Submitted:
04 March 2026
Posted:
05 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
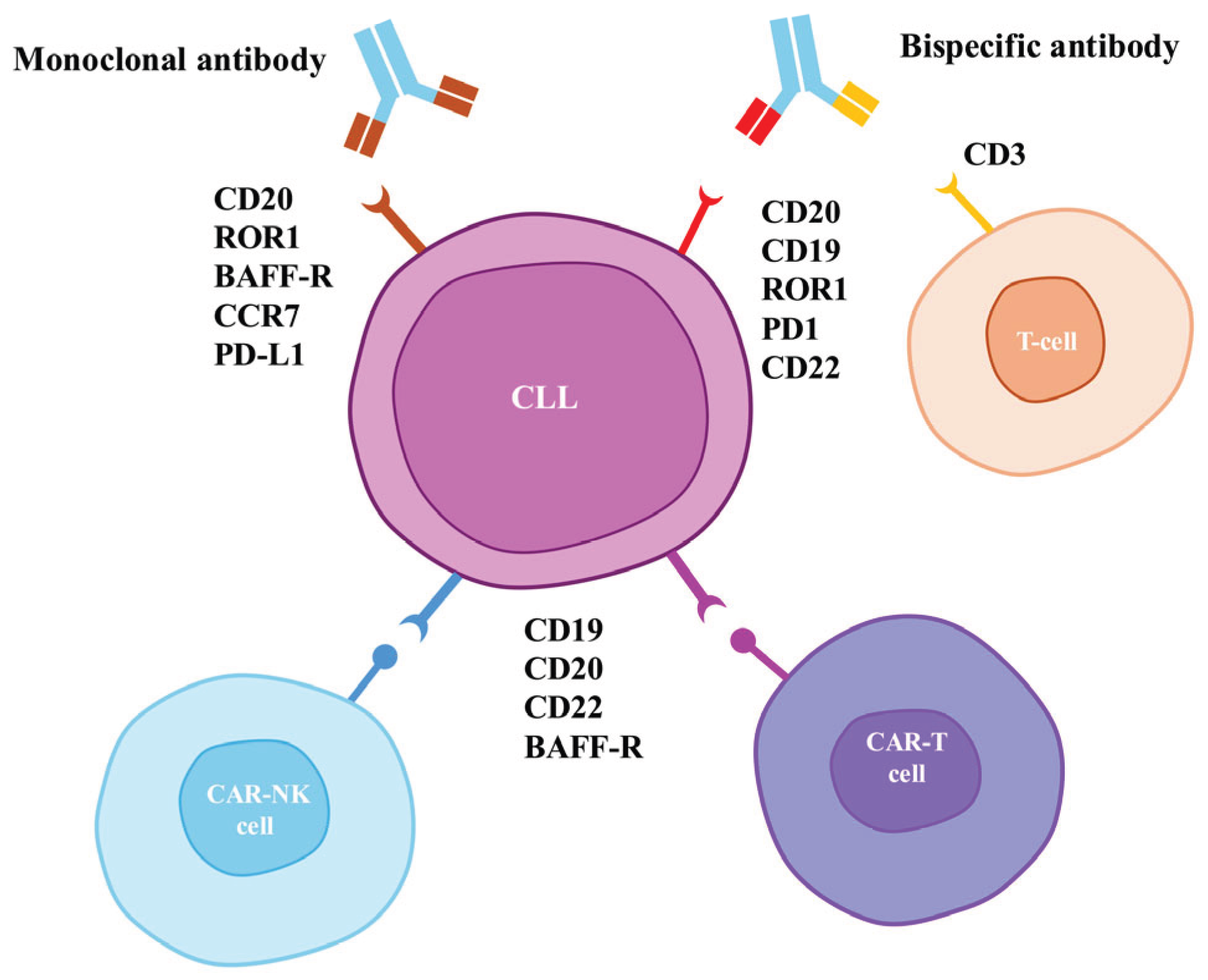
2. Novel Monoclonal Antibodies
2.1. Ianalumab
2.2. CAP-100
2.3. Belimumab
2.4. Tislelizumab
2.5. Pembrolizumab
2.6. Atezolizumab
2.7. ABBV-319
2.8. Zilovertamab
2.9. Zilovertamab Vedotin
| Agent | Phase | Antibody target | No. of participants | ORR | CR/CRi | PFS | OS | Most Common Grade ≥ 3 AEs [%] |
Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Ianalumab + ibrutinib | Ib | BAFF-R | 39 (expansion n=24) | N/A | 37,5% | N/A | N/A | Gr. ≥3 TEAEs 41.0% TRAEs: 23.1% |
[23] |
| Tislelizumab + zanubrutinib | II | PD-1 | 59 (enrolled); 48 (comprised in full analysis set) | 58,3% | 18,8% | Median: 10 months 46.9% at 1 year |
Median: not reached 74,7% at 12 months |
Gr. ≥1 TEAEs 98.2% (n=57) Gastrointestinal disorders: 56.1% Infections: 78.9% with UTI 21.1% Pyrexia: 19.3% Peripheral edema: 17.5% Anaemia: 19.3% Neutropenia: 21.1% Thrombocytopenia: 19.3% |
[27] |
| Pembrolizumab + dinaciclib | Ib | PD-1 | 72 (enrolled); 17 (R/R CLL), 38 (R/R DLBCL), 17 (R/R MM) | 29.4% (R/R CLL) | N/A | Median: 5.2 months (R/R CLL) |
Median: 21.7 months (R/R CLL) | Gr. ≥3 TEAEs: 32% (n= 12); Lymphopenia:13%; Neutropenia: 11%; Thrombocytopenia: 8%; Leukopenia: 8%; Laboratory TLS: 5% |
[28,29] |
| Atezolizumab + obinutuzumab | II |
PD-L1 | 37 (enrolled) | N/A | N/A | 94% at 2 years; 89% at 4 years |
94% at 2 years 94% at 4 years |
Gr. ≥3 TEAEs: Neutropenia: 59%; Thrombocytopenia: 32% |
[30] |
| Zilovertamab + ibrutinib | I/II | ROR1 | 70 (enrolled), 34 (CLL), 26 (R/R MCL) |
91.2% | 8.8% | Median: not reached | N/A | Gr. ≥3 TEAEs Hypertension:10.6%; Pneumonia: 7.1%; Neutropenia: 5.9%; Atrial fibrillation: 5.9%; Fatigue: 5.9% TEAEs due to Zilo: 23.5% (CLL) |
[33] |
| Zilovertamab vedotin + nemtabrutinib | I | ROR1 | 56 (enrolled); 17 (DLBCL); 17 (MCL); 7 (RT) |
57% (RT) |
14% (RT) | Median: 4.7 months (RT) | Median: 19.4 months (RT) | Gr. ≥3 TEAEs: 48% (n=27); Neutropenia: 32%; Thrombocytopenia: 11% |
[34] |
3. Bispecific Antibodies
3.1. Epcoritamab
3.2. NVG-111
3.3. Mosunetuzumab
3.4. Glofitamab
3.5. IGLV3-21R110-Directed Bispecific Antibody (R110-bsAB)
| Agent & Target |
Agent characteristics | Clinical Trial Number | Trial characteristics | No. of participants |
|---|---|---|---|---|
| Epcoritamab | BsAb CD20×CD3 approved for DLBCL, FL and high-grade B-cell Lymphoma | NCT04623541 (EPCORE CLL-1) |
Phase I/II study on epcoritamab monotherapy in R/R CLL. ORR: 61%, CR: 39%, mPFS: 12.8 months, mOS: not reached [41]. | 23 |
| Phase I/II study on epcoritamab monotherapy in RT. ORR: 47.6%, CR: 40%, mPFS: 3.0 months, mOS: 13.0 months [39]. | 42 | |||
| Phase I/II study on epcoritamab with lenalidomide in RT. ORR: 82%, CR: 73%, mPFS: 5.7 months, mOS: not reached [40]. | 11 | |||
| Phase I/II study on epcoritamab with R-CHOP in RT ORR: 61%, CR: 39%, mPFS: 12.8 months, OS: not reached [40]. | 30 | |||
| NCT07108998 | Phase II study oCLL/SLL (consolidation therapy for 2nd generation BTKi +/- obinutuzumab) |
22 (estimated) | ||
| NCT05791409 | Phase I/II study on epcoritamab with venetoclax in R/R CLL/SLL. Estimated study completion in 2032. | 112 (estimated) | ||
| NCT06676033 | Phase I study on epcoritamab in CLL and RT. Estimated study completion in 2027. | 5 | ||
| NCT07218510 (LonGEVity Trial) |
Phase II study on epicoritamab as a consolidation therapy for venetoclax and obinutuzumab in previously untreated CLL/SLL. Estimated study completion in 2029. | 33 (estimated) | ||
| Mosunetuzumab | humanized BsAb CD3×CD20 approved for R/R FL | NCT05091424 | Phase I study of mosunetuzumab alone or in combination with venetoclax in R/R CLL. Estimated study completion in 2030. | 137 (estimated) |
| NCT06926205 | Phase II study of mosunetuzumab with CHOP as a first line in RT. Estimated study completion in 2028. | 34 (estimated) | ||
| NCT07052695 | Phase I/II study of mosunetuzumab alone or in combination with BTKi in CLL/SLL. Estimated study completion in 2032. | 40 (estimated) | ||
| NCT02500407 | Phase I/II study on mosunetuzumab monotherapy and with atezolizumab in R/RCLL and B-cell NHL. Trial completed. | 713 | ||
| GB261 CD20×CD3 |
BsAb CD20×CD3 computationally designed to maintain Fc effector function | NCT04923048* | Phase I/II study in CLL and R/R B-cell NHL. Unknown status. | 460 (estimated) |
| AZD0486 (Surovatamig) |
