Submitted:
05 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
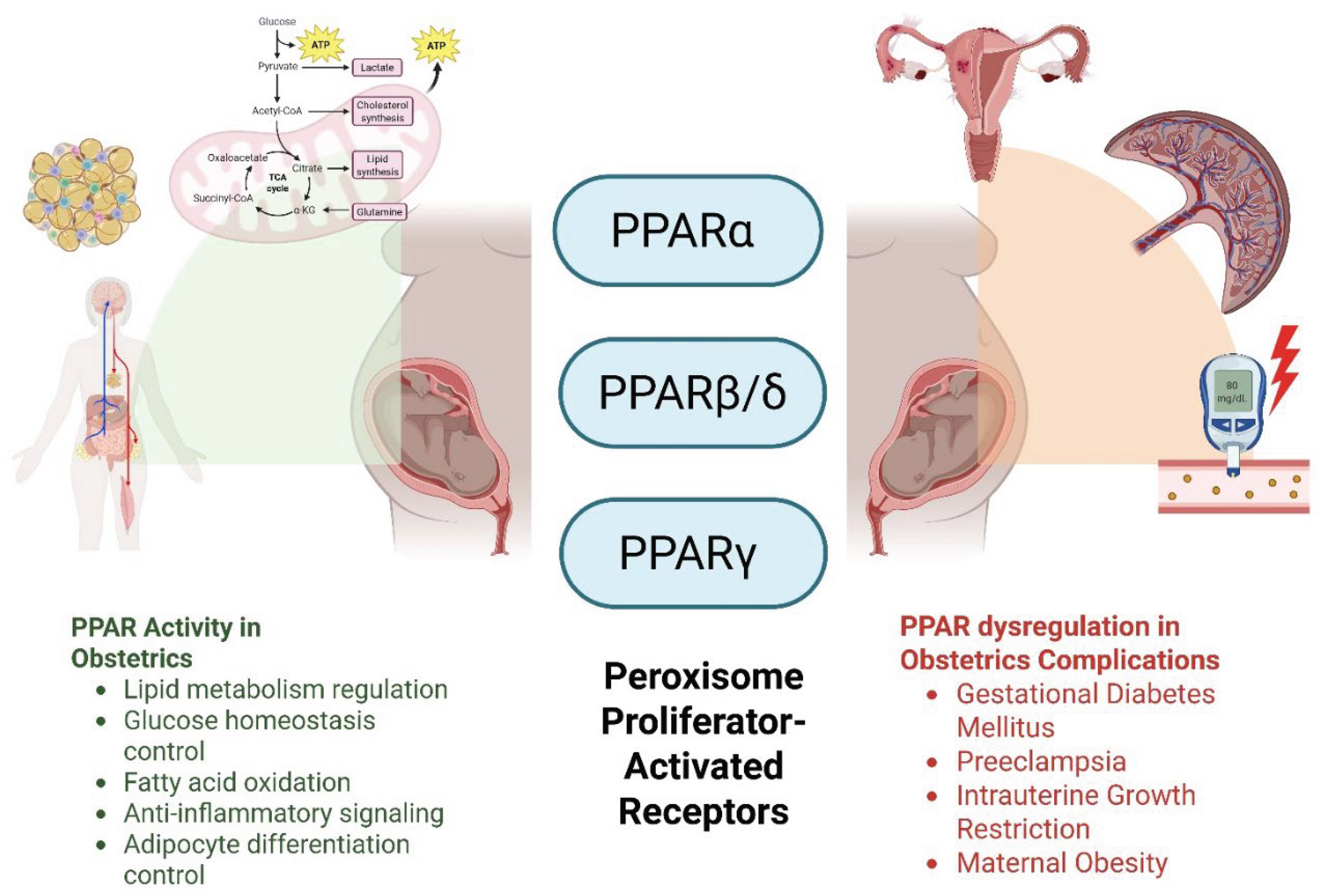
1. Introduction
2. Molecular Structure, Ligand Activation, and Mechanism of Action
3. Pparα Expression and Target Gene Networks in the Human Placenta
4. Pparα and Placental Fatty Acid Metabolism: Central Role in Feto-Maternal Lipid Homeostasis
5. PPARα as a Placental Anti-Inflammatory Regulator
6. PPARα in Fetal Metabolic Programming and Epigenetic Regulation
7. PPARα Dysregulation in Obstetric Complications
7.1. Gestational Diabetes Mellitus
7.2. Preeclampsia
7.3. Intrauterine Growth Restriction
7.4. Maternal Obesity and Environmental Exposures
8. Therapeutic Perspectives and Future Research Priorities
9. Conclusions
Author Contributions
Funding
Ethics Statement
Acknowledgments
Conflicts of Interest
References
- Bidne, K; Uhlson, C; Palmer, C; Zemski-Berry, K; Powell, T. Human placental lipid content and lipid metabolic enzyme abundance in obesity and across gestation Available to Purchas. Clin Sci. 2022, 136(19), 1389–1404. [Google Scholar] [CrossRef]
- Michalik, L; Auwerx, J; Berger, J; Chatterjee, V; Glass, C; et al. International Union of Pharmacology. LXI. Peroxisome proliferator-activated receptors. Pharmacol Rev. 2006, 58(4), 726–741. [Google Scholar] [CrossRef] [PubMed]
- Fournier, T; Tsatsaris, V; Handschuh, K; Evain-Brion, D. PPARs and the Placenta. Placenta 2007, 28(2-3), 65–76. [Google Scholar] [CrossRef] [PubMed]
- Barak, Y; Nelson, M; Ong, E; et al. PPAR gamma is required for placental, cardiac, and adipose tissue development. Mol Cell. 1999, 4, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Nadra, K; Anghel, SI; Joye, E; et al. Differentiation of Trophoblast Giant Cells and Their Metabolic Functions Are Dependent on Peroxisome Proliferator-Activated Receptor β/δ. Mol Cell Biol. 2006, 26(8), 3266–3281. [Google Scholar] [CrossRef]
- Tahri-Joutey, M; Andreoletti, P; Surapureddi, S; Nasser, B; Cherkaoui-Malki, M; Latruffe, N. Mechanisms mediating the regulation of peroxisomal fatty acid beta-oxidation by pparα. Int J Mol Sci. 2021, 22(16). [Google Scholar] [CrossRef]
- Holdsworth-Carson, S; Lim, R; Mitton, A; et al. Peroxisome proliferator-activated receptors are altered in pathologies of the human placenta: Gestational diabetes mellitus, intrauterine growth restriction and preeclampsia. Placenta 2010, 31(3), 222–229. [Google Scholar] [CrossRef]
- Kersten, S; Desvergne, B; Wahli, W. Roles of PPARs in health and disease. Nature 2000, 405, 421–424. [Google Scholar] [CrossRef]
- Tugwood, J; Issemann, I; Anderson, R; Bundell, K; McPheat, W; Green, S. The mouse peroxisome proliferator activated receptor recognizes a response element in the 5’ flanking sequence of the rat acyl CoA oxidase gene. EMBO J 1992, 11(2), 433–439. [Google Scholar] [CrossRef]
