Submitted:
04 March 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Protocol and Registration
2.2. Eligibility Criteria, Information Sources, Search Strategy
2.3. Data Extraction and Assessment of Bias
2.4. Data Synthesis and Analysis
3. Results
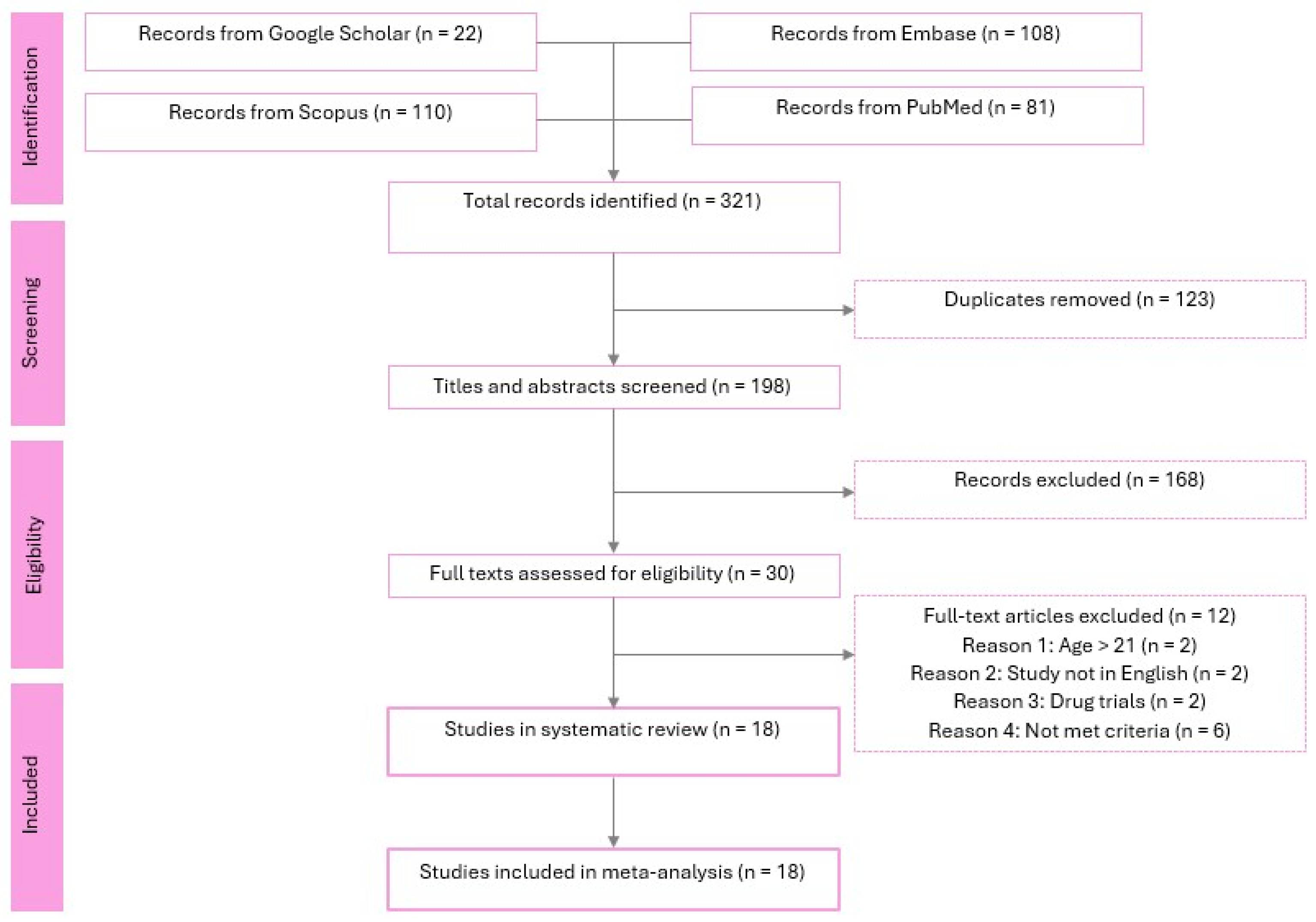
3.1. Study Selection
3.2. Study Characteristics
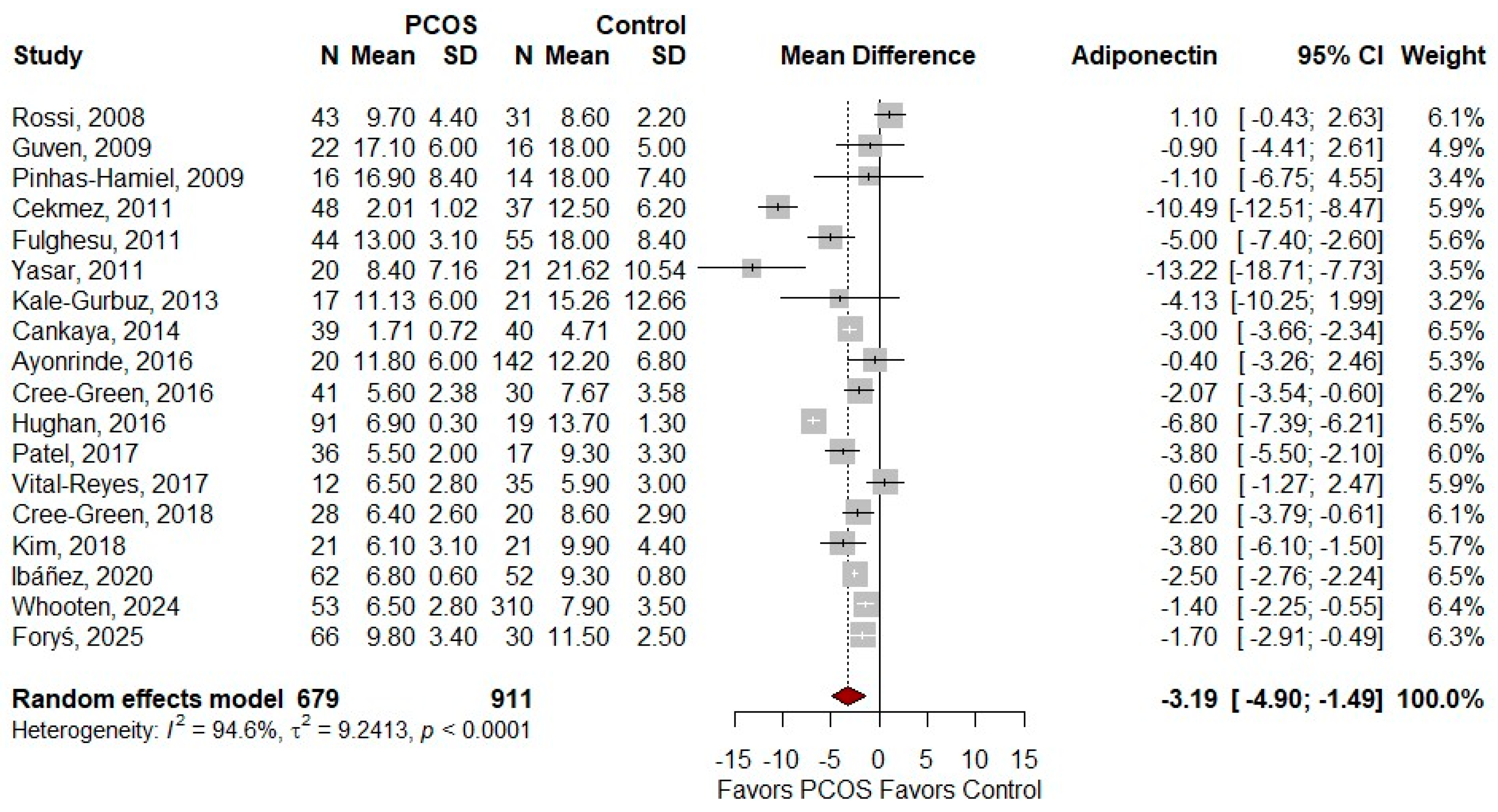
3.3. Meta-Analysis
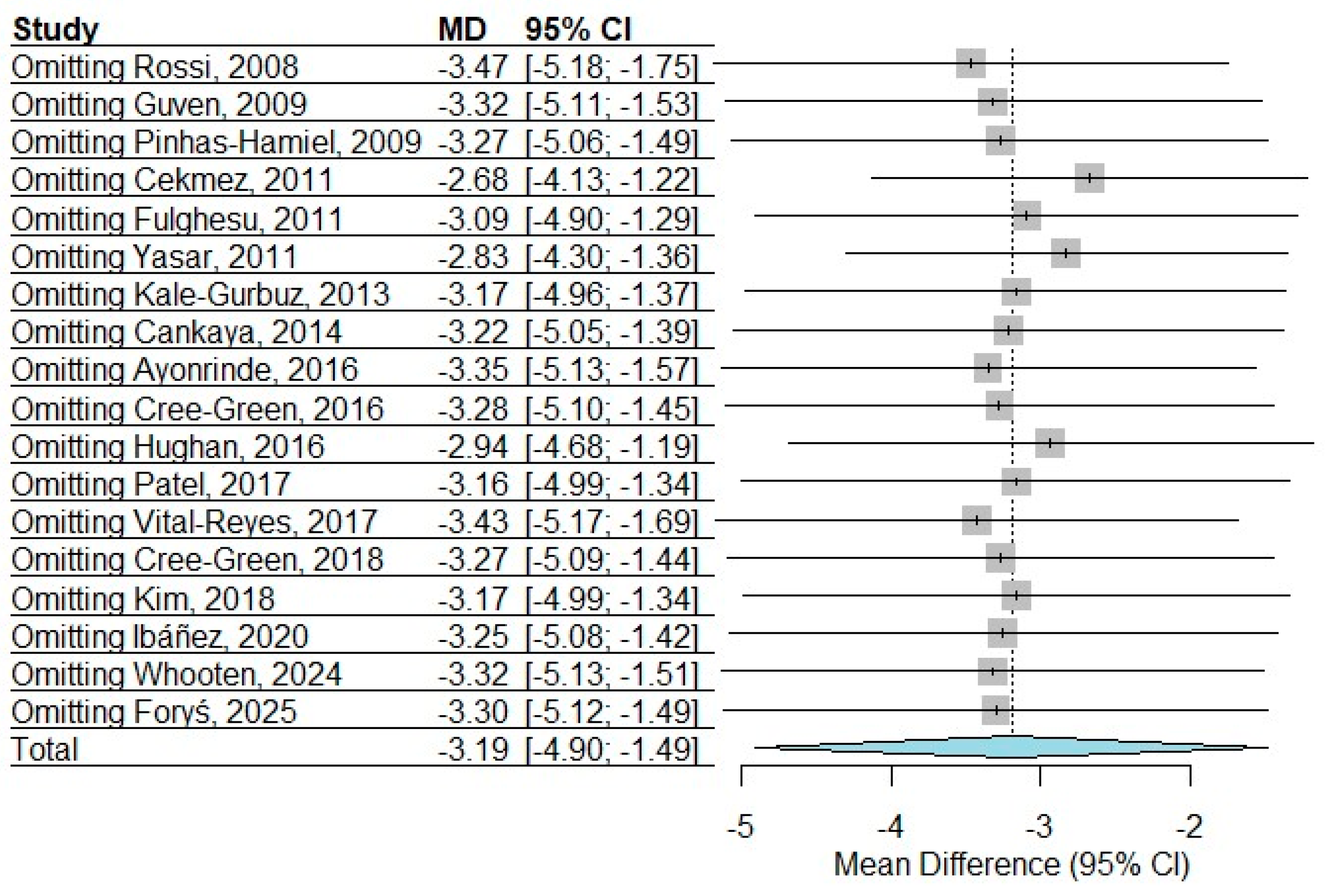
3.4. Sensitivity Analysis
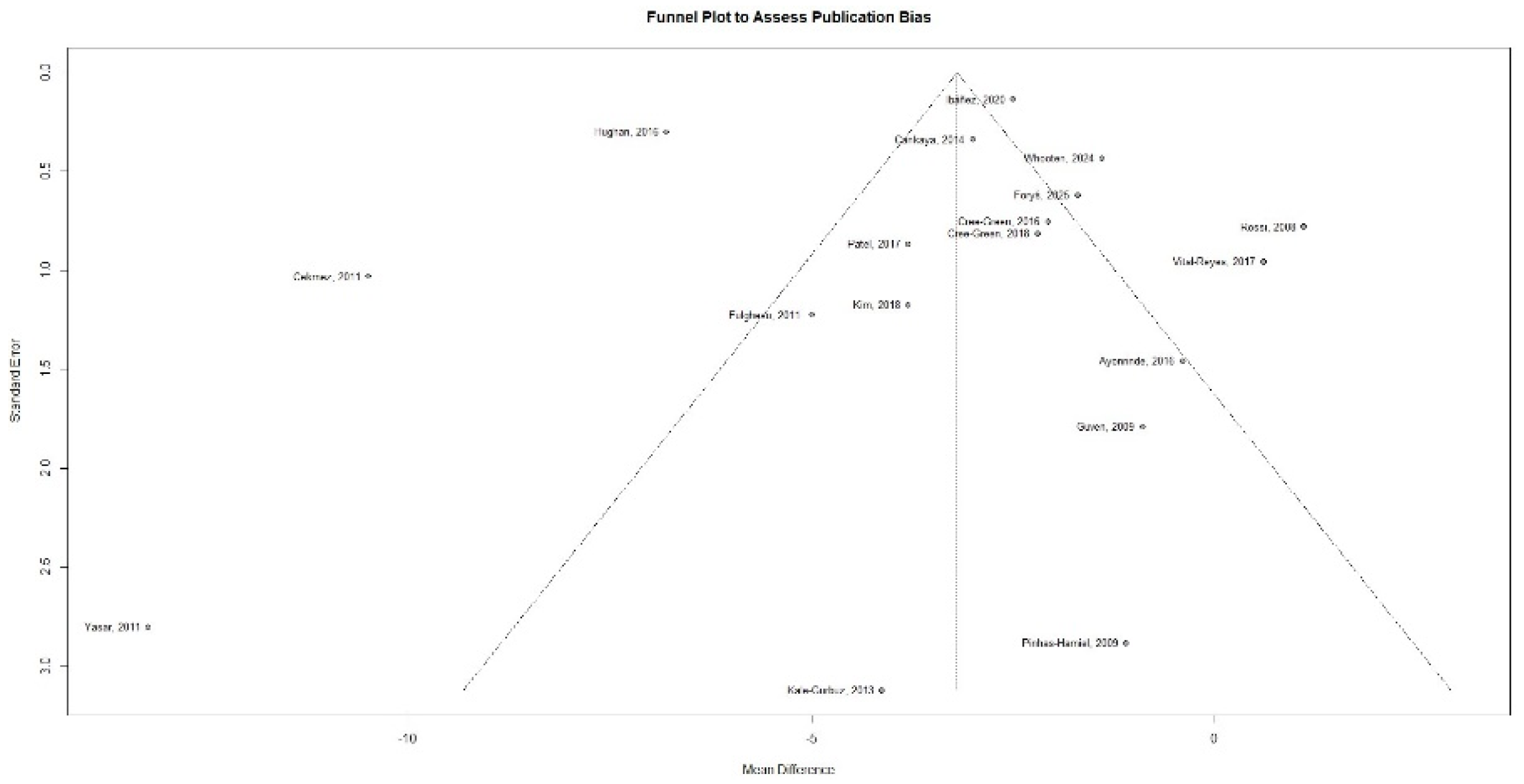
3.5. Publication Bias
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PCOS | Polycystic Ovary Syndrome |
