Submitted:
04 March 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Precursors
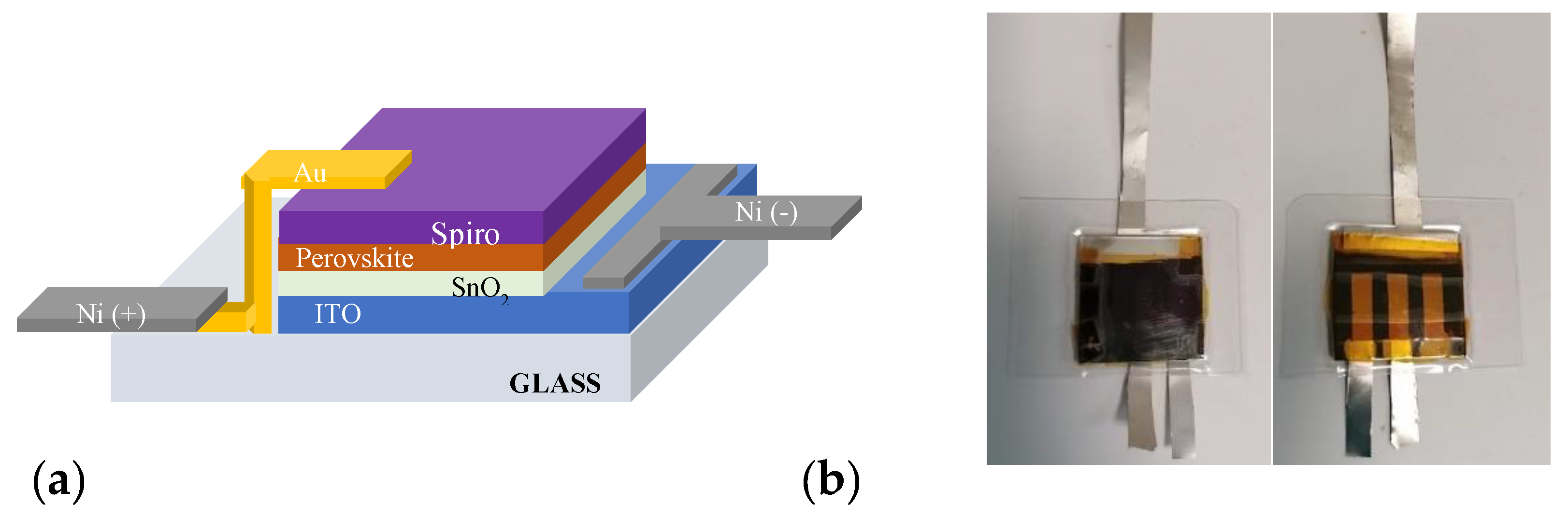
2.3. Perovskite Based Solar Cell and Photoanode Fabrication
3. Results and discussion
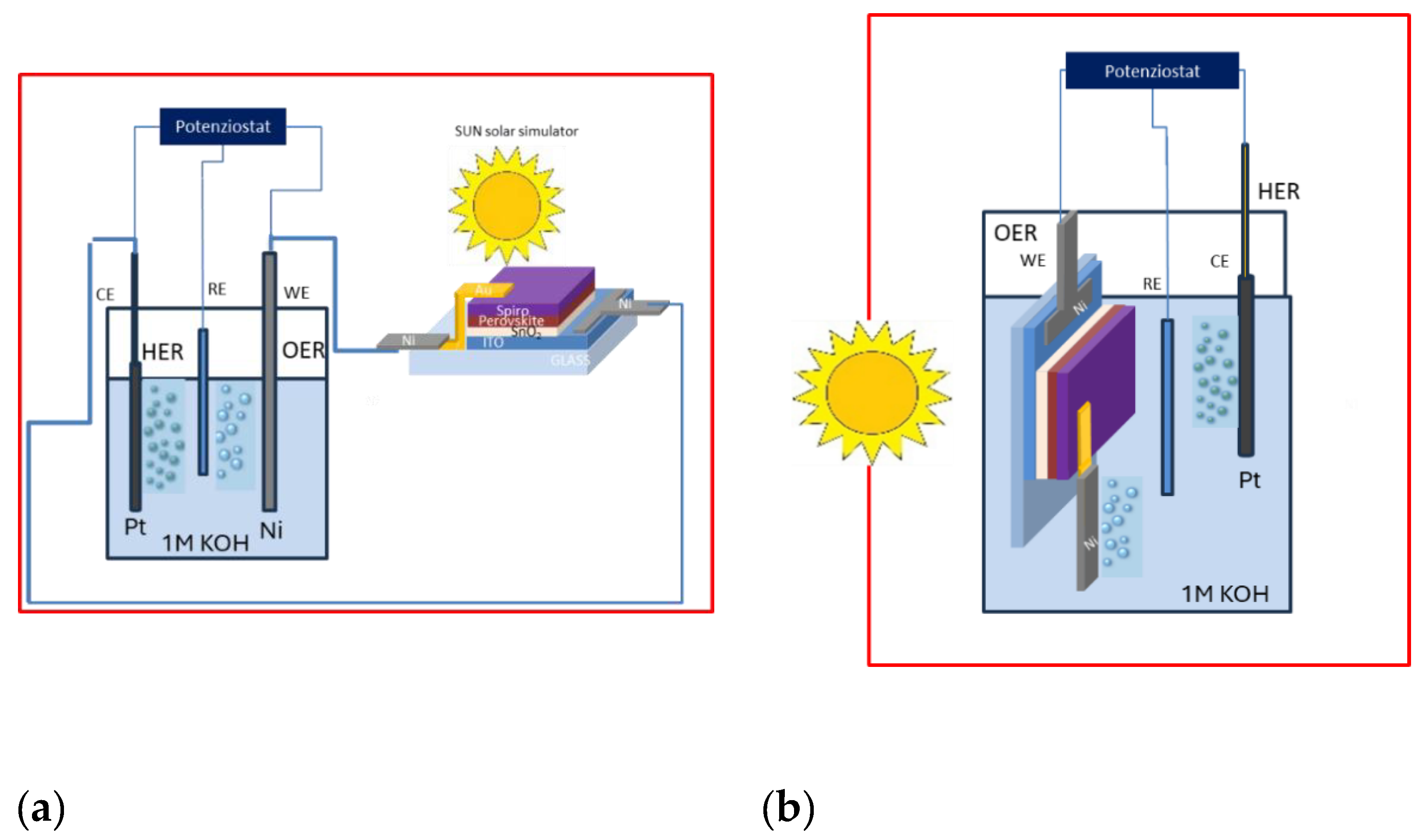
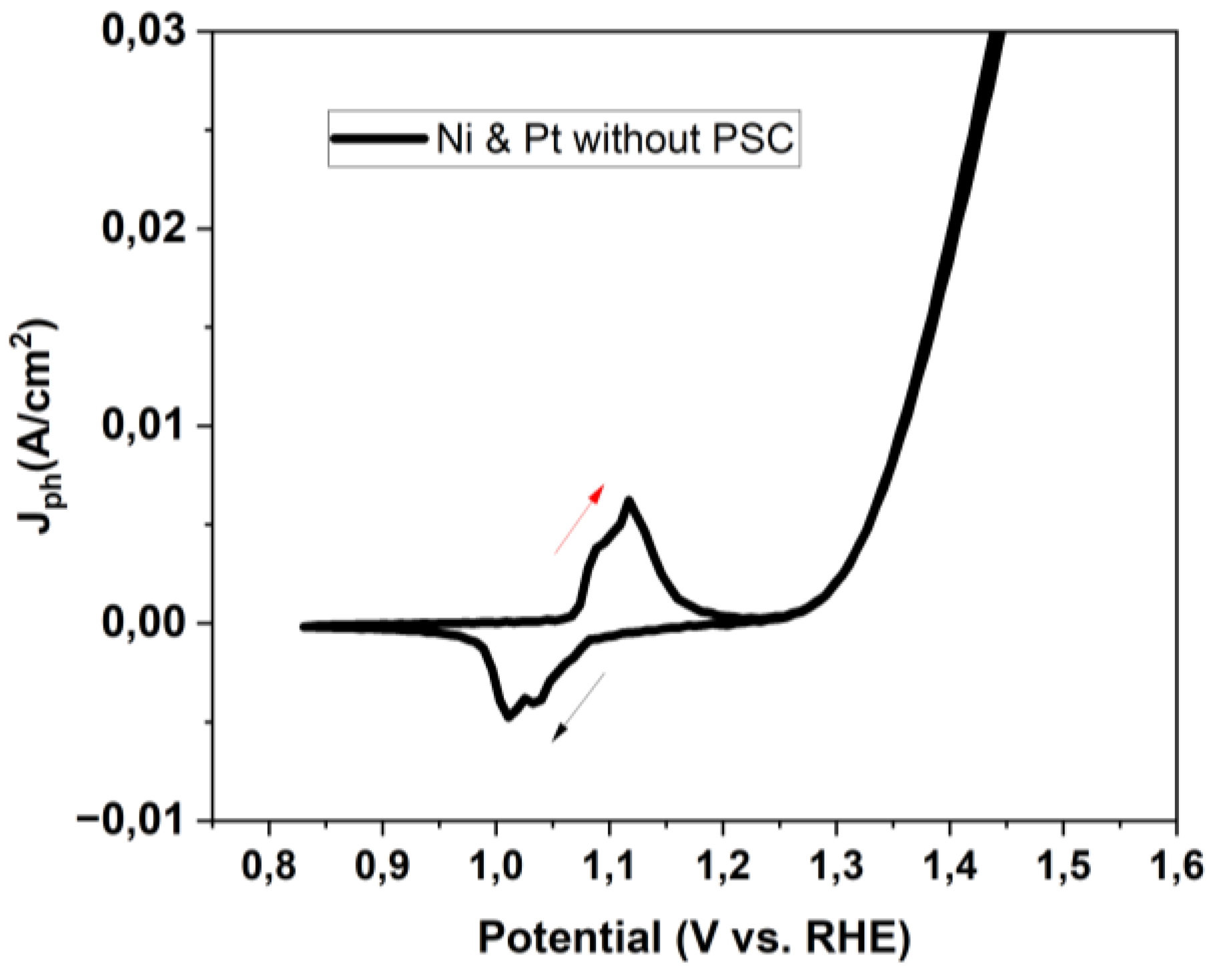
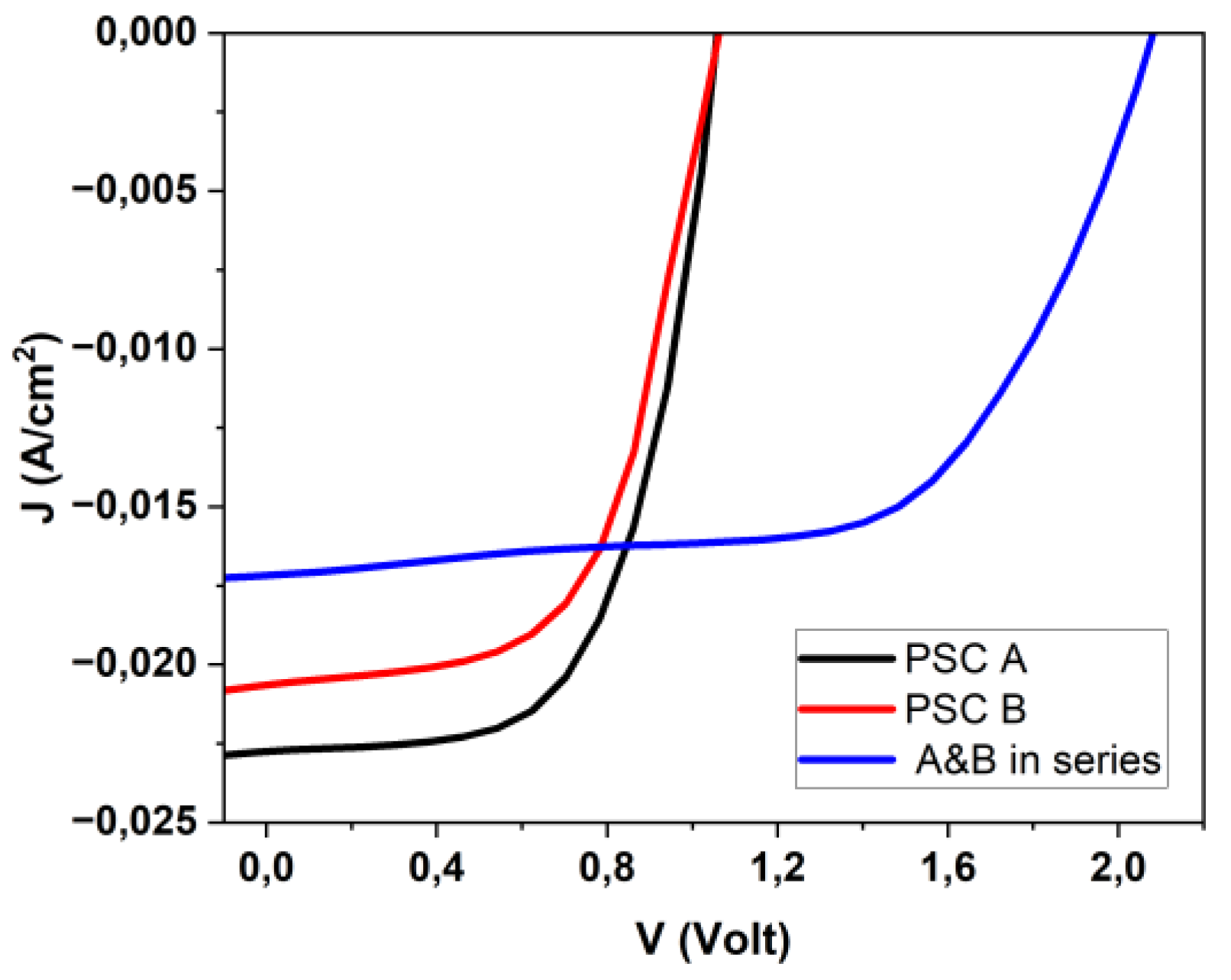
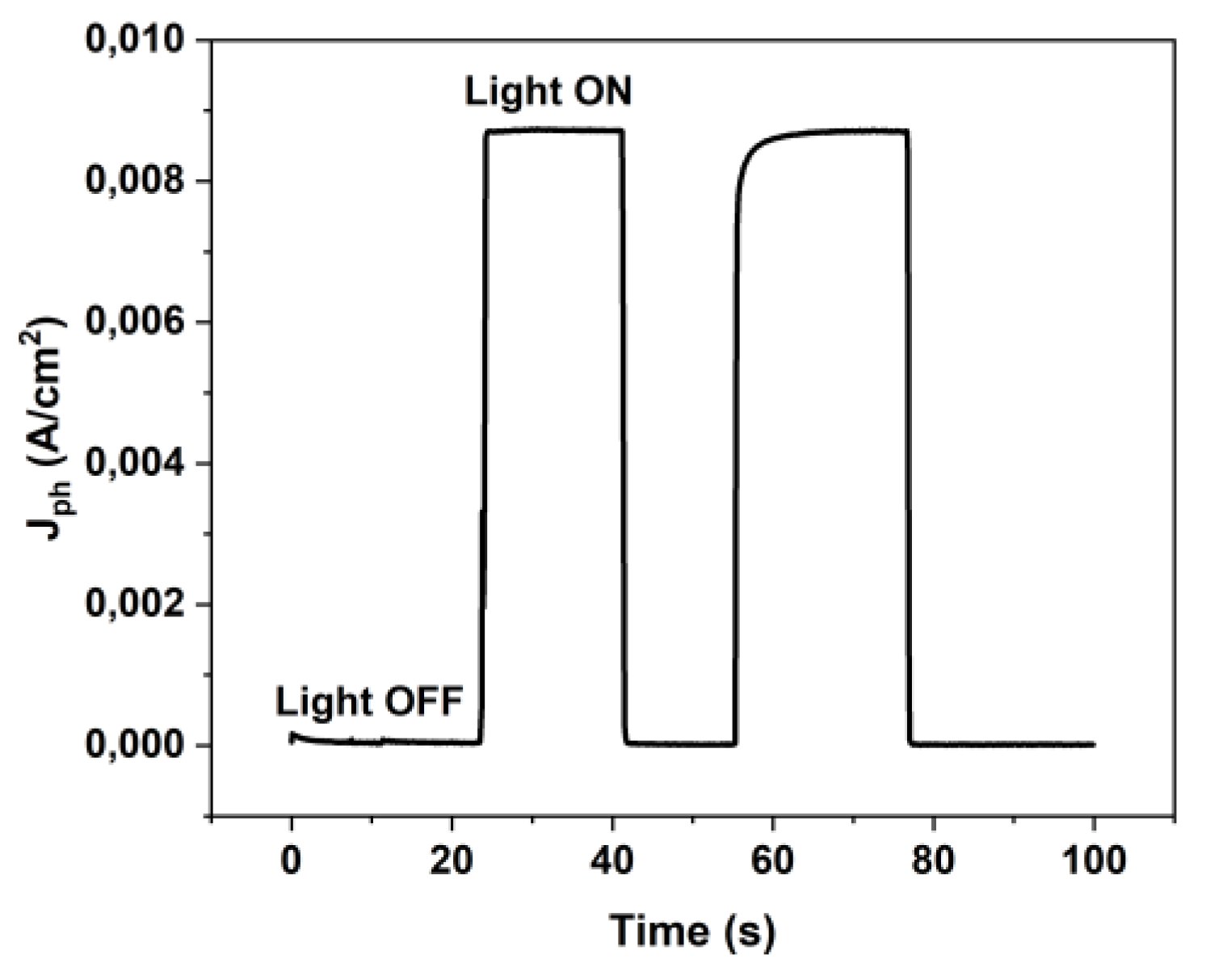
3.1. Perovskite-based PEC Configurations and characterization of Standard Electrochemical Cell

