Submitted:
04 March 2026
Posted:
06 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Fundamentals of Sodium-Ion Batteries
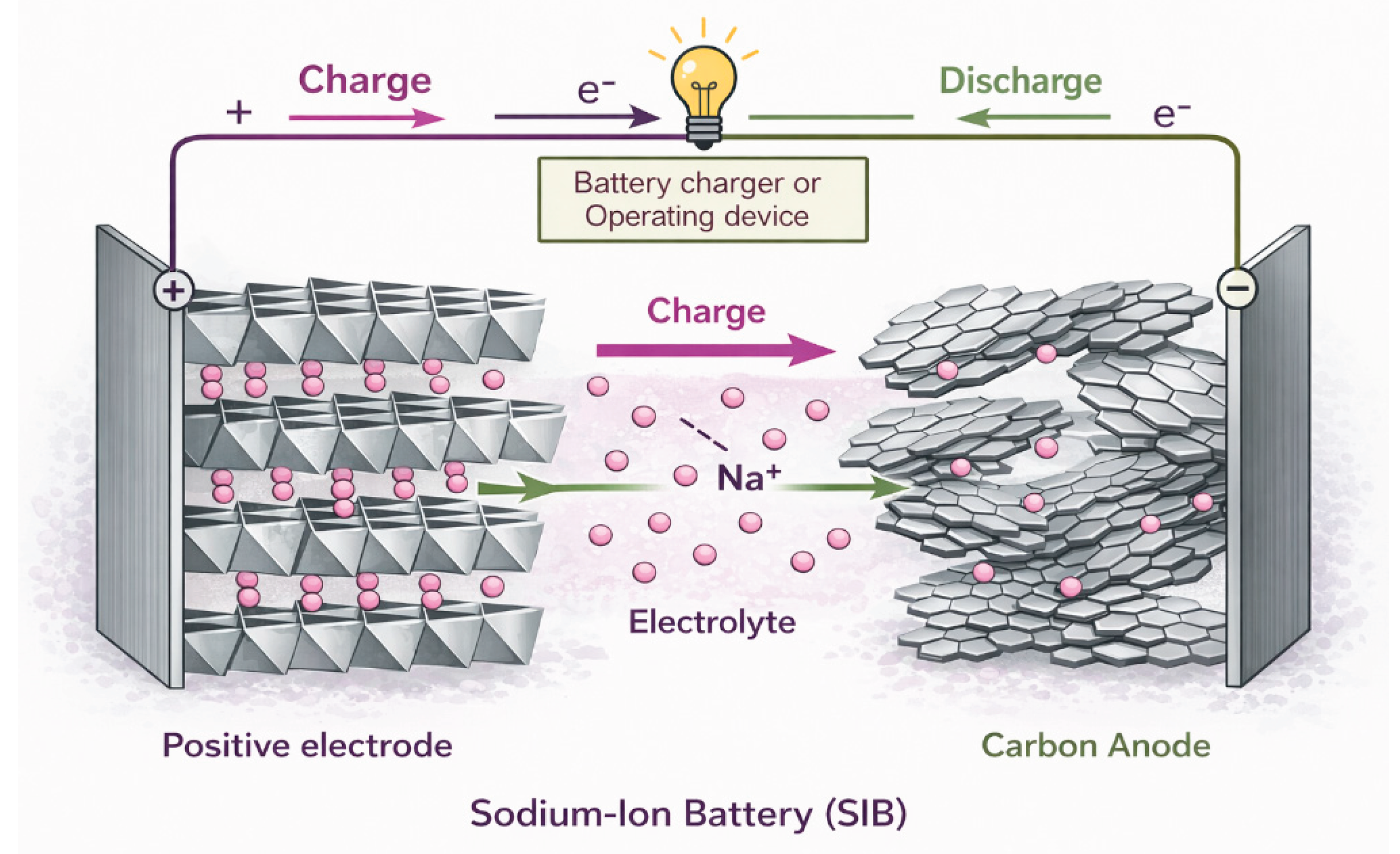
2.1. Electrochemical operating principle
2.2. Comparison with lithium-ion batteries
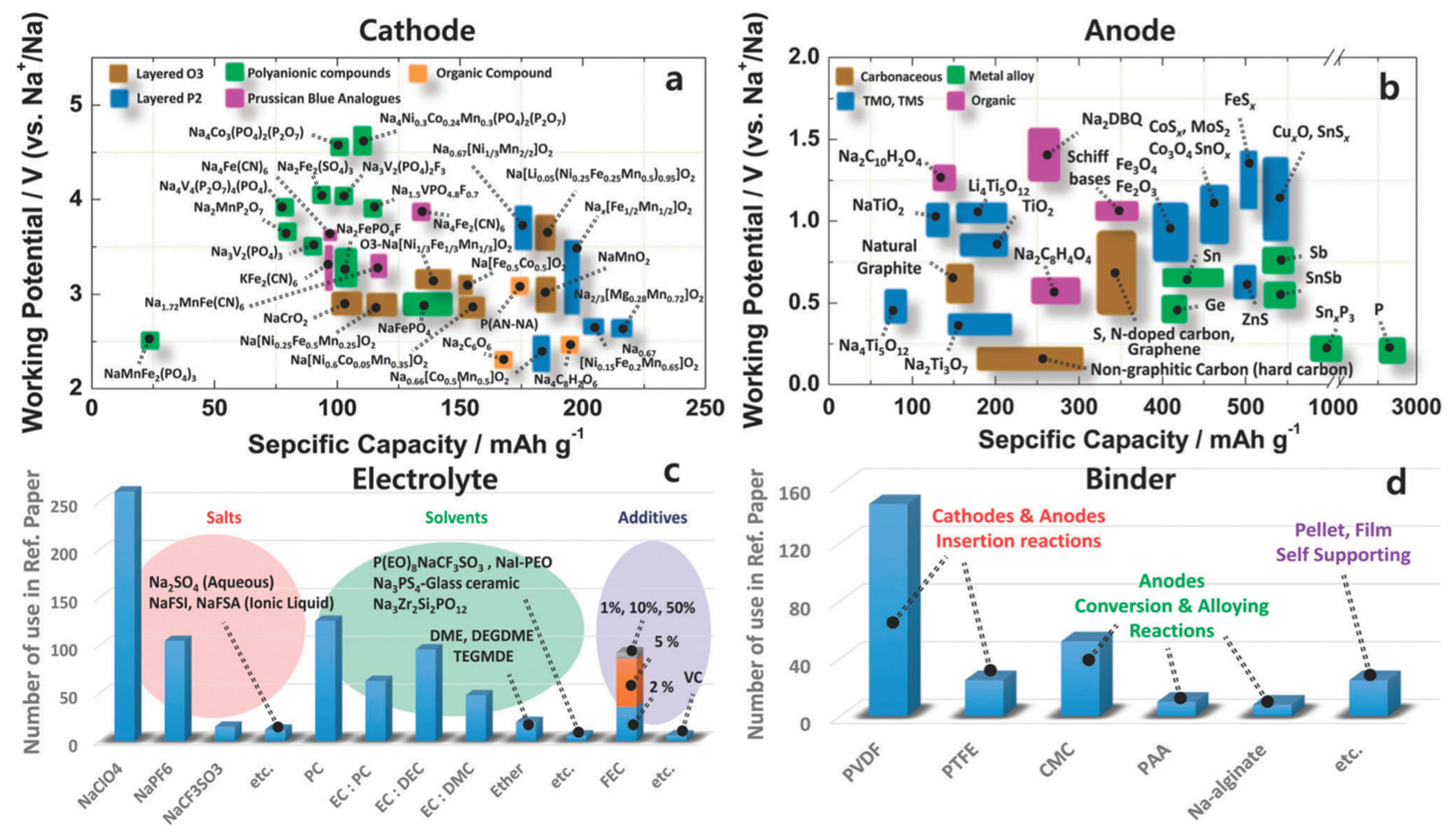
3. Anode Materials
3.1. Carbon-based anodes
3.2. Alloy-type anodes (Sn, Sb, Bi, P)
3.3. Conversion-Type Anodes
3.4. Electrolyte/Interphase Considerations for Anodes (SEI)
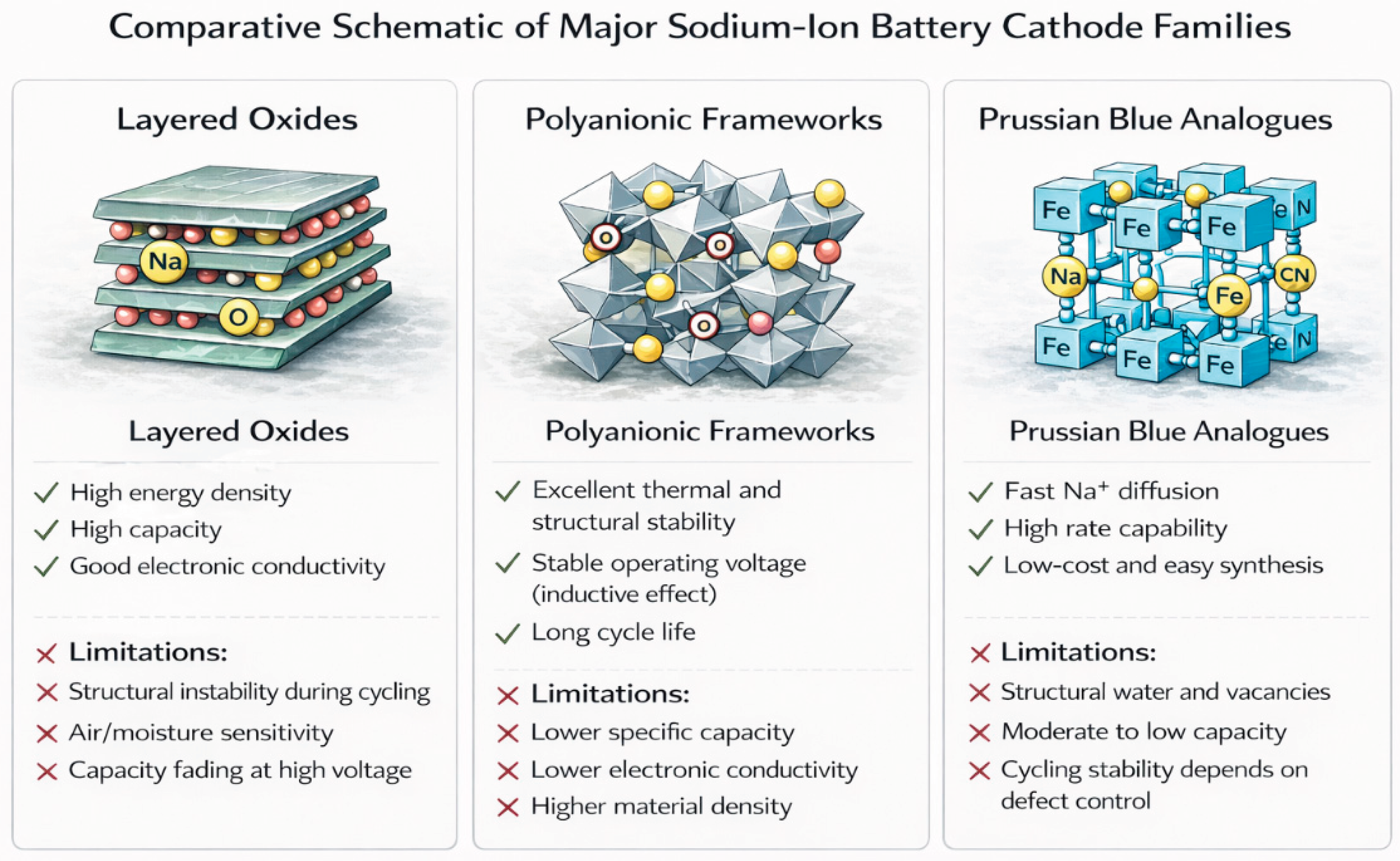
4. Cathode Materials
4.1. Layered transition-metal oxides (NaMO2)
4.2. Polyanionic compounds, including sulfate frameworks
4.3. Prussian blue analogues (PBAs)
4.4. Iron-based sulfate cathodes
4.5. Organic/polymer redox cathodes
5. Electrolytes and Interphases
5.1. Liquid electrolytes: salts, solvents, and formulation principles
