Submitted:
27 February 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
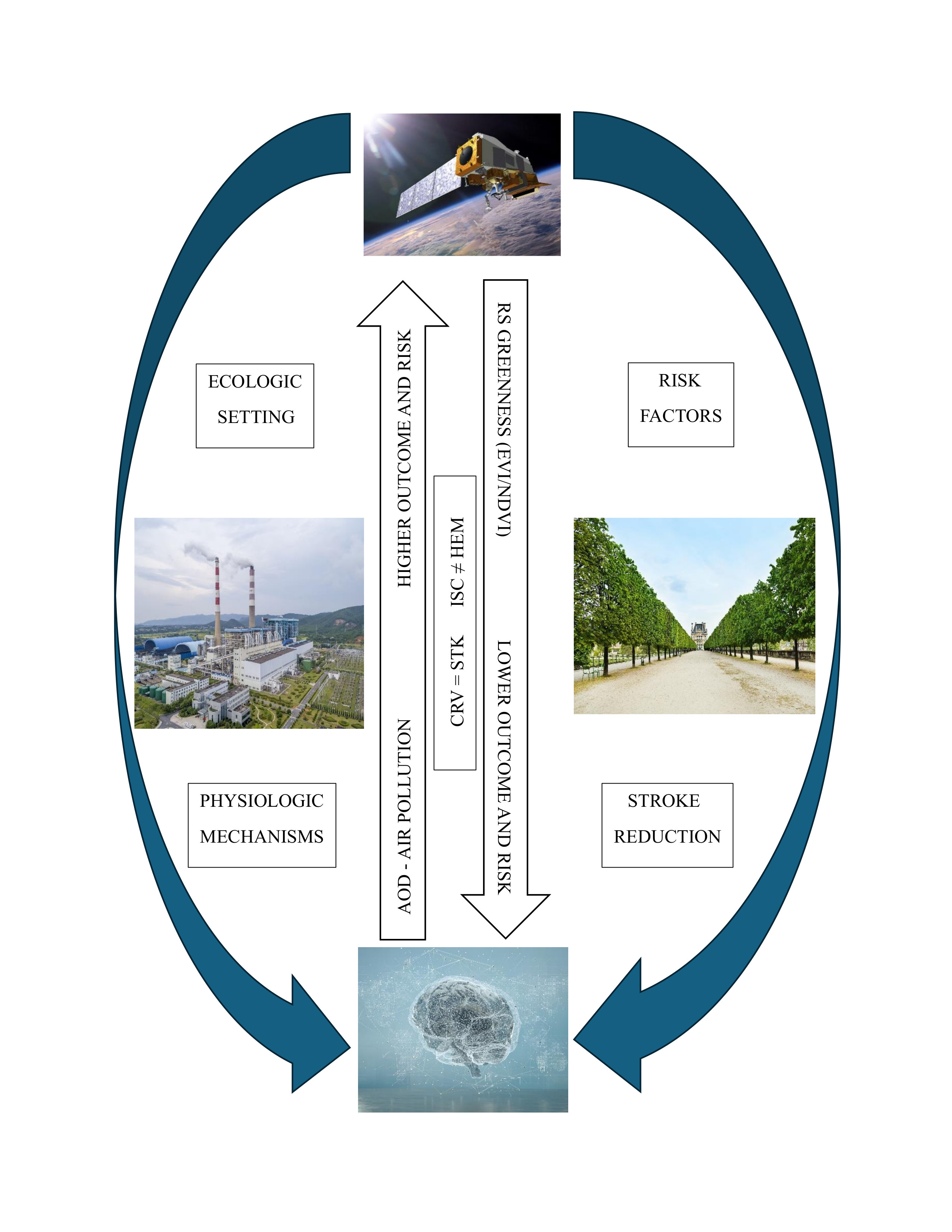
Keywords:
1. Introduction
2. Methods
2.1. Development of the Literature Search Strategy
2.2. Implementation of the Literature Search Strategy
2.3. Analysis of Publications and Publication-Extracted Records
2.4. Identified Research and Review Articles
3.0. Results
3.1. Publication and Record Variable Attributes
3.2. Ambient AOD-Air Pollutant and RS Greenness Values and Accuracy Estimates
3.3. Concentration-Response Functions and Stroke Subtype Prevalence and Incidence Worldwide
3.4. Significant Differences by Variable Outcomes
3.5. Stroke Subtype and Risk Factors
3.6. RS Greenness and Stroke Subtype
3.7. Physiologic Mechanisms and Stroke Subtypes
3.8. Descriptive-Explanatory and Stroke Subtype Physiologic Mechanism Differences
3.9. Stroke Subtype Intervention Programs
4.0. Discussion
5. Conclusions
6. Future Directions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, Q.; Yang, S.; Chen, H. Global trends and hotspots in the study of the effects of PM2.5 on ischemic stroke. Journal of Health, Population and Nutrition 2024, 43, 133. [Google Scholar] [CrossRef] [PubMed]
- Sogno, P.; Traidl-Hoffmann, C.; Kuenzer, C. Earth Observation Data Supporting Non-Communicable Disease Research: A Review. Remote Sensing 2020, 12, 2541. [Google Scholar] [CrossRef]
- Crouse, D.L.; Peters, P.A.; van Donkelaar, A.; Goldberg, M.S.; Villeneuve, P.J.; Brion, O.; Khan, S.; Atari, D.O.; Jerrett, M.; Pope, C.A.; et al. Risk of nonaccidental and cardiovascular mortality in relation to long-term exposure to low concentrations of fine particulate matter: a Canadian national-level cohort study. Environmental health perspectives 2012, 120, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Kloog, I.; Coull, B.A.; Zanobetti, A.; Koutrakis, P.; Schwartz, J.D. Acute and chronic effects of particles on hospital admissions in New-England. PLoS One 2012, 7, e34664. [Google Scholar] [CrossRef]
- Cai, M.; Lin, X.; Wang, X.; Zhang, S.; Qian, Z.; McMillin, S.E.; Aaron, H.E.; Lin, H.; Wei, J.; Zhang, Z.; et al. Ambient particulate matter pollution of different sizes associated with recurrent stroke hospitalization in China: A cohort study of 1.07 million stroke patients. Science of The Total Environment 2023, 856, 159104. [Google Scholar] [CrossRef]
- Chen, S.; Lin, X.; Du, Z.; Zhang, Y.; Zheng, L.; Ju, X.; Guo, T.; Wang, X.; Chen, L.; Jiang, J.; et al. Potential causal links between long-term ambient particulate matter exposure and cerebrovascular mortality: Insights from a large cohort in southern China. Environmental pollution (Barking, Essex: 1987) 2023, 328, 121336. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, Y.; Wang, Y.; Lawrence, W.R.; Rhee, J.; Guo, T.; Chen, S.; Du, Z.; Wu, W.; Li, Z.; et al. Long-term particulate matter exposure and the risk of neurological hospitalization: Evidence from causal inference of a large longitudinal cohort in South China. Chemosphere 2023, 345, 140397. [Google Scholar] [CrossRef]
- Jiang, D.; Wang, L.; Han, X.; Pan, Z.; Wang, Z.; Wang, Y.; Li, J.; Guo, J.; Liu, Y.; Huang, S.; et al. Short-term effects of ambient oxidation, and its interaction with fine particles on first-ever stroke: A national case-crossover study in China. Science of The Total Environment 2024, 907, 168017. [Google Scholar] [CrossRef]
- Li, M.; Edgell, R.C.; Wei, J.; Li, H.; Qian, Z.; Feng, J.; Tian, F.; Wang, X.; Xin, Q.; Cai, M.; et al. Air pollution and stroke hospitalization in the Beibu Gulf Region of China: A case-crossover analysis. Ecotoxicology and environmental safety 2023, 255, 114814. [Google Scholar] [CrossRef]
- Li, W.; Tian, A.; Shi, Y.; Chen, B.; Ji, R.; Ge, J.; Su, X.; Pu, B.; Lei, L.; Ma, R.; et al. Associations of long-term fine particulate matter exposure with all-cause and cause-specific mortality: results from the ChinaHEART project. The Lancet Regional Health – Western Pacific 2023, 41. [Google Scholar] [CrossRef]
- Xu, R.; Wang, Q.; Wei, J.; Lu, W.; Wang, R.; Liu, T.; Wang, Y.; Fan, Z.; Li, Y.; Xu, L.; et al. Association of short-term exposure to ambient air pollution with mortality from ischemic and hemorrhagic stroke. Eur J Neurol 2022, 29, 1994–2005. [Google Scholar] [CrossRef]
- Braggio, J.T.; Hall, E.S.; Weber, S.A.; Huff, A.K. Contribution of Satellite-Derived Aerosol Optical Depth PM2.5 Bayesian Concentration Surfaces to Respiratory-Cardiovascular Chronic Disease Hospitalizations in Baltimore, Maryland. Atmosphere (Basel) 2020, 11, 209. [Google Scholar] [CrossRef] [PubMed]
- Nabizadeh, R.; Yousefian, F.; Moghadam, V.K.; Hadei, M. Characteristics of cohort studies of long-term exposure to PM(2.5): a systematic review. Environmental science and pollution research international 2019, 26, 30755–30771. [Google Scholar] [CrossRef] [PubMed]
- Whyte, M.; Douwes, J.; Ranta, A. Green space and stroke: A scoping review of the evidence. J Neurol Sci 2024, 457, 122870. [Google Scholar] [CrossRef] [PubMed]
- Carver, A.; Beare, R.; Knibbs, L.D.; Mavoa, S.; Grocott, K.; Wheeler, A.J.; Srikanth, V.; Andrew, N.E. Exploring associations of greenery, air pollution and walkability with cardiometabolic health in people at midlife and beyond. Geriatr Gerontol Int 2024, 24 Suppl 1, 208–214. [Google Scholar] [CrossRef]
- Silveira, I.H.D.; Junger, W.L. Green spaces and mortality due to cardiovascular diseases in the city of Rio de Janeiro. Rev Saude Publica 2018, 52, 49. [Google Scholar] [CrossRef]