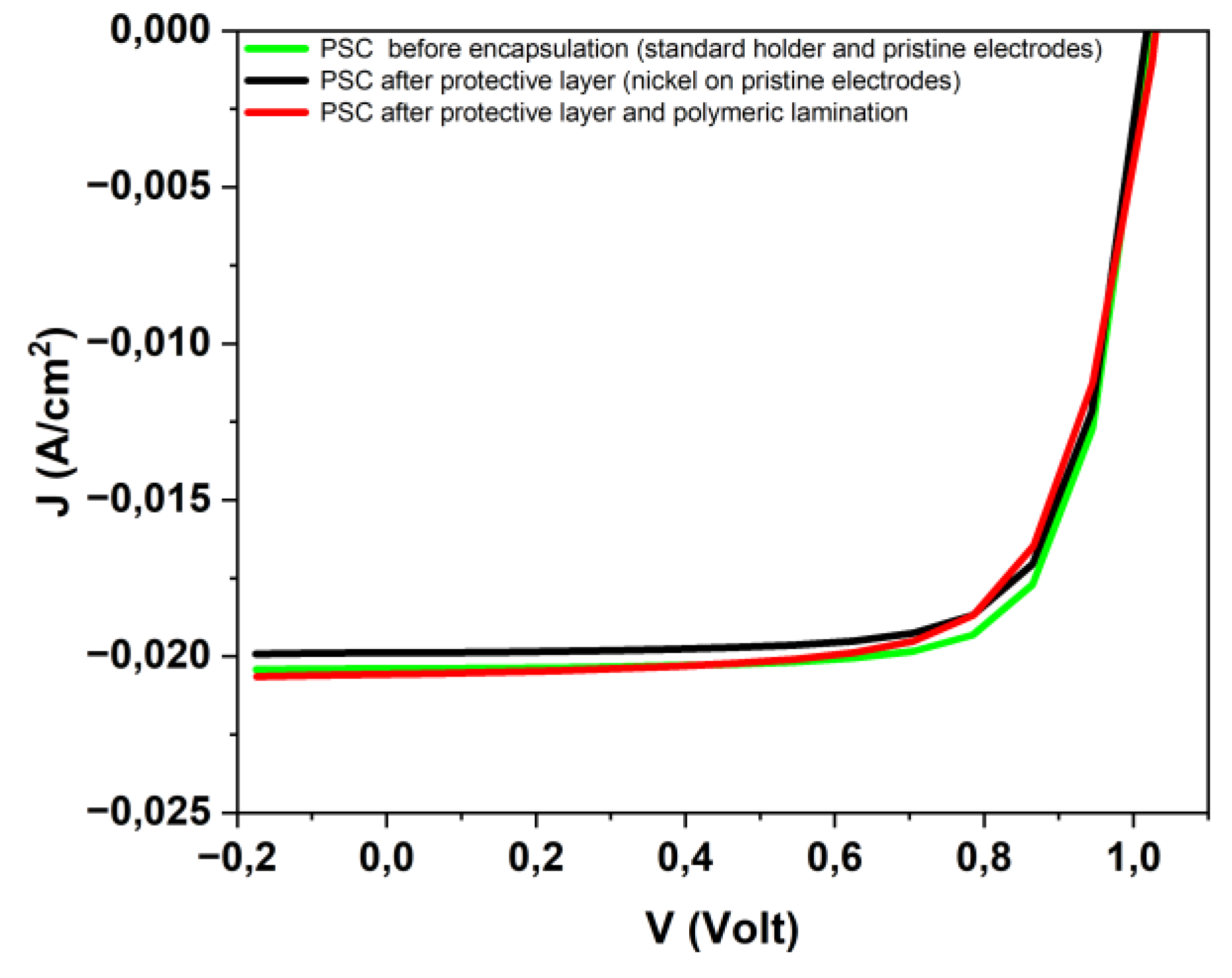
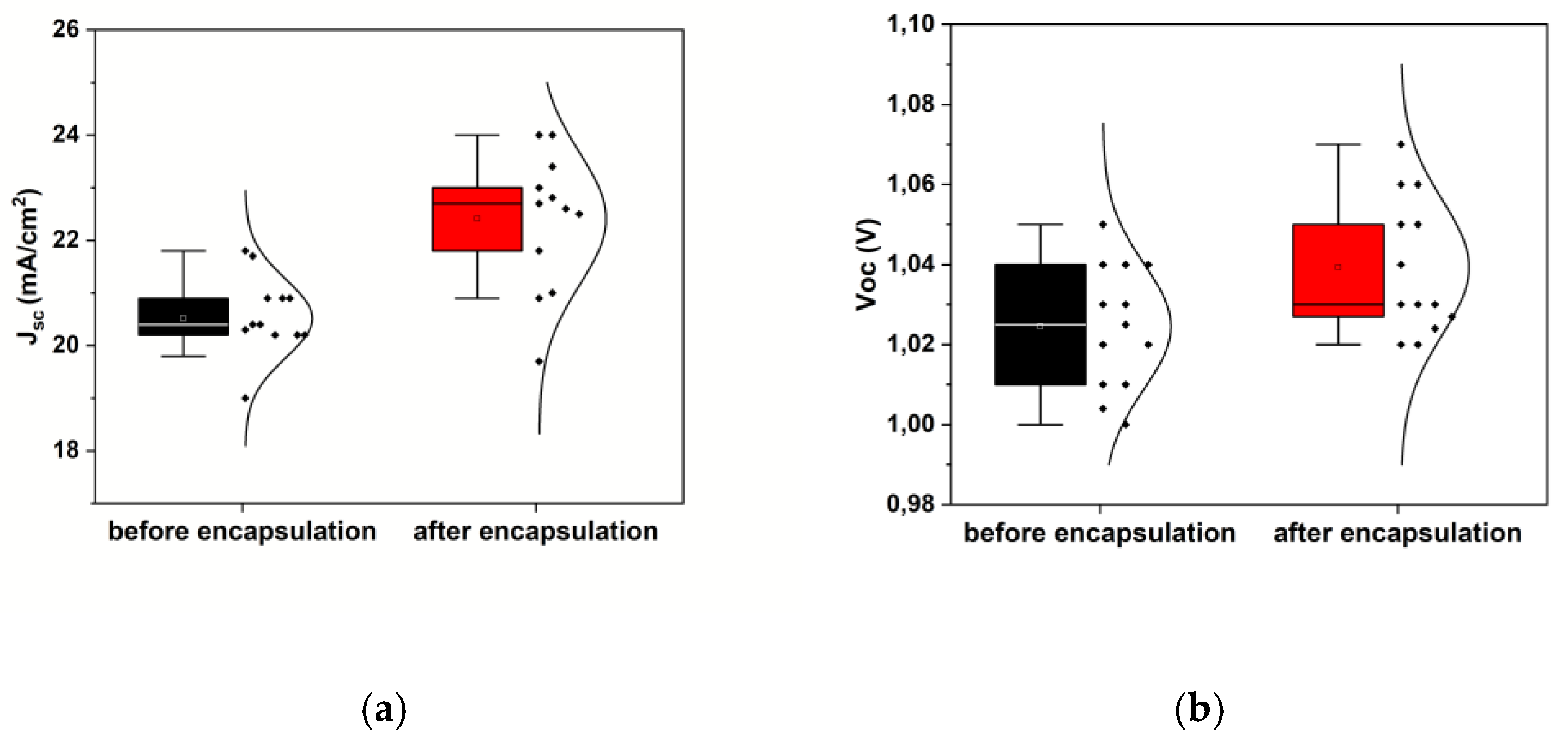
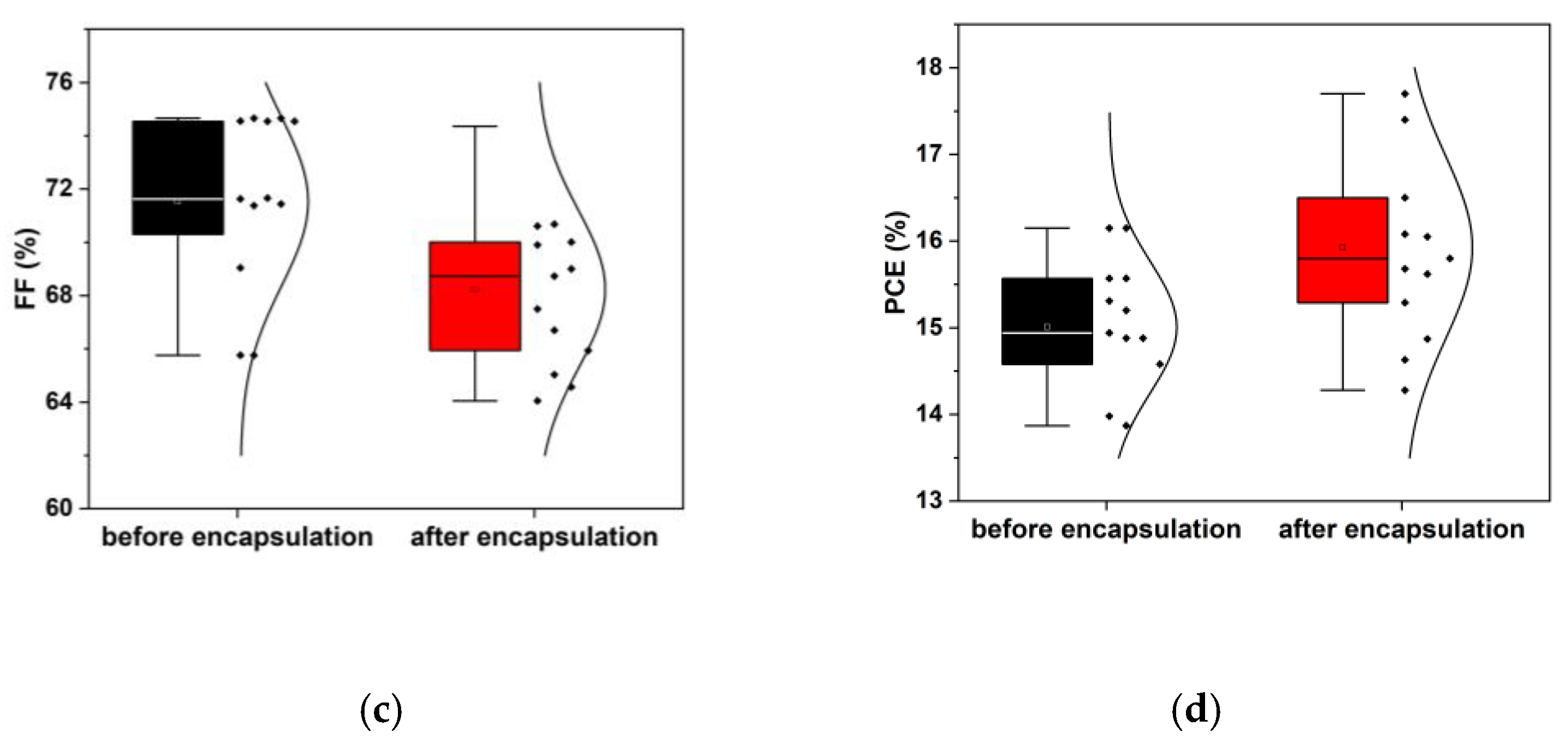
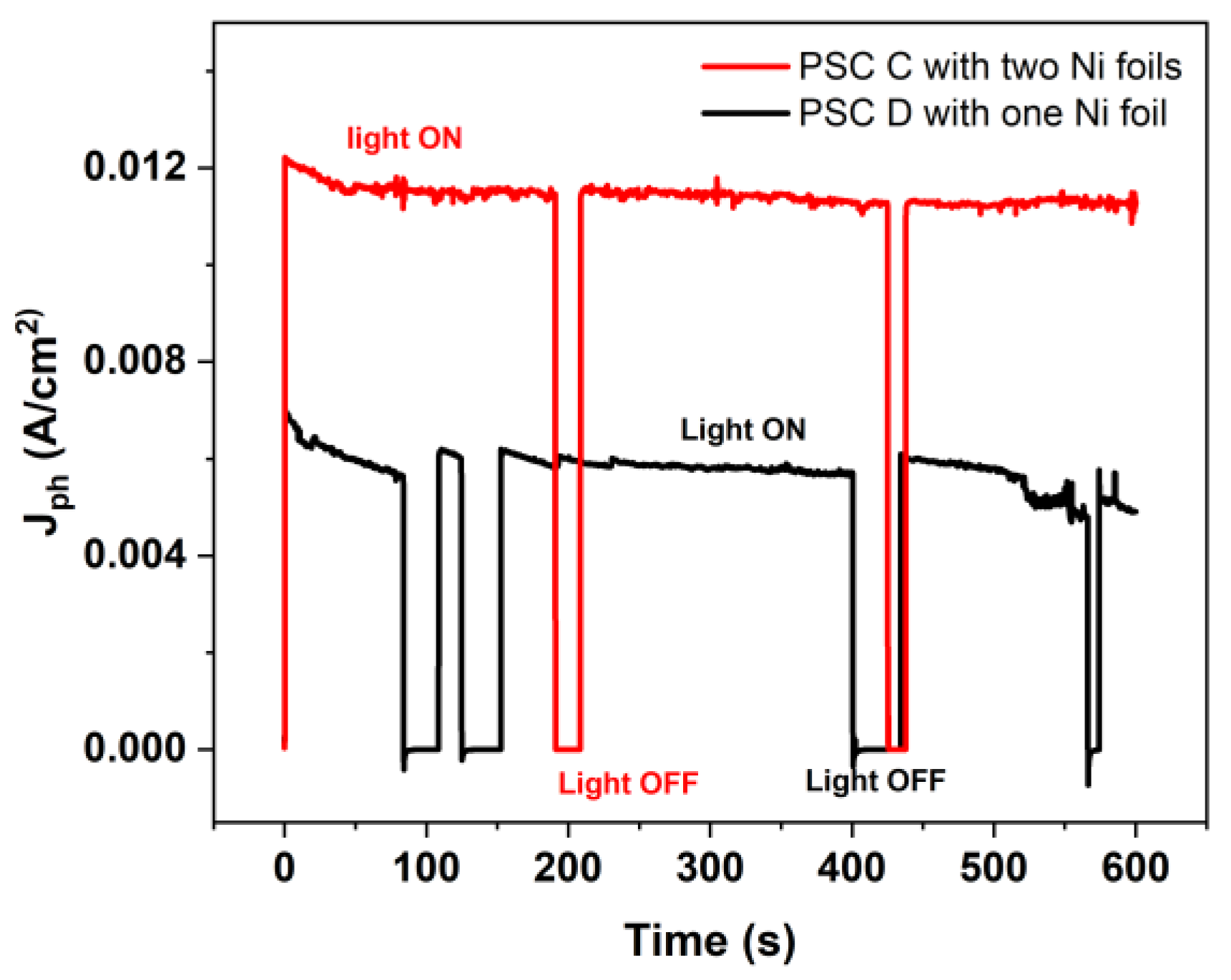
3.2. Encapsulated device characterization for PEC applications
3.3. Experimental Setup for Perovskite Solar Cells in outside and immersed Configurations
3.3.1. Outside configuration
3.3.2. Immersed Configuration
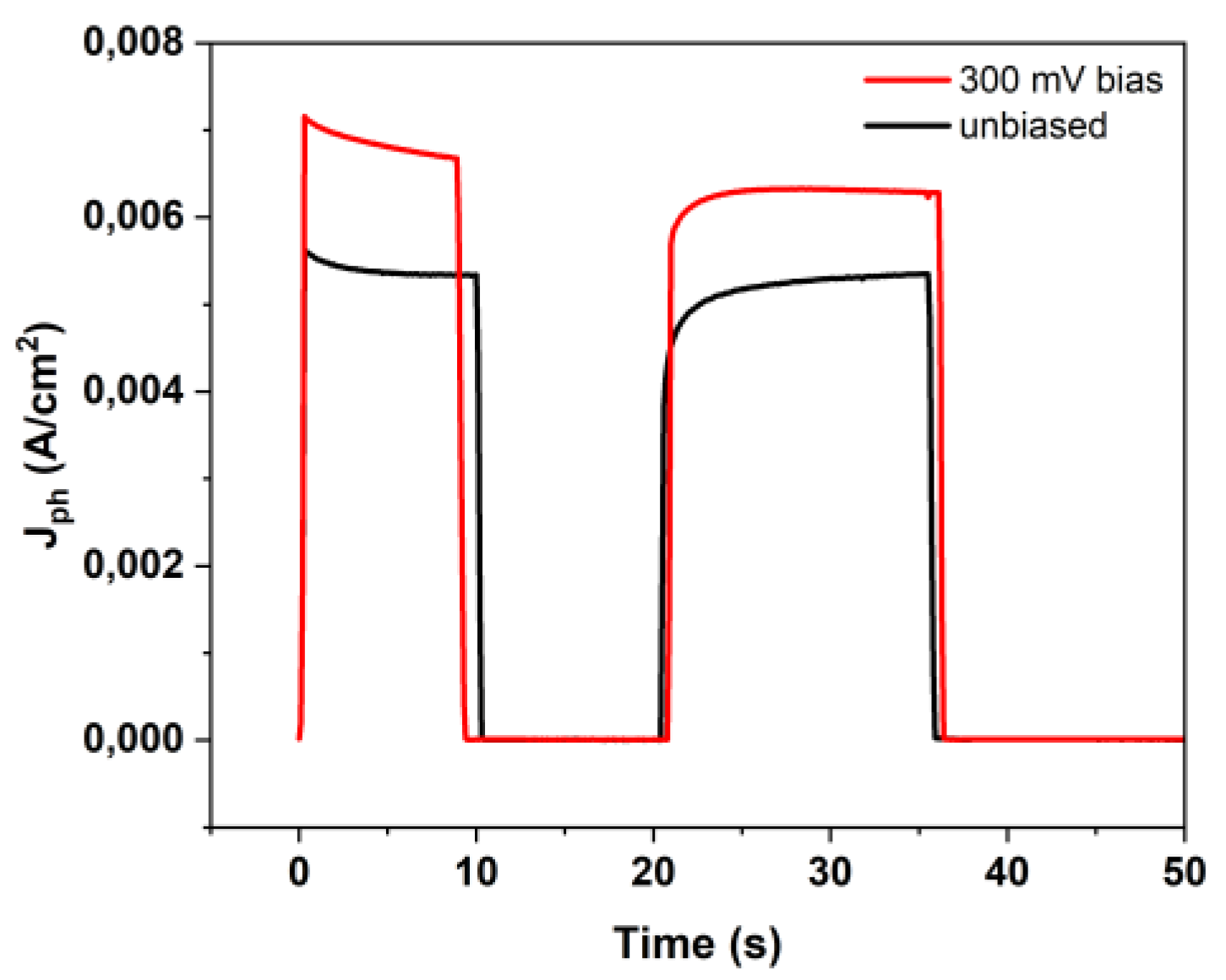
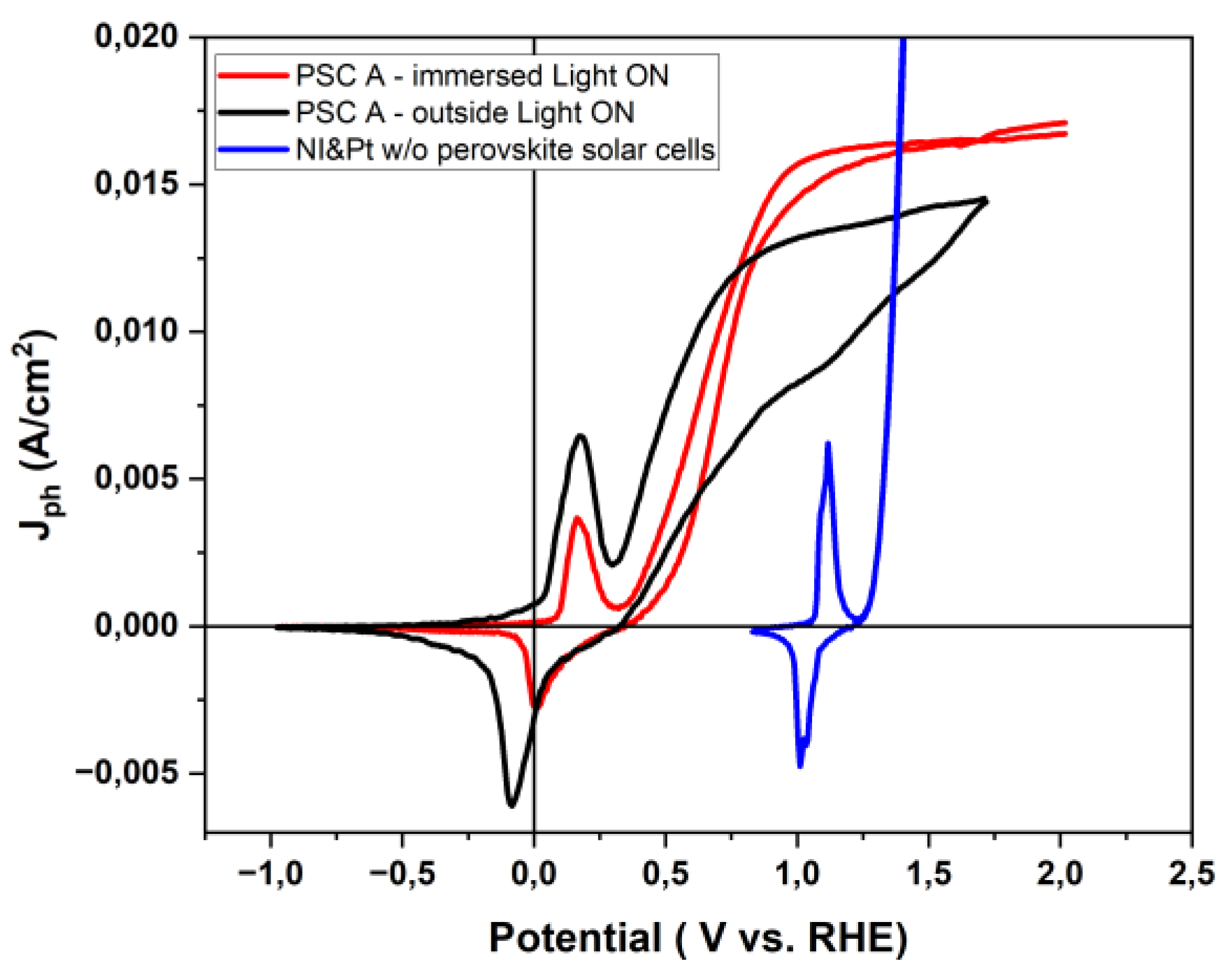