| NOS | Newcastle-Ottawa Scale |
| MD | Mean Difference |
| CI | Confidence Interval |
| N | Sample Size |
| PRISMA | Preferred Reporting Items for Systematic Review and Meta-Analyses |
| NIH | National Institute of Health |
| ESCPG | Endocrine Society Clinical Practice Guideline |
| IEG | International Evidence-based Guideline |
References
- Escobar-Morreale, H.F. Polycystic Ovary Syndrome: Definition, Aetiology, Diagnosis and Treatment. Nat Rev Endocrinol 2018, 14, 270–284. [CrossRef]
- Salari, N.; Nankali, A.; Ghanbari, A.; Jafarpour, S.; Ghasemi, H.; Dokaneheifard, S.; Mohammadi, M. Global Prevalence of Polycystic Ovary Syndrome in Women Worldwide: A Comprehensive Systematic Review and Meta-Analysis. Arch Gynecol Obstet 2024, 310, 1303–1314. [CrossRef]
- Livadas, S.; Anagnostis, P.; Bosdou, J.K.; Bantouna, D.; Paparodis, R. Polycystic Ovary Syndrome and Type 2 Diabetes Mellitus: A State-of-the-Art Review. WJD 2022, 13, 5–26. [CrossRef]
- Gao, L.; Zhao, Y.; Wu, H.; Lin, X.; Guo, F.; Li, J.; Long, Y.; Zhou, B.; She, J.; Zhang, C.; et al. Polycystic Ovary Syndrome Fuels Cardiovascular Inflammation and Aggravates Ischemic Cardiac Injury. Circulation 2023, 148, 1958–1973. [CrossRef]
- Guo, F.; Gong, Z.; Fernando, T.; Zhang, L.; Zhu, X.; Shi, Y. The Lipid Profiles in Different Characteristics of Women with PCOS and the Interaction Between Dyslipidemia and Metabolic Disorder States: A Retrospective Study in Chinese Population. Front. Endocrinol. 2022, 13, 892125. [CrossRef]
- van Andel, M.; Heijboer, A.C.; Drent, M.L. Chapter Five - Adiponectin and Its Isoforms in Pathophysiology. In Advances in Clinical Chemistry; Makowski, G.S., Ed.; Elsevier, 2018; Vol. 85, pp. 115–147 ISBN 0065-2423.