IgG4 fully human BsAb CD19×CD3 | NCT06564038 | Phase I/II study on AZD0486 as monotherapy or in combination with other anticancer agents in R/R CLL/SLL and other mature B-Cell malignancies. Estimated study completion 2028. |
276 (estimated) |
| NVG111 ROR1×CD3 |
first in class, humanized, tandem scFv, ROR1xCD3 BsAb | NCT04763083* | Phase I study in R/R ROR1+ malignancies. Unknown status of completion. | 90 (estimated) |
| ONO4685 PD1×CD3 |
First in class PD1×CD3 BsAb | NCT06547528 | Phase I study in CLL/SLL and T-cell lymphoma. Estimated completion 2029. | 108 (estimated) |
| JNJ 75348780 CD22×CD3 |
CD22×CD3 human BsAb | NCT04540796 | Phase I study in R/R CLL NHL completed in 2025. | 147 |
4. Cell Therapies
4.1. Challenges
4.2. Autologous CD19 CAR-T Cells
4.2.1. Lisocabtagene Maraleucel
4.2.2. Tisagenlecleucel
4.2.3. Axicabtagene Ciloleucel
4.2.4. Brexucabtagene Autoleucel
4.2.5. GLPG5201
4.2.6. huCART19-IL18
4.2.7. Varnimcabtagene Autoleucel
4.2.8. JCAR014
4.2.9. HD-CAR-1
4.2.10. BAFF-R CAR T-Cell Therapy
4.3. Dual-Target CAR-T Cells
4.4. Triple-Target CAR-T Cell Therapy
4.5. Allogeneic CAR-T Cells
4.6. Combination Therapies with CAR-T Cells
4.6.1. Ibrutinib
4.6.2. Lenalidomide
4.6.3. PI3K Inhibition
4.7. Allogeneic CAR-NK Cells
| Agent | Phase | Target | No. of Parti-cipants | ORR | CR / CRi | PFS rate | OS rate | Safety | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Lisocabtagene maraleucel (liso-cel) | I / II | CD19 | 117 | 47% | 18% | Median: 11.9 months | Median 30.3 months | CRS gr. ≥3: 9%; NT gr ≥3: 19% | [21] |
| Tisagenlecleucel (tisa-cel) | I | CD19 | 14 | 57% | 29% | Median: 7 months | Median: 29 months | Neutropenia fever, delayed CRS | [63] |
| Axicabtagene ciloleucel (axi-cel) | I/II | CD19 | 7 | 88% | 63% | N/A | Median OS not reached | N/A | [65] |
| Brexucabtagene autoleucel (brexu-cel) | I | CD19 | 15 | 47% | 13% | N/A | N/A | CRS gr. 4: 7%; NT gr ≥3: 20% | [66] |
| GLPG5201 | I / II | CD19 | 15 | 93% | 66.7% | N/A | N/A | No grade ≥3 CRS or any-grade ICANS were observed | [67] |
| huCART19-IL18 | I | CD19 | 21 (R/R NHL) | 81% at 3 months | 52% | N/A | N/A | CRS: 62% (gr. ≥3: 15%); ICANS: 14% (all gr. 1-2) | [68] |
| Varnimcabtagene autoleucel (ARI-0001) | I | CD19 | 13 (CLL cohort) | 84.6% | 84.6% | 60% at 4 years | Not reached | CRS: 90% (gr ≥3: 3.4%); ICANS gr.2: 6.9% | [69] |
| JCAR014 | I / II | CD19 | 47 | 70% | 17% | Median: 8.9 months | Median: 25 months | CRS: 82% (Gr ≥3: 14%); NT: 33% (Gr ≥3: 27%) | [70] |
| HD-CAR-1 | I / II | CD19 | 9 | 67% | 67% | 30% at 2 years | 69% at 2 years | CRS gr. 3: 9%; no NT | [71] |
| LV20.19 | I | CD20 / CD19 | 14 (RT and CLL) | 92% | 46% | N/A | Median: 15 months | CRS: 100% (gr. ≥3: 14.3%); ICANS: 21% (gr. 3: 14.3%); IEC-HS: 64% (Gr ≥3: 14.3%) | [76] |
| Liso-cel + Ibrutinib | I | CD19 | 19 | 95% | 63% | N/A | N/A | CRS: 74% (Gr ≥3: 5%); NT: 32% (gr ≥3: 16%) | [86] |
| huCART-19 + Ibrutinib | II | CD19 | 19 | 83% | 43% at 3 months | 84% at 4 years | 70% at 4 years | CRS: 94.7% (gr. ≥3: 15.8%); NT: 26.3% (gr. 4: 5.3%) | [87] |
| Anti-CD19 CAR-NK | I / II | CD19 | 37 | 49% | 37.8% | 32% at 1 year | 68% at 1 year | No significant CRS, NT, or GvHD | [95] |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data availability statement
Conflicts of interest
References
- Hallek, M.; Cheson, B.D.; Catovsky, D.; Caligaris-Cappio, F.; Dighiero, G.; Döhner, H.; Hillmen, P.; Keating, M.; Montserrat, E.; Chiorazzi, N.; et al. iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL. Blood 2018, 131, 2745–2760. [Google Scholar] [CrossRef]
- Eichhorst, B.; Robak, T.; Montserrat, E.; Ghia, P.; Niemann, C.U.; Kater, A.P.; Gregor, M.; Cymbalista, F.; Buske, C.; Hillmen, P.; et al. Chronic lymphocytic leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2021, 32, 23–33. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma Version 2025; National Comprehensive Cancer Network (NCCN), 2025. [Google Scholar]
- Hallek, M. Chronic Lymphocytic Leukemia: 2025 Update on the Epidemiology, Pathogenesis, Diagnosis, and Therapy. Am J Hematol 2025, 100, 450–480. [Google Scholar] [CrossRef]
- Robak, T.; Iskierka-Jażdżewska, E.; Puła, B. XXI International Workshop on Chronic Lymphocytic Leukemia in Kraków 2025 — conference report. Acta Haematologica Polonica 2025, 56, 477–482. [Google Scholar] [CrossRef]
