2. Materials and Methods
2.1. Starting Materials
Commercially available water-atomized iron powder (RD grade) was used as the base material for the preparation of gas-phase surface-modified soft magnetic composite powders. According to the supplier’s technical specification and subsequent elemental verification, the powder possessed high iron purity with limited oxygen content attributed to native surface oxidation formed during production and storage.
The morphology of the as-received powder consisted of irregularly shaped particles with developed surface roughness, typical of atomized iron powders. Representative microstructure of the initial powder is shown in
Figure 4a, demonstrating pronounced surface asperities and localized porosity.
To ensure suitability for low-frequency soft magnetic composite applications and to reduce interparticle eddy current pathways, the powder was mechanically classified using size separation. Only fractions with particle sizes exceeding 200 µm were selected for further processing. The use of coarse particles is advantageous in reducing the relative volume fraction of insulating material while maintaining high magnetic induction during subsequent compaction.
The chemical composition of the initial iron powder determined by EDS analysis is summarized in
Table 1.
The results confirm that iron is the dominant constituent of the powder, while the detected oxygen originates from thin native oxide films typically present on commercial iron powders. No additional metallic impurities were detected within the sensitivity limits of the analytical technique, indicating high starting material purity suitable for controlled surface modification.
Prior to gas-phase treatment, the powder was subjected to surface passivation in order to suppress uncontrolled oxidation during thermal processing. A high-temperature-resistant silicone-based additive (0.25 wt.% relative to powder mass) was introduced and mechanically distributed using a laboratory-scale ball mill under ambient conditions. The homogenization process ensured uniform additive coverage without inducing plastic deformation or particle fragmentation.
The ball milling setup used for homogenization is presented in
Figure 1.
The controlled preparation of the starting powder, including particle size classification and surface passivation, represents a critical preliminary step for achieving reproducible coating formation during subsequent gas-phase phosphating.
2.2. Gas-Phase Insulating Coating Procedure
The electrically insulating surface layer was deposited using a controlled gas-phase phosphating process specifically adapted for micron-sized iron powders. The passivated powder fraction (>200 µm) was placed inside a cylindrical stainless-steel reactor equipped with a rotating drum mechanism to ensure continuous particle agitation during treatment. This dynamic configuration enabled homogeneous exposure of the entire particle surface to the reactive gas atmosphere and minimized local deposition gradients.
The experimental reactor setup is shown in
Figure 2.
Prior to heating, the reactor chamber was evacuated using a rotary vacuum pump to achieve a residual pressure in the range of 10⁻²–10⁻³ mmHg. The reduced-pressure environment served two essential purposes: (i) removal of atmospheric oxygen and moisture to suppress uncontrolled oxidation, and (ii) facilitation of uniform vapor-phase transport of the insulating precursor.
After achieving the target vacuum level, the reactor was heated to 200 °C at a controlled heating rate to avoid thermal shock and localized overheating. The selected temperature represents an optimized balance between activation of surface reactions and preservation of the crystalline α-Fe phase. The temperature was maintained throughout the coating cycle to promote controlled surface conversion.
The reactive atmosphere consisted of vapors of a phosphate-forming precursor introduced in combination with a volatile carrier component. Under reduced pressure and elevated temperature, the precursor decomposed and reacted at the iron surface, forming a phosphate-based insulating layer. Continuous mechanical agitation of the powder ensured that particles remained in a quasi-fluidized dynamic state, enabling conformal coating development over complex surface geometries.
To regulate coating thickness, the deposition cycle could be repeated multiple times. Each cycle consisted of:
Vacuum stabilization
Controlled heating to 200 °C
Exposure to reactive vapor phase
Gradual removal of volatile components
Controlled cooling under reduced pressure
This stepwise deposition strategy enabled precise thickness control ranging from thin nanometric films to thicker micrometric insulating layers without inducing sintering or particle fusion.
After completion of the coating cycle, the reactor was cooled to room temperature under vacuum conditions to minimize post-process oxidation. The treated powders were then removed and stored in sealed containers prior to characterization.
The use of a vacuum-assisted gas-phase route provides several advantages compared to conventional wet phosphating, including improved coating uniformity, reduced solvent contamination, enhanced thickness controllability, and compatibility with scalable powder metallurgy workflows.