- Delerive, P; De Bosscher, K; Besnard, S; et al. Peroxisome proliferator-activated receptor α negatively regulates the vascular inflammatory gene response by negative cross-talk with transcription factors NF-κB and AP-1. Journal of Biological Chemistry 1999, 274(45), 32048–32054. [Google Scholar] [CrossRef]
- Duttaroy, AK; Basak, S. Maternal Fatty Acid Metabolism in Pregnancy and Its Consequences in the Feto-Placental Development. Front Physiol. Frontiers Media S.A. 2022;12. [CrossRef]
- Singh, S; Dhar, R; Karmakar, S. Fenofibrate mediated activation of PPARα negatively regulates trophoblast invasion. Placenta 2022, 126, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Rogers, JM; Heintz, MM; Thompson, CM; Haws, LC. A putative adverse outcome network for neonatal mortality and lower birth weight in rodents: Applicability to per- and polyfluoroalkyl substances and relevance to human health. Birth Defects Res. 2023, 115(11), 1011–1062. [Google Scholar] [CrossRef]
- Psilopatis, I; Vrettou, K; Fleckenstein, FN; Theocharis, S. The Role of Peroxisome Proliferator-Activated Receptors in Preeclampsia. Cells. MDPI 2023, 12(4). [Google Scholar] [CrossRef]
- Tarrade, A; Schoonjans, K; Guibourdenche, J; et al. PPARγ/RXRα Heterodimers Are Involved in Human CGβ Synthesis and Human Trophoblast Differentiation. Endocrinology 2001, 142(10), 4504–4514. [Google Scholar] [CrossRef]
- Shekhawat, P; Matern, D; Strauss, A. Fetal Fatty Acid Oxidation Disorders, Their Effect on Maternal Health and Neonatal Outcome: Impact of Expanded Newborn Screening on Their Diagnosis and Management. Pediatr Res. 2005, 57, 78–86. [Google Scholar] [CrossRef]
- Song, S; Attia, R; Connaughton, S; et al. Peroxisome proliferator activated receptor α (PPARα) and PPAR gamma coactivator (PGC-1α) induce carnitine palmitoyltransferase IA (CPT-1A) via independent gene elements. Mol Cell Endocrinol. 2010, 325(1-2), 54–63. [Google Scholar] [CrossRef]
- Rasool, A; Mahmoud, T; Mathyk, B; et al. Obesity downregulates lipid metabolism genes in first trimester placenta. Sci Rep. 2022, 12(1). [Google Scholar] [CrossRef]
- Thomas, MM; Haghiac, M; Grozav, C; Minium, J; Calabuig-Navarro, V; O’Tierney-Ginn, P. Oxidative Stress Impairs Fatty Acid Oxidation and Mitochondrial Function in the Term Placenta. Reproductive Sciences 2019, 26(7), 972–978. [Google Scholar] [CrossRef]
- Kramer, AC; Jansson, T; Bale, TL; Powell, TL. Maternal-fetal cross-talk via the placenta: influence on offspring development and metabolism. Development (Cambridge). Company of Biologists Ltd. 2023;150(20). [CrossRef]
- Devchand, P; Keller, H; Peters, J; Vazquek, M; Gonzalez, F; Wahli, W. The PPARα–leukotriene B4 pathway to inflammation control. Nature 1996, 384, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Martínez, N; Kurtz, M; Capobianco, E; Higa, R; White, V; Jawerbaum, A. PPARα agonists regulate lipid metabolism and nitric oxide production and prevent placental overgrowth in term placentas from diabetic rats. J Mol Endocrinol. 2011, 47(1), 1–12. [Google Scholar] [CrossRef] [PubMed]
