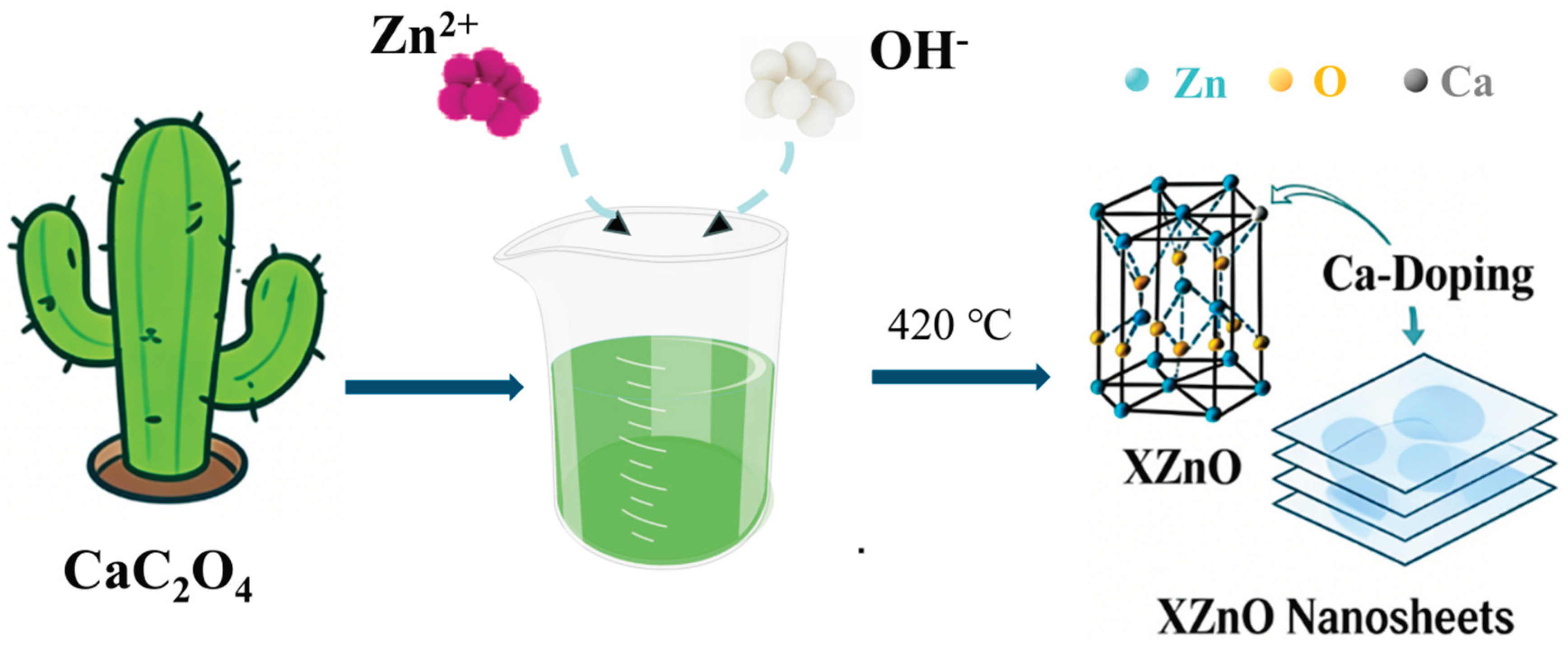
3.1. Material Structure and Morphology
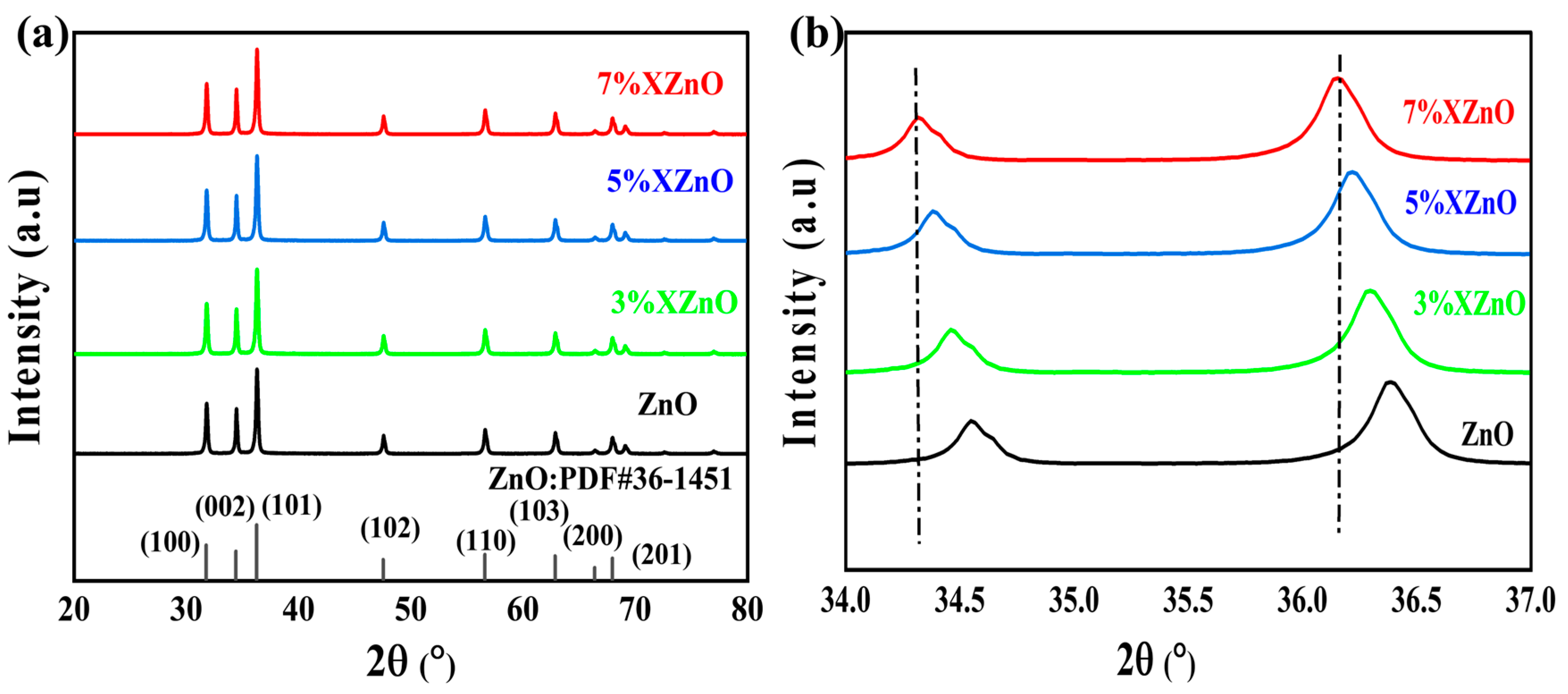
The crystalline structure of the synthesized samples was first investigated by X-ray diffraction (as shown in
Figure 2). All diffraction peaks for the pure and doped ZnO samples are perfectly indexed to the hexagonal wurtzite phase (PDF#36-1451), with no detectable peaks corresponding to CaO or other calcium-based compounds. This indicates that in-situ Ca doping, facilitated by the cactus juice medium, does not alter the fundamental ZnO crystal lattice. A magnified view of the (002) and (101) diffraction peaks (
Figure 2b) reveals a slight shift towards lower angles for the XZnO samples compared to pure ZnO. Consistent with Bragg's law, the decrease in diffraction angles points to an increased lattice spacing. This expansion arises from the partial displacement of native Zn
2+ (ionic radius: 74 pm) by the larger Ca
2+ dopant (100 pm), thereby verifying the effective doping of Ca into the ZnO lattice.
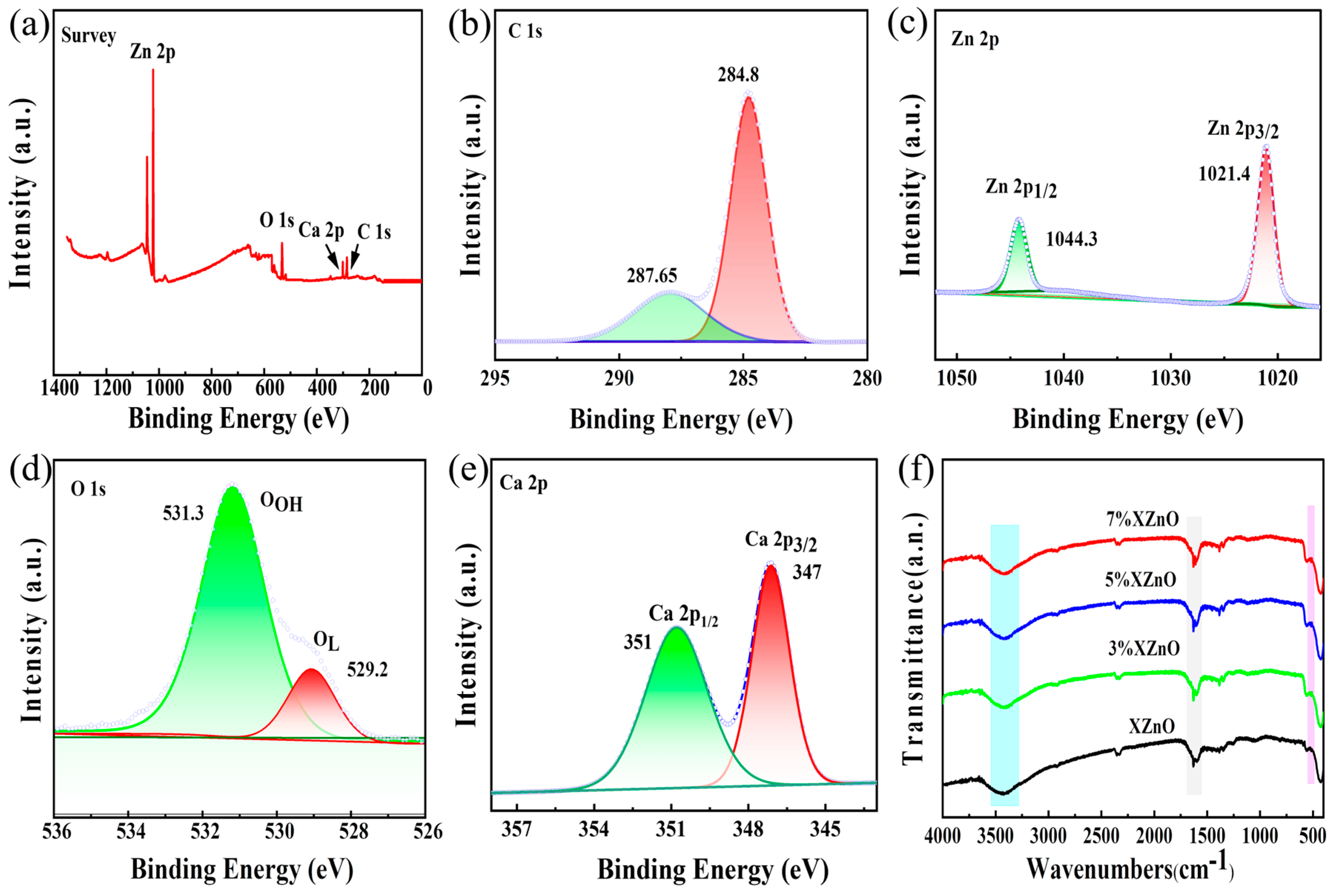
X-ray photoelectron spectroscopy (XPS) was employed to analyze the surface chemical composition and elemental states. The survey spectra in
