. Extracellular vesicles in liquid biopsies: there is hope for oral squamous cell carcinoma. Extracell Vesicles Circ Nucl Acids 2024, 5, 639–659. [Google Scholar] [CrossRef] [PubMed]
- Coumans, F.A.W.; Brisson, A.R.; Buzas, E.I.; Dignat-George, F.; Drees, E.E.E.; El-Andaloussi, S.; Emanueli, C.; Gasecka, A.; Hendrix, A.; Hill, A.F.; et al. Methodological Guidelines to Study Extracellular Vesicles. Circ Res 2017, 120, 1632–1648. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.M.; Mun, Y.J. Sample Preparation and Imaging of Exosomes by Transmission Electron Microscopy. Journal of Visualized Experiments 2018. [Google Scholar] [CrossRef]
- Daroit, B.N.; Prunes, B.D.B.; Maraschin, J.B.; Visioli, F.; Rados, V.P. Microscopia eletrônica de transmissão em citopatologia bucal - um estudo piloto. Revista da Faculdade de Odontologia de Porto Alegre 2022, 63, 56–63. [Google Scholar] [CrossRef]
- Miao, Q.; Li, S.; Lyu, W.; Zhang, J.; Han, Y. Exosomes in Oral Diseases: Mechanisms and Therapeutic Applications. Drug Des Devel Ther 2025, 19, 457–469. [Google Scholar] [CrossRef]
- Mitic, D.; Karisik, J.M.; Lazarevic, M.; Carkic, J.; Zivkovic, E.; Ajtic, M.O.; Milasin, J. Oral Squamous Cell Carcinoma Exosomes Upregulate PIK3/AKT, PTEN, and NOTCH Signaling Pathways in Normal Fibroblasts. Current Issues in Molecular Biology 2025, 47, 568. [Google Scholar] [CrossRef]
- Wang, R.; Lan, C.; Benlagha, K.; Camara, S.O.N.; Miller, H.; Kubo, M.; Heegaard, S.; Lee, P.; Yang, L.; Forsman, H.; et al. The interaction of innate immune and adaptive immune system. MedComm 2024, 5. [Google Scholar] [CrossRef]
- Zhu, X.; Zhu, J. CD4 T Helper Cell Subsets and Related Human Immunological Disorders. International Journal of Molecular Sciences 2020, 21, 8011. [Google Scholar] [CrossRef]
- Menon, P.A.; Moreno, B.; Meraviglia-Crivelli, D.; Nonatelli, F.; Villanueva, H.; Barainka, M.; Zheleva, A.; Santen, V.M.H.; Pastor, F. Modulating T Cell Responses by Targeting CD3. Cancers 2023, 15, 1189. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Hu, X.; Chen, Z.; Wei, W.; Wu, Y. Key links in the physiological regulation of the immune system and disease induction: T cell receptor -CD3 complex. Biochemical Pharmacology 2024, 227, 116441. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Su, Y.; Jiao, A.; Wang, X.; Zhang, B. T cells in health and disease. Signal Transduct Target Ther 2023, 8, 235. [Google Scholar] [CrossRef] [PubMed]
- Koch, F.P.; Kaemmerer, P.W.; Biesterfeld, S.; Kunkel, M.; Wagner, W. Effectiveness of autofluorescence to identify suspicious oral lesions--a prospective, blinded clinical trial. Clin Oral Investig 2011, 15, 975–982. [Google Scholar] [CrossRef]
- Dhatchinamoorthy, K.; Colbert, D.J.; Rock, L.K. Cancer Immune Evasion Through Loss of MHC Class I Antigen Presentation. Frontiers in Immunology 2021, 12. [Google Scholar] [CrossRef]
- Liu, J.; Ye, Y.; Cai, L. Supramolecular attack particle: the way cytotoxic T lymphocytes kill target cells. Signal Transduction and Targeted Therapy 2020, 5. [Google Scholar] [CrossRef]
- Öhman, J.; Mowjood, R.; Larsson, L.; Kovacs, A.; Magnusson, B.; Kjeller, G.; Jontell, M.; Hasseus, B. Presence of CD3-positive T-cells in oral premalignant leukoplakia indicates prevention of cancer transformation. Anticancer Res 2015, 35, 311–317. [Google Scholar]