- Riggs, D.W.; Baumgartner, K.B.; Baumgartner, R.; Boone, S.; Judd, S.E.; Bhatnagar, A. Association Between Residential Greenness and Risk of Stroke by Ecoregions: The REGARDS Study. Journal of the American Heart Association 2025, 14, e037866. [Google Scholar] [CrossRef]
- Paul, L.A.; Hystad, P.; Burnett, R.T.; Kwong, J.C.; Crouse, D.L.; van Donkelaar, A.; Tu, K.; Lavigne, E.; Copes, R.; Martin, R.V.; et al. Urban green space and the risks of dementia and stroke. Environ Res 2020, 186, 109520. [Google Scholar] [CrossRef]
- Crouse, D.L.; Pinault, L.; Balram, A.; Hystad, P.; Peters, P.A.; Chen, H.; van Donkelaar, A.; Martin, R.V.; Ménard, R.; Robichaud, A.; et al. Urban greenness and mortality in Canada's largest cities: a national cohort study. Lancet Planet Health 2017, 1, e289–e297. [Google Scholar] [CrossRef]
- Vivanco-Hidalgo, R.M.; Avellaneda-Gómez, C.; Dadvand, P.; Cirach, M.; Ois, Á.; Gómez González, A.; Rodriguez-Campello, A.; de Ceballos, P.; Basagaña, X.; Zabalza, A.; et al. Association of residential air pollution, noise, and greenspace with initial ischemic stroke severity. Environ Res 2019, 179, 108725. [Google Scholar] [CrossRef]
- Meng, X.; Zhang, L.; Chan, K.H.; Lv, J.; Lam, H.; Liu, C.; Chen, R.; Kartsonaki, C.; Wright, N.; Du, H.; et al. Association of greenness with incidence of cardiovascular disease in China: Evidence from the China Kadoorie Biobank prospective cohort study with 0.5 million adults. Eco Environ Health 2025, 4, 100148. [Google Scholar] [CrossRef] [PubMed]
- Orioli, R.; Antonucci, C.; Scortichini, M.; Cerza, F.; Marando, F.; Ancona, C.; Manes, F.; Davoli, M.; Michelozzi, P.; Forastiere, F.; et al. Exposure to Residential Greenness as a Predictor of Cause-Specific Mortality and Stroke Incidence in the Rome Longitudinal Study. Environmental health perspectives 2019, 127, 27002. [Google Scholar] [CrossRef] [PubMed]
- Braggio, J.T. Remote Sensing Technologies Quantify the Contribution of Ambient Air Pollution to Asthma Severity and Risk Factors in Greenness, Air Pollution, and Wildfire Ecological Settings: A Literature Review. Atmosphere 2024, 15, 1470. [Google Scholar] [CrossRef]
- Braggio, J. Inflammation Describes and Explains the Adverse Effects of Aerosol Optical Depth-Particulate Matter on Cardiovascular Outcomes: A Literature Review Since 2012. Medical Research Archives 2023, 11. [Google Scholar] [CrossRef]
- Abdul-Rahman, T.; Roy, P.; Bliss, Z.S.B.; Mohammad, A.; Corriero, A.C.; Patel, N.T.; Wireko, A.A.; Shaikh, R.; Faith, O.E.; Arevalo-Rios, E.C.E.; et al. The impact of air quality on cardiovascular health: A state of the art review. Curr Probl Cardiol 2024, 49, 102174. [Google Scholar] [CrossRef]
- Brook, R.D.; Rajagopalan, S.; Pope, C.A., 3rd; Brook, J.R.; Bhatnagar, A.; Diez-Roux, A.V.; Holguin, F.; Hong, Y.; Luepker, R.V.; Mittleman, M.A.; et al. Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation 2010, 121, 2331–2378. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, P.; Xia, X.; Wang, L.; Li, X. The underlying mechanism of PM2.5-induced ischemic stroke. Environmental pollution (Barking, Essex 1987) 2022, 310, 119827. [Google Scholar] [CrossRef]
- Guo, Y.; Luo, C.; Cao, F.; Liu, J.; Yan, J. Short-term environmental triggers of hemorrhagic stroke. Ecotoxicology and environmental safety 2023, 265, 115508. [Google Scholar] [CrossRef]
- Irfan, H. Air pollution and cardiovascular health in South Asia: A comprehensive review. Curr Probl Cardiol 2024, 49, 102199. [Google Scholar] [CrossRef]
- Krittanawong, C.; Qadeer, Y.K.; Hayes, R.B.; Wang, Z.; Thurston, G.D.; Virani, S.; Lavie, C.J. PM(2.5) and cardiovascular diseases: State-of-the-Art review. Int J Cardiol Cardiovasc Risk Prev 2023, 19, 200217. [Google Scholar] [CrossRef]
- Kulick, E.R.; Kaufman, J.D.; Sack, C. Ambient Air Pollution and Stroke: An Updated Review. Stroke 2023, 54, 882–893. [Google Scholar] [CrossRef]
- Lamas, G.A.; Bhatnagar, A.; Jones, M.R.; Mann, K.K.; Nasir, K.; Tellez-Plaza, M.; Ujueta, F.; Navas-Acien, A. Contaminant Metals as Cardiovascular Risk Factors: A Scientific Statement From the American Heart Association. Journal of the American Heart Association 2023, 12, e029852. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Lin, G.; Xiao, Z.; Zhang, Y.; Li, B.; Zhou, Y.; Ma, Y.; Chai, E. A review of respirable fine particulate matter (PM(2.5))-induced brain damage. Front Mol Neurosci 2022, 15, 967174. [Google Scholar] [CrossRef] [PubMed]
- Newby, D.E.; Mannucci, P.M.; Tell, G.S.; Baccarelli, A.A.; Brook, R.D.; Donaldson, K.; Forastiere, F.; Franchini, M.; Franco, O.H.; Graham, I.; et al. Expert position paper on air pollution and cardiovascular disease. Eur Heart J 2015, 36, 83–93b. [Google Scholar] [CrossRef] [PubMed]
- Niu, Z.; Liu, F.; Yu, H.; Wu, S.; Xiang, H. Association between exposure to ambient air pollution and hospital admission, incidence, and mortality of stroke: an updated systematic review and meta-analysis of more than 23 million participants. Environ Health Prev Med 2021, 26, 15. [Google Scholar] [CrossRef]
- Rajagopalan, S.; Al-Kindi, S.G.; Brook, R.D. Air Pollution and Cardiovascular Disease: JACC State-of-the-Art Review. Journal of the American College of Cardiology 2018, 72, 2054–2070. [Google Scholar] [CrossRef]
- Zhou, M.; Xin, J.; Chen, J.; Sun, C.; Huo, B.; Zhang, W.; Liu, X. Scientific Landscape of Oxidative Stress in Stroke: From a Bibliometric Analysis to an in-Depth Review. Neurochemical research 2023, 48, 3327–3348. [Google Scholar] [CrossRef]
- SAS. SAS Studio 3.8: User's Guide. 2018.
- SAS. Base SAS 9.4 Procedures Guide: Statistical Procedures, Sixth Edition. 2016.
- SAS. SAS/STAT 15.4 User's Guide. 2025.