2.3. Lubrication and Post-Treatment of Coated Powders
Following gas-phase phosphating, the coated iron powders were subjected to a controlled lubrication and thermal stabilization procedure to improve powder flowability, interparticle rearrangement during compaction, and coating integrity.
A peat-wax-based lubricant was introduced in an amount of 0.15 wt.% relative to the initial powder mass. The lubricant was dissolved in a volatile carrier medium and uniformly distributed over the coated particles using low-energy mechanical mixing in a laboratory-scale ball mill. The mixing was performed under mild conditions to prevent mechanical damage to the phosphate-based insulating layer and to avoid particle fragmentation or coating delamination.
The purpose of lubrication was twofold:
After lubrication, the composite powder underwent thermal post-treatment inside the same cylindrical reactor used for gas-phase coating. The chamber was evacuated to a pressure of 10⁻²–10⁻³ mmHg to remove residual atmospheric gases and volatile components introduced during lubrication.
The reactor was then heated to 200 °C and maintained at this temperature for 20 minutes under reduced pressure. This stabilization step served to:
remove residual solvent traces,
enhance adhesion between the phosphate-based layer and the iron substrate,
promote structural relaxation of the coating,
reduce the risk of microcracking during subsequent mechanical consolidation.
Cooling was carried out under vacuum conditions to minimize re-oxidation of the iron surface. For samples requiring increased insulation thickness, the gas-phase coating and post-treatment cycles were repeated sequentially. The multi-cycle approach enabled systematic control over coating thickness while maintaining crystallographic stability and particle integrity.
