Submitted:
06 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
- Map local patterns of change and identify statistically significant anomalies.
- Examine whether fundamental nuclear effects can be distinguished from artifacts caused by natural isotopic abundance.
- Identify locations in the periodic system where simultaneous changes in multiple properties occur, indicating fundamental atomic reconfigurations.
- Define a multidimensional “fingerprint’’ for each element based on its SSD profile.
- Test whether the numerical periodicity of SSD patterns follows and confirms classical chemical periodicity.
2. Configuration-Space Interpretation of the Periodic System
- n is the principal quantum number of the outer shell,
- l is the dominant orbital type (s,p,d,f),
- f represents a coarse-grained electron filling state of the subshell.
3. Methodology
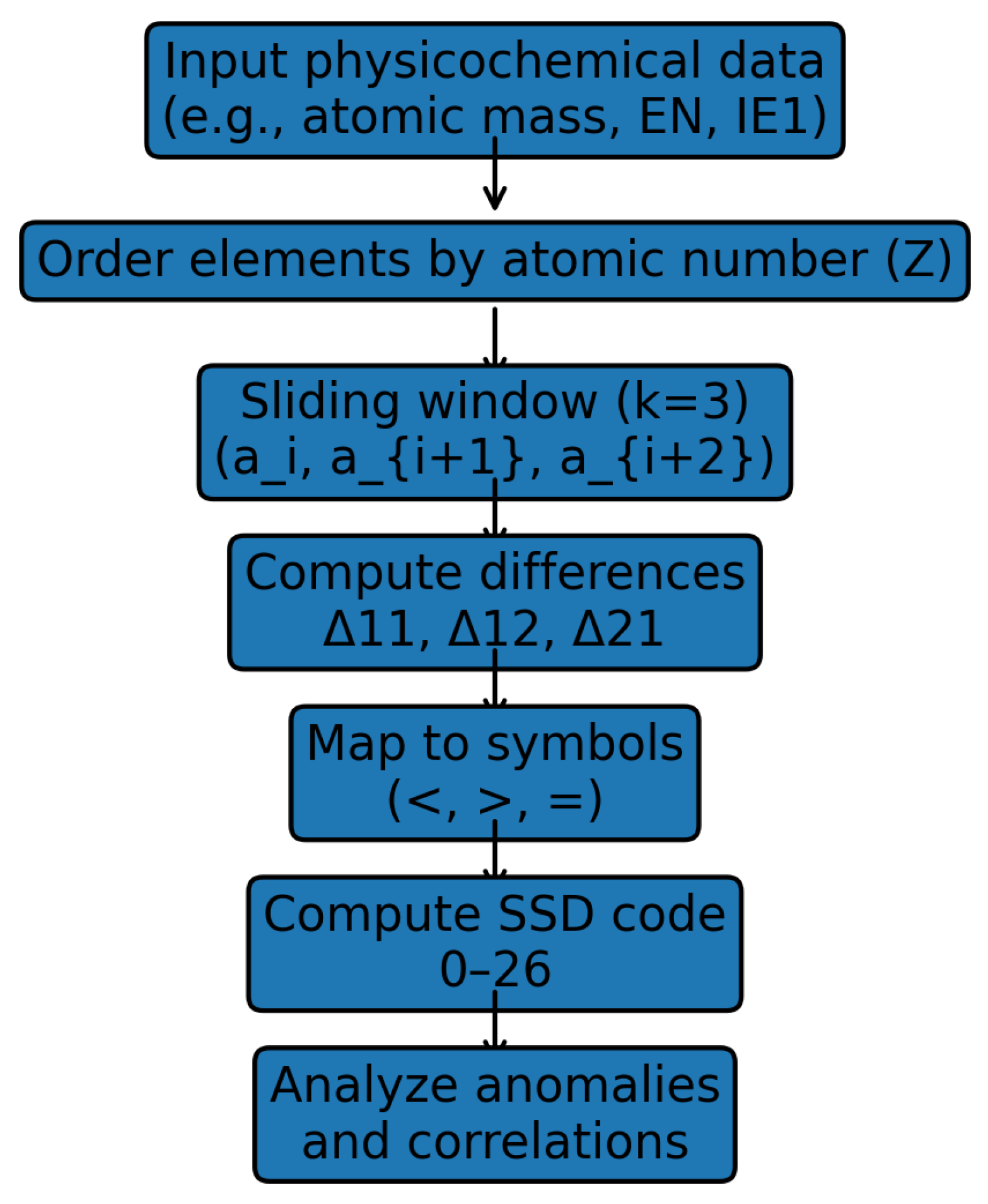
3.1. SSD Algorithm ()
- Symbol < is assigned if the difference is negative (for primary differences) or if (for the secondary difference). Value: .