- Da Silva Rosa, S.C.; Liu, M.; Sweeney, G. Adiponectin Synthesis, Secretion and Extravasation from Circulation to Interstitial Space. Physiology 2021, 36, 134–149. [CrossRef]
- Atanasova Boshku, A. Adiponectin as a Serum Marker of Adipose Tissue Dysfunction in Women with Polycystic Ovary Syndrome: Correlation with Indicators of Metabolic Disturbances. Acta Endo (Buc) 2018, 14, 346–352. [CrossRef]
- Li, S.; Huang, X.; Zhong, H.; Peng, Q.; Chen, S.; Xie, Y.; Qin, X.; Qin, A. Low Circulating Adiponectin Levels in Women with Polycystic Ovary Syndrome: An Updated Meta-Analysis. Tumor Biol. 2014, 35, 3961–3973. [CrossRef]
- Lin, K.; Sun, X.; Wang, X.; Wang, H.; Chen, X. Circulating Adipokine Levels in Nonobese Women With Polycystic Ovary Syndrome and in Nonobese Control Women: A Systematic Review and Meta-Analysis. Front. Endocrinol. 2021, 11, 537809. [CrossRef]
- Guan, Y.; Zuo, F.; Zhao, J.; Nian, X.; Shi, L.; Xu, Y.; Huang, J.; Kazumi, T.; Wu, B. Relationships of Adiponectin to Regional Adiposity, Insulin Sensitivity, Serum Lipids, and Inflammatory Markers in Sedentary and Endurance-Trained Japanese Young Women. Front Endocrinol (Lausanne) 2023, 14, 1097034. [CrossRef]
- Orszulak, D.; Niziński, K.; Matonóg, A.; Zięba-Domalik, M.; Stojko, R.; Drosdzol-Cop, A. Adipokines as Biochemical Marker of Polycystic Ovary Syndrome in Adolescents – Review. Front. Endocrinol. 2025, 16, 1475465. [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [CrossRef]
- Revised 2003 Consensus on Diagnostic Criteria and Long-Term Health Risks Related to Polycystic Ovary Syndrome. Fertil Steril 2004, 81, 19–25. [CrossRef]
- Zawadzki, J.; Dunaif, A.; Givens, J.; Haseltine, F.; Merriam, G. Current Issues in Endocrinology and Metabolism: Polycystic Ovary Syndrome. MA: Blackwell Scientific Publications Cambridge 1992, 10.
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the Sample Mean and Standard Deviation from the Sample Size, Median, Range and/or Interquartile Range. BMC Medical Research Methodology 2014, 14, 135. [CrossRef]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, je; Welch, V. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Case-Control Studies in Meta-Analyses. Eur J Epidemiol 2011, 25, 603–605.
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in Meta-Analysis Detected by a Simple, Graphical Test. BMJ 1997, 315, 629–634. [CrossRef]
- Begg, C.B.; Mazumdar, M. Operating Characteristics of a Rank Correlation Test for Publication Bias. Biometrics 1994, 50, 1088–1101.
- Rossi, B.; Sukalich, S.; Droz, J.; Griffin, A.; Cook, S.; Blumkin, A.; Guzick, D.S.; Hoeger, K.M. Prevalence of Metabolic Syndrome and Related Characteristics in Obese Adolescents with and without Polycystic Ovary Syndrome. The Journal of Clinical Endocrinology & Metabolism 2008, 93, 4780–4786. [CrossRef]
- Güven, A.; Özgen, T.; AliyazicioĞlu, Y. Adiponectin and Resistin Concentrations after Glucose Load in Adolescents with Polycystic Ovary Syndrome. Gynecological Endocrinology 2010, 26, 30–38. [CrossRef]
- Pinhas-Hamiel, O.; Singer, S.; Pilpel, N.; Koren, I.; Boyko, V.; Hemi, R.; Pariente, C.; Kanety, H. Adiponectin Levels in Adolescent Girls with Polycystic Ovary Syndrome (PCOS). Clinical Endocrinology 2009, 71, 823–827. [CrossRef]
- Cekmez, F.; Cekmez, Y.; Pirgon, Ö.; Emre Canpolat, F.; Aydinöz, S.; Metin Ipcioglu, O.; Karademir, F. Evaluation of New Adipocytokines and Insulin Resistance in Adolescents with Polycystic Ovary Syndrome. European Cytokine Network 2011, 22, 32–37. [CrossRef]