- Ai, R.; Tao, Y.; Hao, Y.; Jiang, L.; Dan, H.; Ji, N.; Zeng, X.; Zhou, Y.; Chen, Q. Microenvironmental regulation of the progression of oral potentially malignant disorders towards malignancy. Oncotarget 2017, 8, 81617–81635. [Google Scholar] [CrossRef]
- Bouaoud, J.; Foy, J.P.; Tortereau, A.; Michon, L.; Lavergne, V.; Gadot, N.; Boyault, S.; Valantin, J.; De Souza, G.; Zrounba, P.; et al. Early changes in the immune microenvironment of oral potentially malignant disorders reveal an unexpected association of M2 macrophages with oral cancer free survival. Oncoimmunology 2021, 10, 1944554. [Google Scholar] [CrossRef]
- Deng, S.; Wang, S.; Shi, X.; Zhou, H. Microenvironment in Oral Potentially Malignant Disorders: Multi-Dimensional Characteristics and Mechanisms of Carcinogenesis. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Huang, D.M.; Sakhdari, D.A.; Aiman, D.A.; Guidos, D.C.; Magalhaes, D.M. Characterizing oral dysplasia immune microenvironment (DIME) using imaging mass cytometry. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology 2024, 138, e33–e34. [Google Scholar] [CrossRef]
- Perosa, F.; Favoino, E.; Caragnano, M.A.; Prete, M.; Dammacco, F. CD20: a target antigen for immunotherapy of autoimmune diseases. Autoimmun Rev 2005, 4, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Manríquez, J.; Hernández-Bello, J.; Muñoz-Valle, F.J.; Sifuentes-Franco, S.; Graciano-Machuca, O.; Morales-Núñez, J.J. B cell development: transcriptional regulation and immunological mechanisms in homeostasis. Frontiers in Immunology 2025, 16. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.A. CD20+ T cells: an emerging T cell subset in human pathology. Inflammation Research 2022, 71, 1181–1189. [Google Scholar] [CrossRef]
- Lund, F.E. Cytokine-producing B lymphocytes-key regulators of immunity. Curr Opin Immunol 2008, 20, 332–338. [Google Scholar] [CrossRef]
- Gannot, G.; Gannot, I.; Vered, H.; Buchner, A.; Keisari, Y. Increase in immune cell infiltration with progression of oral epithelium from hyperkeratosis to dysplasia and carcinoma. Br J Cancer 2002, 86, 1444–1448. [Google Scholar] [CrossRef] [PubMed]
- McCarron, M.J.; Park, P.W.; Fooksman, D.R. CD138 mediates selection of mature plasma cells by regulating their survival. Blood 2017, 129, 2749–2759. [Google Scholar] [CrossRef]
- Shetty, K.P.; Gonsalves, N.; Desai, D.; Pandit, S.; Aradhya, C.; Shahid, M.; Shubhalakshmi; Shetty, J.S. Expression of syndecan-1 in different grades of oral squamous cell carcinoma. Journal of Cancer Research and Therapeutics 2022, 18, S191–S196. [Google Scholar] [CrossRef]
- Akkaloori, A.; Saikia, J.; Kuppusamy, A.; Rana, K.; Dashatwar, P.D.; Behura, S.S. Comparison of the IHC Markers CD138 and CD43 in Oral Leukoplakia: An Original Research. J Pharm Bioallied Sci 2023, 15, S209–s212. [Google Scholar] [CrossRef]
- Chistiakov, A.D.; Killingsworth, C.M.; Myasoedova, A.V.; Orekhov, N.A.; Bobryshev, V.Y. CD68/macrosialin: not just a histochemical marker. Laboratory Investigation 2017, 97, 4–13. [Google Scholar] [CrossRef]
- Sutera, S.; Furchì, O.A.; Pentenero, M. Macrophages and the immune microenvironment in OPMDs: a systematic review of the literature. Front Oral Health 2025, 6, 1605978. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Classification of Tumours. In Head and Neck Tumours, 5th ed.; IARC, 2024; Volume 9. [Google Scholar]