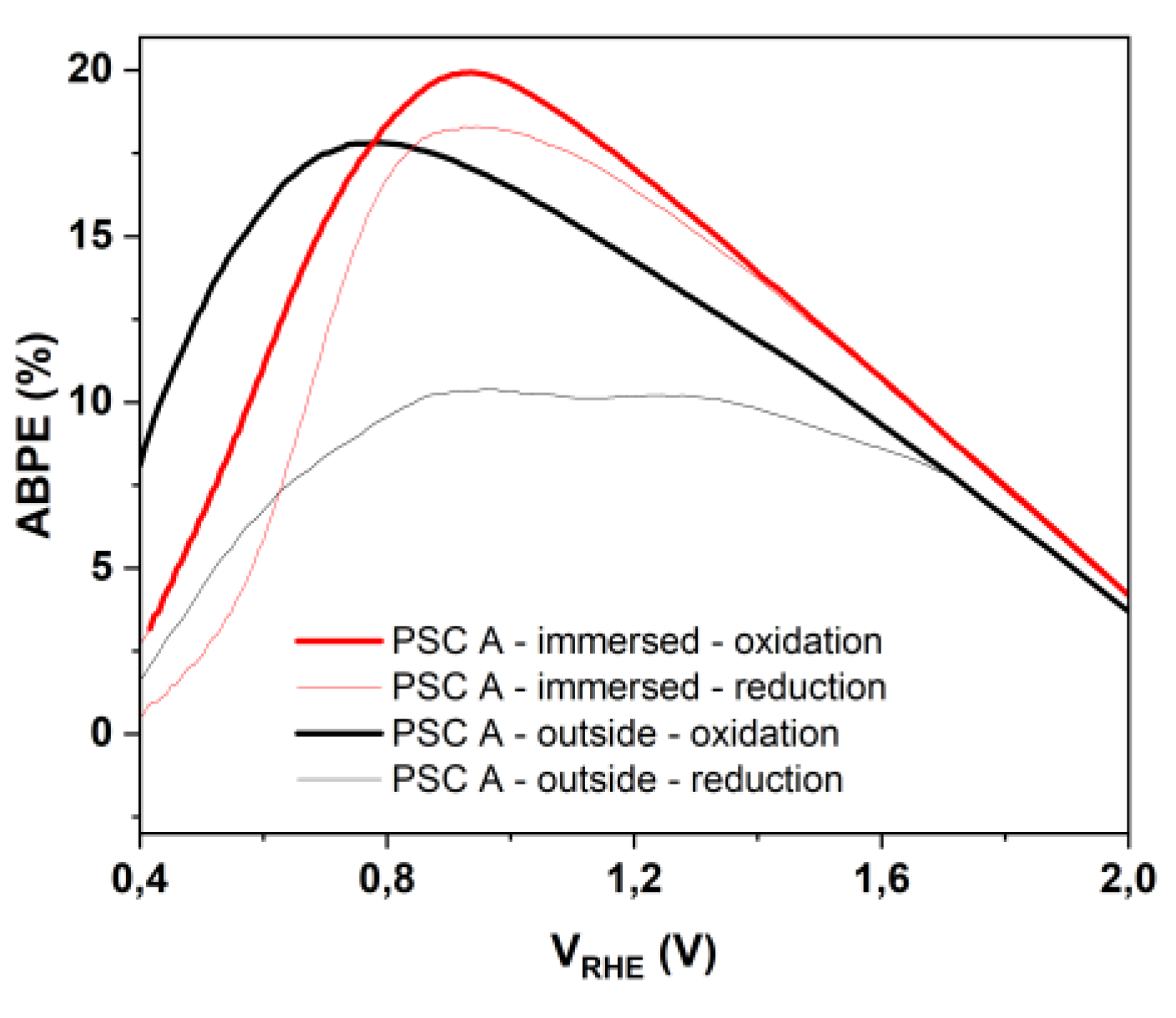
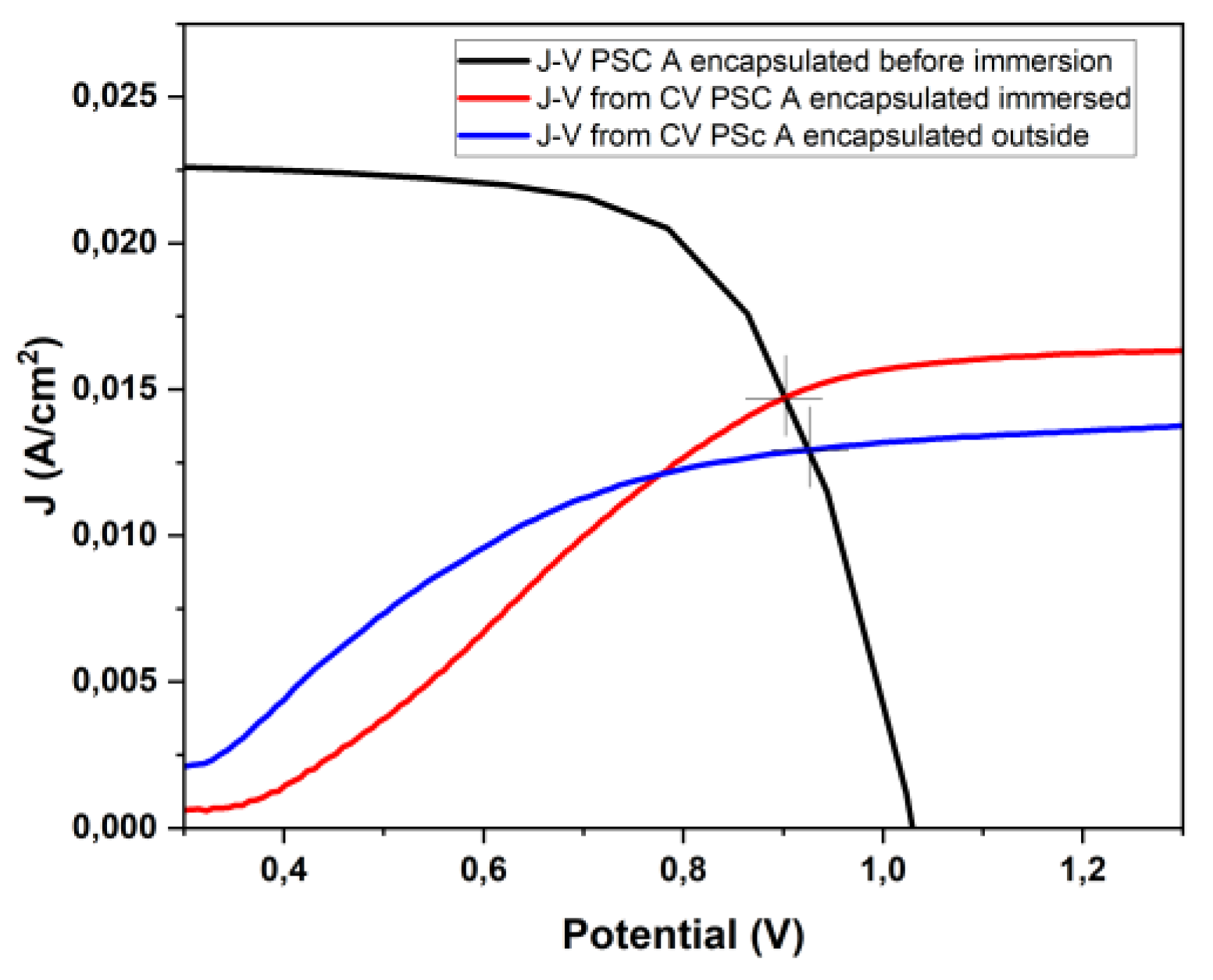
3.4. Photoelectrochemical Response: Outside vs. Immersed Configuration
| Configuration | VOnset (V vs. RHE) |
Vopt (V vs. RHE) |
Jph (mA/cm2) | ABPEmax (%) |
|---|---|---|---|---|
| IS (oxidation curve) | 0.34 | 0.93 | 15.7 | ~19,93 |
| IS (reduction curve) | 0.34 | 0.93 | 15.7 | ~18,31 |
| OS (oxidation curve) | 0.34 | 0.75 | 13.6 | ~17.80 |