- Symbol > is assigned if the difference is positive (for primary differences) or if . Value: .
- Symbol = is assigned if the differences are equal within a defined tolerance. Value: .
3.2. Data Sources
- Standard atomic masses from IUPAC Standard Atomic Weights.
- Monoisotopic masses from the NIST Atomic Weights and Isotopic Compositions database.
- Pauling electronegativities from the CRC Handbook of Chemistry and Physics (100th edition).
- First ionization energies (kJ/mol) from the CRC Handbook.
- Number of unpaired electrons determined from ground-state electron configurations using Hund’s rule.
- Melting points (°C) obtained from the NIST Chemistry WebBook and related sources, supplemented with theoretical estimates for superheavy elements.
3.3. Interpretation of SSD as a Structural Detector
- shell closure,
- subshell half-filling,
- orbital transition (e.g. s–p or p–d),
- relativistic stabilization effects in heavy elements.
4. Results and Analysis
4.1. Distribution of SSD Codes Across Properties
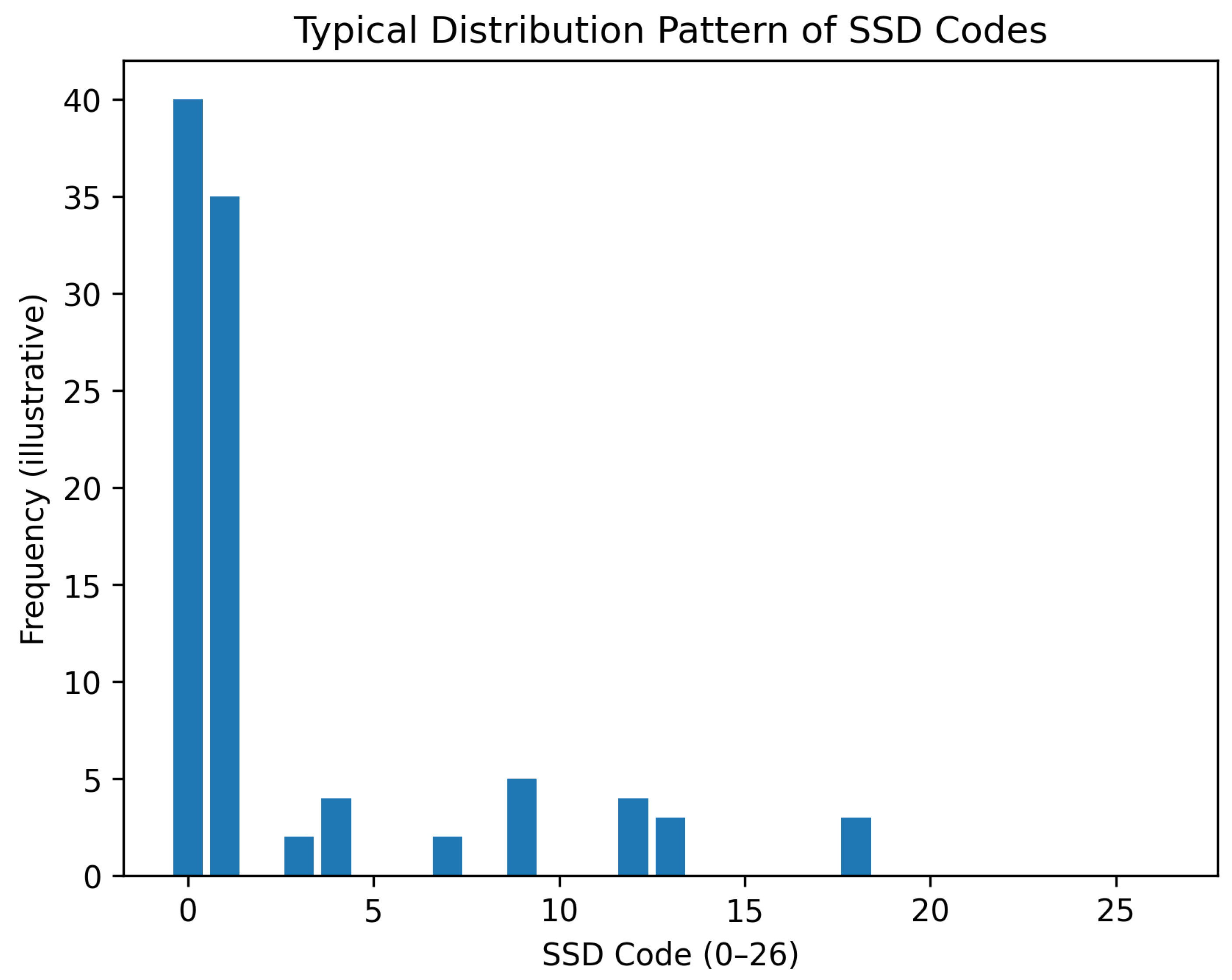
4.2. Topological Classification of Elements
| SSD Group | Characteristic Codes | Key Feature | Example Elements |
| Linear Growers | 0,1 | Stable monotonic increase | Lanthanides |
| Inverters | 4,9 | Reversal of trend | Ar–K–Ca |
| Plateaus | 7,18,2 | Stagnation or slowing | Cm–Bk–Cf |
| Chaotic Jumpers | 3,12,13 | Sudden complex changes | Mn–Fe–Co |
| Perfect Plateaus | 26 | Complete equality | He–Li–Be |
