Submitted:
05 March 2026
Posted:
09 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Population
2.2. Histopathological Examination
2.3. Immunohistochemistry
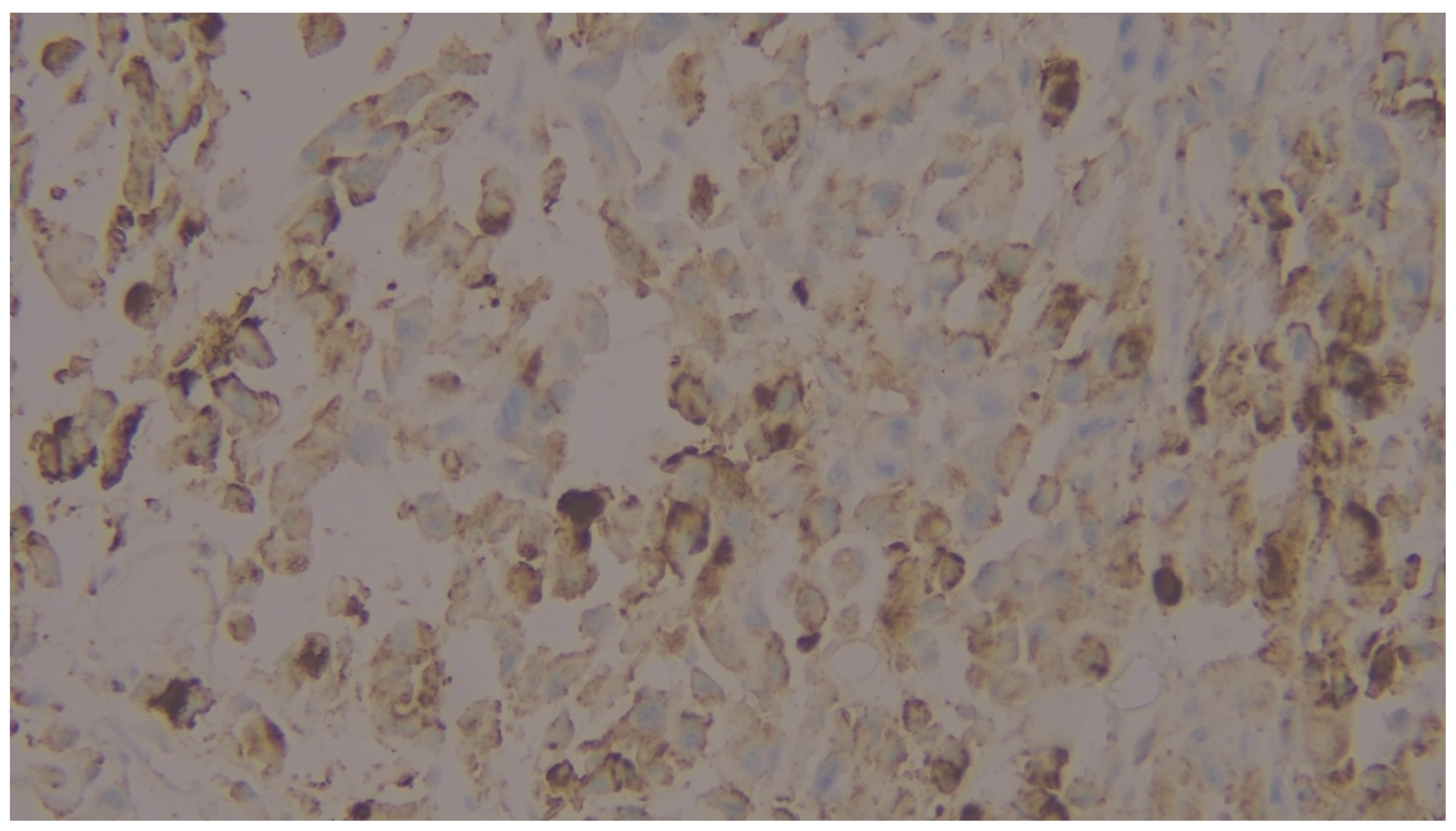
2.4. Evaluation of PD-L1 Expression
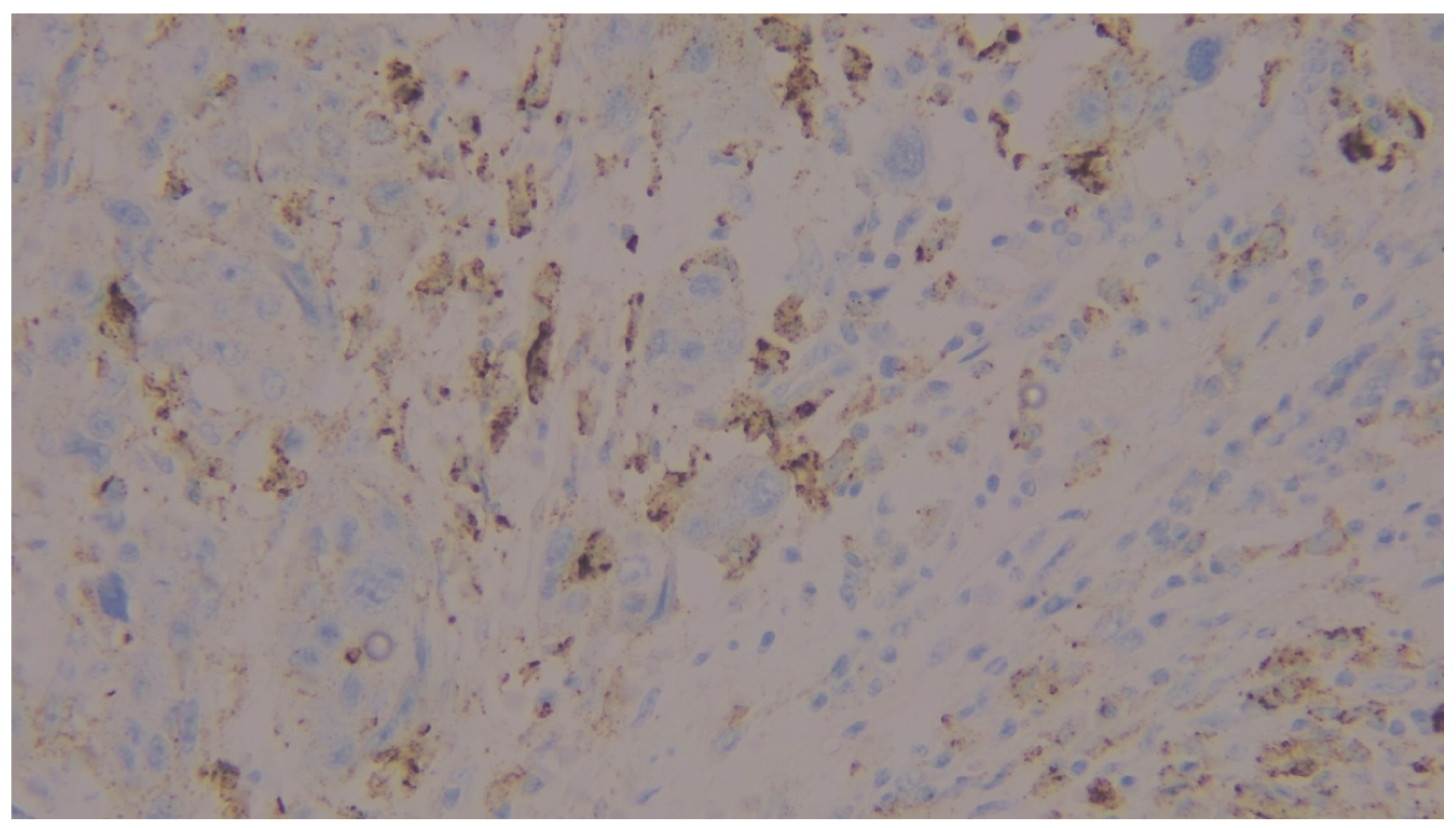
2.5. Evaluation of TILs
2.6. Statistical Analysis
3. Results
4. Discussion
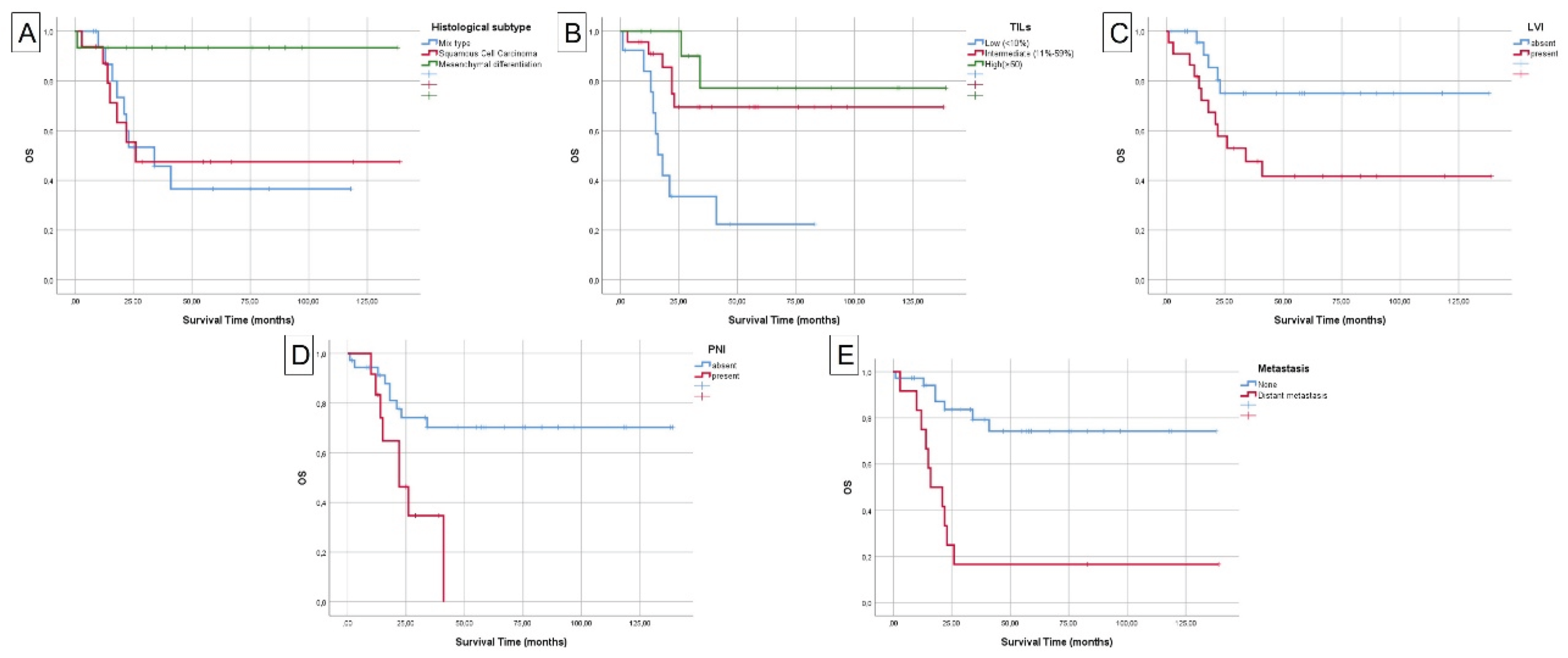
4.1. PD-L1 Expression Rates in Metaplastic Breast Cancer
4.2. Correlation with Clinicopathological Features
4.3. The Paradoxical Inverse Relationship: PD-L1 Negativity and Distant Metastasis
4.4. Clinical Implications of the PD-L1-Metastasis Relationship
4.5. Prognostic Significance of Tumor-Infiltrating Lymphocytes
4.6. Relationship Between PD-L1 Expression and TILs
4.7. Lymphovascular Invasion as an Independent Prognostic Factor
4.8. Perineural Invasion and Other Prognostic Factors
4.9. Metastasis Status and Overall Survival
4.10. Therapeutic Implications for Immunotherapy in MBC
4.11. TILs as a Synergistic Prognostic Biomarker
4.12. Technical Challenges and Assay Standardization
4.13. Study Limitations
- Dynamic Expression: PD-L1 and TILs were assessed only in primary specimens. Given that PD-L1 expression can evolve during metastasis or in response to therapy, paired primary-metastatic analyses are needed [38].
- Biomarker Scope: We did not evaluate genomic signatures (e.g., TMB) or alternative checkpoints such as LAG-3, TIM-3, or CTLA-4. Composite biomarkers integrating genomic and proteomic data likely represent the next frontier in patient stratification [39].
- Immune Characterization: Our assessment was limited to the density of TILs. Future studies utilizing multiplex immunohistochemistry or flow cytometry are necessary to characterize specific T-cell subsets (e.g., CD8+, FoxP3+), thereby providing a more qualitative understanding of the immune response [29].