| OS (reduction curve) | 0.34 | 0.93 | 9.9 | ~10.39 |
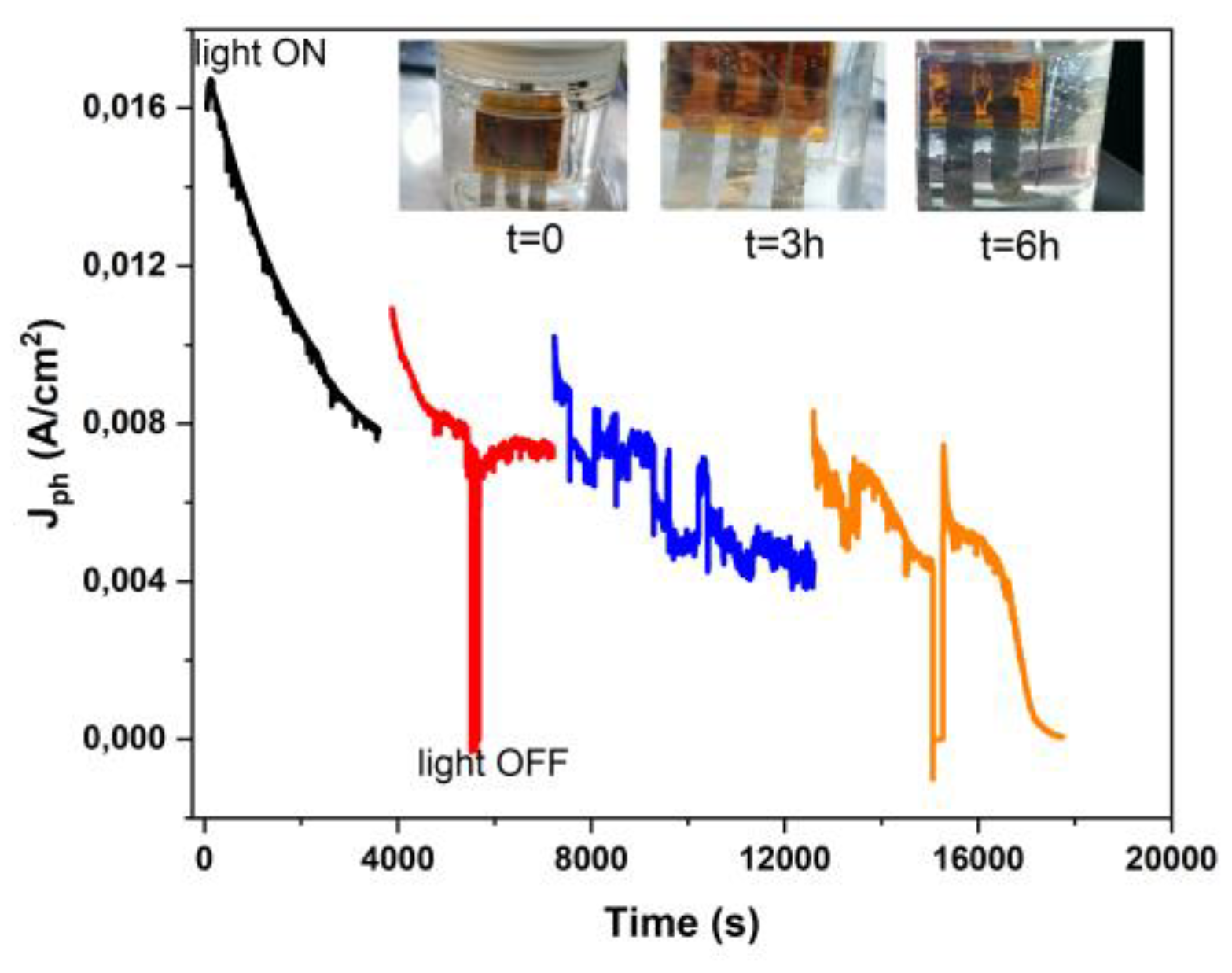
3.5. Stability of Immersed Device
3.6. Comparison with Literature-Reported Approaches
| Photoelectrode configuration | Jph (mA/cm2) at 1.23 V vs. RHE |
ABPE (%) | STH (%) | Aging | Ref. |
|---|---|---|---|---|---|
| Cs0.1(MA0.17FA0.83)0.9Pb(I0.83Br0.17)3 immersed photoanode/Nickel foil |
16.4 |
19.9 |
5 h |
This work | |
| MAPbI3 –BiVO₄ photoanode tandem –outside PSC |
5.01 |