- Barker, DJP. The origins of the developmental origins theory. Journal of Internal Medicine 2007, 261, 412–417. [Google Scholar] [CrossRef]
- Li, L; Zhang, L; Chen, Y; et al. Maternal glucose concentrations and DNA methylation of genes related to the PPAR signaling pathway in human placenta: insights for maternal glucose concentrations’ effects on neonatal anthropometrics. Clin Epigenetics 2025, 17(1). [Google Scholar] [CrossRef] [PubMed]
- Aye, I; Gao, X; Weintraub, S; Jansson, T; Powell, T. Adiponectin inhibits insulin function in primary trophoblasts by PPARα-mediated ceramide synthesis. Molecular Endocrinology 2014, 28(4), 512–524. [Google Scholar] [CrossRef] [PubMed]
- Moody, L; Xu, G; Chen, H; Pan, Y. Epigenetic regulation of carnitine palmitoyltransferase 1 (Cpt1a) by high fat diet. Biochimica et Biophysica Acta (BBA) - Gene Regulatory Mechanisms 2019, 1862(2), 141–152. [Google Scholar] [CrossRef]
- Luo, X; Huang, B; Xu, P; et al. The Placenta Regulates Intrauterine Fetal Growth via Exosomal PPARγ. Advanced Science 2025, 12(15). [Google Scholar] [CrossRef] [PubMed]
- Qin, Y; Bily, D; Aguirre, M; Zhang, K; Xie, L. Understanding PPARγ and Its Agonists on Trophoblast Differentiation and Invasion: Potential Therapeutic Targets for Gestational Diabetes Mellitus and Preeclampsia. Nutrients. MDPI 2023, 15(11). [Google Scholar] [CrossRef]
- Szilagyi, J; Avula, V; Fry, R. Perfluoroalkyl Substances (PFAS) and Their Effects on the Placenta, Pregnancy, and Child Development: a Potential Mechanistic Role for Placental Peroxisome Proliferator–Activated Receptors (PPARs). Curr Environ Health Rep. 2020, 7, 222–230. [Google Scholar] [CrossRef]
| Condition | PPARα Expression | Key Downstream Effects | Functional Consequence | Key References |
|---|---|---|---|---|
| Normal Pregnancy | Abundant; higher in syncytiotrophoblast | CPT1B, MFSD2A, FABP4, ACOX1 upregulated | Balanced FAO; DHA transport; anti-inflammatory tone | [3,16,18] |
| Gestational Diabetes Mellitus | ↓ Protein; normal DNA binding activity | Reduced MCAD, CPT2; epigenetic changes in ACADM, ACAA1 | Lipid accumulation; impaired FAO; altered fetal lipid delivery | [7,24] |
| Preeclampsia | ↓ Protein; preterm placentas | Reduced NF-κB repression; eicosanoid dysbalance | Amplified inflammation; mitochondrial dysfunction; oxidative stress | [7,14] |
| Intrauterine Growth Restriction | ↓ Protein; lowest in PE+IUGR co-morbidity | Impaired FAO; reduced nutrient transport | Fetal energy deficit; impaired adipogenesis; growth restriction | [7,19] |
| Maternal Obesity | ↓ Target gene expression (1st trimester) | ↓ MFSD2A, CPT2, PLIN2 via reduced endogenous ligand | Reduced DHA uptake; lipotoxic risk; male-specific DHA deficit | [18] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