Figure 3a-b confirm the presence of Zn, O, and Ca in the doped samples, with no other impurities detected. The high-resolution Zn 2p spectrum (
Figure 3c) shows two symmetric peaks at 1021.4 eV and 1044.3 eV, corresponding to Zn 2p
3/2 and Zn 2p
1/2, respectively, with a spin-orbit splitting of 22.9 eV, which is characteristic of the Zn
2+ oxidation state. The O 1s spectrum (
Figure 3d) is deconvoluted into two components: a peak at 529.2 eV attributed to lattice oxygen (O
L) and a peak at 531.3 eV associated with surface hydroxyl groups (O
OH) or oxygen-deficient regions. The presence of Ca is unambiguously confirmed by the doublet peaks in the Ca 2p spectrum (
Figure 3e) at approximately 347 eV and 351 eV, corresponding to Ca 2p
3/2 and Ca 2p
1/2, and confirming calcium's presence as Ca
2+.
Fourier-transform infrared (FT-IR) spectroscopy, shown in
Figure 3f, further supports these findings. All samples exhibit a strong absorption band around 500 cm
-1, characteristic of the Zn-O stretching vibration. Notably, this peak becomes sharper and more intense with increasing Ca content, suggesting an enhancement of the local Zn-O bond strength and overall structural rigidity. The broad bands around 3500 cm