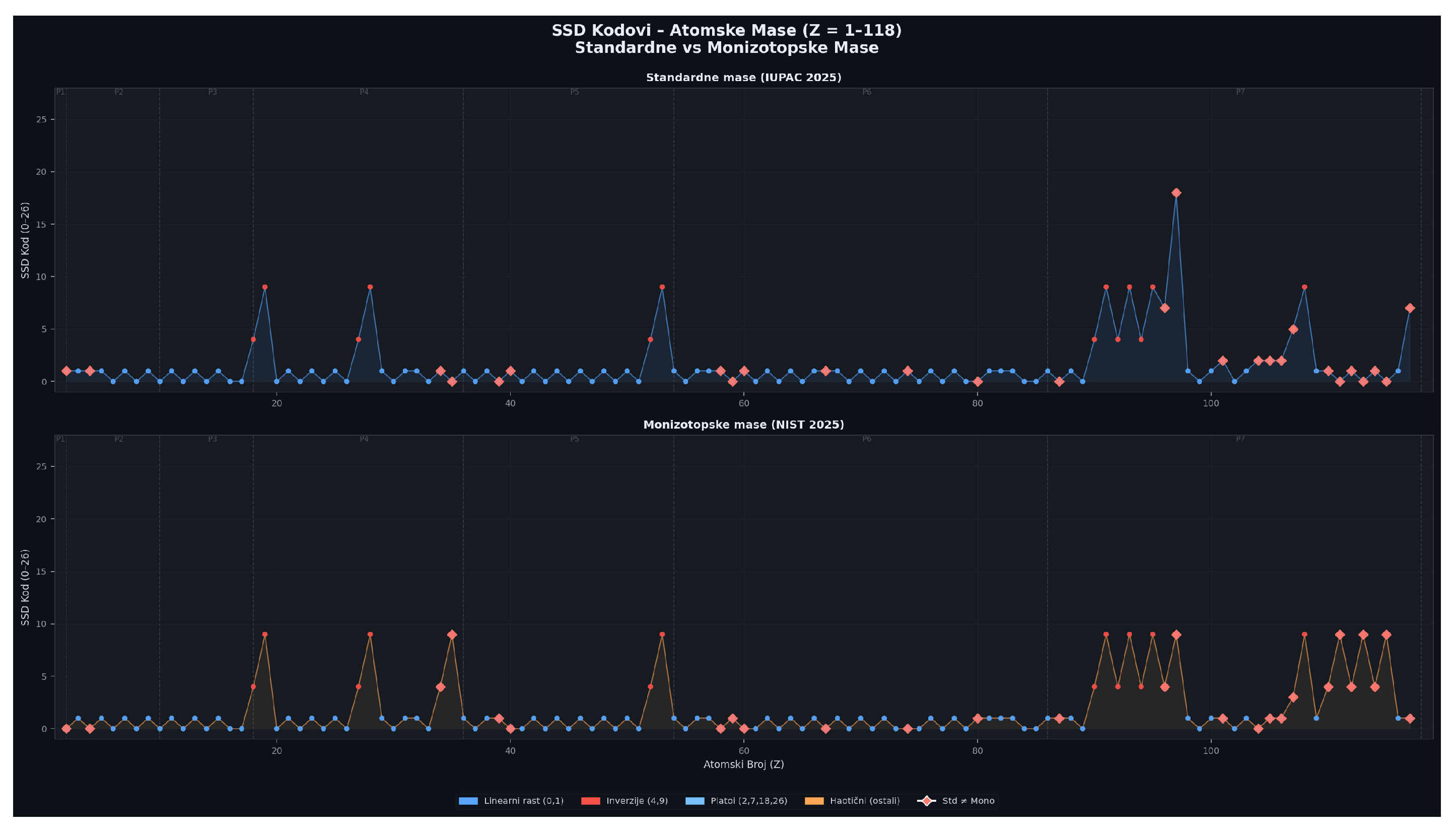
4.3. Critical Points: Atomic Mass Anomalies
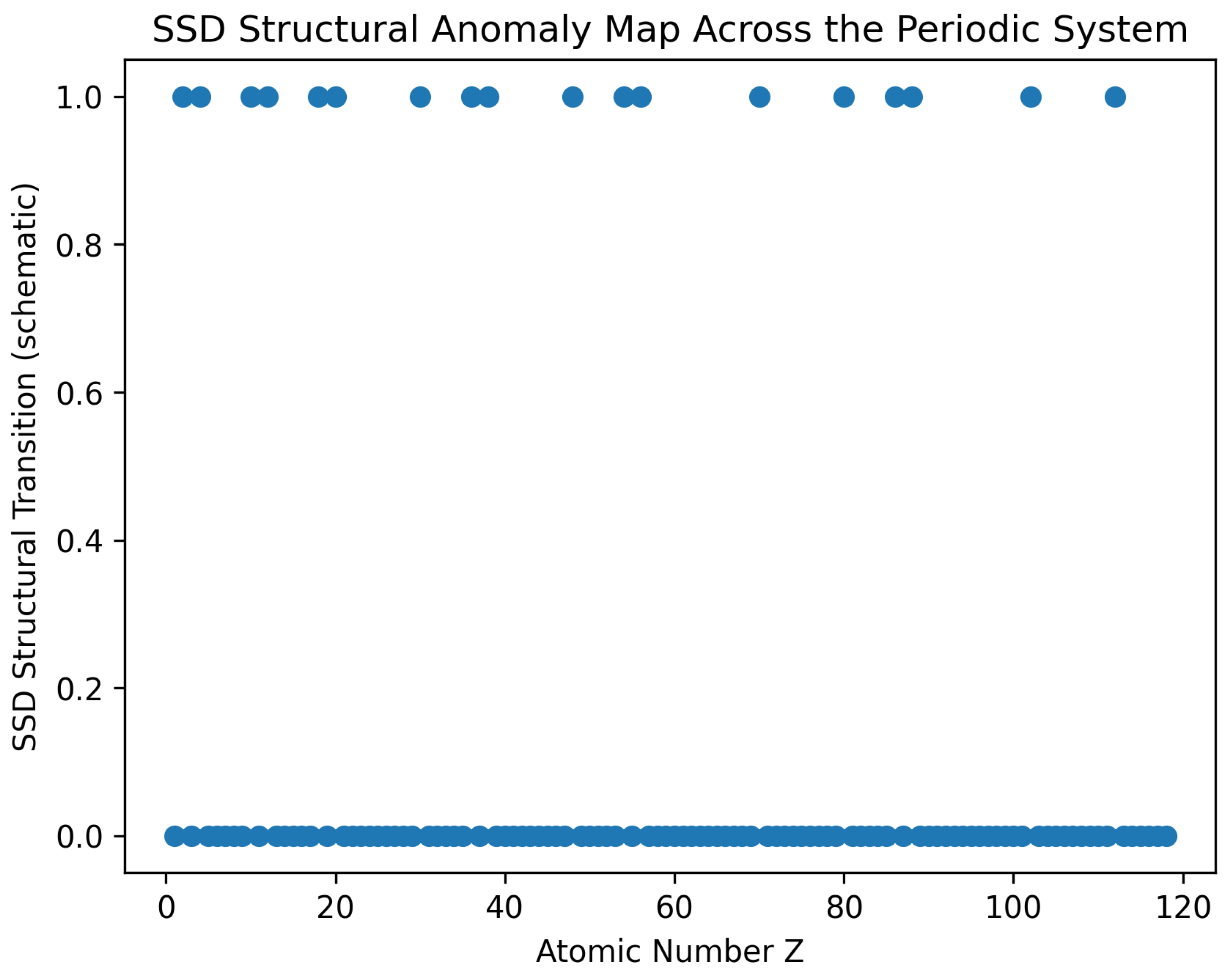
4.4. Taxonomy of SSD Structural Transitions
| Type | Structural Origin | Example |
| T1 | Orbital transition | Be–B |
| T2 | Shell closure | Cl–Ar |
| T3 | Principal shell jump | Ar–K |
| T4 | Half-filled stabilization | Cr–Mn |
| T5 | Subshell reconstruction | Fe–Co–Ni |
5. Advanced Analyses
5.1. Discrimination of Nuclear Anomalies
- Ar–K inversion remains present using monoisotopic masses, confirming a nuclear origin.
- Co–Ni and Te–I inversions also remain robust.
- Actinide anomalies disappear when monoisotopic masses are used, revealing isotopic abundance effects.