- Fulghesu, A.M.; Sanna, F.; Uda, S.; Magnini, R.; Portoghese, E.; Batetta, B. Il-6 Serum Levels and Production Is Related to an Altered Immune Response in Polycystic Ovary Syndrome Girls with Insulin Resistance. Mediators of Inflammation 2011, 2011, 1–8. [CrossRef]
- Yasar, L.; Ekin, M.; Gedikbasi, A.; Erturk, A.D.; Savan, K.; Ozdemir, A.; Temur, M. Serum Adiponectin Levels in High School Girls with Polycystic Ovary Syndrome and Hyperandrogenism. Journal of Pediatric and Adolescent Gynecology 2011, 24, 90–93. [CrossRef]
- Kale-Gurbuz, T.; Akhan, S.E.; Bastu, E.; Telci, A.; Iyibozkurt, A.C.; Topuz, S. Adiponectin, Leptin and Ghrelin Levels in Obese Adolescent Girls with Polycystic Ovary Syndrome. Journal of Pediatric and Adolescent Gynecology 2013, 26, 27–30. [CrossRef]
- Cankaya, S.; Demir, B.; Aksakal, S.E.; Dilbaz, B.; Demirtas, C.; Goktolga, U. Insulin Resistance and Its Relationship with High Molecular Weight Adiponectin in Adolescents with Polycystic Ovary Syndrome and a Maternal History of Polycystic Ovary Syndrome. Fertility and Sterility 2014, 102, 826–830. [CrossRef]
- Ayonrinde, O.T.; Adams, L.A.; Doherty, D.A.; Mori, T.A.; Beilin, L.J.; Oddy, W.H.; Hickey, M.; Sloboda, D.M.; Olynyk, J.K.; Hart, R. Adverse Metabolic Phenotype of Adolescent Girls with Non-alcoholic Fatty Liver Disease plus Polycystic Ovary Syndrome Compared with Other Girls and Boys. J of Gastro and Hepatol 2016, 31, 980–987. [CrossRef]
- Cree-Green, M.; Bergman, B.C.; Coe, G.V.; Newnes, L.; Baumgartner, A.D.; Bacon, S.; Sherzinger, A.; Pyle, L.; Nadeau, K.J. Hepatic Steatosis Is Common in Adolescents with Obesity and PCOS and Relates to De Novo Lipogenesis but Not Insulin Resistance. Obesity 2016, 24, 2399–2406. [CrossRef]
- Hughan, K.S.; Tfayli, H.; Warren-Ulanch, J.G.; Barinas-Mitchell, E.; Arslanian, S.A. Early Biomarkers of Subclinical Atherosclerosis in Obese Adolescent Girls with Polycystic Ovary Syndrome. The Journal of Pediatrics 2016, 168, 104-111.e1. [CrossRef]
- Patel, S.S.; Truong, U.; King, M.; Ferland, A.; Moreau, K.L.; Dorosz, J.; Hokanson, J.E.; Wang, H.; Kinney, G.L.; Maahs, D.M.; et al. Obese Adolescents with Polycystic Ovarian Syndrome Have Elevated Cardiovascular Disease Risk Markers. Vasc Med 2017, 22, 85–95. [CrossRef]
- Vital-Reyes, V.S.; Lopez-Alarcón, M.G.; Inda-Icaza, P.; Márquez-Maldonado, C. Alteraciones metabólicas sutiles en adolescentes con obesidad y síndrome de ovario poliquístico. GMM 2019, 153, 150. [CrossRef]
- Cree-Green, M.; Cai, N.; Thurston, J.E.; Coe, G.V.; Newnes, L.; Garcia-Reyes, Y.; Baumgartner, A.D.; Pyle, L.; Nadeau, K.J. Using Simple Clinical Measures to Predict Insulin Resistance or Hyperglycemia in Girls with Polycystic Ovarian Syndrome. Pediatr Diabetes 2018, 19, 1370–1378. [CrossRef]
- Kim, J.Y.; Tfayli, H.; Michaliszyn, S.F.; Arslanian, S. Impaired Lipolysis, Diminished Fat Oxidation, and Metabolic Inflexibility in Obese Girls With Polycystic Ovary Syndrome. The Journal of Clinical Endocrinology & Metabolism 2018, 103, 546–554. [CrossRef]
- Ibáñez, L.; Díaz, M.; García-Beltrán, C.; Malpique, R.; Garde, E.; López-Bermejo, A.; De Zegher, F. Toward a Treatment Normalizing Ovulation Rate in Adolescent Girls With Polycystic Ovary Syndrome. Journal of the Endocrine Society 2020, 4, bvaa032. [CrossRef]
- Whooten, R.C.; Rifas-Shiman, S.L.; Perng, W.; Chavarro, J.E.; Taveras, E.; Oken, E.; Hivert, M.-F. Associations of Childhood Adiposity and Cardiometabolic Biomarkers With Adolescent PCOS. Pediatrics 2024, 153, e2023064894. [CrossRef]
- Foryś, E.; Drosdzol-Cop, A.; Małecka-Tendera, E.; Gawlik-Starzyk, A.M.; Skrzyńska, K.; Olszanecka-Glinianowicz, M.; Zachurzok, A. Adipokine Profile Signature in Adolescent Girls with Menstrual Disorders and Hyperandrogenism Differs from That of Regularly Menstruating Girls. JCM 2025, 14, 7987. [CrossRef]