4.14. Future Directions and Research Perspectives
4.14.1. Dedicated Prospective Immunotherapy Trials
4.14.2. Investigating the PD-L1/Metastasis Paradox
4.14.3. Transitioning Toward Composite Biomarker Models
4.14.4. Advanced Spatial and Component-Specific Profiling
4.14.5. Strategies for Immunologically “Cold” Tumors
4.14.6. Dynamic and Minimally Invasive Monitoring
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- WHO Classification of Tumors Editorial Board. Breast Tumors, 5th ed.; International Agency for Research on Cancer: Lyon, France, 2019; Volume 2. [Google Scholar]
- Weigelt, B.; Kreike, B.; Reis-Filho, J.S. Metaplastic breast carcinomas are basal-like breast cancers: A genomic profiling analysis. Breast Cancer Res. Treat. 2009, 117, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Rakha, E.A.; Tan, P.H.; Vros, A.; Tse, G.M.; Putti, T.C.; Abdulla, K.H.; Zin, R.R.; Ellis, I.O. Morphological and immunophenotypic analysis of breast carcinomas with choriocarcinomatous differentiation. J. Clin. Pathol. 2007, 60, 1352–1356. [Google Scholar]
- Rayson, D.; Adjei, A.A.; Suman, V.J.; Wold, L.E.; Ingle, J.N. Metaplastic breast cancer: Prognosis and response to systemic therapy. Ann. Oncol. 1999, 10, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Luini, A.; Aguilar, M.; Gatti, G.; Fasani, R.; Botteri, E.; Brito, J.A.; Maisonneuve, P.; Vento, A.R.; Viale, G. Metaplastic carcinoma of the breast, an unusual disease with worse prognosis: The experience of the European Institute of Oncology and review of the literature. Breast Cancer Res. Treat. 2007, 101, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Gatalica, Z.; Joneja, U.; Ghazalpour, A.; Ivanovic, M.; Kimbrough, J.; Reddy, S.; Hanson, J.; Khan, S.; Vranic, S. Mutation and immune profiling of metaplastic breast cancer: Correlation with survival. PLoS ONE 2019, 14, e0224726. [Google Scholar]
- Voutilainen, S.; Heikkilä, P.; Bartkova, J.; Bartek, J.; Bützow, R.; Nevanlinna, H. Markers associated with genomic instability, immunogenicity and immune therapy responsiveness in Metaplastic carcinoma of the breast: Expression of γH2AX, pRPA2, P53, PD-L1 and tumor infiltrating lymphocytes in 76 cases. BMC Cancer 2022, 22, 1355. [Google Scholar] [CrossRef]
- Al-Hilli, Z.; Choong, G.; Keeney, M.G.; Visscher, D.W.; Ingle, J.N.; Goetz, M.P.; Nassar, A. Metaplastic breast cancer: A comprehensive review. Expert Rev. Anticancer Ther. 2021, 21, 305–313. [Google Scholar]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Sharma, P.; Allison, J.P. The future of immune checkpoint therapy. Science 2015, 348, 56–61. [Google Scholar] [CrossRef]
- Topalian, S.L.; Drake, C.G.; Pardoll, D.M. Immune checkpoint blockade: A common denominator approach to cancer therapy. Cancer Cell 2015, 27, 450–461. [Google Scholar] [CrossRef]
- Cortes, J.; Cescon, D.W.; Rugo, H.S.; Nowecki, Z.; Im, S.A.; Yusof, M.M.; Gallardo, C.; Lipatov, O.; Barrios, C.H.; Holgado, E.; et al. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for previously untreated locally recurrent inoperable or metastatic triple-negative breast cancer (KEYNOTE-355): A randomised, placebo-controlled, double-blind, phase 3 clinical trial. Lancet 2020, 396, 1817–1828. [Google Scholar] [CrossRef]
- Schmid, P.; Adams, S.; Rugo, H.S.; Schneeweiss, A.; Barrios, C.H.; Iwata, H.; Diéras, V.; Hegg, R.; Im, S.A.; Shaw Wright, G.; et al. Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer. N. Engl. J. Med. 2018, 379, 2108–2121. [Google Scholar] [CrossRef]