5.2. Electronegativity and Ionization Energy
5.3. Correlation Analysis: Synchronous Ticks
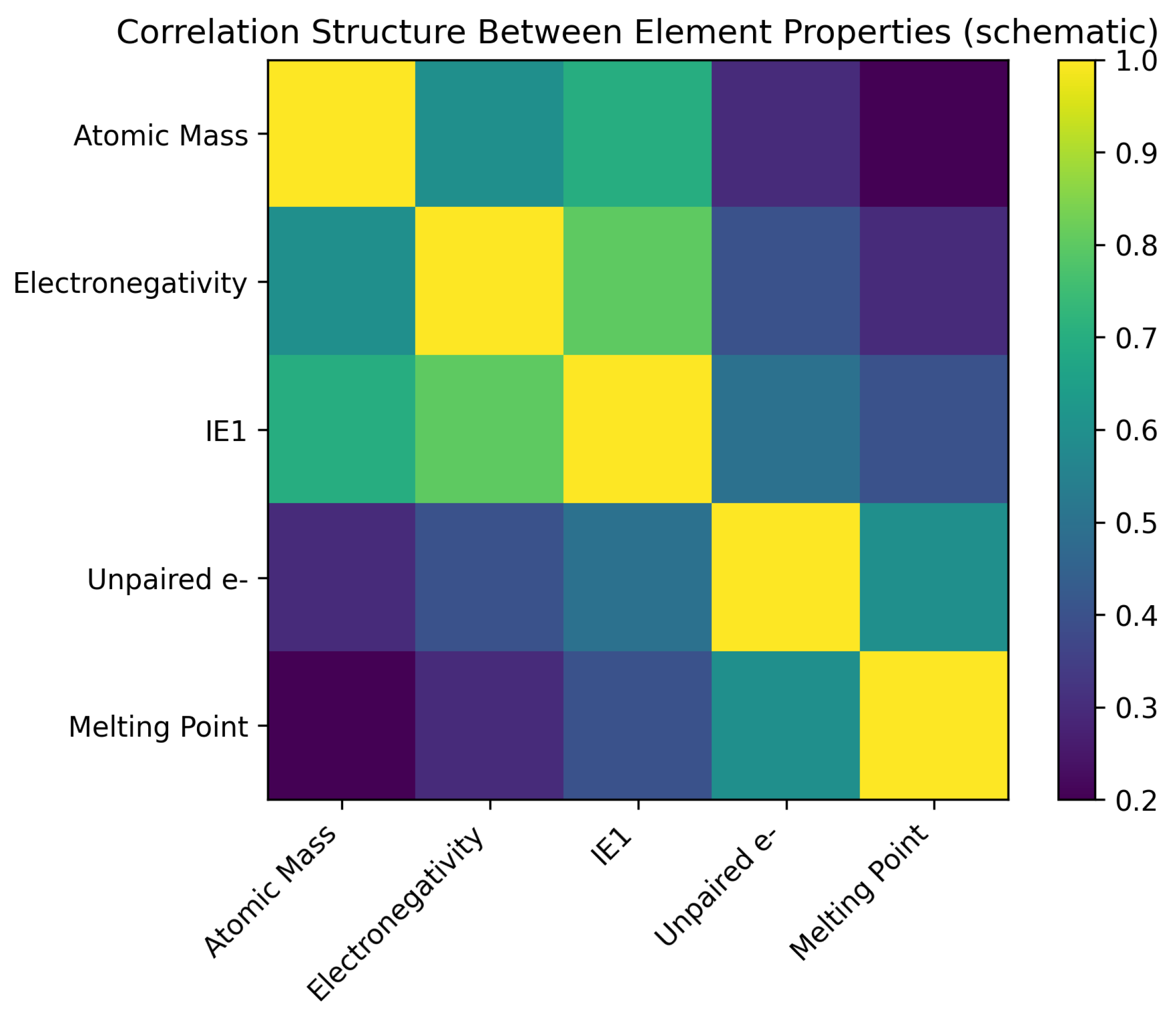
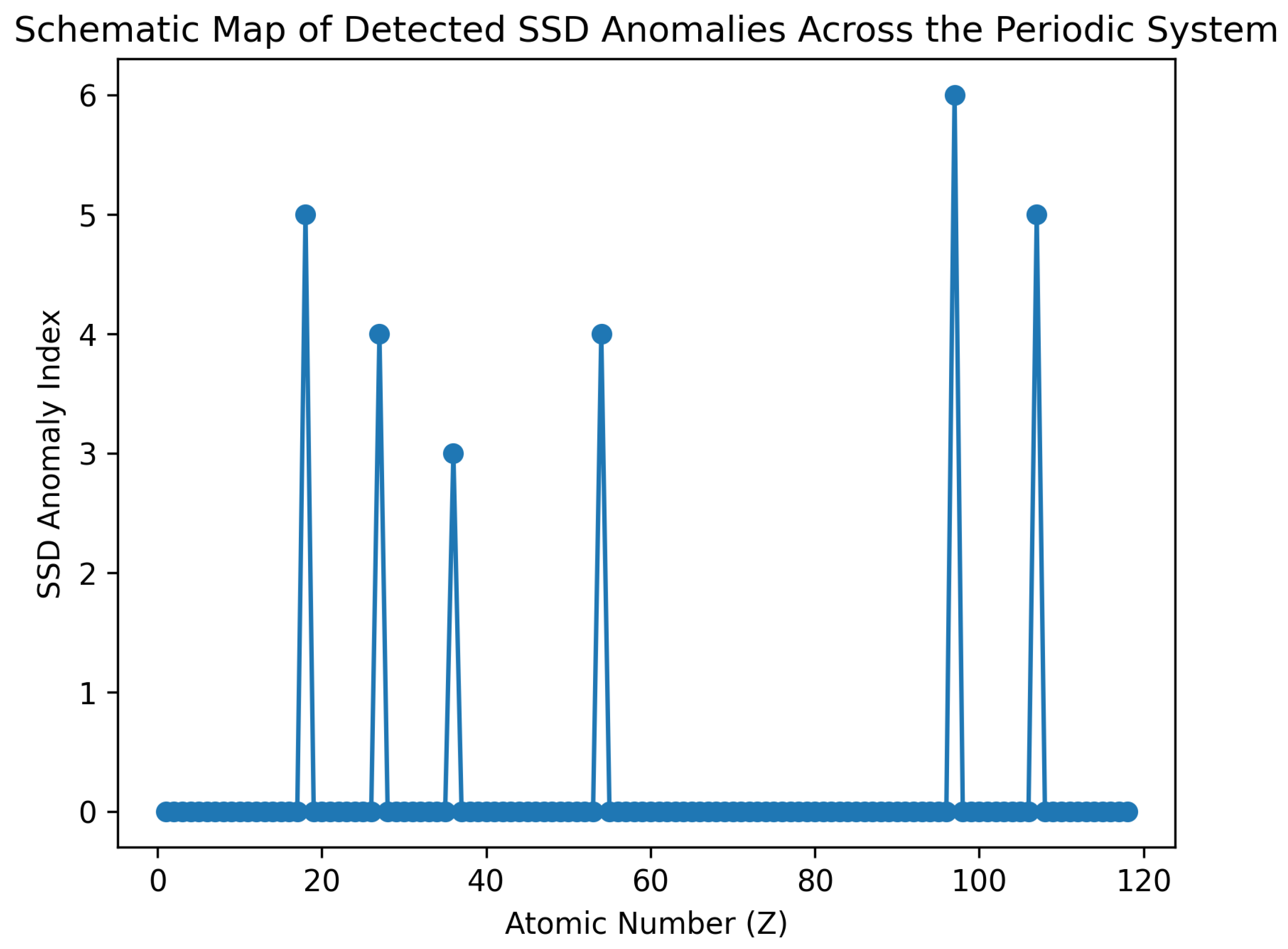
- Cl–Ar–K
- I–Xe–Cs
5.4. Element SSD Fingerprint
6. Predictions for Superheavy Elements
| Element | Predicted Isotope | Expected SSD Code | Behavior |
| Z=119 (Uue) | 297Uue | 18 | Plateau-to-growth transition |
| Z=120 (Ubn) | 302Ubn | 0 | Stable monotonic growth |
7. Discussion
- distinguishing artifacts from genuine nuclear anomalies,
- objectively identifying periodic boundaries,
- providing multidimensional numerical profiles of elements,
- mapping complex non-monotonic properties such as melting points.
- Dependence on data quality, especially for superheavy elements.