- Legro, R.S.; Arslanian, S.A.; Ehrmann, D.A.; Hoeger, K.M.; Murad, M.H.; Pasquali, R.; Welt, C.K. Diagnosis and Treatment of Polycystic Ovary Syndrome: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 2013, 98, 4565–4592. [CrossRef]
- Teede, H.J.; Tay, C.T.; Laven, J.J.E.; Dokras, A.; Moran, L.J.; Piltonen, T.T.; Costello, M.F.; Boivin, J.; Redman, L.M.; Boyle, J.A.; et al. Recommendations from the 2023 International Evidence-Based Guideline for the Assessment and Management of Polycystic Ovary Syndrome. Eur J Endocrinol 2023, 189, G43–G64. [CrossRef]
- Maliqueo, M.; Galgani, J.E.; Pérez-Bravo, F.; Echiburú, B.; De Guevara, A.L.; Crisosto, N.; Sir-Petermann, T. Relationship of Serum Adipocyte-Derived Proteins with Insulin Sensitivity and Reproductive Features in Pre-Pubertal and Pubertal Daughters of Polycystic Ovary Syndrome Women. European Journal of Obstetrics & Gynecology and Reproductive Biology 2012, 161, 56–61. [CrossRef]
- Kadowaki, T. Adiponectin and Adiponectin Receptors in Insulin Resistance, Diabetes, and the Metabolic Syndrome. Journal of Clinical Investigation 2006, 116, 1784–1792. [CrossRef]
- Wang, Y.; Ma, X.L.; Lau, W.B. Cardiovascular Adiponectin Resistance: The Critical Role of Adiponectin Receptor Modification. Trends in Endocrinology & Metabolism 2017, 28, 519–530. [CrossRef]
- Marwitz, S.E.; Gaines, M.V.; Brady, S.M.; Mi, S.J.; Broadney, M.M.; Yanovski, S.Z.; Hubbard, V.S.; Yanovski, J.A. Cross-Sectional and Longitudinal Examination of Insulin Sensitivity and Secretion across Puberty among Non-Hispanic Black and White Children. Endocrinol Metab 2020, 35, 847–857. [CrossRef]
- Sir-Petermann, T.; Codner, E.; Pérez, V.; Echiburú, B.; Maliqueo, M.; Ladrón De Guevara, A.; Preisler, J.; Crisosto, N.; Sánchez, F.; Cassorla, F.; et al. Metabolic and Reproductive Features before and during Puberty in Daughters of Women with Polycystic Ovary Syndrome. The Journal of Clinical Endocrinology & Metabolism 2009, 94, 1923–1930. [CrossRef]
- Ni, M.; Lei, H.; Ye, T.; Wang, Y. Adipose-Androgen Crosstalk in Polycystic Ovary Syndrome: Mechanisms and Therapeutic Implications. Front. Endocrinol. 2025, 16, 1731179. [CrossRef]
- Lin, K.; Sun, X.; Wang, X.; Wang, H.; Chen, X. Circulating Adipokine Levels in Nonobese Women With Polycystic Ovary Syndrome and in Nonobese Control Women: A Systematic Review and Meta-Analysis. Front. Endocrinol. 2021, 11, 537809. [CrossRef]
- Cook, J.R.; Semple, R.K. Hypoadiponectinemia—Cause or Consequence of Human “Insulin Resistance”? The Journal of Clinical Endocrinology & Metabolism 2010, 95, 1544–1554. [CrossRef]
- Houston, E.J.; Templeman, N.M. Reappraising the Relationship between Hyperinsulinemia and Insulin Resistance in PCOS. Journal of Endocrinology 2025, 265, e240269. [CrossRef]
- Naulé, L.; Maione, L.; Kaiser, U.B. Puberty, A Sensitive Window of Hypothalamic Development and Plasticity. Endocrinology 2021, 162, bqaa209. [CrossRef]
- Kuryłowicz, A. Estrogens in Adipose Tissue Physiology and Obesity-Related Dysfunction. Biomedicines 2023, 11, 690. [CrossRef]
- Rosenfield, R.L.; Ehrmann, D.A.; Littlejohn, E.E. Adolescent Polycystic Ovary Syndrome Due to Functional Ovarian Hyperandrogenism Persists into Adulthood. J Clin Endocrinol Metab 2015, 100, 1537–1543. [CrossRef]
- Kara, L.; Cicek, D.; Sarikaya, E.; Gok, E.; Berber, U.; Siraz, U.G.; Kendirci, M.; Hatipoglu, N. Adolescent PCOS and Metabolic Health: An Analysis of Fat, Muscle, and Hormones. European Journal of Obstetrics & Gynecology and Reproductive Biology 2025, 314, 114648. [CrossRef]