5.2. Electrolyte degradation, gas evolution, and lifetime limitations
5.3. High-voltage electrolyte screening and cathode compatibility
5.4. Concentrated electrolytes and transport–stability trade-offs
5.5. Solid and quasi-solid electrolytes
5.6. Interfacial phenomena (SEI/CEI) and cross-talk: a system-level view
5.7. Safety considerations
6. Advanced Cell Architectures and Performance Enhancements
6.1. Binder-free/self-standing electrodes: why architecture matters more at scale
6.2. Nanostructured current collectors and conductive matrices: distributing current and suppressing impedance growth
6.3. Pre-sodiation: compensating Na loss to unlock full-cell energy density
7. Challenges and Failure Mechanisms
7.1. Volume expansion in alloy-type anodes
7.2. Interfacial instability
7.3. Lower energy density compared to Li-ion
7.4. Scalability and cost
7.5. Long-term degradation
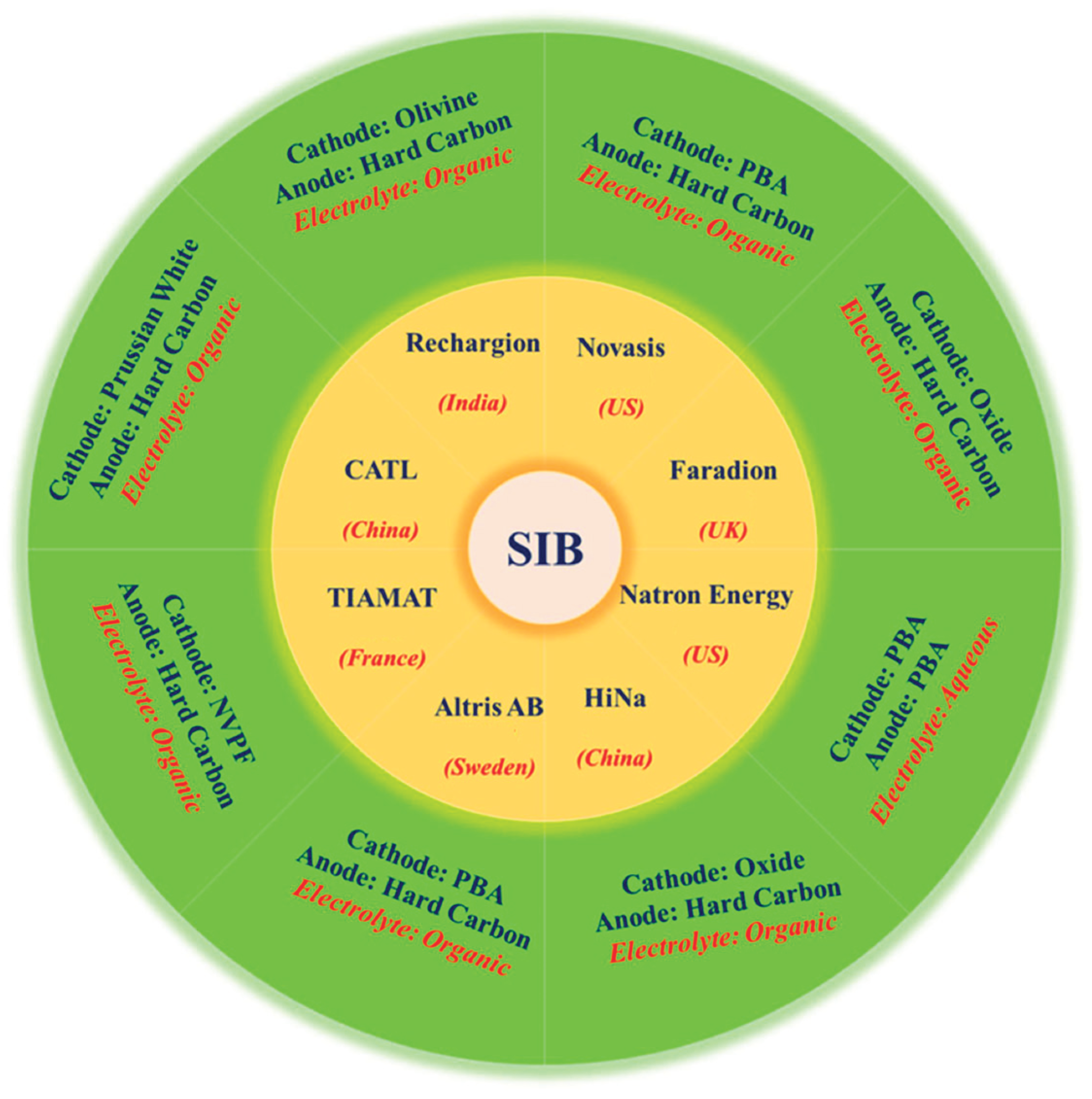
8. Industrial Progress and Commercialization
8.1. Key companies and industrial strategies
8.2. Prototypes and applications
- (i)
- Stationary storage and behind-the-meter systems
- (ii)
- Light mobility and short-range commercial vehicles
- (iii)
- Grid-scale systems and “bankability” constraints
8.3. Benchmarking vs. Li-ion and alternative batteries (Mg, K, Ca, Zn)
- (i)
- Benchmarking vs Li-ion (especially LFP for cost-driven markets)
- a)
- many Li-ion characterization/QA workflows transfer well to SIBs (helpful for ramp and yield learning), and
- b)
- early-generation commercial cells may be power-capable but still exhibit interfacial/electrolyte constraints that limit lifetime claims until formulations mature.