-1 and the peak at 1650 cm
-1 correspond to O-H stretching and bending vibrations of adsorbed water molecules and surface hydroxyl groups. The increase in intensity of the 1650 cm
-1 peak with Ca doping is particularly significant, as a more hydrophilic surface with abundant -OH groups is known to facilitate water adsorption, a crucial first step in the photocatalytic water-splitting reaction.
The morphology and microstructure of the optimal 5%XZnO sample were examined using transmission electron microscopy (TEM). The TEM image in
Figure 4a-c reveal a well-defined, uniformly dispersed nanosheet morphology with minimal agglomeration, highlighting the effectiveness of the cactus-juice-mediated synthesis in controlling particle growth. High-resolution TEM (HRTEM) in
Figure 4d-e displays clear and continuous lattice fringes with an interplanar spacing of 0.25 nm, which corresponds perfectly to the (101) plane of wurtzite ZnO. Energy-dispersive X-ray spectroscopy (EDS) elemental mapping (
Figure 4g-i) displays a homogeneous distribution of Zn, O, and Ca across the entire nanosheet. The absence of any Ca-rich clusters or secondary phases provides compelling evidence for the uniform incorporation of Ca
2+ ions into the ZnO lattice, rather than simple surface adsorption or formation of separate CaO domains.
3.2. Optical Properties and Band Structure Evolution
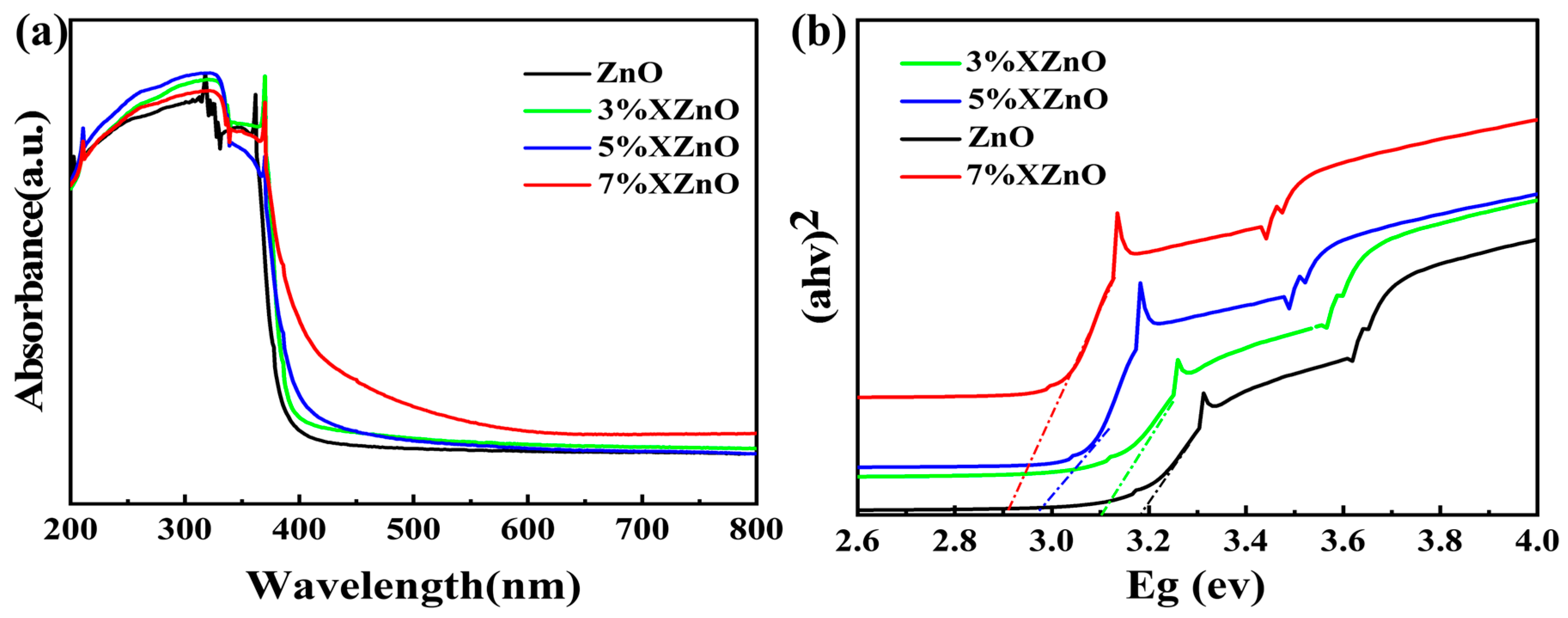
To evaluate the influence of Ca incorporation on the optical characteristics of the prepared samples, UV-vis diffuse reflectance spectroscopy (DRS) was employed. As presented in
Figure 5a, the spectrum of undoped ZnO exhibits a distinct absorption threshold located at around 390 nm within the ultraviolet region, while its response to visible light remains minimal. Upon Ca doping, a progressive red-shift of the absorption edge is observed, accompanied by a significant increase in visible light absorption. The corresponding bandgap energies, determined from Tauc plots (
Figure 5b), decrease monotonically from 3.19 eV for pure ZnO to 2.90 eV for the 7% XZnO sample. This bandgap narrowing is attributed to the formation of impurity energy levels within the ZnO bandgap due to the substitutional Ca