- Hallek, M.; Fischer, K.; Fingerle-Rowson, G.; Fink, A.M.; Busch, R.; Mayer, J.; Hensel, M.; Hopfinger, G.; Hess, G.; von Grünhagen, U.; et al. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Lancet 2010, 376, 1164–1174. [Google Scholar] [CrossRef]
- Fischer, K.; Bahlo, J.; Fink, A.M.; Goede, V.; Herling, C.D.; Cramer, P.; Langerbeins, P.; von Tresckow, J.; Engelke, A.; Maurer, C.; et al. Long-term remissions after FCR chemoimmunotherapy in previously untreated patients with CLL: updated results of the CLL8 trial. Blood 2016, 127, 208–215. [Google Scholar] [CrossRef]
- Stilgenbauer, S.; Zenz, T. Understanding and managing ultra high-risk chronic lymphocytic leukemia. Hematology Am Soc Hematol Educ Program 2010, 2010, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Hus, I.; Giannopoulos, K.; Jamroziak, K.; Wolowiec, D.; Roliński, J.; Robak, T. Diagnostic and therapeutic recommendations of the Polish Society of Haematologists and Transfusiologists and Polish Adult Leukemia Group-CLL for chronic lymphocytic leukemia in 2025. Acta Haematologica Polonica 2025, 56, 143–172. [Google Scholar] [CrossRef]
- Wolska-Washer, A.E.; Robak, T. Acalabrutinib in treatment of patients with chronic lymphocytic leukemia including those at high genetic risk. Acta Haematologica Polonica 2025, 56, 172–186. [Google Scholar] [CrossRef]
- Sharman, J.P.; Egyed, M.; Jurczak, W.; Skarbnik, A.; Pagel, J.M.; Flinn, I.W.; Kamdar, M.; Munir, T.; Walewska, R.; Corbett, G.; et al. Acalabrutinib with or without obinutuzumab versus chlorambucil and obinutuzmab for treatment-naive chronic lymphocytic leukaemia (ELEVATE TN): a randomised, controlled, phase 3 trial. Lancet 2020, 395, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Seymour, J.F.; Kipps, T.J.; Eichhorst, B.; Hillmen, P.; D'Rozario, J.; Assouline, S.; Owen, C.; Gerecitano, J.; Robak, T.; De la Serna, J.; et al. Venetoclax-Rituximab in Relapsed or Refractory Chronic Lymphocytic Leukemia. N Engl J Med 2018, 378, 1107–1120. [Google Scholar] [CrossRef] [PubMed]
- Quartermaine, C.; Ghazi, S.M.; Yasin, A.; Awan, F.T.; Fradley, M.; Wiczer, T.; Kalathoor, S.; Ferdousi, M.; Krishan, S.; Habib, A.; et al. Cardiovascular Toxicities of BTK Inhibitors in Chronic Lymphocytic Leukemia: JACC: CardioOncology State-of-the-Art Review. JACC CardioOncol 2023, 5, 570–590. [Google Scholar] [CrossRef] [PubMed]
- Zygmunciak, P.; Dancewicz, H.; Stróżna, K.; Błażowska, O.; Bieliński, K.; Robak, T.; Puła, B. Double Strike in Chronic Lymphocytic Leukemia-The Combination of BTK and BCL2 Inhibitors in Actual and Future Clinical Practice. Int J Mol Sci 2025, 26. [Google Scholar] [CrossRef]
- Zygmunciak, P.; Robak, T.; Puła, B. Treatment of Double-Refractory Chronic Lymphocytic Leukemia-An Unmet Clinical Need. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Robak, T.; Iskierka-Jażdżewska, E.; Puła, A.; Robak, P.; Puła, B. The Development of Novel Therapies for Chronic Lymphocytic Leukaemia in the Era of Targeted Drugs. J Clin Med 2025, 14. [Google Scholar] [CrossRef]
- Ramsay, A.G.; Gribben, J.G. Immune dysfunction in chronic lymphocytic leukemia T cells and lenalidomide as an immunomodulatory drug. Haematologica 2009, 94, 1198–1202. [Google Scholar] [CrossRef]
- Riches, J.C.; Davies, J.K.; McClanahan, F.; Fatah, R.; Iqbal, S.; Agrawal, S.; Ramsay, A.G.; Gribben, J.G. T cells from CLL patients exhibit features of T-cell exhaustion but retain capacity for cytokine production. Blood 2013, 121, 1612–1621. [Google Scholar] [CrossRef]
- Kipps, T.J. ROR1: an orphan becomes apparent. Blood 2022, 140, 1583–1591. [Google Scholar] [CrossRef]
- Choi, M.Y.; Widhopf, G.F., 2nd; Ghia, E.M.; Kidwell, R.L.; Hasan, M.K.; Yu, J.; Rassenti, L.Z.; Chen, L.; Chen, Y.; Pittman, E.; et al. Phase I Trial: Cirmtuzumab Inhibits ROR1 Signaling and Stemness Signatures in Patients with Chronic Lymphocytic Leukemia. Cell Stem Cell 2018, 22, 951–959.e953. [Google Scholar] [CrossRef]
- Siddiqi, T.; Maloney, D.G.; Kenderian, S.S.; Brander, D.M.; Dorritie, K.; Soumerai, J.; Riedell, P.A.; Shah, N.N.; Nath, R.; Fakhri, B.; et al. Lisocabtagene maraleucel in chronic lymphocytic leukaemia and small lymphocytic lymphoma (TRANSCEND CLL 004): a multicentre, open-label, single-arm, phase 1–2 study. The Lancet 2023, 402, 641–654. [Google Scholar] [CrossRef] [PubMed]
- Casan, J.M.L.; Wong, J.; Northcott, M.J.; Opat, S. Anti-CD20 monoclonal antibodies: reviewing a revolution. Human Vaccines & Immunotherapeutics 2018, 14, 2820–2841. [Google Scholar] [CrossRef]