- Kaufman, J.D.; Elkind, M.S.V.; Bhatnagar, A.; Koehler, K.; Balmes, J.R.; Sidney, S.; Burroughs Peña, M.S.; Dockery, D.W.; Hou, L.; Brook, R.D.; et al. Guidance to Reduce the Cardiovascular Burden of Ambient Air Pollutants: A Policy Statement From the American Heart Association. Circulation 2020, 142, e432–e447. [Google Scholar] [CrossRef]
- Rajagopalan, S.; Brauer, M.; Bhatnagar, A.; Bhatt, D.L.; Brook, J.R.; Huang, W.; Münzel, T.; Newby, D.; Siegel, J.; Brook, R.D. Personal-Level Protective Actions Against Particulate Matter Air Pollution Exposure: A Scientific Statement From the American Heart Association. Circulation 2020, 142, e411–e431. [Google Scholar] [CrossRef]
- Bauwelinck, M.; Casas, L.; Nawrot, T.S.; Nemery, B.; Trabelsi, S.; Thomas, I.; Aerts, R.; Lefebvre, W.; Vanpoucke, C.; Van Nieuwenhuyse, A.; et al. Residing in urban areas with higher green space is associated with lower mortality risk: A census-based cohort study with ten years of follow-up. Environment international 2021, 148, 106365. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, B.; Liu, C.; Zhang, Y.; Zhao, K.; Zhang, P.; Tian, M.; Lu, Z.; Guo, X.; Jia, X. Association of ambient ozone exposure and greenness exposure with hemorrhagic stroke mortality at different times: A cohort study in Shandong Province, China. Ecotoxicology and environmental safety 2024, 278, 116356. [Google Scholar] [CrossRef]
- Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [CrossRef]
- Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol 2021, 20, 795–820. [CrossRef] [PubMed]
- Bo, Y.; Zhu, Y.; Zhang, X.; Chang, H.; Zhang, J.; Lao, X.Q.; Yu, Z. Spatiotemporal trends of stroke burden attributable to ambient PM2. 5 in 204 countries and territories, 1990–2019: a global analysis. Neurology 2023, 101, e764–e776. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Lv, Y.; Zhang, Y.; Suo, H.; Wang, F.; Gao, S. Global trends and burden of stroke attributable to particulate matter pollution from 1990 to 2019. Ecotoxicology and environmental safety 2024, 274, 116205. [Google Scholar] [CrossRef] [PubMed]
- Motairek, I.; Ajluni, S.; Khraishah, H.; AlAhmad, B.; Al-Dulaimi, S.; Abi Khalil, C.; Rajagopalan, S.; Al-Kindi, S. Burden of cardiovascular disease attributable to particulate matter pollution in the eastern Mediterranean region: analysis of the 1990–2019 global burden of disease. European journal of preventive cardiology 2022, 30, 256–263. [Google Scholar] [CrossRef]
- Shaddick, G.; Thomas, M.L.; Green, A.; Brauer, M.; Donkelaar, A.; Burnett, R.; Chang, H.H.; Cohen, A.; Dingenen, R.V.; Dora, C.; et al. Data Integration Model for Air Quality: A Hierarchical Approach to the Global Estimation of Exposures to Ambient Air Pollution. Journal of the Royal Statistical Society Series C: Applied Statistics 2017, 67, 231–253. [Google Scholar] [CrossRef]
- Shaddick, G.; Thomas, M.L.; Amini, H.; Broday, D.; Cohen, A.; Frostad, J.; Green, A.; Gumy, S.; Liu, Y.; Martin, R.V.; et al. Data Integration for the Assessment of Population Exposure to Ambient Air Pollution for Global Burden of Disease Assessment. Environmental science & technology 2018, 52, 9069–9078. [Google Scholar] [CrossRef]
- Cohen, A.J.; Brauer, M.; Burnett, R.; Anderson, H.R.; Frostad, J.; Estep, K.; Balakrishnan, K.; Brunekreef, B.; Dandona, L.; Dandona, R.; et al. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: an analysis of data from the Global Burden of Diseases Study 2015. Lancet 2017, 389, 1907–1918. [Google Scholar] [CrossRef]
- Lelieveld, J.; Haines, A.; Burnett, R.; Tonne, C.; Klingmüller, K.; Münzel, T.; Pozzer, A. Air pollution deaths attributable to fossil fuels: observational and modelling study. Bmj 2023, 383, e077784. [Google Scholar] [CrossRef]
- Shi, Y.; Matsunaga, T.; Yamaguchi, Y.; Zhao, A.; Li, Z.; Gu, X. Long-term trends and spatial patterns of PM(2.5)-induced premature mortality in South and Southeast Asia from 1999 to 2014. The Science of the total environment 2018, 631-632, 1504–1514. [Google Scholar] [CrossRef]
- Yang, X.; Wang, Y.; Zhao, C.; Fan, H.; Yang, Y.; Chi, Y.; Shen, L.; Yan, X. Health risk and disease burden attributable to long-term global fine-mode particles. Chemosphere 2022, 287, 132435. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Shi, Y. Spatio-temporal variations of PM2.5 concentrations and related premature deaths in Asia, Africa, and Europe from 2000 to 2018. Environmental Impact Assessment Review 2023, 99, 107046. [Google Scholar] [CrossRef]
- He, Q.; Gu, Y.; Yim, S.H.L. What drives long-term PM(2.5)-attributable premature mortality change? A case study in central China using high-resolution satellite data from 2003 to 2018. Environment international 2022, 161, 107110. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Cui, L.; Meng, Y.; Zhao, Y.; Fu, H. Satellite-based prediction of daily SO2 exposure across China using a high-quality random forest-spatiotemporal Kriging (RF-STK) model for health risk assessment. Atmospheric Environment 2019, 208, 10–19. [Google Scholar] [CrossRef]
- Liu, M.; Huang, Y.; Ma, Z.; Jin, Z.; Liu, X.; Wang, H.; Liu, Y.; Wang, J.; Jantunen, M.; Bi, J.; et al. Spatial and temporal trends in the mortality burden of air pollution in China: 2004-2012. Environment international 2017, 98, 75–81. [Google Scholar] [CrossRef]
- Liu, M.; Saari, R.K.; Zhou, G.; Li, J.; Han, L.; Liu, X. Recent trends in premature mortality and health disparities attributable to ambient PM(2.5) exposure in China: 2005-2017. Environmental pollution (Barking, Essex: 1987) 2021, 279, 116882. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Liang, X.; Huang, K.; Liu, F.; Chen, S.; Lu, X.; Li, J. Changes on Stroke Burden Attributable to Ambient Fine Particulate Matter in China. Biomedical and Environmental Sciences 2024, 37, 823–833. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, J.; He, M.Z.; Kinney, P.L.; Li, T. A county-level estimate of PM(2.5) related chronic mortality risk in China based on multi-model exposure data. Environment international 2018, 110, 105–112. [Google Scholar] [CrossRef]
- Chowdhury, S.; Dey, S. Cause-specific premature death from ambient PM2.5 exposure in India: Estimate adjusted for baseline mortality. Environment international 2016, 91, 283–290. [Google Scholar] [CrossRef]
- Jain, V.; Dey, S.; Chowdhury, S. Ambient PM(2.5) exposure and premature mortality burden in the holy city Varanasi, India. Environmental pollution (Barking, Essex: 1987) 2017, 226, 182–189. [Google Scholar] [CrossRef]
- Maheshwarkar, P.; Sunder Raman, R. Population exposure across central India to PM(2.5) derived using remotely sensed products in a three-stage statistical model. Sci Rep 2021, 11, 544. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Oh, I.H.; Park, J.H.; Cheong, H.K. Premature Deaths Attributable to Long-term Exposure to Ambient Fine Particulate Matter in the Republic of Korea. J Korean Med Sci 2018, 33, e251. [Google Scholar] [CrossRef] [PubMed]