6.2 | [11] | ||
| FAPbI3/Ni/ NiFeOOH – immersed photoanode |
22.8 |
9.8 |
3 days |
[12] | |
| FAPbBr3/ carbon/graphite/ NiFe immersed Photoanode |
9.12 |
8.5 |
100 h |
[13] | |
| Cs0.05FA0.85MA0.1Pb(I0.95Br0.05)3 & FA(PbI3)0.97(MAPbI3)0.03 immersed photocathode&photoanode Tandem perovskite/silicon phoanode |
20 16.9 10 |
13.4* 20.8* 13.4* |
|
5 h 1 h 102 h |
[14] |
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| OS | Outside |
| IS | Immersed |
| OER | Oxygen Evolution Reaction |
| HER | Hydrogen Evolution Reaction |
| ABPE | Applied Bias Photon-to-Current Efficiency |
| RHE | Reversible Hydrogen Electrode |
| PET–EVA | poly (ethylene terephthalate) - ethylene vinylacetate |
| AM | Air Mass |
| PSC | Perovskite Solar Cell |
| PEC | Photoelectrochemical |
| STH | Solar to Hydrogen |
| CV | Cyclic Voltammetry |
| ITO | Indium tin oxide |
| n-i-p | negative-intrinsic-positive |
| ETL | Electron Transport Layer |
| HTL | Hole Transport Layer |
| J–V | Current density - voltage |
| LED | Light Emitting Diode |
| Jsc | Short circuit current density |
| Voc | Open circuit voltage |
| FF | Fill factor |
| PCE | Power conversion efficiency |
| Jph | Photocurrent density |
| PV | Photovoltaic |
| Vopt | Optimal bias |
References
- NREL Best Research-Cell Efficiency Chart. Available online: https://www.nrel.gov/pv/cell-efficiency.html (accessed on 2026).