- (ii)
- Benchmarking vs post-Li alternatives (Mg, K, Ca, Zn)
8.4. Technology Readiness Levels (TRLs) and where sodium-ion sits today
- a)
- b)
- TRL 8–9 (qualified system / early commercial rollout): high-power niche products where duty cycles are favorable and performance claims can be validated (e.g., NVPF/HC power cells characterized in He 2023, [67] and large-scale manufacturer productization signals (CATL disclosures and production plans).
9. Future Perspectives
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALD | Atomic Layer Deposition |
| AM1.5G | Air Mass 1.5 Global (solar irradiance standard) |
| CDC | Carbide-Derived Carbon |
| CNT(s) | Carbon Nanotube(s) |
References
- Rudola, A.; Rennie, A.J.R.; Heap, R.; Meysami, S.S.; Lowbridge, A.; Mazzali, F.; Sayers, R.; Wright, C.J.; Barker, J. Commercialisation of High Energy Density Sodium-Ion Batteries: Faradion’s Journey and Outlook. J. Mater. Chem. A 2021, 9, 8279–8302. [Google Scholar] [CrossRef]
- Yabuuchi, N.; Kubota, K.; Dahbi, M.; Komaba, S. Research Development on Sodium-Ion Batteries. Chem. Rev. 2014, 114, 11636–11682. [Google Scholar] [CrossRef]
- Hwang, J.-Y.; Myung, S.-T.; Sun, Y.-K. Sodium-Ion Batteries: Present and Future. Chem. Soc. Rev. 2017, 46, 3529–3614. [Google Scholar] [CrossRef] [PubMed]
- Slater, M.D.; Kim, D.; Lee, E.; Johnson, C.S. Sodium-Ion Batteries. Adv Funct Materials 2013, 23, 947–958. [Google Scholar] [CrossRef]
- Palomares, V.; Casas-Cabanas, M.; Castillo-Martínez, E.; Han, M.H.; Rojo, T. Update on Na-Based Battery Materials. A Growing Research Path. Energy Environ. Sci. 2013, 6, 2312. [Google Scholar] [CrossRef]
- Pan, H.; Hu, Y.-S.; Chen, L. Room-Temperature Stationary Sodium-Ion Batteries for Large-Scale Electric Energy Storage. Energy Environ. Sci. 2013, 6, 2338. [Google Scholar] [CrossRef]
- Delmas, C. Sodium and Sodium-Ion Batteries: 50 Years of Research. Advanced Energy Materials 2018, 8, 1703137. [Google Scholar] [CrossRef]
- Chayambuka, K.; Mulder, G.; Danilov, D.L.; Notten, P.H.L. Sodium-Ion Battery Materials and Electrochemical Properties Reviewed. Advanced Energy Materials 2018, 8, 1800079. [Google Scholar] [CrossRef]
- Lee, M.J.; Shin, D.O.; Kim, J.Y.; Oh, J.; Kang, S.H.; Kim, J.; Kim, K.M.; Lee, Y.M.; Kim, S.O.; Lee, Y.-G. Interfacial Barrier Free Organic-Inorganic Hybrid Electrolytes for Solid State Batteries. Energy Storage Materials 2021, 37, 306–314. [Google Scholar] [CrossRef]
- Bača, P.; Libich, J.; Gazdošová, S.; Polkorab, J. Sodium-Ion Batteries: Applications and Properties. Batteries 2025, 11, 61. [Google Scholar] [CrossRef]
- Kundu, D.; Talaie, E.; Duffort, V.; Nazar, L.F. The Emerging Chemistry of Sodium Ion Batteries for Electrochemical Energy Storage. Angew Chem Int Ed 2015, 54, 3431–3448. [Google Scholar] [CrossRef] [PubMed]
- Nekahi, A.; Dorri, M.; Rezaei, M.; Bouguern, M.D.; Madikere Raghunatha Reddy, A.K.; Li, X.; Deng, S.; Zaghib, K. Comparative Issues of Metal-Ion Batteries toward Sustainable Energy Storage: Lithium vs. Sodium. Batteries 2024, 10, 279. [Google Scholar] [CrossRef]
- Nguyen, T.P.; Kim, I.T. Recent Advances in Sodium-Ion Batteries: Cathode Materials. Materials 2023, 16, 6869. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, Z.; Zheng, Y.; Luo, Y.; Jiang, X.; Wang, Y.; Wang, Z.; Wu, Y.; Zhang, Y.; Liu, X.; et al. Sodium-Ion Battery at Low Temperature: Challenges and Strategies. Nanomaterials 2024, 14, 1604. [Google Scholar] [CrossRef]
- Li, P.; Hu, N.; Wang, J.; Wang, S.; Deng, W. Recent Progress and Perspective: Na Ion Batteries Used at Low Temperatures. Nanomaterials 2022, 12, 3529. [Google Scholar] [CrossRef]
- Droese, D.; Luc, P.-M.; Otto, M.; Schlösser, A.; Evans, D.; Kowal, J. Evaluation of Commercial Sodium-Ion Batteries by State-of-the-Art Lithium-Ion Battery Configurations. Batteries 2025, 11, 420. [Google Scholar] [CrossRef]
- Carter, R.; Waller, G.H.; Jacob, C.; Hayman, D.; West, P.J.; Love, C.T. First Look at Safety and Performance Evaluation of Commercial Sodium-Ion Batteries. Energies 2025, 18, 661. [Google Scholar] [CrossRef]
- Bhutia, P.T.; Grugeon, S.; El Mejdoubi, A.; Laruelle, S.; Marlair, G. Safety Aspects of Sodium-Ion Batteries: Prospective Analysis from First Generation Towards More Advanced Systems. Batteries 2024, 10, 370. [Google Scholar] [CrossRef]
- Gulyas, P. Beyond Lithium: Evaluating Sodium-Ion Batteries for the Next Generation of Electric Vehicles. In Proceedings of the SMTS 2025; MDPI, November 7 2025; p. 41.