- Mazeli, M.I.; Pahrol, M.A.; Abdul Shakor, A.S.; Kanniah, K.D.; Omar, M.A. Cardiovascular, respiratory and all-cause (natural) health endpoint estimation using a spatial approach in Malaysia. The Science of the total environment 2023, 874, 162130. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Rueda, D.; Alsufyani, W.; Herbst, C.; AlBalawi, S.; Alsukait, R.; Alomran, M. Ambient particulate matter burden of disease in the Kingdom of Saudi Arabia. Environ Res 2021, 197, 111036. [Google Scholar] [CrossRef]
- Castillo, M.D.; Kinney, P.L.; Southerland, V.; Arno, C.A.; Crawford, K.; van Donkelaar, A.; Hammer, M.; Martin, R.V.; Anenberg, S.C. Estimating Intra-Urban Inequities in PM(2.5)-Attributable Health Impacts: A Case Study for Washington, DC. Geohealth 2021, 5, e2021GH000431. [Google Scholar] [CrossRef]
- Kerr, G.H.; van Donkelaar, A.; Martin, R.V.; Brauer, M.; Bukart, K.; Wozniak, S.; Goldberg, D.L.; Anenberg, S.C. Increasing Racial and Ethnic Disparities in Ambient Air Pollution-Attributable Morbidity and Mortality in the United States. Environmental health perspectives 2024, 132, 37002. [Google Scholar] [CrossRef]
- Brown, P.E.; Izawa, Y.; Balakrishnan, K.; Fu, S.H.; Chakma, J.; Menon, G.; Dikshit, R.; Dhaliwal, R.S.; Rodriguez, P.S.; Huang, G.; et al. Mortality Associated with Ambient PM2.5 Exposure in India: Results from the Million Death Study. Environmental health perspectives 2022, 130, 97004. [Google Scholar] [CrossRef]
- Cai, M.; Lin, X.; Wang, X.; Zhang, S.; Wang, C.; Zhang, Z.; Pan, J.; Lin, H. Long-term exposure to ambient fine particulate matter chemical composition and in-hospital case fatality among patients with stroke in China. The Lancet Regional Health – Western Pacific 2023, 32. [Google Scholar] [CrossRef]
- Cai, M.; Zhang, S.; Lin, X.; Qian, Z.; McMillin, S.E.; Yang, Y.; Zhang, Z.; Pan, J.; Lin, H. Association of Ambient Particulate Matter Pollution of Different Sizes With In-Hospital Case Fatality Among Stroke Patients in China. Neurology 2022, 98, e2474–e2486. [Google Scholar] [CrossRef]
- Chen, G.; Wang, A.; Li, S.; Zhao, X.; Wang, Y.; Li, H.; Meng, X.; Knibbs, L.D.; Bell, M.L.; Abramson, M.J.; et al. Long-Term Exposure to Air Pollution and Survival After Ischemic Stroke. Stroke 2019, 50, 563–570. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, Y.; Lin, Z.; Liu, R.; Zheng, L.; Chen, X.; Lin, S.; Qu, Y.; Hao, C.; Tang, H.; et al. The joint impact of PM2.5 constituents on the risk of cerebrovascular diseases hospitalization: A large community-based cohort study. Environmental Research 2024, 260, 119644. [Google Scholar] [CrossRef]
- Deng, B.; Zhu, L.; Zhang, Y.; Tang, Z.; Shen, J.; Zhang, Y.; Zheng, H.; Zhang, Y. Short-term exposure to PM2.5 constituents, extreme temperature events and stroke mortality. Science of The Total Environment 2024, 954, 176506. [Google Scholar] [CrossRef] [PubMed]
- Gaines, B.; Kloog, I.; Zucker, I.; Ifergane, G.; Novack, V.; Libruder, C.; Hershkovitz, Y.; Sheffield, P.E.; Yitshak-Sade, M. Particulate Air Pollution Exposure and Stroke among Adults in Israel. International journal of environmental research and public health 2023, 20, 1482. [Google Scholar] [CrossRef] [PubMed]
- Guan, T.; Xue, T.; Liu, Y.; Zheng, Y.; Fan, S.; He, K.; Zhang, Q. Differential Susceptibility in Ambient Particle-Related Risk of First-Ever Stroke: Findings From a National Case-Crossover Study. Am J Epidemiol 2018, 187, 1001–1009. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Avila, I.; Rojas-Bracho, L.; Riojas-Rodríguez, H.; Kloog, I.; Just, A.C.; Rothenberg, S.J. Cardiovascular and Cerebrovascular Mortality Associated With Acute Exposure to PM(2.5) in Mexico City. Stroke 2018, 49, 1734–1736. [Google Scholar] [CrossRef]
- Gutiérrez-Avila, I.; Riojas-Rodríguez, H.; Colicino, E.; Rush, J.; Tamayo-Ortiz, M.; Borja-Aburto, V.H.; Just, A.C. Short-term exposure to PM(2.5) and 1.5 million deaths: a time-stratified case-crossover analysis in the Mexico City Metropolitan Area. Environmental health: a global access science source 2023, 22, 70. [Google Scholar] [CrossRef]
- Huang, K.; Jia, J.; Liang, F.; Li, J.; Niu, X.; Yang, X.; Chen, S.; Cao, J.; Shen, C.; Liu, X.; et al. Fine Particulate Matter Exposure, Genetic Susceptibility, and the Risk of Incident Stroke: A Prospective Cohort Study. Stroke 2024, 55, 92–100. [Google Scholar] [CrossRef]
- Huang, K.; Liang, F.; Yang, X.; Liu, F.; Li, J.; Xiao, Q.; Chen, J.; Liu, X.; Cao, J.; Shen, C.; et al. Long term exposure to ambient fine particulate matter and incidence of stroke: prospective cohort study from the China-PAR project. Bmj 2019, 367, l6720. [Google Scholar] [CrossRef]
- Hystad, P.; Larkin, A.; Rangarajan, S.; AlHabib, K.F.; Avezum, Á.; Calik, K.B.T.; Chifamba, J.; Dans, A.; Diaz, R.; du Plessis, J.L.; et al. Associations of outdoor fine particulate air pollution and cardiovascular disease in 157 436 individuals from 21 high-income, middle-income, and low-income countries (PURE): a prospective cohort study. Lancet Planet Health 2020, 4, e235–e245. [Google Scholar] [CrossRef]
- Jalali, S.; Karbakhsh, M.; Momeni, M.; Taheri, M.; Amini, S.; Mansourian, M.; Sarrafzadegan, N. Long-term exposure to PM(2.5) and cardiovascular disease incidence and mortality in an Eastern Mediterranean country: findings based on a 15-year cohort study. Environmental health: a global access science source 2021, 20, 112. [Google Scholar] [CrossRef]
- Jin, J.-Q.; Lin, G.-Z.; Wu, S.-Y.; Zheng, M.-R.; Liu, H.; Liu, X.-Y.; Yan, M.-Q.; Chen, Z.-Y.; Ou, C.-Q. Short-term effects of individual exposure to PM2.5 on hospital admissions for myocardial infarction and stroke: a population-based case-crossover study in Guangzhou, China. Environmental Science and Pollution Research 2023, 30, 78802–78810. [Google Scholar] [CrossRef]
- Kloog, I.; Nordio, F.; Zanobetti, A.; Coull, B.A.; Koutrakis, P.; Schwartz, J.D. Short term effects of particle exposure on hospital admissions in the Mid-Atlantic states: a population estimate. PLoS One 2014, 9, e88578. [Google Scholar] [CrossRef] [PubMed]
- Kongbunkiat, K.; Pattanapipitpaisal, P.; Seoratanaphunt, S.; Peansukwech, U.; Vorasoot, N.; Kasemsap, N.; Tiamkao, S.; Chotmongkol, V.; Nakaphan, T.; Sawanyawisuth, K. An ecological study of PM2.5 and black carbon and acute hemorrhagic stroke: a long term effect study. Environmental Research Communications 2024, 6, 025008. [Google Scholar] [CrossRef]
- Lian, X.; Wang, Z.; Guo, J.; Han, X.; Pan, Z.; Wang, Y.; Xue, T.; Liu, Y.; Huang, S.; Li, J.; et al. Comparison of short-term effects of particulate matter with different particle sizes on first-ever stroke: A national case-crossover study in China. Atmospheric Environment 2023, 313, 120080. [Google Scholar] [CrossRef]
- Liang, R.; Chen, R.; Yin, P.; van Donkelaar, A.; Martin, R.V.; Burnett, R.; Cohen, A.J.; Brauer, M.; Liu, C.; Wang, W.; et al. Associations of long-term exposure to fine particulate matter and its constituents with cardiovascular mortality: A prospective cohort study in China. Environment international 2022, 162, 107156. [Google Scholar] [CrossRef]
- Lin, H.; Guo, Y.; Di, Q.; Zheng, Y.; Kowal, P.; Xiao, J.; Liu, T.; Li, X.; Zeng, W.; Howard, S.W.; et al. Ambient PM(2.5) and Stroke: Effect Modifiers and Population Attributable Risk in Six Low- and Middle-Income Countries. Stroke 2017, 48, 1191–1197. [Google Scholar] [CrossRef]