- Green, M.; Ho-Baillie, A.; Snaith, H. The emergence of perovskite solar cells. Nature Photon 2014, 8, 506–514. [Google Scholar] [CrossRef]
- Park, N.-G.; Zhu, K. Scalable fabrication and coating methods for perovskite solar cells and solar modules. Nat Rev Mater 2020, 5, 333–350. [Google Scholar] [CrossRef]
- Niu, G.; Li, W.; Li, J.; Liang, X.; Wang, L. Enhancement of thermal stability for perovskite solar cells through cesium doping. RSC Adv 2017, 7, 17473–17479. [Google Scholar] [CrossRef]
- Jeong, M.; Choi, I. W.; Go, E. M.; Cho, Y.; Kim, M.; Lee, B.; Jeong, S.; Jo, Y.; Choi, H. W.; Lee, J.; Bae, J. H.; Kwak, S. K.; Kim, D. S.; Yang, C. Stable perovskite solar cells with efficiency exceeding 24.8% and 0.3-V voltage loss. Science 2020, 369, 1615–1620. [Google Scholar] [CrossRef]
- Ahn, N.; Son, D. Y.; Jang, I. H.; Kang, S. M.; Choi, M.; Park, N.-G. Highly reproducible perovskite solar cells with average efficiency of 18.3% and best efficiency of 19.7% fabricated via Lewis base adduct of lead(II) iodide. J Am Chem Soc 2015, 137, 8696–8699. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Song, X.; Liu, H.; Zhang, A.; Cao, J.; Wu, C. Encapsulation-driven stability in perovskite solar cells: suppressing degradation through hermetic sealing. ACS Appl Mater Interfaces 2025, 17, 34119–34128. [Google Scholar] [CrossRef]
- Aitola, K.; Gava Sonai, G.; Markkanen, M.; Kaschuk, J. J.; Hou, X.; Miettunen, K.; Lund, P. D. Encapsulation of commercial and emerging solar cells with focus on perovskite solar cells. Sol Energy 2022, 237, 264–283. [Google Scholar] [CrossRef]
- Dipta, S. S.; Rahim, M. A.; Uddin, A. Encapsulating perovskite solar cells for long-term stability and prevention of lead toxicity. Appl Phys Rev 2024, 11, 021301. [Google Scholar] [CrossRef]
- Chu, Q.-Q.; Sun, Z.; Wang, D.; Cheng, B.; Wang, H.; Wong, C.-P.; Fang, B. Encapsulation: The path to commercialization of stable perovskite solar cells. Matter 2023, 6, 3838–3863. [Google Scholar] [CrossRef]
- Qiu, Y.; Liu, W.; Chen, W.; Zhou, G.; Hsu, P.-C.; Zhang, R.; Liang, Z.; Fan, S.; Zhang, Y.; Cui, Y. Efficient solar-driven water splitting by nanocone BiVO₄–perovskite tandem cells. Sci Adv 2016, 2, e1501764. [Google Scholar] [CrossRef] [PubMed]
- Hansora, D.; Yoo, J. W.; Mehrotra, R.; Byun, W. J.; Lim, D.; Kim, Y. K.; Noh, E.; Lim, H.; Jang, J. W.; Seok, S. I.; Lee, J. S. All-perovskite-based unassisted photoelectrochemical water splitting system for efficient, stable and scalable solar hydrogen production. Nat Energy 2024, 9, 272–284. [Google Scholar] [CrossRef]
- Yang, H.; Liu, Y.; Ding, Y.; Li, F.; Wang, L.; Cai, B.; Zhang, F.; Liu, T.; Boschloo, G.; Johansson, E. M. J.; Sun, L. Monolithic FAPbBr₃ photoanode for photoelectrochemical water oxidation with low onset-potential and enhanced stability. Nat Commun 2023, 14, 5486. [Google Scholar] [CrossRef] [PubMed]
- Fehr, A. M. K.; Agrawal, A.; Mandani, F.; Conrad, C. L.; Jiang, Q.; Park, S. Y.; Alley, O.; Li, B.; Sidhik, S.; Metcalf, I.; Botello, C.; Young, J. L.; Even, J.; Blancon, J.-C.; Deutsch, T. G.; Zhu, K.; Albrecht, S.; Toma, F. M.; Wong, M.; Mohite, A. D. Integrated halide perovskite photoelectrochemical cells with solar-driven water-splitting efficiency of 20.8%. Nat Commun 2023, 14, 3797. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Li, C.; Chen, L.; Dolia, K.; Fu, S.; Sun, N.; Li, Y.; Wyatt, K.; Young, J. L.; Deutsch, T. G.; Yan, Y. All-perovskite tandem photoelectrodes for unassisted solar hydrogen production. ACS Energy Lett 2023, 8, 2611–2619. [Google Scholar] [CrossRef]
- La Ferrara, V.; De Maria, A.; Rametta, G. Green anisole as antisolvent in planar triple-cation perovskite solar cells with varying cesium concentrations. Micromachines 2024, 15, 136. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A. C.; Koper, M. T. M. Effect of saturating the electrolyte with oxygen on the activity for the oxygen evolution reaction. ACS Catal 2018, 8, 9359–9363. [Google Scholar] [CrossRef] [PubMed]
- Tress, W.; Marinova, N.; Inganäs, O.; Nazeeruddin, M. K.; Zakeeruddin, S. M.; Grätzel, M. Understanding the rate-dependent J–V hysteresis, slow time component, and aging in CH₃NH₃PbI₃ perovskite solar cells. Energy Environ Sci 2015, 18 8, 995–1004. [Google Scholar] [CrossRef]
- Ocaña, L.; Montes, C.; González-Díaz, B.; González-Pérez, S.; Llarena, E. Evaluation of ethylene-vinyl acetate, methyl methacrylate, and polyvinylidene fluoride as encapsulating materials for perovskite-based solar cells using the low-temperature encapsulation method in a cleanroom environment. Energies 2024, 17, 60. [Google Scholar] [CrossRef]
- Emery, Q.; Dagault, L.; Khenkin, M.; Kyranaki, N.; Bernardes de Araújo, W. M.; Erdil, U.; Demuylder, M.; Cros, S.; Schlatmann, R.; Stannowski, B.; Ulbrich, C. Tips and tricks for a good encapsulation for perovskite-based solar cells. Prog Photovolt Res Appl 2025, 33, 551–559. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