- Rogers, K.A.; Yan, P.; Flinn, I.W.; Stephens, D.M.; Kipps, T.J.; Larson, S.M.; Martz, L.; Chen, X.; Wang, H.; Hopping, E.; et al. Addition of Ianalumab (VAY736) to Ibrutinib in Patients with Chronic Lymphocytic Leukemia on Ibrutinib Therapy: Results from a Phase Ib Study. Clin Cancer Res 2025, 31, 5145–5158. [Google Scholar] [CrossRef]
- Klein, U.; Tu, Y.; Stolovitzky, G.A.; Mattioli, M.; Cattoretti, G.; Husson, H.; Freedman, A.; Inghirami, G.; Cro, L.; Baldini, L.; et al. Gene expression profiling of B cell chronic lymphocytic leukemia reveals a homogeneous phenotype related to memory B cells. J Exp Med 2001, 194, 1625–1638. [Google Scholar] [CrossRef]
- Mateu-Albero, T.; Marcos-Jimenez, A.; Delgado-Wicke, P.; Terrón, F.; Loscertales, J.; López-Matencio, J.M.S.; Muñoz-Calleja, C.; Cuesta-Mateos, C. Evaluation of the novel therapeutic anti-CCR7 antibody CAP-100 as an add-on therapy in chronic lymphocytic leukemia patients receiving venetoclax. Hematol Oncol 2023, 41, 869–876. [Google Scholar] [CrossRef] [PubMed]
- Tandler, C.; Schmidt, M.; Heitmann, J.S.; Hierold, J.; Schmidt, J.; Schneider, P.; Dörfel, D.; Walz, J.; Salih, H.R. Neutralization of B-Cell Activating Factor (BAFF) by Belimumab Reinforces Small Molecule Inhibitor Treatment in Chronic Lymphocytic Leukemia. Cancers (Basel) 2020, 12. [Google Scholar] [CrossRef]
- Al-Sawaf, O.; Ligtvoet, R.; Robrecht, S.; Stumpf, J.; Fink, A.M.; Tausch, E.; Schneider, C.; Boettcher, S.; Mikusko, M.; Ritgen, M.; et al. Tislelizumab plus zanubrutinib for Richter transformation: the phase 2 RT1 trial. Nat Med 2024, 30, 240–248. [Google Scholar] [CrossRef]
- Gregory, G.P.; Kumar, S.; Wang, D.; Mahadevan, D.; Walker, P.; Wagner-Johnston, N.; Escobar, C.; Bannerji, R.; Bhutani, D.; Chang, J.; et al. Pembrolizumab plus dinaciclib in patients with hematologic malignancies: the phase 1b KEYNOTE-155 study. Blood Adv 2022, 6, 1232–1242. [Google Scholar] [CrossRef]
- Gregory, G.; Walker, P.; Mahadevan, D.; Wang, D.; Chang, J.; Hernandez-Ilizaliturri, F.; Klein, A.; Rybka, W.; Wagner-Johnston, N.; Escobar, C.; et al. ANTITUMOR ACTIVITY OF PEMBROLIZUMAB PLUS DINACICLIB IN PATIENTS WITH DIFFUSE LARGE B CELL LYMPHOMA: THE PHASE 1B KEYNOTE-155 STUDY. Hematological Oncology 2019, 37, 328–329. [Google Scholar] [CrossRef]
- Reville, P.K.; Sajedi, S.; Ferrajoli, A.; Burger, J.A.; Keating, M.J.; Garg, N.; Deng, Q.; Green, M.R.; Abbas, H.A.; Wierda, W.G.; et al. Atezolizumab Combined with Venetoclax and Obinutuzumab for Frontline CLL. Blood 2024, 144, 1014. [Google Scholar] [CrossRef]
- Rose, A.L.; Smith, B.E.; Maloney, D.G. Glucocorticoids and rituximab in vitro: synergistic direct antiproliferative and apoptotic effects. Blood 2002, 100, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.A.; Emberley, E.; D'Souza, A.L.; Zhao, W.; Cosgrove, C.; Parrish, K.; Mitra, D.; Payson, E.; Oleksijew, A.; Ellis, P.; et al. ABBV-319: a CD19-targeting glucocorticoid receptor modulator antibody-drug conjugate therapy for B-cell malignancies. Blood 2024, 144, 757–770. [Google Scholar] [CrossRef]
- Lee, H.J.; Choi, M.Y.; Siddiqi, T.; Rhodes, J.M.; Wierda, W.G.; Isufi, I.; Tuscano, J.M.; Lamanna, N.; Subbiah, S.; Koff, J.L.; et al. Phase 1/2 study of zilovertamab and ibrutinib in mantle cell lymphoma (MCL) or chronic lymphocytic leukemia (CLL). Journal of Clinical Oncology 2022, 40, 7520–7520. [Google Scholar] [CrossRef]
- Wang, M.L.; Mei, M.; Barr, P.M.; Barrientos, J.C.; de Vos, S.; Furman, R.R.; Patel, K.; Wierda, W.G.; Choi, M.Y.; Kallam, A.; et al. Zilovertamab vedotin (MK-2140) in relapsed or refractory (R/R) non-Hodgkin lymphoma (NHL): 14-month follow-up of the phase 1 waveLINE-001 study. Hematological Oncology 2023, 41, 571–572. [Google Scholar] [CrossRef]
- Paszkiewicz-Kozik, E.; Moreira, C.; Turgut, M.; Garrido, M.; Glimelius, I.; Lee, S.T.; Sawalha, Y.; Ren, Y.; Ryland, K.; Ogbu, U.C.; et al. Zilovertamab Vedotin in Combination with Nemtabrutinib for Patients with Relapsed or Refractory Mantle Cell Lymphoma: Cohort C of the Open-Label, Phase 2 Waveline-006 Study. Blood 2024, 144, 3025–3025. [Google Scholar] [CrossRef]
- Nayyar, M.; Menezes, R.C.B.; Ailawadhi, S.; Parrondo, R.D. Chronic Lymphocytic Leukemia: Novel Therapeutic Targets Under Investigation. Cancers (Basel) 2025, 17. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, M.; Basharat, A.; Irfan, S.; Sadiq, M.H.; Amin, M.K.; Jaglal, M.V. Future Landscapes of Bispecific Antibodies in Chronic Lymphocytic Leukemia (CLL). Systematic Review of Ongoing Trials. Blood 2024, 144, 6804–6804. [Google Scholar] [CrossRef]