- Hankinson, P.; Clark, M.; Walsh, H.; Khurram, A.S. A head-to-head comparison of four grading systems for oral epithelial dysplasia. Histopathology 2025, 86, 933–941. [Google Scholar] [CrossRef]
- Pimenta-Barros, A.L.; Ramos-García, P.; González-Moles, Á.M.; Aguirre-Urizar, M.J.; Warnakulasuriya, S. Malignant transformation of oral leukoplakia: Systematic review and comprehensive meta-analysis. Oral Diseases 2025, 31, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Warnakulasuriya, S.; Kujan, O.; Aguirre-Urizar, J.M.; Bagan, J.V.; González-Moles, M.; Kerr, A.R.; Lodi, G.; Mello, F.W.; Monteiro, L.; Ogden, G.R.; et al. Oral potentially malignant disorders: A consensus report from an international seminar on nomenclature and classification, convened by the WHO Collaborating Centre for Oral Cancer. Oral Dis 2021, 27, 1862–1880. [Google Scholar] [CrossRef] [PubMed]
- Warnakulasuriya, S.; Chen, T.H.H. Areca Nut and Oral Cancer: Evidence from Studies Conducted in Humans. Journal of Dental Research 2022, 101, 1139–1146. [Google Scholar] [CrossRef]
- Sari, F.E.; Johnson, W.N.; Mccullough, J.M.; Cirillo, N. Prevalence and risk factors of oral potentially malignant disorders in Indonesia: a cross-sectional study undertaken in 5 provinces. Scientific Reports 2024, 14. [Google Scholar] [CrossRef]
- Liutkutė-Gumarov, V.; Galkus, L.; Petkevičienė, J.; Štelemėkas, M.; Miščikienė, L.; Mickevičienė, A.; Vaitkevičiūtė, J. Illicit Tobacco in Lithuania: A Cross-Sectional Survey. International Journal of Environmental Research and Public Health 2020, 17, 7291. [Google Scholar] [CrossRef]
- Salokannel, M.; Ollila, E. Snus and snus-like nicotine products moving across Nordic borders: Can laws protect young people? Nordic Studies on Alcohol and Drugs 2021, 38, 540–554. [Google Scholar] [CrossRef]
- Miļuna-Meldere, S. The Impact of Smokeless Tobacco, Nicotine Pouches, and Electronic Cigarettes on Oral Health: Summary of the Doctoral Thesis: Sub-Sector – Stomatology. 2025. [Google Scholar] [CrossRef]
- Aguirre-Urizar, J.M.; Lafuente-Ibáñez de Mendoza, I.; Warnakulasuriya, S. Malignant transformation of oral leukoplakia: Systematic review and meta-analysis of the last 5 years. Oral Dis 2021. [Google Scholar] [CrossRef]
- Rubert, A.; Bagán, L.; Bagán, J. Oral leukoplakia, a clinical-histopathological study in 412 patients. Journal of Clinical and Experimental Dentistry 2020, e540–e546. [Google Scholar] [CrossRef] [PubMed]
- Saldivia-Siracusa, C.; González-Arriagada, W.A. Difficulties in the Prognostic Study of Oral Leukoplakia: Standardisation Proposal of Follow-Up Parameters. Front Oral Health 2021, 2, 614045. [Google Scholar] [CrossRef] [PubMed]
- Venkatesh, P.D.; Ramalingam, K.; Ramani, P.; Krishnan, M.; Vadivel, K.J. Epidemiological Trends and Clinicopathological Characteristics of Oral Leukoplakia: A Retrospective Analysis From a Single Institution in Chennai, Tamil Nadu, India. Cureus 2024. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.; Mohsen, A.; Fantozzi, J.P.; Golrang, A.; Podda, M.G.; Rocchetti, F.; Borghetti, L.; Sansotta, L.; Gioia, D.T.R.C.; Romeo, U. Risk Assessment for Malignant Transformation in Patients with Oral Proliferative Leukoplakia: A 10-Year Retrospective Cohort Study. Cancers 2025, 18, 2. [Google Scholar] [CrossRef]