- Jia, Q.; Li, Z.; Ruan, H.; Luo, D.; Wang, J.; Ding, Z.; Chen, L. A Review of Carbon Anode Materials for Sodium-Ion Batteries: Key Materials, Sodium-Storage Mechanisms, Applications, and Large-Scale Design Principles. Molecules 2024, 29, 4331. [Google Scholar] [CrossRef]
- Iordache, M.; Oubraham, A.; Borta, S.; Ungureanu, G.; Marinoiu, A. Testing the Stability of NASICON Solid Electrolyte in Seawater Batteries. Energies 2024, 17, 5241. [Google Scholar] [CrossRef]
- Iordache, M.; Oubraham, A.; Petreanu, I.; Sisu, C.; Borta, S.; Capris, C.; Soare, A.; Marinoiu, A. NASICON Membrane with High Ionic Conductivity Synthesized by High-Temperature Solid-State Reaction. Materials 2024, 17, 823. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.; Yang, H.; Zhang, Z.; Xu, X.; Xu, Y.; Zhou, J.; Zhou, X.; Pan, Z.; Rao, X.; Gu, Y.; et al. The Progress of Hard Carbon as an Anode Material in Sodium-Ion Batteries. Molecules 2023, 28, 3134. [Google Scholar] [CrossRef]
- Ji, Y.; Li, J.; Li, J. Recent Development of Electrolyte Engineering for Sodium Metal Batteries. Batteries 2022, 8, 157. [Google Scholar] [CrossRef]
- Hashimov, M.; Hofmann, A. Deciphering Electrolyte Degradation in Sodium-Based Batteries: The Role of Conductive Salt Source, Additives, and Storage Condition. Batteries 2023, 9, 530. [Google Scholar] [CrossRef]
- Kulova, T.L.; Skundin, A.M. Electrode/Electrolyte Interphases of Sodium-Ion Batteries. Energies 2022, 15, 8615. [Google Scholar] [CrossRef]
- Ahangari, M.; Zhou, M.; Luo, H. Review of Layered Transition Metal Oxide Materials for Cathodes in Sodium-Ion Batteries. Micromachines 2025, 16, 137. [Google Scholar] [CrossRef]
- Drozhzhin, O.A.; Shevchenko, V.A.; Bobyleva, Z.V.; Alekseeva, A.M.; Antipov, E.V. Rational Screening of High-Voltage Electrolytes and Additives for Use in LiNi0.5Mn1.5O4-Based Li-Ion Batteries. Molecules 2022, 27, 3596. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhang, Z.; Jiang, X.; Zhao, Y.; Luo, Y.; Wang, Y.; Wang, Z.; Zhang, Y.; Liu, X.; Fang, B. A Comprehensive Review on Iron-Based Sulfate Cathodes for Sodium-Ion Batteries. Nanomaterials 2024, 14, 1915. [Google Scholar] [CrossRef]
- Kumar, S.; Raghupathy, R.; Vittadello, M. Sodium Polymer Electrolytes: A Review. Batteries 2024, 10, 73. [Google Scholar] [CrossRef]
- Song, W.; Li, Y.; Chen, J.; Min, H.; Wu, X.; Liu, X.; Yang, H. Open-Pore Skeleton Prussian Blue as a Cathode Material to Achieve High-Performance Sodium Storage. Materials 2025, 18, 3174. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-T.; Niu, B.-T.; Wang, Z.-H.; Li, Y.-X.; Xu, Y.-P.; Liu, S.-W.; Chen, Y.-X.; Lin, X.-M. High-Entropy and Component Stoichiometry Tuning Strategies Boost the Sodium-Ion Storage Performance of Cobalt-Free Prussian Blue Analogues Cathode Materials. Molecules 2024, 29, 4559. [Google Scholar] [CrossRef]
- Dong, X.; Wang, H.; Wang, J.; Wang, Q.; Wang, H.; Hao, W.; Lu, F. Preparation of Low-Defect Manganese-Based Prussian Blue Cathode Materials with Cubic Structure for Sodium-Ion Batteries via Coprecipitation Method. Molecules 2023, 28, 7267. [Google Scholar] [CrossRef]
- Li, C.; Xiao, Y.; Zhang, D.; Yuan, X.; Xiao, J.; Zhao, Y.; Gao, H.; Liu, H. Stable Manganese-Based High-Entropy Prussian Blue for Enhanced Sodium-Ion Storage. Batteries 2025, 11, 328. [Google Scholar] [CrossRef]
- Li, Z.; Zhong, S.; Zhou, B.; Chen, D.; Qiu, Z.; Zhang, R.; Zheng, R.; Zhao, C.; Zhou, J. Synthesis of Low-Defect Iron-Based Prussian Blue with Low Water Content for High-Stability Sodium-Ion Batteries. Materials 2025, 18, 1455. [Google Scholar] [CrossRef] [PubMed]
- Karahan Toprakci, H.A.; Toprakci, O. Recent Advances in New-Generation Electrolytes for Sodium-Ion Batteries. Energies 2023, 16, 3169. [Google Scholar] [CrossRef]
- Senthilkumar, K.K.; Thiruvengadathan, R.; Raghava, R.B.T.S. Recent Advancements in Na Super Ionic Conductor-Incorporated Composite Polymer Electrolytes for Sodium-Ion Battery Application. Electrochem 2025, 6, 6. [Google Scholar] [CrossRef]
- Salgueiro, T.A.; Veloso, R.C.; Ventura, J.; Danzi, F.; Oliveira, J. Ionic Conductivity Analysis of NASICON Solid Electrolyte Coated with Polyvinyl-Based Polymers. Batteries 2024, 10, 157. [Google Scholar] [CrossRef]
- Shang, R.; Ma, Y.; Anduaga-Quiros, K.; Briseno, G.; Ning, Y.; Chang, H.-J.; Ozkan, M.; Ozkan, C.S. Powering the Future: Unveiling the Potential of Na, K, and Mg Solid-State Batteries. Nanomaterials 2025, 15, 859. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Jia, G.; Yang, H.; Wang, M. Recent Advances for SEI of Hard Carbon Anode in Sodium-Ion Batteries: A Mini Review. Front. Chem. 2022, 10, 986541. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, Q.; Weng, S.; Ding, F.; Qi, X.; Lu, J.; Li, Y.; Zhang, X.; Rong, X.; Lu, Y.; et al. Interfacial Engineering to Achieve an Energy Density of over 200 Wh Kg−1 in Sodium Batteries. Nat Energy 2022, 7, 511–519. [Google Scholar] [CrossRef]