- Iskierka-Jażdżewska, E.; Puła, B.; Jamroziak, K.; Robak, T. Chronic lymphocytic leukemia: what clinical progress have we seen in the last five years? Expert Opin Investig Drugs 2025, 34, 267–285. [Google Scholar] [CrossRef] [PubMed]
- Kater, A.P.; Janssens, A.; Eradat, H.; Offner, F.; Sandoval-Sus, J.D.; Shadman, M.; Poulsen, C.B.; Christensen, J.H.; Thompson, M.C.; Guan, M.; et al. Epcoritamab monotherapy for Richter transformation (EPCORE CLL-1): findings from a single-arm, multicentre, open-label, phase 1b/2 trial. Lancet Haematol 2026, 13, e8–e21. [Google Scholar] [CrossRef]
- Thompson, P.; Tadmor, T.; Bentzen, H.H.; Cordoba, R.; Snauwaert, S.; Goldschmidt, N.; Simkovic, M.; Christensen, J.; Lopez Jimenez, J.; Benjamini, O.; et al. Epcoritamab combinations demonstrate promising efficacy in patients (pts) with Richter transformation (RT): First results from arms 2B (epcor + lenalidomide [LEN]) and 2C (epcor + R-CHOP) of the phase 1b/2 EPCORE CLL-1 trial. Blood 2025, 146, 1015. [Google Scholar] [CrossRef]
- Danilov, A.; Fakhri, B.; Awan, F.T.; Bentzen, H.H.; Eradat, H.A.; Niemann, C.U.; Offner, F.; Poulsen, C.B.; Hoeyer, T.; Bellido, M.; et al. Epcoritamab Monotherapy in Patients (Pts) with Relapsed or Refractory (R/R) Chronic Lymphocytic Leukemia (CLL): Results from CLL Expansion and Optimization Cohorts of Epcore CLL-1. Blood 2024, 144, 883–883. [Google Scholar] [CrossRef]
- Townsend, W.; Leong, S.; Shah, M.; Batten, T.; Tucker, D.; Pottinger, B.; Paneesha, S.; El-Sharkawi, D.; Granger, D.; O'Donovan, K.; et al. Encouraging long-term efficacy and safety from first-in-human phase 1 study of time-limited NVG-111 in relapsed/refractory CLL and MCL. Blood 2025, 146, 2125–2125. [Google Scholar] [CrossRef]
- Dickinson, M.J.; Carlo-Stella, C.; Morschhauser, F.; Bachy, E.; Corradini, P.; Iacoboni, G.; Khan, C.; Wróbel, T.; Offner, F.; Trněný, M.; et al. Glofitamab for Relapsed or Refractory Diffuse Large B-Cell Lymphoma. New England Journal of Medicine 2022, 387, 2220–2231. [Google Scholar] [CrossRef]
- Carlo-Stella, C.; Hutchings, M.; Offner, F.; Mulvihill, E.; Relf, J.; Byrne, B.; Lundberg, L.; Dickinson, M. GLOFITAMAB MONOTHERAPY INDUCES DURABLE COMPLETE REMISSIONS AND HAS A MANAGEABLE SAFETY PROFILE IN PATIENTS WITH RICHTER’S TRANSFORMATION. Hematological Oncology 2023, 41, 63–65. [Google Scholar] [CrossRef]
- Fischer, C.; Chen, S.-S.; Nimmerfroh, J.; Eugster, A.; Stücheli, S.; Schultheiß, C.; Widmer, C.; Heim, D.; Kasenda, B.; Passweg, J.; et al. IGLV3-21R110-directed bispecific antibodies activate T cells and promote killing in a high-risk subset of chronic lymphocytic leukemia. Haematologica 2026, 111, 572–582. [Google Scholar] [CrossRef]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; Lekakis, L.J.; Miklos, D.B.; Jacobson, C.A.; Braunschweig, I.; Oluwole, O.O.; Siddiqi, T.; Lin, Y.; et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. New England Journal of Medicine 2017, 377, 2531–2544. [Google Scholar] [CrossRef] [PubMed]
- Schuster, S.J.; Bishop, M.R.; Tam, C.S.; Waller, E.K.; Borchmann, P.; McGuirk, J.P.; Jäger, U.; Jaglowski, S.; Andreadis, C.; Westin, J.R.; et al. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. New England Journal of Medicine 2019, 380, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, C.A.; Chavez, J.C.; Sehgal, A.R.; William, B.M.; Munoz, J.; Salles, G.; Munshi, P.N.; Casulo, C.; Maloney, D.G.; de Vos, S.; et al. Axicabtagene ciloleucel in relapsed or refractory indolent non-Hodgkin lymphoma (ZUMA-5): a single-arm, multicentre, phase 2 trial. The Lancet Oncology 2022, 23, 91–103. [Google Scholar] [CrossRef]
- Wang, M.; Munoz, J.; Goy, A.; Locke, F.L.; Jacobson, C.A.; Hill, B.T.; Timmerman, J.M.; Holmes, H.; Jaglowski, S.; Flinn, I.W.; et al. KTE-X19 CAR T-Cell Therapy in Relapsed or Refractory Mantle-Cell Lymphoma. New England Journal of Medicine 2020, 382, 1331–1342. [Google Scholar] [CrossRef]
- Abramson, J.S.; Palomba, M.L.; Gordon, L.I.; Lunning, M.; Wang, M.; Arnason, J.; Purev, E.; Maloney, D.G.; Andreadis, C.; Sehgal, A.; et al. Two-year follow-up of lisocabtagene maraleucel in relapsed or refractory large B-cell lymphoma in TRANSCEND NHL 001. Blood 2024, 143, 404–416. [Google Scholar] [CrossRef]