2+ ions, effectively lowering the energy required for electronic transitions and extending the light absorption range into the visible spectrum.
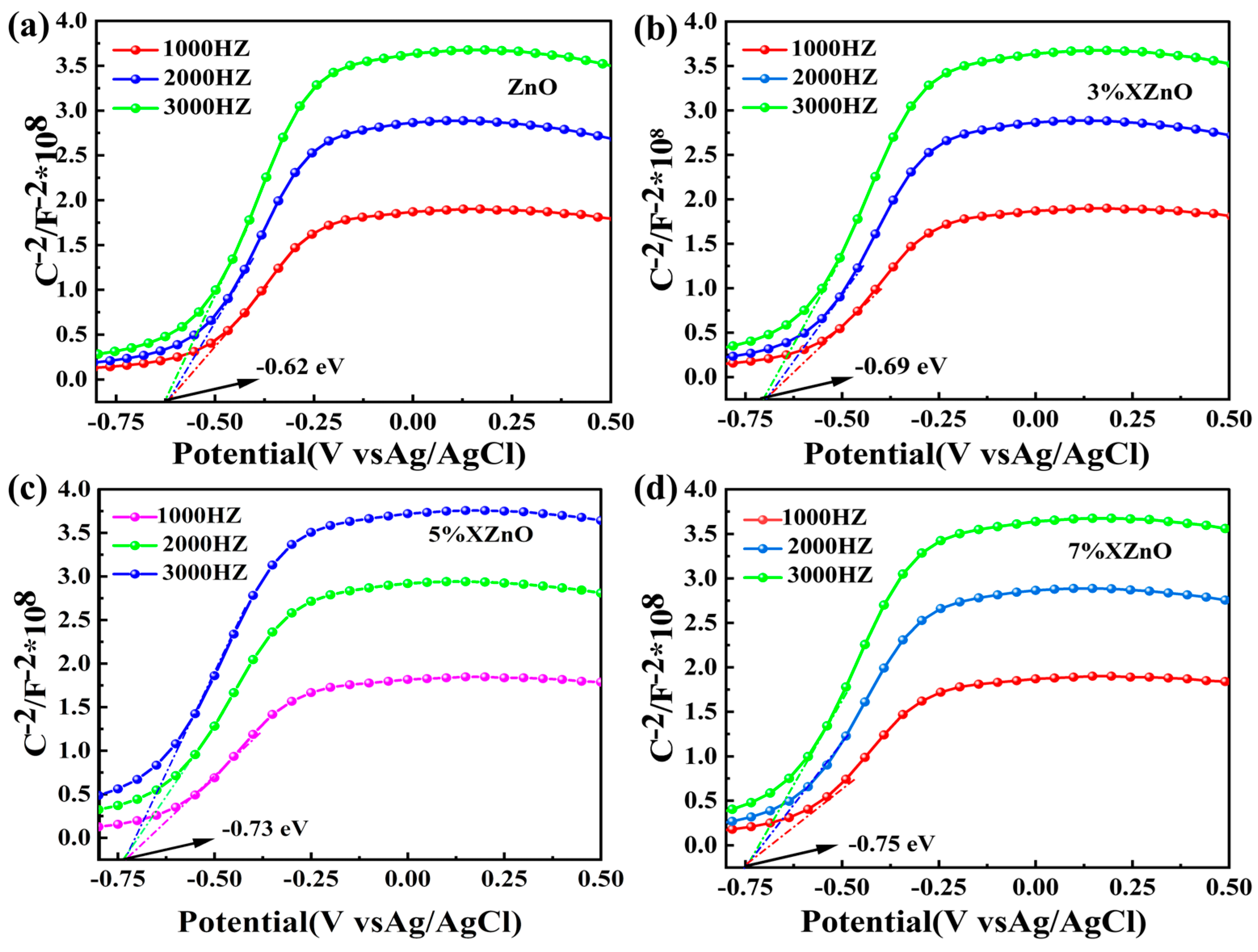
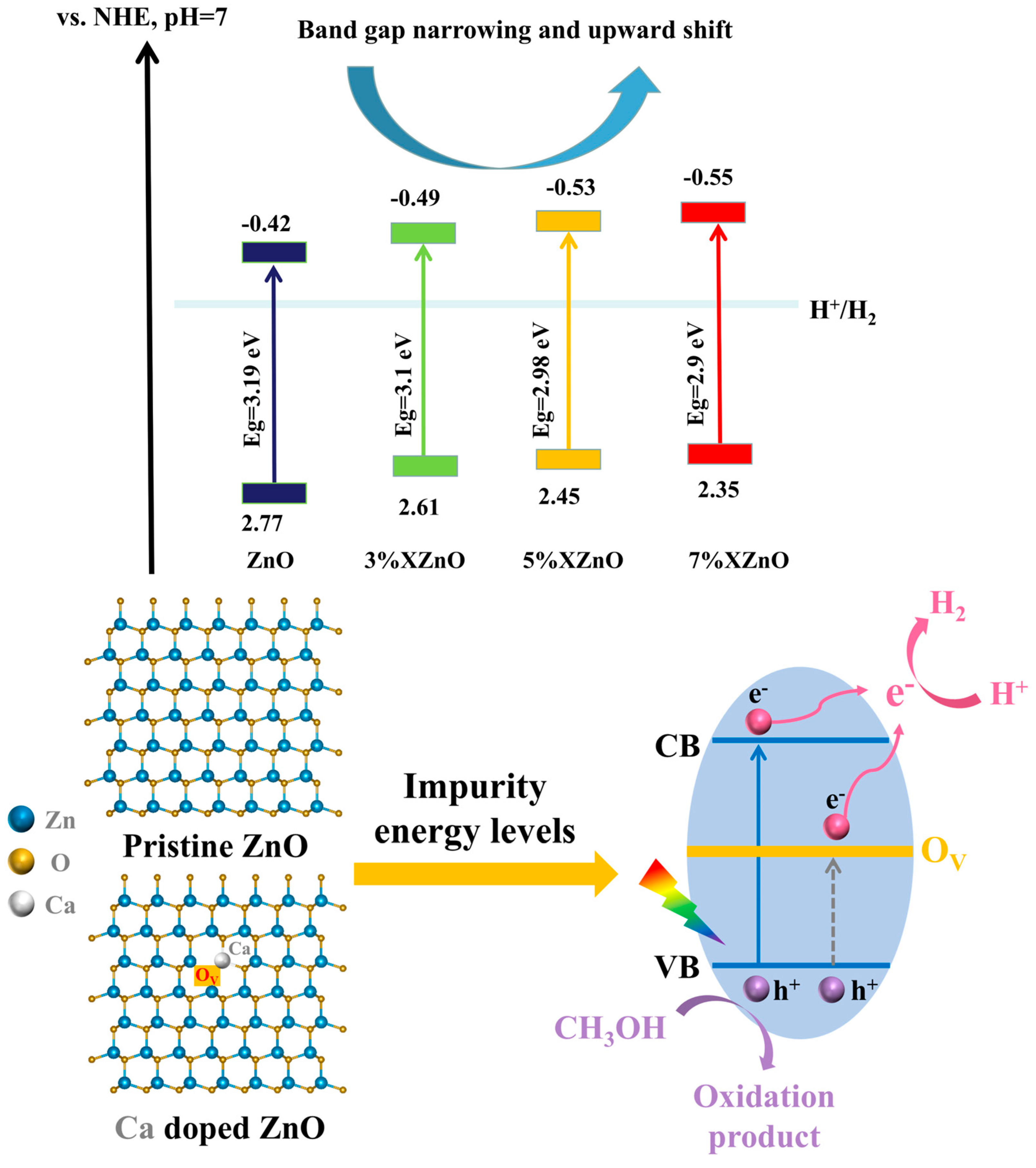
To elucidate the absolute band edge positions, Mott-Schottky (M-S) measurements were performed (
Figure 6). The positive slopes of the linear plots confirm the n-type semiconductor nature. The flat band potentials (E
FB), which for n-type semiconductors are approximately equal to the conduction band minimum (E
CB), were determined from the x-intercepts. After conversion to the standard hydrogen electrode (SHE) scale, the E
CB values are -0.42 V for pure ZnO, -0.49 V for 3%XZnO, -0.53 V for 5%XZnO, and -0.55 V for 7% XZnO. Using the bandgap energies (E
g) obtained from DRS, the valence band maxima (E
VB = E
CB + E
g) were calculated. The progressive negative shift of E
CB with increasing Ca content is particularly noteworthy, as it implies that the photogenerated electrons possess a stronger thermodynamic driving force for the proton reduction reaction (H
+/H
2), a key factor in enhancing photocatalytic hydrogen production.