- Liang, X.; Song, X.; Sun, H.H.; Kim, H.; Kim, M.-C.; Sun, Y.-K. High-Energy and Long-Life O3-Type Layered Cathode Material for Sodium-Ion Batteries. Nat Commun 2025, 16, 3505. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xiong, B.; Gao, S.; Li, J.; Li, D.; Deng, W.; Hou, H.; Zou, G.; Ji, X. Presodiation Technology: Progress, Strategy and Prospects of Sacrificial Cathode Additives in Sodium-Based Energy Storage Systems. Chem. Commun. 2025, 61, 5386–5394. [Google Scholar] [CrossRef]
- Niu, Y.; Guo, Y.; Yin, Y.; Zhang, S.; Wang, T.; Wang, P.; Xin, S.; Guo, Y. High-Efficiency Cathode Sodium Compensation for Sodium-Ion Batteries. Advanced Materials 2020, 32, 2001419. [Google Scholar] [CrossRef]
- Deng, J.; Luo, W.; Lu, X.; Yao, Q.; Wang, Z.; Liu, H.; Zhou, H.; Dou, S. High Energy Density Sodium-Ion Battery with Industrially Feasible and Air-Stable O3-Type Layered Oxide Cathode. Advanced Energy Materials 2018, 8, 1701610. [Google Scholar] [CrossRef]
- Xiao, Y.; Xiao, J.; Zhao, H.; Li, J.; Zhang, G.; Zhang, D.; Guo, X.; Gao, H.; Wang, Y.; Chen, J.; et al. Prussian Blue Analogues for Sodium-Ion Battery Cathodes: A Review of Mechanistic Insights, Current Challenges, and Future Pathways. Small 2024, 20, 2401957. [Google Scholar] [CrossRef]
- Luo, W.; Shen, F.; Bommier, C.; Zhu, H.; Ji, X.; Hu, L. Na-Ion Battery Anodes: Materials and Electrochemistry. Acc. Chem. Res. 2016, 49, 231–240. [Google Scholar] [CrossRef]
- Hou, H.; Jing, M.; Yang, Y.; Zhu, Y.; Fang, L.; Song, W.; Pan, C.; Yang, X.; Ji, X. Sodium/Lithium Storage Behavior of Antimony Hollow Nanospheres for Rechargeable Batteries. ACS Appl. Mater. Interfaces 2014, 6, 16189–16196. [Google Scholar] [CrossRef]
- Chen, Z.; Li, Y.; Wang, L.; Zhang, J.; Dang, R.; Yu, H.; Wang, B.; Xu, C.; Li, H.; Li, Y.; et al. Durable Alloy Anode for Na-Ion Batteries with High Volumetric Energy Density. Nat Energy 2026. [Google Scholar] [CrossRef]
- Lim, H.; Yu, S.; Chang, W.; Chung, K.Y.; Choi, W.; Kim, S. Boosting the Sodiation Kinetics of Sn Anode Using a Yolk–Shell Nanohybrid Structure for High-Rate and Ultrastable Sodium-Ion Batteries. Advanced Science 2024, 11, 2408450. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sun, J.; Zhang, S.; Zhang, H.; Wang, P.; Guo, J.; Geng, S. Research Progress on Phosphorus/Carbon Anode Materials for Sodium-Ion Batteries. ACS Omega 2025, 10, 32511–32531. [Google Scholar] [CrossRef] [PubMed]
- Dahbi, M.; Nakano, T.; Yabuuchi, N.; Fujimura, S.; Chihara, K.; Kubota, K.; Son, J.; Cui, Y.; Oji, H.; Komaba, S. Effect of Hexafluorophosphate and Fluoroethylene Carbonate on Electrochemical Performance and the Surface Layer of Hard Carbon for Sodium-Ion Batteries. ChemElectroChem 2016, 3, 1856–1867. [Google Scholar] [CrossRef]
- Gogoi, P.; Ge, X.; Janpandit, M.; Prakash, S.; Kalita, N.; Li, Y.C. Investigations on Electrolyte Additives and Formation Mechanism of the Solid Electrolyte Interphase for Sodium Ion Batteries. ACS Appl. Mater. Interfaces 2025, 17, 13980–13987. [Google Scholar] [CrossRef]
- Li, Y.; Liu, G.; Che, J.; Chen, L.; Wang, X.; Wang, G.; Lei, L.; Hou, J.; Li, S.; Wang, J.; et al. Review on Layered Oxide Cathodes for Sodium-ion Batteries: Degradation Mechanisms, Modification Strategies, and Applications. Interdisciplinary Materials 2025, 4, 24–51. [Google Scholar] [CrossRef]
- Kuang, Y.; Wu, Y.; Zhang, H.; Sun, H. Interface Issues of Layered Transition Metal Oxide Cathodes for Sodium-Ion Batteries: Current Status, Recent Advances, Strategies, and Prospects. Molecules 2024, 29, 5988. [Google Scholar] [CrossRef]
- Yao, A.; Benson, S.M.; Chueh, W.C. Critically Assessing Sodium-Ion Technology Roadmaps and Scenarios for Techno-Economic Competitiveness against Lithium-Ion Batteries. Nat Energy 2025, 10, 404–416. [Google Scholar] [CrossRef]
- Kühn, J.; Schmidt, F.; Seete, P.; Boenke, T.; Hoffmann, F.S.; Dupuy, A.; Schumm, B.; Abendroth, T.; Althues, H.; Kaskel, S. Towards Scalable Production of Sodium-Ion Batteries: Solvent-Free Layered-Oxide Cathodes and Aqueous-Processed Hard Carbon Anodes for Cost-Effective Full-Cell Manufacturing. Batteries & Supercaps 2025, 8, e202400572. [Google Scholar] [CrossRef]