- DB, M.; DrS, G.; PC, H. Oral leukoplakia: an update for dental practitioners. Journal of the Irish Dental Association 2024. [Google Scholar] [CrossRef]
- Aguirre-Urizar, J.M. Proliferative multifocal leukoplakia better name that proliferative verrucous leukoplakia. World J Surg Oncol 2011, 9, 122. [Google Scholar] [CrossRef]
- Sperandio, M.; Warnakulasuriya, S.; Soares, A.B.; Passador-Santos, F.; Mariano, F.V.; Lima, C.S.P.; Scarini, J.F.; Dominguete, M.H.L.; de Camargo Moraes, P.; Montalli, V.A.M.; et al. Oral epithelial dysplasia grading: Comparing the binary system to the traditional 3-tier system, an actuarial study with malignant transformation as outcome. J Oral Pathol Med 2023, 52, 418–425. [Google Scholar] [CrossRef]
- de Freitas Silva, B.S.; Batista, D.C.R.; de Souza Roriz, C.F.; Silva, L.R.; Normando, A.G.C.; Dos Santos Silva, A.R.; Silva, M.A.G.; Yamamoto-Silva, F.P. Binary and WHO dysplasia grading systems for the prediction of malignant transformation of oral leukoplakia and erythroplakia: a systematic review and meta-analysis. Clin Oral Investig 2021, 25, 4329–4340. [Google Scholar] [CrossRef]
- Yan, F.; Reddy, P.D.; Nguyen, S.A.; Chi, A.C.; Neville, B.W.; Day, T.A. Grading systems of oral cavity pre-malignancy: a systematic review and meta-analysis. Eur Arch Otorhinolaryngol 2020, 277, 2967–2976. [Google Scholar] [CrossRef]
- Solomon, C.M.; Chandrashekar, C.; Kulkarni, S.; Shetty, N.; Pandey, A. Exosomes: Mediators of cellular communication in potentially malignant oral lesions and head and neck cancers. F1000Research 2023, 12, 58. [Google Scholar] [CrossRef]
- Erovic, B.M.; Pammer, J.; Hollemann, D.; Woegerbauer, M.; Geleff, S.; Fischer, M.B.; Burian, M.; Frommlet, F.; Neuchrist, C. Motility-related protein-1/CD9 expression in head and neck squamous cell carcinoma. Head Neck 2003, 25, 848–857. [Google Scholar] [CrossRef] [PubMed]
- Brosseau, C.; Colas, L.; Magnan, A.; Brouard, S. CD9 Tetraspanin: A New Pathway for the Regulation of Inflammation? Front Immunol 2018, 9, 2316. [Google Scholar] [CrossRef] [PubMed]
- Mckelvey, J.K.; Powell, L.K.; Ashton, W.A.; Morris, M.J.; Mccracken, A.S. Exosomes: Mechanisms of Uptake. Journal of Circulating Biomarkers 2015, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Zlotogorski-Hurvitz, A.; Dayan, D.; Chaushu, G.; Korvala, J.; Salo, T.; Sormunen, R.; Vered, M. Human saliva-derived exosomes: comparing methods of isolation. J Histochem Cytochem 2015, 63, 181–189. [Google Scholar] [CrossRef]
- Weber, M.; Wehrhan, F.; Baran, C.; Agaimy, A.; Büttner-Herold, M.; Öztürk, H.; Neubauer, K.; Wickenhauser, C.; Kesting, M.; Ries, J. Malignant transformation of oral leukoplakia is associated with macrophage polarization. Journal of Translational Medicine 2020, 18. [Google Scholar] [CrossRef]
- Buim, C.E.M.; Lourenço, V.S.; Carvalho, C.K.; Cardim, R.; Pereira, C.; Carvalho, L.A.; Fregnani, H.J.; Soares, A.F. Downregulation of CD9 protein expression is associated with aggressive behavior of oral squamous cell carcinoma. Oral Oncology 2010, 46, 166–171. [Google Scholar] [CrossRef]