- Fowler, N.H.; Dickinson, M.; Dreyling, M.; Martinez-Lopez, J.; Kolstad, A.; Butler, J.; Ghosh, M.; Popplewell, L.; Chavez, J.C.; Bachy, E.; et al. Tisagenlecleucel in adult relapsed or refractory follicular lymphoma: the phase 2 ELARA trial. Nature Medicine 2022, 28, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Maude, S.L.; Laetsch, T.W.; Buechner, J.; Rives, S.; Boyer, M.; Bittencourt, H.; Bader, P.; Verneris, M.R.; Stefanski, H.E.; Myers, G.D.; et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. New England Journal of Medicine 2018, 378, 439–448. [Google Scholar] [CrossRef]
- Shah, B.D.; Ghobadi, A.; Oluwole, O.O.; Logan, A.C.; Boissel, N.; Cassaday, R.D.; Leguay, T.; Bishop, M.R.; Topp, M.S.; Tzachanis, D.; et al. KTE-X19 for relapsed or refractory adult B-cell acute lymphoblastic leukaemia: phase 2 results of the single-arm, open-label, multicentre ZUMA-3 study. The Lancet 2021, 398, 491–502. [Google Scholar] [CrossRef]
- Riches, J.C.; Davies, J.K.; McClanahan, F.; Fatah, R.; Iqbal, S.; Agrawal, S.; Ramsay, A.G.; Gribben, J.G. T cells from CLL patients exhibit features of T-cell exhaustion but retain capacity for cytokine production. Blood 2013, 121, 1612–1621. [Google Scholar] [CrossRef]
- Roessner, P.M.; Seiffert, M. T-cells in chronic lymphocytic leukemia: Guardians or drivers of disease? Leukemia 2020, 34, 2012–2024. [Google Scholar] [CrossRef] [PubMed]
- Borogovac, A.; Siddiqi, T. Advancing CAR T-cell therapy for chronic lymphocytic leukemia: exploring resistance mechanisms and the innovative strategies to overcome them. Cancer Drug Resist 2024, 7, 18. [Google Scholar] [CrossRef] [PubMed]
- Fraietta, J.A.; Lacey, S.F.; Orlando, E.J.; Pruteanu-Malinici, I.; Gohil, M.; Lundh, S.; Boesteanu, A.C.; Wang, Y.; O’Connor, R.S.; Hwang, W.-T.; et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nature Medicine 2018, 24, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Taghiloo, S.; Asgarian-Omran, H. Cross-talk between leukemic and immune cells at the tumor microenvironment in chronic lymphocytic leukemia: An update review. European Journal of Haematology 2024, 113, 4–15. [Google Scholar] [CrossRef]
- Burger, J.A.; Tsukada, N.; Burger, M.; Zvaifler, N.J.; Dell'Aquila, M.; Kipps, T.J. Blood-derived nurse-like cells protect chronic lymphocytic leukemia B cells from spontaneous apoptosis through stromal cell–derived factor-1. Blood 2000, 96, 2655–2663. [Google Scholar] [CrossRef]
- Jitschin, R.; Braun, M.; Büttner, M.; Dettmer-Wilde, K.; Bricks, J.; Berger, J.; Eckart, M.J.; Krause, S.W.; Oefner, P.J.; Le Blanc, K.; et al. CLL-cells induce IDOhi CD14+HLA-DRlo myeloid-derived suppressor cells that inhibit T-cell responses and promote TRegs. Blood 2014, 124, 750–760. [Google Scholar] [CrossRef]
- Teoh, J.; Brown, L.F. Developing lisocabtagene maraleucel chimeric antigen receptor T-cell manufacturing for improved process, product quality and consistency across CD19+ hematologic indications. Cytotherapy 2022, 24, 962–973. [Google Scholar] [CrossRef]
- Siddiqi, T.; Maloney, D.G.; Kenderian, S.S.; Brander, D.M.; Dorritie, K.; Soumerai, J.; Riedell, P.A.; Shah, N.N.; Nath, R.; Fakhri, B.; et al. Lisocabtagene Maraleucel (liso-cel) in R/R CLL/SLL: 24-Month Median Follow-up of TRANSCEND CLL 004. Blood 2023, 142, 330–330. [Google Scholar] [CrossRef]
- Porter, D.L.; Hwang, W.-T.; Frey, N.V.; Lacey, S.F.; Shaw, P.A.; Loren, A.W.; Bagg, A.; Marcucci, K.T.; Shen, A.; Gonzalez, V.; et al. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Science Translational Medicine 2015, 7, 303ra139–303ra139. [Google Scholar] [CrossRef] [PubMed]
- Melenhorst, J.J.; Chen, G.M.; Wang, M.; Porter, D.L.; Chen, C.; Collins, M.A.; Gao, P.; Bandyopadhyay, S.; Sun, H.; Zhao, Z.; et al. Decade-long leukaemia remissions with persistence of CD4(+) CAR T cells. Nature 2022, 602, 503–509. [Google Scholar] [CrossRef]
- Cappell, K.M.; Sherry, R.M.; Yang, J.C.; Goff, S.L.; Vanasse, D.A.; McIntyre, L.; Rosenberg, S.A.; Kochenderfer, J.N. Long-Term Follow-Up of Anti-CD19 Chimeric Antigen Receptor T-Cell Therapy. Journal of Clinical Oncology 2020, 38, 3805–3815. [Google Scholar] [CrossRef]
- Davids, M.S.; Kenderian, S.S.; Flinn, I.; Hill, B.T.; Maris, M.; Ghia, P.; Byrne, M.; Bartlett, N.L.; Pagel, J.M.; Zheng, Y.; et al. ZUMA-8: a phase 1 study of brexucabtagene autoleucel in patients with relapsed/refractory chronic lymphocytic leukemia. Blood 2025, 146, 938–943. [Google Scholar] [CrossRef]