3.3. Photocatalytic Hydrogen Production Performance
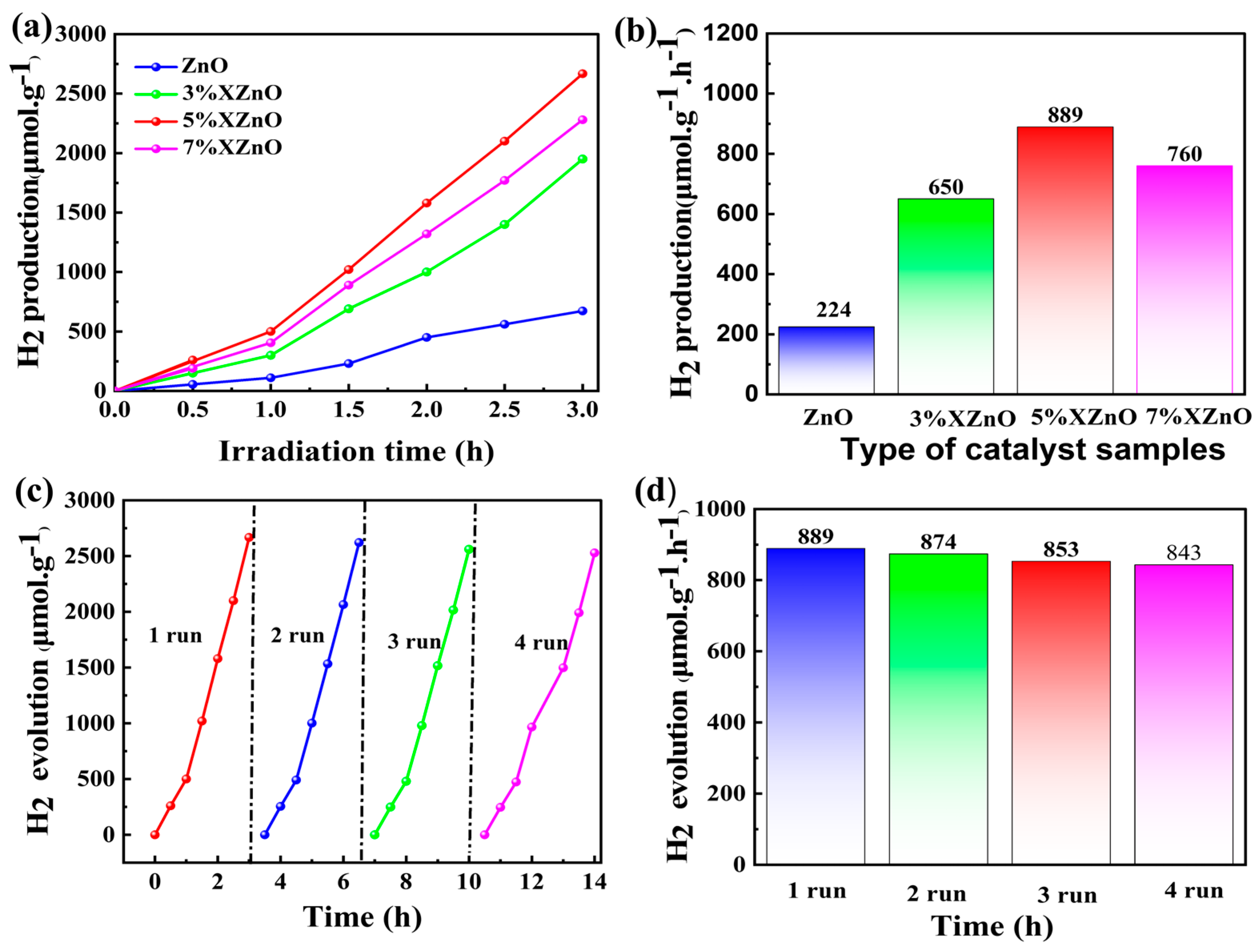
The photocatalytic activity of the synthesized samples for hydrogen evolution was evaluated under visible light irradiation (λ > 420 nm). As shown in
Figure 7a and
Figure 7b, pure ZnO exhibits negligible H
2 production, consistent with its wide bandgap and inability to absorb visible light. In stark contrast, all Ca-doped XZnO samples demonstrate significant and sustained H
2 evolution. The hydrogen evolution rate follows a volcano-type trend with doping concentration, initially increasing, reaching a maximum, and then decreasing. The 5%XZnO sample achieves the highest performance, with an impressive H
2 evolution rate of 889 μmol·g
-1·h
-1, which is about 4 times of the pristine ZnO. The hydrogen production performance of this work is compared with that of other similar published studies in
Table S1. This optimal doping level represents a balance between enhanced light absorption and charge separation and the introduction of excessive defects that can act as recombination centers, as observed for the 7%XZnO sample.
The stability of a photocatalyst is paramount for practical applications. The 5%XZnO sample was subjected to four consecutive cycling runs, totaling 14 hours of illumination (
Figure 7c). The catalyst exhibited remarkable stability, retaining 94.8% of its initial H
2 evolution rate in the fourth cycle (
Figure 7d). Post-reaction characterization (XRD and FT-IR, as shown in
Figure S1) confirmed that the crystal structure and chemical functional groups of the catalyst remained unchanged, underscoring its excellent resistance to photocorrosion and structural degradation, a key advantage conferred by the robust, Ca-doped lattice.