- Gatalica, Z.; Joneja, U.; Ghazalpour, A.; Ivanovic, M.; Kimbrough, J.; Reddy, S.; Hanson, J.; Khan, S.; Vranic, S. Comprehensive Profiling of Metaplastic Breast Carcinoma Reveals Frequent Over-Expression of PD-L1. Cancer Res. 2015, 75 (Suppl. S9). [Google Scholar]
- Lien, H.C.; Hsiao, Y.H.; Lin, Y.S.; Yao, Y.T.; Juan, H.F.; Kuo, W.H.; Hung, M.C.; Chang, K.J.; Hsieh, F.J. Molecular signatures of metaplastic carcinoma of the breast by large-scale transcriptional profiling: Identification of genes potentially related to epithelial-mesenchymal transition. Oncogene 2007, 26, 7859–7871. [Google Scholar] [CrossRef]
- Joneja, U.; Vranic, S.; Swensen, J.; Feldman, R.; Chen, W.; Kimbrough, J.; Xiao, N.; Reddy, S.; Palazzo, J.; Gatalica, Z. Evaluation of immune density, PD-L1, and CXCR4 expressions in metaplastic breast carcinoma to predict potential immunotherapy benefit. Med. Oncol. 2024, 41, 22. [Google Scholar]
- Basho, R.K.; Yam, C.; Gilcrease, M.; Murthy, R.K.; Helgason, T.; Karp, D.D.; Meric-Bernstam, F.; Hess, K.R.; Valero, V.; Albarracin, C.; et al. Comparative Effectiveness of an mTOR-Based Systemic Therapy Regimen in Advanced, Metaplastic and Nonmetaplastic Triple-Negative Breast Cancer. Oncologist 2018, 23, 1300–1309. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.S.; Badve, S.; Wang, K.; Sheehan, C.E.; Boguniewicz, A.B.; Otto, G.A.; Yelensky, R.; Lipson, D.; Ali, S.; Morosini, D.; et al. Genomic profiling of advanced-stage, metaplastic breast carcinoma by next-generation sequencing reveals frequent, targetable genomic abnormalities and potential new treatment options. Arch. Pathol. Lab. Med. 2015, 139, 642–649. [Google Scholar] [CrossRef] [PubMed]
- Salgado, R.; Denkert, C.; Demaria, S.; Sirtaine, N.; Klauschen, F.; Pruneri, G.; Wienert, S.; Van den Eynden, G.; Baehner, F.L.; Penault-Llorca, F.; et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: Recommendations by an International TILs Working Group 2014. Ann. Oncol. 2015, 26, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Loi, S.; Drubay, D.; Adams, S.; Pruneri, G.; Francis, P.A.; Lacroix-Triki, M.; Joensuu, H.; Dieci, M.V.; Badve, S.; Demaria, S.; et al. Tumor-infiltrating lymphocytes and prognosis: A pooled individual patient analysis of early-stage triple-negative breast cancers. J. Clin. Oncol. 2019, 37, 559–569. [Google Scholar] [CrossRef]
- Edenfield, J.; Schammel, D.; Collins, J.; Schammel, C.; Edenfield, W.J. Metaplastic breast cancer: Molecular typing and identification of potential targeted therapies at a single institution. Clin. Breast Cancer 2017, 17, e1–e10. [Google Scholar] [CrossRef]
- Bagbudar, S.; Aydogdu, O.; Akyol, M.; Gokoz Dogu, G.; Yilmaz, E.; Aktas, S.; Sezgin Goksu, S. Evaluation of immune density, PD-L1, and CXCR4 expressions in metaplastic breast carcinoma to predict potential immunotherapy benefit. Med. Oncol. 2023, 41, 18. [Google Scholar] [CrossRef]
- Grabenstetter, A.; Jungbluth, A.A.; Frosina, D.; Bay, C.; Zeck, J.; Merghoub, T.; Shoushtari, A.N.; Snuderl, M.; Desrichard, A.; Havel, J.J.; et al. PD-L1 Expression in Metaplastic Breast Carcinoma Using the PD-L1 SP142 Assay and Concordance Among PD-L1 Immunohistochemical Assays. Am. J. Surg. Pathol. 2021, 45, 1612–1621. [Google Scholar] [CrossRef] [PubMed]