- Wang; Wang, L.; Yang, H.; Lu, H.; Zhang, J.; Li, N.; Guo, C.B. Development and validation of nomogram for prediction of malignant transformation in oral leukoplakia: A large-scale cohort study. J Oral Pathol Med 2019, 48, 491–498. [Google Scholar] [CrossRef]
- Čēma, I.; Dzudzilo, M.; Kleina, R.; Franckevica, I.; Svirskis, Š. Correlation of Soluble CD44 Expression in Saliva and CD44 Protein in Oral Leukoplakia Tissues. Cancers 2021, 13, 5739. [Google Scholar] [CrossRef]
- Baghban, N.; Kodam, S.P.; Ullah, M. Role of CD9 Sensing, AI, and Exosomes in Cellular Communication of Cancer. Int J Stem Cell Res Ther 2023, 10. [Google Scholar] [CrossRef]
- Xing, C.; Xu, W.; Shi, Y.; Zhou, B.; Wu, D.; Liang, B.; Zhou, Y.; Gao, S.; Feng, J. CD9 knockdown suppresses cell proliferation, adhesion, migration and invasion, while promoting apoptosis and the efficacy of chemotherapeutic drugs and imatinib in Ph+ ALL SUP-B15 cells. Mol Med Rep 2020, 22, 2791–2800. [Google Scholar] [CrossRef]
- Dzudzilo, M. Detecting Early Signs of Malignancy of Oral Leukoplakia in its Tissues and Saliva: Summary of the Doctoral Thesis: Sub-Sector – Dentistry. Rīga Stradiņš University, Riga, 2024. [Google Scholar]
- Buim, M.E.; Lourenço, S.V.; Carvalho, K.C.; Cardim, R.; Pereira, C.; Carvalho, A.L.; Fregnani, J.H.; Soares, F.A. Downregulation of CD9 protein expression is associated with aggressive behavior of oral squamous cell carcinoma. Oral Oncol 2010, 46, 166–171. [Google Scholar] [CrossRef]
- Dzudzilo, M.; Kleina, R.; Čēma, I.; Dabuzinskiene, A.; Svirskis, Š. Expression and Localisation of CD44 Antigen as a Prognostic Factor of Oral Leukoplakia. Proceedings of the Latvian Academy of Sciences. Section B. Natural, Exact, and Applied Sciences 2021, 75, 68–74. [Google Scholar] [CrossRef]
- Bouquot, E.J.; Speight, M.P.; Farthing, M.P. Epithelial dysplasia of the oral mucosa—Diagnostic problems and prognostic features. Current Diagnostic Pathology 2006, 12, 11–21. [Google Scholar] [CrossRef]
- Yousafzai, N.A.; Jin, H.; Ullah, M.; Wang, X. Recent advances of SIRT1 and implications in chemotherapeutics resistance in cancer. Am J Cancer Res 2021, 11, 5233–5248. [Google Scholar] [PubMed]
- Wang, Y.; Mao, J.; Wang, R.; Duan, D.; Liu, Z.; Hu, X.; Yu, Z.; Shi, X. Macrophage-induced immunomodulation in oral tissue repair and regeneration: Recent advances and future perspectives. J Adv Res 2025. [Google Scholar] [CrossRef]
- Shigeoka, M.; Koma, Y.I.; Nishio, M.; Komori, T.; Yokozaki, H. CD163+ macrophages infiltration correlates with the immunosuppressive cytokine interleukin 10 expression in tongue leukoplakia. Clinical and Experimental Dental Research 2019, 5, 627–637. [Google Scholar] [CrossRef]
- Liu, C.; Xiao, K.; Xie, L. Advances in the Regulation of Macrophage Polarization by Mesenchymal Stem Cells and Implications for ALI/ARDS Treatment. Frontiers in Immunology 2022, 13. [Google Scholar] [CrossRef]
- Hanania, R.; Sun, H.S.; Xu, K.; Pustylnik, S.; Jeganathan, S.; Harrison, R.E. Classically activated macrophages use stable microtubules for matrix metalloproteinase-9 (MMP-9) secretion. J Biol Chem 2012, 287, 8468–8483. [Google Scholar] [CrossRef]
- Feltraco, K.L.; Fernandes, R.J.; Diel, F.L.; Bittencourt, S.D.L.; Bernardi, L.; Lamers, L.M. Macrophages infiltration and oral potentially malignant disorders: A systematic review and meta-analysis. Oral Oncology Reports 2024, 11, 100559. [Google Scholar] [CrossRef]