- Liu, C.; Chan, K.H.; Lv, J.; Lam, H.; Newell, K.; Meng, X.; Liu, Y.; Chen, R.; Kartsonaki, C.; Wright, N.; et al. Long-Term Exposure to Ambient Fine Particulate Matter and Incidence of Major Cardiovascular Diseases: A Prospective Study of 0.5 Million Adults in China. Environmental science & technology 2022, 56, 13200–13211. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Yang, Z.; Luo, S.; Zhang, Y. Long-term exposure to fine particulate constituents and cardiovascular diseases in Chinese adults. J Hazard Mater 2021, 416, 126051. [Google Scholar] [CrossRef]
- Loop, M.S.; Kent, S.T.; Al-Hamdan, M.Z.; Crosson, W.L.; Estes, S.M.; Estes, M.G., Jr.; Quattrochi, D.A.; Hemmings, S.N.; Wadley, V.G.; McClure, L.A. Fine particulate matter and incident cognitive impairment in the REasons for Geographic and Racial Differences in Stroke (REGARDS) cohort. PLoS One 2013, 8, e75001. [Google Scholar] [CrossRef]
- Luo, J.; Craver, A.; Jin, Z.; Zheng, L.; Kim, K.; Polonsky, T.; Olopade, C.O.; Pinto, J.M.; Ahsan, H.; Aschebrook-Kilfoy, B. Contextual Deprivation, Race and Ethnicity, and Income in Air Pollution and Cardiovascular Disease. JAMA Netw Open 2024, 7, e2429137. [Google Scholar] [CrossRef]
- McClure, L.A.; Loop, M.S.; Crosson, W.; Kleindorfer, D.; Kissela, B.; Al-Hamdan, M. Fine Particulate Matter (PM(2.5)) and the Risk of Stroke in the REGARDS Cohort. J Stroke Cerebrovasc Dis 2017, 26, 1739–1744. [Google Scholar] [CrossRef] [PubMed]
- Nethery, R.C.; Josey, K.; Gandhi, P.; Kim, J.H.; Visaria, A.; Bates, B.; Schwartz, J.; Robinson, D.; Setoguchi, S. Air Pollution and Cardiovascular and Thromboembolic Events in Older Adults With High-Risk Conditions. American Journal of Epidemiology 2023, 192, 1358–1370. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Cai, R.; Su, X.; Li, Q.; Jin, S.; Shi, W.; Chen, R.; Wang, C.; He, J. Residential Nitrogen Dioxide Exposure and Cause-Specific Cerebrovascular Mortality: An Individual-Level, Case-Crossover Study. Toxics 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Su, X.; Yu, H.; Li, Q.; Jin, S.; Cai, R.; Shi, W.; Shi, S.; Meng, X.; Zhou, L.; et al. Differentiating the impact of fine and coarse particulate matter on cause-specific cerebrovascular mortality: An individual-level, case-crossover study. Ecotoxicology and environmental safety 2024, 279, 116447. [Google Scholar] [CrossRef]
- Qiu, H.; Sun, S.; Tsang, H.; Wong, C.M.; Lee, R.S.; Schooling, C.M.; Tian, L. Fine particulate matter exposure and incidence of stroke: A cohort study in Hong Kong. Neurology 2017, 88, 1709–1717. [Google Scholar] [CrossRef]
- Riggs, D.W.; Baumgartner, K.B.; Baumgartner, R.; Boone, S.; Judd, S.E.; Bhatnagar, A. Long-term exposure to air pollution and risk of stroke by ecoregions: The REGARDS study. Environmental Pollution 2024, 345, 123367. [Google Scholar] [CrossRef]
- Yitshak Sade, M.; Novack, V.; Ifergane, G.; Horev, A.; Kloog, I. Air Pollution and Ischemic Stroke Among Young Adults. Stroke 2015, 46, 3348–3353. [Google Scholar] [CrossRef]
- Sahu, S.K.; Sharma, S.; Zhang, H.; Chejarla, V.; Guo, H.; Hu, J.; Ying, Q.; Xing, J.; Kota, S.H. Estimating ground level PM(2.5) concentrations and associated health risk in India using satellite based AOD and WRF predicted meteorological parameters. Chemosphere 2020, 255, 126969. [Google Scholar] [CrossRef]
- Shamsa, E.H.; Song, Z.; Kim, H.; Shamsa, F.; Hazlett, L.D.; Zhang, K. The links of fine airborne particulate matter exposure to occurrence of cardiovascular and metabolic diseases in Michigan, USA. PLOS Glob Public Health 2022, 2, e0000707. [Google Scholar] [CrossRef]
- Shin, S.; Burnett, R.T.; Kwong, J.C.; Hystad, P.; van Donkelaar, A.; Brook, J.R.; Goldberg, M.S.; Tu, K.; Copes, R.; Martin, R.V.; et al. Ambient Air Pollution and the Risk of Atrial Fibrillation and Stroke: A Population-Based Cohort Study. Environmental health perspectives 2019, 127, 87009. [Google Scholar] [CrossRef]
- Stafoggia, M.; Renzi, M.; Forastiere, F.; Ljungman, P.; Davoli, M.; De' Donato, F.; Gariazzo, C.; Michelozzi, P.; Scortichini, M.; Solimini, A.; et al. Short-term effects of particulate matter on cardiovascular morbidity in Italy: a national analysis. European journal of preventive cardiology 2022, 29, 1202–1211. [Google Scholar] [CrossRef]
- Sun, D.; Liu, C.; Ding, Y.; Yu, C.; Guo, Y.; Sun, D.; Pang, Y.; Pei, P.; Du, H.; Yang, L.; et al. Long-term exposure to ambient PM(2·5), active commuting, and farming activity and cardiovascular disease risk in adults in China: a prospective cohort study. Lancet Planet Health 2023, 7, e304–e312. [Google Scholar] [CrossRef] [PubMed]
- Tapia, V.; Steenland, K.; Sarnat, S.E.; Vu, B.; Liu, Y.; Sánchez-Ccoyllo, O.; Vasquez, V.; Gonzales, G.F. Time-series analysis of ambient PM(2.5) and cardiorespiratory emergency room visits in Lima, Peru during 2010-2016. J Expo Sci Environ Epidemiol 2020, 30, 680–688. [Google Scholar] [CrossRef] [PubMed]
- To, T.; Zhu, J.; Villeneuve, P.J.; Simatovic, J.; Feldman, L.; Gao, C.; Williams, D.; Chen, H.; Weichenthal, S.; Wall, C.; et al. Chronic disease prevalence in women and air pollution--A 30-year longitudinal cohort study. Environment international 2015, 80, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Valdez, R.B.; Al-Hamdan, M.Z.; Tabatabai, M.; Hood, D.B.; Im, W.; Wilus, D.; Nori-Sarma, A.; Ramesh, A.; Donneyong, M.M.; Langston, M.A.; et al. Association of Cardiovascular Disease and Long-Term Exposure to Fine Particulate Matter (PM(2.5)) in the Southeastern United States. Atmosphere (Basel) 2021, 12, 1–12. [Google Scholar] [CrossRef]
- Wang, M.; Han, Y.; Wang, C.J.; Xue, T.; Gu, H.Q.; Yang, K.X.; Liu, H.Y.; Cao, M.; Meng, X.; Jiang, Y.; et al. Short-term effect of PM2.5 on stroke in susceptible populations: A case-crossover study. Int J Stroke 2023, 18, 312–321. [Google Scholar] [CrossRef]
- Wettstein, Z.S.; Hoshiko, S.; Fahimi, J.; Harrison, R.J.; Cascio, W.E.; Rappold, A.G. Cardiovascular and Cerebrovascular Emergency Department Visits Associated With Wildfire Smoke Exposure in California in 2015. Journal of the American Heart Association 2018, 7. [Google Scholar] [CrossRef]
- Wong, C.M.; Lai, H.K.; Tsang, H.; Thach, T.Q.; Thomas, G.N.; Lam, K.B.; Chan, K.P.; Yang, L.; Lau, A.K.; Ayres, J.G.; et al. Satellite-Based Estimates of Long-Term Exposure to Fine Particles and Association with Mortality in Elderly Hong Kong Residents. Environmental health perspectives 2015, 123, 1167–1172. [Google Scholar] [CrossRef]
- Wu, Y.; Shen, P.; Yang, Z.; Yu, L.; Xu, L.; Zhu, Z.; Li, T.; Luo, D.; Lin, H.; Shui, L.; et al. Outdoor Light at Night, Air Pollution, and Risk of Cerebrovascular Disease: A Cohort Study in China. Stroke 2024, 55, 990–998. [Google Scholar] [CrossRef]
- Yang, L.; Wang, M.; Xuan, C.; Yu, C.; Zhu, Y.; Luo, H.; Meng, X.; Shi, S.; Wang, Y.; Chu, H.; et al. Long-term exposure to particulate matter pollution and incidence of ischemic and hemorrhagic stroke: A prospective cohort study in Eastern China. Environmental pollution (Barking, Essex 1987) 2024, 358, 124446. [Google Scholar] [CrossRef]