- Ortiz-Maldonado, V.; Martínez-Cibrián, N.; Alserawan, L.; Betriu, S.; Triguero, A.; Blum, S.; Faes, M.; Liefaard, M.C.; Pont, M.J.; Spoon, M.; et al. Euplagia-1: A Phase 1/2 Trial of GLPG5201, a Fresh Stem-like Early Memory CD19 CAR T-Cell Therapy with a 7-Day Vein-to-Vein Time, in Patients with Relapsed/Refractory CLL and RT. Blood 2024, 144, 3452–3452. [Google Scholar] [CrossRef]
- Svoboda, J.; Landsburg, D.J.; Gerson, J.; Nasta, S.D.; Barta, S.K.; Chong, E.A.; Cook, M.; Frey, N.V.; Shea, J.; Cervini, A.; et al. Enhanced CAR T-Cell Therapy for Lymphoma after Previous Failure. N Engl J Med 2025, 392, 1824–1835. [Google Scholar] [CrossRef]
- Albiol N., M.-C.N.; Franch, A; Navarro, A.; África Martín-López, A. 1905 - Academic CD19-directed CAR-T ARI-0001 (varnimcabtagene autoleucel) for patients with relapsed/refractory chronic lymphocytic leukemia and Richter’s transformation. In Proceedings of the XXII International Workshop on CLL, Kraków, 2025. [Google Scholar]
- Liang, E.C.; Albittar, A.; Huang, J.J.; Hirayama, A.V.; Kimble, E.L.; Portuguese, A.J.; Chapuis, A.; Shadman, M.; Till, B.G.; Cassaday, R.D.; et al. Factors associated with long-term outcomes of CD19 CAR T-cell therapy for relapsed/refractory CLL. Blood Advances 2023, 7, 6990–7005. [Google Scholar] [CrossRef]
- Derigs, P.; Schubert, M.-L.; Dreger, P.; Schmitt, A.; Yousefian, S.; Haas, S.; Röthemeier, C.; Neuber, B.; Hückelhoven-Krauss, A.; Brüggemann, M.; et al. Third-generation anti-CD19 CAR T cells for relapsed/refractory chronic lymphocytic leukemia: a phase 1/2 study. Leukemia 2024, 38, 2419–2428. [Google Scholar] [CrossRef]
- Thompson, J.S.; Bixler, S.A.; Qian, F.; Vora, K.; Scott, M.L.; Cachero, T.G.; Hession, C.; Schneider, P.; Sizing, I.D.; Mullen, C.; et al. BAFF-R, a Newly Identified TNF Receptor That Specifically Interacts with BAFF. Science 2001, 293, 2108–2111. [Google Scholar] [CrossRef] [PubMed]
- Rodig, S.J.; Shahsafaei, A.; Li, B.; Mackay, C.R.; Dorfman, D.M. BAFF-R, the major B cell–activating factor receptor, is expressed on most mature B cells and B-cell lymphoproliferative disorders. Human Pathology 2005, 36, 1113–1119. [Google Scholar] [CrossRef]
- Qie, Y.; Gadd, M.E.; Shao, Q.; To, T.; Liu, A.; Li, S.; Rivera-Valentin, R.; Yassine, F.; Murthy, H.S.; Dronca, R.; et al. Targeting chronic lymphocytic leukemia with B-cell activating factor receptor CAR T cells. MedComm (2020) 2024, 5, e716. [Google Scholar] [CrossRef]
- Hug, S.; Phely, L.; Riedel, A.; Killinger, S.; Hensen, L.; Faul, C.; Bethge, W.A.; Lengerke, C. Dual CD19/CD22-Targeting CAR-T Cells for Richter Transformation: A Case Series. Blood 2024, 144, 6544–6544. [Google Scholar] [CrossRef]
- Shah, N.N.; Atallah, E.L.; Abedin, S.; Murthy, G.S.G.; Runaas, L.; Michaelis, L.C.; Longo, W.; Furqan, F.; Bucklan, D.; Szabo, A.; et al. Phase 1 Trial of LV20.19 CAR T-Cells for Relapsed, Refractory CLL and Richter's Transformation. Transplantation and Cellular Therapy 2024, 30, S35–S36. [Google Scholar] [CrossRef]
- Wang, X.; Dong, Z.; Awuah, D.; Chang, W.C.; Cheng, W.A.; Vyas, V.; Cha, S.C.; Anderson, A.J.; Zhang, T.; Wang, Z.; et al. CD19/BAFF-R dual-targeted CAR T cells for the treatment of mixed antigen-negative variants of acute lymphoblastic leukemia. Leukemia 2022, 36, 1015–1024. [Google Scholar] [CrossRef] [PubMed]
- Mansoori, S.; Noei, A.; Maali, A.; Seyed-Motahari, S.S.; Sharifzadeh, Z. Recent updates on allogeneic CAR-T cells in hematological malignancies. Cancer Cell International 2024, 24, 304. [Google Scholar] [CrossRef]
- Lussana, F.; Magnani, C.F.; Galimberti, S.; Gritti, G.; Gaipa, G.; Belotti, D.; Cabiati, B.; Napolitano, S.; Ferrari, S.; Moretti, A.; et al. Donor-derived CARCIK-CD19 cells engineered with Sleeping Beauty transposon in acute lymphoblastic leukemia relapsed after allogeneic transplantation. Blood Cancer J 2025, 15, 54. [Google Scholar] [CrossRef] [PubMed]
- Lekakis, L.J.; Locke, F.L.; Tees, M.; Neelapu, S.S.; Malik, S.A.; Hamadani, M.; Frank, M.J.; Popplewell, L.L.; Abramson, J.S.; de Vos, S.; et al. ALPHA2 Study: ALLO-501A Allogeneic CAR T in LBCL, Updated Results Continue to Show Encouraging Safety and Efficacy with Consolidation Dosing. Blood 2021, 138, 649. [Google Scholar] [CrossRef]
- Ghobadi, A.; McGuirk, J.P.; Shaughnessy, P.; Tam, C.S.; Allen, M.; Pan, C.; Li, Z.; Stevens, W.N.; Weaver, A.; Cheah, C.Y. CTX112, a Next-Generation Allogeneic CRISPR-Cas9 Engineered CD19 CAR T Cell with Novel Potency Edits: Data from Phase 1 Dose Escalation Study in Patients with Relapsed or Refractory B-Cell Malignancies. Blood 2024, 144, 4829–4829. [Google Scholar] [CrossRef]