- Akbarian, M.; Chen, S.-H. Instability Challenges and Stabilization Strategies of Pharmaceutical Proteins. Pharmaceutics 2022, 14, 2533. [Google Scholar] [CrossRef]
- Chaurasia, A.; Brigi, C.; Daghrery, A.; Asa’Ad, F.; Spirito, F.; Hasuike, A.; González-Alva, P.; Kojic, D.D.; Ünsal, K.B.R.; Sivaramakrishnan, G. Tumour-Associated Macrophages in Oral Squamous Cell Carcinoma. Oral Diseases 2025, 31, 1546–1563. [Google Scholar] [CrossRef]
- Soukka, T.; Pohjola, J.; Inki, P.; Happonen, R.P. Reduction of syndecan-1 expression is associated with dysplastic oral epithelium. Journal of Oral Pathology & Medicine 2000, 29, 308–313. [Google Scholar] [CrossRef]
- Lakkam, B.; Majage, B.; Astekar, M.; Gugwad, R.S.; Giri, G.; Ramasahayam, S. Immunohistochemical expression of syndecan-1 in oral dysplastic epithelium. J Cancer Res Ther 2014, 10, 103–106. [Google Scholar] [CrossRef] [PubMed]
- AS, T.; VH, S.; SM, I.; MT, A.; PR, H.; AT, B.Z. Expressions of CD 138 and CD43 in Oral Leukoplakia. Oral Maxillofac Pathol J 2022, 13(1), 1–5. [Google Scholar]
- Basharat, S.; Javaid, F.; Sajjad, A.; Khattak, M.; Ali, N.; Khan, M. Comparison of CD-138 Expression in Different Grades of Oral Squamous Cell Carcinoma. Life and Science 2024, 5, 06. [Google Scholar] [CrossRef]
- Kind, S.; Merenkow, C.; Büscheck, F.; Möller, K.; Dum, D.; Chirico, V.; Luebke, M.A.; Höflmayer, D.; Hinsch, A.; Jacobsen, F.; et al. Prevalence of Syndecan-1 (CD138) Expression in Different Kinds of Human Tumors and Normal Tissues. Disease Markers 2019, 2019, 1–11. [Google Scholar] [CrossRef]
- Kaur, H.; Mishra, D.; Roychoudhury, A.; Bhalla, S.A.; Ramteke, S.P.P.; Kumar, L. Plasma cells in oral lesion. Journal of Oral and Maxillofacial Pathology 2022, 26, 591. [Google Scholar] [CrossRef]
- Kamath, P.; Babu, C.; Tamgadge, A.; Pereira, T. Multiple myeloma with presentation in the oral cavity. Journal of Oral and Maxillofacial Pathology 2022, 26, 104–107. [Google Scholar] [CrossRef]
- Máthé, M.; Suba, Z.; Németh, Z.; Tátrai, P.; Füle, T.; Borgulya, G.; Barabás, J.; Kovalszky, I. Stromal syndecan-1 expression is an adverse prognostic factor in oral carcinomas. Oral Oncol 2006, 42, 493–500. [Google Scholar] [CrossRef]
- Mukunyadzi, P.; Liu, K.; Hanna, Y.E.; Suen, Y.J.; Fan, C.-Y. Induced Expression of Syndecan-1 in the Stroma of Head and Neck Squamous Cell Carcinoma. Modern Pathology 2003, 16, 796–801. [Google Scholar] [CrossRef]
- Löning, T.; Burkhardt, A. Plasma cells and immunoglobulin-synthesis in oral precancer and cancer. Virchows Archiv A Pathological Anatomy and Histology 1979, 384, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, M.; Soria, A.S.; Pires, R.J.; Sant’Ana, P.C.A.; Freire, M. Natural and induced immune responses in oral cavity and saliva. BMC Immunology 2025, 26. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Oh, J.-H.; Park, W.C.; Kim, H.; Kang, G.Y.; Oh, E.D.; Lee, J.H.; Kim, H.J.; Sung, O.C. Recombinant antibodies from clonally expanded cancer-associated plasma cells. Cancer Immunology, Immunotherapy 2025, 74. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.-Y.; Lin, C.-L.; Chang, M.-C.; Chen, H.-M.; Kok, S.-H.; Chang, S.-H.; Kuo, Y.-S.; Hahn, L.-J.; Chan, C.-P.; Lee, J.-J.; et al. Differences in oral habit and lymphocyte subpopulation affect malignant transformation of patients with oral precancer. Journal of the Formosan Medical Association 2016, 115, 263–268. [Google Scholar] [CrossRef]