- Yang, L.; Zhu, Y.; Zhao, B.; Wan, W.; Shi, S.; Xuan, C.; Yu, C.; Mao, W.; Yan, J. Long-term cardiometabolic effects of ambient ozone pollution in a large Chinese population. Ecotoxicology and environmental safety 2023, 261, 115115. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, L.; Chen, X.; Liu, F.; Shan, A.; Liang, F.; Li, X.; Wu, H.; Yan, M.; Ma, Z.; et al. Long-term exposure to ambient PM(2.5) and stroke mortality among urban residents in northern China. Ecotoxicology and environmental safety 2021, 213, 112063. [Google Scholar] [CrossRef] [PubMed]
- Yitshak-Sade, M.; Bobb, J.F.; Schwartz, J.D.; Kloog, I.; Zanobetti, A. The association between short and long-term exposure to PM(2.5) and temperature and hospital admissions in New England and the synergistic effect of the short-term exposures. The Science of the total environment 2018, 639, 868–875. [Google Scholar] [CrossRef] [PubMed]
- Yitshak-Sade, M.; Kloog, I.; Novack, V. Do air pollution and neighborhood greenness exposures improve the predicted cardiovascular risk? Environment international 2017, 107, 147–153. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, C.; Lin, C.; Wu, Y.; Wei, J.; Lu, J.; Chen, B.; Wu, C.; Zhang, X.; Yang, Y.; et al. Association of long-term exposure to ozone with cardiovascular mortality and its metabolic mediators: evidence from a nationwide, population-based, prospective cohort study. The Lancet Regional Health – Western Pacific 2024, 52. [Google Scholar] [CrossRef]
- Zheng, M.; Yin, Z.; Wei, J.; Yu, Y.; Wang, K.; Yuan, Y.; Wang, Y.; Zhang, L.; Wang, F.; Zhang, Y. Submicron particle exposure and stroke hospitalization: An individual-level case-crossover study in Guangzhou, China, 2014-2018. The Science of the total environment 2023, 886, 163988. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, X.; Fouxi, Z.; Yao, C.; Wang, Y.; Tang, E.; Wang, K.; Yu, L.; Zhou, Z.; Wei, J.; et al. Rural-urban difference in the association between particulate matters and stroke incidence: The evidence from a multi-city perspective cohort study. Environ Res 2024, 261, 119695. [Google Scholar] [CrossRef]
- Zhu, L.; Fang, J.; Yao, Y.; Yang, Z.; Wu, J.; Ma, Z.; Liu, R.; Zhan, Y.; Ding, Z.; Zhang, Y. Long-term ambient ozone exposure and incident cardiovascular diseases: National cohort evidence in China. J Hazard Mater 2024, 471, 134158. [Google Scholar] [CrossRef]
- Brown, S.C.; Aitken, W.W.; Lombard, J.; Wang, K.; Rundek, T.; Byrne, M.M.; Toro, M.; Nardi, M.I.; Kardys, J.; Parrish, A. Precision greenness and stroke/transient ischemic attack in 249,405 US Medicare beneficiaries. Journal of stroke 2023, 25, 173–176. [Google Scholar] [CrossRef]
- Jia, X.; Yu, Y.; Xia, W.; Masri, S.; Sami, M.; Hu, Z.; Yu, Z.; Wu, J. Cardiovascular diseases in middle aged and older adults in China: the joint effects and mediation of different types of physical exercise and neighborhood greenness and walkability. Environ Res 2018, 167, 175–183. [Google Scholar] [CrossRef]
- Jia, X.; Zhang, B.; Yu, Y.; Xia, W.; Lu, Z.; Guo, X.; Xue, F. Greenness mitigate cause-specific mortality associated with air pollutants in ischemic and hemorrhagic stroke patients: An ecological health cohort study. Environmental Research 2024, 251, 118512. [Google Scholar] [CrossRef]
- Liu, C.; Yu, Y.; Liu, C.; Tang, L.; Zhao, K.; Zhang, P.; He, F.; Wang, M.; Shi, C.; Lu, Z.; et al. Effect of neighbourhood greenness on the association between air pollution and risk of stroke first onset: A case-crossover study in shandong province, China. International journal of hygiene and environmental health 2023, 254, 114262. [Google Scholar] [CrossRef]
- Song, S.; Cheng, C.; Liu, Y.; Duan, Y.; Zuo, H.; Xi, R.; Ni, Z.; Liang, K.; Li, S.; Cui, F.; et al. Associations between short-term exposure to fine particulate matter with ischemic stroke mortality and the role of green space: a time-series study in Zibo, China. J Glob Health 2025, 15, 04068. [Google Scholar] [CrossRef]
- Wang, H.; Tassinary, L.G. Association between greenspace morphology and prevalence of non-communicable diseases mediated by air pollution and physical activity. Landscape and Urban Planning 2024, 242, 104934. [Google Scholar] [CrossRef]
- Zhao, K.; He, F.; Zhang, B.; Liu, C.; Hu, Y.; Dong, Y.; Zhang, P.; Liu, C.; Wei, J.; Lu, Z.; et al. Short-term ozone exposure on stroke mortality and mitigation by greenness in rural and urban areas of Shandong Province, China. BMC public health 2024, 24, 2955. [Google Scholar] [CrossRef]
- Wang, H.; Tassinary, L.G.; Newman, G.D. Developing the health effect assessment of landscape (HEAL) Tool: Assessing the health effects of community greenspace morphology design on non-communicable diseases. Landscape and Urban Planning 2024, 244, 104990. [Google Scholar] [CrossRef]
- Cai, L.; Yang, J.; Cosky, E.; Xin, R.; Geng, X.; Ding, Y. Enhanced Cerebral Microbleeds by Long-Term Air Pollution Exposure in Spontaneously Hypertensive Rats. Neurol Res 2022, 44, 196–205. [Google Scholar] [CrossRef]

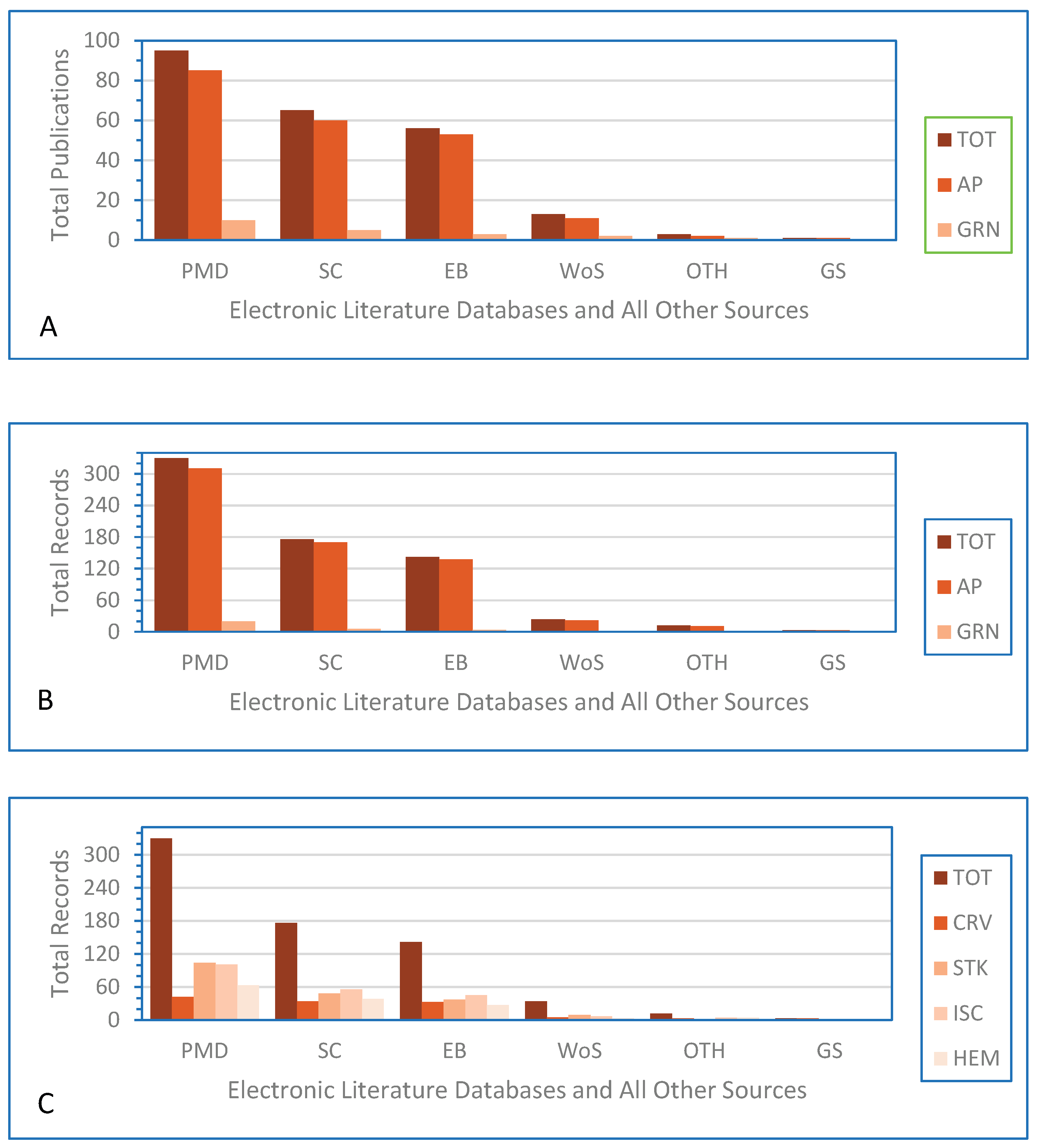
| Variables | Publications1 | Records1 |
| N (%) or Mean (95% CI)2 | N (%) or Mean (95% CI)2 | |
| Research Publications or Extracted Records | 99 (100.00) | 343 (100.00) |
| Stroke Participant Totals | 7,555,646 | 26,513,512 |
| Age Mean (95% CI)2 | 62.21 (59.02-66.39) | 42.8 (39.4-46.2) |
| Publication Year Category | ||
| 2012-2018 | 25 (25.25)b | 20 (5.83)b |
| 2019-2025 | 74 (74.75) | 323 (94.17) |
| Remote Sensing | ||
| Air Pollution | 88 (88.89)b | 315 (93.75)b |
| Greenness | 11 (11.11) | 21 (6.25) |
| Publication Year by Remote Sensing | ||
| 2012-2018 Greenness | 3 (3.03) | 3 (0.87) |
| 2012-2018 Air Pollution | 22 (22.22) | 37 (10.79) |
| 2019-2025 Greenness | 8 (8.08) | 17 (4.96) |
| 2019-2025 Air Pollution | 66 (66.67) | 286 (83.38) |
| Country | ||
| China | 47 (47.47)b | 240 (69.97)b |
| United States | 16 (16.16) | 38 (11.08) |
| 2+ | 9 (9.09) | 13 (3.79) |
| Canada | 5 (5.05) | 10 (2.92) |
| Indonesia | 5 (5.05) | 5 (1.46) |
| Israel | 3 (3.03) | 9 (2.62) |
| Italy | 2 (2.02) | 6 (1.75) |
| Mexico | 2 (2.02) | 6 (1.75) |
| Australia | 1 (1.01) | 3 (0.87) |
| Belgium | 1 (1.01) | 1 (0.29) |
| Brazil | 1 (1.01) | 1 (0.29) |
| Iran | 1 (1.01) | 1 (0.29) |
| Malysia | 1 (1.01) | 2 (0.58) |
| Peru | 1 (1.01) | 1 (0.29) |
| Republic of Korea | 1 (1.01) | 2 (0.58) |
| Saudi Arabia | 1 (1.01) | 2 (0.58) |
| Spain | 1 (1.01) | 1(0.29) |
| Thailand | 1 (1.01) | 2 (0.58) |
| Stroke Subtype | ||
| Cerebrovascular | 22 (22.22)b | 47 (13.70)b |
| Stroke | 47 (47.47) | 111 (32.36) |
| Ischemic | 23 (23.23) | 102 (29.74) |
| Hemorrhagic | 7 (7.07) | 83 (24.20) |
| Record Format | ||
| One | 73 (73.74)b | 292 (85.13)b |
| 2+ | 5 (5.05) | 15 (4.37) |
| Concentration-Response Function | 20 (20.20) | 35 (10.20) |
| Z | 1 (1.01) | 1 (0.29) |
| ICD | ||
| Nine | 11 (11.11)b | 29 (8.45)b |
| Ten | 43 (43.43) | 224 (65.31) |
| Both | 6 (6.06) | 17 (4.96) |
| Other | 19 (19.19) | 40 (11.66) |
| Z | 20 (20.20) | 33 (9.62) |