- Dhokte, S.; Czaja, K. Visceral Adipose Tissue: The Hidden Culprit for Type 2 Diabetes. Nutrients 2024, 16, 1015. [CrossRef]
- Castro-Barquero, S.; Casas, R.; Rimm, E.B.; Tresserra-Rimbau, A.; Romaguera, D.; Martínez, J.A.; Salas-Salvadó, J.; Martínez-González, M.A.; Vidal, J.; Ruiz-Canela, M.; et al. Loss of Visceral Fat Is Associated with a Reduction in Inflammatory Status in Patients with Metabolic Syndrome. Molecular Nutrition & Food Research 2023, 67, 2200264. [CrossRef]
- Reneau, J.; Goldblatt, M.; Gould, J.; Kindel, T.; Kastenmeier, A.; Higgins, R.; Rengel, L.R.; Schoyer, K.; James, R.; Obi, B.; et al. Effect of Adiposity on Tissue-Specific Adiponectin Secretion. PLoS ONE 2018, 13, e0198889. [CrossRef]
- Luo, Y.; Liu, M. Adiponectin: A Versatile Player of Innate Immunity. J Mol Cell Biol 2016, 8, 120–128. [CrossRef]
- Rudnicka, E.; Suchta, K.; Grymowicz, M.; Calik-Ksepka, A.; Smolarczyk, K.; Duszewska, A.M.; Smolarczyk, R.; Meczekalski, B. Chronic Low Grade Inflammation in Pathogenesis of PCOS. Int J Mol Sci 2021, 22. [CrossRef]
- Wu, Y.; Zhong, L.; Li, G.; Han, L.; Fu, J.; Li, Y.; Li, L.; Zhang, Q.; Guo, Y.; Xiao, X.; et al. Puberty Status Modifies the Effects of Genetic Variants, Lifestyle Factors and Their Interactions on Adiponectin: The BCAMS Study. Front Endocrinol (Lausanne) 2021, 12, 737459. [CrossRef]
- Liu, Z.; Liang, S.; Que, S.; Zhou, L.; Zheng, S.; Mardinoglu, A. Meta-Analysis of Adiponectin as a Biomarker for the Detection of Metabolic Syndrome. Front. Physiol. 2018, 9, 1238. [CrossRef]
- Kim, J.Y.; Barua, S.; Jeong, Y.J.; Lee, J.E. Adiponectin: The Potential Regulator and Therapeutic Target of Obesity and Alzheimer’s Disease. Int J Mol Sci 2020, 21. [CrossRef]
- Ghadge, A.A.; Khaire, A.A.; Kuvalekar, A.A. Adiponectin: A Potential Therapeutic Target for Metabolic Syndrome. Cytokine & Growth Factor Reviews 2018, 39, 151–158. [CrossRef]
- Zhao, D.; Sohouli, M.H.; Rohani, P.; Fotros, D.; Velu, P.; Ziamanesh, F.; Fatahi, S.; Shojaie, S.; Li, Y. The Effect of Metformin on Adipokines Levels: A Systematic Review and Meta-Analysis of Randomized-Controlled Trials. Diabetes Research and Clinical Practice 2024, 207, 111076. [CrossRef]
| Study | Country | Study Design | Criteria for PCOS1 | Sample Size | NOS2 Score |
| Rossi, 2008[20] | USA | Case-Control | NIH3 | 74 | 8 |
| Guven, 2009[21] | Türkiye | Case-Control | Rotterdam | 38 | 8 |
| Pinhas-Hamiel, 2009[22] | Israel | Case-Control | Rotterdam | 30 | 4.5 |
| Cekmez, 2011[23] | Türkiye | Case-Control | Rotterdam | 85 | 5 |
| Fulghesu, 2011[24] | Italy | Case-Control | Rotterdam | 99 | 8.5 |
| Yasar, 2011[25] | Türkiye | Case-Control | Rotterdam | 41 | 7 |
| Kale-Gurbuz, 2013[26] | Türkiye | Case-Control | Rotterdam | 38 | 4.5 |
| Cankaya, 2014[27] | Türkiye | Case-Control | Rotterdam | 79 | 7 |
| Ayonrinde, 2016[28] | Australia | Case-Control | NIH | 162 | 7 |
| Cree-Green, 2016[29] | USA | Case-Control | NIH | 71 | 6 |
| Hughan, 2016[30] | USA | Case-Control | ESCPG4 | 110 | 8 |
| Patel, 2017[31] | USA | Case-Control | NIH | 53 | 5 |
| Vital-Reyes, 2017[32] | Mexico | Case-Control | Rotterdam | 47 | 7 |
| Cree-Green, 2018[33] | USA | Case-Control | NIH | 48 | 6.5 |
| Kim, 2018[34] | USA | Case-Control | ESCPG | 42 | 4.5 |
| Ibáñez, 2020[35] | Spain | Prospective Cohort | Not Stated | 114 | 7 |
| Whooten, 2024[36] | USA | Prospective Cohort | IEG5 | 363 | 5.5 |
| Foryś, 2025[37] | Poland | Cross-sectional | IEG | 96 | 8.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