3.4. Photoelectrochemical Properties and Mechanism for Enhanced Activity
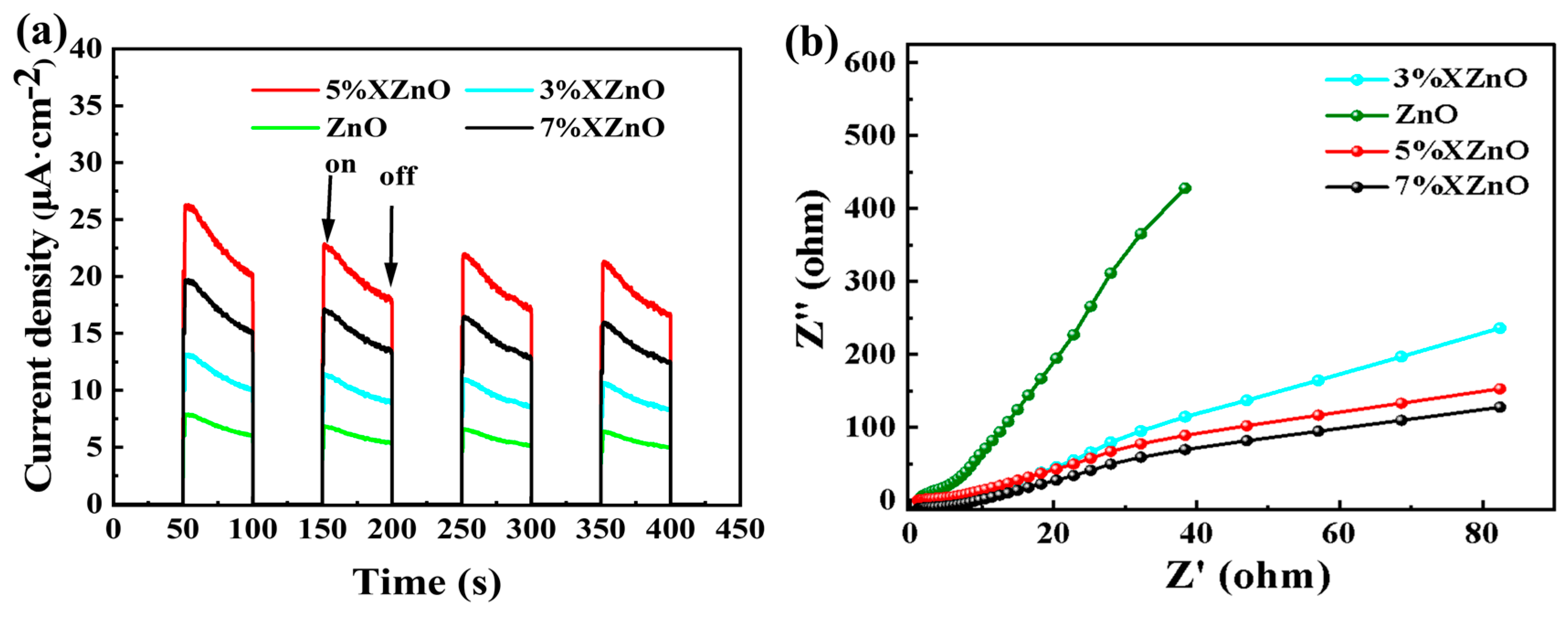
To gain deeper insight into the charge carrier dynamics, transient photocurrent responses and electrochemical impedance spectroscopy (EIS) were performed.
Figure 8a exhibits the photocurrent responses under intermittent visible light irradiation. The 5%XZnO sample generates the highest photocurrent density, which is more than five times that of pure ZnO. This directly demonstrates that optimal Ca doping dramatically enhances the efficiency of photogenerated electron-hole pair separation and collection. The EIS Nyquist plots in
Figure 8b further corroborate this finding.With the smallest arc radius in the EIS measurement, the 5%XZnO composition demonstrates superior conductivity, as it offers the least resistance to electron transfer across the electrode/electrolyte boundary. This implies faster interfacial charge migration kinetics, allowing more photogenerated electrons to participate in the surface reduction reaction for H
2 evolution [
12,
13,
14].