The integration of lubrication and thermal stabilization into the processing route ensures compatibility of the coated powders with conventional powder metallurgy workflows, particularly cold pressing and low-temperature consolidation methods.
2.4. Structural and Microstructural Characterization
2.4.1. X-Ray Diffraction (XRD) Analysis
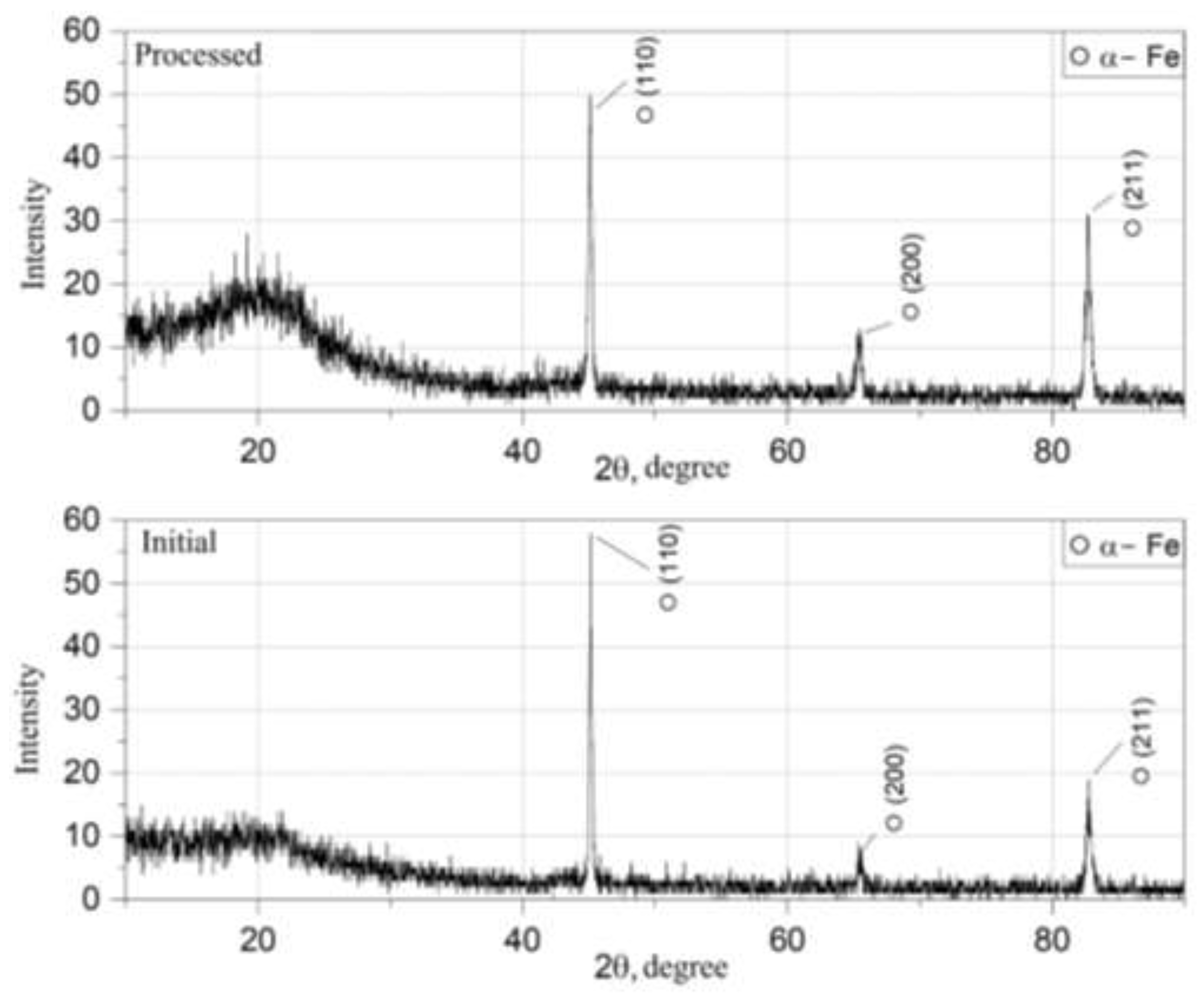
Phase composition and crystallographic stability of both initial and gas-phase treated powders were analyzed using X-ray diffraction (XRD). Measurements were performed using Cu Kα radiation (λ = 1.5406 Å) over a 2θ range of 20°–90°, with a scanning step of 0.02° and a counting time sufficient to ensure adequate signal-to-noise ratio.
Diffraction patterns of the initial and treated powders are presented in
Figure 3.
The diffraction data were analyzed to identify possible phase transformations, oxide formation, or secondary crystalline compounds induced by thermal exposure during phosphating. Peak positions were compared with standard reference data for body-centered cubic α-Fe. Particular attention was given to potential reflections corresponding to iron oxides or crystalline phosphate phases.
Preservation of the α-Fe phase and absence of additional crystalline peaks were used as criteria for structural stability during gas-phase treatment.
2.4.2. Scanning Electron Microscopy (SEM)
Surface morphology and coating development were investigated using scanning electron microscopy (SEM). Micrographs were acquired at multiple magnifications to evaluate particle shape, surface roughness, and coating continuity.
Representative SEM images of the initial and treated powders are shown in
Figure 4.
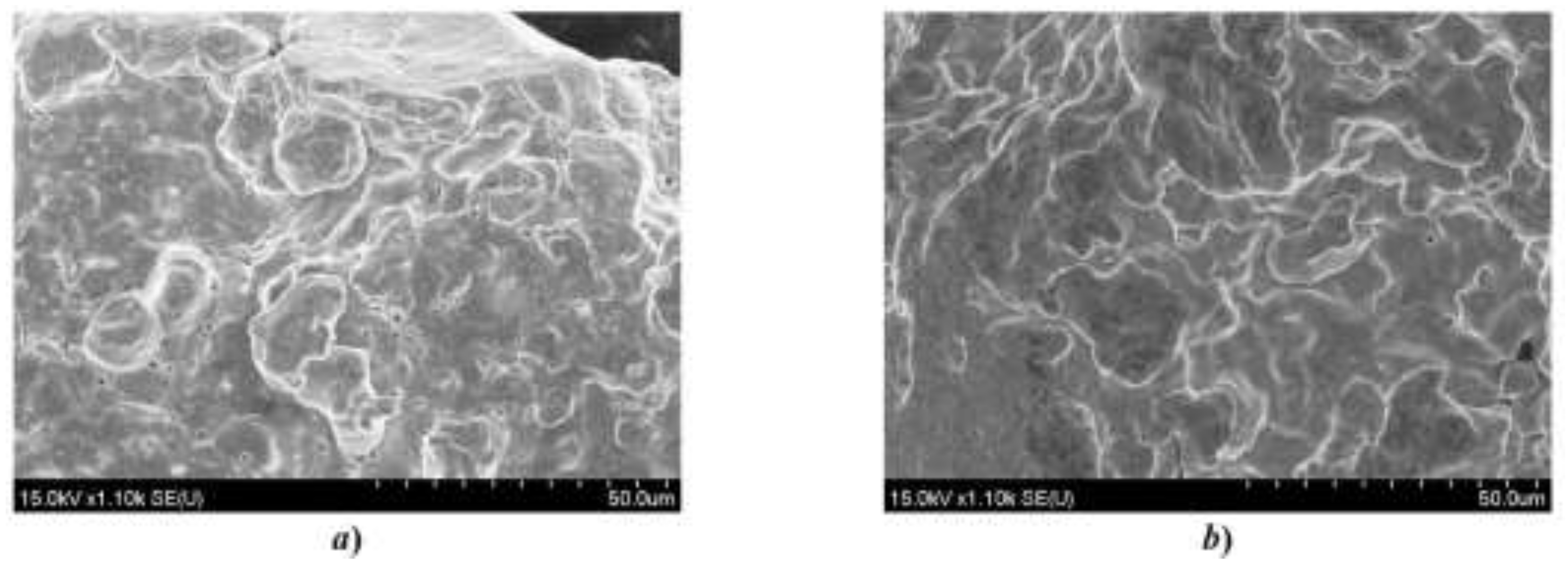
Figure 4.