- Lv, Z.; Yang, R.; Zhang, K.; Wang, R.; Shi, X.; Wu, J.; Liu, L.; Jiao, J. The dual immunomodulatory role of B cells in tumorigenesis: mechanisms, microenvironment crosstalk, and therapeutic implications. Frontiers in Immunology 2025, 16. [Google Scholar] [CrossRef]
- Sá, D.S.R.; Galvis, M.M.; Mariz, A.L.A.B.; Leite, A.A.; Schultz, L.; Almeida, P.O.; Santos-Silva, R.A.; Pinto, L.A.C.; Vargas, A.P.; Gollob, J.K.; et al. Increased Tumor Immune Microenvironment CD3+ and CD20+ Lymphocytes Predict a Better Prognosis in Oral Tongue Squamous Cell Carcinoma. Frontiers in Cell and Developmental Biology 2021, 8. [Google Scholar] [CrossRef]
- Xue, D.; Hu, S.; Zheng, R.; Luo, H.; Ren, X. Tumor-infiltrating B cells: Their dual mechanistic roles in the tumor microenvironment. Biomed Pharmacother 2024, 179, 117436. [Google Scholar] [CrossRef]
- Jiang, F.; Zheng, Q.; Chang, L.; Li, X.; Wang, X.; Gu, X. Pro-oncogene Pokemon Promotes Prostate Cancer Progression by Inducing STRN4 Expression. J Cancer 2019, 10, 1833–1845. [Google Scholar] [CrossRef]
- Öhman, J.; Magnusson, B.; Telemo, E.; Jontell, M.; Hasséus, B. Langerhans Cells and T Cells Sense Cell Dysplasia in Oral Leukoplakias and Oral Squamous Cell Carcinomas – Evidence for Immunosurveillance. Scandinavian Journal of Immunology 2012, 76, 39–48. [Google Scholar] [CrossRef]
- Hladíková, K.; Koucký, V.; Bouček, J.; Laco, J.; Grega, M.; Hodek, M.; Zábrodský, M.; Vošmik, M.; Rozkošová, K.; Vošmiková, H.; et al. Tumor-infiltrating B cells affect the progression of oropharyngeal squamous cell carcinoma via cell-to-cell interactions with CD8(+) T cells. J Immunother Cancer 2019, 7, 261. [Google Scholar] [CrossRef]
- Pellicioli, A.C.A.; Bingle, L.; Farthing, P.; Lopes, A.M.; Martins, D.M.; Vargas, A.P. Immunosurveillance profile of oral squamous cell carcinoma and oral epithelial dysplasia through dendritic and T-cell analysis. Journal of Oral Pathology & Medicine 2017, 46, 928–933. [Google Scholar] [CrossRef]
- Pretscher, D.; Distel, L.V.; Grabenbauer, G.G.; Wittlinger, M.; Buettner, M.; Niedobitek, G. Distribution of immune cells in head and neck cancer: CD8+ T-cells and CD20+ B-cells in metastatic lymph nodes are associated with favourable outcome in patients with oro- and hypopharyngeal carcinoma. BMC Cancer 2009, 9, 292. [Google Scholar] [CrossRef]
- Sharma, D.; Thomas, M.A.; Kwatra, S.K.; Koshy, G.; Mashon, S.R. Assessment of immune infiltrate in oral cancer: An immunohistochemical study. Journal of Oral and Maxillofacial Pathology 2025, 29, 66–75. [Google Scholar] [CrossRef]
- Seenivasagam, K.R.; Singh, A.; Gowda, N.V.; Poonia, R.D.; Majumdar, S.K.; Abhinav, T.; Kaul, P.; Panuganti, A.; Kailey, S.V.; Kumar, R.; et al. Clinico-Pathological Significance of Tumor Infiltrating Immune Cells in Oral Squamous Cell Carcinoma—Hope or Hype? Head & Neck 2025, 47, 1706–1716. [Google Scholar] [CrossRef]
- Jäwert, F.; Pettersson, H.; Jagefeldt, E.; Holmberg, E.; Kjeller, G.; Öhman, J. Clinicopathologic factors associated with malignant transformation of oral leukoplakias: a retrospective cohort study. International Journal of Oral and Maxillofacial Surgery 2021, 50, 1422–1428. [Google Scholar] [CrossRef]