- Vinay, D.S.; Ryan, E.P.; Pawelec, G.; Talib, W.H.; Stagg, J.; Elkord, E.; Lichtor, T.; Decker, W.K.; Whelan, R.L.; Kumara, H.M.; et al. Immune evasion in cancer: Mechanistic basis and therapeutic strategies. Semin. Cancer Biol. 2015, 35 (Suppl.), S185–S198. [Google Scholar] [CrossRef] [PubMed]
- Beatty, G.L.; Gladney, W.L. Immune escape mechanisms as a guide for cancer immunotherapy. Clin. Cancer Res. 2015, 21, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef]
- Taube, J.M.; Anders, R.A.; Young, G.D.; Xu, H.; Sharma, R.; McMiller, T.L.; Chen, S.; Klein, A.P.; Pardoll, D.M.; Topalian, S.L.; et al. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci. Transl. Med. 2012, 4, 127ra37. [Google Scholar] [CrossRef]
- Teng, M.W.; Ngiow, S.F.; Ribas, A.; Smyth, M.J. Classifying Cancers Based on T-cell Infiltration and PD-L1. Cancer Res. 2015, 75, 2139–2145. [Google Scholar] [CrossRef]
- Chao, X.; Liu, L.; Sun, P.; Yang, C.; Deng, Q.; Liu, Q.; Mao, W. Immune parameters associated with survival in metaplastic breast cancer. Breast Cancer Res. 2020, 22, 92. [Google Scholar] [CrossRef]
- Kim, I.; Rajamanickam, V.; Bernard, B.; Abramson, R.G.; Pockaj, B.; Maughan, B.L.; Agarwal, N. A Case Series of Metastatic Metaplastic Breast Carcinoma Treated With Anti-PD-1 Therapy. Front. Oncol. 2021, 11, 635237. [Google Scholar] [CrossRef]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.J.; Cowey, C.L.; Lao, C.D.; Schadendorf, D.; Dummer, R.; Smylie, M.; Rutkowski, P.; et al. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N. Engl. J. Med. 2015, 373, 23–34. [Google Scholar] [CrossRef]
- Galluzzi, L.; Buqué, A.; Kepp, O.; Zitvogel, L.; Kroemer, G. Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 2017, 17, 97–111. [Google Scholar] [CrossRef] [PubMed]
- André, F.; Ciruelos, E.; Rubovszky, G.; Campone, M.; Loibl, S.; Rugo, H.S.; Iwata, H.; Conte, P.; Mayer, I.A.; Kaufman, B.; et al. Alpelisib for PIK3CA-Mutated, Hormone Receptor-Positive Advanced Breast Cancer. N. Engl. J. Med. 2019, 380, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Fridman, W.H.; Pagès, F.; Sautès-Fridman, C.; Galon, J. The immune contexture in human tumors: Impact on clinical outcome. Nat. Rev. Cancer 2012, 12, 298–306. [Google Scholar] [CrossRef]
- Mohammed, R.A.; Martin, S.G.; Gill, M.S.; Green, A.R.; Paish, E.C.; Ellis, I.O. Improved methods of detection of lymphovascular invasion demonstrate that it is the predominant method of vascular invasion in breast cancer and has important clinical consequences. Am. J. Surg. Pathol. 2007, 31, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, P.; Shekarian, T.; Alcazer, V.; Valladeau-Guilemond, J.; Valsesia-Wittmann, S.; Amigorena, S.; Caux, C.; Depil, S. Cold Tumors: A Therapeutic Challenge for Immunotherapy. Front. Immunol. 2019, 10, 168. [Google Scholar] [CrossRef] [PubMed]
- Paver, E.C.; Cooper, W.A.; Colebatch, A.J.; Ferguson, P.M.; Hill, S.K.; Lum, T.; Shin, J.S.; O’Toole, S.; Anderson, L.; Scolyer, R.A.; et al. Programmed death ligand-1 (PD-L1) as a predictive marker for immunotherapy in solid tumors: A guide to immunohistochemistry implementation and interpretation. Pathology 2021, 53, 141–156. [Google Scholar] [CrossRef]
- Madore, J.; Vilain, R.E.; Menzies, A.M.; Kakavand, H.; Wilmott, J.S.; Hyman, J.; Yearley, J.H.; Kefford, R.F.; Thompson, J.F.; Long, G.V.; et al. PD-L1 expression in melanoma shows marked heterogeneity within and between patients: Implications for anti-PD-1/PD-L1 clinical trials. Pigment Cell Melanoma Res. 2015, 28, 245–253. [Google Scholar] [CrossRef]