| Surveillance Type | ||
| Prevalence | 28 (28.28)b | 87 (25.36)b |
| Incidence | 60 (60.61) | 241 (70.26) |
| Both | 1 (1.01) | 1 (0.29) |
| Other | 8 (8.08) | 12 (3.50) |
| Z | 2 (2.02) | 2 (0.58) |
| Outcome | ||
| Morbidity | 46 (46.46)b | 159 (46.36)b |
| Mortality | 45 (45.45) | 162 (47.23) |
| Both | 8 (8.08) | 22 (6.41) |
| Design | ||
| Case Control | 2 (2.02)b | 3 (0.87)b |
| Case Crossover | 14 (14.14) | 93 (27.11) |
| Concentration Response Function | 18 (18.18) | 27 (7.87) |
| Cross Sectional | 8 (8.08) | 17 (4.96) |
| Other | 3 (3.03) | 3 (0.87) |
| Panel | 1 (1.01) | 8 (2.33) |
| Prospective Cohort | 34 (34.34) | 120 (34.99) |
| Retrospective Cohort | 11 (11.11) | 55 (16.03) |
| Time Series | 8 (8.08) | 17 (4.96) |
| Physiologic Mechanism in Introduction | ||
| No | 80 (80.81)b | 263 (76.68)b |
| Yes | 19 (19.19) | 80 (23.32) |
| Aerosol Optical Depth | ||
| No | 16 (16.16)b | 32 (9.33)b |
| Yes | 82 (82.83) | 310 (90.38) |
| Z | 1 (1.01) | 1 (0.29) |
| Monitor | ||
| No | 11 (11.96)b | 22 (7.42)b |
| Yes | 79 (85.87) | 285 (91.94) |
| Z | 2 (2.17) | 2 (0.65) |
| Geocode | ||
| Address | 40 (40.40)6 | 207 (60.35)b |
| Block Group | 1 (1.01) | 1 (0.29) |
| Census Block | 2 (2.02) | 9 (2.68) |
| Census Tract | 3 (3.03) | 4 (1.17) |
| Zip Code | 11 (11.11) | 20 (5.83) |
| Neighborhood | 6 (6.06) | 15 (4.37) |
| City | 9 (9.09) | 28 (8.16) |
| County | 6 (6.06) | 21 (6.12) |
| State | 3 (3.03) | 4 (1.17) |
| Country | 6 (6.06) | 16 (4.66) |
| World region | 3 (3.03) | 4 (1.17) |
| Other | 5 (5.05) | 7 (2.08) |
| Z | 6 (6.06) | 6 (1.75) |
| Remote Sensing Air Pollution and Greenness | ||
| <1 Year | 32 (32.32)b | 140 (44.82)b |
| ≥1 Year | 67 (67.68) | 203 (59.18) |
| Remote Sensing Air Pollution and Greenness | ||
| <1 Year Greenness | 2 (2.02) | 3 (0.87)a |
| <1 Year Air Pollution | 30 (30.30) | 137 (39.94) |
| 1+ Years Greenness | 9 (9.09) | 17 (4.96) |
| 1+ Years Air Pollution | 58 (58.59) | 186 (54.23) |
| Outcome | ||
| Not significant | 15 (15.15)b | 82 (23.91)b |
| Significantly higher | 54 (54.55) | 205 (59.77) |
| Significantly lower | 10 (10.10) | 28 (8.16) |
| Other | 3 (3.03) | 3 (0.87) |
| Z | 17 (17.17) | 25 (7.29) |
| Variable1 | N2 (%) | Exposure3 | Accuracy (r2%)4 | ||
| Mean | 95% CI5 | Mean | 95% CI4 | ||
| AOD-PM10 | 35 (12.15) | 68.1 | 59.6-76.7 | 84.7 | 82.1-87.3 |
| AOD-PM2.5 | 165 (57.29) | 32.8 | 29.2-36.4 | 75.1 | 72.0-78.2 |
| AOD-PM1 | 17 (5.90) | 29.6 | 24.2-34.9 | 82.2 | 79.2-85.1 |
| AOD-O3 | 20 (6.94) | 89.3 | 78.4-100.3 | 86.7 | 85.9-87.5 |
| AOD-NO2 | 24 (8.33) | 29.6 | 25.5-33.8 | 78.6 | 75.6-81.6 |
| AOD-SO2 | 10 (3.47) | 23.3 | 18.1-28.4 | 83.2 | 79.0-87.4 |
| EVI | 2 (0.69) | 0.3 | . | . | . |
| NDVI | 15 (5.21) | 0.4 | 0.3-0.5 | . | . |
| AREA1 |
RS2 | Type3 | CRF4 | Outcomes |
| 2+ [47] | AOD-PM2.5 | STK, ISC | GBD | In 2019 there were 1.14 million incident STK deaths, which included 516,000 ISC stroke deaths. |
| 2+ [52] | AOD-PM2.5 | CRV | GBD | In 2015 there were 898,100 CRV deaths. |
| 2+ [53] | AOD-O3, AOD-PM2.5 | STK |
RR | In 2019 there were 1, 332,000 STK deaths. |
| 2+ [48] | AOD-PM2.5 | STK | GBD | In 2019 there were 1.14 million STK deaths. |
| 2+ [54] | AOD-PM2.5 | STK | ERF | In South-Southeast Asia, between 1999-2014, there were 556,400 premature STK deaths per year. |
| 2+ [55] | AOD-PM2.5 | STK | ERF | Between 2000-2018 STK deaths increased in the world by 0.67% per year, and 2.78% Southeast Asia (>4% in 2000-2006). |
| 2+ [56] | AOD-PM2.5 | STK | GBD | Between 2000-2018, STK death percentages were highest in Asia (40.2%, n=909,000/2,261,000), intermediate in Africa (31.8%, n=54,000/170,000), and lowest in Europe (30.9%, n= 120,000/ 388,000). |
| CN [57] | AOD-PM2.5 | STK | GBD | Between 2003-2018, in central China STK mortality decreased from 143,000 to 103,000. |
| CN [58] | AOD-SO2 | CRV | RF-STKR | Between May 2014 and May 2015 there were 2,188 avoidable premature CRV stroke deaths. |
| CN [59] | AOD-PM2.5 | STK | GBD | Premature STK deaths increased from 589,000 in 2004 and to 761,000 in 2012. |
| CN [60] | AOD-PM2.5 | STK | GEM | Between 2005-2017 there were 580,000 premature STK deaths, representing 36% of all PM2.5 deaths. |
| CN [61] | AOD-PM2.5 | STK | GBD | STK age-standardized disability-adjusted life year rates were 3.99 in 1995-2001, -1.31 in 2001-2004 and -1.98 in 2012-219. |
| CN [62] | AOD-PM2.5 | STK | GBD | In this county-level 2010 study there were 670,906 STK deaths; this total represents 52.90% of all PM2.5 impacted premature deaths. |
| IN [63] | AOD-PM2.5 | STK | NLP | There were 88,700 premature STK deaths. |
| IN [64,65] | AOD-PM2.5 | STK | GBD | Between 2001-2015, in Veranasi, there were 1,000 premature STK deaths per year. |
| IN [65] | AOD-PM2.5 | STK | GBD | Between 2018-2019, in central India (Madhya Pradesh) there were 37,468 STK deaths. |
| KR [66] | AOD-PM2.5 | HEM, ISC | GBD | Between 1990-2013 there were 5,382 ISC stroke and 3,452 HEM stroke deaths. |
| MA [67] | AOD-PM2.5 | STK | RR | There were 1,329 STK deaths in 2,000, 1,062 in 2008, and 2,745 in 2013. |
| SA [68] | AOD-PM2.5 | STK | GBD | STK deaths increased from 554 in 1990, to 986 in 2010, and then to 1,056 in 2017. |
| US [69] | AOD-PM2.5 | STK | RR | In 2000-2015 there were 10 STK deaths in Washington, DC. |
| US [70] | AOD-PM2.5 |
STK | GBD | Between 2010-2018 there were approximately 7,500 STK deaths per year, with higher totals among Black persons and lower totals among White persons. |
| Variable1 | Outcome2-3 | |||
| Higher | Lower | NS | Total | |
| Stroke Subtypeb | ||||
| Cerebrovascular | 24 (10.48) |
0 (0.00) |
6 (2.62) |
30 (13.10) |
| Stroke | 48 (20.96) |
3 (1.31) |
10 (4.37) |
61 (26.64) |
| Ischemic | 65 (28.38) |
2 (0.87) |
10 (4.37) |
77 (33.62) |
| Hemorrhagic | 33 (14.41) |
8 (3.49) |
20 (8.73) |
61 (26.64) |
| Geocoding Specificityb | ||||
| Residential Address | 125 (54.59) |
4 (1.75) |
27 (11.79) |
156 (68.12) |
| Other | 45 (19.65) |
9 (3.93) |
19 (8.30) |
73 (31.88) |
| Spatial Resolution | ||||
| <10 km2 | 92 (42.20) |
9 (4.13) |
34 (15.60) |
135 (61.93) |
| ≥10 km2 | 67 (30.73) |
4 (1.83) |
12 (5.50) |
83 (38.07) |
| Remote Sensing Air Pollutantsb | ||||
| AOD-Black Carbon (BC) | 1 (0.44) |
0 (0.00) |
0 (0.00) |
1 (0.44) |
| AOD-Carbon Monoxide (CO) | 5 (2.18) |
0 (0.00) |
4 (1.75) |
9 (3.93) |
| AOD-Nitrogen Dioxide (NO2) | 16 (6.99) |
3 (1.31) |
4 (1.75) |
23 (10.04) |
| AOD-Ozone (O3) | 10 (4.37) |
5 (2.18) |
4 (1.75) |
19 (8.30) |
| AOD-Ultrafine PM (PM1) | 15 (6.55) |
1 (0.44) |
1 (0.44) |
17 (7.42) |
| AOD-Fine PM (PM2.5) | 88 (38.43) |
1 (0.44) |
22 (9.61) |
111 (48.47) |
| AOD-Intermediate PM (PM2.5-10) | 2 (0.87) |
0 (0.00) |
4 (1.75) |
6 (2.62) |
| AOD-Coarse PM (PM10) | 28 (12.23) |
1 (0.44) |
5 (2.18) |
34 (14.85) |
| AOD-Sulfur Dioxide (SO2) | 5 (2.18) |
2 (0.87) |
2 (0.87) |
9 (3.93) |
| AOD-Air Pollution Readings | ||||
| <37.60 µg/m3 | 43 (18.78) |
1 (0.44) |
11 (4.80) |
55 (24.02) |
| ≥37.60 µg/m3 | 127 (55.46) |
12 (5.24) |
35 (15.28) |
174 (75.98) |
| AOD-Air Pollution Exposure | ||||
| <1 Year | 85 (37.12) |
10 (4.37) |
29 (12.66) |
124 (54.15) |