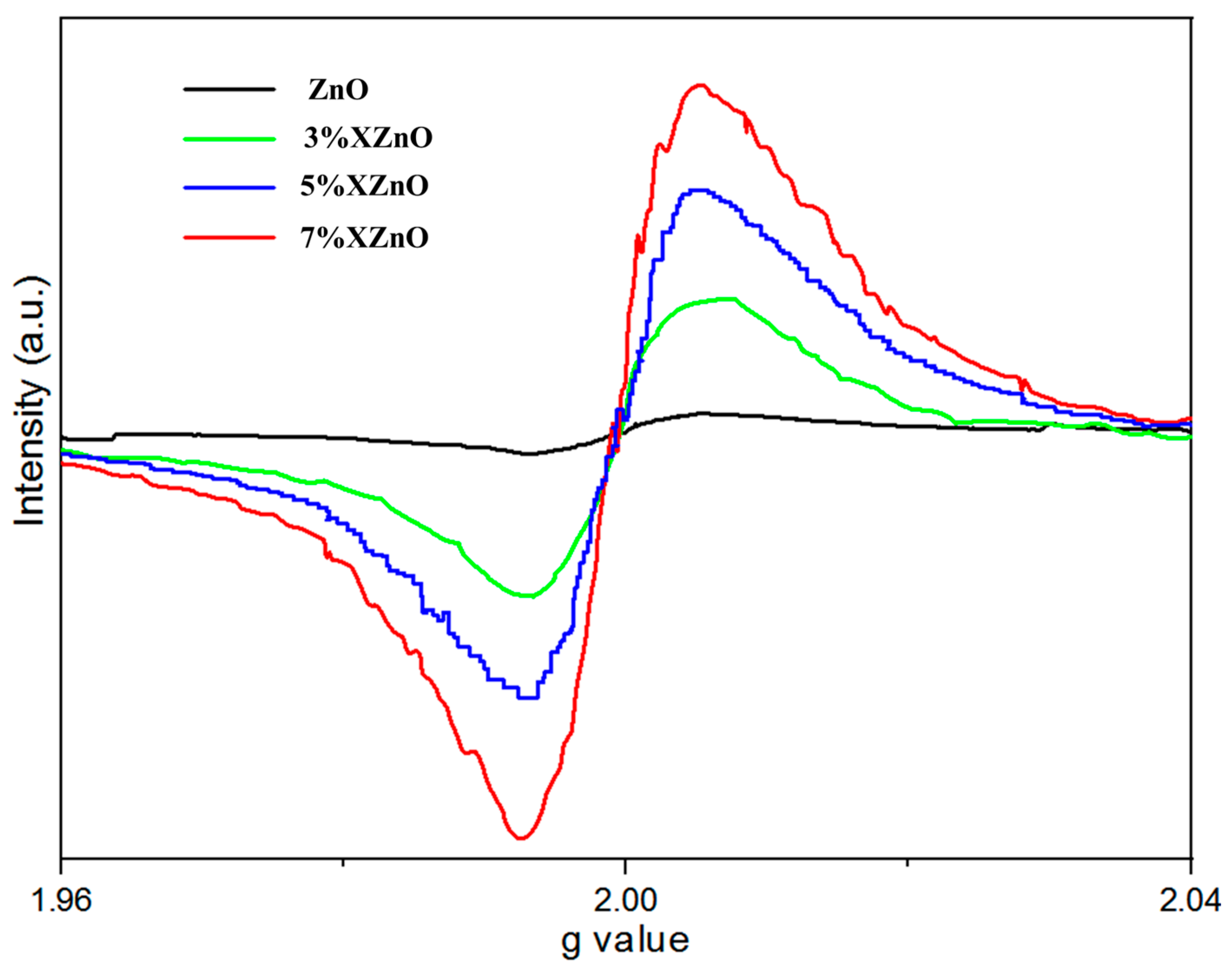
To directly identify and semi-quantify oxygen vacancies, electron paramagnetic resonance (EPR) spectroscopy was employed at 77 K, with the results shown in
Figure 9. A distinct signal at g ≈ 2.0, characteristic of singly ionized oxygen vacancies (O
v), is observed for all samples. Crucially, the intensity of this signal increases with Ca doping, being weakest for pure ZnO and strongest for the 5%XZnO sample, before slightly decreasing for the 7%XZnO sample. This trend indicates that the substitution of Zn
2+ by Ca
2+ introduces a degree of charge imbalance and lattice strain, which facilitates the formation of oxygen vacancies. The higher concentration of oxygen vacancies in the optimally doped sample can act as shallow electron donors and efficient trap sites, temporarily immobilizing photogenerated electrons and thereby prolonging their lifetime by suppressing rapid recombination with holes. The subsequent slight decrease in the 7% sample may be due to an overabundance of defects that begin to aggregate or form different types of recombination centers. These findings provide direct evidence that the enhanced performance of the Ca-doped samples is linked not only to bandgap narrowing but also to the strategic introduction of beneficial oxygen vacancies.
Based on the comprehensive experimental evidence, a plausible mechanism for the enhanced photocatalytic activity of Ca-doped ZnO nanosheets is proposed, as illustrated in
Figure 10. The enhanced performance is a result of several synergistic effects: (i) Extended light absorption: Substitutional Ca
2+ doping narrows the bandgap, enabling the absorption of visible light and generating more electron-hole pairs. (ii) Enhanced charge separation: The incorporation of Ca
2+ induces lattice distortion and promotes the formation of oxygen vacancies. These vacancies can serve as efficient electron traps, temporarily immobilizing electrons and significantly prolonging their lifetime by impeding rapid recombination with holes in the valence band. (iii) Favorable band edge positions: Ca doping shifts the conduction band minimum to more negative potentials, increasing the reducing power of the photogenerated electrons. (iv) Improved surface reactivity: The increased surface hydroxyl groups, as evidenced by FT-IR, enhance the catalyst's hydrophilicity and its ability to adsorb water molecules, the primary reactant. These combined factors funnel a greater number of long-lived, highly energetic electrons to the surface-active sites to efficiently reduce adsorbed protons to molecular hydrogen. Furthermore, optimal Ca doping suppresses the formation of lattice defects that can act as recombination centers, improving the lifetime and migration efficiency of photogenerated charge carriers. These synergistic effects collectively contribute to the efficient and stable photocatalytic water splitting for hydrogen production by in-situ Ca-doped ZnO under visible light.