- Kujan, O.; Oliver, R.J.; Khattab, A.; Roberts, S.A.; Thakker, N.; Sloan, P. Evaluation of a new binary system of grading oral epithelial dysplasia for prediction of malignant transformation. Oral Oncol 2006, 42, 987–993. [Google Scholar] [CrossRef]
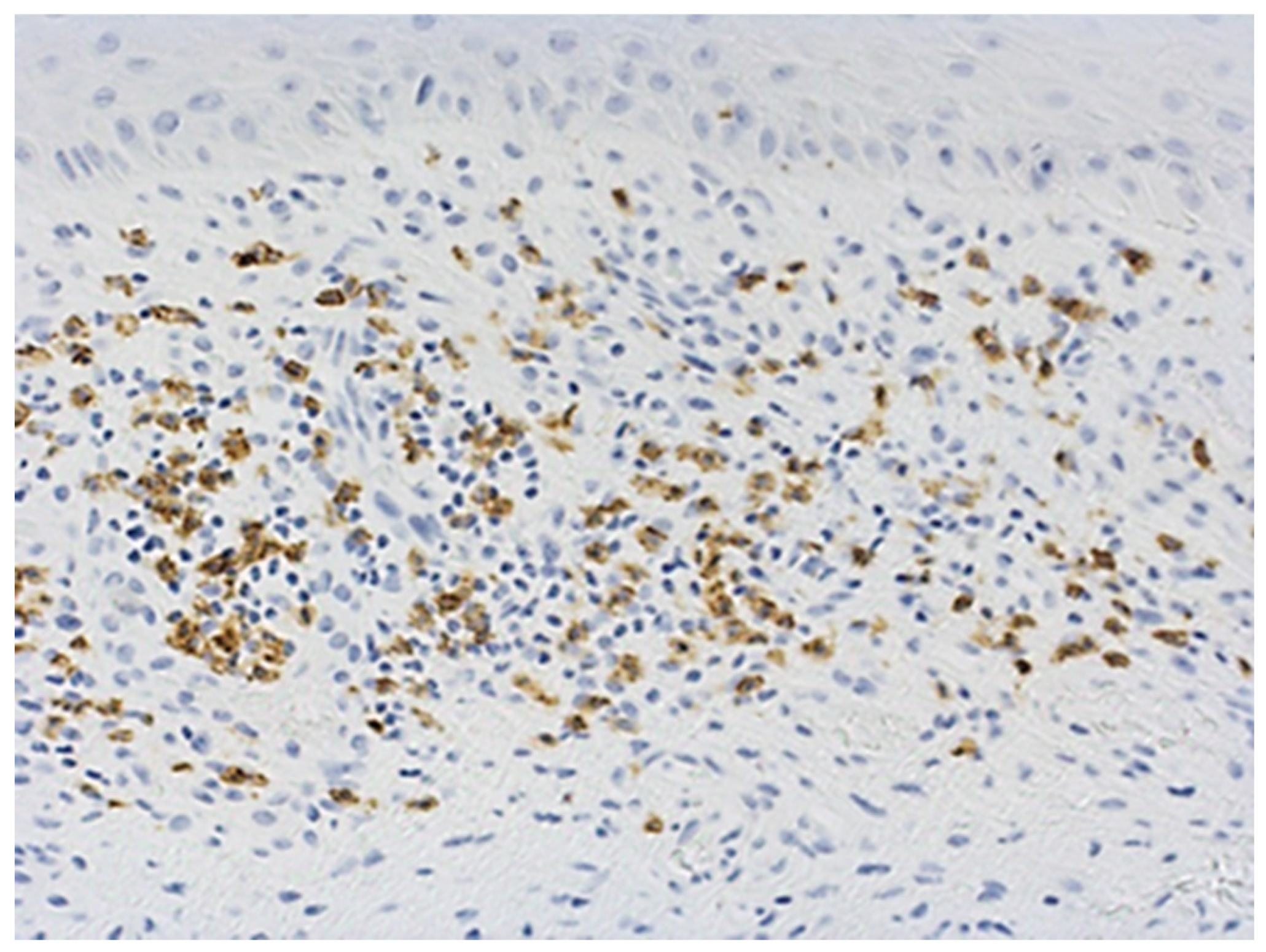
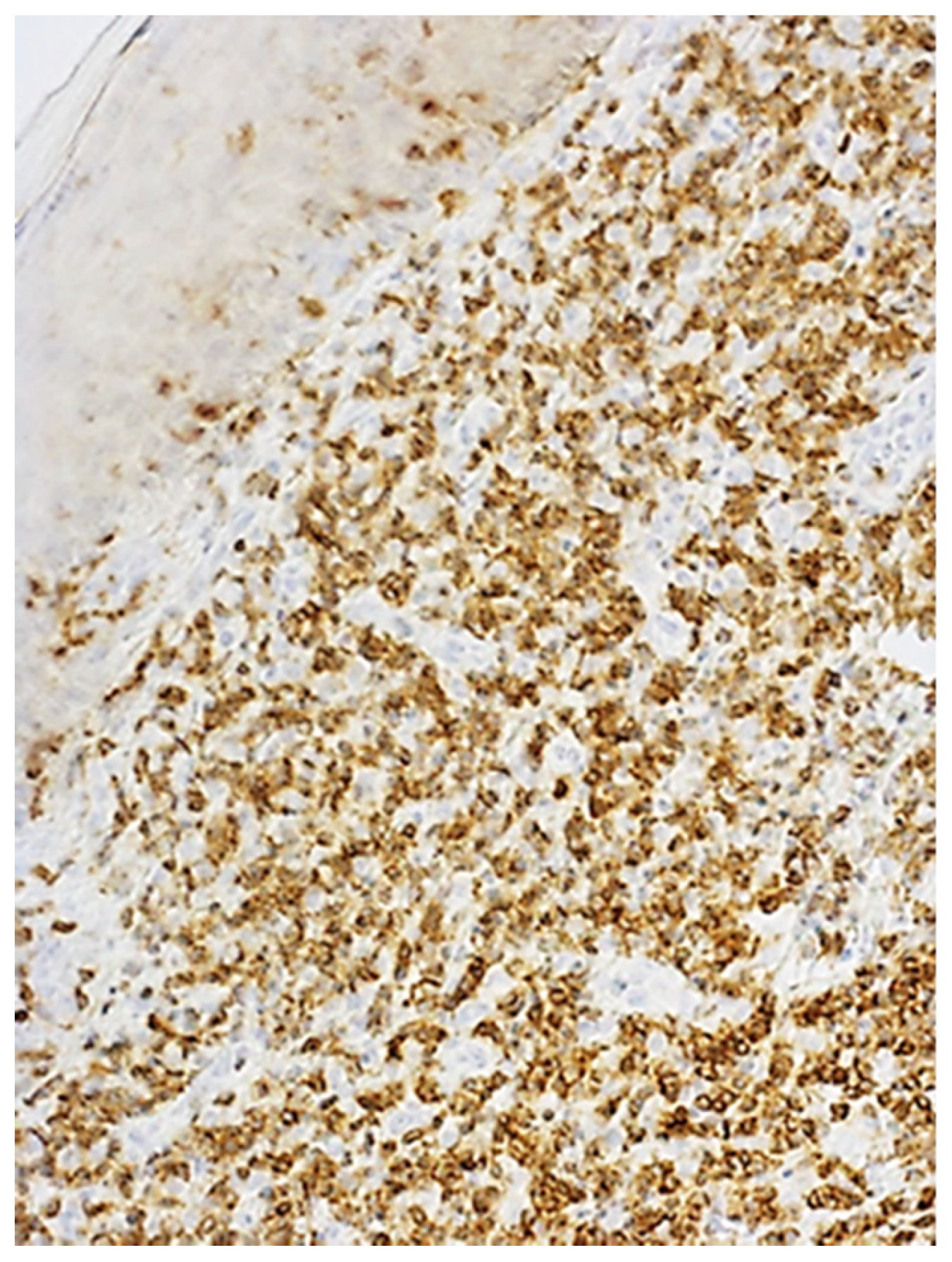
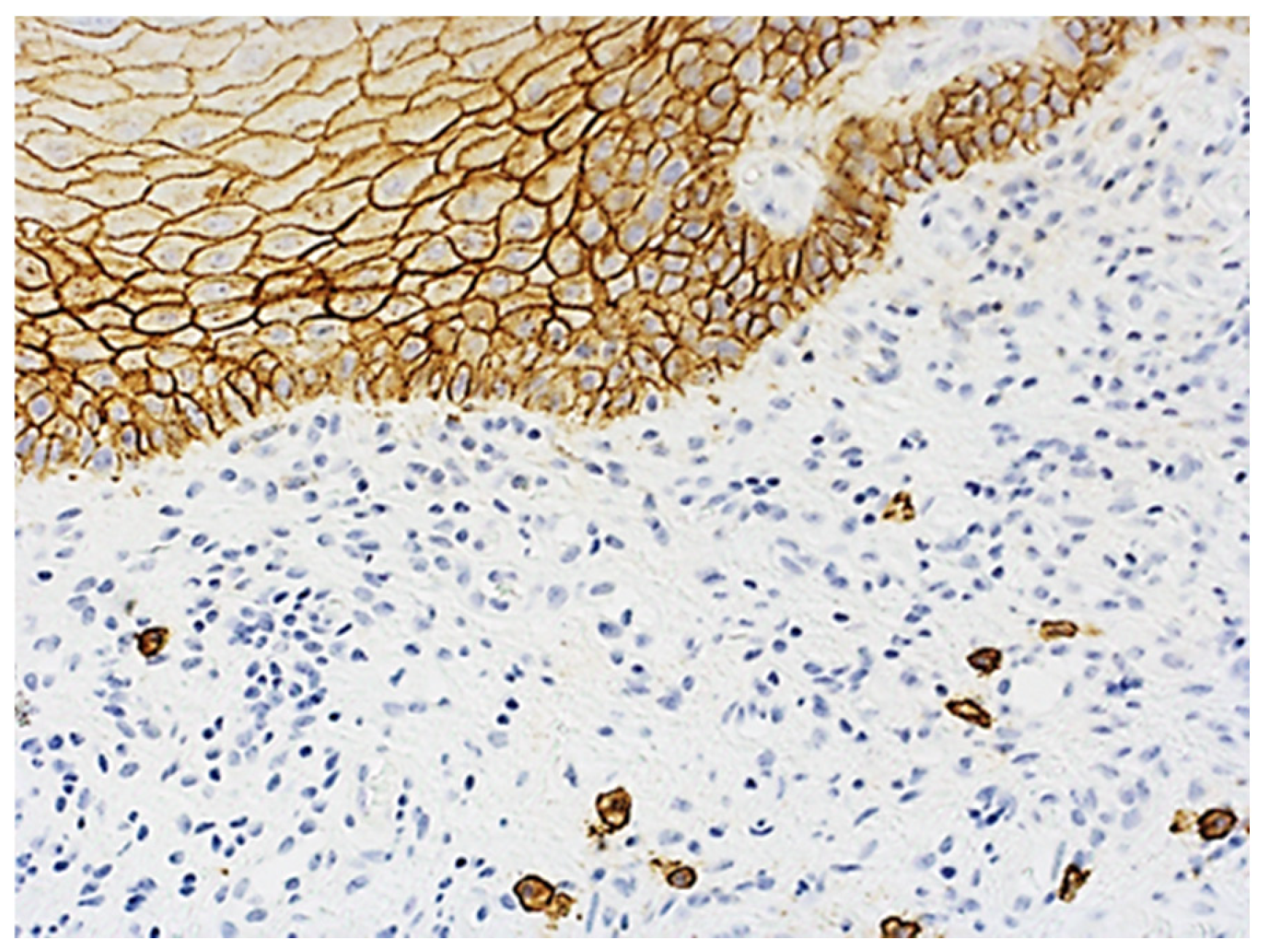
| Characteristic | Total (N=50) | Homogeneous (N=18) | Non-homogeneous (N=32) | p-value |
|---|---|---|---|---|
| Age, years (Mean ± SD) | 57.0 (±14.1) | 54.8 (±13.7) | 58.2 (±14.5) | 0.41 |
| Gender, n (%) | ||||
| Male | 29 (58.0%) | 9 (50.0%) | 20 (62.5%) | 0.575 |
| Female | 21 (42.0%) | 9 (50.0%) | 12 (37.5%) | |
| Localization, n (%) | ||||
| Buccal mucosa | 18 (36.0%) | 8 (44.4%) | 10 (31.2%) | 0.194 |
| Lateral border of tongue | 17 (34.0%) | 4 (22.2%) | 13 (40.6%) | |
| Floor of mouth | 11 (22.0%) | 3 (16.7%) | 8 (25.0%) | |
| Other (Lip, Alveolar ridge) | 4 (8.0%) | 3 (16.7%) | 1 (3.1%) | |
| Dysplasia status, n (%) | ||||
| Presence | 33 (66.0%) | 7 (38.9%) | 26 (81.2%) | 0.006 |
| Marker | Parameters | df | F | p | Partial η2 |
|---|---|---|---|---|---|
| CD138 | Model | 5 | 33.95 | < 0.001 | 0.794 |
| Clinical type | 1 | 10.54 | 0.002 | 0.193 | |
| Dysplasia | 1 | 72.94 | < 0.001 | 0.624 | |
| Clinical type x dysplasia | 1 | 9.96 | 0.003 | 0.185 | |
| Age | 1 | 0.70 | 0.407 | 0.016 | |
| Gender | 1 | 0.09 | 0.768 | 0.002 | |
| CD20 | Model | 5 | 14.27 | < 0.001 | 0.618 |
| Clinical type | 1 | 8.37 | 0.006 | 0.160 | |
| Dysplasia | 1 | 19.00 | < 0.001 | 0.302 | |
| Clinical type x dysplasia | 1 | 8.87 | 0.005 | 0.168 | |
| Age | 1 | 0.11 | 0.747 | 0.002 | |
| Gender | 1 | 0.06 | 0.811 | 0.001 | |
| CD3 | Model | 5 | 12.57 | < 0.001 | 0.588 |
| Clinical type | 1 | 14.39 | < 0.001 | 0.246 | |
| Dysplasia | 1 | 9.14 | 0.004 | 0.172 | |
| Clinical type x dysplasia | 1 | 8.95 | 0.005 | 0.169 | |
| Age | 1 | 0.00 | 0.974 | 0.000 | |
| Gender | 1 | 0.00 | 0.966 | 0.000 | |
| CD68 | Model | 5 | 30.47 | < 0.001 | 0.776 |
| Clinical type | 1 | 12.47 | < 0.001 | 0.221 | |
| Dysplasia | 1 | 57.08 | < 0.001 | 0.565 | |
| Clinical type x dysplasia | 1 | 12.93 | < 0.001 | 0.227 | |
| Age | 1 | 0.00 | 0.964 | 0.000 | |
| Gender | 1 | 0.16 | 0.687 | 0.004 |
| Marker | Oral Leukoplakia: clinical types | Effect of dysplasia (Estimate) | 95% CI | p |
|---|---|---|---|---|
| CD138 | Homogeneous | 0.999 | 0.800 – 1.182 | < 0.001 |
| Non-homogeneous | 2.075 | 1.758 – 2.385 | < 0.001 | |
| CD20 | Homogeneous | 0.419 | 0.006 – 0.951 | 0.284 |
| Non-homogeneous | 1.952 | 1.487 – 2.383 | < 0.001 | |
| CD3 | Homogeneous | 0.043 | -0.363 – 0.419 | 0.894 |
| Non-homogeneous | 1.312 | 0.922 – 1.667 | < 0.001 | |
| CD68 | Homogeneous | 0.885 | 0.509 – 1.178 | 0.005 |
| Non-homogeneous | 2.336 | 1.953 – 2.656 | < 0.001 |
| Antibody | Clone | Manufacturer | Dilution |
|---|---|---|---|
| CD3 | F7.2.38 | DAKO | Ready-to-use |
| CD20 | L26 | DAKO | Ready-to-use |
| CD138 | M15 | DAKO | Ready-to-use |
| CD68 | KP1 | DAKO | Ready-to-use |
| CD9 | No.4H7B9 | Proteintech | 1:1000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