- Keiner, D.; Jasper, F.; Bogdanov, D.; Lopez, G.; Peters, J.; Baumann, M.; Breyer, C.; Weil, M. Sodium-Ion Battery Cost Projections and Their Impact on the Global Energy System Transition until 2050. Journal of Energy Storage 2026, 146, 119861. [Google Scholar] [CrossRef]
- Li, X.; Chen, X.; Li, M.; Wei, H.; Yang, X.; Ye, S.; Li, L.; Chen, J.; Ren, X.; Ouyang, X.; et al. Mechanisms and Mitigation Strategies of Gas Generation in Sodium-Ion Batteries. Nano-Micro Lett. 2025, 17, 177. [Google Scholar] [CrossRef]
- Xu, B.; Li, J.; Xie, Y.; Gui, Q.; Wu, Q.; Liu, W.; Mao, L. Capacity Degradation Study of NaNi1/3Fe1/3Mn1/3O2 Cathode Sodium-Ion Batteries Induced by Overcharge. Journal of Power Sources 2024, 623, 235448. [Google Scholar] [CrossRef]
- Chang, X.; Chen, J.; Xu, W.; Li, M.; Zhou, Q.; Dai, B.; Zhou, H.; Liu, Y.; Qiao, Y. Designing Robust Electrolytes for Extreme Environments: Overcoming Voltage and Thermal Constraints in Sodium-Ion Batteries. Energy Storage Materials 2025, 81, 104526. [Google Scholar] [CrossRef]
- Deng, W.; Du, X.; Xu, G.; Wang, S.; Du, L.; Dong, T.; Wu, R.; Li, C.; Lv, Z.; Ju, J.; et al. Constructing Dissolution–Resistant Interphases for Long-Life Sodium-Ion Batteries at Elevated Temperatures. Advanced Science 2025, 12, 2502860. [Google Scholar] [CrossRef]
- Yadav, P.; Shelke, V.; Patrike, A.; Shelke, M. Sodium-Based Batteries: Development, Commercialization Journey and New Emerging Chemistries. Oxford Open Materials Science 2023, 3, itac019. [Google Scholar] [CrossRef]
- Hu, Y.; Komaba, S.; Forsyth, M.; Johnson, C.; Rojo, T. A New Emerging Technology: Na-Ion Batteries. Small Methods 2019, 3, 1900184. [Google Scholar] [CrossRef]
- Chayambuka, K.; Mulder, G.; Danilov, D.L.; Notten, P.H.L. From Li-Ion Batteries toward Na-Ion Chemistries: Challenges and Opportunities. Advanced Energy Materials 2020, 10, 2001310. [Google Scholar] [CrossRef]
- CATL Unveils Its Latest Breakthrough Technology by Releasing Its First Generation of Sodium-Ion Batteries 2026.
- He, M.; Mejdoubi, A.El.; Chartouni, D.; Morcrette, M.; Troendle, P.; Castiglioni, R. High Power NVPF/HC-Based Sodium-Ion Batteries. Journal of Power Sources 2023, 588, 233741. [Google Scholar] [CrossRef]
- Sodium Ion Batteries. A Technology Brief 2026.
- Laufen, H.; Klick, S.; Ditler, H.; Quade, K.L.; Mikitisin, A.; Blömeke, A.; Schütte, M.; Wasylowski, D.; Sonnet, M.; Henrich, L.; et al. Multi-Method Characterization of a Commercial 1.2 Ah Sodium-Ion Battery Cell Indicates Drop-in Potential. Cell Reports Physical Science 2024, 5, 101945. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, R.; Wang, C.; Mao, J.; Chao, D.; Zhang, C.; Zhang, S.; Guo, Z. Zinc Ion Batteries: Bridging the Gap from Academia to Industry for Grid-Scale Energy Storage. Angew Chem Int Ed 2024, 63, e202400045. [Google Scholar] [CrossRef]
- Jian, R.; Mashhadian, A.; Wang, C.; Hwang, T.; Hao, Y.; Cho, K.; Xiong, G. Reassessing the Commercialization of Aqueous Zinc-Ion Batteries Based on Laboratory Achievements. ACS Appl. Energy Mater. 2025, 8, 12460–12479. [Google Scholar] [CrossRef]
- Li, M.; Wang, C.; Wang, C.; Lyu, Y.; Wang, J.; Xia, S.; Mao, J.; Guo, Z. 10 Years Development of Potassium-Ion Batteries. Advanced Materials 2025, 37, 2416717. [Google Scholar] [CrossRef]
- Herodotou, P.; Hao, R.; E․ Georghiou, G.; Yagi, S. Rechargeable Magnesium Batteries: System-Level Opportunities and Challenges for Battery Energy Storage Applications. Electrochimica Acta 2026, 555, 148315. [Google Scholar] [CrossRef]
- Taghavi-Kahagh, A.; Roghani-Mamaqani, H.; Salami-Kalajahi, M. Powering the Future: A Comprehensive Review on Calcium-Ion Batteries. Journal of Energy Chemistry 2024, 90, 77–97. [Google Scholar] [CrossRef]
- Lin, H.; Meng, J.; Guo, W.; Li, R.; Yi, Y.; Ma, Y.; Cheung, C.F.; Aurbach, D.; Xu, Z.-L. Deciphering the Dynamic Interfacial Chemistry of Calcium Metal Anodes. Energy Environ. Sci. 2024, 17, 6548–6558. [Google Scholar] [CrossRef]
- Cui, Z.; Liu, C.; Manthiram, A. A Perspective on Pathways Toward Commercial Sodium-Ion Batteries. Advanced Materials 2025, 37, 2420463. [Google Scholar] [CrossRef] [PubMed]
- Thackeray, M.M.; Wolverton, C.; Isaacs, E.D. Electrical Energy Storage for Transportation—Approaching the Limits of, and Going beyond, Lithium-Ion Batteries. Energy Environ. Sci. 2012, 5, 7854. [Google Scholar] [CrossRef]
- Dunn, B.; Kamath, H.; Tarascon, J.-M. Electrical Energy Storage for the Grid: A Battery of Choices. Science 2011, 334, 928–935. [Google Scholar] [CrossRef]