- Danaher, P.; Warren, S.; Dennis, L.; D’Amico, L.; White, A.; Disis, M.L.; Geller, M.A.; Odunsi, K.; Beechem, J.; Fling, S.P. Gene expression markers of Tumor Infiltrating Leukocytes. J. Immunother. Cancer 2017, 5, 18. [Google Scholar] [CrossRef]
| n | % | ||
| Histological subtype | Mix type | 17 | 35.4 |
| Squamous Cell Carcinoma | 16 | 33.3 | |
| Mesenchymal differentiation | 15 | 31.3 | |
| PD-L1 22C3 CPS category | Negative | 13 | 27.1 |
| Positive | 35 | 72.9 | |
| Tumor-infiltrating lymphocytes (TILs) | Low (<10%) | 13 | 27.1 |
| Intermediate (11%-59%) | 23 | 47.9 | |
| High(≥60) | 12 | 25.0 | |
| Ductal carcinoma in situ | Absent | 42 | 87.5 |
| Present | 6 | 12.5 | |
| Pathological category | 0-20 | 4 | 8.3 |
| 21-50 | 24 | 50.0 | |
| >50 | 20 | 41.7 | |
| T staging |
1 | 2 | 4.2 |
| 2 | 26 | 54.2 | |
| 3 | 13 | 27.1 | |
| 4 | 7 | 14.6 | |
| Lymph node category |
Negative | 26 | 54.2 |
| 1-3 | 13 | 27.1 | |
| 4-10 | 7 | 14.6 | |
| >10 | 2 | 4.2 | |
| Lymphovascular invasion |
Absent | 25 | 52.1 |
| Present | 23 | 47.9 | |
| N staging |
Negative | 26 | 54.2 |
| 1 | 13 | 27.1 | |
| 2 | 7 | 14.6 | |
| 3 | 2 | 4.2 | |
| Neoadjuvant therapy | Absent | 28 | 58.3 |
| Present | 20 | 41.7 | |
| Response to neoadjuvant therapy | No response | 5 | 25.0 |
| Partial response | 12 | 60.0 | |
| Complete response | 3 | 15.0 | |
| Surgical lymph node dissection |
Absent | 15 | 31.3 |
| Present | 33 | 68.8 | |
| ERBB2 (HER2) |
0 | 37 | 77.1 |
| 1 | 2 | 4.2 | |
| 2 | 7 | 14.6 | |
| 3 | 2 | 4.2 | |
| Perineural invasion | Absent | 36 | 75.0 |
| Present | 12 | 25.0 | |
| Adjuvant therapy | Absent | 18 | 37.5 |
| Present | 30 | 62.5 | |
| Adjuvant radiotherapy |
Absent | 33 | 68.8 |
| Present | 15 | 31.3 | |
| Metastasis | Distant metastasis | 12 | 25.0 |
| None | 36 | 75.0 | |
| PD-L1 | p | |||
| Negative | Positive | |||
| n(%) | n(%) | |||
| Histological subtype | Mix type | 3(23.1) | 14(40) | 0.439 |
| Squamous Cell Carcinoma | 6(46.2) | 10(28.6) | ||
| Mesenchymal differentiation | 4(30.8) | 11(31.4) | ||
| Squamous component | Absent | 4(30.8) | 11(31.4) | 0.965 |
| Present | 9(69.2) | 24(68.6) | ||
| TILs | Low (<10/%) | 4(30.8) | 9(25.7) | 0.719 |
| Intermediate (11/%-59/%) | 5(38.5) | 18(51.4) | ||
| High(≥60) | 4(30.8) | 8(22.9) | ||
| pT Stage | 1 | 1(7.7) | 1(2.9) | 0.655 |
| 2 | 8(61.5) | 18(51.4) | ||
| 3 | 2(15.4) | 11(31.4) | ||
| 4 | 2(15.4) | 5(14.3) | ||
| LVI | Absent | 5(38.5) | 21(60) | 0.210 |
| Present | 8(61.5) | 14(40) | ||
| Lymph node count | Negative | 5(38.5) | 21(60) | 0.531 |
| 1-3 | 5(38.5) | 8(22.9) | ||
| 4-10 | 2(15.4) | 5(14.3) | ||
| >10 | 1(7.7) | 1(2.9) | ||
| Metastasis | None | 7(53.8) | 29(82.9) | 0.039 |
| Distant metastasis | 6(46.2) | 6(17.1) | ||
| Status | Alive | 7(53.8) | 24(68.6) | 0.498 |
| Ex | 6(46.2) | 11(31.4) | ||
| TILs | p | ||||
| Low (<10%) |
Intermediate (11%-59/%) |
High (≥60) |
|||
| n(%) | n(%) | n(%) | |||
| Histological subtype | Mix type | 7(53.8) | 7(30.4) | 3(25.0) | 0.158 |
| Squamous Cell Carcinoma | 2(15.4) | 7(30.4) | 7(58.3) | ||
| Mesenchymal differentiation | 4(30.8) | 9(39.2) | 2(16.7) | ||
| Squamous component | Absent | 4(30.8) | 9(39.1) | 2(16.7) | 0.396 |
| Present | 9(69.2) | 14(60.9) | 10(83.3) | ||
| pT Stage | 1 | 0(0.0) | 2(8.7) | 0(0.0) | 0.625 |