- Long, M.; Beckwith, K.; Do, P.; Mundy, B.L.; Gordon, A.; Lehman, A.M.; Maddocks, K.J.; Cheney, C.; Jones, J.A.; Flynn, J.M.; et al. Ibrutinib treatment improves T cell number and function in CLL patients. The Journal of Clinical Investigation 2017, 127, 3052–3064. [Google Scholar] [CrossRef]
- Fraietta, J.A.; Beckwith, K.A.; Patel, P.R.; Ruella, M.; Zheng, Z.; Barrett, D.M.; Lacey, S.F.; Melenhorst, J.J.; McGettigan, S.E.; Cook, D.R.; et al. Ibrutinib enhances chimeric antigen receptor T-cell engraftment and efficacy in leukemia. Blood 2016, 127, 1117–1127. [Google Scholar] [CrossRef]
- Ruella, M.; Kenderian, S.S.; Shestova, O.; Klichinsky, M.; Melenhorst, J.J.; Wasik, M.A.; Lacey, S.F.; June, C.H.; Gill, S. Kinase inhibitor ibrutinib to prevent cytokine-release syndrome after anti-CD19 chimeric antigen receptor T cells for B-cell neoplasms. Leukemia 2017, 31, 246–248. [Google Scholar] [CrossRef]
- Gauthier, J.; Hirayama, A.V.; Purushe, J.; Hay, K.A.; Lymp, J.; Li, D.H.; Yeung, C.C.S.; Sheih, A.; Pender, B.S.; Hawkins, R.M.; et al. Feasibility and efficacy of CD19-targeted CAR T cells with concurrent ibrutinib for CLL after ibrutinib failure. Blood 2020, 135, 1650–1660. [Google Scholar] [CrossRef]
- Wierda, W.G.; Dorritie, K.A.; Munoz, J.; Stephens, D.M.; Solomon, S.; Gillenwater, H.H.; Gong, L.; Yang, L.; Ogasawara, K.; Thorpe, J.; et al. TRANSCEND CLL 004: PHASE 1 COHORT OF LISOCABTAGENE MARALEUCEL (LISO-CEL) COMBINED WITH IBRUTINIB (IBR) FOR PATIENTS (PTS) WITH R/R CLL/SLL. Hematological Oncology 2021, 39. [Google Scholar] [CrossRef]
- Gill, S.; Vides, V.; Frey, N.V.; Hexner, E.O.; Metzger, S.; O'Brien, M.; Hwang, W.T.; Brogdon, J.L.; Davis, M.M.; Fraietta, J.A.; et al. Anti-CD19 CAR T cells in combination with ibrutinib for the treatment of chronic lymphocytic leukemia. Blood Adv 2022, 6, 5774–5785. [Google Scholar] [CrossRef]
- Tettamanti, S.; Rotiroti, M.C.; Giordano Attianese, G.M.P.; Arcangeli, S.; Zhang, R.; Banerjee, P.; Galletti, G.; McManus, S.; Mazza, M.; Nicolini, F.; et al. Lenalidomide enhances CD23.CAR T cell therapy in chronic lymphocytic leukemia. Leuk Lymphoma 2022, 63, 1566–1579. [Google Scholar] [CrossRef]
- Funk, C.R.; Wang, S.; Chen, K.Z.; Waller, A.; Sharma, A.; Edgar, C.L.; Gupta, V.A.; Chandrakasan, S.; Zoine, J.T.; Fedanov, A.; et al. PI3Kδ/γ inhibition promotes human CART cell epigenetic and metabolic reprogramming to enhance antitumor cytotoxicity. Blood 2022, 139, 523–537. [Google Scholar] [CrossRef]
- Sportoletti, P.; De Falco, F.; Del Papa, B.; Baldoni, S.; Guarente, V.; Marra, A.; Dorillo, E.; Rompietti, C.; Adamo, F.M.; Ruggeri, L.; et al. NK Cells in Chronic Lymphocytic Leukemia and Their Therapeutic Implications. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Herrera, L.; Santos, S.; Vesga, M.A.; Anguita, J.; Martin-Ruiz, I.; Carrascosa, T.; Juan, M.; Eguizabal, C. Adult peripheral blood and umbilical cord blood NK cells are good sources for effective CAR therapy against CD19 positive leukemic cells. Scientific Reports 2019, 9, 18729. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.A.; Leveson-Gower, D.B.; Gill, S.; Baker, J.; Beilhack, A.; Negrin, R.S. NK cells mediate reduction of GVHD by inhibiting activated, alloreactive T cells while retaining GVT effects. Blood 2010, 115, 4293–4301. [Google Scholar] [CrossRef]
- Liu, E.; Tong, Y.; Dotti, G.; Shaim, H.; Savoldo, B.; Mukherjee, M.; Orange, J.; Wan, X.; Lu, X.; Reynolds, A.; et al. Cord blood NK cells engineered to express IL-15 and a CD19-targeted CAR show long-term persistence and potent antitumor activity. Leukemia 2018, 32, 520–531. [Google Scholar] [CrossRef]
- Liu, E.; Marin, D.; Banerjee, P.; Macapinlac, H.A.; Thompson, P.; Basar, R.; Nassif Kerbauy, L.; Overman, B.; Thall, P.; Kaplan, M.; et al. Use of CAR-Transduced Natural Killer Cells in CD19-Positive Lymphoid Tumors. N Engl J Med 2020, 382, 545–553. [Google Scholar] [CrossRef]
- Marin, D.; Li, Y.; Basar, R.; Rafei, H.; Daher, M.; Dou, J.; Mohanty, V.; Dede, M.; Nieto, Y.; Uprety, N.; et al. Safety, efficacy and determinants of response of allogeneic CD19-specific CAR-NK cells in CD19(+) B cell tumors: a phase 1/2 trial. Nat Med 2024, 30, 772–784. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