- Schütte, M.; Laufen, H.; Luder, D.; Ditler, H.; Kern, J.; Klick, S.; Junker, M.; Stahl, G.; Frie, F.; Sauer, D.U. First Full Cell Parameterization of a Commercial Layered Oxide/Hard Carbon Sodium-Ion 18650 Battery Cell for a Physico-Chemical Model. Journal of Energy Storage 2025, 107, 114931. [Google Scholar] [CrossRef]
- Zhao, C.; Andersen, P.B.; Træholt, C.; Hashemi, S. Grid-Connected Battery Energy Storage System: A Review on Application and Integration. Renewable and Sustainable Energy Reviews 2023, 182, 113400. [Google Scholar] [CrossRef]
- Vaalma, C.; Buchholz, D.; Weil, M.; Passerini, S. A Cost and Resource Analysis of Sodium-Ion Batteries. Nat Rev Mater 2018, 3, 18013. [Google Scholar] [CrossRef]
- Tarascon, J.-M. Na-Ion versus Li-Ion Batteries: Complementarity Rather than Competitiveness. Joule 2020, 4, 1616–1620. [Google Scholar] [CrossRef]
- Nozarijouybari, Z.; Fathy, H.K. Machine Learning for Battery Systems Applications: Progress, Challenges, and Opportunities. Journal of Power Sources 2024, 601, 234272. [Google Scholar] [CrossRef]
- Wang, Z.; Zeier, W.G.; You, F. Toward AI Ecosystems for Electrolyte and Interface Engineering in Solid-State Batteries. Sci. Adv. 2025, 11, eaea0638. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.; Buchholz, D.; Passerini, S.; Weil, M. Life Cycle Assessment of Sodium-Ion Batteries. Energy Environ. Sci. 2016, 9, 1744–1751. [Google Scholar] [CrossRef]
- Peters, J.F.; Baumann, M.; Binder, J.R.; Weil, M. On the Environmental Competitiveness of Sodium-Ion Batteries under a Full Life Cycle Perspective – a Cell-Chemistry Specific Modelling Approach. Sustainable Energy Fuels 2021, 5, 6414–6429. [Google Scholar] [CrossRef]
- Wickerts, S.; Arvidsson, R.; Nordelöf, A.; Svanström, M.; Johansson, P. Prospective Life Cycle Assessment of Sodium-ion Batteries Made from Abundant Elements. J of Industrial Ecology 2024, 28, 116–129. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Y.; Kang, Y.; Du, H.; Jia, T.; Xu, J.; Huang, Y.; Zhao, Y.; Kang, F.; Li, B. Cathode Recycling of Spent Sodium Ion Batteries. Energy Mater Adv 2024, 5, 0128. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, Y.; Chen, C.; Lin, Z.; Zhang, S.; Lu, J. Sustainability-Inspired Cell Design for a Fully Recyclable Sodium Ion Battery. Nat Commun 2019, 10, 1965. [Google Scholar] [CrossRef]
| Anode Family | Dominant storage mechanisms | Key practical bottlenecks / design levers | Reference |
|---|---|---|---|
| Hard carbon | Mixed: defect/surface storage (sloping) + low-voltage plateau (nanopore filling), SEI-dependent | Low ICE (Na consumption in SEI); microstructure/porosity control; densification vs wetting/transport trade-off; cold-temperature polarization | [14,15,20,23] |
| Graphitic/soft carbon | Limited Na intercalation in carbonate electrolytes; improved storage in some ether systems via co-intercalation | Poor capacity with standard electrolytes; requires specialized formulations; interphase control | [3,14,15] |
| Alloy-type (Sn, Sb, Bi, P) | Na alloying/dealloying with multiple NaxM phases | Large volume change; particle fracture; repeated SEI rupture; mitigation via nanoconfinement/void engineering/elastic matrices/SEI additives | [5,8,25,26] |
| Conversion-type | Multielectron conversion reactions with extensive interfacial area evolution during cycling | High hysteresis; continuous surface renewal; SEI instability; partial mitigation via confinement/coatings/electrolyte engineering | [5,8,25,26] |
| Family | Strengths | Main limitations | Common mitigations | Reference |
|---|---|---|---|---|
| Layered oxides | High energy-density potential, broad compositional tunability | Phase transitions, surface and CEI instability at higher voltages, sensitivity to processing and handling | Doping, particle engineering, surface coatings, high-voltage electrolyte screening | [27,28] |
| Polyanionic cathodes, including sulfates | Robust frameworks, generally favorable safety, good structural stability | Lower gravimetric energy density, limited electronic conductivity, polarization at high loading | Particle control, conductive coatings and networks, electrode architecture optimization | [2,29] |
| PBAs | Fast Na⁺ kinetics, scalable synthesis routes, strong rate capability | Defects and vacancies, structural water management, reproducibility constraints | Low-defect synthesis, water control, stoichiometry tuning, surface and interphase stabilization | [31,32,33,34,35] |
| Organic cathodes | Sustainability; flexible molecular design | Dissolution; hysteresis; low conductivity | Polymerization/anchoring; electrolyte optimization; conductive scaffolds | [29] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