| 2 | 7(53.8) | 11(47.8) | 8(66.6) | ||
| 3 | 5(38.5) | 6(26.1) | 2(16.7) | ||
| 4 | 1(7.7) | 4(17.4) | 2(16.7) | ||
| LVI | Absent | 6(46.2) | 16(69.6) | 4(33.3) | 0.099 |
| Present | 7(53.8) | 7(30.4) | 8(66.7) | ||
| Lymph node count | Negative | 6(46.1) | 16(69.6) | 4(33.3) | 0.282 |
| 1-3 | 3(23.1) | 4(17.4) | 6(50) | ||
| 4-10 | 3(23.1) | 2(8.7) | 2(16.7) | ||
| >10 | 1(7.7) | 1(4.3) | 0(0.0) | ||
| Metastasis | None | 7(53.8) | 19(82.6) | 10(83.3) | 0.119 |
| Distant metastasis | 6(46.2) | 4(17.4) | 2(16.7) | ||
| Status | Alive | 4(30.8) | 17(73.9) | 10(83.3) | 0.006 |
| Ex | 9(69.2) | 6(26.1) | 2(16.7) | ||
| Non-survivors | Survivors | p | ||
| n(%) | n(%) | |||
| Histological subtype | Mix type | 8(25.80) | 9(52.90) | 0,017 |
| Squamous Cell Carcinoma | 9(29.00) | 7(41.20) | ||
| Mesenchymal differentiation | 14(45.20) | 1(5.90) | ||
| PD-L1 22C3 CPS category | negative | 7(22.60) | 6(35.30) | 0,498 |
| positive | 24(77.40) | 11(64.70) | ||
| Tumor-Infiltrating Lymphocytes (TILs) | Low (<10%) | 4(12.90) | 9(52.90) | 0,006 |
| Intermediate (11/%-59/%) | 17(54.80) | 6(35.30) | ||
| High(≥60) | 10(32.30) | 2(11.80) | ||
| Ductal carcinoma in situ | absent | 27(87.10) | 15(88.20) | 0,909 |
| present | 4(12.90) | 2(11.80) | ||
| Tumor size | 0-20 | 3(9.70) | 1(5.90) | 0,203 |
| 21-50 | 18(58.10) | 6(35.30) | ||
| >50 | 10(32.30) | 10(58.80) | ||
| Multifocal | 0 | 31(100.00) | 15(88.20) | 0,121 |
| 1 | 0(0.00) | 2(11.80) | ||
| T staging | 1 | 1(3.20) | 1(5.90) | 0,115 |
| 2 | 20(64.50) | 6(35.30) | ||
| 3 | 8(25.80) | 5(29.40) | ||
| 4 | 2(6.50) | 5(29.40) | ||
| Lymphovascular invasion (LVI) | Absent | 21(67.70) | 4(29.10) | 0,011 |
| Present | 10(32.30) | 12(70.60) | ||
| N staging | 0 | 21(67.7) | 5(29.4) | 0,004 |
| 1 | 8(25.8) | 5(29.4) | ||
| 2 | 1(3.2) | 6(35.3) | ||
| 3 | 1(3.2) | 1(5.9) | ||
| Neoadjuvant therapy | Absent | 18(58.10) | 10(58.80) | 0,959 |
| Present | 13(41.90) | 7(41.20) | ||
| Response to neoadjuvant therapy | No response | 3(23.10) | 2(28.60) | 0,964 |
| Partial response | 8(61.50) | 4(57.10) | ||
| Complete response | 2(15.40) | 1(14.30) | ||
| Localization / Tumor location | Left | 16(51.60) | 7(41.20) | 0,489 |
| Right | 15(48.40) | 10(58.80) | ||
| ERBB2 (HER2) | 0 | 27(87.10) | 10(58.80) | 0,006 |
| 1 | 2(6.50) | 0(0.00) | ||
| 2 | 2(6.50) | 5(29.40) | ||
| 3 | 0(0.00) | 2(11.80) | ||
| HER2 FISH | Negative | 31(100.00) | 15(88.20) | 0,038 |
| Positive | 0(0.00) | 2(11.80) | ||
| Perineural invasion (PNI) | Absent | 27(87.10) | 9(52.90) | 0,015 |
| Present | 4(12.90) | 8(47.10) | ||
| Adjuvant therapy | Absent | 9(29.00) | 9(52.90) | 0,102 |
| Present | 22(71.00) | 8(47.10) | ||
| Adjuvant radiotherapy | Absent | 23(74.20) | 10(58.80) | 0,272 |
| Present | 8(25.80) | 7(41.20) | ||
| Metastasis | None | 29(93.50) | 7(41.20) | <0,001 |
| Distant metastasis | 2(6.50) | 10(58.80) | ||
| HR | 95% CI for HR (Lower-Upper) |
p | |||
| Histological subtype | MD—reference | 0.390 | |||
| SCC | 4.508 | .494 | 41.170 | 0.182 | |
| Mixed | 4.637 | .448 | 48.022 | 0.198 | |
| TILs | High—reference | 0.046 | |||
| Low | 9.659 | 1.538 | 60.674 | 0.016 | |
| Intermediate | 7.444 | 1.166 | 47.531 | 0.034 | |
| LVI | 3.841 | 1.019 | 14.475 | 0.047 | |
| PNI | 1.394 | .464 | 4.184 | 0.554 | |
| Metastasis | 4.397 | 1.223 | 15.807 | 0.023 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