SEM micrographs of iron powder particles: (a) initial powder and (b) gas-phase treated powder. Scale bar: 50 µm.
Figure 4.
SEM micrographs of iron powder particles: (a) initial powder and (b) gas-phase treated powder. Scale bar: 50 µm.
The initial powder exhibited irregular particle morphology with pronounced surface asperities typical of atomized iron. After gas-phase phosphating, morphological modification was observed, including smoother surface features and indications of conformal surface layer formation.
Qualitative comparison of micrographs was used to assess coating uniformity and detect possible microstructural defects such as cracking, delamination, or localized oxidation.
2.4.3. Energy-Dispersive X-ray Spectroscopy (EDS)
Elemental composition and spatial distribution of chemical elements were determined using energy-dispersive X-ray spectroscopy (EDS) integrated with SEM analysis. Both point analysis and elemental mapping were performed to evaluate phosphorus incorporation and oxygen variation after gas-phase treatment.
The chemical composition of the initial powder is summarized in
Table 1, while the composition of the treated powder is presented in
Table 2.
Elemental mapping was used to verify coating homogeneity across particle surfaces. Uniform phosphorus distribution without localized enrichment was considered evidence of conformal layer formation. Comparative analysis between initial and treated powders enabled evaluation of chemical modification efficiency.
2.4.4. Data Reliability and Comparative Assessment
All structural and compositional measurements were performed on multiple randomly selected powder regions to ensure statistical representativeness. Comparative evaluation between untreated and treated samples allowed establishment of process–structure correlations.
The integration of XRD, SEM, and EDS provided complementary information regarding crystallographic integrity, surface morphology evolution, and elemental redistribution, forming the basis for the subsequent discussion of coating formation mechanisms.
2.5. Coating Thickness Evaluation and Statistical Analysis
To quantitatively assess the effectiveness of the gas-phase phosphating process, coating thickness and surface coverage were evaluated using cross-sectional SEM observations and image analysis techniques.
Representative cross-sectional micrographs were obtained by embedding treated powder particles in conductive resin followed by mechanical polishing. The thickness of the phosphate-based surface layer was measured at multiple randomly selected locations on at least 20 individual particles to ensure statistical reliability.
The average coating thickness (
) was calculated as:
where
represents the local coating thickness measured at a given location and
n is the total number of measurements.
Standard deviation values were determined to evaluate coating uniformity and reproducibility of the deposition process. A low deviation was considered indicative of homogeneous vapor-phase deposition.
Surface coverage was qualitatively assessed by comparing treated and untreated particle surfaces using high-magnification SEM imaging (see
Figure 4b). The absence of exposed metallic regions under optimized processing conditions confirmed conformal layer formation.
In addition to thickness evaluation, elemental mapping results were analyzed to verify spatial homogeneity of phosphorus distribution. Regions exhibiting consistent phosphorus intensity across the particle surface were interpreted as uniform coating zones, whereas localized intensity peaks were considered indicators of possible inhomogeneous nucleation.
All measurements were performed in triplicate batches to ensure repeatability of the gas-phase process. The combined structural, morphological, and statistical analyses provide a quantitative basis for correlating processing parameters with coating formation efficiency.