| 1+ Years | 85 (37.12) |
3 (1.31) |
17 (7.42) |
105 (45.85) |
| AOD-Air Pollutant Accuracy (r2%)b | ||||
| <84.00% | 68 (29.69) |
1 (0.44) |
27 (11.79) |
96 (41.92) |
| ≥84.00% | 102 (44.54) |
12 (5.24) |
19 (8.30) |
133 (58.08) |
| Country | ||||
| Canada | 7 (3.06) |
0 (0.00) |
1 (0.44) |
8 (3.49) |
| China | 133 (58.08) |
12 (5.24) |
34 (14.85) |
179 (78.17) |
| Israel | 5 (2.18) |
1 (0.44) |
3 (1.31) |
9 (3.93) |
| Mexico | 3 (1.31) |
0 (0.00) |
3 (1.31) |
6 (2.62) |
| United States | 12 (5.24) |
0 (0.00) |
2 (0.87) |
14 (6.11) |
| Other | 10 (4.37) |
0 (0.00) |
3 (1.31) |
13 (5.68) |
| Publication Year Category | ||||
| 2012-2018 | 15 (6.55) |
0 (0.00) |
7 (3.06) |
22 (9.61) |
| 2019-2025 | 155 (67.69) |
13 (5.68) |
39 (17.03) |
207 (90.39) |
| Risk Factor1 |
Stroke Subtype2 | total | |||
| ISCb, b | STKb, ab | HEMb, a | CRVb | ||
| Environmentalb | 122 (20.37) |
87 (14.52) |
70 (11.69) |
50 (8.35) |
329 (54.92) |
| Otherb | 23 (3.84) |
46 (7.68) |
12 (2.00) |
9 (1.50) |
90 (15.03) |
| Agea | 21 (3.51) |
19 (3.17) |
8 (1.34) |
9 (1.50) |
57 (9.52) |
| Gender | 16 (2.67) |
15 (2.50) |
10 (1.67) |
8 (1.34) |
49 (8.18) |
| Geographic | 14 (2.34) |
14 (2.34) |
9 (1.50) |
5 (0.83) |
42 (7.01) |
| Education-Income | 4 (0.67) |
9 (1.50) |
1 (0.17) |
6 (1.00) |
20 (3.34) |
| Ethnicity | 2 (0.33) |
6 (1.00) |
0 (0.00) |
0 (0.00) |
8 (1.34) |
| Race | 2 (0.33) |
2 (0.33) |
0 (0.00) |
0 (0.00) |
4 (0.67) |
| Total | 204 (34.06) |
198 (33.06) |
110 (18.36) |
87 (14.52) |
599 (100.00) |
| AREA1 |
RS2 | Type3 | Outcomes4 |
| AU [15] | AOD-PM2.5 AOD-NO2 NDVI |
STK | In an ecologic cross-sectional study of 699 neighborhoods, AOD-PM2.5 and AOD-NO2 were positively and significantly associated with self-reported STK prevalence, while NDVI greenness was negatively and significantly associated with STK prevalence. |
| BE [43] | NDVI |
CRV | In a prospective study of 23,003 CRV adults at least 30 years old, NDVI greenness did not significantly decrease CRV mortality. |
| BR [16] | NDVI | CRV | In an ecologic study of census tracts, study participants exposed to fourth quartile NDVI values showed significantly lower CRV mortality. |
| CA [18] | NDVI | STK | In a retrospective cohort study of 89,958 persons, an interquartile NDVI increase produced a significant decrease in STK incidence, even after controlling for SES and ambient air pollution. |
| CA [19] | NDVI | CRV | In a cohort of 5,955 non-immigrant persons in 30 CA cities, higher NDVI values were negatively and significantly associated with lower CRV mortality after controlling for ambient air pollutants. |
| CN [127] | AOD-PM2.5 NDVI |
ISC | The AOD-PM2.5 produced increase in ISC stroke mortality was significant in an area with a lower NDVI value of 0.31, but not in an area with a higher NDVI value of 0.57. |
| CN [125] | AOD-PM2.5 AOD-NO2 EVI |
HEM ISC |
Compared to the first two EVI tertiles, the highest EVI tertile significantly decreased the contribution of AOD-PM2.5 and AOD-NO2 to increased HEM stroke and ISC stroke mortality risk. |
| CN [126] | AOD-PM2.5 AOD-NO2 AOD-O3 AOD-SO2 AOD-CO EVI |
HEM ISC STK |
Results from a case-crossover design showed that higher neighborhood EVI values significantly decreased AOD-NO2 risk of first HEM stroke, ISC stroke, and STK. |
| CN [44] | AOD-O3 EVI NDVI |
HEM | AOD-O3 significantly increased HEM stroke mortality only during the warm season, but higher NDVI and EVI values significantly decreased HEM stroke mortality during the warm season and the entire year. |
| CN [129] | AOD-O3 EVI |
HEM ISC |
In urban counties only, higher EVI values significantly mitigated the AOD-O3 produced increase in ISC stroke mortality but not HEM stroke mortality. |
| CN [124] | NDVI | STK | In a cross-sectional study, higher movement activity or higher NDVI values were negatively and significantly associated with 25% and 45% decreased stroke risk, respectively. |
| CN [21] | NDVI | HEM ISC STK |
In a prospective study of adults who were 30-79 years old, a 0.1 increase in the NDVI maximum value was inversely and significantly associated with lower STK and ISC stroke risk but not HEM stroke risk. |
| ES [20] | NDVI | ISC | In Barcelona, higher third and fourth quartile NDVI values were negatively and significantly associated with lower acute ISC stroke hospitalization risk. |
| IT [22] | NDVI | CRV STK |
In Rome, 5th NDVI quintile values were negatively and significantly associated with lower CRV mortality and STK incidence. |
| US [123] | NDVI | STK/TIA | In a cross-sectional study completed in Miami-Dade County, Florida, higher census block NDVI values were negatively and significantly associated with decreased STK/TIA risk after controlling for SES. |
| US [128] | GRN MAPS | STK | In Los Angeles, a one interquartile increase in greenspace aggregation was associated with a 0.44% decrease in STK prevalence. |
| US [17] | EVI NDVI |
STK | The Reasons for Geographic and Racial Differences in Stroke cohort study found significant inverse associations between higher EVI and NDVI values and lower STK incidence risk in the Stroke Belt. |
| PHYSIOLOGIC MECHANISMS1,3 | Stroke Subtype2, 3-4 | ||||
| CRVb, ab | HEMb, ac | ISCb, bd | STKb, cd | Total | |
| IM | 0 (0.00) | 5 (1.49) | 7 (2.08) | 8 (2.38) | 20 (5.95) |
| IM-IN | 2 (0.60) | 2 (0.60) | 2 (0.60) | 1 (0.30) | 7 (2.08) |
| IM-IN-OS | 0 (0.00) | 6 (1.79) | 6 (1.79) | 9 (2.68) | 21 (6.25) |
| IM-OS | 0 (0.00) | 3 (0.89) | 2 (0.60) | 1 (0.30) | 6 (1.79) |
| INb | 6 (1.79) | 18 (5.36) | 28 (8.33) | 13 (3.87) | 65 (19.35) |
| IN-OSb | 11 (3.27) | 32 (9.52) | 35 (10.42) | 32 (9.52) | 110 (32.74) |
| OS | 3 (0.89) | 0 (0.00) | 4 (1.19) | 6 (1.79) | 13 (3.87) |
| OTHa | 24 (7.14) | 17 (5.06 | 17 (5.06) | 36 (10.71) | 94 (27.98) |
| Total | 46 (13.69) | 83 (24.70) | 101 (30.06) | 106 (31.55) | 336 (100.00) |
| Activities | Available Options2 |
| Development Stage | |
| Stroke Subtype | CRV, HEM, ISC, STK |
| Risk Factors | Environmental, Study participant Attributes, OTH |
| Pollutants | Ambient AOD-air pollutant, Industrial, or Indoor |
| Constituents | Lead, Dust. Soil, OTH |
| Physiologic Mechanisms | IM, IN, OS, OTH, 2+ |
| Greenness (Protective) | EVI, NDVI |
| Resources | Public health infrastructure, Health care system |
| Objectives | Define outcomes as measurable endpoints |
| Implementation Stage | |
| Infrastructure | Hire and train employees |
| Location | Identify intervention catchment area |
| Recruitment | Enroll intervention study participants |
| Duration | Establish starting and ending dates |
| Subgoals | Internal assessment of milestone attainment |
| Evaluation Stage | |
| Accomplishments | Statistically evaluate intervention outcomes |
| Confirmation | Agreement with previously reported outcome |
| Unique Outcomes | Describe importance of new findings |
| Dissemination Stage | |
| Study Participants | Describe intervention results |
| Study Area | Present program results to community members |
| Professionals | Conference presentations |
| Scientific Journals | Publish results |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