- Local nature of the method due to window size .
- Descriptive character; SSD detects anomalies but does not explain them.
8. Conclusion
- SSD analysis can distinguish genuine nuclear anomalies from isotopic artifacts.
- Synchronous SSD ticks across multiple properties identify critical periodic boundaries.
- The SSD vector provides a unique multidimensional fingerprint for each element.
- Numerical periodicity of SSD patterns independently confirms classical periodicity.
- The method successfully quantifies complex properties such as melting points.
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Scerri, E. The Periodic Table: Its Story and Its Significance. Oxford University Press.
- Jensen, W.B. The Origin of the Periodic Table. Journal of Chemical Education.
- Pyykkö, P. Relativistic Effects in Chemistry. Chemical Reviews.
- Schwarz, W.H.E. The Full Story of the Electron Configurations of the Periodic Table. Journal of Chemical Education.
- IUPAC (2025). Standard Atomic Weights. https://www.iupac.org/atomic-weights/.
- NIST (2025). Atomic Weights and Isotopic Compositions.
- Rumble, J.R. (Ed.) CRC Handbook of Chemistry and Physics, 100th Edition.
- NIST Chemistry WebBook SRD 69.
- NIST Atomic Weights and Isotopic Composition Database.
- Pangarić, Z. Symbolic Geometry of the Number π: Structures, Statistics, and Security. Preprints (2026).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



